Introduction
Radical treatment of gastric cancer is possible if
the disease is diagnosed at an early stage, prior to metastasis.
Gastric cancer causes approximately 800,000 deaths worldwide
annually (1). Prognosis is poor and
the 5-year survival rate is <5–15%, as most patients present
with advanced disease. Advanced gastric cancer is not particularly
sensitive to chemotherapy (2) as
the benefits of these different drugs, independently or in
combination remain to be elucidated. Resistance of tumor cells
towards chemotherapeutic drugs and tumor metastasis have been
observed. Of patients presenting with metastatic cancer ~90% become
resistant to chemotherapy (3). This
occurs readily in cancers of the breast, prostate, lung, pancreas
and colon (4–8). Therefore, it is necessary to
re-evaluate the drugs commonly used in the treatment of gastric
cancer.
Ara-C, a deoxycytidine analog affecting DNA
synthesis, is an effective inhibitor that changes into diphosphate
or triphosphare cytarabine via phosphatase in vivo.
Triphosphare cytarabine can be a powerful inhibitor of the
synthesis of DNA polymerase, which can inhibit the conversion of
cytidine diphosphate into deoxycytidine diphosphate, thus
inhibiting polymerization and synthetic DNA and interfering with
cell proliferation (9). Due to its
inhibitory effect on cell proliferation, Ara-C as a routine
anticancer drug is widely and effectively used in the clinical
treatment of leukemia (10). The
treatment of gastric cancer with Ara-C as well as the underlying
mechanism involved remain to be elucidated.
In the present study, we found that Ara-C did not
inhibit gastric cancer cell proliferation. Conversely, Ara-C was
able to activate the ERK signaling pathway, resulting in the high
expression of CD-147-MMP-2/MMP-9 and an increase in the
aggressiveness of gastric cancer cells.
Materials and methods
Reagents
Ara-C was obtained from Sigma (St. Louis, MO, USA).
It was dissolved in sterile H2O to a stock concentration
of 0.4 g/ml. U-0126 was obtained from Cell Signaling Technology
(Beverly, MA, USA) and dissolved in DMSO (Sigma) at a stock
concentration of 10 mM. The stock solutions were wrapped in foil
and maintained at 4°C or −20°C.
Cell cultures
NB4 and Jurkat cell lines and the AGS and MKN-45
gastric cancer cell line were purchased from the Shanghai
Institutes for Biological Sciences (Shanghai, China). The cells
were maintained in humidified room air containing 5% CO2
at 37°C, NB4 and Jurkat were cultured in RPMI-1640 medium and AGS
and MKN-45 cells were cultured in DMEM medium supplemented with 10%
fetal bovine serum (FBS) and 1% penicillin-streptomycin. RPMI-1640,
DMEM and FBS were purchased from Gibco-BRL (Gibco, Long Island, NY,
USA). Cells in the logarithmic phase of growth were used in all the
experiments.
Cell Counting Kit-8
The cells were seeded at a concentration of
2×103 cells/200 μl/well in 96-well culture plates for a
cell proliferation assay with Cell Counting Kit-8 reagent (CCK-8;
Dojindo Molecular Technologies, Inc., Kumamoto, Japan). Briefly,
the cultured wells were treated with 20 μl/well of CCK-8 for 2 h
prior to the end of incubation and the optical density of wells was
measured at 450 nm using a microplate reader (Bio-Rad, Hercules,
CA, USA). Results of the cell proliferation measurement were
expressed as the absorbance at OD450.
Gelatinase zymography
Gelatinase zymography was performed in 10% Novex
pre-cast SDS polyacrylamide gel in the presence of 0.1% gelatin
under non-reducing conditions. Culture media (20 μl) were mixed
with sample buffer and loaded with SDS-PAGE with Tris glycine SDS
buffer, according to the manufacturer’s instructions (Novex). The
samples were not boiled prior to electrophoresis. Following
electrophoresis the gels were washed twice in 2.5% Triton X-100 for
30 min at room temperature to remove SDS. The gels were then
incubated at 37°C overnight in substrate buffer containing 50 mM
Tris HCl and 10 mM CaCl2 at pH 8.0, stained with 0.5%
Coomassie blue R250 in 50% methanol and 10% glacial acetic acid for
30 min, and destained. Protein standards were run concurrently and
approximate molecular weights were determined by plotting the
relative mobilities of known proteins.
