Introduction
The global incidence of kidney cancer is increasing
year by year, and it is highly invasive and metastatic. The most
common type of kidney cancer is kidney renal clear cell carcinoma
(KIRC) (1,2). Surgery remains the first choice for
early treatment due to the fact that KIRC is insensitive to
conventional radiotherapy and chemotherapy (3); however, the disease has an insidious
onset, progresses rapidly and is poorly treated with late-stage
surgery, resulting in an extremely low late-stage survival rate
(4,5). Despite the promising results of
targeted therapies (6), the issue
of resistance to targeted therapies has arisen. For example,
Chatterjee and Bivona (7) found
that reversible proteomic and epigenetic mechanisms, tumor
microenvironment-mediated mechanisms, and tumor heterogeneity may
all contribute to the emergence of resistance, thereby affecting
the therapeutic efficacy of cancer treatment.
The use of immunotherapy in cancer has provided
novel ideas for the treatment of KIRC, which exhibits a stronger
immune response compared with other cancers (8–12).
Immunotherapy is effective in prolonging the overall survival (OS)
of patients and tumor, node and metastasis (TNM) staging is
considered to be the most appropriate prognostic indicator
(13–21). However, there are few studies on
KIRC immune infiltration and its biomarkers (22). Hence, the search for specific immune
biomarkers holds great clinical significance to provide more
personalized and precise treatments to improve the prognosis of
patients with KIRC.
Thioredoxin-interacting protein (TXNIP), a
multifunctional protein that inhibits the production of glucose
transporter proteins, enzymes involved in glycolysis and associated
genes, is crucial in preventing tumor aerobic glycolysis (23–25).
TXNIP is associated with the cell cycle process and its
upregulation inhibits the function of the cell cycle protein A
promoter, thereby suppressing the cell cycle (26). Under oxidative stress, TXNIP in the
nucleus is transported to the mitochondria, where it binds to
thioredoxin-2, which in turn triggers apoptosis and inhibits the
proliferation of tumor cells (27,28).
Additionally, TXNIP is closely associated with inflammatory
immune responses, in which TXNIP binds to the nucleotide-binding
oligomerization domain-like receptor family pyrin domain containing
3 inflammasome to induce inflammation (29). Although there are many studies on
TXNIP, information on immune infiltration and clinical
prognosis is scarce (30). Previous
studies have reported the relationship between TXNIP and
angiogenesis, as well as clinical prognosis in KIRC (31,32);
however, the relationship between the expression level of
TXNIP and immune infiltration in KIRC has rarely been
reported (33).
In the present study, the mRNA expression level of
TXNIP in patients with KIRC were assessed using The Cancer
Genome Atlas (TCGA) database to evaluate the association with
overall survival and clinicopathological characteristics.
Additionally, the correlation between TXNIP expression
level, immune cell infiltration and prognosis was assessed using
CIBERSORT and univariate and multivariate Cox regression
analysis.
Materials and methods
Data gathering
Using the TCGA database (https://portal.gdc.cancer.gov/), gene expression
patterns and clinical information from 542 patients with KIRC and
72 normal kidney tissue samples were obtained from the TCGA-KIRC
dataset (34). The Tumor Immune
Estimation Resource (TIMER) database was used to determine the mRNA
expression levels of TXNIP in 33 different cancer types
(https://cistrome.shinyapps.io/timer/). The Human
Protein Atlas (HPA) database (http://www.proteinatlas.org) was used to obtain
immunohistochemical data on protein expression of TXNIP in KIRC and
normal tissues.
Identification and enrichment analysis
of genes co-expressed with TXNIP
A total of five genes co-expressed with TXNIP
were screened, with P<0.001 used as a significant correlation
cutoff. Gene Ontology (GO) and Kyoto Encyclopedia of Genes and
Genomes (KEGG) analyses of co-expressed genes were performed using
the R package clusterProfiler v4.6.2. with P<0.05 considered the
significance cutoff (35,36). The files ‘c2.cp.kegg.v7.4.symbols’
and ‘c5.go.v7.4.symbols’ were used for gene set variation analysis
(GSVA). The ‘limma’ R package v3.54.2 was used to identify
biological functions (https://bioinf.wehi.edu.au/limma/). A GSVA score
t-value >2 was considered significantly altered.
Evaluation of immune infiltration
The proportion of infiltrating immune cells in 542
tumor samples was assessed using the CIBERSORT database (http://cibersort.stanford.edu/), and the
CIBERSORT R v1.03 and LM22 R software packages were used as tools
for algorithmic ensembles (37).
Based on the median TXNIP mRNA expression level of the
patients with KIRC, the patients were divided into TXNIP low
and high expression groups (P<0.05 was considered as a
statistically significant screening condition), and the level of
infiltration of the different immune cells was subsequently
confirmed using the TIMER 2.0 algorithm (38).
Immune checkpoint correlation
A significant correlation of P<0.001 was used as
a screening condition and the R package ‘corrplot’ v0.92
(https://github.com/taiyun/corrplot)
was used to assess the correlation between the expression data of
immune checkpoint-related genes and TXNIP mRNA
expression.
Expression levels of TXNIP at the
single cell level
The Tumor Immune Single-Cell Hub (http://tisch.comp-genomics.org/home/) is
a publicly available and comprehensive web resource site. The
KIRC_GSE139555 and KIRC_GSE111360 datasets were selected in the
‘Datasets’ module to visualize and assess the variations in
TXNIP expression at the single-cell level between different
immune cells.
Drug susceptibility analysis
Drug-related data were obtained from the CellMiner
database (39), which includes
records of drug sensitivity analysis of drugs validated by clinical
trials and approved by the U.S. Food and Drug Administration.
Subsequently, Pearson correlation coefficients were used to analyze
the relationship between mRNA expression levels of TXNIP and
drug sensitivity in the TCGA-KIRC dataset.
Cell culture and transfection
Human kidney cancer A498 cells (cat. no. CL-0254)
and normal kidney tissue HK-2 cells (cat. no. CM-0109) were
obtained from Procell Life Science & Technology Co., Ltd. The
cells were resuscitated and cultured with complete minimal
essential medium, including MEM basal medium (cat. no. PM150410;
Life Science & Technology Co., Ltd.), 1% penicillin mixture
(cat. no. P1400; Beijing Solarbio Science & Technology Co.,
Ltd.), and 10% neonatal fetal bovine serum [cat. no. CF-01P-02;
Cell-Box (HK) Biological products Trading Co., Ltd.]. The cell
cultures were kept at 37°C in a 5% CO2 cell incubator.
Before transfection, the cells were cultured and cultivated until
they reached ~70% confluence. A498 cells were then transfected with
4 µg each of TXNIP-overexpression plasmid (A498-LV-TXNIP) or
empty vector plasmid (A498-LV-Empty). The plasmids were purchased
from GeneCopoeia, Inc. The TXNIP overexpression and empty
vector plasmids were added into MEM basal medium and HighGene plus
transfection reagent (cat. no. RM09014P; ABclonal Biotech Co.,
Ltd.) was then added into the wells containing cells after thorough
mixing. After transfection, the cells were placed in a 5%
CO2 cell culture incubator at 37°C for 4–6 h, and then
half of the medium was replaced and the cells were incubated again
for 24–48 h before the cells were used for subsequent
experiments.
RNA extraction and reverse
transcription (RT)-quantitative (q)PCR
An RNA Fast Small Extraction Kit (cat. no. TR154-50;
Jianshi Biotechnology Co., Ltd) was used to extract total RNA and
cDNA was synthesized using the SureScript™ First-Strand cDNA
Synthesis Kit (cat. no. QP056; GeneCopoeia, Inc.). mRNA expression
levels were determined using the LightCycler® 96
Instrument (SW 1.1; Roche Diagnostics GmBH) and the BlazeTaq™ SYBR
Green qPCR Mix 2.0 kit (cat. no. QP031; GeneCopoeia, Inc.). RNA
extraction, cDNA synthesis and qPCR were performed according to the
manufacturers' protocols. The synthesis of cDNA was performed at
25°C for 5 min, 42°C for 15 min and 85°C for 5 min, and then
annealed at 4°C to finish. qPCR was performed at 95°C for 10 min,
followed by 40 cycles at 95°C for 10 sec, 60°C for 20 sec and
extension at 72°C for 15 sec, with a final extension step at 72°C
for 10 min. GAPDH was used as an endogenous control and the results
were quantified using the 2−∆∆Cq method (40). A498 cells (~5×105 cells)
transfected with empty vector and TXNIP-overexpression
plasmids were used as control and treatment groups, respectively,
and this experiment was repeated three times. The primer sequences
(Shanghai Sangon Pharmaceutical Co., Ltd.) used were as follows:
GAPDH forward (F), 5′-GGTGAAGGTCGGAGTCAACG-3′ and GAPDH reverse
(R), 5′-CAAAGTTGTCATGGATGACC-3′; TXNIP F,
5′-GGCAATCATATTATCTCAGGGAC-3′ and TXNIP R,
5′-CAGGAACGCTAACATAGATCAGTAA-3′; CD25 F, 5′-TTCGTGGTGGGGCAGATGGT-3′
and CD25 R, 5′-TCTTCCCGTGGGTCATTTTG-3′; and cytotoxic
T-lymphocyte-associated protein 4 (CTLA4) F,
5′-AACCTACATGATGGGGAATGAG-3′ and CTLA4 R,
5′-AGGTAGTATGGCGGTGGGTAC-3′.
