Introduction
Ovarian cancer is highly aggressive and the most
malignant gynecologic neoplasm in women (1). Ovarian epithelial cancer accounts for
90% of all ovarian cancers and is the leading cause of death
(2). Despite the initial response
to surgery and chemotherapy as carboplatin and paclitaxel, most
tumors ultimately become drug resistant, which causes relapse for
the patients (3,4). Eventually the majority of the patients
die from their disease, highlighting the need for the development
of novel treatment to improve upon the current therapeutic
options.
It is generally accepted that the activation of
signaling pathways PI3K/Akt (5),
Wnt/β-catenin (6,7), Notch (8,9) and
Hedgehog (10) contributes to the
pathogenesis of ovarian carcinomas. Among them, Notch signaling
pathway has received special attention due to its crucial role in
controlling cell differentiation, proliferation and apoptosis
(11). In normal tissue, Notch
signaling pathway is under strict temporal and spatial control
during development (11). In
contrast, Notch has been found to be aberrantly expressed in
several malignancies (12), such as
T-ALL (13), lung cancer (14), breast cancer (15) and ovarian cancer (16). However, the biological importance of
Notch in carcinoma goes well beyond that. Notch3 overexpression may
be related to chemoresistance, and inactivation of Notch3 by
γ-secretase inhibitor might abrogate drug resistance (17). Therefore, therapeutic new drugs
targeting Notch signaling pathway might be a promising approach for
the treatment of ovarian cancer.
Mangiferin, a C-glucosylxanthone
(1,3,6,7-tetrahydroxyxanthone-C2-β-D-glucoside), is isolated
from the leaves, stem bark, fruit peels and root of Mangifera
indica L. (18). Previous
studies revealed the antioxidant (19), antitumor (20), anti-bacteria, antiviral and
immunomodulatory activities (21)
of mangiferin. Furthermore, studies on cancer therapy showed
anti-neoplastic effects of mangiferin on lung cancer (22,23),
colon cancer (23,24), leukemia (22,25–28),
lung cancer (22,28) and prostate cancer (22). Studies on the mechanism of
mangiferin-induced apoptosis of breast cancer suggested that the
downregulation of MMP-7 and −9 might contribute to the anti-tumor
effect of mangiferin. Besides the activation of β-catenin pathway
in breast cancer cells was also inhibited by mangiferin (29). Our previous results demonstrated
that mangiferin possessed anti-neoplastic effect by inducing
caspase-dependent apoptosis and regulating signaling pathways of
NF-κB and cyclin B1 (30) in human
lung carcinomas. However, up to now, no data on the anti-neoplastic
effect of mangiferin on ovarian cancer have been reported. Herein,
we show that mangiferin treatment could induce apoptosis via
blocking or silencing Notch3, which is the crux of multiple cancer
survival and proliferation pathways. Therefore, our current study
on mangiferin offers a potential new strategy for the therapeutic
intervention in ovarian cancer.
Materials and methods
Reagents
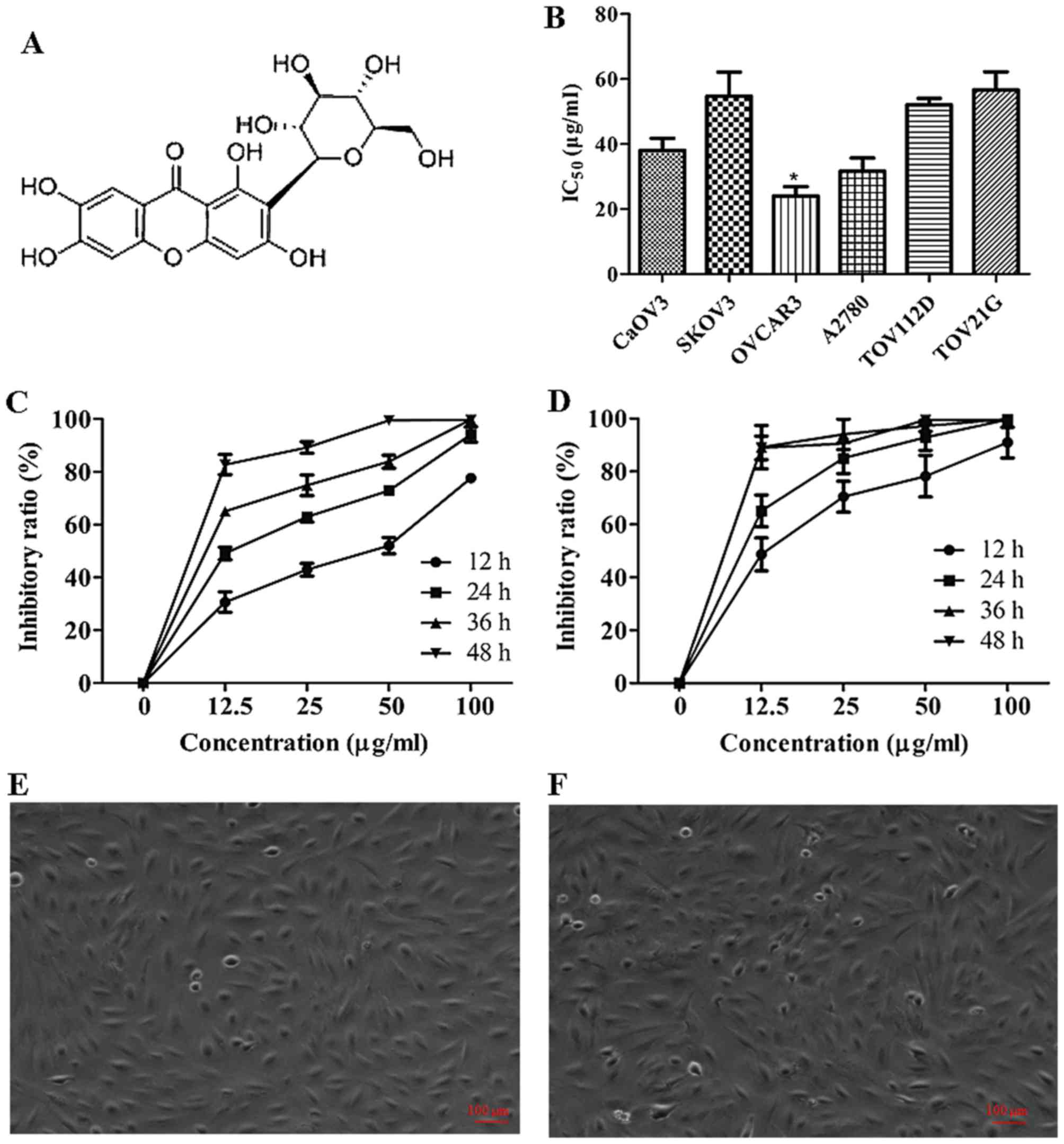
Mangiferin was purchased from Shanghai PureOne
Technology (Shanghai, China). The purity of mangiferin was >95%,
and the chemical structure is shown on Fig. 1A. Human Embryonic Kidney 293T cells,
human ovarian adenocarcinoma OVCAR3 cells, CaOV3 cells, SKOV3
cells, endometrioid carcinoma TOV-112D cells, and clear cell
carcinoma TOV-21G were purchased from American Type Culture
Collection (ATCC, Manassas, VA, USA). Human ovarian adenocarcinoma
A2780 cells were purchased from Procell Co. (Wuhan, China).
