Introduction
Colorectal cancer (CRC) is one of the most common
malignant tumors and ranked the second leading cause of
cancer-associated mortalities worldwide in 2018 (1). Current treatment methods targeting CRC
primarily include surgery assisted by radiotherapy and
chemotherapy; however, high metastasis and recurrence rates remain
primary causes of the high mortality associated with CRC (2). Cancer immunotherapy has received much
attention and has become the focus of cancer therapy.
Cytokine-induced killer (CIK) cell therapy has demonstrable
therapeutic benefits, preventing cancer recurrence, increasing the
quality of life of patients with cancer and extending the
progression-free survival period; therefore, it has been
extensively studied and applied in cancer therapies (3).
CIK cells are a group of heterogeneous cells
obtained from the coculture of human peripheral blood mononuclear
cells (PBMCs) and various cytokines, such as CD3McAb, interleukin
(IL)-2, interferon (INF)-γ and IL-1α (4). CIK cells expressing CD3 and CD56
membrane proteins function as natural killer (NK) T cells. These
CIK cells possess both the strong antitumor activity of T
lymphocytes and the non-major histocompatibility complex restricted
tumor-killing activity of NK cells (5–8). CIK
cells can specifically target the tumor and this may be associated
with the corresponding association between the chemokine expression
profile of cancer cells and the chemokine receptor (CKR) expression
profile in CIK cells (9).
Chemokines have similar structures and functions and
their molecular weights primarily range from 8–14 kDa. Chemokines
stimulate chemotactic functions in several cell types, including
neutrophils, lymphocytes and monocytes (10,11).
During the antitumor immune response of the body, chemokines can
mediate the targeted migration of immune cells in the blood and
lymph nodes to tumor locations to function in the tumor immune
response. This targeted migration has become a new focus of
research (12).
The present study investigated the concordance
between the chemokine expression profiles of tumor tissues from
patients with CRC and the CKR expression profiles of the surface of
CIK cells obtained from the peripheral blood from patients with
CRC. The present study aimed to increase the tumor-targeted
migration ability of CIK cells through regulation of the
chemokine-CKR axis in CRC.
Materials and methods
Clinical tumor and normal tissue
samples
Tissue samples were collected from a total of 36
patients with histologically confirmed CRC at The Affiliated
Hospital of Kunming University of Science and Technology, China.
Tumor stage was classified according to the 7th edition of the
Union for International Cancer Control (UICC)/American Joint
Committee on Cancer TNM staging system for CRC (13). Among the 36 patients, 24 patients
diagnosed with stage I–II and 12 patients diagnosed with stage
III–IV. Tumor tissue and the corresponding normal tissue were
collected at the same time from 7 patients who underwent surgical
resection. The remaining 29 patients underwent digestive endoscopic
resection and only tumor tissues were collected (Table I). Fresh tissues were washed with
RPMI-1640 medium within 30 min of removal (HyClone; GE Healthcare
Life Sciences) to remove traces of blood and the samples were cut
into pieces for RNA extraction immediately, or stored in liquid
nitrogen at −196°C. Blood samples from 20 patients with CRC
(male:female, 12:8; median age ± standard deviation, 57±19 years)
and 10 from healthy donors (male:female,5:5; median age ± standard
deviation, 55±10 years) were also collected to cultivate CIK cells
(Table I). The present study was
approved by The Ethics Committee of the First People's Hospital of
Yunnan Province and all patients provided written informed
consent.
 | Table I.Clinicopathological characteristics
of patients with CRC and healthy control individuals. |
Table I.
Clinicopathological characteristics
of patients with CRC and healthy control individuals.
|
| Sample used for
chemokine detection | Sample used for CIK
cell culture and chemokine receptor detection |
|---|
|
|
|
|
|---|
| Characteristic | Patients with CRC
(n=36) | Patients with CRC
(n=20) | Healthy control
(n=10) |
|---|
| Age, n (%) |
|
|
|
| ≤60
years | 20 (55.6) | 11 (55.0) | 6 (60.0) |
| >60
years | 16 (44.4) | 9 (45.0) | 4 (40.0) |
| Sex, n (%) |
|
|
|
|
Male | 18 (50.0) | 12 (60.0) | 5 (50.0) |
|
Female | 18 (50.0) | 8 (40.0) | 5 (50.0) |
| Tumor site, n
(%) |
|
|
|
|
Colon | 14 (38.9) | 6 (30.0) | – |
|
Rectum | 22 (61.1) | 14 (70.0) | – |
| UICC stage, n
(%) |
|
|
|
|
I–II | 24 (.66.7) | 12 (60.0) | – |
|
III–IV | 12 (33.3) | 8 (40.0) | – |
| Histologic grade, n
(%) |
|
|
|
|
Well | 1 (2.8) | 0 (0.0) | – |
|
Moderate | 24 (66.7) | 14 (70.0) | – |
|
Poor | 11 (30.5) | 6 (30.0) | – |
| Tumor size, cm, n
(%) |
|
|
|
| ≤5
cm | 30 (83.3) | 16 (80.0) | – |
| >5
cm | 6 (116.7) | 4 (20.0) | – |
| Lymph node
metastasis, n (%) |
|
|
|
|
None | 32 (88.9) | 18 (90.0) | – |
|
Present | 4 (11.1) | 2 (10.0) | – |
CRC cell lines
The CRC cell lines DLD1, HCT116, SW480, SW620, HT29,
LOVO and LS174T were purchased from the Typical Culture
Preservation Committee Kunming Cell Bank, Kunming Institute of
Zoology, Chinese Academy of Sciences. All of the cell lines were
cultured in RPMI-1640 (Hyclone; GE Healthcare) containing 10% fetal
bovine serum (FBS; Biological industries) and antibiotics (100 U/ml
penicillin and 100 µg/ml streptomycin, Biological Industries) in a
humidified incubator with 5% CO2 at 37°C.
RNA extraction and detection of
chemokine expression profiles in tissues and cells using
RT-qPCR
Total RNA was extracted from tissue samples and
cells using TRIzol reagent (Invitrogen; Thermo Fisher Scientific,
Inc.). The concentration and purity of total RNA samples were
verified using a NanoDrop 2000 (Thermo Fisher Scientific, Inc.).
RNA was reverse transcribed into cDNA using a GoScriptTM Reverse
Transcription system (Promega Corporation) according to the
supplied instructions. In brief, 5 µg RNA was mixed with the
primers and nuclease-free water completely and heated in a 70°C
heat block for 5 min. The RNA was then placed in ice water for ≥5
min. Then the reverse transcription reaction mixture was prepared.
