Introduction
Cervical cancer (CC) is a serious public health
problem in China, with ~102,000 new cases estimated according to
the National Central Cancer Registry in 2013 (1). In addition, CC is the second most
common malignant tumor in women worldwide (2,3). It is
widely known that human papillomavirus (HPV) infection (4), family history of CC (5) and HIV infection (6) contribute to the development of CC. HPV
infection is one of the main causes of CC. Nearly all cases of CC
are causally associated with HPV infection (7,8),
particularly high-risk types of HPV, including HPV 16 and 18
(9–11). Simultaneously, sexual contact is the
dominant risk factor for HPV infection, particularly in those with
multiple sexual partners (MSP) (12). Thus, there is an indirect link
between MSP and cervical intraepithelial neoplasia (CIN)/CC.
CIN is a precancerous lesion characterized by
abnormal cell growth in the lining of the cervix. CIN can be
divided into three stages, according to the degree of the lesion:
CIN-I, CIN-II and CIN-III. CIN has the potential to progress to CC
if left untreated; this usually takes >10 years (13). Moreover, having MSP has been reported
as an independent risk factor of CC after adjusting for HPV
infection (14,15). Furthermore, the link between MSP and
CIN/CC is considered to be partially associated with geographical
region, as a meta-analysis of epidemiological studies revealed
(16). Another consequence of MSP is
an imbalance in the vaginal microecology, which theoretically
directly leads to bacterial vaginosis (BV). A previous study
reported a positive association between BV and cervical HPV
infection (17). Dysregulated
vaginal microbiota can enhance the risk of transient and persistent
HPV infections (18), thus BV and
HPV may jointly increase the risk of CIN. It was reported that BV
is a cofactor of HPV and the development of CIN (19), and a significantly increased risk of
CIN has been detected when BV is present (20,21).
However, controversial results also exist, for example, a study in
1995 reported that, in women with dyskaryotic cervical smears, the
prevalence of bacterial vaginosis did not seem to be increased, and
bacterial vaginosis did not influence the histologic changes. In
addition, in women with low grade squamous intraepithelial lesion,
aerobic vaginitis is very common but is not an indicator of HPV
infection (22,23). Since previous studies have only
included a small number of samples (17–21), and
the relationship among MSP, BV, HPV and CIN/CC development in
Chinese women remains unclear, the present study was designed to
clarify their association. Notably, the current study indicated
that when information regarding MSP has been obtained, it is
important to assess the severity of BV to better predict HPV and
CIN/CC development in Chinese women.
Materials and methods
Patients
The present study retrospectively analyzed 549
female patients who had visited the physical examination center,
outpatient and inpatient departments of Fujian Provincial Hospital
(Fuzhou, China) from January 2017 to December 2019. The inclusion
criteria were as follows: i) Aged between 20 and 55 years old, with
regular menstruation and sexual activity history; ii) suspicion of
high risk of HPV infection and BV infection; iii) 1 week before
diagnosis, there was no history of sexual activity, the use of
vaginal plugs, vaginal irrigation or other treatments. The
exclusion criteria were: i) Menopause; ii) a history of total
hysterectomy due to other diseases; iii) being pregnant; or iv)
presenting with severe malignant tumors, AIDS and other
immunodeficiencies.
The MSP information was acquired using a
questionnaire; accordingly, recruited patients were divided into
two groups: No MSP and MSP. The MSP group consisted of patients who
had more than one male sexual partner in the last 6 months. The no
MSP group comprised: i) Patients with a fixed male sexual partner,
ii) patients who lived alone and had not had sex in the last 6
months and iii) those who lived alone but occasionally had sex with
a fixed male.
Vaginal microecology test
The severity of BV was determined using a
gram-stained slide and was detected with the Cro vaginal
microbiological evaluation system (ShanDong Shida Si Biological
Industry Co., Ltd.). The results were hierarchically represented by
the Nugent Gram stain scoring system (24–27). A
Nugent score of 7 to 10 was defined as severe BV, a score of 4 to 6
was defined as mild BV, and a score of 1 to 3 was defined as
negative BV.
Cobas high-risk HPV test
The fully automated Cobas ×480 nucleic acid
extraction instrument and Cobas z480 PCR analyzer instrument [both
from Roche Diagnostics (Shanghai) Co., Ltd.] were utilized
according to the manufacturer's protocol to detect HPV types 16, 18
and 12 other high-risk subtypes (types 31, 33, 35, 39, 45, 51, 52,
56, 58, 59, 66 and 68).