RNA extraction and reverse
transcriptase-polymerase chain reaction (RT-PCR)
Total RNA was extracted from the cells using RNAiso™
plus (Takara Bio, Otsu, Japan). The concentration and purity of RNA
were determined by absorbance at 260/280 nm. cDNA was synthesized
from 1-μg total RNA using an RNA PCR kit (both from Takara Bio,
Shiga, Japan). Total RNA was reverse transcribed (10 μl of total
volume) at 42°C for 30 min, 99°C for 5 min and 4°C for 5 min. The
total volume of PCR reaction was 20 μl. PCR conditions were 1 cycle
at 94°C for 2 min, 30 cycles at 94°C for 30 sec, 55°C for 30 sec,
and 72°C for 2 min. The primers used were: GAPDH forward: 5′-TGG
ACT CTG GAA TCC ATT CTG-3′ and reverse: 5′-AAA ATC CCT GTT CCC ACT
CA-3′, CD-147 forward: 5′-CCA TGC TGG TCT GCA AGT CAG-3′ and
reverse: 5′-CCG TTC ATG AGG GCC TTG TC-3′, MMP-2 forward: 5′-GAA
GGC TGT GTT CTT TGC AG-3′ and reverse: 5′-AGG CTG GTC AGT GGC
TTG-3′ and MMP-9 forward: 5′-TGC CAG TTT CCA TTC ATC TTC CAA-3′ and
reverse: 5′-CTG CGG TGT GGT GGT GGT T-3′. PCR was performed on
Bio-Rad MyCycler and PCR products were separated by 1.5% agarose
gel electrophoresis. Images were captured using a gel imaging
system (Bio-Rad, Hercules, CA, USA).
Short-interfering RNA (siRNA)
The human CD-147 siRNA and negative control siRNA
were purchased from Genepharma (Shanghai, China). The siRNA
sequence for CD-147 was: forward: 5′-GGU UCU UCG UGA GUU CCU CTT-3′
and reverse: 5′-GAG GAA CUC ACG AAG AAC CTG-3′. The negative
control siRNA sequence was: forward: 5′-UUC UCC GAA CGU GUC ACG
UTT-3′ and reverse: 5′-ACG UGA CAC GUU CGG AGA ATT-3′ for NC.
DharmaFect 4 transfection reagent was purchased from Dharmacon,
Inc. (Lafayette, Co, USA). AGS cells were seeded in 6-well plates
at a density of 2.5×105 cells/well in MEM and grown for
16 h. Transfection was performed according to the instructions
provided by Dharmacon using 4 μl of DharmaFect-4 and a 100 nM/well
final siRNA concentration. The cells were cultured for another 24
h. Total protein extracts were isolated and analyzed using
anti-CD-147 (Invitrogen, Carlsbad, CA, USA) and GAPDH (Kangcheng,
Shanghai, China) antibody immunoblotting. The cells were collected,
washed twice with pre-cold PBS, and invasion was measured.
Western blot analysis
Cells were lysed in SDS lysis buffer on ice for 30
min. Cell debris was removed by centrifugation at 14,000 × g at 4°C
for 5 min, and protein contents of the cell lysates were determined
using a Bio-Rad protein assay kit (Hercules, CA, USA). Cell lysates
with equal protein content were then loaded and separated by 12%
SDS-PAGE. The protein bands were electrotransferred onto PVDF
membrane and blocked with 5% non-fat milk in TBS/T buffer (20
mmol/l Tris base, 135 mmol/l NaCl, 0.1% Tween-20, pH 7.6) for 1 h.
After incubation of the membrane with the appropriate antibodies,
i.e., anti-phosphorylated or non-phosphorylated ERK, anti-CD-147,
MMP-2 and MMP-9 (all from Cell Signaling Technology, MA, USA), for
at least 4 h, specific protein bands were visualized using
SuperSignal West Pico/Fico chemiluminescent substrate (Millipore
Co., Bedford, MA, USA). As an internal control, the GAPDH contents
in the samples were also immunoblotted using polyclonal anti-actin
antibody as the primary antibody.
Invasion assay
Invasion of gastric cancer cells was measured using
the BD BioCoat™ BD Matrigel™ invasion chamber (BD Biosciences, San
Jose, USA) according to the manufacturer’s instructions. Briefly,
AGS and MKN-45 cells were treated with the inhibitor or siRNA for
24 h. The cells were seeded in the membrane of the upper chamber of
the Transwell at a concentration of 3–5×105 cells/ml in
2 ml of DMEM medium. The medium in the upper chamber was
serum-free. The medium in the lower chamber contained 5% fetal calf
serum as a source of chemoattractants. The cells that passed
through the Matrigel-coated membrane were counted.
