Introduction
Primary central nervous system lymphoma (PCNSL) is a
malignant tumor confined to the brain, pia mater, spinal cord or
eye (1). A study at the
Massachusetts General Hospital from 1958 to 1989 showed that a
total of >90% of patients with PCNSL have diffuse large B cell
lymphoma (2), which exhibits high
invasion rates and poor prognosis. Poor prognostic factors include
age, Karnofsky score, serum lactate dehydrogenase (LDH),
cerebrospinal fluid protein, B cell lymphoma 6 protein expression
(3) and reduced lymphocytes
(4).
PCNSL cells usually present with diffuse
infiltration. Therefore, it is difficult to completely remove by
surgery; and its recurrence rate is high (5). Previously, whole brain radiotherapy was
performed as a first-line therapy; however, treatment with
radiotherapy alone confers disease recurrence and neurotoxicity
(6,7), which limits its clinical application.
High-dose methotrexate (HD-MTX) has an affirmed curative effect,
but the complications, such as neurotoxicity, limit its dose
(8,9). In addition, the 2-year overall survival
(OS) rate of HD-MTX monotherapy is only 61–63% (10,11),
which is not satisfying. At present, the preferred regimen is the
combined treatment based on HD-MTX (combined with cytarabine,
rituximab and idarubicin) (12–18).
However, the combination of HD-MTX based chemotherapy is still
under debate. Teniposide (Vm26) has high liposolubility, which
makes it is able to pass the blood-brain barrier, and it has a
slower elimination rate (19). Fan
et al (20) Revealed that
MTX+Vm26 did not significantly improve the median progression-free
survival of patients, but complete remission (CR) and overall
response (OR) were significantly increased. In the present study,
the clinical curative effects of HD-MTX+Vm26 vs. Vm26 alone were
compared, with the aim of providing a useful reference for clinical
practice.
Materials and methods
Data acquisition
A total of 56 patients with PCNSL who were admitted,
but had not been treated, to the Department of Hematology of
Huashan Hospital Affiliated to Fudan University (Shanghai, China)
from January 2009 to December 2014 were enrolled into the present
study. Due to the retrospective nature of the present study, the
requirement for ethical approval was waived by the Ethics Committee
of Huashan Hospital (Shanghai, China). The cohort of 56 patients
comprised 38 males and 18 females. The age of these patients ranged
between 25–74 years, with a median age of 54.5 years. A total of 13
patients (23.21%) were ≥60 years.
According to the treatment regimen received,
patients were divided into two groups: A HD-MTX+Vm26 group and
HD-MTX group (n=28 in each group). A retrospective analysis method
was adopted. Inclusion criteria were as follows: i) Patients who
were first diagnosed with PCNSL; ii) patients with a negative HIV
test result; iii) patients with diagnosis supported by pathological
evidences; iv) patients who had completed at least three courses of
chemotherapy; v) patients with changes in intracranial lesions that
had been followed up by imaging tests (MRI or PET-CT) prior to
diagnosis and subsequent to chemotherapy; and vi) patients who were
diagnosed based on the 2008 World Health Organization
classification of hematopoietic and lymphoid neoplasms (21). Exclusion criteria were as follows:
Congenital or acquired immunodeficiency, including patients with
previous organ transplantation, concurrent treatment with
immunosuppressive drugs, and AIDS-related PCNSL; disease confined
to the eye without another localization in the central nervous
system (CNS); the presence or history of systemic lymphoma; any
prior malignancy with the exception of adequately treated
nonmelanoma skin cancer and carcinoma in situ of the cervix
uteri; a serious impairment of cardiac, renal, or liver function;
pregnancy; any severe uncontrolled infection; prior chemotherapy,
with the exception of corticosteroids, for a maximum period of 6
weeks before and after diagnosis or surgery; and Burkitt's
lymphomas of low-grade T-cell lymphomas.
Tissue and reagents
56 cases of excised tissue or biopsy tissue of brain
tumor were collected, fixed by 3.7% neutral formaldehyde at room
temperature for 6–12 h, until further processing, routinely
dehydrated, embedded by paraffin, sliced into 3-µm thick sections,
and then subjected to H&E staining (stained with Gill's
hematoxylin for 2–3 min and eosin for 10–20 sec at room
temperature) and immunohistochemical staining. In
immunohistochemical staining, the tissue was embedded by paraffin,
sliced into 4-µm thick sections, dewaxed with 100% xylene and
concentration gradients of alcohol (100% EtOH for 5 min twice, 95%
EtOH for 4 min twice, 80% EtOH for 4 min) according to the standard
operation process, and repaired in citrate buffer (pH 6.0; Shanghai
Xin Yu Biotech Co., Ltd, Shanghai, China) and boiled for 10 min.
After natural cooling, the endogenous peroxidase activity was
blocked using 3% hydrogen peroxide at room temperature for 10 min,
and the sections were blocked using 10% goat serum/PBS at room
temperature for 30 min, then and treated with the primary
antibodies and incubated at 4°C overnight: CD10 (1:50; cat. no.
ab951), CD19 (1:200; cat. no. ab134114), CD38 (1:200; cat. no.
ab108403), CD3 (1:30; cat. no. ab16669), Bcl-6 (undiluted; cat. no.
ab172610) and Bcl-2 (1:100; cat. no. ab32124), which were purchased
from Abcam (Cambridge, UK); MPO (1:50; cat. no. TC66701-2), CD5
(1:100; cat. no. M364101-2), CD38 (1:50; cat. no. TC67401-2), CD56
(1:50; cat. no. TC67401-2), CD4 (1:10; cat. no. FR86850-2), CD8
(1:10; cat. no. F076501-2), CD23 (1:50; cat. no. IS78130-2), Kappa
(1:2,500; cat. no. A019102-2) and Lambda (1:2,500; cat. no.
239222), which were purchased from Dako; Agilent Technologies, Inc.
(Santa Clara, CA, USA); CD20 (1:100; cat. no. MA513141), CD79a
(1:100; cat. no. 186490) and MIB-1 (1:100; cat. no. PA5-16446),
which were purchased from Zymed; TDT (undiluted; cat. no.
PL0403179) and CyclinD1 (1:50; cat. no. or677046), which were
purchased from Neomarkers (https://univ.biomart.cn/). After washing with PBS, the
tissue sections were placed to react with biotin-labeled rabbit
anti-guinea pig IgG secondary antibody (1:1,000; cat. no. QN2726;
Beijing Baiaoleboke Co., Ltd., Beijing, China) at 37°C for 30 min.
