Introduction
Cervical cancer is one of the most common causes of
cancer-associated mortality among women worldwide. Many patients
with cervical cancer present with advanced stage disease at the
time of diagnosis. However, available treatment options are limited
and patient prognosis is usually poor (1). Therefore, there is an urgent need to
develop novel and effective therapeutic regimens for women with
advanced cervical cancer.
The PI3K/AKT pathway is one of the most important
signaling cascades, which regulates numerous essential
physiological functions and cellular processes (2,3).
Alterations that affect the activation of certain components of the
PI3K pathway occur frequently in various types of cancer, including
cervical cancer (1,2,4), making
them attractive targets for drug development. However, pan-PI3K
inhibitors have been shown to exert serious adverse effects
following long-term treatment and have not exhibited significant
results in recent clinical trials (5). An increasing number of studies have
demonstrated that isoform-selective PI3K inhibitors may be safer
and achieve superior therapeutic efficacy than pan-class I PI3K
inhibitors (6,7). PIK3CA, which encodes the p110α
catalytic isoform of class IA PI3Ks, is one of the most frequently
mutated oncogenes in cervical cancer (1,4),
indicating that PI3K p110α may be a potential therapeutic target
for cervical cancer. BYL719, a PI3K p110α-specific inhibitor
(8), is currently being assessed in
phase I/II clinical trials for the treatment of various types of
cancer (https://www.clinicaltrials.gov). A BYL719 phase I
study demonstrated a marked antineoplastic effect and a promising
safety profile in various types of tumor with PIK3CA
mutations (9). In addition, a phase
Ib study revealed that the combined use of BYL719 and letrozole
yielded favorable results in estrogen-receptor-positive breast
cancer, independent of the PIK3CA mutation status (10).
Poly(ADP-ribose) polymerase (PARP) inhibitors are a
promising class of novel drugs. PARP inhibitors were designed to
exploit synthetic lethality, a situation in which tumors carrying
deleterious BRCA1/2 mutations were sensitive to PARP inhibitors due
to a defect in the homologous recombination repair pathway
(11,12). Three PARP inhibitors have been
approved by the Food and Drug Administration for BRCA1/2: Olaparib,
rucaparib (for mutated advanced ovarian cancer) and niraparib (for
recurrent epithelial ovarian, fallopian tube or primary peritoneal
cancer) (13–15). The primary cytotoxic effect of PARP
inhibitors has also been associated with the trapping of cytotoxic
PARP1-DNA complexes (16). PARP
inhibitors used in a clinical setting differ in their ability to
trap PARP1 at DNA damage sites. BMN673 (talazoparib) exhibits ~100
times more potent PARP1-trapping activity than niraparib, rucaparib
or olaparib (17), inducing marked
cytotoxic effects (18–20).
A phase I clinical trial of veliparib, a PARP
inhibitor, in combination with cisplatin and paclitaxel
demonstrated favorable effects in persistent and recurrent cervical
cancer (21). A recent preclinical
study also reported that olaparib sensitizes human cervical cancer
cells to cisplatin and exhibits the anti-invasive properties of
cervical cancer cells (22),
indicating that PARP inhibitors have therapeutic potential in
cervical cancer.
The present study aimed to investigate whether the
combined use of PI3K inhibitors with PARP inhibitors can
effectively treat homologous recombination repair (HRR)-proficient
cervical cancer cells. The results revealed that the PI3K
p110α-specific inhibitor BYL719, rather than the pan-PI3K inhibitor
BKM120, synergized with BMN673, a PARP inhibitor with superior
PARP-trapping activity, to effectively suppress cervical cancer
cell growth, migration and invasion. The potential mechanism
underlying the therapeutic effect conferred by the combined
inhibition of p110α by BYL719 and PARP by BMN673 was also assessed.
Additionally, the effect of the proposed drug combination on ex
vivo cultured patient-derived cervical tumor explants was
assessed.
Materials and methods
Cell culture and reagents
CaSki, HeLa and SiHa human cervical cancer cell
lines and the H8 human cervical epithelial cell line were obtained
from the American Type Culture Collection (ATCC). The ME180 cell
line was purchased from the Cell Bank of the Chinese Academy of
Sciences. The cells were cultured at 37°C as follows: CaSki and H8
cells were cultured in RPMI-1640 with 10% fetal bovine serum (FBS),
HeLa cells were cultured in DMEM containing 10% FBS, ME180 cells
were cultured in McCoy's 5A (Gibco; Thermo Fisher Scientific, Inc.)
supplemented with 10% FBS and SiHa cells were cultured in MEM
(Gibco; Thermo Fisher Scientific, Inc.) with 10% FBS. All media
were supplemented with 100 U/ml penicillin and streptomycin
(HyClone; GE Healthcare Life Sciences). The PI3K p110α inhibitor,
BYL719 and the PARP inhibitor, BMN673 were purchased from
MedChemExpress.
Drug combination analysis
The drug combination was determined using a Cell
Counting Kit-8 (CCK-8) viability assay (Dojindo Molecular
Technologies, Inc.). The cells were seeded at a density of 2,000 or
700 cells per well for 3 or 7 days under the treatment of different
concentrations of drugs at 37°C. CCK-8 solution (10 µl) and 90 µl
of medium were added to each well of the plate. The plate was
incubated for 2 h at 37°C. The absorbance at a wavelength of 450 nm
was measured by an automated reader. The combination index (CI) was
calculated to determine the combinational effects using CalcuSyn
software version 2.0 (Biosoft) as previously described (23). Synergistic effects were indicated by a
CI value <1.
Three-dimensional (3D) sphere
culture
The 3D sphere culture experiments were performed as
previously described (24). The
cervical cancer cells were seeded on plates coated with 50%
Matrigel (BD Biosciences) at a density of 1,500-2,000 cells
depending on the size of the cells and 50% of the aforementioned
respective mediums without serum. The cells were then cultured in
their respective mediums supplemented with 5% FBS and 2% Matrigel.
After 72 h, the cells were treated with BYL719 (500 nM) and BMN673
(50 nM) for 8 days at 37°C, as indicated, and media was replaced
every 2 days. Images of the 3D cell cultures were captured using an
inverted phase-contrast microscope (Leica Microsystems) and scored
according to the diameter of the 3D spheres.