The reverse transcription mix was combined with 5 µl of RNA and
primer mix and the following temperature protocol were used:
Annealing at 25°C for 5 min, extension at 42°C for 1h and then
inactivation of reverse transcriptase at 70°C for 15 min. The cDNA
was then used to measure the expression levels of CCL3, CCL5,
CCL17, CCL19, CCL21, CCL22, CXCL10, CXCL11 and CXCL12 in CRC tumor
and adjacent samples, as well as in CRC cells using SYBR Fast qPCR
Master mix (Kapa Biosystems; Roche Diagnostics) and a Roche
LightCycler 480 (Roche Diagnostics). The sequences of the primers
used for RT-qPCR are shown in Table
II (Takara Biotechnology Co., Ltd.). GAPDH was used for data
normalization or reference of chemokines expression level in the
CRC cell lines. The qPCR mixture was comprised of 10 µl 2× KAPA
SYBR FAST qPCR Master mix, 0.4 µM each primer, 0.5 µl cDNA and
PCR-grade water in a final reaction volume of 20 µl. The
thermocycling conditions were as follows: Pre-incubation at 95°C
for 3 min, amplification at 95°C for 10 sec, 56°C for 20 sec and
extension/data acquisition at 72°C for 30 sec, for a total of 40
cycles. The melt curve was as follows: 95°C for 5 sec, 65°C for 1
min, with a continuous increase in temperature from 65–97°C at the
rate of 0.02°C/sec with 10 signal acquisitions per degree; and
cooling at 40°C for 10 sec. The 2−ΔΔCq method was used
for data analysis (14).
 | Table II.Primers used for reverse
transcription-quantitative PCR. |
Table II.
Primers used for reverse
transcription-quantitative PCR.
| Gene | Sequence,
5′→3′ |
|---|
| GAPDH |
|
Forward |
TGACTTCAACAGCGACACCCA |
|
Reverse |
CACCCTGTTGCTGTAGCCAAA |
| CCL3 |
|
Forward |
TGTTGCCAAACAGCCACAC |
|
Reverse |
CAGAGCAAACAATCACAAACACAC |
| CCL5 |
|
Forward |
TCCCACAGGTACCATGAAGGTC |
|
Reverse |
GCAATGTAGGCAAAGCAGCAG |
| CCL17 |
|
Forward |
CAGAGGGACCTGCACACAGA |
|
Reverse |
TTCAGCTTTCTAAGGGGAATGG |
| CCL19 |
|
Forward |
ACCAGGCTTCCAGCTCCTCT |
|
Reverse |
ACCAGGCGGCTTTATTGGT |
| CCL21 |
|
Forward |
GCCACACTCTTTCTCCTGCTTT |
|
Reverse |
ACTCTCCCTCCTCGGTCTCTCT |
| CCL22 |
|
Forward |
GGTATTTGAACCTGTGGAATTGGAG |
|
Reverse |
CAGGCCCTGGATGACACTGA |
| CXCL10 |
|
Forward |
TGCAAGCCAATTTTGTCCAC |
|
Reverse |
GACCTTTCCTTGCTAACTGCTTTC |
| CXCL11 |
|
Forward |
GCTGTGATATTGTGTGCTACAGTTG |
|
Reverse |
TTGGGTACATTATGGAGGCTTTC |
| CXCL12 |
|
Forward |
CCCTGCTTACCCGCAAAA |
|
Reverse |
CTTCAGAGGCAATCACAAAACC |
Extraction of CIK cells
Blood samples from healthy donors or patients with
CRC were processed using Ficoll-Hypaque density gradient
centrifugation (Beijing Solarbio Science and Technology Co., Ltd.)
to obtain PBMCs. After washing in RPMI-1640 medium,
2×106 cells were resuspended in RPMI-1640 medium
containing 10% FBS, 2 mM glutamine, 100 IU/ml penicillin and 100
IU/ml streptomycin in a cell culture flask. After incubation for 24
h with 1,500 IU/ml INF-γ (Shanghai ChemoWanbang Biopharma Co.,
Ltd.; cat. no. S10980084), 5,000 IU/ml IL-2 (Beijing T&L
Biotechnology Co., Ltd; cat. no. TL-104) and 100 ng/ml anti-CD3
antibodies (1:10,000; Wuhan Institute of Biological Products Co.,
Ltd.; cat. no. S19990012) were added and maintained at 37°C in
humidified atmosphere of 5% CO2 for 2 days. Then fresh
culture medium containing 5,000 IU/ml recombinant human (rh)IL-2
was added every 2 to 3 days. In some assays, the recombinant
chemokine ligand CCL21 (100 ng/ml) or CXCL11 (10 ng/ml) (PeproTech,
Inc.) was added during the culture process. The cells were
maintained at 37°C in a humidified atmosphere of 5% CO2.
After 13 days, cells were harvested for flow cytometry or Transwell
analysis.
Detection of the targeted migration
ability of CIK cells using a Transwell assay
To assess the effects of CRC cells with different
expression levels of chemokines on the targeted migration ability
of CIK cells, DLD1 (low expression of chemokine CCL21 and CXCL11),
SW480 (high expression of chemokine CCL21) and HT29 (high
expression chemokine of CXCL11) were used. In brief, 200 µl CIK
cell suspension containing 5×105 cells was inoculated in
the top chamber of a 24-well Transwell plate (5 µm; Corning Inc.)
and 600 µl CRC cell suspension (DLD1, SW480 or HT29) containing
5×105 cells was added to the bottom chamber and placed
in an incubator at 37°C with 5% CO2 for 24 h. Then the
number of CIK cells that had migrated to the bottom chamber was
imaged and counted using a flow cytometer. To assess the effects of
different concentrations of recombinant human chemokine proteins on
the targeted migration ability of CIK cells, 200 µl CIK cell
suspension containing 5×105 cells was inoculated in the
top chamber of a Transwell plate and 600 µl of RPMI-1640 contained
10% FBS and either rhCCL21 (final concentration 100 or 10 ng/ml) or
rhCXCL11 (final concentration 10 or 0.1 ng/ml) was added to the
bottom chamber. To assess the effects of the chemokine pretreatment
of CIK cells on the expression levels of CKRs and cell-targeted
migration ability, after culturing the CIK cells for 12 days,
either rhCCL21, (final concentration of 100 ng/ml) or rhCXCL11
(final concentration 10 ng/ml) was added to the CIK cell culture
system. After culturing the cells for another 2 days, 200 µl CIK
cell suspension containing 5×105 cells was collected and
inoculated in the top chamber of a Transwell plate and 600 µl of
RPMI-1640 containing 10% FBS and either rhCCL21 (final
concentration 100 ng/ml) or rhCXCL11 (final concentration 10 ng/ml)
was added to the bottom chamber. The control was composed of CIK
cells without pretreatment with the recombinant chemokine. Cells
were placed in an incubator at 37°C with 5% CO2 for 24
h. After the top chamber of the Transwell was discarded, the number
of CIK cells that had migrated to the bottom chamber was imaged and
counted using a flow cytometer.