Cervical conization pathology
(CCP)
Colposcopy was ordered if a patient had abnormal
thin-layer cytology test results or was highly possibly with CIN/CC
development. The colposcope magnified the visual field, and acetic
acid was used to swab the vaginal surface to discolor abnormal
tissues and easily identify lesions. When abnormal areas were
identified, colposcopy was followed by cervical conization to
determine the final diagnosis (24,25).
Briefly, a cone-shaped tissue sample around and below the zone of
the cervical squamocolumnar junction was resected. The resection
included normal tissue ranging from 3 to 5 mm outside the abnormal
tissue, and the cone depth ranged from 15 to 25 mm. Cone cutting
depth was determined by the site of the lesion. The tissue samples
were then immediately sent for pathological examination. According
to the CCP results, these patients were subdivided into CIN-I,
CIN-II, CIN-III and CC groups. The remaining patients who had
normal outcomes and did not receive CCP were divided into the
No-Need-to-Test group.
Statistical analysis
All categorical data were expressed in frequencies
and percentages; continuous data were expressed as the mean ±
standard deviation. SPSS software (version 25.0; IBM Corp.) was
used for statistical analysis and data visualization. The
χ2 test was used to compare frequencies in different
groups, and one-way ANOVA was used for comparison of continuous
data. The logistic regression model was established to predict the
outcomes of different levels of CCP outcomes (No-need-to-test,
CIN-I, CIN-II, CIN-III and CC). In addition, receiver operating
characteristic (ROC) curves were generated for the prediction of
CCP-positive results, and the area under the curve (AUC), as well
as recommended threshold values in distinguishing different CCP
outcomes, were calculated. P<0.05 was considered to indicate a
statistically significant difference.
Results
Age and MSP distribution
The demographic characteristics, including age and
MSP status, are presented in Table
I. A total of 549 women were included in the present study,
with an average age of 36.01±8.07 years. Of these, 309 women tested
negative for BV, and among women that tested positive for BV, 141
were in the BV mild group and 99 were in the BV severe group,
according to the Nugent score. The three groups displayed a similar
age distribution with no significant differences across BV groups,
whereas MSP status was distinct among patients with different
levels of BV severity. For women who did not have MSP, the number
in the BV-negative group was 298, whereas the number in the BV mild
group was 116, and in the BV severe group was 75. For those with
MSP, 11 women tested negative for BV, 25 women had mild BV and 24
women had severe BV. The percentage of patients with mild and
severe BV were significantly increased in the MSP group
(P<0.001).
 | Table I.Age and MSP status of enrolled
patients. |
Table I.
Age and MSP status of enrolled
patients.
| Index | Total (n=549) | BV negative
(n=309) | BV mild (n=141) | BV severe (n=99) | F or
χ2 | P-value |
|---|
| Age | 36.01±8.07 | 36.20±7.92 | 35.44±7.92 | 36.24±8.75 |
0.476 |
0.622 |
| MSP, n (%) |
|
|
|
| 41.964 | <0.001 |
| No | 489 (89.1%) | 298 (96.4%) | 116 (82.3%) | 75 (75.8%) |
|
|
|
Yes | 60 (10.9) | 11 (3.6%) | 25
(17.7%) | 24 (24.2%) |
|
|
Association between MSP, BV severity
and HPV infection outcomes
Next, the present study investigated the
relationship between BV severity and HPV infection. As BV severity
progressed, the HPV-positive ratio increased (Table II). This association was consistent
across all HPV groups, including total HPV, HPV 16, HPV 18 and
other high-risk HPV infections. Furthermore, MSP was significantly
associated with a HPV-positive outcome; this association was
observed for HPV 16, HPV 18 and other high-risk HPV infections
(Table III).
 | Table II.Association between BV severity and
HPV infection. |
Table II.