The sections were washed with PBS for 5 min, repeated three times,
then incubated with VECTA STAIN Elite ABC standard kit (Vector
Laboratories, Inc., Burlingame, CA, USA) according to the
manufacturer's protocol at 37°C for 30 min, finally the signals
were visualized with a chromogenic kit (Vector Laboratories, Inc.)
according to the manufacturer's protocol or by microscopic
examination: Optical microscope (Olympus BX41, Olympus Corporation,
Tokyo, Japan) was used, and ×4, ×10 and ×40 objectives were used
for observation, respectively.
Data acquisition included the following aspects: i)
Basic information; ii) diagnostic information; iii) clinical
manifestations; iv) imaging manifestations; v) histopathology and
laboratory examination indices; vi) treatment situations; vii)
Eastern Cooperative Oncology Group (ECOG) score (22), and; viii) curative effect.
Treatment regimens and treatment
response assessment
The assessment of the treatment response was based
on the consensus standards developed by the International Primary
CNS Lymphoma Collaborative Group (IPCG) (23), consist of complete response (CR),
partial response (PR), progressive disease (PD), and stable disease
(SD). Overall response (OR) includes PR and CR. The summary of the
assessment criteria is presented in Table I. The follow-up deadline was December
31, 2015.
 | Table I.Criteria for assessment of
therapeutic effects. |
Table I.
Criteria for assessment of
therapeutic effects.
| Treatment
response | Imaging
examination | Dosage of
glucocorticoid | Ophthalmologic
examination | Cerebrospinal fluid
examination |
|---|
| CR | No enhancement | Nil | Normal | Negative |
| CRu | No enhancement | Any | Normal | Negative |
|
| No abnormal
lesions | Any | Micro RPE was
abnormal | Negative |
| PR | Enhanced lesion was
reduced by 50% | No association | Micro RPE was
abnormal or normal | Negative |
| PD | No enhancement | No association | Vitreous cell or
retinal infiltration, reduction | Persistent positive
or suspicious positive |
|
| Tumor was reduced
by 25% or new lesion appeared | No association | Recurrence or new
eye lesions | Recurrence or
positive |
There were XX courses of treatment. In each course
of treatment, the total dose of MTX was 3.0 g/m2, while
the total dose of Vm26 was 300 mg, while the total dose of Vm26 in
every course of treatment was 300 mg. During the intravenous
administration of MTX, patients were required to undergo hydration
and alkalization, and to receive leucovorin rescue. The interval
between chemotherapy sessions was three weeks. Treatment regimens
were altered if patients suffered from disease progression, or if
unacceptable toxic reactions occurred.
Statistical analysis
The collected data were analyzed using SPSS 20.0
software (IMB Corp., Armonk, NY, USA). Overall survival (OS) time
was defined as the period from the time the patient was diagnosed
to the time the patient succumbed, or up to the follow-up deadline.
Progression-free survival (PFS) was defined as the period from the
time the patient was diagnosed to disease recurrence/progression or
last point of contact with the patient. Univariate analysis was
conducted using a log-rank test, survival analysis was conducted
using Kaplan-Meier curves and log-rank test and multivariate
analysis was conducted using a Cox's regression. P<0.05 was
considered to indicate a statistically significant difference.
Results
Patients
The cohort of 56 patients comprised 38 males and 18
females. The age of these patients ranged between 25–74 years, with
a median age of 54.5 years. A total of 13 patients (23.21%) were
≥60 years.
Clinical characteristics of patients
Clinical symptoms
The common symptoms of PCNSL include elevated
intracranial pressure (53.6%), somatic dysfunction (53.6%),
cognitive dysfunction (33.9%), impaired vision (30.3%) and language
disorder (17.9%).
Imaging tests
All patients underwent cranial magnetic resonance
imaging, and 17 additionally underwent positron emission
tomography-computed tomography. Patients with multiple lesions
accounted for 60.7% of the total cohort, and patients with deep
lesions accounted for 60.7%. The frontal lobe was the most common
lesion site, accounting for 46.4% patients, followed by the
parietal lobe (25%) and basal ganglia (23.2%).
ECOG score results
A total of 12 patients received 1 point, 13 patients
received 2 points, 26 patients received 2 points, 4 patients
received 3 points and 2 patients received 5 points. Patients who
received a score within 1–3 points accounted for 91% of the total
cohort.
Histopathology and laboratory tests
Histopathology
The histopathology of all 56 patients was diffuse
large B cell lymphoma.
Cerebrospinal fluid examination
Cerebrospinal fluid examination was performed on 26
patients. Among 26 patients, a Pandy's test exhibited a positive
result in 19 patients (73.1%), the protein content increased in 18
patients (69.1%), and spinal cord involvement, such as chest-back
or lower-back pain, weakness of lower limbs, paraplegia and
superficial sensory dullness or disappearance, occurred in 11
patients (42.3%). Spinal cord involvement: i) The patients had
periodical lumbar pain and compression symptoms; ii) Enhanced MRI
revealed lesions; iii) The lesions shrunk after treatment.
Serum LDH and serum and urinary
β2-microglobulin (MG) examination
Serum LDH (normal range from 109 to 245 U/l) was
increased in 21 patients (37.5%), serum β2-MG (normal range from 1
to 2.8 µg/ml) was increased in 32 patients (57.1%), and urinary
β2-MG (normal range from 0 to 0.3 µg/ml) was increased in 21
patients (37.5%).
Treatment protocol
Treatment regimen
Treatment regimens were divided into three
categories: i) Radiotherapy + chemotherapy (radiotherapy followed
by chemotherapy); ii) chemotherapy + radiotherapy (chemotherapy
followed by radiotherapy); iii) chemotherapy alone. All had been
done. 7 patients received radiotherapy + chemotherapy, 20 patients
received chemotherapy + radiotherapy, and 29 patients received
chemotherapy alone. MTX group: 3 patients received radiotherapy +
chemotherapy, 13 patients received chemotherapy + radiotherapy, and
13 patients received chemotherapy alone.
The 56 patients in the present study all received
chemotherapy regimens with HD-MTX. Among these patients, 51.8%
patients were treated with chemotherapy alone, and the remaining
48.2% patients were treated with chemotherapy and radiotherapy. The
majority of the patients received radiotherapy followed by
chemotherapy; only 7 patients (12.5%) received chemotherapy
followed by radiotherapy. These results revealed that the median
PFS was 20 months and the median OS time was 31 months in patients
who received chemotherapy alone. The median PFS and median OS were
21 and 37 months, respectively, in patients who received
chemotherapy followed by radiotherapy; while median PFS and median
OS were 22 and 52 months, respectively, in patients who received
radiotherapy followed by chemotherapy. Between patients who
received chemotherapy alone and the patients who received
chemotherapy combined with radiotherapy, the difference in PFS was
not statistically significant.