Detection of the CKR expression
profiles in CIK cells using flow cytometry
For CIK cells harvested on days 7, 14, 21 and 28
were washed twice with washing buffer (PBS buffer containing 0.5%
BSA), blocked with blocking buffer (PBS containing 2% BSA) for 10
min at 4°C and washed twice again. Then the washed CIK cells were
stained with 5 µl of the following monoclonal antibodies in 100 µl
blocking buffer (PBS containing 1% BSA) for 30 min at 4°C, washed
twice, resuspended in 100 µl PBS buffer and analyzed via flow
cytometry. The following antibodies were used and diluted 1:20 in
blocking buffer (PBS containing 1% BSA): CD3-FITC (cat. no.
11-0036-42), CD56-APC (cat. no. 17-0567-42), CCR4-PE (cat. no.
12-1949-41), CCR5-PE (cat. no. 12-1956-41), CCR7-PE (cat. no.
12-1979-42), CXCR3-PE (cat. no. 12-1839-42) and CXCR4-PE (cat. no.
12-9999-41) antibodies, as well as the corresponding isotype
controls IgG2a-PE (cat. no. 12-4321-80), IgG1a-PE (cat. no.
12-4714-82), IgG1a-FITC (cat. no. 11-4714-81) and IgG1a-APC (cat.
no. 17-4714-82). All antibodies were purchased from eBioscience;
Thermo Fisher Scientific, Inc. Data were obtained using a MoFlo
flow cytometer (BeckmanCoulter, Inc.) and analyzed using Summit
version 5.2 (Beckman Coulter, Inc.) and FlowJo version 10 software
(Becton, Dickinson and Company).
Statistical analysis
GraphPad software version 5.0 (GraphPad Software)
was used for statistical analysis. Analyses were performed using an
unpaired Student's t-test with Welch's correction and one-way or
two-way ANOVA with Bonferroni's correction or Tukey-Kramer post-hoc
tests where appropriate. The statistical test used for each figure
is described in the corresponding figure legend. P<0.05 was
considered to indicate a statistically significant difference.
Results
Different levels of chemokine
expression were detected in CRC tissues
The RT-qPCR results revealed significantly higher
expression levels of the chemokine ligands CCL3, CCL17, CCL22 and
CXCL10 in cancer tissues (n=36) compared with adjacent normal
tissues (n=7) (P<0.05, P<0.01, P<0.001 and P<0.01,
respectively), whereas other chemokine ligands (CCL5, CCL19, CCL21,
CXCL11 and CXCL12) exhibited no significant differences between CRC
tumor tissues and adjacent normal tissues (Fig. 1).
Chemokine expression profiles in
different patients with CRC and CRC cell lines showed both common
characteristics and donor-to-donor variation
The chemokine expression levels were evaluated in
both the colon tumor tissues and corresponding adjacent normal
tissues of 7 patients with CRC. In addition, 7 CRC cell lines
(174T, HCT116, DLD1, SW620, LOVO, SW480 and HT29) were also
evaluated. The data demonstrated that the expression levels of
certain chemokines were concordant in the cancer tissues of
different patients with CRC and different CRC cell lines. For
example, CXCL11 expression levels were significantly higher in 6/7
cases of cancer tissues from patients with CRC compared with normal
tissues (all P<0.001; Fig. 2A)
and highly expressed in 5/7 CRC cell lines chemokines were
upregulated more than 5-fold compared with GAPDH (Fig. 2B). Similarly, the expression levels
of CCL3 were significantly higher in 4/7 cancer tissues compared
with adjacent normal tissues (P<0.001; Fig. 2A) and 3/7 CRC cell lines relative to
GAPDH expression (>2-fold of the CCL3/GAPDH expression ratio was
the cut off value used. The expression ratio of CCL3/GAPDH in
SW620, SW480 and HT29 was 2.00±0.15, 8.85±0.35 and 3.45±0.39,
respectively; Fig 2B). In addition,
CXCL10 expression levels were higher in 7/7 cases of CRC compared
with adjacent normal tissue (CRC tissue vs. adjacent normal tissue;
6/7 samples P<0.001 and 1/7 sample P<0.01) and 3/7 CRC cell
lines (LOVO, SW480 and HT29) compared with GAPDH exhibited a 2-fold
change in the CCL3/GAPDH expression ratio. The ratio of
CXCL10/GAPDH in LOVO, SW480 and HT29 was 3.69±0.23, 3.42±0.37 and
2.41±0.09, respectively. The expression levels of CCL19 did not
differ between all the cancer tissues and normal tissues. The same
result was also observed in 6/7 CRC cell lines (with the exception
of SW480). However, the chemokine expression profiles in cancer
tissues from different patients with CRC and in different CRC cells
showed heterogeneity (Fig. 2A and
B).
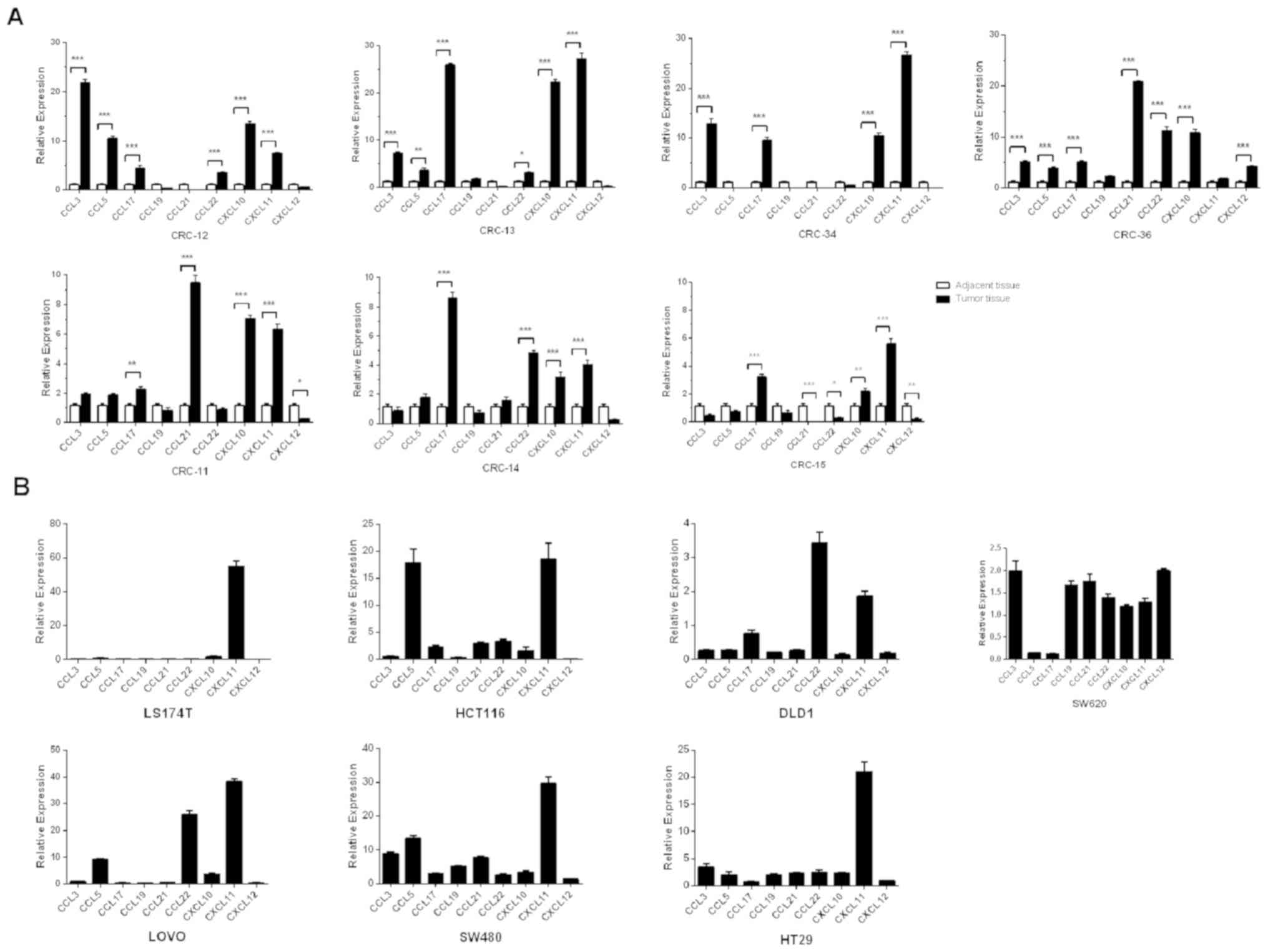 | Figure 2.Chemokine expression profiles of
patients with CRC and CRC cell lines. The expression levels of 9
chemokine ligands were detected in tumor and the paired adjacent