Association between BV severity and
HPV infection.
| HPV infection | BV negative
(%) | BV mild (%) | BV severe (%) | χ2 | P-value |
|---|
| Positive |
|
|
| 60.833 | <0.001 |
| No | 174 (56.3) | 45 (31.9) | 15 (15.2) |
|
|
|
Yes | 135 (43.7) | 96 (68.1) | 84 (84.8) |
|
|
| HPV 16 |
|
|
| 39.000 | <0.001 |
| No | 289 (93.5) | 111 (78.7) | 70 (70.7) |
|
|
|
Yes | 20 (6.5) | 30 (21.3) | 29 (29.3) |
|
|
| HPV 18 |
|
|
| 20.946 | <0.001 |
| No | 298 (96.4) | 128 (90.8) | 82 (82.8) |
|
|
|
Yes | 11 (3.6) | 13 (9.2) | 17 (17.2) |
|
|
| Other HPV
types |
|
|
| 41.225 | <0.001 |
| No | 204 (66.0) | 70 (49.6) | 30 (30.3) |
|
|
|
Yes | 105 (34.0) | 71 (50.4) | 69 (69.7) |
|
|
 | Table III.Association between MSP and HPV
infection. |
Table III.
Association between MSP and HPV
infection.
| HPV infection | No MSP (n=489)
(%) | MSP (n=60) (%) | χ2 | P-value |
|---|
| Positive |
|
| 23.630 | <0.001 |
| No | 226 (46.2) | 8 (13.3) |
|
|
|
Yes | 263 (53.8) | 52 (86.7) |
|
|
| HPV 16 |
|
| 31.348 | <0.001 |
| No | 433 (88.5) | 37 (61.7) |
|
|
|
Yes | 56 (11.5) | 23 (38.3) |
|
|
| HPV 18 |
|
| 24.536 | <0.001 |
| No | 462 (94.5) | 46 (76.7) |
|
|
|
Yes | 27 (5.5) | 14 (23.3) |
|
|
| Other HPV
types |
|
| 15.320 | <0.001 |
| No | 285 (58.3) | 19 (31.7) |
|
|
|
Yes | 204 (41.7) | 41 (68.3) |
|
|
Association between MSP, BV severity
and CIN/CC development
Analysis of the association between MSP and CCP
outcome demonstrated that the MSP group had a significantly higher
percentage of positive CCP outcomes (Table IV). Notably, the MSP group had a
higher proportion of CIN-II and CIN-III cases. Similarly, Nugent
score classification was significantly associated with the CCP
outcome (Table V); higher BV
severity was associated with a more severe CIN/CC progression. To
understand the association between MSP, BV and CCP outcome, six
groups were established according to BV severity and MSP status:
No-MSP-BV-negative, No-MSP-BV-mild, No-MSP-BV-severe,
MSP-BV-negative, MSP-BV-mild and MSP-BV-severe. Positive CCP
outcomes were significantly associated with MSP and BV; the
percentage of positive CCP results was successively increased from
No-MSP-BV-negative, to No-MSP-BV-mild, MSP-BV-negative,
No-MSP-BV-severe, MSP-BV-mild and MSP-BV-severe (Table VI). These univariate results
suggested that the combination of MSP and the Nugent score level
may be helpful to predict the final CCP results.
 | Table IV.Association between MSP and CCP
outcome. |
Table IV.
Association between MSP and CCP
outcome.
| CCP outcome | No MSP (n=489)
(%) | MSP (n=60) (%) | χ2 | P-value |
|---|
| Result |
|
| 38.683 | <0.001 |
|
Negative | 445 (91.0) | 38 (63.3) |
|
|
|
Positive | 44 (9.0) | 22 (36.7) |
|
|
| Level |
|
| 39.991 | <0.001 |
|
No-need-to-test | 443 (90.6) | 38 (63.3) |
|
|
|
CIN-I | 2 (0.4) | 0 (0.0) |
|
|
|
CIN-II | 24 (4.9) | 12 (20.0) |
|
|
|
CIN-III | 19 (3.9) | 10 (16.7) |
|
|
| CC | 1 (0.2) | 0 (0.0) |
|
|
 | Table V.Association between Nugent score
level and CCP outcome. |
Table V.
Association between Nugent score
level and CCP outcome.
| CCP outcome | BV negative
(%) | BV mild (%) | BV severe (%) | χ2 | P-value |
|---|
| Result |
|
|
| 74.886 | <0.001 |
|
Positive | 297 (96.1) | 123 (87.2) | 63 (63.6) |
|
|
|
Negative | 12 (3.9) | 18 (12.8) | 36 (36.4) |
|
|
| Level |
|
|
| 89.104 | <0.001 |
|
No-need-to-test | 297 (96.1) | 123 (87.2) | 61 (61.6) |
|
|
|
CIN-I | 0 (0.0) | 0 (0.0) | 2 (2.0) |
|
|
|
CIN-II | 8 (2.6) | 11 (7.8) | 17 (17.2) |
|
|
|
CIN-III | 4 (1.3) | 7 (5.0) | 18 (18.2) |
|
|
| CC | 0 (0.0) | 0 (0.0) | 1 (1.0) |
|
|
 | Table VI.Frequencies of positive cervical
conization pathology outcome in six subgroups. |
Table VI.