Western blotting
Total proteins were extracted from HK-2, A498,
A498-LV-TXNIP and A498-LV-Empty cells (~5.5×106 cells)
using RIPA lysis buffer (Beyotime Institute of Biotechnology) mixed
with phenylmethanesulfonyl fluoride (cat. no. P6730; Beijing
Solarbio Science & Technology Co., Ltd.) at a ratio of 1:100.
The protein concentration was assessed using a BCA protein assay
kit (cat. no. PC0020; Beijing Solarbio Science & Technology
Co., Ltd.). Subsequently, 70 µg protein/lane were separated on 10%
precast gels using SDS-PAGE (Invitrogen; Thermo Fisher Scientific,
Inc.) and transferred to PVDF transfer membranes (Biosharp Life
Sciences). The membranes were blocked using 5% skim milk powder
(cat. no. 1172GR100; BioFroxx; neoFroxx GmbH) for 2 h at room
temperature and then incubated with anti-TXNIP (1:1,000; cat. no.
A11682; Nature Biosciences Ltd.) and GAPDH antibodies (1:1,000;
cat. no. RA0003; Nature Biosciences Ltd.) overnight at 4°C. The
following day, these membranes were washed 3 times using TBST
(contains 0.05% Tween) and incubated again with HRP-conjugated goat
anti-rabbit IgG (H+L) secondary antibodies (1:3,000; cat. no.
AS014; ABclonal Biotech Co., Ltd.) for 1 h at room temperature.
Luminescence development was performed using MonPro™ ECL
Ultrasensitive Substrate Pro (cat. no. PW30701S; Monad Biotech Co.,
Ltd.) and a Tanon-5200Multi gel imager was used to capture protein
images (Tanon Science and Technology Co., Ltd.). Protein expression
levels were semi-quantified using ImageJ v1.8.0 (National
Institutes of Health). The GAPDH signal was used to normalize the
TXNIP band intensity and the experiment was repeated three
times.
Cell Counting Kit (CCK)-8 cell
proliferation assay
The CCK-8 was purchased from Dojindo Laboratories,
Inc. A498-LV-Empty and A498-LV-TXNIP cells (~2×104
cells) were placed in 96-well plates (3×103 cells per
well) and five wells of each were replicated and cultured in an
incubator at 37°C for 0, 24, 48, 72 and 96 h. CCK-8 reagent (10 µl)
was added to each well and then the cells were incubated for
another 2 h. Finally, using an ELISA microplate reader [Biobase
Biodusty (Shandong) Co., Ltd.], the absorbance values were
determined at 450 nm. The experiment was repeated three times.
Statistical analysis
Data handling and statistical analysis was performed
using R software version 4.2.1 (https://www.r-project.org/), Strawberry Perl version
5.30.1.1 (https://strawberryperl.com/), SPSS
version 25 (IBM Corp.) and GraphPad Prism version 9.0 (Dotmatics).
To determine if there were significant differences in TXNIP,
CD25 and CTLA4 mRNA expression levels between subgroups, analysis
was performed using the unpaired t-test. Kruskal-Wallis and Dunn's
test were used to analyze the relationship between KIRC
clinicopathological variables and TXNIP mRNA expression
levels. Statistical analysis of Kaplan-Meier and other survival
analyses were performed using Log-rank tests. For survival
analyses, univariate and multivariate Cox regression models were
used. P<0.05 was considered to indicate a statistically
significant difference.
Results
mRNA expression of TXNIP in certain
cancers
The differences in mRNA level expression of
TXNIP in 33 tumor and normal tissues were compared using
data from TCGA database and the TIMER online web tool. TCGA
database results showed that the mRNA expression level of
TXNIP in KIRC was significantly lower than that in normal
tissues (Fig. 1A). The 605 samples
of KIRC were further evaluated using the TIMER online database, in
which the mRNA expression level of TXNIP was significantly
reduced in 533 tumor samples, which was consistent with the results
shown in TCGA database (Fig. 1B).
The aforementioned results indicated that TXNIP mRNA levels
were expressed at a low level in most cancers.
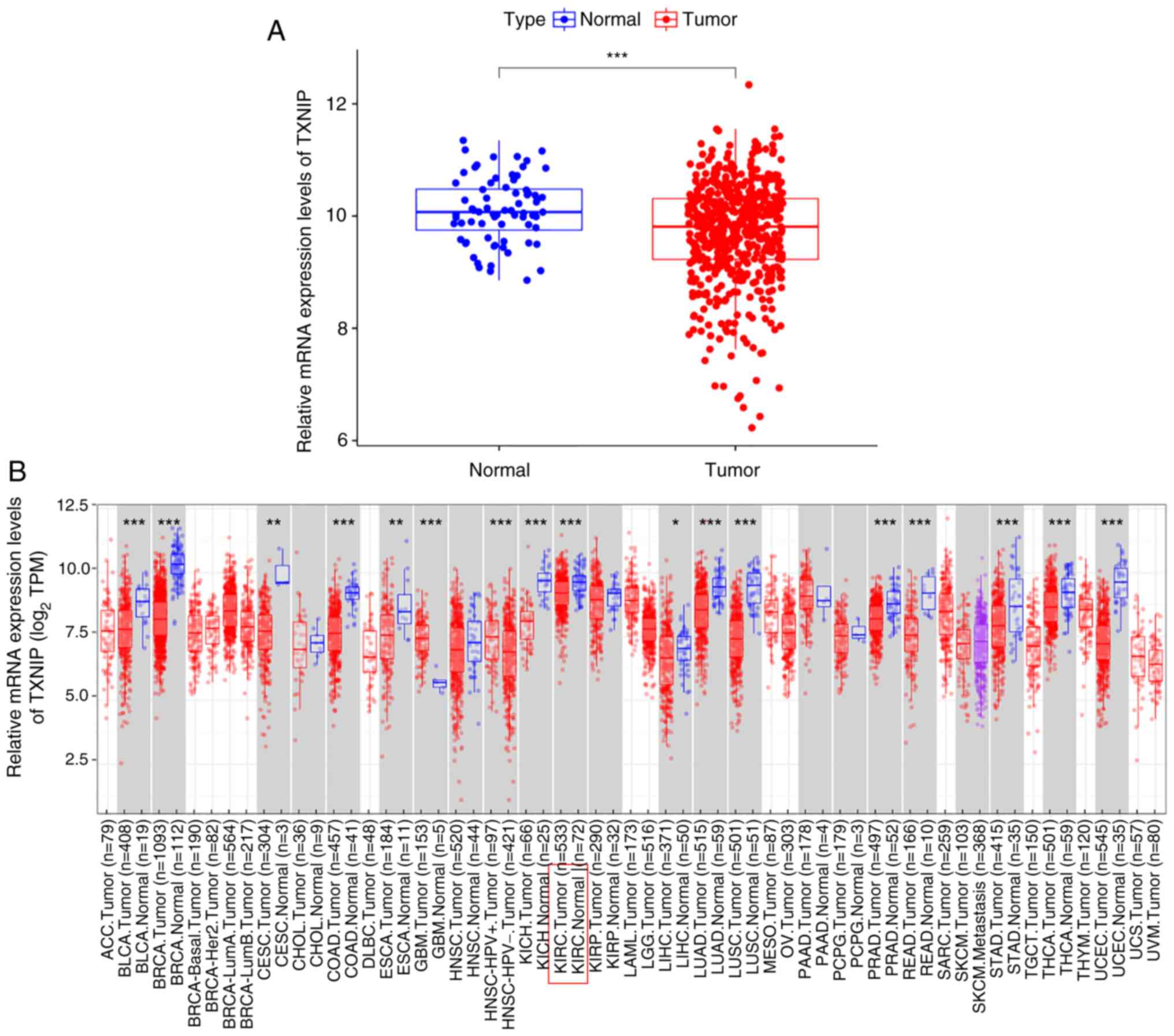 | Figure 1.Differential expression of
TXNIP in different cancers. (A) Differential expression of
TXNIP in KIRC tumor and normal tissues. (B) Analysis of the
differential expression of TXNIP in 39 tumors based data
from the Tumor Immune Estimation Resource database. *P<0.05,
**P<0.01 and ***P<0.001. TXNIP, thioredoxin-interacting
protein; KIRC, kidney renal clear cell carcinoma; TPM, transcripts
per million; ACC, adrenocortical carcinoma; BLCA, bladder
urothelial carcinoma; BRCA, breast invasive carcinoma; CESC,
cervical squamous cell carcinoma; CHOL, cholangiocarcinoma; COAD,
colon adenocarcinoma; DLBC, lymphoid neoplasm diffuse large B-cell
lymphoma; ESCA, esophageal carcinoma; GBM, glioblastoma multiforme;
HNSC, head and neck squamous cell carcinoma; KICH, kidney
chromophobe; KIRP, kidney renal papillary cell carcinoma; LAML,
acute myeloid leukemia; LGG, brain lower grade glioma; LIHC, liver
hepatocellular carcinoma; LUAD, lung adenocarcinoma; LUSC, lung
squamous cell carcinoma; MESO, mesothelioma; OV, ovarian serous
cystadenocarcinoma; PAAD, pancreatic adenocarcinoma; PCPG,
pheochromocytoma and paraganglioma; PRAD, prostate adenocarcinoma;
READ, rectum adenocarcinoma; SARC, sarcoma; SKCM, skin cutaneous
melanoma; STAD, stomach adenocarcinoma; TGCT, testicular germ cell
tumors; THCA, thyroid carcinoma; THYM, thymoma; UCEC, uterine
corpus endometrial carcinoma; UCS, uterine carcinosarcoma; UVM,
uveal melanoma. |
Patients with KIRC were matched for clinical data
and separated into low and high TXNIP expression groups
according to their median TXNIP mRNA expression level.