Curative effect analysis
Treatment response
The evaluation of disease progression was performed
following three courses of chemotherapy for all patients. In the
MTX+Vm26 group, 12 patients (42.85%) achieved CR, 10 patients
(35.71%) achieved PR and the OR rate was 78.57%. In the MTX group,
7 patients (25%) achieved CR, 11 patients (39.29%) achieved PR and
the OR rate was 64.3%. Differences in CR and OR between these two
groups were not statistically significant (Table II).
 | Table II.Comparison of the curative effects
between treatments groups. |
Table II.
Comparison of the curative effects
between treatments groups.
| Group | MTX + Vm26 | MTX | χ2 | P-value |
|---|
| CR | 12 (42.85%) | 7 (25%) | 1.991 | 0.158 |
| PR | 10 (35.71%) | 11 (39.29%) | 0.076 | 0.783 |
| SD | 4 (14.29%) | 4 (14.29%) | 0.000 | 1.000 |
| PD | 2 (7.14%) | 6 (21.43%) | 1.313 | 0.252 |
| OR | 22 (78.57%) | 18 (64.3%) | 1.400 | 0.237 |
| Median PFS,
months | 22 | 12 | 5.462 | 0.019 |
| Median OS,
months | 57 | 28 | 6.196 | 0.013 |
PFS and OS
At the end of the follow-up, 26 patients succumbed,
29 patients survived, and 1 patient in the MTX+Vm26 group was lost
to follow-up subsequent to confirmation of relapse. The median PFS
were 22 and 12 months in the MTX+Vm26 and MTX groups, respectively;
the median OS rates were 57 and 28 months, respectively. These two
indices were significantly increased in the MTX+Vm26 group compared
with the MTX group (P<0.05; Table
II). The median OS was 40 months in 55 patients (total patient
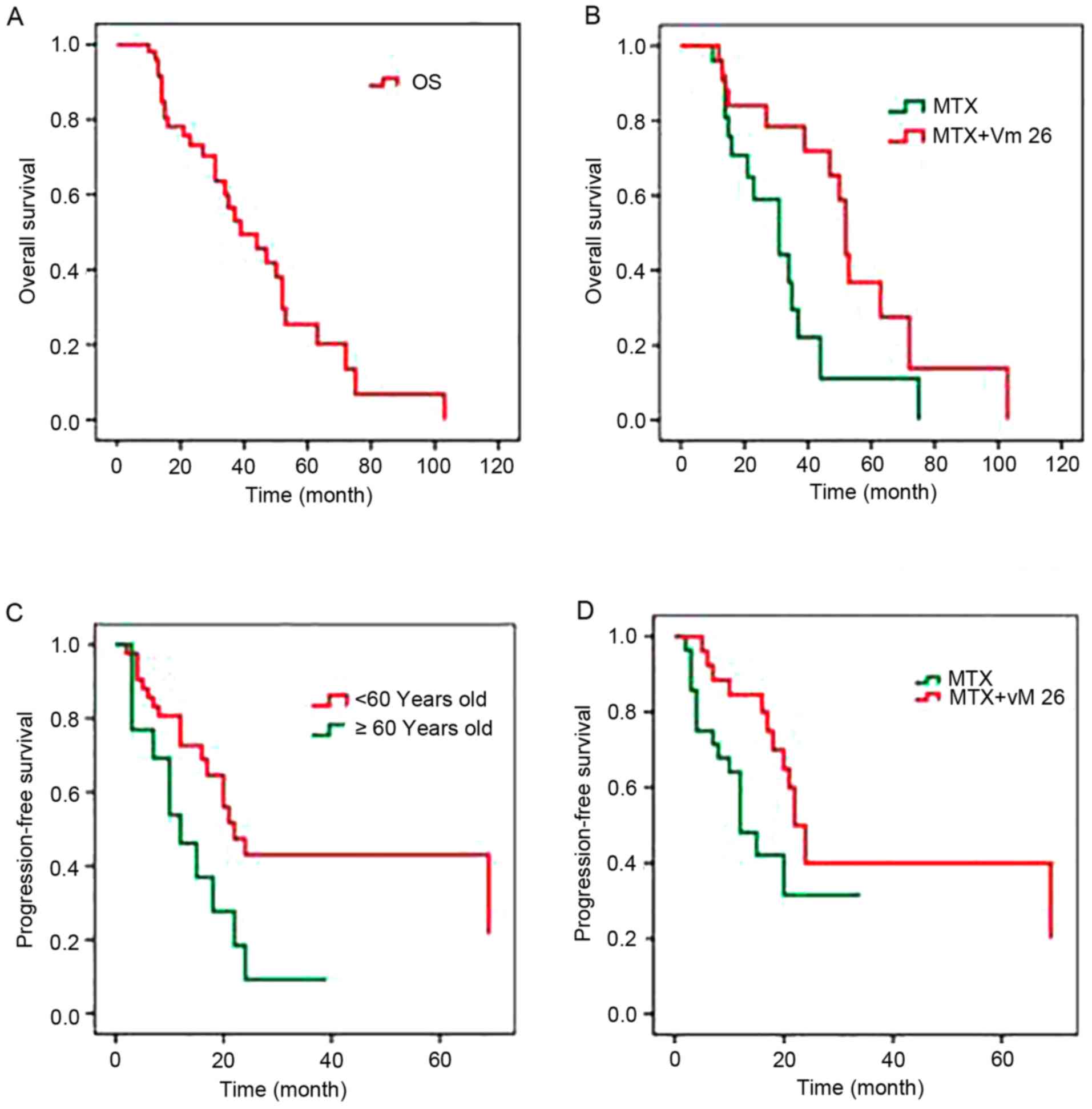
cohort minus 1 patient lost to follow up) (Fig. 1A). Comparison of the median OS
between the two treatment groups revealed that the OS of the
MTX+Vm26 group was significantly increased compared with the MTX
group (Fig. 1B).
Mortality
Overall, mortality was 47.3% at the end of
follow-up, in which 13 patients (47.3%) were from the MTX group and
13 patients (48.1%) were from the MTX+Vm26 group, and the
difference between the mortality rates between the two treatment
groups was not statistically significant (P=0.898).
Disease progression
A total of 30 patients exhibited disease progression
at the end of the follow-up, in which 16 patients (57.14%) were
from the MTX group and 14 patients (50%) were from the MTX+Vm26
group; the difference between these two groups was not
statistically significant (P=0.592).
Prognosis analysis
Univariate analysis
Univariate analysis was performed to analyze factors
that may be associated with the prognosis of disease. The disease
progression curve was evaluated using the Kaplan-Meier method and
the factors were compared using a log-rank test. Detailed results
are indicated in Table III.