RPMI-1640 medium and fetal bovine serum (FBS) was purchased from
Gibco BRL (Grand Island, NY, USA). 3-(4,5-Dimetrylthiazol-2-yl)-2,
5-diphenyltetrazolium bromide (MTT, cat. no. M5655), dimethyl
sulfoxide (DMSO, cat. no. D2650), paraformaldehyde (PFA, cat. no.
16005), cisplatin (USP grade, cat. no. 1134357), Annexin V-FITC
Apoptosis detection kit (cat. no. APOAF), HEPES (cat. no. H3375),
Triton X-100 (cat. no. H9284), 2 mmol/l sodium orthovanadate (cat.
no. S6508), sodium fluoride (cat. no. S7920), 1 mmol/l edetic acid
(cat. no. E9884), PMSF (cat. no. 78830), aprotinin (cat. no.
A11530), leupeptin (cat. no. L2884) and rhodamine-123 were
purchased from Sigma-Aldrich Chemical (St. Louis, MO, USA). Hoechst
33342 (cat. no. C1025) was purchased from Beyotime Chemicals
(Suzhou, China). Rabbit polyclonal pro-caspase-3 (cat. no. sc-7148;
1:1,000 dilution), mouse monoclonal pro-caspase-9 (cat. no.
sc-56073; 1:1,000 dilution), rabbit polyclonal Bax (cat. no.
sc-493; 1:500 dilution), mouse monoclonal Bid (cat. no. sc- 135847;
1:1,000 dilution), rabbit polyclonal Bcl-2 (cat. no. sc-492;
1:1,000 dilution), mouse monoclonal Bcl-XL (cat. no.
sc-8392; 1:1,000 dilution), rabbit polyclonal PARP (cat. no.
sc-7150; 1:1,000 dilution), rabbit polyclonal cytochrome c (cat.
no. sc-7159; 1:1,000 dilution), mouse monoclonal cyclin D1 (cat.
no. sc-450; 1:1,000 dilution), mouse monoclonal β-catenin (cat. no.
sc-59737; 1:1,000 dilution), rabbit polyclonal Notch3 (NICD domain,
cat. no. sc-5593; 1:1,000 dilution), mouse monoclonal Notch1 (cat.
no. sc-376403; 1:1,000 dilution) and mouse monoclonal β-actin (cat.
no. sc-47778; 1:5,000 dilution) purchased from Santa Cruz
Biotechnology, Inc. (Santa Cruz, CA, USA). Mouse monoclonal PI3K
p85 (cat. no. ab189403; 1:500 dilution), rabbit polyclonal
phosphor-PI3K p85 (cat. no. ab182651; 1:500 dilution), rabbit
polyclonal mTOR (cat. no. ab2732; 1:500 dilution) and rabbit
polyclonal phospho-mTOR (S2448, cat. no. ab109268; 1:500 dilution)
antibodies were from Abcam (Cambridge, MA, USA). Mouse monoclonal
Akt (pan, cat. no. 2920; 1:500 dilution), rabbit polyclonal
phospho-Akt (S473, cat. no. 4060; 1:500 dilution), rabbit
polyclonal cleaved caspase-3 (cat. no. 9661; 1:500 dilution) and
rabbit polyclonal cleaved caspase-9 (cat. no. 9509; 1:1,000
dilution), were purchased from Cell Signaling Technologies
(Danvers, MA, USA). Horseradish peroxidase (HRP)-conjugated goat
anti-mouse IgG polyclonal antibody (cat. no. 115-035-003) and
HRP-conjugated goat anti-rabbit polyclonal IgG (cat. no.
111-035-003) were purchased from Jackson ImmunoResearch
Laboratories.
Cell culture
Human Embryonic Kidney 293T (HEK-293T) cells,
ovarian cancer cell lines CaOV3 and SKOV3 were cultured in DMEM
containing 10% FBS, 100 µg/ml streptomycin (Life Technologies,
Rockville, MD, USA), 100 U/ml penicillin (Life Technologies), and
0.03% L-glutamine (Sigma-Aldrich, St. Louis, MO, USA). TOV-112D and
TOV-21G cells were cultured in RPMI-1640 containing 15% FBS, 100
µg/ml streptomycin, 100 U/ml penicillin and 0.03% L-glutamine.
OVCAR3 cells were cultured in RPMI-1640 containing 20% FBS, 10
µg/ml bovine insulin, 100 µg/ml streptomycin, 100 U/ml penicillin
and 0.03% L-glutamine. CHO cells were cultured in F-12K containing
10% FBS, 100 µg/ml streptomycin, 100 U/ml penicillin and 0.03%
L-glutamine.
MTT colormetric assay
OVCAR3 cells at logarithmic growth phase were seeded
in a 96-well plate and incubated at 37°C for 24 h, and different
dosage of mangiferin (12.5, 25, 50 and 100 µg/ml) were added and
incubated for 12, 24, 36 and 48 h, respectively. MTT [0.05 mg (10
µl of 5 mg/ml)] was added to each well and incubated at 37°C for 4
h, and then medium was removed and shaked thoroughly for 1 h.
Finally, termination buffer was added into each well. The
absorbance at 570 nm was measured with a spectrophoto-meter (Model
3550 Microplate Reader, Bio-Rad Laboratories, Hercules, CA, USA).
Cell viability (%) = [OD 570 nm (drug)/OD 570 nm (control)]
×100%.
Cell morphology observation
OVCAR3 cells were seeded into a 6-well culture plate
at a density of 4×105 cells/well and cultured for 24 h.