Kaplan-Meier survival curves and TIMER data (Fig. 2A) demonstrated a positive
association between the mRNA expression level of TXNIP and
cumulative survival. The mRNA expression levels of TXNIP in
542 KIRC samples and 72 normal samples were evaluated using TCGA
data, and the results showed that KIRC patients with high
TXNIP expression had significantly improved DSS, OS and
progression-free survival (PFS) compared to those with low
TXNIP mRNA expression levels (Fig. 2B-D). Additionally, the levels of
TXNIP expression significantly decreased with higher tumor
grade, stage and TNM stage (Fig.
2E-I). Thus, it can be seen that the high expression of
TXNIP could improve the survival rate of patients with KIRC,
and that its expression level was negatively associated with the
clinicopathological stage.
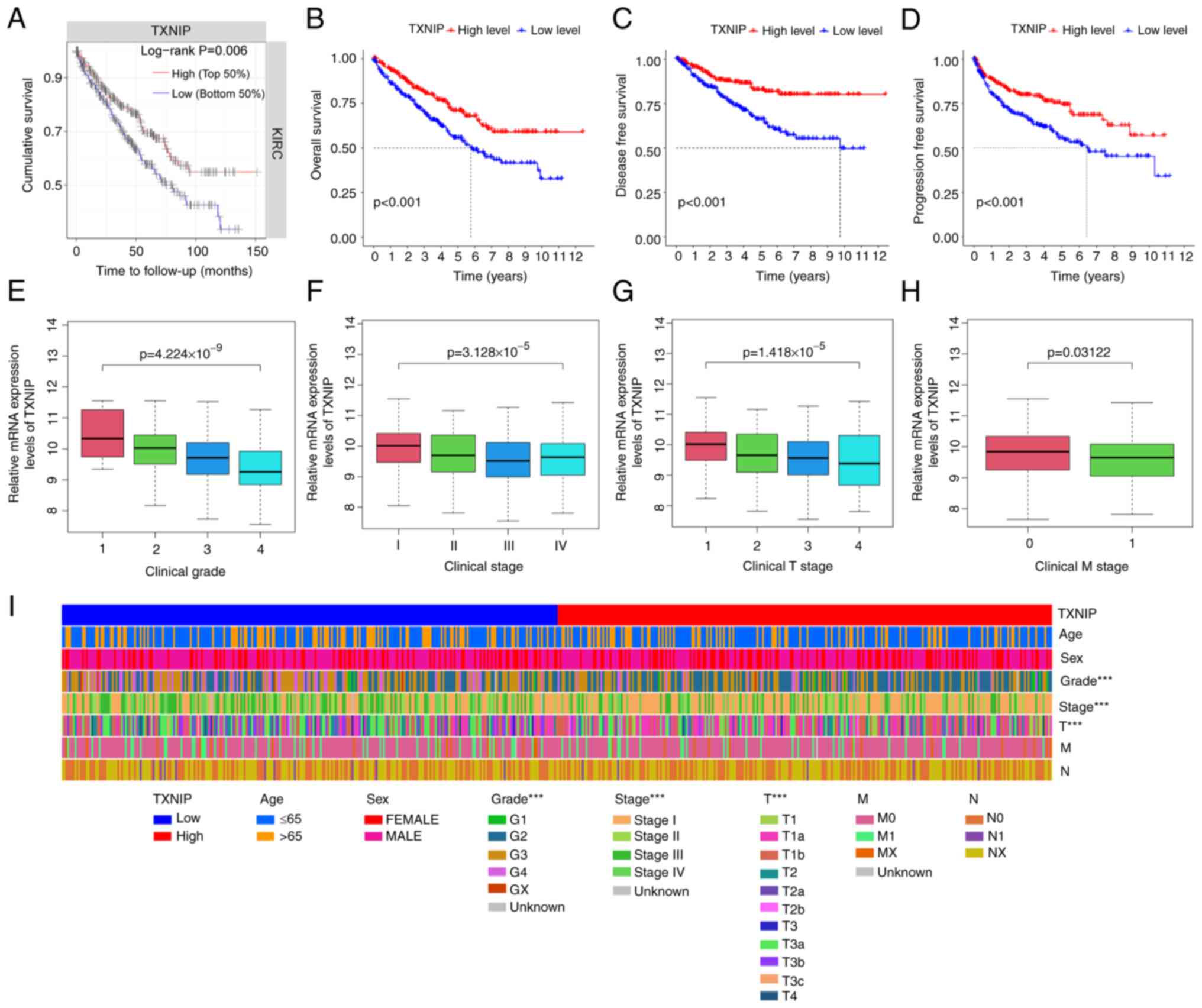 | Figure 2.Association between TXNIP
expression with clinicopathological characteristics of KIRC. (A)
Association between OS and the level of TXNIP expression
using data from the Tumor Immune Estimation Resource database.
Reduced expression of TXNIP was significantly associated
with worse (B) OS, (C) DSS and (D) PFS in patients with KIRC,
compared with those with increased expression. TXNIP
expression significantly decreased with increasing (E) grade, (F)
stage, (G) T stage and (H) the occurrence of distant metastases.
(I) Relationship between low TXNIP expression and age, sex,
grade, stage and metastasis. ***P<0.001. TXNIP,
thioredoxin-interacting protein; KIRC, kidney renal clear cell
carcinoma; OS, overall survival; G, grade; T, tumor; M, metastasis;
N, node. |
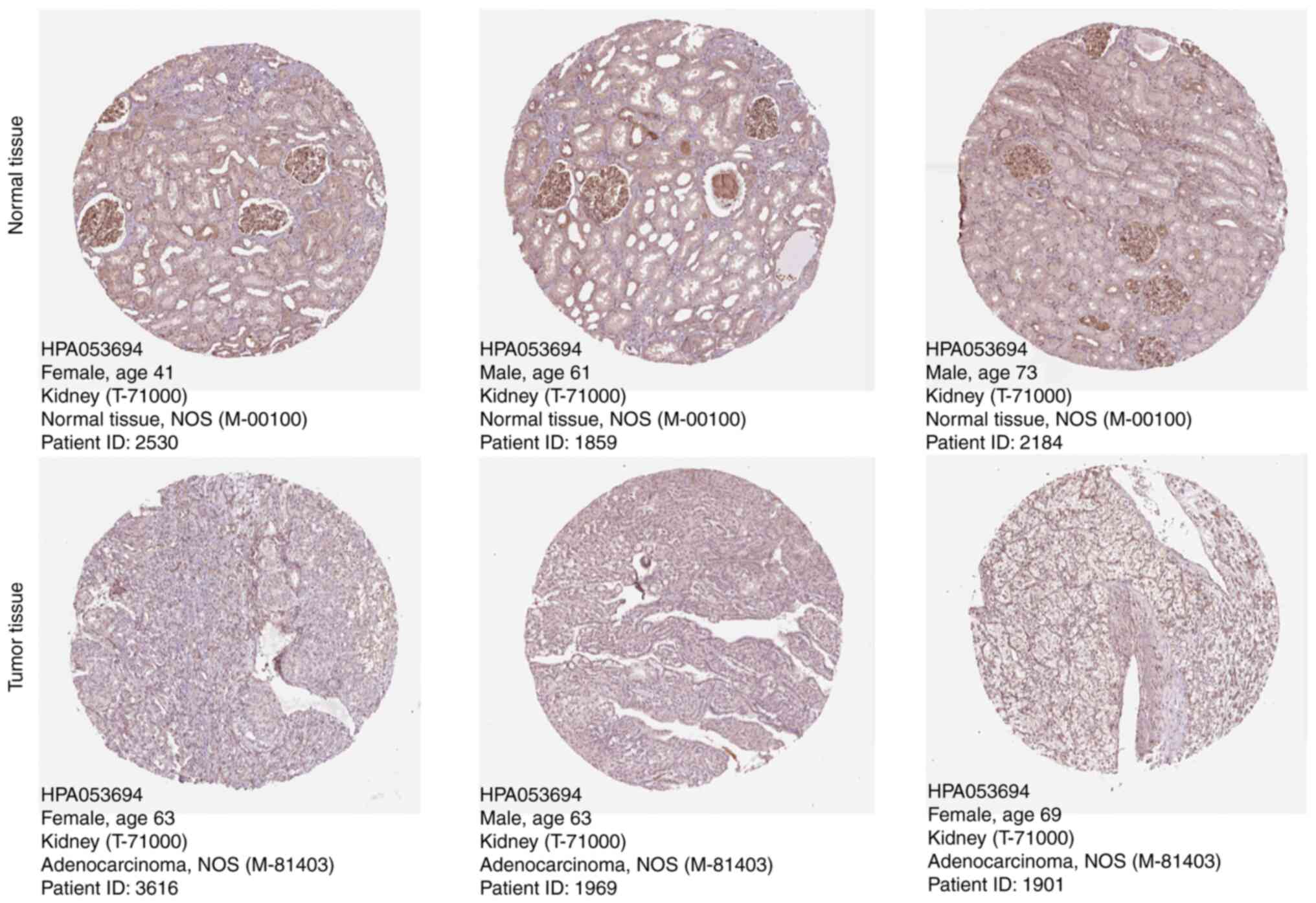
Protein expression of TXNIP in two
types of tissue samples
The TXNIP protein expression level and its clinical
significance was assessed using micrographs from patients with KIRC
which were included HPA database. TXNIP protein expression was
observed to be markedly lower in KIRC tissue samples compared with
normal kidney tissue samples, based on immunohistochemistry
analysis data from the HPA database (Fig. 3). This result indicates that the
protein level of TXNIP in KIRC was also lower than that in normal
kidney tissue compared with the mRNA level.