Statistical analysis
Each sample was analyzed in triplicate, and
experiments were repeated three times. In all figures, error bars
are standard deviations. Statistical analyses were performed using
Microsoft Office Excel 2003 (Microsoft, Albuquerque, New Mexico,
USA) and Statisca ver. 10 (StatSoft, Tulsa, OK, USA). Differences
between mean values were evaluated by the unpaired t-test.
Differences were considered statistically significant at
P<0.05.
Results
Ara-C did not induce the inhibition of
proliferation in gastric cancer cells
AGS and MKN-45 gastric cancer cells were treated
with Ara-C (0, 1, 2, 4 μg) for 24 h (Fig. 1). The results indicated that Ara-C
did not inhibit the proliferation of gastric cancer cells (Fig. 1A and B). NB4 and Jurkat
Ara-C-sensitive cells were treated with Ara-C (0, 1, 2 and 4 μg)
for 24 h. The data showed that Ara-C inhibited the proliferation of
NB4 and Jurkat cells in a concentration gradient-dependent manner
(Fig. 1C and D).
Ara-C induces potentialization of
invasiveness and the activation of MMP-2 and -9 in gastric cancer
cells
After the treatment of AGS and MKN-45 cells with
Ara-C (0, 1, 2 and 4 μg) for 24 h, the invasion of these gastric
cancer cells was detected. The data showed that Ara-C (0, 1, 2 and
4 μg) did not suppress AGS and MKN-45 cell invasion, but increased
invasion depending on the Ara-C dose (Fig. 2A and C). The activity of MMP-2 and
-9 was analyzed by gelatinase zymography experiments. The results
indicated that the activity of MMP-2 and -9 was enhanced by Ara-C
concentration in a gradient-dependent manner in the supernatant of
AGS and MKN-45 cells (Fig. 2B and
D).
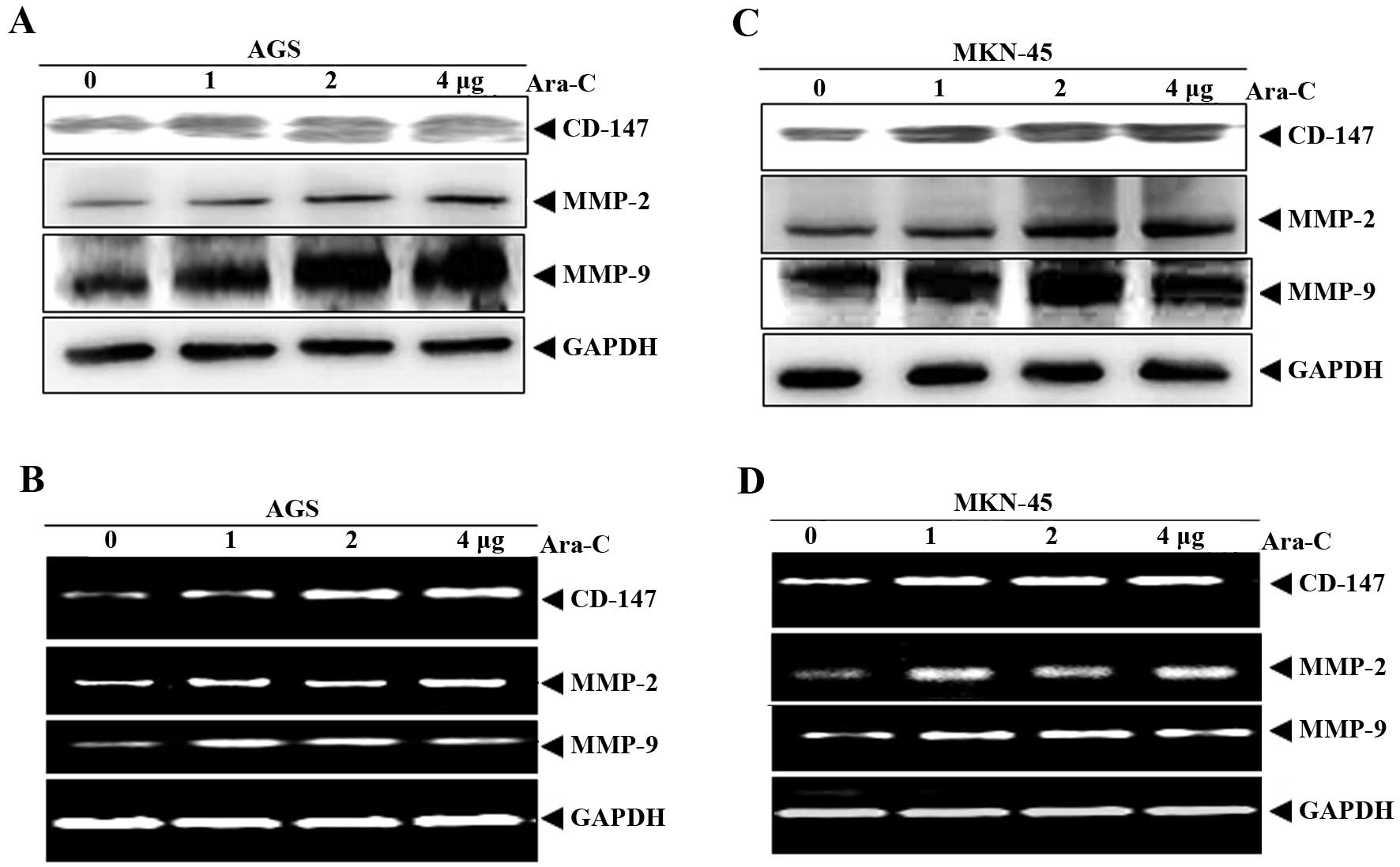
Ara-C induces the expression of MMP-2 and
-9, and CD147 in AGS and MKN-45 cells
Ara-C-induced MMP-2 and -9 activity allows us to
determine whether CD-147 is involved in this process, as the
literature supports that CD-147 is involved in the regulation of
MMP-2 and -9 in tumor cell invasion (11–13).
CD-147 expression was evaluated after AGS and MKN-45 cells were
treated with Ara-C (0, 1, 2 and 4 μg) for 24 h. The results showed