Transwell migration and invasion
assay
The assays were performed in Boyden chambers with
8-µm pore filter inserts for 24-well plates (BD Biosciences). The
cervical cancer cells at a density of 105 cells in 200
µl of serum-free medium were added to the upper chamber with or
without Matrigel pre-coated filters (BD Biosciences). The lower
chamber was filled with 600 µl of medium containing 10% FBS as an
attractant. Following drug treatment with BYL719 (500 nM) and
BMN673 (50 nM) for 24 h in the Transwell migration assay and for 48
h in the invasion assay at 37°C, cells that had migrated through
the pores on the underside of the filter were fixed and stained
with crystal violet. Images of the cells on the underside of the
filter were captured with a Leica optical microscope (Leica
Microsystems) and the cell numbers were counted in 10 independent
visual fields.
Western blotting and antibodies
The cells were washed in PBS and lysed using RIPA
buffer (Sigma-Aldrich; Merck KGaA) supplemented with protease and
phosphatase inhibitors (Roche Diagnostics). A total of 40 µg
protein concentration was then determined using a BCA kit (Tiangen
Biochemical Technology Co., Ltd., Beijing, China). Blocking buffer
(5% skim milk) was added and shaken gently for ~2 h at 25°C,
separated via SDS-PAGE and transferred onto PVDF membranes. The
blots were probed with the following antibodies: Phosphorylated
(p-)AKT (Ser473; cat. no. 700392; 1:1,000 dilution; Cell Signaling
Technology, Inc.), p-S6RP (Ser235/236; cat. no. 4858S; 1:1,000
dilution; Cell Signaling Technology, Inc.), cleaved-PARP (cat. no.
5625S; 1:1,000 dilution; Cell Signaling Technology, Inc.), PARP1
(cat. no. 9542S; 1:1,000 dilution; Cell Signaling Technology,
Inc.), histone H3 (cat. no. 17168-1-AP; 1:1,000 dilution;
ProteinTech Group, Inc.) and vinculin (cat. no. V9264; 1:10,000
dilution; Sigma-Aldrich; Merck KGaA). The secondary antibodies
included goat anti-mouse IgG (cat. no. A11031; 1:5,000 dilution;
Invitrogen; Thermo Fisher Scientific, Inc.) and goat anti-rabbit
IgG (cat. no. A11034; 1:5,000 dilution; Invitrogen; Thermo Fisher
Scientific, Inc.) shaken gently for at least 1 h at 25°C. The blots
were detected with LI-COR Odyssey (LI-COR Biosciences, Lincoln, NE,
USA).
Comet assay
Following drug treatment with BYL719 (500 nM) and
BMN673 (50 nM) for 48 h, a comet assay was performed, as previously
described (25). Following
electrophoresis, the cells were stained with ethidium bromide, and
images were captured with a fluorescence microscope (Leica
Microsystems) and analyzed using CaspLab 1.0.0 software. For each
group, 200 randomly selected cells were assessed, and the degree of
DNA damage was calculated as the percentage of DNA in the tail.
Immunofluorescence analysis
Immunofluorescent staining was performed as
previously described (25). Following
drug treatment with BYL719 (500 nM) and BMN673 (50 nM) for 48 h,
the cells were incubated with anti-RAD51 (cat. no. sc-53428; 1:800
dilution; Santa Cruz Biotechnology, Inc.) or anti-γH2AX (Ser139;
cat. no. 9718S; 1:500 dilution; Cell Signaling Technology, Inc.)
primary antibodies overnight at 4°C and fluorescence-conjugated
secondary antibodies for 1 h at 25°C. Images of the cells were
subsequently captured using a fluorescence microscope (Leica
Microsystems). For each group, 200 randomly selected cells were
assessed and cells containing more than five foci were scored as
positive.
Subcellular fractionating and PARP
trapping assay
Subcellular chromatin fractionating was performed as
previously described (16). The cell
pellets were lysed using cytoplasmic extraction buffer and membrane
extraction buffer. The collected nuclei were then further lysed in
nuclear extraction buffer to isolate the nuclear soluble fraction.
Micrococcal nuclease (5 units; Thermo Fisher Scientific, Inc.) was
used to release chromatin-bound proteins in the remaining nuclear
insoluble fraction. PARP binding in chromatin was subsequently
assessed via western blotting.
Patient information, tissue
preparation and ex vivo culture of patient tumor tissues
Two cases of cervical cancer were included in the
present study. The first is a 60-year-old patient with newly
diagnosed primary high-grade cervical squamous cell carcinoma. The
second is a 63-year-old patient with newly diagnosed primary
high-grade cervical adenocarcinoma. Neither patient had received
any treatment prior to hysterectomy. The acquisition of tumor
tissues was performed under an Institutional Review Board protocol
approved by the First Hospital of Dalian Medical University
(Dalian, China).
The tumor tissues were sectioned into ~200-µm
sections using an automated vibratome (Leica Biosystems). The
sections were then randomized and cultured in RPMI-1640 medium
supplemented with 10% FBS, 1% antibiotic/antimycotic, 1 mg/100 ml
hydrocortisone and 1 mg/100 ml insulin. Following plating, the
sections were allowed to recover for 2 h and were subsequently
treated with BYL719 (500 nM) and BMN673 (50 nM) as single-agents or
in combination for 24 h. Each treatment condition was examined
using at least three tumor sections. All sections were cultured at
37°C and 5% CO2 until harvest. The tissues were then
fixed with paraformaldehyde and processed for histology and
immunohistochemical (IHC) analyses.
Histology and IHC analysis
The histological and IHC analyses were performed as
described previously (25).
Formalin-fixed and paraffin-embedded tumor blocks were sectioned
and stained with hematoxylin for 10 min and eosin for 1 min
(H&E) at room temperature. IHC was subsequently performed using
the following antibodies: Ki67 (cat. no. LM13785; 1:200 dilution;
Vector Laboratories, Inc.), cleaved-caspase-3 (cat. no. 9664S;
1:1,000 dilution; Cell Signaling Technology, Inc.), p-AKT (Ser473;
cat. no. 700392; 1:200 dilution; Invitrogen; Thermo Fisher
Scientific, Inc.), p-S6RP (cat. no. 4858S; 1:400 dilution; Cell
Signaling Technology, Inc.) and γH2AX (cat. no. 9718S; 1:500
dilution; Cell Signaling Technology, Inc.). Images were captured
and quantified using an upright optical microscope (Leica
Microsystems). At least five random 40X fields were assessed. Image
Pro Plus 6.0 software (Media Cybernetics, Inc.) was used to
quantify the protein levels of p-AKT and p-S6RP.