Independent prognostic value of TXNIP
in KIRC
Univariate Cox regression analysis demonstrated a
significant association between grade and risk scores with OS in
529 patients with KIRC with TXNIP, age, grade and stage
being statistically significant in KIRC (P<0.001), showing good
prognostic value (Fig. 4A).
Multivariate Cox regression analysis showed that grade and risk
scores were independent prognostic indicators for KIRC, and
TXNIP had a high prognostic value in KIRC (P=0.012), while
age, grade and stage still maintained good prognostic value
(P<0.001) (Fig. 4B).
Additionally, nomogram plots were constructed that incorporated
age, TNM stage, stage, sex and grade to forecast the survival of
patients with KIRC at 1, 3 and 5 years, and survival rate was found
to decrease significantly with time (Fig. 4C and D). The results suggest that
TXNIP can be used as an independent prognostic indicator, as
well as age, T staging and stage.
TXNIP co-expression analysis
By analyzing 211 genes co-expressed with
TXNIP, the co-expressed genes were screened using P<0.001
and corFilter=0.7 as threshold conditions (where P<0.001 was an
indication of a significant correlation and corFilter=0.7 was used
as a criterion for identifying co-expressed genes to filter out
irrelevant genes). The results demonstrated that the gene
expression of tudor domain containing 7, cold shock domain
containing E1, enhancer of polycomb homolog 2, round spermatid
basic protein 1 and ribonuclease L had strong and significant
positive correlations with TXNIP mRNA expression (Fig. 5A-F). Furthermore, Kaplan-Meier
analysis demonstrated that a high expression of these five genes
was significantly associated with a good prognosis, compared with a
low expression (Fig. 5G-K).
 | Figure 5.Identification and functional
enrichment analysis of co-expressed genes. (A) Degree of
association between 11 genes with a strong correlation with
TXNIP (gene-gene associations are connected by shading, with
red representing the degree of positive correlation and green
representing the degree of negative correlation). Correlation
between TXNIP and (B) TDRD7, (C) EPC2, (D) RNASEL, (E) RSBN1
and (F) CSDE1. Kaplan-Meier curves of the five co-expressed genes:
(G) TDRD7, (H) EPC2, (I) RNASEL, (J) RSBN1 and (K) CSDE1. (L)
Results of Gene Ontology enrichment analysis of co-expressed genes.
(M) Results of Kyoto Encyclopedia of Genes and Genomes enrichment
analysis of co-expressed genes (q-value is the optimized P-value).
(N) The gene set enrichment analysis results show the five selected
representative pathways. TXNIP, thioredoxin-interacting protein;
TDRD7, tudor domain containing 7; CSDE1, cold shock domain
containing E1; EPC2, enhancer of polycomb homolog 2; RSBN1, round
spermatid basic protein 1; RNASEL, ribonuclease L. |
GO analysis results demonstrated that the function
of TXNIP was mainly enriched in the ‘acute-phase response’,
‘anatomical structure maturation’ and ‘acute-inflammatory
response’. It was also associated with ‘endopeptidase activity’ and
‘serine-type endopeptidase activity’ (Fig. 5L). Moreover, the KEGG analysis
showed there was a high association between the mRNA expression
level of TXNIP and ‘neuroactive ligand-receptor
interactions’ (Fig. 5M). KEGG
genomic analyses demonstrated that five signaling pathways,
including chemokine and T-cell receptor signaling pathways, were
markedly differentially enriched at high mRNA expression levels of
TXNIP (Fig. 5N). This
finding suggests that TXNIP and its co-expressed genes have
some association with the immune response of the organism.
Relationship between mRNA expression
of TXNIP and KIRC immune cell infiltration
Fig. 6A demonstrates
the distribution of the 21 tumor-infiltrating immune cells (TIICs)
in the TCGA-KIRC dataset samples. Additionally, a comparative
analysis of the TXNIP high and low expression groups was
performed to assess the proportion of TIICs in the two groups. The
results showed that seven TIICs differed significantly between the
two groups (Fig. 6B). Among them,
in the TXNIP high expression group, macrophages M1
(P=0.00045), dendritic cells resting (P=0.00028), monocytes
(P=0.0014), macrophages M2 (P=0.0076), neutrophils (P=0.008), T
cells CD4 memory resting (P<0.001) and mast cells resting
(P<0.001) had significantly increased levels of infiltration,
compared with that in the low expression group. In the TXNIP
low expression group, the infiltration levels of macrophages M0
(P<0.001), T cells regulatory (Tregs; P<0.001) and T cells
follicular helper (P<0.001) were significantly increased,
compared with that in the high expression group. Furthermore,
correlation analysis demonstrated that macrophages M1, mast cells
resting, T cells CD4 memory resting and dendritic cells resting
showed a significant positive association with TXNIP
expression, and T cells follicular helper, Tregs and macrophages M0
exhibited a strong negative association with TXNIP
expression (Fig. 6C-I). Fig. 6J not only reaffirms the high
association of TXNIP mRNA expression levels with the immune
cells aforementioned, but also visualizes the positive and negative
association between various types of immune cells and TXNIP
expression. These results indicated that TXNIP was a key
player that regulated the immunological microenvironment of
KIRC.
 | Figure 6.Relationship between TXNIP
expression and immune cell infiltration in KIRC. (A)
Characteristics of 21 tumor-infiltrating immune cells in the
TCGA-KIRC samples. (B) Differences in immune infiltrating cells
between high and low TXNIP expression subgroups. Correlation
between TXNIP expression (C) T cells follicular helper, (D)
T cells regulatory, (E) Macrophages M0, (F) T cells CD4 memory
resting, (G) Macrophages M1, (H) Dendritic cells resting and (I)
mast cells resting. (J) Correlation of TXNIP expression with
21 immune cells. *P<0.05, **P<0.01 and ***P<0.001. TXNIP,
thioredoxin-interacting protein; KIRC, kidney renal clear cell
carcinoma; Abs, absolute value; Cor, correlation coefficient. |
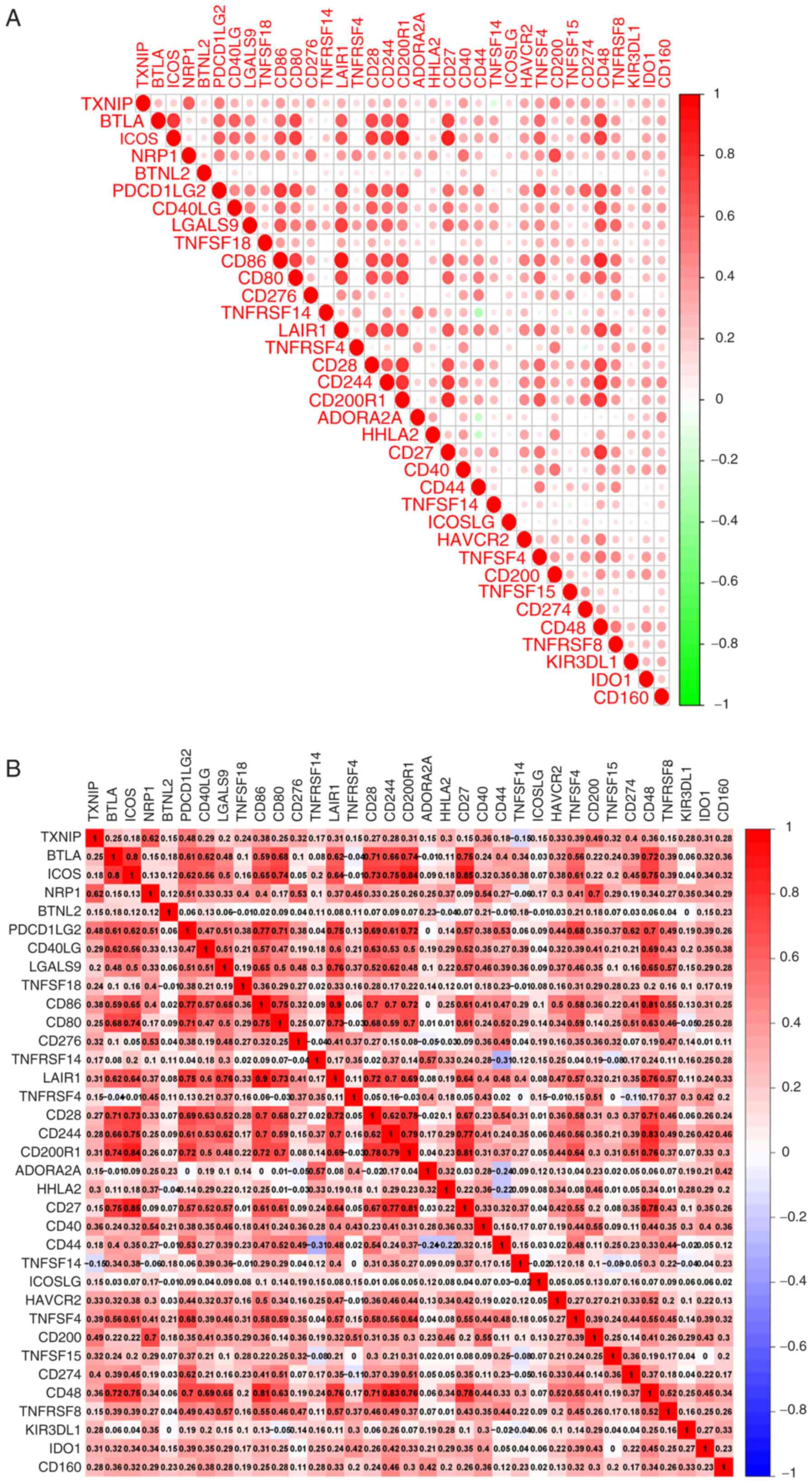
Correlation analysis of immune
checkpoints
Following correlation analysis of the expression
levels of immune checkpoint genes and TXNIP expression, 34
immune checkpoint-related genes were found to be strongly
associated with the mRNA expression levels of TXNIP. With
the exception of tumor necrosis factor superfamily member 14, a
positive correlation between the mRNA expression levels of
TXNIP and almost all the immune checkpoint genes was
demonstrated. neuropilin 1, programmed cell death 1 ligand 2, CD200
and CD274 had the most notable positive associations with
TXNIP expression of those genes assessed (Fig. 7A and B). The aforementioned results
suggest that TXNIP is closely associated with the expression
of most immune checkpoint-related genes.