Univariate analysis revealed that age was a prognostic factor in
patients with PCNSL, and the median PFS of patients with an age
>60 years was 10 months shorter compared with that in patients
with an age of ≤60 years (Fig. 1C).
These are consistent with the results of previous studies (24–26). The
median PFS was 69 months in patients who achieved CR following
three courses of treatment, and was 20 months in patients who did
not achieve CR. The difference between these two was marked, but
was not statistically significant (P=0.061). The median PFS was 12
months in the MTX group, and 22 months in the HD-MTX+Vm26 group,
and the difference between these two groups was statistically
significant (P=0.033; Fig. 1D).
 | Table III.Univariate analysis. |
Table III.
Univariate analysis.
| Potential
prognostic factors | PD/total cases | Median PFS,
months | χ2 | P-value |
|---|
| Age, years |
| 20 | 6.021 | 0.014 |
|
≤60 | 19/43 | 22 |
|
|
|
>60 | 11/13 | 12 |
|
|
| ECOG score,
points |
|
| 0.017 | 0.896 |
|
0–1 | 6/12 | 22 |
|
|
| ≥1 | 24/44 | 20 |
|
|
| Deep lesions
accumulating |
|
| 1.117 | 0.291 |
| No | 14/22 | 16 |
|
|
|
Yes | 16/34 | 20 |
|
|
| Number of
lesions |
|
| 0.741 | 0.389 |
|
Single | 14/22 | 16 |
|
|
|
Multiple | 16/34 | 20 |
|
|
| Serum LDH |
|
| 1.100 | 0.294 |
| Normal
(106–211 U/l) | 15/35 | 24 |
|
|
|
Abnormally increasing | 15/21 | 18 |
|
|
| Blood
β2-MG |
|
| 0.793 | 0.373 |
| Normal
(0.70–1.80 mg/l) | 14/24 | 17 |
|
|
|
Abnormally increasing | 16/32 | 22 |
|
|
| Urine
β2-MG |
|
| 0.076 | 0.783 |
| Normal
(<0.25 mg/l) | 19/35 | 18 |
|
|
|
Abnormally increasing | 11/21 | 21 |
|
|
| CSF; Pandy's
test |
|
| 1.670 | 0.196 |
|
Negative | 4/6 | 10 |
|
|
|
Positive | 13/20 | 20 |
|
|
| CSF protein
quantification |
|
| 0.948 | 0.330 |
| Normal
(150–450 mg/l) | 5/8 | 10 |
|
|
|
Abnormally increasing | 12/18 | 20 |
|
|
| CSF glucose
level |
|
| 0.160 | 0.689 |
| Normal
(2.5–4.4 mmol/l) | 12/20 | 20 |
|
|
|
Abnormal | 5/6 | 20 |
|
|
| CSF chloride |
|
| 0.915 | 0.339 |
| Normal
(2.7–3.9 mmol/l) | 12/14 | 20 |
|
|
|
Abnormal | 5/12 | 22 |
|
|
| CSF white blood
cell count |
|
| 2.296 | 0.130 |
| Normal
(0–5/mm3) | 12/19 | 24 |
|
|
|
Abnormally increasing | 5/7 | 20 |
|
|
| Lesion resection by
surgery |
|
| 0.562 | 0.454 |
|
Yes | 12/31 | 20 |
|
|
| No | 18/25 | 22 |
|
|
| Received CR |
|
| 3.519 | 0.061 |
| No | 22/37 | 20 |
|
|
|
Yes | 8/19 | 69 |
|
|
| Chemotherapy
regiments |
|
| 4.557 | 0.033 |
|
HD-MTX | 16/28 | 12 |
|
|
| HD-MTX
+ Vm26 | 14/28 | 22 | 0.920 | 0.631 |
| Therapeutic
schedule |
|
|
|
|
|
Chemotherapy | 16/29 | 18 |
|
|
|
Chemotherapy +
radiotherapy | 4/7 | 21 |
|
|
|
Radiotherapy +
chemotherapy | 10/20 | 22 |
|
|
Multivariate analysis
As identified from the univariate analysis, those
progression-associated factors with a P<0.05, and factors with a
P>0.05 with a PFS presenting a difference, were analyzed using
multivariate analysis. These results revealed that age and
treatment regimen were independent risk factors that affect the
progression of PCNSL (Table
IV).
 | Table IV.Multivariate analysis. |
Table IV.
Multivariate analysis.
| Factor | B | SE | Wald test | D.f. | P-value | Exp (B) | 95% CI |
|---|
| Age | −0.886 | 0.400 | 4.902 | 1 | 0.027 | 0.412 | 0.188–0.903 |
| Treatment
remission | 0.595 | 0.428 | 1.928 | 1 | 0.165 | 1.812 | 0.783–4.195 |
| Treatment
regimen | 0.951 | 0.412 | 5.321 | 1 | 0.021 | 2.587 | 1.154–5.803 |
Toxic reaction
A total of 8 patients (28.57%) in the Vm26+MTX group
and 6 patients (21.43%) in the MTX group exhibited grade 3/4
hematologic leukopenia. A total of 5 patients in the MTX+Vm26 group
(17.86%) and 3 patients in the MTX group (10.71%) exhibited
agranulocytosis and fever. A total of 7 patients (25%) developed
pneumonia. In addition, 5 patients in the MTX+Vm26 (17.86%) and 4
patients in the MTX group (14.29%) exhibited grade 2 liver function
impairment (Table V). No patients
exhibited ≥grade 3 hyperpyrexia and cardiac toxicity.
 | Table V.Comparison of toxic reactions between
MTX + Vm26 and MTX treatment groups. |
Table V.