Statistical analyses
For in vitro analyses, each experiment was
repeated at least three times. For analyzing quantitative results,
Student's t-test and χ2 test were used for two-group
comparisons, and one-way analysis of variance (ANOVA) and the
Student-Newman-Keuls post hoc test were used for multiple
comparisons. P<0.05 was considered to indicate a statistically
significant difference. All statistical analyses were performed
using GraphPad Prism 5.0 software (GraphPad Software, Inc.).
Results
Combined use of BMN673 and BYL719
synergistically inhibits cervical cancer cell growth, migration and
invasion
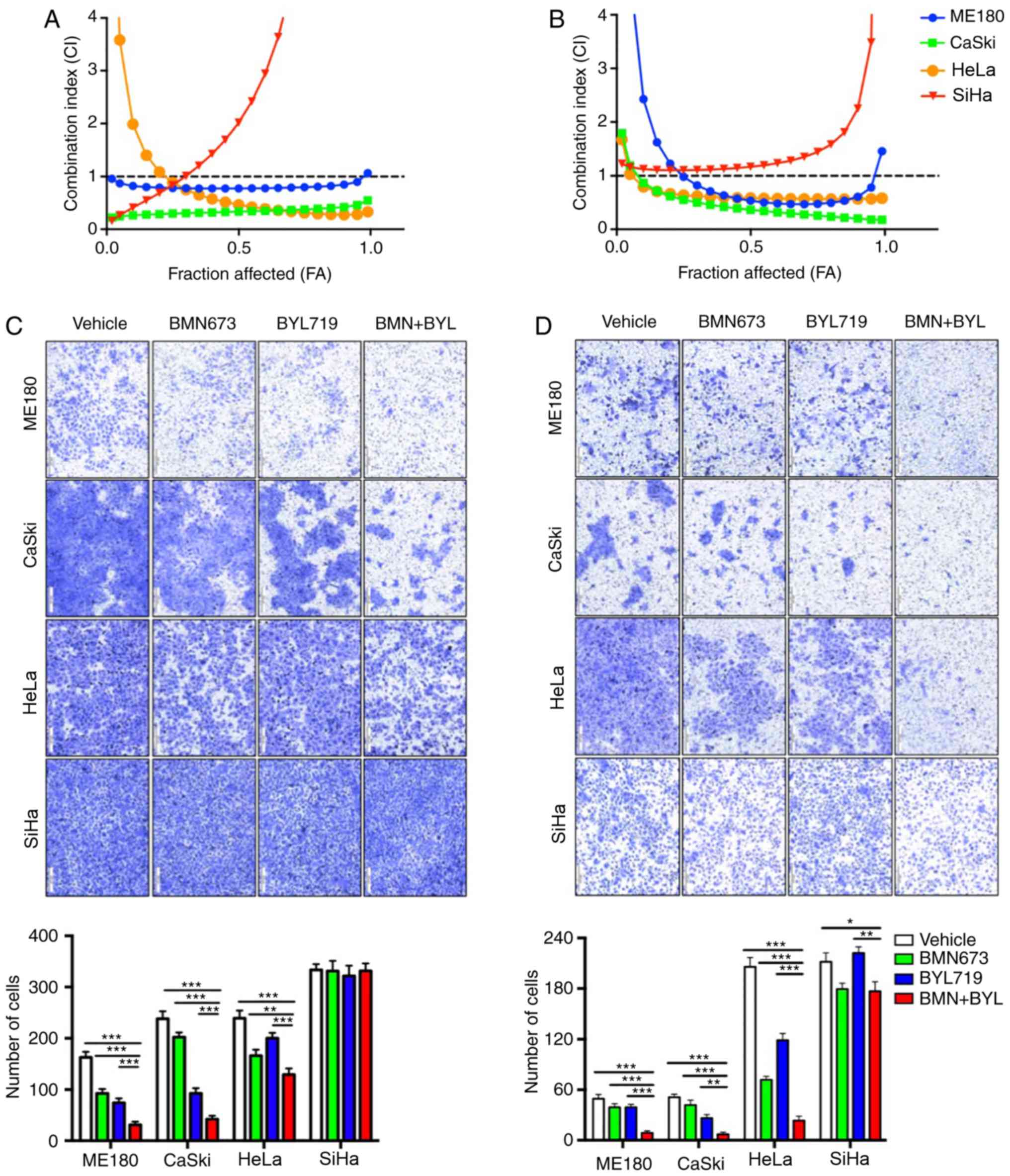
The growth of four cervical cancer cell lines
(ME180, CaSki, HeLa and SiHa) was assessed following treatment with
certain PI3K inhibitors, including BKM120 and BYL719, alone or in
combination with BMN673. The cervical cancer cells were treated
with increasing drug concentrations for 72 h and the cytotoxic
effects were analyzed via a CCK-8 assay followed by median-effect
analysis. Although none of the four cervical cancer cell lines
appeared to respond to the combination of BKM120 and BMN673 (data
not shown), synergistic inhibitory effects on the proliferation of
cultured ME180, CaSki and HeLa cells were observed following the
combined treatment of BYL719 and BMN673. However, no effect was
demonstrated in SiHa cells (Fig. 1A).
The synergistic inhibitory effect of combined BYL719 and BMN673
treatment was also observed when the ME180, CaSki and HeLa cells
were subjected to longer drug exposure for 7 days (Fig. 1B). However, the SiHa cells remained
unresponsive. These results indicated that further analysis was
required to determine the therapeutic potential of the combined
inhibition of p110α and PARP in cervical cancer.
The inhibition of migration and metastasis remains a
challenge for cervical cancer treatment (26). To assess the effect of drug
combinations on cell migration, a Transwell migration assay was
performed with confluent monolayers of cervical cancer cells.
Treatment with BMN673 and BYL719 as single agents led to partially
filled spaces in the migration assay, whereas combinational
treatment synergistically attenuated the migration of ME180, CaSki
and HeLa cells, but not SiHa cells (Fig.
1C). A Matrigel invasion assay was subsequently performed to
analyze the effect of the BMN673 and BL719 combined treatment on
the invasion of cervical cancer cells. As hypothesized, BMN673 and
BYL719 when used alone only moderately inhibited cell invasion,
whereas dual treatment with BMN673 and BYL719 markedly inhibited
the invasion of ME180, CaSki and HeLa cells (Fig. 1D). However, combinational treatment
did not lead to a significant synergistic inhibitory effect on SiHa
cell invasion (Fig. 1D). These
results indicated that dual treatment with BMN673 and BYL719
effectively inhibited the migration and invasion of certain
cervical cancer cells.