normal samples from (A) 7 patients with CRC(CRC-11, CRC-12, CRC-13,
CRC-14, CRC-15, CRC-34 and CRC-36) and (B) 7CRC cell lines (DLD1,
HCT116, SW620, SW480, HT29, LOVO and LS174T) using reverse
transcription-quantitative PCR. The expression levels of 3
chemokine ligands (CXCL11, CCL3 and CCL19) were consistently
upregulated in the majority of the CRC patients compared with the
adjacent normal samples and also in CRC cell lines relative to the
house-keeping gene GAPDH. Expression levels of CCL19 were not
significantly different in tumor vs. paired adjacent normal samples
and CRC cell lines relative to the house-keeping gene GAPDH.
However, the expression profiles of chemokines between patients
with CRC and CRC cell lines were heterogeneous. The comparisons
were performed using two-way ANOVA analysis and Bonferroni's
correction. *P<0.05; **P<0.01; ***P<0.001. CRC, colorectal
cancer. |
CXCR3 and CXCR4 were more highly
expressed on the surface of CIK cells derived from patients with
CRC compared with those derived from healthy controls
On days7, 14, 21 and 28 of culture of peripheral
blood CIK cells from patients with CRC (n=20) and healthy
individuals (n=10), CKR expression levels in CIK cells were
detected using flow cytometry. The results indicated that the CIK
cells derived from patients with CRC or healthy individuals showed
similar CKR expression profiles. CXCR3 and CXCR4 had higher
expression levels on CIK cells compared with isotype, while the
expression profiles of CCR4, CCR5 and CCR7 did not show significant
changes compared with isotype (Fig.
3A). At the 4 time points examined, the expression levels of
both CXCR3 and CXCR4 were significantly higher in CIK cells from
patients with CRC compared with those from healthy individuals
(P<0.001). However, the expression levels of CCR4, CCR5 and CCR7
in CIK cells from patients with CRC and healthy individuals
differed over time; CCR4 expression levels on day 7 (P<0.01) and
day 28 (P<0.05), CCR5 on day 28 (P<0.05) and CCR7 on day 7
(P<0.01), 21 (P<0.05) and 28 (P<0.05) were significantly
higher in CIK cells from patients with CRC compared with those from
healthy individuals (Fig. 3B).
 | Figure 3.Expression levels of chemokine
receptors on CIK cells generated from patients with CRC and healthy
donors. (A) Analysis of the expression levels of CCR4, CCR5, CCR7,
CXCR3 and CXCR4 on CIK cells via flow cytometry revealed that CXCR3
and CXCR4 had higher expression levels on CIK cells compared with
isotype, expression profiles of CCR4, CCR5 and CCR7 did not show
significant changes compared with isotype. (B) Dynamic changes of
chemokine receptors CCR4, CCR5, CCR7, CXCR3 and CXCR4 of CIK cells
on D7, 14, 21 and 28 were detected in patients with CRC and HD CIK
cells via flow cytometry. The result revealed that expression
levels of CXCR3 and CXCR4 were significantly higher on CIK cells
cultured from CRC compared with HD at all the time points analyzed,
while the expression levels of CCR4, CCR5 and CCR7 were
significantly higher on CIK cells cultured from CRC compared with
HD at different time points analyzed (CCR4 on D7 and D28; CCR5 on
D28; CCR7 on D7, D21 and D28). All comparisons were performed using
unpaired Student's t-test with Welch's correction. *P<0.05;
**P<0.01; ***P<0.001. CRC, colorectal cancer; HD, healthy
donors; CIK, cytokine-induced killer cell. |
Expression levels of CKRs on the
surface of CIK cells decreased gradually during the expansion
process
Analyses of the changing trends of the CKR
expression profiles of CIK cells from patients with CRC (n=20) over
time showed that CCR4 and CXCR4 expression levels peaked on day 7
(11.06±2.020 and 53.89±5.539%, respectively); then, CCR4 and CXCR4
expression levels gradually decreased and reached their minimum
values on day 28 (3.697±0.839 and 14.630±2.112%, respectively). The
changes in expression levels of day 28 and day 7 in CCR4 and CXCR4,
respectively were significant (P=0.015 for CCR4 and P<0.001 for
CXCR4). Expression levels of CCR5, CCR7 and CXCR3 were detected on
day 7 (5.165±1.673, 8.663±1.810 and 57.040±5.486%, respectively)
and then these expression levels reached their peak on day 14
(7.855±2.521, 9.620±2.410 and 75.140±5.319%, respectively) and then
gradually decreased. On day 28, the expression levels of CCR5, CCR7
and CXCR3 returned to levels similar to those on day 7
(4.913±1.006, 6.209±1.415 and 61.060±3.577%, respectively);
however, the changes in expression levels of these 3 CKRs over time
were not significant (Fig. 4).