Comparison of toxic reactions between
MTX + Vm26 and MTX treatment groups.
| Toxicity | MTX+Vm26 (%) | MTX (%) | χ2 | P-value |
|---|
| Hematologic |
| Grade
3–4 leukopenia | 8 (28.57) | 6 (21.43) | 0.381 | 0.537 |
| Grade
3–4 anemia | 3 (10.71) | 2 (7.14) | 0.220 | 0.639 |
| Grade
3–4 PLT decrease | 4 (14.29) | 2 (7.14) | 0.747 | 0.388 |
|
Non-hematologic |
| Grade 2
abnormal liver function | 5 (17.86) | 4 (14.29) | 0.132 | 0.716 |
| Grade 2
renal function damage | 4 (14.29) | 5 (17.86) | 0.132 | 0.716 |
| Grade 2
numbness | 3 (10.71) | 2 (7.14) | 0.220 | 0.639 |
Discussion
PCNSL is a rare type of extra-nodal lymphoma. The
initial symptoms include changes in cognitive function, increased
intracranial pressure and hemiplegia. During the early stage,
lesions are confined to the CNS; and in the late stage, <10% of
patients exhibit tumors that spread outside the CNS (27). Although the cancer cells originates
from lymphocytes, PCNSL usually is considered as a brain tumor in
practice due to its therapeutic challenges, such as drug delivery
through blood-brain barrier, and cerebral toxicity (28). Due to these unique characteristics of
the clinical symptoms and biological behaviors of the tumor cells
in PCNSL, the 2008 WHO Hematopoietic and Lymphoid Classification
lists it as an independent disease entity (21). In the past three decades,
epidemiological data in Western countries, such as Norway and
Sweden revealed that the incidence of PCNSL continues to increase
(29–31). The prognosis of patients with PCNSL
is relatively poor, and the prognosis of patients with HIV
infection is particularly poor (32). In the present study, no patient had
HIV infection.
Statistical data from the United States of America
between 2000–2004 revealed that the average age of patients
diagnosed with PCNSL was 60 years, and the age of the majority of
patients ranged within 50–65 years (5,33,34). A
study conducted in Spain revealed that within the period 1990–2010,
the average age of patients diagnosed with PCNSL was 59.3 years
(35). In the present study, the
average age of patients was 53.5 years, which was decreased
compared with that in other countries.
PCNSL exhibits three pathological types: DLBCL, T
cell lymphoma and other types of B cell lymphoma. DLBCL accounts
for >90% of PCNSL (36). The
pathological types of the 56 patients were all DLBCL in the present
study.
The clinical symptoms of patients with PCNSL were
associated with the location of tumor lesions (37,38). A
study in Spain revealed that patients with cognitive function
decline accounted for 33.4%, and those with cephalea accounted for
25% (35). The present study
revealed that increased intracranial pressure and physical movement
disorders were common, and the rates for the two symptoms were
53.6%; followed by cognitive dysfunction (33.9%), vision impairment
(30.3%) and language disorder (17.9%).
Statistics from the guidelines of the National
Comprehensive Cancer Network (NCCN) revealed that >90% of
patients with PCNSL exhibited lesions affecting the brain
parenchyma, and over one-half of these patients possessed multiple
lesions (39). Fraser et al
(40) demonstrated that 35% of
patients with PCNSL exhibited multiple lesions, and up to 38% of
patients exhibited lesions in the cerebral hemispheres. In the
present study, frontal lobe involvement (46.4%) accounted for the
largest proportion of lesion sites. The proportion of patients with
deep involvement was 60.7%. The majority of patients exhibited
multiple lesions (60.7%).
The physical fitness score of patients was one of
the factors that affected the choice of treatment regimens. It was
not recommended that patients with poor physical condition use the
HD-MTX+Vm26 regimen (41). In the
present study, the ECOG scores of patients were concentrated within
1–3 points.
Results of the cerebrospinal fluid examination in 26
patients indicated that the Pandy's test demonstrated a positive
result in 19 patients (73.1%), and that the protein level in
cerebrospinal fluid was increased in 18 patients (not examined
using a Pandy's test) (69.2%). The examinations of serum LDH, serum
β2-MG and urine β2-MG revealed that 37.5% of patients exhibited
increased serum LDH, 57.1% of patients exhibited increased serum
β2-MG and 37.5% of patients exhibited increased urinary β2-MG.
The feasibility and effectiveness of HD-MTX in the
treatment of PCNSL have been recognized. It may be used for
induction therapy and salvage therapy (1). Results of a study conducted by Prica
et al (42) revealed that
compared with chemotherapy alone, chemotherapy combined with
radiotherapy was an improved induction regimen in younger patients
(aged <60 years). However, due to toxic side effects, it is
recommended that elderly patients (>60 years old) should receive
chemotherapy alone (43,44). A study conducted by Muirhead et
al (45) revealed that for
patients who were not able to tolerate large-dose chemotherapy,
3-year survival rates of salvage radiotherapy were increased
compared with that of the second-line chemotherapy (60 vs. 33%).
Salvage radiotherapy may be a choice for young patients with
recurrent/refractory PCNSL (7). The
use of chemotherapy or radiotherapy should be determined on an
individual patient basis. The 56 patients in the present study all
received chemotherapy regimens with HD-MTX. Among these patients,
51.8% patients were treated with chemotherapy alone, and the
remaining 48.2% patients were treated with chemotherapy and
radiotherapy. Between patients who received chemotherapy alone and
the patients who received chemotherapy combined with radiotherapy,
the difference in PFS was not statistically significant.
Additionally, whilst the difference in median OS was notable, it
was not statistically significant (P=0.24). This may be associated
with not enough sample size between chemotherapy alone group and
chemotherapy combined with radiotherapy group.
Total cranial irradiation may cause toxic side
effects, such as the decrease in cognitive function. Ongoing
clinical trials are being conducted on replacing radiotherapy
alone, with combined chemotherapy (46). On the basis of HD-MTX, these regimens
contain 1–3 chemotherapeutic drugs with different mechanisms, such
as HD-Ara-C, temozolomide, rituximab, idarubicin and etoposide
(12). Vm26 is a type of
semi-synthetic derivative of podophyllotoxin, which has a high
liposolubility and may effectively pass through the blood-brain
barrier (19). However, it remains
unknown whether this drug, in combination with HD-MTX, may improve
the curative effect in the treatment of PCNSL.
In the present study, in the HD-MTX+Vm26 group, 12
patients (42.85%) achieved CR and 10 patients (35.71%) achieved PR,
while in the HD-MTX group, 7 patients (25%) achieved CR, 11
patients (39.29%) achieved PR, and the OR rate was 64.3%. The
difference of CR and OR between these two groups was notable, but
not statistically significant. This may be associated with the
small number of cases in the two groups. The median PFS was 22
months in the HD-MTX+Vm26 group and 12 months in the HD-MTX group,
and the difference between the two groups was statistically
significant (P=0.019). The median OS was 57 months in the
HD-MTX+Vm26 group and 28 months in the HD-MTX group (P=0.013). This
suggests that HD-MTX combined with Vm26 may effectively improve the
long-term curative effect of the treatment of patients with
PCNSL.
Ferreri et al (33) conducted a multicenter retrospective
study. Their results revealed 5 factors that were associated with
poor prognosis: i) Age >60 years old; ii) ECOG grades 2–4; iii)
increased serum LDH; iv) increased protein level in cerebrospinal
fluid; and v) tumors located in the deep areas of brain tissues,
including periventricular tissues, basal ganglia, brainstem and
cerebellum. Hu et al (47)
demonstrated that solitary or multiple tumor lesions were
independent factors that affected prognosis. Abrey et al
(48) indicated that only age and
physical condition were of significance for prognosis.