The effect of drug combinations on the growth of 3D
spheroids cultured in Matrigel was assessed in the present study,
as it closely mimics the tumor microenvironment (24). Single-agent treatment with BMN673 or
BYL719 exerted minor inhibitory effects, whereas dual treatment
with BMN673 and BYL719 markedly attenuated the growth of ME180 and
CaSki cells, and to a lesser extent, HeLa cells, in 3D Matrigel
(Fig. 1E). Similar to observations
made following two-dimensional (2D) monoculture, the growth of SiHa
cell 3D spheroids remained intact in all treatment groups. These
results indicated that the combined use of BYL719 and BMN673 may be
an effective strategy for the treatment of cervical cancer.
Combined use of BYL719 and BMN673
synergistically inhibits the PI3K/AKT/mTOR signaling pathway and
induces G2/M arrest
The effect of dual treatment with BYL719 and BMN673
on the PI3K/AKT pro-survival signaling pathway was subsequently
assessed. The results of western blot analysis revealed that BYL719
used alone or in combination with BMN673 markedly reduced the
phosphorylation of AKT and S6RP proteins in the ME180, CASKI and
HeLa cells (Fig. 2A). This was
consistent with the inhibitory effect of BYL719 on PI3K p110α. In
SiHa cells, the combined treatment markedly reduced the
phosphorylation of S6RP protein, and p110α inhibition induced the
activation of pro-survival AKT (Fig.
2A). This may explain the lack of synergy between BYL719 and
BMN673 in this cell line. Additionally, western blotting revealed
that drugs used in combination did not yield a synergistic increase
in cleaved PARP (an apoptotic marker) in any of the four cervical
cell lines (Fig. 2B).
The G2/M DNA damage checkpoint is known to prevent
genomic DNA-damaged cells from entering mitosis (27). To further assess the effect of drugs
in this regard, cell cycle analysis was performed via flow
cytometry. Compared with its effect as a single-agent, BMN673 used
in combination with BYL719 induced more marked G2/M phase arrest in
the ME180, CaSki and HeLa cells (Fig.
2C). Concordant with the lack of observable effects in SiHa
cells, the additional use of BYL719 did not lead to further
increases in G2/M arrest caused by BMN673 single-agent treatment
(Fig. 2C).
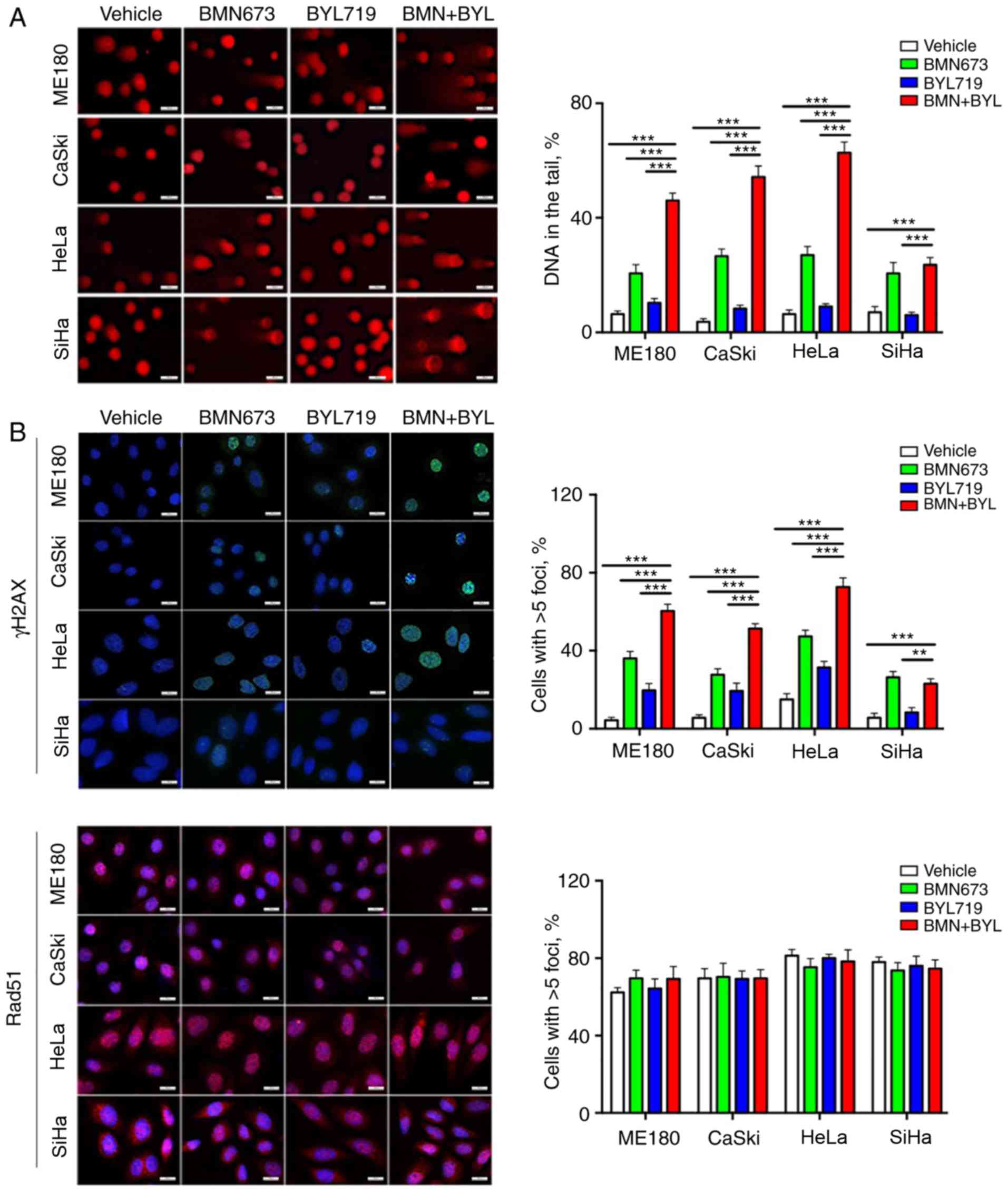
Combined use of BMN673 and BYL719
induces DNA damage
The molecular mechanisms underlying the sensitivity
of cervical cancer cells to combined treatment with BMN673 and
BYL719 were determined. Previous studies have indicated that PI3K
inhibition has the potential to sensitize non-BRCA1/2 mutant or
HRR-proficient tumors to PARP inhibitors (23,25,28,29).