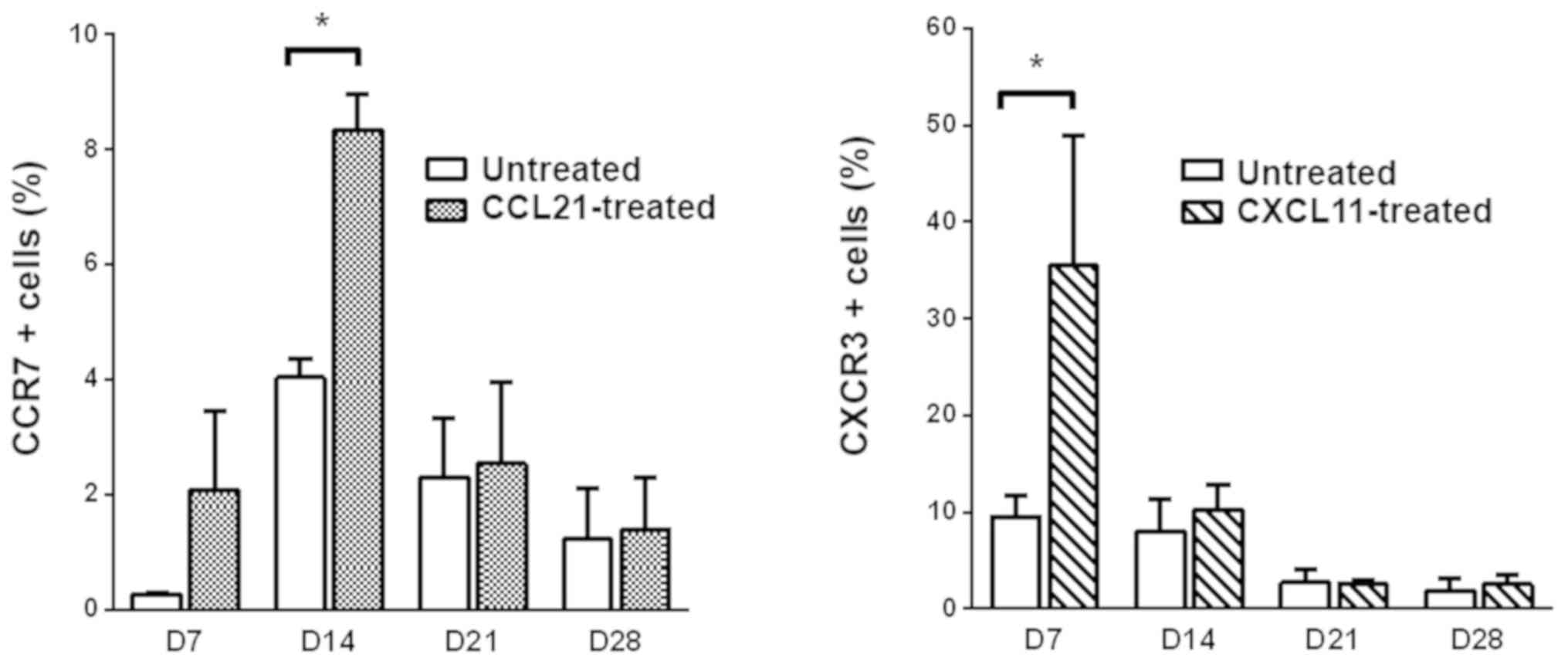
CKR expression levels could be boosted
by adding exogenous recombinant chemokines
CCL21 and CXCL11 were separately added to the CIK
cell culture system on days 5, 12, 19 and 26. After culturing for
another 2 days (to days 7, 14, 21 and28, respectively), the CIK
cells were collected and the expression levels of CCR7 (the
corresponding ligand of CCL21) and CXCR3 (the corresponding ligand
of CXCL11) (15) were detected using
flow cytometry. CCR7 expression levels on day 14 were significantly
higher in CIK cells pretreated with CCL21 compared with CIK cells
without pretreatment (8.350 vs. 4.235%; P<0.05), whereas CCR7
expression levels were not significantly increased at the 3other
time points. CXCR3 expression levels were significantly higher on
day 7 in CIK cells pretreated with CXCL11 compared with CIK cells
without pretreatment (35.500 vs. 9.570%; P<0.05), whereas CXCR3
expression levels were not significantly increased at the 3 other
time points (Fig. 5).
Boosting CKR expression levels
enhances the tumor-targeted trafficking ability of CIK cells
To explore the trafficking ability of CIK cells, a
Transwell assay was performed and the CIK cells that migrated into
the lower chamber were observed under a microscope (Fig. 6A) or counted using flow cytometry
(Fig. 6B-E). When the bottom chamber
of the Transwell was inoculated with DLD1 cells with low CCL21
expression levels (CCL2-L) and SW480 cells with high CCL21
expression levels (CCL2-H), DLD1 cells had a significantly reduced
effect on the chemotactic ability of CIK cells compared with that
of SW480 cells (41,430±575 vs. 101,300±8,250; P<0.05; Fig. 6B). Similar results were obtained in
experiments using different concentrations of recombinant CCL21 to
influence the chemotactic ability of CIK cells. When the CCL21
concentrations in the bottom chamber of the Transwell were 10 ng/ml
and 100 ng/ml, 10 ng/ml CCL21 had a significantly lower effect on
the chemotactic ability of CIK cells compared with 100 ng/ml CCL21
(5,400±900 vs. 28,450±3,050; P<0.05; Fig. 6C). When the CCL21 concentration in
the bottom chamber of the Transwell was 100 ng/ml, a significantly
greater number of CIK cells cultured to day 14 migrated from the
top to the bottom chamber pretreated with exogenous CCL21 vs.
without CCL21 pretreatment (132,000±12,000 vs. 28,450±3,050;
P<0.05; Fig. 6C).
When DLD1 cells with low CXCL11 expression levels
(CXCL11-L) and HT29 cells with high CXCL11 expression levels
(CXCL11-H) were separately inoculated in the bottom chamber in the
Transwell assay, DLD1 cells had a significantly reduced effect on
the chemotactic ability of CIK cells compared with HT29 cells
(40,500±1,500 vs.79,250±2,750; P<0.01; Fig. 6D). Similar results were also obtained
using different concentrations of recombinant CXCL11 to influence
the chemotactic ability of CIK cells. The addition of 0.1 ng/ml
CXCL11 to the bottom chamber had a significantly reduced effect on
the chemotactic ability of CIK cells compared with of 10 ng/ml
CXCL11 (51,250±1,750 vs. 84,000±600; P<0.05; Fig. 6E). At a CXCL11 concentration of 10
ng/ml in the bottom chamber, there was no significant increase in
the number of CIK cells that migrated to the bottom from the top
chamber pretreated with exogenous CXCL11 and cultured to day 14 vs.
without CXCL11 pretreatment (114,500±11,500 vs. 84,000±6,000,
respectively; P>0.05) (Fig.
6E).
Discussion
Due to their strong cell-killing capacity and broad
antitumor spectrum, CIK cells can effectively kill
chemotherapy-resistant tumor cells. CIK cells exhibit clear
efficacy regarding the clearance of minimal residual lesions of
tumors and have few side effects; therefore, CIK cells have become
an extensively used treatment for adoptive cellular immunotherapy
(16).