TXNIP-specific expression in
conventional CD4+ T cells (CD4Tconv)
The association between TXNIP mRNA expression
and several immune cells was assessed using the TISCH public
database. The results demonstrated that TXNIP was mainly
expressed in CD4Tconv cells (Fig. 8A
and B). In addition, it was demonstrated that TXNIP was
predominantly expressed in immune cells and stromal cells in KIRC
(Fig. 8C). The aforementioned
results suggest that TXNIP is associated with
CD4+ T cells to a greater extent than other immune
cells.
 | Figure 8.Characterization of TXNIP
expression in different immune cells of KIRC. UMAP graphs
presenting the distribution of immune cell populations (left) and
their degree of association with TXNIP (right) analyzed
through the (A) KIRC-GSE139555 and (B) KIRC-GSE111360 datasets
using the TISCH database. (C) Degree of relationship between
TXNIP expression and immune cells in different data sets.
TXNIP, thioredoxin-interacting protein; KIRC, kidney renal clear
cell carcinoma; TPM, transcipts per million; CD4Tconv, CD4T
conventional lymphocytes; Treg, regulatory T cells; Tprolif,
proliferating T cells; CD8Tex, exhausted CD8T cells; NK, natural
killer cells; B, blymphocytes; Mono/Macro,
monocytes/macrophages. |
Drug sensitivity analysis of
TXNIP
The CellMiner TM database was used to analyze
whether there was a correlation between TXNIP expression and
drug sensitivity. A total of 12 drugs were screened from 46 drugs,
which were found to be significantly associated with the mRNA
expression level of TXNIP, and the drug sensitivity was
significantly enhanced in the group with high TXNIP
expression compared to the group with low TXNIP expression
(Fig. 9). Among these, afuresertib,
ipatasertib and MK-2206 are AKT kinase inhibitors (41), entinostat and vorinostat are histone
deacetylase (HDAC) inhibitors (42,43),
and WIKI4 and XAV939 are tankyrase inhibitors and have inhibitory
effects on the Wnt/β-catenin signaling pathway (44,45).
Therefore, we hypothesized that the increased expression of
TXNIP may have some effect on the presence of the AKT, HDAC,
tankyrase and Wnt/β-catenin signaling pathways compared to the
generally low expression of TXNIP in cancer.
Cellular experimental validation
TXNIP expression levels in HK-2 and A498
cells differed substantially. The mRNA and protein expression
levels of TXNIP in A498 cells were significantly lower than
those in HK-2 cells, and these levels were significantly increased
following the overexpression of TXNIP, compared with that in
the A498-LV-Empty cells (Fig.
10A-C). Furthermore, the mRNA levels of CD25 and CTLA4, which
are surface markers of CD4+ T cells (46), were significantly reduced after the
overexpression of TXNIP, compared with that in the
A498-LV-Empty cells (Fig. 10C).
Additionally, results from the CCK-8 assay showed that TXNIP
overexpression significantly reduced the capacity of A498 cells to
proliferate (Fig. 10D). These
findings indicate that TXNIP acts as an important oncogene
in KIRC, exerting inhibitory effects on immune escape and the rapid
proliferation of cancer cells.
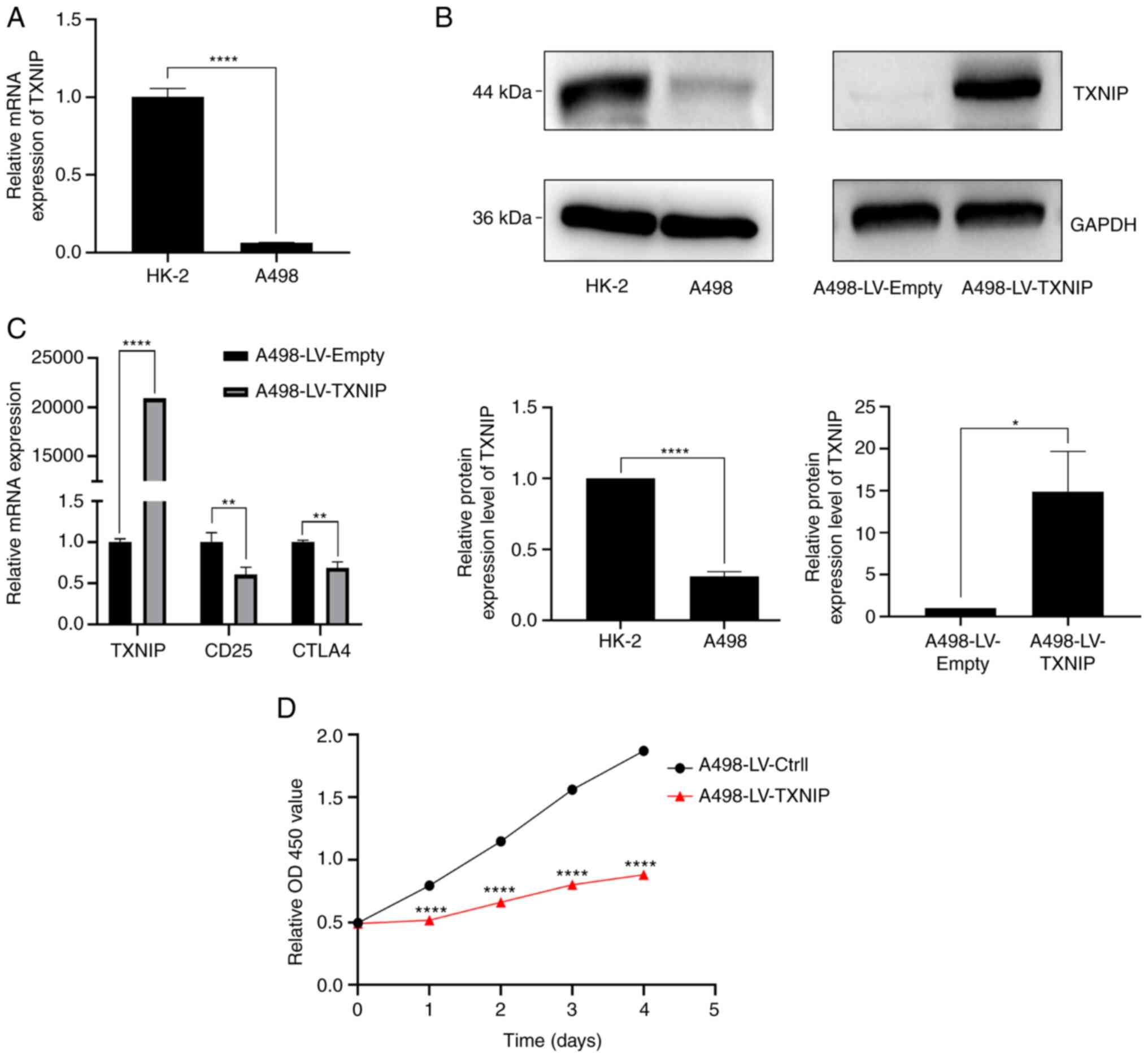 | Figure 10.Cellular assay validation in KIRC.