Results of the associated prognostic factors in the
present study revealed that age was an independent prognostic
factor in patients with PCNSL in the univariate and multivariate
analyses. The time to disease progression in patients with an age
of >60 years was ~10 months shorter compared with that in
patients with an age of ≤60 years. This was consistent with the
results of the majority of studies conducted outside China
(33). In addition, the present
study revealed that the difference in chemotherapy regimen is also
an independent prognostic factor. Compared with HD-MTX alone, the
HD-MTX+Vm26 regimen was not only useful in improving the CR rate
for its short-term curative effect (three courses), it was also
able to improve its long-term curative effect.
The present study revealed that the physical fitness
scores were not associated with prognosis. This may be due to the
small sample size in the present study. In addition, early studies
conducted in countries outside China primarily focused on the
association between physical fitness score and OS. However, the
present study focused on PFS duration. This may also lead to
different conclusions.
A previous study revealed that serum LDH level was
an independent prognostic factor for PCNSL: The prognosis of
patients with increased serum LDH level was poor (33). Results of the present study revealed
that serum LDH level exhibited no significant effect on PFS in
patients. Although there have been multiple studies on the
association between the survival time and the serum and urinary
β2-MG levels in patients with PCNSL worldwide, no consensus has
been obtained (49). The results of
the present study revealed that increased serum and urinary β2-MG
levels did not significantly affect the PFS of patients.
Increased protein levels in the cerebrospinal fluid
in patients with PCNSL suggest that these lesions may have affected
the cerebral pia mater (24).
However, it may also be induced by increased tumor load. These two
factors may lead to poor prognosis in patients (33). The present study revealed that the
increase in cerebrospinal fluid protein level led to a significant
difference in PFS, which were 20 and 10 months, respectively.
However, this is probably due to the small sample size, and the
difference was not statistically significant.
Results of a study conducted by Ferreri et al
(33) revealed that the 2-year
survival rate of patients with PCNSL who did not exhibit deep
structure involvement was ~42%, and that of patients who
demonstrated deep structure involvement was 28%. In the present
study, 60.7% of patients exhibited lesions that affected the deep
structures. Furthermore, statistical data revealed that this had no
significant association with the PFS of patients.
A previous study suggested that surgery did not
confer significant benefits to patients with PCNSL, and had risk of
damage to neural functions (28).
Surgical removal is not recommended in the majority of cases
(27). Cases of surgical operation
in patients with PCNSL were few, and little recent publications
have demonstrated that surgical resection could be established as a
prognostic factor in PCNSL (32,37). In
2012, the results of a German PCNSL SG-1 trial revealed that
surgical removal may extend the PFS of patients (50–52).
Fraser et al (40) considered
that surgical removal was advantageous to rapidly relieving
symptoms and decreasing glucocorticoid dose, which eliminated the
production of drug-resistant tumor clones. In addition, due to
modern surgical positioning technology, in 2012 a German primary
central nervous system lymphoma (PCNSL) SG-1 test revealed that,
surgical resection of PCNSL tumor lesions can prolong the
progression free survival time of patients (53,54). The
results of the present study revealed that median PFS was 20 months
in patients who underwent the surgical removal of lesions, while
PFS was 22 months in patients who underwent directional puncture
biopsy. The difference was not statistically significant between
these two surgical methods.
A previous study identified that patients with
multiple lesions exhibited shorter survival times (24), but this was not recognized. An early
study revealed that the number of lesions would affect the survival
time of patients, and the survival time of patients with a large
number of lesions is relatively shorter (42). However, this research result has not
been recognized. In the present study, univariate analysis of the
number of lesions also revealed that the difference in PFS between
patients with multiple lesions and patients with single lesion was
not statistically significant.
In the present study, the univariate analysis on the
number of lesions revealed that the difference in PFS between
patients with multiple lesions and patients with a single lesion
was not statistically significant.
In summary, the results of the present study
revealed that age and the treatment regimen were independent
prognostic factors of PCNSL. Compared with patients >60 years
old, patients who were ≤60 years old exhibited a significantly
prolonged PFS. At the same time, the PFS of patients who received
the combined treatment of Vm25+HD-MTX was significantly prolonged
when comparing with HD-MTX alone treatment. The difference in the
toxic reaction of chemotherapy was not statistically significant.
However, the sample size of the present study was small, and this
single-center retrospective study has limitations. Therefore, the
conclusions of the present study should be confirmed through
prospective multicenter clinical trials in the future.
The clinical data of the 56 patients with PCNSL were
retrospectively analyzed. It was revealed that: i) Age is an
independent prognostic factor in patients with PCNSL, in which
patients who were ≤60 years old exhibited longer PFS compared with
patients who were >60 year old; ii) the median PFS time was 69
months in patients who achieved CR following three courses of
treatment and 20 months in patients who did not achieve remission,
and the difference between these two was marked but not
statistically significant (P=0.061); iii) PFS time was 22 months in
the HD-MTX+Vm26 group and was 12 months in the MTX group, and
median OS time was 57 months in the HD-MTX+Vm26 group and was 28
months in the MTX group. Both PFS time and median OS time were
significantly different between these two groups. Therefore,
compared with HD-MTX alone, the combined treatment of HD-MTX+Vm26
exhibited a significantly improved long-term curative effect in the
treatment of PCNSL.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
YXW contributed to the conception and design of the
study and drafted the manuscript. YXW, YH, XPX, BBC, ZGL, YM, TLD
and QW acquired, analyzed and interpreted the data and revised the
manuscript critically for important intellectual content. All
authors read and approved the final version of the manuscript.
Ethics approval and consent to
participate
This study was conducted in accordance with the
declaration of Helsinki. This study was conducted with approval
from the Ethics Committee of Huashan Hospital Affiliated to Fudan
University. Written informed consent was obtained from the
participants.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ferreri AJ, Abrey LE, Blay JY, Borisch B,
Hochman J, Neuwelt EA, Yahalom J, Zucca E, Cavalli F, Armitage J
and Batchelor T: Summary statement on primary central nervous
system lymphomas from the Eighth International Conference on
Malignant Lymphoma, Lugano, Switzerland, June 12 to 15, 2002. J
Clin Oncol. 21:2407–2414. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Miller DC, Hochberg FH, Harris NL, Gruber
ML, Louis DN and Cohen H: Pathology with clinical correlations of
primary central nervous system non-Hodgkin's lymphoma. The
Massachusetts General Hospital experience 1958–1989. Cancer.