that Ara-C significantly upregulated CD-147 mRNA and protein levels
(Fig. 3A–D). The expression of
MMP-2 and MMP-9 was also analyzed, and the results showed that
Ara-C significantly upregulated MMP-2 and -9 mRNA and protein
levels (Fig. 3A–D).
CD147 siRNAs inhibit Ara-C induced
invasion and restore its anti-proliferation activity in gastric
cancer cells
To determine whether Ara-C mediates the
CD-147-MMP-2/MMP-9 signaling pathway involved in gastric cancer
cell invasiveness, CD-147 siRNAs were transfected into AGS cells
for 24 h. The cells were treated with or without 4 μg Ara-C, and
western blotting revealed that Ara-C-inducing MMP-2/MMP-9
expression was inhibited by CD-147 siRNAs (Fig. 4A). Invasive in vitro
experiments showed that CD-147 siRNAs completely blocked off the
Ara-C increased gastric cancer cell invasiveness. At the same time,
CD-147 siRNAs also restored the growth inhibitory effect of Ara-C
in gastric cancer cells (Fig. 4B and
C).
Ara-C induces the expression of MMP-2 and
-9, and CD-147 via ERK activated in gastric cancer cells
The activation of ERK signaling pathway is closely
associated with activation of the CD-147-MMP-2/MMP-9 signaling
pathway (14,15). In order to determine the role of ERK
signaling molecules in Ara-C-induced CD-147 and its downstream
MMP-2 and -9, AGS cells were treated with 4 μg Ara-C at the
indicated time points. MMP-2 and -9, and CD-147 protein levels were
upregulated after activation of ERK signaling molecules (Fig. 5A). U-0126, the ERK signaling
molecule inhibitor (16–18), was used to pre-treat gastric cancer
cells was found to block Ara-C-induced ERK-CD-147 -MMP-2/MMP-9
activation (Fig. 5B). Accordingly,
U-0126 allowed Ara-C to inhibit gastric cancer cell proliferation
activity (Fig. 5C). In invasive
in vitro experiments it was observed that 4 μg Ara-C
increased the invasiveness of gastric cancer cells, while the ERK
inhibitor U-0126 reduced gastric cancer cell invasion. More
importantly, U-0126 completely blocked off the Ara-C increased
gastric cancer cell invasiveness (Fig.
5D).