(A) Differences in TXNIP mRNA expression levels in HK-2 and
A498 cells, assessed using RT-qPCR. (B) Differences in protein
expression levels of TXNIP in HK-2 and A498 cells, and its protein
expression changes in A498 after overexpression of TXNIP,
assessed using western blotting. (C) mRNA expression changes of
CD25 and CTLA4, the surface markers of CD4+ T cells,
after overexpression of TXNIP, assessed using RT-qPCR. (D)
Effect of overexpression of TXNIP on the proliferation of
kidney cancer A498 cells, assessed using the Cell Counting Kit-8
assay. *P<0.05; **P<0.01; ****P<0.0001. TXNIP,
thioredoxin-interacting protein; KIRC, kidney renal clear cell
carcinoma; RT-qPCR, reverse transcription-quantitative PCR; CTLA4,
cytotoxic T-lymphocyte-associated protein 4; OD, optical
density. |
Discussion
KIRC is a heterogeneous disease with a poor
prognosis (47). The limited
predictors to assess the risk of KIRC may result in inaccurate
grading, lowering the survival rate of patients with KIRC (48). Hence, the identification of reliable
biomarkers for the prognosis and treatment of KIRC is urgently
required. In the present study, high-throughput RNA sequencing data
from the TCGA database was used and was further validated using
RT-qPCR and western blot analyses. The findings revealed that
TXNIP expression was significantly associated with OS, DSS
and PFS, as well as the infiltration levels of TIICs. These
findings highlight the potential for the use of TXNIP as a
prognostic biomarker and therapeutic target for KIRC.
The primary structure of TXNIP includes an
inhibitor-like N-terminus (10–152 aa) and a C-terminus (175–298
aa), indicating its role in the inhibition of the function of
binding proteins (49–51). TXNIP is a recognized oncogene
repressor in several types of breast cancer (52,53).
Moreover, TXNIP expression has been reported to be
significantly downregulated in breast, liver and lung cancers
(54–56). In the present study, the expression
of TXNIP in 33 tumor types was demonstrated, and it was
compared with that of normal tissues using data from online
databases. The results revealed that TXNIP was significantly
downregulated in most cancers (Fig.
1B). Furthermore, RT-qPCR and western blotting demonstrated
that TXNIP expression was significantly reduced in KIRC.
The significance of TXNIP as a prognostic
factor has been demonstrated in patients with cancer. In
hepatocellular carcinoma, lower TXNIP expression was notably
associated with a worse prognosis (55). TXNIP was also identified as
an independent prognostic factor for distant metastasis-free
survival and OS in gastroesophageal adenocarcinoma (57) and the prognostic and predictive
value of TXNIP have been established in human breast cancer
(58,59). The present study found that reduced
mRNA expression of TXNIP was associated with unfavorable
clinicopathological features, including high histological grade,
stage and the occurrence of distant metastases (Fig. 2E-I). Multivariate Cox regression
analysis demonstrated that TXNIP was an independent
prognostic factor that significantly impacted PFS, DSS and OS.
Reduced TXNIP expression was also associated with adverse
clinical outcomes in KIRC. Furthermore, the analyses, stratified by
sex, age, TNM stage, stage and grade, demonstrated that patients
with high mRNA expression levels of TXNIP expression
experienced significantly greater OS in comparison with those
patients with low levels of expression. The findings by Gao et
al (32) similarly showed that
reduced TXNIP mRNA expression was significantly associated
with clinical stage. In addition, the study also showed that
TXNIP was an independent prognostic factor for KIRC by
univariate and multivariate Cox analysis (32). In contrast to this study, the
present study not only used TCGA, the TIMER and HPA databases and
related experiments (Fig. 10A and
B) for further assessment to ensure the reliability of the
results, but also screened for co-expressed genes and performed
Kaplan-Meier analyses on the co-expressed genes (Fig. 5G-K) to evaluate their association
with prognosis.
In the tumor microenvironment, the ecosystem
established by TIICs serves a crucial role in the regulation of
cancer progression in KIRC, and the immune responses are a critical
determinant of survival outcomes. Hence, the proportion of TIICs in
cancer potentially has prognostic value (60–62).
Further analysis using TCGA and CIBERSORT revealed that certain
immune cells associated with TXNIP expression were
significantly associated with the survival of patients with KIRC.
Increased TXNIP mRNA expression was associated with an
increase in macrophages M1, T cells CD4 memory resting, mast cells
resting and dendritic cells resting. Conversely, there was a
decrease in the proportion of T cells follicular helper, Tregs and
macrophages M0 (Fig. 6B). The
ability of the chemokine signaling pathway to control T cell
migration toward chemokine sources has been demonstrated. In the
context of cancer, ligands for C-C chemokine receptor type 5 and
CXC motif chemokine receptor 3 have also been reported to be
associated with the degree of tumor-infiltrating lymphocyte (TIL)
infiltration in cancer (63–65).
Reactive oxygen species (ROS) metabolism is controlled by the
essential component TXNIP and cytosolic ROS in
CD4+ T lymphocytes rapidly decrease during the
contraction phase. Once CD4+ T cells activation is
attenuated, TXNIP has been reported to be quickly
upregulated, and furthermore, TXNIP expression has also been
directly associated with the development of allergen-specific
memory Th2 cells (66). Results
from the present study demonstrated that the T-cell receptor and
chemokine pathways were notably differentially enriched in KIRC
with high TXNIP mRNA expression, according to the gene set
enrichment analysis data (Fig. 5N).
Additionally, the outcomes of RT-qPCR assays revealed that
TXNIP mRNA expression was significantly associated with
CD4+ T cells in KIRC (Fig.
10C). These findings demonstrate that TXNIP regulates
immune infiltration and may affect the levels of ROS in patients
with KIRC.
Additionally, the present study found a significant
association between the mRNA levels of TXNIP and sensitivity
to 12 antitumor drugs (Fig. 9).
WIKI4 and XAV-939 are known to suppress tankyrase activity,
inhibiting Wnt/β-catenin signaling pathway-mediated transcription
(44,45). Afuresertib, ipatasertib and MK-2206
are key antitumor drugs as they potentially inhibit AKT kinase
(41). Elevated levels of
TXNIP expression can suppress the activity of the
Wnt/β-catenin and PI3K/AKT/mTOR signaling pathways (67–69).
This mechanism may therefore be related to enhanced drug
sensitivity, but further evaluation is needed.
Whilst the present study offers insights on the
potential function of TXNIP on KIRC prognosis and immune
infiltration, certain limitations merit attention. Primarily, the
sample size was only 614 cases and a larger data set is needed to
confirm the accuracy of the findings. Additionally, further
experimental studies are needed to confirm the functional role of
TXNIP in KIRC.
In summary, the present study indicated that there
is a strong association between decreased TXNIP expression
levels, insufficient immune cell infiltration and poor prognosis in
patients with KIRC, and that the reduction in TXNIP
expression levels may impair the antitumor activity of the immune
system in patients with KIRC. These findings provide insights that
may prove beneficial in the development of novel immunotherapeutic
approaches.
Acknowledgements
Not applicable.
Funding
Funding was received from the Guangxi Zhuang Autonomous Region
Health and Family Planning Commission (grant. no. Z-L20221837).
Availability of data and materials
The datasets used and/or analyzed during the
current study are available from the corresponding author on
reasonable request. The data accessed in the present study may be
found in the TCGA (https://portal.gdc.cancer.gov/), HPA (http://www.proteinatls.org), and TIMER (https://cistrome.shinyapps.io/timer/)
databases.
Authors' contributions
WL, ZX and MD contributed to the development of the
statistical analysis strategies to collect, evaluate and organize
data from the databases; design, implementation and processing of
experimental data; analysis of data; and drafting and revision of
the manuscript. XL and ZH designed and directed the study, provided
experimental design ideas, guidance on data processing and advice
on manuscript revision. WL and ZX wrote the main manuscript text
and prepared Fig. 1, Fig. 2, Fig.
3, Fig. 4. MD prepared Fig. 5, Fig.
6, Fig. 7, Fig. 8, Fig.