74:1383–1397. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Rubenstein JL, Hsi ED, Johnson JL, Jung
SH, Nakashima MO, Grant B, Cheson BD and Kaplan LD: Intensive
chemotherapy and immunotherapy in patients with newly diagnosed
primary CNS lymphoma: CALGB 50202 (Alliance 50202). J Clin Oncol.
31:3061–3068. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Jang JE, Kim YR, Kim SJ, Cho H, Chung H,
Lee JY, Park H, Kim Y, Cheong JW, Min YH and Kim JS: A new
prognostic model using absolute lymphocyte count in patients with
primary central nervous system lymphoma. Eur J Cancer. 57:127–135.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Joerger M, Huitema AD, Krähenbühl S,
Schellens JH, Cerny T, Reni M, Zucca E, Cavalli F and Ferreri AJ:
Methotrexate area under the curve is an important outcome predictor
in patients with primary CNS lymphoma: A
pharmacokinetic-pharmacodynamic analysis from the IELSG no. 20
trial. Br J Cancer. 102:673–677. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Hottinger AF, Alentorn A and Hoang-Xuan K:
Recent developments and controversies in primary central nervous
system lymphoma. Curr Opin Oncol. 27:496–501. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hoang-Xuan K, Bessell E, Bromberg J,
Hottinger AF, Preusser M, Rudà R, Schlegel U, Siegal T, Soussain C,
Abacioglu U, et al: Diagnosis and treatment of primary CNS lymphoma
in immunocompetent patients: Guidelines from the European
Association for Neuro-Oncology. Lancet Oncol. 16:e322–e332. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bhojwani D, Sabin ND, Pei D, Yang JJ, Khan
RB, Panetta JC, Krull KR, Inaba H, Rubnitz JE, Metzger ML, et al:
Methotrexate-induced neurotoxicity and leukoencephalopathy in
childhood acute lymphoblastic leukemia. J Clin Oncol. 32:949–959.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Cohen IJ: Neurotoxicity after high-dose
methotrexate (MTX) is adequately explained by insufficient folinic
acid rescue. Cancer Chemother Pharmacol. 79:1057–1065. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cher L, Glass J, Harsh GR and Hochberg FH:
Therapy of primary CNS lymphoma with methotrexate-based
chemotherapy and deferred radiotherapy: Preliminary results.
Neurology. 46:1757–1759. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Guha-Thakurta N, Damek D, Pollack C and
Hochberg FH: Intravenous methotrexate as initial treatment for
primary central nervous system lymphoma: Response to therapy and
quality of life of patients. J Neurooncol. 43:259–268. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Citterio G, Reni M and Ferreri AJ: Present
and future treatment options for primary CNS lymphoma. Expert Opin
Pharmacother. 16:2569–2579. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ferreri AJ, Reni M, Foppoli M, Martelli M,
Pangalis GA, Frezzato M, Cabras MG, Fabbri A, Corazzelli G,
Ilariucci F, et al: High-dose cytarabine plus high-dose
methotrexate versus high-dose methotrexate alone in patients with
primary CNS lymphoma: A randomised phase 2 trial. Lancet.
374:1512–1520. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Crivellari D, Lombardi D, Spazzapan S,
Veronesi A and Toffoli G: New oral drugs in older patients: A
review of idarubicin in elderly patients. Crit Rev Oncol Hematol.
49:153–163. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Geffen DB and Man S: New drugs for the
treatment of cancer, 1990–2001. Isr Med Assoc J. 4:1124–1131.
2002.PubMed/NCBI
|
|
16
|
Xu G, Yang M, Tong H, Mao L and Jin J:
High-dose methotrexate plus temozolomide as a salvage treatment in
metastatic central nervous system Lymphoma: Two cases report and
review of literature. Int J Clin Exp Med. 8:8222–8225.
2015.PubMed/NCBI
|
|
17
|
Ferreri AJ, Cwynarski K, Pulczynski E, et
al: The addition of thiotepa and rituximab to antimetabolites
significantly improves outcome in primary CNS lymphoma: The first
randomization of the IELSG-32 trial. Int Conference Malignant
Lymphoma. 2015.
|
|
18
|
Ponzoni M, Issa S, Batchelor TT and
Rubenstein JL: Beyond high-dose methotrexate and brain
radiotherapy: Novel targets and agents for primary CNS lymphoma.
Ann Oncol. 25:316–322. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Muggia FM: Teniposide: Overview of its
therapeutic potential in adult cancers. Cancer Chemother Pharmacol.
34 (Suppl):S127–S133. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Fan N, Chen BB, Zhu C, Xu XP, Lin ZG, Ma Y
and Zhang J: Comparison of the efficacy of MTX-alone and combined
with other drugs in treating CNS DLBCL. Fudan Univ J Med Sci.
41:81–87. 2014.
|
|
21
|
Swerdlow SH, Campo E, Harris NL, Jaffe ES,
Pileri SA, Stein H, Thiele J and Vardiman JW: WHO Classification of
tumors of Hematopoietic and Lymphoid Tissues. IARC; Lyon: pp.
4392008
|
|
22
|
Oken MM, Creech RH, Tormey DC, Horton J,
Davis TE, McFadden ET and Carbone PP: Toxicity and response
criteria of the Eastern Cooperative Oncology Group. Am J Clin
Oncol. 5:649–655. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Abrey LE, Batchelor TT, Ferreri AJ,
Gospodarowicz M, Pulczynski EJ, Zucca E, Smith JR, Korfel A,
Soussain C, DeAngelis LM, et al: Report of an international
workshop to standardize baseline evaluation and response criteria
for primary CNS lymphoma. J Clin Oncol. 23:5034–5043. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Jellinger KA and Paulus W: Primary central
nervous system lymphomas-an update. J Cancer Res Clin Oncol.
119:7–27. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Reni M, Ferreri AJ, Garancini MP and Villa
E: Therapeutic management of primary central nervous system
lymphoma in immunocompetent patients: Results of a critical review
of the literature. Ann Oncol. 8:227–234. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Michalski JM, Garcia DM, Kase E, Grigsby
PW and Simpson JR: Primary central nervous system lymphoma:
Analysis of prognostic variables and patterns of treatment failure.