To determine this mechanism in HRR proficient cervical cancer, a
comet assay was performed to assess DNA damage in the different
cell lines. BMN673 monotherapy induced a moderate degree of DNA
damage, whereas the combined use of BYL719 and BMN673 induced a
more pronounced increase of DNA in comet tails in the ME180, CaSki
and HeLa cells. However, no observable effect was observed in SiHa
cells (Fig. 3A). By contrast, BYL719
had minimal impact on DNA integrity in any of the four cervical
cancer cell lines.
To further assess the effect of combined treatment
on DNA damage, nuclear γH2AX foci, which are indicators of DNA
double-strand breaks (DSBs), were assessed via immunofluorescence
staining. BMN673 treatment alone induced a moderate increase in
DSBs, as evaluated by γH2AX nuclear foci, whereas BYL719 treatment
exhibited a weak effect. Consistent with the results from the comet
assay (Fig. 3B), the combined use of
BYL719 with BMN673 resulted in significantly more γH2AX nuclear
foci than single-agent treatment in the ME180, CaSki and HeLa
cells, but not in the SiHa cells (Fig.
3B).
Whether deficient homologous recombination DNA
repair contributed to the cytotoxic effects induced by the combined
inhibition of p110α and PARP was determined. The impact of drug
treatment was therefore assessed on the nuclear foci formation of
RAD51, a key homologous recombination DNA repair protein (30). The immunofluorescence staining
analysis revealed that all four cell lines retained strong nuclear
staining of RAD51 across treatment groups, which was indicative of
competent homologous recombination DNA damage repair (Fig. 3B). Furthermore, unlike the potent
inhibitory effect of combined BYL719 and BMN673 treatment, no
synergistic cytotoxic effect was detected between BYL719 and
olaparib in the cervical cancer cell lines examined (Fig. 3C). The results indicated that the
combined use of BYL719 with BMN673 markedly induced DNA damage via
a mechanism that is independent of deficient homologous
recombination DNA repair. In order to clarify the specificity of
this combination and demonstrate whether this treatment confers
toxicity to normal cells, the effects of the combination of BYL719
and BMN673 on the growth of normal cervical epithelial cells (H8)
was assessed (Fig. 3D and E).
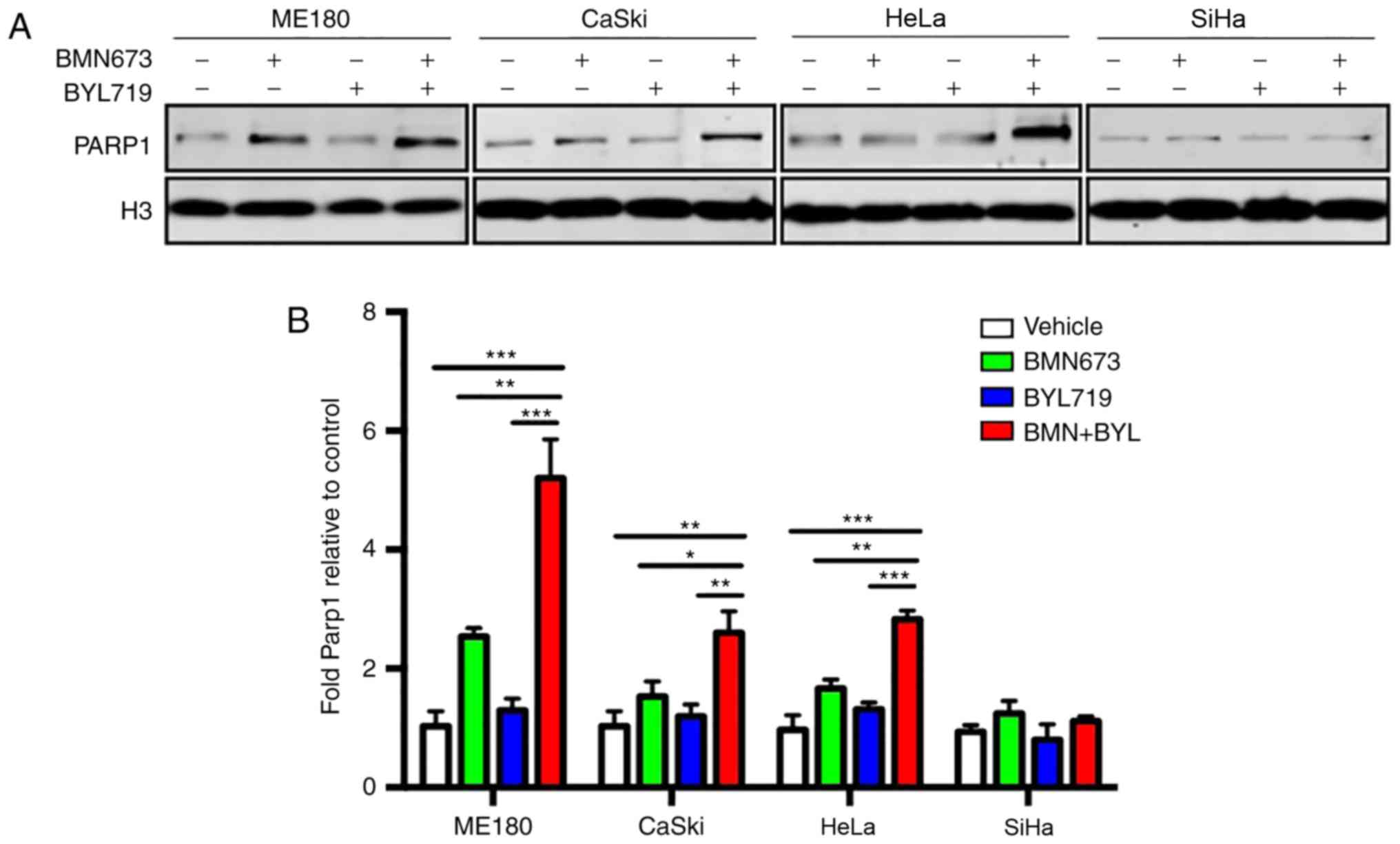
Combined treatment with BYL719 and
BMN673 increases the trapping of PARP1 on chromatin
BMN673 possesses a more potent ability to trap
PARP1/2 at DNA damage sites to form cytotoxic PARP1/2-chromatin
complexes than other PARP inhibitors, including olaparib (17,19).