CRC is a common malignant tumor with global high
morbidity and mortality rates. Most patients with CRC are already
in the late stage at the time of diagnosis and because late-stage
tumor cells have already spread or metastasized in patients with
late-stage CRC, conventional surgery have little therapeutic
benefit. In addition, patients may not be able to tolerate
traditional radio-or chemotherapy; therefore, CIK cell treatment
may improve the quality of life of these middle- to late-stage
patients and extend patient survival time (17,18). In
the clinic, however, patients with CRC who have received CIK cell
treatment usually present with different clinic efficacies, whereas
a number of patients do not experience any therapeutic benefit
(3).
How to effectively promote the infiltration of
immune-reactive cells into tumors is an important focus of cancer
research in order to increase the efficacy of antitumor
immune-therapies. Successful antitumor immunotherapy enhances the
tumor-targeted trafficking ability of immune cells but requires
effective methods for in vitro activation of these cells (19).
Chemokines are important regulatory factors that
direct immune-reactive cells to tumors. By binding to corresponding
CKRs on the target cell membrane, chemokines can induce the
targeted migration of target cells (20). The interaction between chemokines
released in an abnormal cancer microenvironment and CKRs on the
surface of CIK cells is an important factor that affects the
tumor-targeted migration ability of CIK cells (21). The concordance between these two
variables directly affects the treatment effects of CIK cells
(22–24). Currently, however, the CKR expression
profiles of CIK cells derived from patients with autologous CRC and
the underlying molecular mechanisms influencing the tumor-targeted
migration ability of CIK cells via CKR expression profiles remain
unclear. In the present study, the chemokine expression profiles in
tumor tissues from patients with CRC and the CKR expression
profiles of the surface of CIK cells derived from the same donors
were detected. The corresponding association between these profiles
was analyzed to understand the effects of the concordance between
the chemokine expression profiles in the cancer microenvironment
and the CKR expression profiles on CIK cells on the tumor-targeted
migration ability of CIK cells. In addition, a simple modification
of the chemokine-CKR axis for increasing the tumor-targeted
migration ability of CIK cells was also investigated.
First, the chemokine expression profiles in CRC
tissues and adjacent normal tissues were examined, as well as in
CRC cell lines. The results reported that the chemokine expression
profiles in the tumor tissues from patients with CRC and different
CRC cell lines had common characteristics. CXCL10, CXCL11 and CCL3
were overexpressed in most CRC tumor samples and CRC cell lines. In
contrast, CCL19 was expressed at low levels in both the tumor and
adjacent normal samples. This effect might be due to the regulation
of cancer cell proliferation and invasion through the interaction
with CXCR3 and CXCR7 (25). However,
the chemokine expression profiles of tumor tissues from patients
with CRC and in different CRC cell lines had significant
heterogeneity. Due to the fact that the different CRC cell lines
were derived from CRC tumor tissues from different patients with
CRC, the high heterogeneity of chemokine expression profiles in
patients with CRC may be associated with pathological factors, such
as tumor stage and metastasis. Whether this heterogeneity affects
the response to CIK cell treatment requires evidence from clinical
observational data obtained from large sample sizes.
Next, the present study examined the CKR expression
profiles on the surface of CIK cells from patients with CRC and
healthy individuals. The dynamic changes in CKR expression levels
in CIK cells during the expansion process were also monitored. The
results showed that CIK cells from the two sources had similar CKR
expression profiles: CXCR3 and CXCR4 were notably more highly
expressed on the surface of CIK cells compared with CCR4, CCR5 and
CCR7. In addition, the CKR expression levels on the surface of CIK
cells derived from patients with CRC were significantly higher
compared with those derived from healthy individuals. The
differences in expression levels of CXCR3 and CXCR4 were the most
notable. These results were inconsistent with those of our previous
work (26), which demonstrated that
the increased expression levels of CKR on the surface of CIK cells
did not differ significantly between patients with CRC and healthy
individuals. It was hypothesized that certain factors, such as the
culture condition, ethnicity and pathological stage, might be
associated with these differences. The present results also
differed from those described by Wang et al (9), who reported a reduction in the
expression levels of CKR on the surface of CIK cells in patients
with CRC compared with cells derived from healthy individuals. It
was hypothesized that this discrepancy between the present study
and the aforementioned study may be due to the disparate in vitro
activation times of the CIK cells used for CKR detection, donor
resources, such as UICC stage and other parameters. Therefore,
future studies with larger sample sizes are needed.
It is noteworthy that all the CKR expression levels
declined during the CIK cell culture process in both the present
study and in the other two aforementioned previous reports
(9,26). Therefore, due to these consistent
results, the present study aimed to enhance CKR expression levels
during the course of CIK cell culture and enhance CIK cell
trafficking ability. Further analyses between the chemokine
expression profiles in tumor tissues from patients with CRC and the
CKR expression profiles on the surface of CIK cells derived from
the same patients demonstrated that the chemokine and CKR
expression profiles were associated. CXCR3 expression levels were
higher on the surface of CIK cells and the expression of its
corresponding ligand, CXCL10, was also higher in CRC tumor tissues
compared with normal tissues. In addition, the expression levels of
CCR4 were higher on the surface of CIK cells and the expression
levels of its corresponding ligands, CCL3 and CCL22, were also
higher in CRC tumor tissues compared with adjacent normal tissues.
It was hypothesized that the corresponding association between
chemokines and CKRs was important for allowing CIK cells to migrate
to tumor tissue in patients with CRC. Consistent with the present
study, Wang et al (9)
demonstrated that expression levels CXCL10 was elevated in CRC
tumor tissues compared with paracancerous tissues and that the
expression levels of its corresponding ligand, CXCR3, were also
increased in CIK cells derived from patients with CRC compared with
PBMCs before activation. However, no corresponding association
between chemokine and CKR expression profiles was observed in the
present study. For example, CXCR4 expression levels were elevated
on the surface of CIK cells but the expression levels of its
corresponding ligand, CXCL12, were lower in CRC tumor tissues
compared with paracancerous tissues. In addition, CCR7 expression
levels were higher on the surface of CIK cells, but the expression
levels of its corresponding ligands, CCL19 and CCL21, were not
significantly different between CRC tumor tissues and normal
tissues. The discrepancy between cytokine expression levels from
tumor tissue and CKRs from CIK cells might impair CIK cell
tumor-targeted migration and limit the clinic efficacy of CIK cells
in CRC.
The expression levels of chemokines in CRC cells
partially determine the targeted migration ability of CIK cells.
Several studies have indicated that chemokines induce the targeted
migration of lymphocytes (27–30).