Frequencies of positive cervical
conization pathology outcome in six subgroups.
| Group | Negative (%) | Positive (%) | χ2 | P-value |
|---|
|
No-MSP-BV-negative | 287 (96.3 | 11 (3.7) | 95.301 | <0.001 |
| No-MSP-BV-mild | 106 (91.4) | 10 (8.6) |
|
|
|
No-MSP-BV-severe | 52 (69.3) | 23 (30.7) |
|
|
|
MSP-BV-negative | 10 (90.9) | 1 (9.1) |
|
|
| MSP-BV-mild | 17 (68.0) | 8 (32.0) |
|
|
| MSP-BV-severe | 11 (45.8) | 13 (54.2) |
|
|
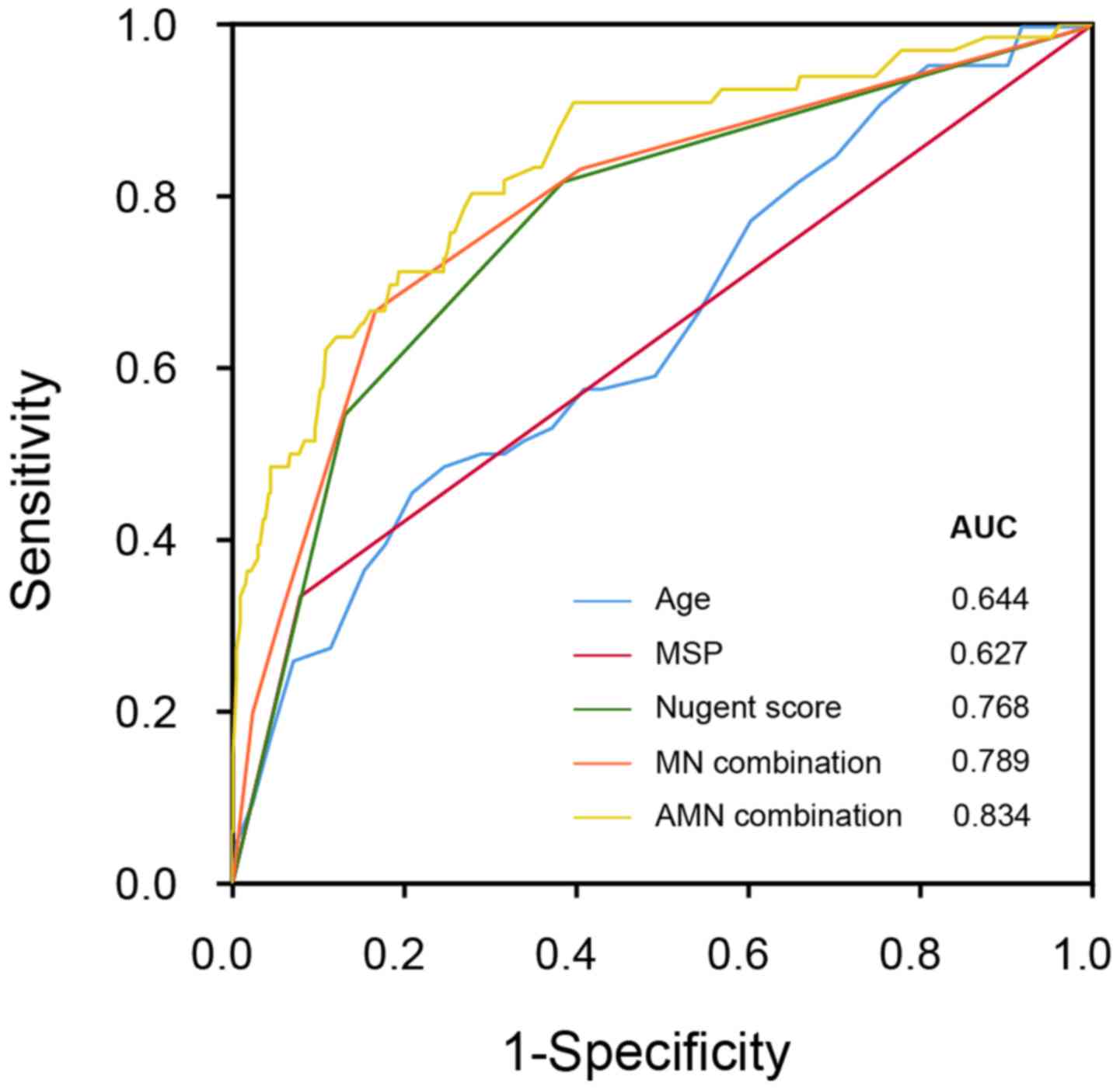
Next, we used for the logistic two-ends regression
model (Table VII) based on age
(continuous variable), MSP status (0, no; 1, yes) and the Nugent
score level (1, negative; 2, mild; 3, severe) in order to predict
the CCP outcome (negative or positive). The calculated value of the
logistic regression model using three variables (age, MSP and
Nugent score, namely AMN) revealed an AUC of 0.834 in the ROC
curve. This predictor had a sensitivity level of 0.803 and
specificity level of 0.702 at the cutoff value of 0.0818. When only
two indexes (MSP and Nugent score, namely MN) were applied, an AUC
of 0.789 was revealed. Meanwhile, the AUC of the single index
Nugent score was 0.768, which highlights the informative role of BV
examination (Fig. 1). Together, the
severity of BV infection may be used to predict CIN/CC development,
with satisfactory sensitivity and accuracy, and vaginal
microecology may strongly impact infection and cervical
carcinogenesis.
 | Table VII.Variables in the logistic regression
model of cervical conization pathology outcomes. |
Table VII.
Variables in the logistic regression
model of cervical conization pathology outcomes.
| Variable | B | Wald | P-value | Odd ratio | 95% CI of odd
ratio |
|---|
| Age | 0.095 | 22.282 | <0.0001 | 1.099 | 1.057–1.143 |
| MSP or not | 1.708 | 20.551 | <0.0001 | 5.518 | 2.637–11.548 |
| BV severity
levels |
| 44.181 | <0.0001 |
|
|
| Mild vs.
negative | 1.070 | 6.779 | 0.0090 | 2.915 | 1.303–6.523 |
| Severe vs.
negative | 2.466 | 41.582 | <0.0001 | 11.780 | 5.566–24.930 |
Discussion
Sexually transmitted infections, including HPV, have
been reported to be associated with the etiology of cervical
carcinogenesis. Although HPV infection can be eliminated within 2
years without any complications, 1% of HPV-positive women develop
CC (in the United States of America) (4). Thus, other potential risk factors need