Therefore, the present study assessed whether the enhanced trapping
of the PARP1-chromatin complex accounted for the therapeutic effect
exerted by BYL719 and BMN673 used in combination. The results
revealed that BYL719 used as a single agent did not induce PARP1
trapping on chromatin (Fig. 4A and
B). However, BMN673 induced moderate PARP1 trapping.
Furthermore, the combined use of BYL719 and BMN673 resulted in a
significantly higher number of PARP1-DNA complexes compared with
that following BMN673 treatment alone in ME180, CaSki and HeLa
cells, but not in SiHa cells (Fig. 4A and
B). These results indicated that enhanced PARP1 trapping on
chromatin may, at least in part, account for the cytotoxic effect
of dual BMN673 and BYL719 treatment on cervical cancer cells.
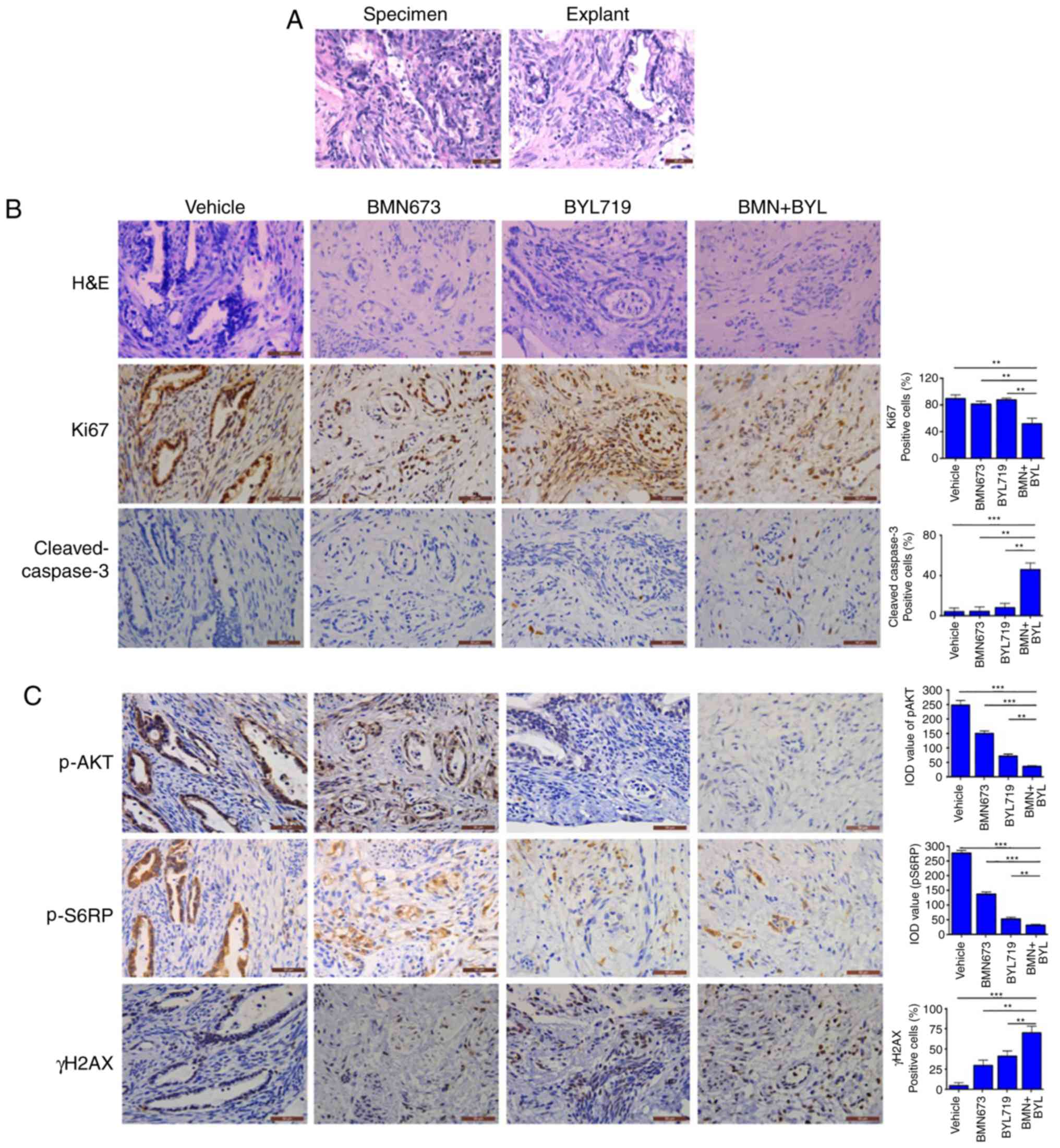
Combined use of BMN673 and BYL719
effectively treats ex vivo explants of cervical cancer harboring
aberrant activation of PI3K signaling
An ex vivo explant culture model of cervical
cancer was utilized to evaluate the therapeutic effect of BYL719
and BMN673 dual treatment. Fresh surgical specimens of a primary
cervical adenocarcinoma and a primary cervical squamous cell
carcinoma were dissected into small sections. The tumor sections
were subsequently cultured on an absorbable gelatin sponge for 2 h
and then treated with BMN673 and BYL719 either alone or in
combination for 24 h. The untreated explants exhibited similar
architecture and cellularity to those of primary tumors (Figs. 5A and 6A), whereas BMN673 and BYL719 used as single
agent treatments exerted minor effects. However, dual BMN673 and
BYL719 treatment resulted in disrupted cellular integrity in both
cases, indicating a promising therapeutic effect induced by the
co-targeting PARP and PI3K p110α (Figs.
5B and 6B). Concordantly, when
compared with the vehicle or single-agent treatment groups, the
combinational treatment group exhibited significantly reduced
proliferation (determined via Ki67 nuclear staining) and enhanced
apoptotic cell death (determined via cleaved caspase-3 staining;
Figs. 5B and 6B). In vivo signaling analysis using
IHC staining revealed high AKT phosphorylation and S6RP signals in
the two vehicle-treated clinical samples, which were indicative of
pro-survival PI3K/AKT/mTOR signaling pathway activation (Figs. 5C and 6C). BYL719 used alone or in combination with
BMN673 abrogated PI3K/AKT/mTOR signaling, as indicated by markedly
reduced levels of pAKT and pS6RP (Figs.