Control groups were treated with 0.05% DMSO, and mangiferin groups
were treated with 25 µg/ml mangiferin. Hoechst 33342 staining was
applied to further detect viable cells. In brief, cells were fixed
with 4% PFA for 30 min at room temperature after 48 h incubation
with either mangiferin or 0.05% DMSO, and then cells were washed
twice with PBS. Hoechst 33342 (5 µg/ml) was added and stained for
15 min, and then the cells were washed and analyzed immediately
with a fluorescence microscope (Olympus, Tokyo, Japan). All
experiments were carried out in triplicate. Experiments were
repeated three times, and data are representative of replicate
experiments.
Annexin V-FITC/PI flow cytometry
To study the effects of mangiferin on cell
apoptosis, OVCAR3 cells were treated with 25 µg/ml mangiferin for
24 h. Detached and adherent cells were then collected and labeled
for 15 min at room temperature with Annexin V-FITC (1 µg/ml) and
prodidium iodide (PI, 40 µg/ml) after washing away unbound
fluorescent dye, cells were immediately analyzed on a FACS Aria II
cytometer (Becton Dickinson, Franklin Lakes, NJ, USA). Cells
(1×105) were measured for each sample. All experiments
were carried out in triplicate. Experiments were repeated three
times, and data are representative of replicate experiments.
Caspase-3 activity and mitochondrial
membrane potential assay
Mangiferin-induced apoptosis of OVCAR3 cells was
also assessed by measuring the activity of caspase-3. OVCAR3 cells
were seeded into a 6-well culture plate at a density of
4×105 cells/well for 16 h incubation. Subsequently,
cells were treated with 25 µg/ml mangiferin for 0, 12, 24, 36, 48
h. Caspase-3 activity was measured by using a caspase-3 activity
kit (Beyotime Institute of Biotechnology, Haimen, China). In brief,
cells were lysed, the supernatant was collected, quantified, and
incubated with the caspase-3-specific color substrate Ac-DEVD-pNA.
Caspase-3 activity was determined by measuring optical density at
OD400 nm.
Mitochondrial membrane potential was measured by
fluorescent dye rhodamine-123. After treatment with 25 µg/ml
mangiferin for 12, 24, 36 and 48 h, cells were collected and
suspended in 1 ml of PBS containing 1 µg/ml rhodamine-123 and
incubated at 37°C for 15 min. The fluorescence intensity of the
cells was analyzed on a FACS Aria II cytometer (Becton Dickinson).
All experiments were carried out in triplicate. Experiments were
repeated three times, and data are representative of replicate
experiments.
Western blot analysis
OVCAR3 cells were treated with 0, 12.5, 25, 50 and
100 µg/ml mangiferin for 24 h, respectively, and then both adherent
and floating cells were collected. The cell pellets were
resuspended with lysis buffer and lysed at 4°C for 15 min, and the
lysis buffer consisted of 50 mmol/l HEPES (pH 7.4), 1% Triton
X-100, 2 mmol/l sodium orthovanada, 100 mmol/l sodium fluoride, 1
mmol/l edetic acid, 1 mmol/l PMSF, 10 mg/l aprotinin and 10 mg/l
leupeptin. After 12,000 × g centrifugation for 15 min, the protein
content of supernatant was determined by Bradford protein assay
(cat. no. P0006, Beyotime, Suzhou, China). Equal amounts of the
total protein were separated by 4–12% NuPAGE Bis-Tris gels (cat.
no. NP0327BOX, Life Technologies, Carlsbad, CA, USA) and
transferred to PVDF membranes (cat. no. ISEQ00010, Millipore,
Bedford, MA, USA), the membranes were soaked in blocking buffer (5%
BSA, cat. no. V900933, Sigma-Aldrich Chemicals). Then proteins were
detected using primary and secondary antibodies and visualized with
ECL (cat. no. 345818, Millipore). Experiments were repeated three
times, and data are representative of replicate experiments.
Lentivirus preparation and
transduction
The PCR product of the Notch3 intracellular domain
(NICD) was cloned into pTY linkers. Third-generation vectors were
used in this experiment. NICD lentiviral vector were transient
transfected into HEK-293T cells. Briefly, HEK-293T cells were
co-transfected with appropriate amounts of vector plasmids,
including helper construct, envelope plasmid, tat plasmid and pTY
linker containing NICD. The viruses were collected, concentrated,
and 1×106 lentiviral particles were transduced to OVCAR3
cells in the presence of 5 µg/ml Polybrene (cat. no. sc-134220,
Santa Cruz Biotechnology).
Acute toxicity testing
Acute toxicity testing was performed to determine
median lethal dosing (LD50) of mangiferin. After 16 h
fasting, BALB/c mice were randomly divided into 6 groups (n=10).
Graded doses of mangiferin (10, 20, 50, 100, 200, and 500 mg/kg)
were separately administered intraperitoneally to the mice. All
mice were allowed free access to food and water, and then mortality
in each group was assessed after 24, 48 and 72 h administration of
mangiferin. Percentage mortality in each group was calculated and
plotted against log10 of mangiferin dose. A regression
line was fitted by the method of least squares, and LD50
values were calculated. Animal handling was in accordance with the
ethics committee of the Sichuan Academy of Medical Science and
Sichuan Provincial People's Hospital, and all animals were kept in
12 h light/dark cycle with free access to water and food, which is
in consistent with IVC requirement in Sichuan Academy of Medical
Science and Sichuan Provincial People's Hospital.
Tumor xenograft study
The in vivo efficacy of mangiferin was
studied in xenograft models. In brief, to initiate tumor
xenografts, 5×106 OVCAR3 cells were injected into BALB/c
nude female mice. After implantation, tumors were allowed to grow
to a size of 100–550 mm3 before starting daily
treatment. Mice were randomly divided into five groups: 1) Blank
control group: mice intraperitoneally administered PBS after tumor
formation by OVCAR3 cells; 2) High dose mangiferin group: mice
intraperitoneally administered 100 mg/kg mangiferin after tumor
formation by OVCAR3 cells; 3) Medium dose mangiferin group: mice
intraperitoneally administered 50 mg/kg mangiferin after tumor
formation by OVCAR3 cells; 4) Low dose mangiferin group: mice
intraperitoneally administered 10 mg/kg mangiferin after tumor
formation by OVCAR3 cells; 5) Combined therapy group: mice
intraperitoneally administered both cisplatin (10 mg/kg) and
mangiferin (50 mg/kg) after tumor formation by OVCAR3 cells.