to be investigated to better predict CC. It is known that the
normal vaginal microecology plays an indispensable role in
preventing HPV infection and accelerating HPV clearance (26,27).
Abnormal vaginal microecology may be a synergistic factor of
persistent HPV infection (28),
which ultimately increases the risk of CIN/CC. A study with a twin
cohort reported that increased vaginal microbiota diversity was
closely related to HPV infection (29). HPV is a sexually transmitted
infection, and increased multiple-partner sexual contact may
enhance HPV transmission and CC risk (30). A collaborative study that collected
data from >15,000 women with CIN-III/CC confirmed the
relationship between major indicators of sexual behavior and the
risk of CC (31). Given that
regional disparity can influence the relationship between MSP, BV
severity, HPV and CIN/CC (16), it
is worth exploring the association between these variables in
Western and Eastern countries. A Spanish study reported that MSP
increased the risk of HPV infection (12). In addition, a recent study revealed
that marital status, age at first sexual intercourse, number of
lifetime sexual partners and condom use were related to the
presence of cervical pathology and the presence of oral HPV
(32). Still, there have not been
sufficient studies in other countries, particularly Eastern
countries, investigating these associations. In the current study,
a cohort of the Chinese population was used to confirm that MSP and
BV were associated with CIN/CC in Chinese women.
MSP has been associated with an increased risk of
CIN/CC. One of the key causes of CIN/CC development is long-term
infection with HPV (33). In
addition, vaginal microbiota impact the acquisition, persistence
and clearance of HPV, and hence affect the risk of developing CC
(34). MSP status contributes to the
abnormality of vaginal microecology, and this abnormal intravaginal
microbial environment can be the basis for CIN/CC development. In
addition, MSP may result in the introduction of other sexually
transmitted pathogens, such as HIV, which is known to increase the
risk of CIN/CC (35).
In the present study, patients potentially infected
with HPV and BV were enrolled to clarify the relationships between
MSP, BV and CIN/CC development. MSP and different levels of BV
severity were found to be associated with HPV infection and CIN/CC.