5C and 6C). Consistent with
previous observations that cervical cancer cell line models harbor
aberrantly activated PI3K/AKT signaling, BMN673 and BYL719 dual
treatment resulted in a substantial increase in the formation of
γH2AX nuclear foci (Figs. 5C and
6C), indicating an accumulation of
DSBs. These data may provide insights into the subset of cervical
cancer cells that may be effectively treated with the combination
of BMN673 and BYL719.
 | Figure 5.Response of primary cervical squamous
cell carcinoma explants to BYL719 and BMN673 as single agents or in
combination (A) Representative H&E staining between surgical
specimens and cultured tumor explants. Representative images of (B)
H&E staining and (C) immunohistochemical staining analyses of
Ki67, cleaved-caspase 3, p-AKT, p-S6RP and γH2AX in tumor explants
treated with inhibitors as indicated (50 nM BMN673, 500 nM BYL719;
scale bar, 50 µm. Data are presented as the mean ± SEM. *P<0.05,
**P<0.01 and ***P<0.001 (analysis of variance and
Student-Newman-Keuls post hoc test). H&E, hematoxylin and
eosin; p-, phosphorylated; BYL, BYL719; BMN, BMN673. |
Discussion
There is an urgent need to develop an effective
therapeutic regimen for advanced cervical cancer. The inhibition of
PARP1 causes synthetic lethality in ovarian tumor cells with
deleterious BRCA1/2 mutations or deficiency in homologous
recombination DNA repair (31,32).
Unlike ovarian cancer, with >50% of cases lacking HRR
proficiency (33), the majority of
cervical cancer cases retain intact HRR ability, thus limiting the
application of PARP inhibitors in this disease. In previous years,
intensive preclinical efforts have been made to develop novel PARP
inhibitor-based combinations against HRR proficient tumors
(29,34–36). For
example, PI3K inhibition with BKM120, a pan-class I PI3K inhibitor,
has been revealed to result in impaired HR DNA repair, thus
synergizing with the PARP inhibitor, olaparib, in a number of
cancer types with BRCA proficiency, including triple negative
breast cancer (29), prostate cancer
(37), ovarian cancer (23,28), lung
cancer (38) and endometrial cancer
(25,39). Although the aforementioned studies
reported the effective use of BKM120 in combination with olaparib,
to the best of our knowledge, the present study demonstrated for
the first time that BYL719 instead of BKM120 enhanced the antitumor
effect of BMN673, a PARP inhibitor with potent PARP1-trapping
activity.
PARP1 is known to be recruited to damaged DNA sites
and can be covalently trapped by PARP inhibitors specifically at
single-strand DNA breaks (16,17). The
results of the present study revealed that the observed synergistic
cytotoxicity of combined BYL719 and BMN673 most likely occurs by
increasing PARP1 binding to chromatin and thus inducing DNA damage.
However, future investigations are necessary to investigate the
molecular mechanisms by which dual BYL719 and BMN673 treatment
increases the retention of PARP1 at DNA damage sites, inducing
cytotoxicity.
The three cervical cancer cell lines (ME180, CaSki
and HeLa) that responded well to combined BYL719 and BMN673
treatment harbored PI3K pathway alterations, with ME180 and CaSki
cells carrying PIK3CA mutations and HeLa cells carrying a mutation
in EGFR upstream of the PI3K/AKT signaling pathway. Therefore, it
was hypothesized that the mutational status of the PI3K pathway may
explain the sufficient attenuation of the AKT/mTOR signaling
pathway in response to p110α inhibition by BYL719. By contrast,
SiHa cells, which did not respond to drug combinations, did not
carry PI3K/AKT pathway alterations (https://cancer.sanger.ac.uk). Furthermore, BYL719
treatment in SiHa cells resulted in the induction, rather than a
reduction, of pro-survival AKT signaling, indicating that the
PI3K/AKT signaling pathway was altered in response to PI3K
inhibition. These results indicated that abrogated PI3K/AKT
signaling in response to the combined use of BYL719 and BMN673 may
predict therapeutic responses in cervical cancer. The results of
the present study also revealed that BKM120 did not phenocopy the
effect of BYL719 and that BKM120 in combination with BMN673 did not
yield synergistic effects, indicating the distinct roles of class
IA PI3K isoforms in regulating the response of cervical cancer
cells to PARP inhibitors.
To assess drug efficacy in an in vivo
setting, primary tumor explant culture models of surgically
resected primary cervical adenocarcinoma and primary cervical
squamous cell carcinoma were used in the present study, both of
which harbor activated PI3K/AKT signaling. The results demonstrated
that these ex vivo explant culture models recapitulated
several features of primary cervical tumors, with preserved tissue
architecture and tumor microenvironments. Therefore, the treatment
of these tumor explant culture models may reveal drug responses
that would be concordant with their respective primary tumors,
representing a promising method for evaluating therapeutic
sensitivities for personalized treatment. In congruence with the
drug responses of cervical cancer cell lines cultured in 2D
monoculture and 3D Matrigel, the two cervical tumor explant culture
models harboring activated PI3K/AKT signaling revealed markedly
attenuated proliferation and induced apoptosis following short-term
exposure to BYL719 and BMN673. In conclusion, the results of the
present study indicated that the combined use of BMN673 and BYL719
may represent a promising therapeutic strategy for cases of
HRR-proficient cervical cancer harboring aberrant PI3K pathway
activation.
Acknowledgements
The authors would like to thank the Pathology
Department of the First Affiliated Hospital of Dalian Medical
University (Nanjing, China) for their technical support in IHC and
staining evaluation.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed supporting
conclusions are included in this published article.
Authors' contributions
PC and SL conceived the study; PC contributed to the
methodology of the study; PC, YW, YL, NJ, LZ and JG validated the
data; PC and YW performed the formal analysis; PC, YW, XM, XX, DD
and LL conducted the investigation; PC performed the writing and
the original draft preparation; SL wrote, reviewed, edited and
supervised the manuscript. All authors have read and approved the
final manuscript and agree to be accountable for all aspects of the
research in ensuring that the accuracy or integrity of any part of
the work are appropriately investigated and resolved.
Ethics approval and consent to
participate
The present study was performed with the approval of
the Ethics Committee of the First Affiliated Hospital of Dalian
Medical University (Nanjing, China; approval no. 2006-SRFA-054).
Written informed consent was obtained from all patients.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ojesina AI, Lichtenstein L, Freeman SS,
Pedamallu CS, Imaz-Rosshandler I, Pugh TJ, Cherniack AD, Ambrogio
L, Cibulskis K, Bertelsen B, et al: Landscape of genomic
alterations in cervical carcinomas. Nature. 506:371–375. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Fruman DA and Rommel C: PI3K and cancer:
Lessons, challenges and opportunities. Nat Rev Drug Discov.