Therefore, strategies to alter the local concentration of
chemokines in tumors, increase the expression levels of CKR on the
surface of immune cells and utilize the chemokine-CKR axis to
increase tumor-targeted migration and infiltration of immune cells,
such as lymphocytes, NK cells and CIK cells, into tumors may
improve our theoretical understanding for treatment of cancer. For
example, Pevida et al (31)
added the chemokine ligands CCL3 and CCL5 to NCTC2472 mouse
fibrosarcoma cells to increase the expression levels of the
corresponding receptor CCR1 on the cell surface. In our previous
work (26), CD3/CD28 magnetic beads
were added to a CIK culture system to stimulate the expression of
CCR5, CCR7 and CXCR3 on the CIK cell surface.
As aforementioned, almost all the CKRs declined
during the expansion process in the present study. To boost CKR
expression levels in CIK cells, 2 chemokines (CCL21 and CXCL11)
were selected. The results indicated that CCL21 or CXCL11
pretreatment significantly increased the expression levels of CCR7
or CXCR3 on the CIK cell surface. These results suggest that the
addition of exogenous recombinant chemokines during the culture
process of CIK cells might increase the expression levels of
ligands corresponding to chemokines on the surface of CIK cells.
However, it was also observed that the expression levels of CCR7
were significantly higher at day 14 on the surface of CIK cells
with CCL21 pretreatment vs. without CCL21 pretreatment, whereas
CXCR3 expression levels were significantly higher at day 7 on the
surface of CIK cells with CXCL11 pretreatment vs. without
pretreatment. These results suggested that when adding exogenous
recombinant chemokines to increase the expression levels of ligands
corresponding to chemokines on the surface of CIK cells, different
time points should be selected based on disparate chemokines.
Furthermore, the present study reported that
increased CKR expression levels on the surface of CIK cells could
enhance the tumor-targeted migration ability of CIK cells. A
significantly greater number of CIK cells had migrated to the
bottom Transwell chamber after pretreatment with CCL21 vs. without
treatment. However, CIK cells cultured to day 14 pretreated with
CXCL11 did not exhibit increased CXCR3 expression levels because
the CXCR3 expression levels in CIK cells can be boosted
significantly with CXCL11 on day7 but no statistical difference was
observed at other time points. Therefore, the observed chemotactic
ability was not significantly increased. These results further
demonstrated that the tumor-targeted migration ability of CIK cells
is chemokine-CKR dependent and that it was feasible to increase the
tumor-targeted migration ability of CIK cells with chemokine
pretreatment during the CIK culture process at the proper time
point.
There are some limitations of the present study. CIK
cells are a heterogeneous population, which compose various
subsets, such as CD3(+)CD56(+) CIK, CD3(+) CD56(−) T and
CD3(−)CD56(+) NK cells (32).
CD3(+)CD56(+) CIK cells appear to possess the most potent
cytotoxicity and high impact on adoptive cellular immunotherapy
(33). However, other subtypes of
CIK cells may have different immunologic functions. For example,
alloreactivity against human leukocyte antigen-mismatched PBMC is
restricted to CD3(+)CD56(−) CIKs (34). Therefore, the association between
chemokines and CKRs from various subsets of total CIK cells should
be considered in future research. Then, the relative expression
levels of chemokines in CRC cell lines were compared with GAPDH as
a reference in the present study. However, GAPDH at the mRNA level
in CRC sample was increased significantly compared with the paired
non-cancerous part (35). Therefore,
it will be better to use a normal control cell line to measure
differential CKR expression in CRC cell lines. Next, 2 chemokines
(CCL21 and CXCL11) were selected to boost CKR expression levels
during CIK cell culture process in the present study. However,
whether these two chemokines affect the proliferation of CIK cells
requires further study. Finally, the present study only measured
chemokine expression levels in CRC and adjacent normal tissue using
RT-qPCR. It would be better to perform western blots to verify the
results of the present study at the protein expression level.
Taken together, the results of the study
demonstrated that CXCL10, CXCL11 and CCL3 expression levels were
significantly higher in CRC tumor tissues compared with adjacent
normal tissue. However, the levels of all CKRs of CIK cells,
especially CCR4, CXCR4 and CXCR3, decreased considerably during the
course of CIK cell expansion. Notably, re-stimulating CIK cells
with chemokines CCL21 at day 14 and CXCL11 at day 7 significantly
increased the corresponding CKR expression levels on the surface of
CIK cells and enhanced tumor-targeted trafficking in vitro. To the
best of our knowledge, the present study is the first to show that,
although adding exogenous recombinant chemokines to increase the
impaired CKRs expression on the surface of CIK cells, different
time points should be selected based on disparate chemokines.
Therefore, evaluating the expression levels of chemokines in the
CRC tumors and stimulating with proper exogenous recombinant
chemokines at proper time point could increase corresponding CKR
expression levels, enhance CIK cell tumor-targeted trafficking and
improve clinic efficacy.
Acknowledgements
Not applicable.
Funding
The present study was supported by The National
Natural Science Foundation of China (grant nos. 81460463 and
81502556), The Medical Academic Talents Cultivation Foundation for
Health Commission of Yunnan Province (grant no. D-201642) and The
Kunming Key Laboratory of Tumor Molecular and Immune Prevention
Foundation (grant no. 2018-1-A-17334).
Availability of data and materials
All the datasets generated and analyzed in the
present study are included in this published article.
Authors' contributions
YZ and HT designed the study and wrote the
manuscript. XY, HT and QG carried out the concepts, definition of
intellectual content and reviewed the manuscript. YZ, JL and JW
performed the experiments. LW and JZ collected the data. XY, DL and
JF analyzed and interpreted the data. All the authors read and
approved the final manuscript.
Ethics approval and consent to
participate
The present study was approved by The Ethics
Committee of the First People's Hospital of Yunnan Province and all
patients gave written informed consent. The approval number was
2018GJ111.
Patient consent for publication
Not applicable.
Competing interests
All the authors declare that they have no competing
interests.
References
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Dekker E, Tanis PJ, Vleugels JLA, Kasi PM
and Wallace MB: Colorectal Cancer. Lancet. 394:1467–1480. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hontscha C, Borck Y, Zhou H, Messmer D and
Schmidt-Wolf IG: Clinical trials on CIK cells: First report of the
international registry on CIK cells (IRCC). J Cancer Res Clin
Oncol. 137:305–310. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Giancola R, Olioso P, Di Riti M, Capone A,
Contento A, Pompetti F and Iacone A: Evaluation of an automated
closed fluid management device for processing expanded
cytokine-induced killer cells to use in immunotherapy programs for
cancer. Transfusion. 4:629–639. 2008. View Article : Google Scholar
|
|
5
|
Baker J, Verneris MR, Ito M, Shizuru JA
and Negrin RS: Expansion of cytolytic CD8(+) natural killer T cells
with limited capacity for graft-versus-host disease induction due
to interferon gamma production. Blood. 97:2923–2931. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lu PH and Negrin RS: A novel population of
expanded human CD3+CD56+ cells derived from T cells with potent in
vivo antitumor activity in mice with severe combined
immunodeficiency. J Immunol. 153:1687–1696. 1994.PubMed/NCBI
|
|
7
|
Leemhuis T, Wells S, Scheffold C, Edinger
M and Negrin RS: A phase I trial of autologous cytokine-induced
killer cells for the treatment of relapsed Hodgkin disease and non-
Hodgkin lymphoma. Biol Blood Marrow Transplant. 11:181–187. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Verneris MR, Karimi M, Baker J, Jayaswal A
and Negrin RS: Role of NKG2D signaling in the cytotoxicity of
activated and expanded CD8+ T cells. Blood. 103:3065–3072. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wang D, Li J, Liu JY, Li F, Wang LP, Huang
L, Li JY, Chen XF, Liu JB, Wu CC, et al: Modification of chemokine
receptor expression to enhance levels of trafficking receptors on
autologous cytokine-induced killer cells derived from patients with
colorectal cancer. Biomed Pharmacother. 68:551–556. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Horuk R: Chemokines. Scientific World
Journal. 19:224–232. 2007. View Article : Google Scholar
|
|
11
|
Kufareva I, Salanga CL and Handel TM:
Chemokine and chemokine receptor structure and interactions:
Implications for therapeutic strategies. Immunol Cell Biol.