The administration of mangiferin lasted for another
two weeks. Before and after the mangiferin treatment, the body
weight of the mice was measured daily then, 5 mice in each group
were sacrificed, and 5 mice in each group were raised for survival
assay. For the sacrificed mice, the subcutaneous tumors were peeled
off and weighed. The volume of tumors was determined in three
dimensions with vernier calipers according to the formula: Tvol =
length × width × depth ×0.5, and then relative tumor volume was
calculated as relative increase or decrease in mean tumor volume
from initiation of treatment (V0) up to value at a given time (Vt)
and RTV = Vt/V0. Inhibitory rate of tumor volume = (Vcontrol ×
Vt)/Vcontrol ×100%. After 14 days treatment, mice were sacrificed
by cervical dislocation, and subcutaneous tumor masses were
determined. Inhibitory level of tumor weight = Wcontrol -
Wt)/Wcontrol ×100%.
Statistical analysis of the data
All data were expressed as mean values ± SEM from at
least three independent experiments. Statistical significance was
determined by two-way ANOVA and Student's t-test. Experiments were
repeated three times, and data are representative of replicate
experiments.
Results
Mangiferin inhibits viability of
OVCAR3 cells
Based on our previous study on mangiferin (30), we hypothesized that mangiferin might
inhibit the viability of ovarian cancer cells. Therefore, to
substantiate our hypothesis and to explore the inhibitory role of
mangiferin in ovarian cancer cells, we performed MTT assay on
ovarian adenocarcinoma cell lines (CaOV3, SKOV3, OVCAR3 and A2780),
endometrioid carcinoma cell line TOV-112D and clear cell carcinoma
cell line TOV-21G (Fig. 1B). The
IC50 values were 38.14 µg/ml for CaOV3, 57.67 µg/ml for
SKOV3, 31.67 µg/ml for A2780, 24.13 µg/ml for OVCAR3, 53.65 µg/ml
for TOV-112D and 56.69 µg/ml for TOV-21G. These data strongly
suggested that mangiferin could inhibit the viability of ovarian
carcinoma cell lines, and OVCAR3 cells were most sensitive to
mangiferin treatment. To ascertain whether mangiferin treatment was
dose- and time-dependent, OVCAR3 cells were treated with different
dosage of mangiferin (0, 12.5, 25, 50,100 µg/ml) for 12, 24, 36,
and 48 h (Fig. 1C). As predicted,
accompanied with increased dosage and extended treatment, the
inhibitory ratio increased, suggesting that mangiferin inhibited
the proliferation of OVCAR3 cells in a dose- and time-dependent
manner. In addition, it has been widely acknowledged that the
standard first-line treatment for advanced stage ovarian carcinoma
is the combined chemotherapy of platinum based drugs as cisplatin
coupled with paclitaxel (31).
However, most patients will relapse due to drug resistance.
Therefore to validate whether mangiferin treatment could enhance
the sensitivity of OVCAR3 cells to cisplatin, we evaluated the
inhibitory rates of combined therapy of both mangiferin and
cisplatin. Simultaneously with 1 µg/ml cisplatin, different dosages
of mangiferin (0, 12.5, 25, 50 and 100 µg/ml) were added into the
culture medium of OVCAR3 cells (Fig.
1D). As expected, increased inhibitory ratios were observed,
suggesting that mangiferin treatment could improve the sensitivity
of ovarian carcinoma cells to cisplatin in dose- and time-dependent
manner. However, 100 µg/ml mangiferin did not result in apparent
apoptotic morphology in non-cancerous Chinese Hamster Ovary (CHO)
cells after 24 or 48 h treatment (Fig.
1E and F). These results suggested that mangiferin might
selectively induce OVCAR3 cells apoptosis but could not trigger
apoptosis of normal ovary cells.
Mangiferin induces apoptosis
To address whether mangiferin could induce
apoptosis, we observed the cell morphology under phase contrast
microscopy (Fig. 2A). It was
obvious that cells treated with 25 µg/ml mangiferin for 48 h showed
shrinkage of cytoplasm and nucleus, membrane blebbing, and
shattering, which are typical in apoptotic morphology. Furthermore,
less viable cells were observed in the cisplatin plus mangiferin
treated group. To substantiate our observations, we preformed
Hoechst 33342 staining, and found that the remaining cells in
mangiferin treated group showed condensed nuclei, indicating those
cells were underging apoptosis (Fig.
2B). In order to determine the apoptotic percentages of each
group, cells were treated with 25 µg/ml mangiferin for 24 h, and
then sorted by flow cytometry. As shown in Fig. 2C, there were more early apoptotic
(Annexin V+/PI−) cells and late apoptotic
cells (Annexin V+/PI+) in mangiferin treated
group compared to those of the controls. Statistics on the early
apoptotic cells, late apoptotic cells and viable cells are in
accordance further demonstrating that mangiferin induced apoptosis
of OVCAR3 cells and increased the sensitivity of OVCAR3 cells to
cisplatin treatment (Fig. 2D).
Taken together, based on the qualitative and quantitative analysis
of the apoptotic events, mangiferin was able to induce apoptosis in
ovarian carcinoma OVCAR3 cells.
Mangiferin induces apoptosis via
caspase pathway
To unravel the mechanism of mangiferin-induced
apoptosis, and to determine whether it relied on the caspase
pathway, a series of western blotting were performed. Accumulating
evidence revealed that during apoptosis, cytochrome c is released
from mitochondria to cytosoplasm (32). We, therefore, assessed the cytosol
and mitochondria cytochrome c, and found that mangiferin treatment
could promote the translocation of cytochrome c from mitochondria.
Furthermore, activation of caspase plays a central role in
apoptosis, and caspase-3 serves as the convergence point of
different apoptotic signaling pathway. As shown in Fig. 3A, caspase-3 and −9 were activated
upon mangiferin treatment with increased cleaved caspase-3 and −9.
As Bid induces conformational change of Bax, and Bax induces the
release of cytochrome c from mitochondria during apoptosis, we
found mangiferin treated cells had higher expression level of Bax
and Bid compared with that of the control, suggesting more
apoptotic events mediated by mangiferin. However, as the protein
Bcl-2 interferes with the activation of caspases by preventing the
release of cytochrome c, cells treated with mangiferin showed a
comparatively lower expression level of Bcl-2. Moreover, decreased
Bcl-XL and increased PARP cleavage were found in
mangiferin treated cells. To further validate that caspase pathway
was involved in mangiferin-induced apoptosis, we analyzed the
caspase-3 activity and determined the integrity of mitochondria
membrane. As expected, caspase activity increased in accordance
with the dosage of mangiferin (Fig.