Discussion
In this study, we used Ara-C treated gastric cancer
cells to determine whether Ara-C inhibited cell proliferation and
induced apoptosis in gastric cancer cells. The results show that
Ara-C did not inhibit gastric cancer cells. By contrast, Ara-C
promoted gastric cancer cell invasiveness. Furthermore, we revealed
that the activation of ERK involved in Ara-C upregulated
CD-147-MMP-2/MMP-9 expression which could enhance the invasive
properties of gastric cancer cells.
CD-147 is a highly glycosylated transmembrane
protein belonging to the immunoglobulin superfamily, which can
induce the production of its downstream MMP-2 and -9 when activated
by certain molecules. These molecules are involved in cell
migration and invasiveness (19–21).
Notably, Ara-C can increase the expression of CD-147 in gastric
cancer cells. We also observed that Ara-C activated MMP-2 and MMP-9
expressions. SiRNA-CD-147 resisted Ara-C-induced invasiveness
increase in gastric cancer cells. Following this, Ara-C-induced
MMP-2 and -9 was restored by siRNA-CD-147. These findings suggest
that Ara-C enhanced gastric cancer cell invasiveness by
CD-147-MMP-2/MMP-9 signaling molecules.
As the abnormal activation of ERK signaling pathway
may trigger tumor cell CD-147 signaling pathway (22,23),
whether Ara-C can induce CD-147 signaling molecules by ERK in
gastric cancer cells remains to be elucidated. Treatment of gastric
cancer cells using Ara-C at different concentrations for 24 h
showed that MMP-2 and -9, and CD-147 were upregulated in an
Ara-C-dependent manner when the activation of ERK was observed. It
was hypothesized that Ara-C upregulated CD-147-MMP-2/MMP-9
expression through the ERK activation of Ara-C. This observation
was confirmed by U-0126, the ERK inhibitor, which blocked the ERK
activation of Ara-C, following restoration of MMP-2 and -9, and
CD-147 upregulation in Ara-C-treated gastric cancer cells. U-0126
inhibited gastric cancer cell invasion of Ara-C induction,
indicating that the Ara-C induction increased gastric cancer cell
invasion by activation of ERK. The manner in which Ara-C activated
ERK, as well as the activation of CD-147-MMP-2/MMP-9 require
further investigation.
In summary, the results suggest that Ara-C did not
inhibit gastric cancer cell proliferation and viability. By
contrast, Ara-C activated the ERK signaling pathway in gastric
cancer cells, which is involved in gastric cancer cell
proliferation resistance and activated the invasiveness of gastric
cancer cells via CD-147-MMP-2/MMP-9 signaling molecules. These
results show that Ara-C alone may not be used for the treatment of
patients with gastric cancer, and application of Ara-C treatment
for gastric cancer patients points may involve potential risks.
Acknowledgements
This study was supported in part by grants from
National Natural Science Foundation of China (81172322, 81101642),
Science and Technology Commission of Shanghai Municipality
(11ZR1421000), Science and Technology Fund of Shanghai Jiao Tong
University School of Medicine (YZ1027).
References
|
1
|
Li R, Chen WC, Pang XQ, et al: Combined
effect of sCD40L and PI3K siRNA on transplanted tumours growth and
microenvironment in nude mice with gastric cancer. Mol Biol Rep.
39:8755–8761. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Pietrantonio F, De Braud F, Da Prat V, et
al: A review on biomarkers for prediction of treatment outcome in
gastric cancer. Anticancer Res. 33:1257–1266. 2013.PubMed/NCBI
|
|
3
|
Braun S, Kentenich C, Janni W, et al: Lack
of effect of adjuvant chemotherapy on the elimination of single
dormant tumor cells in bone marrow of high-risk breast cancer
patients. J Clin Oncol. 18:80–86. 2000.PubMed/NCBI
|
|
4
|
Aogi K, Rai Y, Ito Y, et al: Efficacy and
safety of ixabepilone in taxane-resistant patients with metastatic
breast cancer previously treated with anthracyclines: results of a
phase II study in Japan. Cancer Chemother Pharmacol. 71:1427–1433.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Harland S, Staffurth J, Molina A, et al:
Effect of abiraterone acetate treatment on the quality of life of
patients with metastatic castration-resistant prostate cancer after
failure of docetaxel chemotherapy. Eur J Cancer. 49:3648–3657.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Sawai K, Goi T, Koneri K, et al: Partial
response after transcatheter arterial infusion chemotherapy in a
patient with systemic chemotherapy-resistant unresectable colon
cancer and hepatic metastasis: (case report). World J Surg Oncol.