Through logistic regression analysis of CCP outcomes, MSP status
and Nugent score were revealed to be risk factors for CIN/CC
prediction. Using two-parameter (MN) or three-parameter (AMN)
prediction models, results obtained showed a satisfactory accuracy
and sensitivity for the prediction of CIN/CC.
Overall, the findings of the current study are
consistent with previously published articles. According to a study
of 211 cervical samples, BV and inflammatory response were
independently associated with the severity of cervical neoplasia in
HPV-positive women (36). A
meta-analysis also provided the same association between BV and
uterine cervical HPV infection (17). The mechanisms underlying this
association may include the production of carcinogenic nitrosamines
and an altered inflammatory cytokine profile based on higher
vaginal pH in BV-infected patients with persistent HPV (37,38).
However, more mechanisms need to be explored. It has been suggested
that HPV examination provides 60–70% higher protection against
invasive CC and it is usually regarded as a more direct way of
early warning of CC than cytological methods (39). The present work highlights the role
of vaginal microecological testing in the monitoring of CIN/CC. In
addition, it was confirmed that BV might be involved in the
pathological process of CIN/CC. In the BV mild stage, the
percentage of positive CCP cases sharply increased in comparison
with BV negative. Therefore, even for patients with mild BV, timely
detection and treatment should be conducted to halt the development
of CIN/CC.
There are some limitations in the present study.
Firstly, this cross-sectional study lacks longitudinal data and the
causality of the MSP-BV-HPV-CIN axis (as well as other potential
causal chains) remains unclear. Secondly, the single-index MSP used
in this study is not informative enough, which only concerns the
total number of sexual partners. Other sexual behaviors, such as
age at first intercourse and the definite number of sexual
partners, were not included in this study. Moreover, the MSP index
only focused on sexual activity with men, but not women. Finally,
HPV 16 and HPV 18 were primarily used as the first-choice risk
factors, but other high-risk HPV strains, such as HPV 31, HPV 33
and HPV 35 that are implicated in 99% of CC cases have not been
analyzed (9,40).
In conclusion, MSP and BV severity may be the main
risk factors for HPV and CC development among Chinese women. A
combination of MSP information and BV examination could provide a
rapid, economic and accurate prediction of CIN/CC. Therefore,
appropriate sexual health education, and prompt detection and
treatment of BV could reduce the risk of CC.
Acknowledgements
Not applicable.
Funding
This work was funded by The Natural Science
Foundation of Fujian Province (grant no. 2017J01254).
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
XW, YL and WL collected data. YH, XW and YL analyzed
data and made the figures. JL and BS designed the study and wrote
the paper. All authors read and approved the final study.
Ethics approval and consent to
participate
This study was approved by the Hospital Ethics
Committee of Shengli Clinical Medical College of Fujian Medical
University (approval no. K2017-03-002). Written informed consent
was obtained from all participants, and the consent was in strict
accordance with The Declaration of Helsinki.
Patients consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Gu XY, Zheng RS, Sun KX, Zhang SW, Zeng
HM, Zou XN, Chen WQ and He J: Incidence and mortality of cervical
cancer in China, 2014. Zhonghua Zhong Liu Za Zhi. 40:241–246.
2018.(In Chinese). PubMed/NCBI
|
|
2
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wardak S: Human Papillomavirus (HPV) and
cervical cancer. Med Dosw Mikrobiol. 68:73–84. 2016.PubMed/NCBI
|
|
4
|
Khan MJ, Partridge EE, Wang SS and
Schiffman M: Socioeconomic status and the risk of cervical
intraepithelial neoplasia grade 3 among oncogenic human
papillomavirus DNA-positive women with equivocal or mildly abnormal
cytology. Cancer. 104:61–70. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Negri E, La Vecchia C, Bosetti C,
Franceschi S and Parazzini F: Risk of cervical cancer in women with
a family history of breast and female genital tract neoplasms. Int
J Cancer. 117:880–881. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Abraham AG, D'Souza G, Jing Y, Gange SJ,
Sterling TR, Silverberg MJ, Saag MS, Rourke SB, Rachlis A,
Napravnik S, et al: Invasive cervical cancer risk among
HIV-infected women: A North American multicohort collaboration
prospective study. J Acquir Immune Defic Syndr. 62:405–413. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Joung JG, June S and Zhang BT: Protein
sequence-based risk classification for human papillomaviruses.