3B). Whereas, mitochondria membrane integrity was decreased
with the increased dosage, suggesting that mangiferin stimulated
the release of cytochrome c (Fig.
3C).
Mangiferin induces apoptosis through
the inhibition of Notch3
As discussed above, because Notch signaling pathway
plays a crucial role in the pathogenesis and chemoresistance of
ovarian cancer, we evaluated whether mangiferin could inhibit the
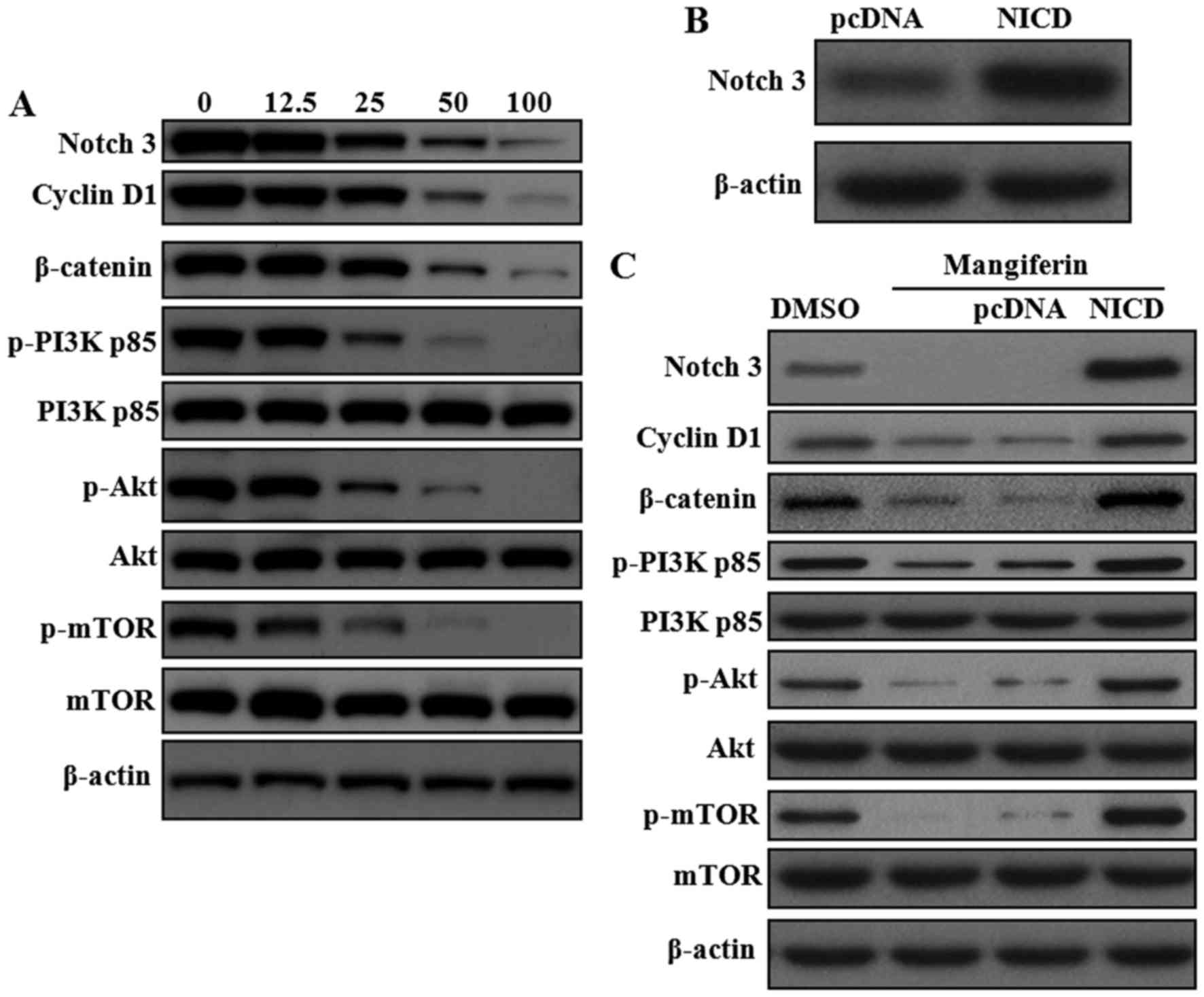
expression of Notch1 and Notch3. As shown in Fig. 4A, mangiferin could reduce protein
levels of Notch3, but has no effect on Notch1 (data not shown).
Besides, as amplification and overexpression of cyclin D1 have been
found in several human malignancies (33), the expression levels of cyclin D1
and β-catenin were determined and were also found to be inhibited
by mangiferin. Previous report clearly showed crosstalk between
Notch and PI3K/Akt (34),
therefore, we examined whether mangiferin has any inhibitory effect
on PI3K/Akt. As expected, decreased phosphorylation of PI3K p85
(Y607), Akt (S473) and mTOR (S2448) were found in mangiferin
treated cells. To further substantiate the inhibitory role of
mangiferin on Notch signaling pathway, Notch3 intracellular domain
(NICD) were overexpressed in OVCAR3 cells by lentivirus infection
(Fig. 4B). Then we found that
mangiferin-induced inhibition of cyclin D1, β-catenin and
PI3K/Akt/mTOR could be partially abrogated by Notch3
overexpression, highlighting the critical role of Notch3 in the
anti-neoplastic effect of mangiferin (Fig. 4C).
Further studies on cell morphology revealed that
Notch3 overexpression played a protective role from apoptosis for
cells treated with mangiferin. Cell treated with mangiferin showed
shrinking and bubbling compared with cells treated with both
mangiferin and Notch3 (NICD) overexpressed lentivirus (Fig. 5A). Besides, fluorescent staining
revealed that cells showed more condensed nuclei after mangiferin
treatment. In contrast, more viable cells were found in mangiferin
treated Notch3 overexpressed cells (Fig. 5B). Quantification of apoptotic cells
by flow cytometry clearly showed that overexpression of Notch3
rescued the cells from apoptosis (Fig.
5C and D).