11:2032013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Oyamada Y, Maeshima A, Takeuchi K and Kato
R: A case of lung cancer resistant to pemetrexed responding to S-1
monotherapy. Gan To Kagaku Ryoho. 40:781–784. 2013.(In Japanese).
PubMed/NCBI
|
|
8
|
Neuzillet C, Hentic O, Rousseau B, et al:
FOLFIRI regimen in metastatic pancreatic adenocarcinoma resistant
to gemcitabine and platinum-salts. World J Gastroenterol.
18:4533–4541. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Galmarini CM, Mackey JR and Dumontet C:
Nucleoside analogues and nucleobases in cancer treatment. Lancet
Oncol. 3:415–424. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Löwenberg B: Sense and nonsense of
high-dose cytarabine for acute myeloid leukemia. Blood. 121:26–28.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Fan X, Wu W, Shi H and Han J: RNA
interference targeting CD147 inhibits the invasion of human
cervical squamous carcinoma cells by downregulating MMP-9. Cell
Biol Int. 37:737–741. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhao S, Ma W, Zhang M, et al: High
expression of CD147 and MMP-9 is correlated with poor prognosis of
triple-negative breast cancer (TNBC) patients. Med Oncol.
30:3352013. View Article : Google Scholar
|
|
13
|
Piao S, Zhao S, Guo F, et al: Increased
expression of CD147 and MMP-9 is correlated with poor prognosis of
salivary duct carcinoma. J Cancer Res Clin Oncol. 138:627–635.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kim JY, Kim WJ, Kim H, et al: The
stimulation of CD147 induces MMP-9 expression through ERK and
NF-kappaB in macrophages: implication for atherosclerosis. Immune
Netw. 9:90–97. 2009. View Article : Google Scholar
|
|
15
|
Farabegoli F, Papi A and Orlandi M:
(−)-Epigallocatechin-3-gallate down-regulates EGFR, MMP-2, MMP-9
and EMMPRIN and inhibits the invasion of MCF-7 tamoxifen-resistant
cells. Biosci Rep. 31:99–108. 2011. View Article : Google Scholar
|
|
16
|
Chow S, Patel H and Hedley DW: Measurement
of MAP kinase activation by flow cytometry using phospho-specific
antibodies to MEK and ERK: potential for pharmacodynamic monitoring
of signal transduction inhibitors. Cytometry. 46:72–78. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Fukazawa H, Noguchi K, Murakami Y and
Uehara Y: Mitogen-activated protein/extracellular signal-regulated
kinase kinase (MEK) inhibitors restore anoikis sensitivity in human
breast cancer cell lines with a constitutively activated
extracellular-regulated kinase (ERK) pathway. Mol Cancer Ther.
1:303–309. 2002.PubMed/NCBI
|
|
18
|
Sunayama J, Matsuda K, Sato A, et al:
Crosstalk between the PI3K/mTOR and MEK/ERK pathways involved in
the maintenance of self-renewal and tumorigenicity of glioblastoma
stem-like cells. Stem Cells. 28:1930–1939. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hao JL, Cozzi PJ, Khatri A, et al:
CD147/EMMPRIN and CD44 are potential therapeutic targets for
metastatic prostate cancer. Curr Cancer Drug Targets. 10:287–306.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kanekura T and Chen X: CD147/basigin
promotes progression of malignant melanoma and other cancers. J
Dermatol Sci. 57:149–154. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Nabeshima K, Iwasaki H, Koga K, et al:
Emmprin (basigin/ CD147): matrix metalloproteinase modulator and
multifunctional cell recognition molecule that plays a critical
role in cancer progression. Pathol Int. 56:359–367. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Boulos S, Meloni BP, Arthur PG, et al:
Evidence that intracellular cyclophilin A and cyclophilin A/CD147
receptor-mediated ERK1/2 signalling can protect neurons against in
vitro oxidative and ischemic injury. Neurobiol Dis. 25:54–64. 2007.
View Article : Google Scholar
|
|
23
|
Kim K, Kim H, Jeong K, et al: Release of
overexpressed CypB activates ERK signaling through CD147 binding
for hepatoma cell resistance to oxidative stress. Apoptosis.
17:784–796. 2012. View Article : Google Scholar : PubMed/NCBI
|