4:372–383. 2015. View Article : Google Scholar
|
|
12
|
van der Vorst EP, Döring Y and Weber C:
Chemokines. Arterioscler Thromb Vasc Biol. 35:e52–e56. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sobin LH, Gospodarowicz MK and Wittekind
C: TNM classification of malignant tumors. 7thed. Oxford.
Wiley–Blackwell. 2010.
|
|
14
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2-ΔΔCq method. Methods. 25:402–408. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lazennec G and Richmond A: Chemokines and
chemokine receptors: New insights into cancer-related inflammation.
Trends Mol Med. 3:133–144. 2010. View Article : Google Scholar
|
|
16
|
Jäkel CE and Schmidt-Wolf IG: An update on
new adoptive immunotherapy strategies for solid tumors with
cytokine-induced killer cells. Expert Opin Biol Ther. 7:905–916.
2014. View Article : Google Scholar
|
|
17
|
Peng H, Yao M, Fan H, Song L, Sun J, Zhou
Z, Du Y, Lu K, Li T, Yin A, et al: Effects of autologous
cytokine-induced killer cells infusion in colorectal cancer
patients: A prospective study. Cancer Bionter Radiopharm.
6:221–226. 2017.
|
|
18
|
Kim JS, Kin YG, Park EJ, Kim B, Lee HK,
Hong JT, Kim Y and Han SB: Cell-based immunotherapy for colorectal
cancer with cytokine-induced killer cells. Immune Netw. 2:99–108.
2016. View Article : Google Scholar
|
|
19
|
Anandappa AJ, Wu CJ and Ott PA: Directing
traffic: How to effectively drive T cells into tumors. Cancer
Discov. 2:185–197. 2020. View Article : Google Scholar
|
|
20
|
Lippitz BE: Cytokine patterns in patients
with cancer: A systematic review. Lancet Oncol. 14:e218–e228. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Cremonesi E, Governa V, Garzon JFG, Mele
V, Amicarella F, Muraro MG, Trella E, Galati-Fournier V, Oertli D,
Däster SR, et al: Gut microbiata modulate T cell trafficking into
human colorectal cancer. Gut. 11:1984–1994. 2018. View Article : Google Scholar
|
|
22
|
Zipin-Roitman A, Meshel T, Sagi-Assif O,
Shalmon B, Avivi C, Pfeffer RM, Witz IP and Ben-Baruch A: CXCL10
promotes invasion-related properties in human colorectal carcinoma
cells. Cancer Res. 67:3396–3405. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Al-Haidari AA, Syk I and Thorlacius H:
HMG-CoA reductase regulates CCL17-induced colon cancer cell
migration via geranylgeranylation and RhoA activation. Biochem
Biophys Res Commun. 1:68–72. 2014. View Article : Google Scholar
|
|
24
|
Al-haidari AA, Syk I, Jirström K and
Thorlacius H: CCR4 mediates CCL17 (TARC)-induced migration of human
colon cancer cells via RhoA/Rho-kinase signaling. Int J Colorectal
Dis. 28:1479–1487. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Singh AK, Arya RK, Trivedi AK, Sanyal S,
Baral R, Dormond O, Briscoe DM and Datta D: Chemokine receptor
trio: CXCR3, CXCR4 and CXCR7 crosstalk via CXCL11 and CXCL12.
Cytokine Growth Factor Rev. 24:41–49. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zou Y, Li F, Hou W, Sampath P, Zhang Y and
Thorne SH: Manipulating the expression ofchemokine
receptorsenhances delivery and activity of cytokine-induced killer
cells. Br J Cancer. 110:1992–1999. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Mauro C, Fu H and Marelli-Berg FM: T cell
trafficking and metabolism: Novel mechanisms and targets for
immunomodulation. Curr Opin Pharmacol. 12:452–457. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Abastado JP: The next challenge in cancer
immunotherapy: Controlling T-cell traffic to the tumor. Cancer Res.
72:2159–2161. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Franciszkiewicz K, Boissonnas A, Boutet M,
Combadière C and Mami-Chouaib F: Role of chemokines and chemokine
receptors in shaping the effector phase of the antitumor immune
response. Cancer Res. 72:6325–6332. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Bryant J, Ahern DJ and Brennan FM: CXCR4
and vascular cell adhesion molecule 1 are key chemokine/adhesion
receptors in the migration of cytokine-activated T cells. Arthritis
Rheum. 64:2137–2146. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Pevida M, Lastra A, Meana Á, Hidalgo A,
Baamonde A and Menéndez L: The chemokine CCL5 induces CCR1-mediated
hyperalgesia in mice inoculated with NCTC 2472 tumoral cells.
Neuroscience. 259:113–125. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Tingting J, Changping W and Binfeng L:
Cytokine-induced killer cells promote antitumor immunity. J Transl
Med. 83:1–9. 2013.
|
|
33
|
Linn YC, Lau SK, Liu BH, Ng LH, Yong HX
and Hui KM: Characterization of recognition and functional
heterogeneity exhibited by cytokine-induced killer cell subsets
against acute myeloid leukaemia target cell. Immunology. 3:423–435.
2009. View Article : Google Scholar
|
|
34
|
Sangiolo D, Martinuzzi E, Todorovic M,
Vitaggio K, Vallario A, Jordaney N, Carnevale-Schianca F, Capaldi
A, Geuna M, Casorzo L, et al: Alloreactivity and anti-tumor
acitivity segregate within two distinct subsets of cytokine-induced
killer (CIK) cells: Implications for their infusion across major
HLA barriers. Int Immunol. 20:841–848. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Guo C, Liu S and Sun MZ: Novel insight
into the role of GAPDH playing in tumor. Clin Transl Oncol.
15:167–172. 2013. View Article : Google Scholar : PubMed/NCBI
|