Mangiferin inhibits tumor growth in
vivo
Based on our in vitro results, we decided to
evaluate the anti-neoplastic properties of mangiferin in an in
vivo tumor xenograft model system. Therefore, OVCAR3 cells were
xenografted into nude mice, and then examined the growth rates of
these tumors with or without mangiferin treatment. On the basis of
caliper measurements (Fig. 6A),
after 14 days of treatment, with the increase dose of mangiferin,
the xenograft tumor volume decreased significantly compared to that
of the blank control group. Similarly, mangiferin plus cisplatin
treated mice showed reduced xenograft tumor volume, suggesting
mangiferin could increase the sensitivity of ovarian cells to
cisplatin. Moreover, tumor weights were also decreased after
mangiferin therapy (Fig. 6B). In
addition, calculation of the relative tumor volume demonstrated
that mangiferin treatment could greatly inhibit tumor growth
(Fig. 6C). Furthermore, judged from
the survival curve, mangiferin could remarkably prolong the life
span of OVCAR3 cell xenograft mice compared with blank control
group (Fig. 6D). Therefore,
accompanied with decreased tumor size, body weights of mice
receiving mangiferin treatment, showed gradual but remarkable
recovery after mangiferin treatment (Fig. 6E). In summary, our in vivo
data clearly indicated that mangiferin could inhibit tumor growth,
supporting the potential use of mangiferin as a chemotherapeutic
agent in ovarian cancer.
Discussion
Based on recently published statistics ovarian
cancer is the fifth leading cause of cancer death in women
(1). Partially due to the
nonspecific symptom, most patients are diagnosed at an advanced
stage, which ultimately lead to the high mortality of this disease.
Most patients are initially sensitive to surgery and platinum based
therapy, but the majority will relapse and become drug resistant.
Therefore, it is well established that the development of effective
therapeutic agents for ovarian cancer is of great importance. Given
our observation of mangiferin on human lung carcinoma, we
hypothesized that mangiferin could induce apoptosis in ovarian
cancer. In the current study, we found that mangiferin remarkably
inhibited the viability of ovarian carcinoma cells, and markedly
increased tumor sensitivity to cisplatin both in vitro and
in vivo. We further showed that caspase-dependent apoptosis
pathway was activated by mangiferin. Of note, we observed that
Notch signaling pathway was involved in mangiferin-induced
apoptosis, and crosstalk between Notch and Wnt, PI3K/Akt was also
regulated by mangiferin. In addition, cell morphology observation
and apoptosis assays by Annexin V and PI revealed that Notch3
(NICD) overexpression blocked the inhibitory effect of mangiferin.
Furthermore, data collected from tumor growth and survival curve in
animal models indicated a strong response of xenograft tumors to
mangiferin treatment.
Previous studies on mangiferin revealed the
anti-cancer effects of mangiferin by inhibiting tumor necrosis
factor (TNF)-induced activation of NF-κB (35). Besides, mangiferin treatment
enhanced the detoxification activity of enzymes, including
glutathione transferase, quinone reductase and uridin
5′-diphosphate-glucuronosyl transferase, and reduced DNA damage in
lung cancer-bearing animals (36).
In addition, mangiferin was reported to inhibit telomerase activity
and to induce apoptosis in K562 cells (8,9), and
Mangifera indica extract also initiated
G0/G1 phase cell cycle arrest (10). Our previous studies showed that
mangiferin induced apoptosis of human lung carcinoma A549 cells by
the activation of NF-κB and through caspase-dependent pathway.
Although, the primary finding from our present study was that
mangiferin was able to induce apoptosis of ovarian cancer cells via
caspase-dependent pathway, the mechanisms underlying the mangiferin
induced apoptosis need to be further explored.
Notch signaling pathway is a highly conserved and
strictly regulated playing a critical role in the maintenance of
tissue homeostasis by the regulation of proliferation,
differentiation and apoptosis in normal tissue. In contrast, recent
evidence revealed that the misregulated Notch was tightly
correlated with tumorigenesis (37). Therefore, beyond its basic
regulatory role of self-renewal and cell-fate determination,
aberrantly deregulated Notch is thought to be involved in a wide
variety of human malignancies and it is deemed as a therapeutic
target in cancer (38). Studies by
Rose et al showed that Notch1 was overexpressed in ovarian
cancer cell lines and human ovarian cancer clinical samples, and
depletion of Notch1 led to the growth inhibition (9). In contrast, blocking the activity of
Notch1 contributed to cell growth inhibition and apoptosis in
ovarian cancer cells A2780 (39).
Therefore, studies on the pytomedicine xanthohumol revealed that
xanthohumol induced the growth inhibition by downregulation of
Notch1 transcription and protein expression (40). Besides, epigenetic silencing of
microRNA-199b-5p was associated with acquired chemoresistance in
ovarian cancer via the activation of JAG1-Notch1 signaling,
suggesting that Notch1 signaling was correlated with drug
resistance (41). Unfortunately, we
did not detect significant decrease of Notch1 expression after
mangiferin treatment, indicating that mangiferin might selectively
inhibit Notch3, and may have no influence on Notch1.
Studies by Park et al showed that Notch3 gene
copy number is increased and is correlated with ovarian cancer
(8). Noteworthy, our data show that
mangiferin exerted its anti-neoplastic effect by the inhibition of
Notch signaling pathway in a dose-dependent manner. Furthermore,
Park and his group also found that aberrant expression of Notch3
was related to the recurrence and participated in development of
chemoresistance of OVCAR3 cells to carboplatin (42). Studies on the mechanism of
chemoresistance revealed that Notch3 might induce epithelial to
mesenchymal transition, which led to the carboplatin resistance in
ovarian cancer cells (43).
Therefore, for advanced stage chemoresistant ovarian cancer, Notch3
could be a potential therapeutic target (17). Herein, our studies strongly support
the notion that by targeting Notch3 activation, mangiferin
sensitized ovarian cancer cells to cisplatin, which further
substantiated that Notch3 is related with chemoresistance. However,
given that Notch3 (NICD) overexpression rescued cells from
mangiferin-induced apoptosis, the anti-neoplastic effects of
mangiferin in sensitizing ovarian tumors to cisplatin therapy was
Notch pathway specific. Moreover, McAuliffe et al found that
Notch3 was also activated in ovarian cancer stem cells, and
inhibition of Notch will increase the sensitivity of ovarian tumors
to platinum therapy (44). Besides,
Notch3 ligand Jagged-1 was also involved in the regulation of
ovarian tumor growth (45), and its
expression was regulated by Notch3 and Wnt/β-catenin (46), indicating crosstalk between Notch
and Wnt in ovarian cancer. We confirmed that the inhibition of
Notch3 by mangiferin interferes with the activation of
Wnt/β-catenin pathway. Not only crosstalk between Notch and
Wnt/β-catenin pathway was influenced by mangiferin, but also the
PI3K/Akt/mTOR pathway was regulated by mangiferin. Surprisingly,
exogenously overexpressed Notch3 abrogated the inhibitory effect of
mangiferin on the activation of PI3K/Akt/mTOR pathway. Besides its
apoptosis-inducing effect, these results confirmed and extended our
previous findings with the regulatory and chemosensitization
function of mangiferin.