Radiology. 176:855–860. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Gerstner ER and Batchelor TT: Primary
central nervous system lymphoma. Arch Neurol. 67:291–297. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Low S and Batchelor TT: Primary central
nervous system lymphoma. Semin Neurol. 38:86–94. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Haldorsen IS, Krossnes BK, Aarseth JH,
Scheie D, Johannesen TB, Mella O and Espeland A: Increasing
incidence and continued dismal outcome of primary central nervous
system lymphoma in Norway 1989–2003: Time trends in a 15-year
national survey. Cancer. 110:1803–1814. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Makino K, Nakamura H, Kino T, Takeshima H
and Kuratsu J: Rising incidence of primary central nervous system
lymphoma in Kumamoto, Japan. Surg Neurol. 66:503–506. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Eloranta S, Branvall E, Celsing F,
Papworth K, Ljungqvist M, Enblad G and Ekström-Smedby K: Increasing
incidence of primary central nervous system lymphoma but no
improvement in survival in Sweden 2000–2013. Eur J Haematol.
100:61–68. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Bayraktar S, Bayraktar UD, Ramos JC,
Stefanovic A and Lossos IS: Primary CNS lymphoma in HIV positive
and negative patients: Comparison of clinical characteristics,
outcome and prognostic factors. J Neurooncol. 101:257–265. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ferreri AJ, Blay JY, Reni M, Pasini F,
Spina M, Ambrosetti A, Calderoni A, Rossi A, Vavassori V, Conconi
A, et al: Prognostic scoring system for primary CNS lymphomas: The
International Extranodal Lymphoma Study Group experience. J Clin
Oncol. 21:266–272. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Bataille B, Delwail V, Menet E,
Vandermarcq P, Ingrand P, Wager M, Guy G and Lapierre F: Primary
intracerebral malignant lymphoma: Report of 248 cases. J Neurosurg.
92:261–266. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Gelabert-González M, Castro Bouzas D,
Serramito-García R, Frieiro Dantas C and Aran Echabe E: Primary
central nervous system lymphoma. Neurologia. 28:283–293. 2013.(In
English, Spanish). View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Deckert M, Engert A, Brück W, Ferreri AJ,
Finke J, Illerhaus G, Klapper W, Korfel A, Küppers R, Maarouf M, et
al: Modern concepts in the biology, diagnosis, differential
diagnosis and treatment of primary central nervous system lymphoma.
Leukemia. 25:1797–1807. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Thiel E, Korfel A, Martus P, Kanz L,
Griesinger F, Rauch M, Röth A, Hertenstein B, von Toll T,
Hundsberger T, et al: High-dose methotrexate with or without whole
brain radiotherapy for primary CNS lymphoma (G-PCNSL-SG-1): A phase
3, randomised, non-inferiority trial. Lancet Oncol. 11:1036–1047.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
O'Brien P, Roos D, Pratt G, Liew K, Barton
M, Poulsen M, Olver I and Trotter G: Phase II multicenter study of
brief single-agent methotrexate followed by irradiation in primary
CNS lymphoma. J Clin Oncol. 18:519–526. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Nabors LB, Portnow J, Ammirati M, Baehring
J, Brem H, Brown P, Butowski N, Chamberlain MC, Fenstermaker RA,
Friedman A, et al: Central nervous system cancers, version 1.2015.
J Natl Compr Canc Netw. 13:1191–1202. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Fraser E, Gruenberg K and Rubenstein JL:
New approaches in primary central nervous system lymphoma. Chin
Clin Oncol. 4:112015.PubMed/NCBI
|
|
41
|
Kim JE, Yoon DH, Kim S, Lee DH, Kim JH,
Yoon YH, Chi HS, Lee SW, Park CS, Huh J and Suh C: Relapse pattern
and prognostic factors for patients with primary central nervous
system lymphoma. Korean J Hematol. 47:60–66. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Prica A, Chan K and Cheung M: Combined
modality therapy versus chemotherapy alone as an induction regimen
for primary central nervous system lymphoma: A cost-effectiveness
analysis. Neuro Oncol. 16:1384–1391. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Correa DD, Maron L, Harder H, Klein M,
Armstrong CL, Calabrese P, Bromberg JE, Abrey LE, Batchelor TT and
Schiff D: Cognitive functions in primary central nervous system
lymphoma: Literature review and assessment guidelines. Ann Oncol.
18:1145–1151. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Batchelor TT: Primary central nervous
system lymphoma. Hematol Am Soc Hematol Educ Program. 2016:379–385.
2016. View Article : Google Scholar
|
|
45
|
Muirhead R, Murray EC, Bell SL, Stewart W
and James A: Is there a role for radiotherapy in the primary
management of primary central nervous system lymphoma? A
single-centre case series. Clin Oncol (R Coll Radiol). 25:400–405.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Swinnen LJ, O'Neill A, Imus PH, Gujar S,
Schiff D, Kleinberg LR, Advani RH, Dunbar EM, Moore D and Grossman
SA: Phase II study of rituximab given in conjunction with standard
chemotherapy in primary central nervous system lymphoma (PCNSL): A
trial of the ECOG-ACRIN cancer research group (E1F05). Oncotarget.
9:766–773. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Hu Y, Feng FY, Shi YK, Zhou LQ, Gu DZ and
Wang QL: Primary central nervous system lymphoma: A report of 28
patients. Zhonghua Zhong Liu Za Zhi. 26:375–378. 2004.(In Chinese).
PubMed/NCBI
|
|
48
|
Zhang ET, Inman CB and Weller RO:
Interrelationships of the pia mater and the perivascular
(Virchow-Robin) spaces in the human cerebrum. J Anat. 170:111–123.
1990.PubMed/NCBI
|
|
49
|
Abrey LE, Ben-Porat L, Panageas KS,
Yahalom J, Berkey B, Curran W, Schultz C, Leibel S, Nelson D, Mehta
M and DeAngelis LM: Primary central nervous system lymphoma: The
Memorial Sloan-Kettering Cancer Center prognostic model. J Clin
Oncol. 24:5711–5715. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Patrick LB and Mohile NA: Advances in
primary central nervous system lymphoma. Curr Oncol Rep. 17:602015.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
DeAngelis LM, Yahalom J, Heinemann MH,
Cirrincione C, Thaler HT and Krol G: Primary CNS lymphoma: Combined
treatment with chemotherapy and radiotherapy. Neurology. 40:80–86.
1990. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Bierman PJ: Surgery for primary central
nervous system lymphoma: Is it time for reevaluation? Oncology
(Williston Park). 28:632–637. 2014.PubMed/NCBI
|
|
53
|
Weller M, Martus P, Roth P, Thiel E and
Korfel A; German PCNSL Study Group, : Surgery for primary CNS
lymphoma? Challenging a paradigm. Neuro Oncol. 14:1481–1484. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Brastianos PK and Batchelor TT: Primary
central nervous system lymphoma: Overview of current treatment
strategies. Hematol Oncol Clin North Am. 26:897–916. 2012.
View Article : Google Scholar : PubMed/NCBI
|