9, Fig. 10. All authors have
read and approved the final manuscript. WL and ZX confirm the
authenticity of all the raw data.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
ACC
|
adrenocortical carcinoma
|
|
BLCA
|
bladder urothelial carcinoma
|
|
BRCA
|
breast invasive carcinoma
|
|
CESC
|
cervical squamous cell carcinoma and
endocervical adenocarcinoma
|
|
CHOL
|
cholangiocarcinoma
|
|
COAD
|
colon adenocarcinoma
|
|
DLBC
|
lymphoid neoplasm diffuse large
B-cell lymphoma
|
|
ESCA
|
esophageal carcinoma
|
|
GBM
|
glioblastoma multiforme
|
|
HNSC
|
head and neck squamous cell
carcinoma
|
|
KICH
|
kidney chromophobe
|
|
KIRC
|
kidney renal clear cell carcinoma
|
|
KIRP
|
kidney renal papillary cell
carcinoma
|
|
LAML
|
acute myeloid leukemia
|
|
LGG
|
brain lower grade glioma
|
|
LIHC
|
liver hepatocellular carcinoma
|
|
LUAD
|
lung adenocarcinoma
|
|
LUSC
|
lung squamous cell carcinoma
|
|
MESO
|
mesothelioma
|
|
OV
|
ovarian serous cystadenocarcinoma
|
|
PAAD
|
pancreatic adenocarcinoma
|
|
PCPG
|
pheochromocytoma and
paraganglioma
|
|
PRAD
|
prostate adenocarcinoma
|
|
READ
|
rectum adenocarcinoma
|
|
SARC
|
sarcoma
|
|
SKCM
|
skin cutaneous melanoma
|
|
STAD
|
stomach adenocarcinoma
|
|
TGCT
|
testicular germ cell tumor
|
|
THCA
|
thyroid carcinoma
|
|
THYM
|
thymoma
|
|
UCEC
|
uterine corpus endometrial
carcinoma
|
|
UCS
|
uterine carcinosarcoma
|
|
UVM
|
uveal melanoma
|
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2019. CA Cancer J Clin. 69:7–34. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Shuch B, Amin A, Armstrong AJ, Eble JN,
Ficarra V, Lopez-Beltran A, Martignoni G, Rini BI and Kutikov A:
Understanding pathologic variants of renal cell carcinoma:
Distilling therapeutic opportunities from biologic complexity. Eur
Urol. 67:85–97. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sonpavde G, Choueiri TK, Escudier B,
Ficarra V, Hutson TE, Mulders PF, Patard JJ, Rini BI, Staehler M,
Sternberg CN and Stief CG: Sequencing of agents for metastatic
renal cell carcinoma: Can we customize therapy? Eur Urol.
61:307–316. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bella L, Zona S, Nestal de Moraes G and
Lam EW: Foxm1: A key oncofoetal transcription factor in health and
disease. Semin Cancer Biol. 29:32–39. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Mitchell TJ, Turajlic S, Rowan A, Nicol D,
Farmery JHR, O'Brien T, Martincorena I, Tarpey P, Angelopoulos N,
Yates LR, et al: Timing the landmark events in the evolution of
clear cell renal cell cancer: Tracerx Renal. Cell. 173:611–623.e17.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bedke J, Gauler T, Grünwald V, Hegele A,
Herrmann E, Hinz S, Janssen J, Schmitz S, Schostak M, Tesch H, et
al: Systemic therapy in metastatic renal cell carcinoma. World J
Urol. 35:179–188. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chatterjee N and Bivona TG: Polytherapy
and targeted cancer drug resistance. Trends Cancer. 5:170–182.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Noessner E, Brech D, Mendler AN, Masouris
I, Schlenker R and Prinz PU: Intratumoral alterations of
dendritic-cell differentiation and CD8(+) T-cell anergy are immune
escape mechanisms of clear cell renal cell carcinoma.
Oncoimmunology. 1:1451–1453. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Choueiri TK, Fishman MN, Escudier B,
McDermott DF, Drake CG, Kluger H, Stadler WM, Perez-Gracia JL,
McNeel DG, Curti B, et al: Immunomodulatory activity of nivolumab
in metastatic renal cell carcinoma. Clin Cancer Res. 22:5461–5471.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lalani AA, McGregor BA, Albiges L,
Choueiri TK, Motzer R, Powles T, Wood C and Bex A: Systemic
treatment of metastatic clear cell renal cell carcinoma in 2018:
Current paradigms, use of immunotherapy, and future directions. Eur
Urol. 75:100–110. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Carlo MI, Voss MH and Motzer RJ:
Checkpoint inhibitors and other novel immunotherapies for advanced
renal cell carcinoma. Nat Rev Urol. 13:420–431. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Gill DM and Agarwal N: Cancer
immunotherapy: A paradigm shift in the treatment of advanced
urologic cancers. Urol Oncol. 35:676–677. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Martínez-Salamanca JI, Huang WC, Millán I,
Bertini R, Bianco FJ, Carballido JA, Ciancio G, Hernández C,
Herranz F, Haferkamp A, et al: Prognostic impact of the 2009
UICC/AJCC TNM staging system for renal cell carcinoma with venous
extension. Eur Urol. 59:120–127. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Brahmer JR, Drake CG, Wollner I, Powderly
JD, Picus J, Sharfman WH, Stankevich E, Pons A, Salay TM, McMiller
TL, et al: Phase I study of single-agent anti-programmed death-1
(MDX-1106) in refractory solid tumors: Safety, clinical activity,
pharmacodynamics, and immunologic correlates. J Clin Oncol.
28:3167–3175. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lipson EJ, Sharfman WH, Drake CG, Wollner
I, Taube JM, Anders RA, Xu H, Yao S, Pons A, Chen L, et al: Durable
cancer regression off-treatment and effective reinduction therapy
with an anti-PD-1 antibody. Clin Cancer Res. 19:462–468. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Topalian SL, Hodi FS, Brahmer JR,
Gettinger SN, Smith DC, McDermott DF, Powderly JD, Carvajal RD,
Sosman JA, Atkins MB, et al: Safety, activity, and immune
correlates of anti-PD-1 antibody in cancer. N Engl J Med.
366:2443–2454. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Motzer RJ, Rini BI, McDermott DF, Redman
BG, Kuzel TM, Harrison MR, Vaishampayan UN, Drabkin HA, George S,
Logan TF, et al: Nivolumab for metastatic renal cell carcinoma:
Results of a randomized phase ii trial. J Clin Oncol. 33:1430–1437.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Motzer RJ, Escudier B, McDermott DF,
George S, Hammers HJ, Srinivas S, Tykodi SS, Sosman JA, Procopio G,
Plimack ER, et al: Nivolumab versus everolimus in advanced
renal-cell carcinoma. N Engl J Med. 373:1803–1813. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Brahmer JR, Tykodi SS, Chow LQ, Hwu WJ,
Topalian SL, Hwu P, Drake CG, Camacho LH, Kauh J, Odunsi K, et al:
Safety and activity of anti-PD-L1 antibody in patients with
advanced cancer. N Engl J Med. 366:2455–2465. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
McDermott DF, Sosman JA, Sznol M, Massard
C, Gordon MS, Hamid O, Powderly JD, Infante JR, Fassò M, Wang YV,
et al: Atezolizumab, an anti-programmed death-ligand 1 antibody, in
metastatic renal cell carcinoma: Long-term safety, clinical
activity, and immune correlates from a phase Ia study. J Clin
Oncol. 34:833–842. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yang JC, Hughes M, Kammula U, Royal R,
Sherry RM, Topalian SL, Suri KB, Levy C, Allen T, Mavroukakis S, et
al: Ipilimumab (anti-CTLA4 antibody) causes regression of
metastatic renal cell cancer associated with enteritis and
hypophysitis. J Immunother. 30:825–830. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sharpe AH and Pauken KE: The diverse
functions of the PD1 inhibitory pathway. Nat Rev Immunol.
18:153–167. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chen KS and DeLuca HF: Isolation and
characterization of a novel cDNA from HL-60 cells treated with
1,25-dihydroxyvitamin D-3. Biochim Biophys Acta. 1219:26–32. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wu N, Zheng B, Shaywitz A, Dagon Y, Tower
C, Bellinger G, Shen CH, Wen J, Asara J, McGraw TE, et al:
Ampk-dependent degradation of TXNIP upon energy stress leads to
enhanced glucose uptake via GLUT1. Mol Cell. 49:1167–1175. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Shen L, O'Shea JM, Kaadige MR, Cunha S,
Wilde BR, Cohen AL, Welm AL and Ayer DE: Metabolic reprogramming in
triple-negative breast cancer through Myc suppression of TXNIP.
Proc Natl Acad Sci USA. 112:5425–5430. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Han SH, Jeon JH, Ju HR, Jung U, Kim KY,
Yoo HS, Lee YH, Song KS, Hwang HM, Na YS, et al: Vdup1 upregulated
by TGF-beta1 and 1,25-dihydorxyvitamin d3 inhibits tumor cell
growth by blocking cell-cycle progression. Oncogene. 22:4035–4046.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Saxena G, Chen J and Shalev A:
Intracellular shuttling and mitochondrial function of
thioredoxin-interacting protein. J Biol Chem. 285:3997–4005. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhou R, Yazdi AS, Menu P and Tschopp J: A
role for mitochondria in NLRP3 inflammasome activation. Nature.
469:221–225. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhou R, Tardivel A, Thorens B, Choi I and
Tschopp J: Thioredoxin-interacting protein links oxidative stress
to inflammasome activation. Nat Immunol. 11:136–140. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Jiao D, Huan Y, Zheng J, Wei M, Zheng G,
Han D, Wu J, Xi W, Wei F, Yang AG, et al: UHRF1 promotes renal cell
carcinoma progression through epigenetic regulation of TXNIP.
Oncogene. 38:5686–5699. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Meszaros M, Yusenko M, Domonkos L, Peterfi
L, Kovacs G and Banyai D: Expression of TXNIP is associated with
angiogenesis and postoperative relapse of conventional renal cell
carcinoma. Sci Rep. 11:172002021. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Gao Y, Qi JC, Li X, Sun JP, Ji H and Li
QH: Decreased expression of TXNIP predicts poor prognosis in
patients with clear cell renal cell carcinoma. Oncol Lett.
19:763–770. 2020.PubMed/NCBI
|
|
33
|
Pan M, Zhang F, Qu K, Liu C and Zhang J:
TXNIP: A double-edged sword in disease and therapeutic outlook.