Comput Biol Med. 36:656–667. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
de Villiers EM, Fauquet C, Broker TR,
Bernard HU and zur Hausen H: Classification of papillomaviruses.
Virology. 324:17–27. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Boulet G, Horvath C, Vanden Broeck D,
Sahebali S and Bogers J: Human papillomavirus: E6 and E7 oncogenes.
Int J Biochem Cell Biol. 39:2006–2011. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
zur Hausen H: Human papillomaviruses in
the pathogenesis of anogenital cancer. Virology. 184:9–13. 1991.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Walboomers JM, Jacobs MV, Manos MM, Bosch
FX, Kummer JA, Shah KV, Snijders PJ, Peto J, Meijer CJ and Muñoz N:
Human papillomavirus is a necessary cause of invasive cervical
cancer worldwide. J Pathol. 189:12–19. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Vaccarella S, Franceschi S, Herrero R,
Muñoz N, Snijders PJ, Clifford GM, Smith JS, Lazcano-Ponce E,
Sukvirach S, Shin HR, et al: Sexual behavior, condom use, and human
papillomavirus: Pooled analysis of the IARC human papillomavirus
prevalence surveys. Cancer Epidemiol Biomarkers Prev. 15:326–333.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bos AB, van Ballegooijen M, van
Oortmarssen GJ, van Marle ME, Habbema JD and Lynge E:
Non-progression of cervical intraepithelial neoplasia estimated
from population-screening data. Br J Cancer. 75:124–130. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Herrero R, Brinton LA, Reeves WC, Brenes
MM, Tenorio F, de Britton RC, Gaitan E, Garcia M and Rawls WE:
Sexual behavior, venereal diseases, hygiene practices, and invasive
cervical cancer in a high-risk population. Cancer. 65:380–386.
1990. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang PD and Lin RS: Risk factors for
cervical intraepithelial neoplasia in Taiwan. Gynecol Oncol.
62:10–18. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Liu ZC, Liu WD, Liu YH, Ye XH and Chen SD:
Multiple sexual partners as a potential independent risk factor for
cervical cancer: A meta-analysis of epidemiological studies. Asian
Pac J Cancer Prev. 16:3893–3900. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Gillet E, Meys JF, Verstraelen H, Bosire
C, De Sutter P, Temmerman M and Broeck DV: Bacterial vaginosis is
associated with uterine cervical human papillomavirus infection: A
meta-analysis. BMC Infect Dis. 11:102011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Brotman RM, Shardell MD, Gajer P, Tracy
JK, Zenilman JM, Ravel J and Gravitt PE: Interplay between the
temporal dynamics of the vaginal microbiota and human
papillomavirus detection. J Infect Dis. 210:1723–1733. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Platz-Christensen JJ, Sundstrom E and
Larsson PG: Bacterial vaginosis and cervical intraepithelial
neoplasia. Acta Obstet Gynecol Scand. 73:586–588. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Platz-Christensen JJ, Larsson PG,
Sundstrom E and Bondeson L: Detection of bacterial vaginosis in
Papanicolaou smears. Am J Obstet Gynecol. 160:132–133. 1989.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Verbruggen BS, Boon ME and Boon LM:
Dysbacteriosis and squamous (pre)neoplasia of immigrants and Dutch
women as established in population-based cervical screening. Diagn
Cytopathol. 34:377–381. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
22
|
Peters N, Van Leeuwen AM, Pieters WJ,
Hollema H, Quint WG and Burger MP: Bacterial vaginosis is not
important in the etiology of cervical neoplasia: A survey on women
with dyskaryotic smears. Sex Transm Dis. 22:296–302. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Jahic M, Mulavdic M, Hadzimehmedovic A and
Jahic E: Association between aerobic vaginitis, bacterial vaginosis
and squamous intraepithelial lesion of low grade. Med Arch.
67:94–96. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Grubisić G, Klarić P, Jokanović L,
Soljacić Vranes H, Grbavac I and Bolanca I: Diagnostic approach for
precancerous and early invasive cancerous lesions of the uterine
cervix. Coll Antropol. 33:1431–1436. 2009.PubMed/NCBI
|
|
25
|
Partridge EE, Abu-Rustum N, Campos S,
Fahey PJ, Greer BE, Lele SM, Lieberman RW, Lipscomb GH, Morgan M,
Nava ME, et al: Cervical cancer screening. J Natl Compr Canc Netw.