To determine whether mangiferin treatment could
eradicate tumors, mice bearing established OVCAR3 tumors (100
mm3) were treated with mangiferin at 10, 50, and 100
mg/kg/day for consecutive 14 days. On the basis of caliper
measurements, although complete tumor regression did not occur in
all mice, the tumors treated with the efficacy dose (50 and 100
mg/kg/day) did not re-grow during a 100-day observation period
after the end of treatment. However, mice treated with only 10
mg/kg/day mangiferin showed a remarkable longer survival during the
initial one month. However, with the retreat of mangiferin, tumors
re-grew, which ultimately lead to mortality of the mice. Therefore,
as we expected, in the murine models, mangiferin substantially
inhibited tumor growth. In future studies, in order to collect more
pre-clinical data, we plan to explore the anti-angiogenesis and
anti-invasion effect of mangiferin on ovarian cancer cells.
Besides, the toxicity and safety of mangiferin should be carefully
investigated before its clinical application.
In summary, based on our previous and current
research, there is great hope that mangiferin, functioning as a
molecularly targeted therapy of Notch3, will offer a new and
substantially different drug to the treatment of human cancers. We
anticipate that with the gradually clarification of the molecular
mechanisms of mangiferin, our work will pave the way in the near
future for developing mangiferin as a new therapeutic drug by
targeting cell apoptosis pathways and enhancing the response to
cisplatin treatment for ovarian cancer therapy.
Acknowledgements
This study was supported by Sichuan Health and
Family Planning Commission Funding (16ZD0253), Sichuan National
Science Research Funding (2015JY0183), Funding from Sichuan Academy
of Medical Science and Sichuan Provincial People's Hospital, and
Sichuan Scientific Research Foundation of the Returned Overseas
Chinese Scholars for Y.W.
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2016. CA Cancer J Clin. 66:7–30. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Choi JH, Wong AS, Huang HF and Leung PC:
Gonadotropins and ovarian cancer. Endocr Rev. 28:440–461. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ozols RF: Systemic therapy for ovarian
cancer: Current status and new treatments. Semin Oncol. 33:(Suppl
6). S3–S11. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Agarwal R and Kaye SB: Ovarian cancer:
Strategies for overcoming resistance to chemotherapy. Nat Rev
Cancer. 3:502–516. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Mabuchi S, Kuroda H, Takahashi R and
Sasano T: The PI3K/AKT/mTOR pathway as a therapeutic target in
ovarian cancer. Gynecol Oncol. 137:173–179. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Arend RC, Londoño-Joshi AI, Straughn JM Jr
and Buchsbaum DJ: The Wnt/β-catenin pathway in ovarian cancer: A
review. Gynecol Oncol. 131:772–779. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Takebe N, Harris PJ, Warren RQ and Ivy SP:
Targeting cancer stem cells by inhibiting Wnt, Notch, and Hedgehog
pathways. Nat Rev Clin Oncol. 8:97–106. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Park JT, Li M, Nakayama K, Mao TL,
Davidson B, Zhang Z, Kurman RJ, Eberhart CG, Shih IeM and Wang TL:
Notch3 gene amplification in ovarian cancer. Cancer Res.
66:6312–6318. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Rose SL, Kunnimalaiyaan M, Drenzek J and
Seiler N: Notch 1 signaling is active in ovarian cancer. Gynecol
Oncol. 117:130–133. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Liao X, Siu MK, Au CW, Wong ES, Chan HY,
Ip PP, Ngan HY and Cheung AN: Aberrant activation of hedgehog
signaling pathway in ovarian cancers: Effect on prognosis, cell
invasion and differentiation. Carcinogenesis. 30:131–140. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Artavanis-Tsakonas S, Rand MD and Lake RJ:
Notch signaling: Cell fate control and signal integration in
development. Science. 284:770–776. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Dang TP: Notch, apoptosis and cancer. Adv
Exp Med Biol. 727:199–209. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Weng AP, Ferrando AA, Lee W, Morris JP IV,
Silverman LB, Sanchez-Irizarry C, Blacklow SC, Look AT and Aster
JC: Activating mutations of NOTCH1 in human T cell acute
lymphoblastic leukemia. Science. 306:269–271. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Westhoff B, Colaluca IN, D'Ario G,
Donzelli M, Tosoni D, Volorio S, Pelosi G, Spaggiari L, Mazzarol G,
Viale G, et al: Alterations of the Notch pathway in lung cancer.
Proc Natl Acad Sci USA. 106:22293–22298. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Stylianou S, Clarke RB and Brennan K:
Aberrant activation of notch signaling in human breast cancer.
Cancer Res. 66:1517–1525. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Rose SL: Notch signaling pathway in
ovarian cancer. Int J Gynecol Cancer. 19:564–566. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Rahman MT, Nakayama K, Rahman M, Katagiri
H, Katagiri A, Ishibashi T, Ishikawa M, Iida K, Nakayama S, Otsuki
Y, et al: Notch3 overexpression as potential therapeutic target in
advanced stage chemoresistant ovarian cancer. Am J Clin Pathol.
138:535–544. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sanchez GM, Re L, Giuliani A, Núñez-Sellés
AJ, Davison GP and León-Fernández OS: Protective effects of
Mangifera indica L. extract, mangiferin and selected
antioxidants against TPA-induced biomolecules oxidation and
peritoneal macrophage activation in mice. Pharmacol Res.
42:565–573. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Dar A, Faizi S, Naqvi S, Roome T,
Zikr-ur-Rehman S, Ali M, Firdous S and Moin ST: Analgesic and
antioxidant activity of mangiferin and its derivatives: The
structure activity relationship. Biol Pharm Bull. 28:596–600. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Guha S, Ghosal S and Chattopadhyay U:
Antitumor, immunomodulatory and anti-HIV effect of mangiferin, a
naturally occurring glucosylxanthone. Chemotherapy. 42:443–451.
1996. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Duang XY, Wang Q, Zhou XD and Huang DM:
Mangiferin: A possible strategy for periodontal disease to therapy.