Oxid Med Cell Longev. 2022:78051152022. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Hutter C and Zenklusen JC: The cancer
genome atlas: Creating lasting value beyond its data. Cell.
173:283–285. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Subramanian A, Tamayo P, Mootha VK,
Mukherjee S, Ebert BL, Gillette MA, Paulovich A, Pomeroy SL, Golub
TR, Lander ES and Mesirov JP: Gene set enrichment analysis: A
knowledge-based approach for interpreting genome-wide expression
profiles. Proc Natl Acad Sci USA. 102:15545–15550. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Yu G, Wang LG, Han Y and He QY:
clusterProfiler: An R package for comparing biological themes among
gene clusters. OMICS. 16:284–287. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Chen B, Khodadoust MS, Liu CL, Newman AM
and Alizadeh AA: Profiling tumor infiltrating immune cells with
CIBERSORT. Methods Mol Biol. 1711:243–259. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Li B, Severson E, Pignon JC, Zhao H, Li T,
Novak J, Jiang P, Shen H, Aster JC, Rodig S, et al: Comprehensive
analyses of tumor immunity: Implications for cancer immunotherapy.
Genome Biol. 17:1742016. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Shankavaram UT, Varma S, Kane D, Sunshine
M, Chary KK, Reinhold WC, Pommier Y and Weinstein JN: CellMiner: A
relational database and query tool for the NCI-60 cancer cell
lines. BMC Genomics. 10:2772009. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Wu JH, Limmer AL, Narayanan D, Doan HQ,
Simonette RA, Rady PL and Tyring SK: The novel AKT inhibitor
afuresertib suppresses human Merkel cell carcinoma MKL-1 cell
growth. Clin Exp Dermatol. 46:1551–1554. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Trapani D, Esposito A, Criscitiello C,
Mazzarella L, Locatelli M, Minchella I, Minucci S and Curigliano G:
Entinostat for the treatment of breast cancer. Expert Opin Investig
Drugs. 26:965–971. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Athira KV, Sadanandan P and Chakravarty S:
Repurposing Vorinostat for the treatment of disorders affecting
brain. Neuromolecular Med. 23:449–465. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
James RG, Davidson KC, Bosch KA, Biechele
TL, Robin NC, Taylor RJ, Major MB, Camp ND, Fowler K, Martins TJ
and Moon RT: WIKI4, a novel inhibitor of tankyrase and
wnt/ß-catenin signaling. PLoS One. 7:e504572012. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Yu J, Liu D, Sun X, Yang K, Yao J, Cheng
C, Wang C and Zheng J: CDX2 inhibits the proliferation and tumor
formation of colon cancer cells by suppressing Wnt/β-catenin
signaling via transactivation of GSK-3β and Axin2 expression. Cell
Death Dis. 10:262019. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Haddadi MH and Negahdari B: Clinical and
diagnostic potential of regulatory T cell markers: From bench to
bedside. Transplant Immunol. 70:1015182022. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Wang L, Zhu Y, Ren Z, Sun W, Wang Z, Zi T,
Li H, Zhao Y, Qin X, Gao D, et al: An immunogenic cell
death-related classification predicts prognosis and response to
immunotherapy in kidney renal clear cell carcinoma. Front Oncol.
13:11478052023. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Sun Z, Tao W, Guo X, Jing C, Zhang M, Wang
Z, Kong F, Suo N, Jiang S and Wang H: Construction of a
Lactate-related prognostic signature for predicting prognosis,
tumor microenvironment, and immune response in kidney renal clear
cell carcinoma. Front Immunol. 13:8189842022. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Zhou J, Yu Q and Chng WJ: TXNIP (VDUP-1,
TBP-2): A major redox regulator commonly suppressed in cancer by
epigenetic mechanisms. Int J Biochem Cell Biol. 43:1668–1673. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Patwari P, Higgins LJ, Chutkow WA,
Yoshioka J and Lee RT: The interaction of thioredoxin with Txnip.
Evidence for formation of a mixed disulfide by disulfide exchange.
J Biol Chem. 281:21884–21891. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Zhang P, Wang C, Gao K, Wang D, Mao J, An
J, Xu C, Wu D, Yu H, Liu JO and Yu L: The ubiquitin ligase itch
regulates apoptosis by targeting thioredoxin-interacting protein
for ubiquitin-dependent degradation. J Biol Chem. 285:8869–8879.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Iqbal MA, Chattopadhyay S, Siddiqui FA, Ur
Rehman A, Siddiqui S, Prakasam G, Khan A, Sultana S and Bamezai RN:
Silibinin induces metabolic crisis in triple-negative breast cancer
cells by modulating EGFR-MYC-TXNIP axis: Potential therapeutic
implications. FEBS J. 288:471–485. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Chen D, Dang BL, Huang JZ, Chen M, Wu D,
Xu ML, Li R and Yan GR: Mir-373 drives the
epithelial-to-mesenchymal transition and metastasis via the
mir-373-TXNIP-HIF1α-TWIST signaling axis in breast cancer.
Oncotarget. 6:32701–32712. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Cadenas C, Franckenstein D, Schmidt M,
Gehrmann M, Hermes M, Geppert B, Schormann W, Maccoux LJ, Schug M,
Schumann A, et al: Role of thioredoxin reductase 1 and thioredoxin
interacting protein in prognosis of breast cancer. Breast Cancer
Res. 12:R442010. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Hamilton JP, Potter JJ, Koganti L, Meltzer
SJ and Mezey E: Effects of vitamin D3 stimulation of
thioredoxin-interacting protein in hepatocellular carcinoma.
Hepatol Res. 44:1357–1366. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Hong SY, Yu FX, Luo Y and Hagen T:
Oncogenic activation of the PI3K/AKT pathway promotes cellular
glucose uptake by downregulating the expression of
thioredoxin-interacting protein. Cell Signal. 28:377–383. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Woolston CM, Madhusudan S, Soomro IN, Lobo
DN, Reece-Smith AM, Parsons SL and Martin SG: Thioredoxin
interacting protein and its association with clinical outcome in
gastro-oesophageal adenocarcinoma. Redox Biol. 1:285–291. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Yang MH, Wu MZ, Chiou SH, Chen PM, Chang
SY, Liu CJ, Teng SC and Wu KJ: Direct regulation of TWIST by
HIF-1alpha promotes metastasis. Nat Cell Biol. 10:295–305. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Sheth SS, Bodnar JS, Ghazalpour A,
Thipphavong CK, Tsutsumi S, Tward AD, Demant P, Kodama T, Aburatani
H and Lusis AJ: Hepatocellular carcinoma in Txnip-deficient mice.
Oncogene. 25:3528–3536. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Grivennikov SI, Greten FR and Karin M:
Immunity, inflammation, and cancer. Cell. 140:883–899. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Picard E, Verschoor CP, Ma GW and Pawelec
G: Relationships between immune landscapes, genetic subtypes and
responses to immunotherapy in colorectal cancer. Front Immunol.
11:3692020. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Wang SS, Liu W, Ly D, Xu H, Qu L and Zhang
L: Tumor-infiltrating B cells: Their role and application in
anti-tumor immunity in lung cancer. Cell Mol Immunol. 16:6–18.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Ribas A and Wolchok JD: Cancer
immunotherapy using checkpoint blockade. Science. 359:1350–1355.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Denkert C, von Minckwitz G, Brase JC, Sinn
BV, Gade S, Kronenwett R, Pfitzner BM, Salat C, Loi S, Schmitt WD,
et al: Tumor-infiltrating lymphocytes and response to neoadjuvant
chemotherapy with or without carboplatin in human epidermal growth
factor receptor 2-positive and triple-negative primary breast
cancers. J Clin Oncol. 33:983–991. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Ding Q, Lu P, Xia Y, Ding S, Fan Y, Li X,
Han P, Liu J, Tian D and Liu M: CXCL9: Evidence and contradictions
for its role in tumor progression. Cancer Med. 5:3246–3259. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Kokubo K, Hirahara K, Kiuchi M, Tsuji K,
Shimada Y, Sonobe Y, Shinmi R, Hishiya T, Iwamura C, Onodera A and
Nakayama T: Thioredoxin-interacting protein is essential for memory
T cell formation via the regulation of the redox metabolism. Proc
Natl Acad Sci USA. 120:e22183451202023. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Zhu J and Han S: Histone deacetylase 10
exerts anti-tumor effects on cervical cancer via a novel
microRNA-223/TXNIP/Wnt/β-catenin pathway. IUBMB Life. Jan
22–2021.(Epub ahead of print). doi: 10.1002/iub.2448. View Article : Google Scholar
|
|
68
|
Dong F, Dong S, Liang Y, Wang K, Qin Y and
Zhao X: Mir-20b inhibits the senescence of human umbilical vein
endothelial cells through regulating the Wnt/β-catenin pathway via
the TXNIP/NLRP3 axis. Int J Mol Med. 45:847–857. 2020.PubMed/NCBI
|
|
69
|
Ao H, Li H, Zhao X, Liu B and Lu L: TXNIP
positively regulates the autophagy and apoptosis in the rat müller
cell of diabetic retinopathy. Life Sci. 267:1189882021. View Article : Google Scholar : PubMed/NCBI
|