6:58–82. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Champer M, Wong AM, Champer J, Brito IL,
Messer PW, Hou JY and Wright JD: The role of the vaginal microbiome
in gynaecological cancer. BJOG. 125:309–315. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Liang Y, Chen M, Qin L, Wan B and Wang H:
A meta-analysis of the relationship between vaginal microecology,
human papillomavirus infection and cervical intraepithelial
neoplasia. Infect Agent Cancer. 14:292019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Di Paola M, Sani C, Clemente AM, Iossa A,
Perissi E, Castronovo G, Tanturli M, Rivero D, Cozzolino F,
Cavalieri D, et al: Characterization of cervico-vaginal microbiota
in women developing persistent high-risk human papillomavirus
infection. Sci Rep. 7:102002017. View Article : Google Scholar
|
|
29
|
Lee JE, Lee S, Lee H, Song YM, Lee K, Han
MJ, Sung J and Ko G: Association of the vaginal microbiota with
human papillomavirus infection in a Korean twin cohort. PLoS One.
8:e635142013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
International Collaboration of
Epidemiological Studies of Cervical Cancer, . Comparison of risk
factors for invasive squamous cell carcinoma and adenocarcinoma of
the cervix: Collaborative reanalysis of individual data on 8,097
women with squamous cell carcinoma and 1,374 women with
adenocarcinoma from 12 epidemiological studies. Int J Cancer.
120:885–891. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
International Collaboration of
Epidemiological Studies of Cervical Cancer, . Cervical carcinoma
and sexual behavior: Collaborative reanalysis of individual data on
15,461 women with cervical carcinoma and 29,164 women without
cervical carcinoma from 21 epidemiological studies. Cancer
Epidemiol Biomarkers Prev. 18:1060–1069. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Sánchez-Siles M, Remezal-Solano M,
López-López AM and Camacho-Alonso F: Prevalence of human
papillomavirus in the saliva of sexually active women with cervical
intraepithelial neoplasias. Med Oral Patol Oral Cir Bucal.
25:e195–e204. 2020.PubMed/NCBI
|
|
33
|
Kjellberg L, Hallmans G, Ahren AM,
Johansson R, Bergman F, Wadell G, Angström T and Dillner J:
Smoking, diet, pregnancy and oral contraceptive use as risk factors
for cervical intra-epithelial neoplasia in relation to human
papillomavirus infection. Br J Cancer. 82:1332–1338. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Mitra A, MacIntyre DA, Marchesi JR, Lee
YS, Bennett PR and Kyrgiou M: The vaginal microbiota, human
papillomavirus infection and cervical intraepithelial neoplasia:
What do we know and where are we going next? Microbiome. 4:582016.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Silverberg MJ, Lau B, Justice AC, Engels
E, Gill MJ, Goedert JJ, Kirk GD, D'Souza G, Bosch RJ, Brooks JT, et
al: Risk of anal cancer in HIV-infected and HIV-uninfected
individuals in North America. Clin Infect Dis. 54:1026–1034. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
de Castro-Sobrinho JM, Rabelo-Santos SH,
Fugueiredo-Alves RR, Derchain S, Sarian LO, Pitta DR, Campos EA and
Zeferino LC: Bacterial vaginosis and inflammatory response showed
association with severity of cervical neoplasia in HPV-positive
women. Diagn Cytopathol. 44:80–86. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Pavić N: Is there a local production of
nitrosamines by the vaginal microflora in anaerobic
vaginosis/trichomoniasis? Med Hypotheses. 15:433–436. 1984.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Hudson MM, Tidy JA, McCulloch TA and
Rogstad KE: When is bacterial vaginosis not bacterial vaginosis?-a
case of cervical carcinoma presenting as recurrent vaginal
anaerobic infection. Genitourin Med. 73:306–307. 1997.PubMed/NCBI
|
|
39
|
Ronco G, Dillner J, Elfström KM, Tunesi S,
Snijders PJ, Arbyn M, Kitchener H, Segnan N, Gilham C, Giorgi-Rossi
P, et al: Efficacy of HPV-based screening for prevention of
invasive cervical cancer: Follow-up of four European randomised
controlled trials. Lancet. 383:524–532. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Bruni L, Diaz M, Castellsague X, Ferrer E,
Bosch FX and de Sanjose S: Cervical human papillomavirus prevalence
in 5 continents: Meta-analysis of 1 million women with normal
cytological findings. J Infect Dis. 202:1789–1799. 2010. View Article : Google Scholar : PubMed/NCBI
|