Med Hypotheses. 76:486–488. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
García-Rivera D, Delgado R, Bougarne N,
Haegeman G and Berghe WV: Gallic acid indanone and mangiferin
xanthone are strong determinants of immunosuppressive anti-tumour
effects of Mangifera indica L. bark in MDA-MB231 breast
cancer cells. Cancer Lett. 305:21–31. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Noratto GD, Bertoldi MC, Krenek K, Talcott
ST, Stringheta PC and Mertens-Talcott SU: Anticarcinogenic effects
of polyphenolics from mango (Mangifera indica) varieties. J
Agric Food Chem. 58:4104–4112. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chieli E, Romiti N, Rodeiro I and Garrido
G: In vitro effects of Mangifera indica and polyphenols
derived on ABCB1/P-glycoprotein activity. Food Chem Toxicol.
47:2703–2710. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Cheng P, Peng ZG, Yang J and Song SJ: The
effect of mangiferin on telomerase activity and apoptosis in
leukemic K562 cells. Zhong Yao Cai. 30:306–309. 2007.(In Chinese).
PubMed/NCBI
|
|
26
|
Peng ZG, Luo J, Xia LH, Chen Y and Song
SJ: CML cell line K562 cell apoptosis induced by mangiferin.
Zhongguo Shi Yan Xue Ye Xue Za Zhi. 12:590–594. 2004.(In Chinese).
PubMed/NCBI
|
|
27
|
Percival SS, Talcott ST, Chin ST, Mallak
AC, Lounds-Singleton A and Pettit-Moore J: Neoplastic
transformation of BALB/3T3 cells and cell cycle of HL-60 cells are
inhibited by mango (Mangifera indica L.) juice and mango
juice extracts. J Nutr. 136:1300–1304. 2006.PubMed/NCBI
|
|
28
|
Chari NS, Pinaire NL, Thorpe L, Medeiros
LJ, Routbort MJ and McDonnell TJ: The p53 tumor suppressor network
in cancer and the therapeutic modulation of cell death. Apoptosis.
14:336–347. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Li H, Huang J, Yang B, Xiang T, Yin X,
Peng W, Cheng W, Wan J, Luo F, Li H, et al: Mangiferin exerts
antitumor activity in breast cancer cells by regulating matrix
metalloproteinases, epithelial to mesenchymal transition, and
β-catenin signaling pathway. Toxicol Appl Pharmacol. 272:180–190.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Shi W, Deng J, Tong R, Yang Y, He X, Lv J,
Wang H, Deng S, Qi P, Zhang D, et al: Molecular mechanisms
underlying mangiferin-induced apoptosis and cell cycle arrest in
A549 human lung carcinoma cells. Mol Med Rep. 13:3423–3432.
2016.PubMed/NCBI
|
|
31
|
Ozols RF, Bundy BN, Greer BE, Fowler JM,
Clarke-Pearson D, Burger RA, Mannel RS, DeGeest K, Hartenbach EM,
Baergen R, et al: Gynecologic Oncology Group: Phase III trial of
carboplatin and paclitaxel compared with cisplatin and paclitaxel
in patients with optimally resected stage III ovarian cancer: A
Gynecologic Oncology Group study. J Clin Oncol. 21:3194–3200. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Luo X, Budihardjo I, Zou H, Slaughter C
and Wang X: Bid, a Bcl2 interacting protein, mediates cytochrome c
release from mitochondria in response to activation of cell surface
death receptors. Cell. 94:481–490. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Worsley SD, Ponder BA and Davies BR:
Overexpression of cyclin D1 in epithelial ovarian cancers. Gynecol
Oncol. 64:189–195. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Yao J and Qian C: Inhibition of Notch3
enhances sensitivity to gemcitabine in pancreatic cancer through an
inactivation of PI3K/Akt-dependent pathway. Med Oncol.
27:1017–1022. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Sarkar A, Sreenivasan Y, Ramesh GT and
Manna SK: beta-D-Glucoside suppresses tumor necrosis factor-induced
activation of nuclear transcription factor kappaB but potentiates
apoptosis. J Biol Chem. 279:33768–33781. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Rajendran P, Ekambaram G and Sakthisekaran
D: Protective role of mangiferin against Benzo(a)pyrene induced
lung carcinogenesis in experimental animals. Biol Pharm Bull.
31:1053–1058. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Allenspach EJ, Maillard I, Aster JC and
Pear WS: Notch signaling in cancer. Cancer Biol Ther. 1:466–476.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Nickoloff BJ, Osborne BA and Miele L:
Notch signaling as a therapeutic target in cancer: A new approach
to the development of cell fate modifying agents. Oncogene.
22:6598–6608. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wang M, Wu L, Wang L and Xin X:
Down-regulation of Notch1 by gamma-secretase inhibition contributes
to cell growth inhibition and apoptosis in ovarian cancer cells
A2780. Biochem Biophys Res Commun. 393:144–149. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Drenzek JG, Seiler NL, Jaskula-Sztul R,
Rausch MM and Rose SL: Xanthohumol decreases Notch1 expression and
cell growth by cell cycle arrest and induction of apoptosis in
epithelial ovarian cancer cell lines. Gynecol Oncol. 122:396–401.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Liu MX, Siu MK, Liu SS, Yam JW, Ngan HY
and Chan DW: Epigenetic silencing of microRNA-199b-5p is associated
with acquired chemoresistance via activation of JAG1-Notch1
signaling in ovarian cancer. Oncotarget. 5:944–958. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Park JT, Chen X, Tropè CG, Davidson B,
Shih IeM and Wang TL: Notch3 overexpression is related to the
recurrence of ovarian cancer and confers resistance to carboplatin.
Am J Pathol. 177:1087–1094. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Gupta N, Xu Z, El-Sehemy A, Steed H and Fu
Y: Notch3 induces epithelial-mesenchymal transition and attenuates
carboplatin-induced apoptosis in ovarian cancer cells. Gynecol
Oncol. 130:200–206. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
McAuliffe SM, Morgan SL, Wyant GA, Tran
LT, Muto KW, Chen YS, Chin KT, Partridge JC, Poole BB, Cheng KH, et
al: Targeting Notch, a key pathway for ovarian cancer stem cells,
sensitizes tumors to platinum therapy. Proc Natl Acad Sci USA.
109:E2939–E2948. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Choi JH, Park JT, Davidson B, Morin PJ, Ie
M Shih and Wang TL: Jagged-1 and Notch3 juxtacrine loop regulates
ovarian tumor growth and adhesion. Cancer Res. 68:5716–5723. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Chen X, Stoeck A, Lee SJ, Shih IeM, Wang
MM and Wang TL: Jagged1 expression regulated by Notch3 and
Wnt/β-catenin signaling pathways in ovarian cancer. Oncotarget.
1:210–218. 2010. View Article : Google Scholar : PubMed/NCBI
|