13:140–156. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
3
|
Liu P, Cheng H, Roberts TM and Zhao JJ:
Targeting the phosphoinositide 3-kinase pathway in cancer. Nat Rev
Drug Discov. 8:627–644. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Cancer Genome Atlas Research Network:
Albert Einstein College of Medicine; Analytical Biological
Services; Barretos Cancer Hospital; Baylor College of Medicine;
Beckman Research Institute of City of Hope; Buck Institute for
Research on Aging; Canada's Michael Smith Genome Sciences Centre;
Harvard Medical Schoo; Helen F. Graham Cancer Center & Research
Institute at Christiana Care Health Services et al, . Integrated
genomic and molecular characterization of cervical cancer. Nature.
543:378–384. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rodon J, Dienstmann R, Serra V and
Tabernero J: Development of PI3K inhibitors: Lessons learned from
early clinical trials. Nat Rev Clin Oncol. 10:143–153. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Okkenhaug K, Graupera M and Vanhaesebroeck
B: Targeting PI3K in cancer: impact on tumor cells, their
protective stroma, angiogenesis, and immunotherapy. Cancer Discov.
6:1090–1105. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Thorpe LM, Yuzugullu H and Zhao JJ: PI3K
in cancer: Divergent roles of isoforms, modes of activation and
therapeutic targeting. Nat Rev Cancer. 15:7–24. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Furet P, Guagnano V, Fairhurst RA,
Imbach-Weese P, Bruce I, Knapp M, Fritsch C, Blasco F, Blanz J,
Aichholz R, et al: Discovery of NVP-BYL719 a potent and selective
phosphatidylinositol-3 kinase alpha inhibitor selected for clinical
evaluation. Bioorg Med Chem Lett. 23:3741–3748. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Juric D, Rodon J, Tabernero J, Janku F,
Burris HA, Schellens JHM, Middleton MR, Berlin J, Schuler M,
Gil-Martin M, et al: Phosphatidylinositol 3-Kinase α-selective
inhibition with alpelisib (BYL719) in PIK3CA-altered solid tumors:
Results from the first-in-human study. J Clin Oncol. 36:1291–1299.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Mayer IA, Abramson VG, Formisano L, Balko
JM, Estrada MV, Sanders ME, Juric D, Solit D, Berger MF, Won HH, et
al: A Phase Ib study of alpelisib (BYL719), a PI3Kα-specific
inhibitor, with letrozole in ER+/HER2-metastatic breast cancer.
Clin Cancer Res. 23:26–34. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kaufman B, Shapira-Frommer R, Schmutzler
RK, Audeh MW, Friedlander M, Balmaña J, Mitchell G, Fried G,
Stemmer SM, Hubert A, et al: Olaparib monotherapy in patients with
advanced cancer and a germline BRCA1/2 mutation. J Clin Oncol.
33:244–250. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Scott CL, Swisher EM and Kaufmann SH:
Poly(ADP-ribose) polymerase inhibitors: Recent advances and future
development. J Clin Oncol. 33:1397–1406. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Balasubramaniam S, Beaver JA, Horton S,
Fernandes LL, Tang S, Horne HN, Liu J, Liu C, Schrieber SJ, Yu J,
et al: FDA approval summary: Rucaparib for the treatment of
patients with deleterious BRCA mutation-associated advanced ovarian
cancer. Clin Cancer Res. 23:7165–7170. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kim G, Ison G, McKee AE, Zhang H, Tang S,
Gwise T, Sridhara R, Lee E, Tzou A, Philip R, et al: FDA approval
summary: Olaparib monotherapy in patients with deleterious germline
BRCA-mutated advanced ovarian cancer treated with three or more
lines of chemotherapy. Clin Cancer Res. 21:4257–4261. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Scott LJ: Niraparib: First global
approval. Drugs. 77:1029–1034. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Murai J, Huang SY, Das BB, Renaud A, Zhang
Y, Doroshow JH, Ji J, Takeda S and Pommier Y: Trapping of PARP1 and
PARP2 by clinical PARP inhibitors. Cancer Res. 72:5588–5599. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Murai J, Huang SY, Renaud A, Zhang Y, Ji
J, Takeda S, Morris J, Teicher B, Doroshow JH and Pommier Y:
Stereospecific PARP trapping by BMN 673 and comparison with
olaparib and rucaparib. Mol Cancer Ther. 13:433–443. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Shen Y, Rehman FL, Feng Y, Boshuizen J,
Bajrami I, Elliott R, Wang B, Lord CJ, Post LE and Ashworth A: BMN
673, a novel and highly potent PARP1/2 inhibitor for the treatment
of human cancers with DNA repair deficiency. Clin Cancer Res.
19:5003–5015. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Shen Y, Aoyagi-Scharber M and Wang B:
Trapping Poly (ADP-Ribose) polymerase. J Pharmacol Exp Ther.
353:446–457. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Pommier Y, O'Connor MJ and de Bono J:
Laying a trap to kill cancer cells: PARP inhibitors and their
mechanisms of action. Sci Transl Med. 8:362ps172016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Thaker PH, Salani R, Brady WE, Lankes HA,
Cohn DE, Mutch DG, Mannel RS, Bell-McGuinn KM, Di Silvestro PA,
Jelovac D, et al: A phase I trial of paclitaxel, cisplatin, and
veliparib in the treatment of persistent or recurrent carcinoma of
the cervix: An NRG oncology study (NCT#01281852). Ann Oncol.
28:505–511. 2017.PubMed/NCBI
|
|
22
|
Prasad CB, Prasad SB, Yadav SS, Pandey LK,
Singh S, Pradhan S and Narayan G: Olaparib modulates DNA repair
efficiency, sensitizes cervical cancer cells to cisplatin and
exhibits anti-metastatic property. Sci Rep. 7:128762017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wang D, Li C, Zhang Y, Wang M, Jiang N,
Xiang L, Li T, Roberts TM, Zhao JJ, Cheng H and Liu P: Combined
inhibition of PI3K and PARP is effective in the treatment of
ovarian cancer cells with wild-type PIK3CA genes. Gynecol Oncol.
142:548–556. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lee GY, Kenny PA, Lee EH and Bissell MJ:
Three-dimensional culture models of normal and malignant breast
epithelial cells. Nat Methods. 4:359–365. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Bian X, Gao J, Luo F, Rui C, Zheng T, Wang
D, Wang Y, Roberts TM, Liu P, Zhao JJ and Cheng H: PTEN deficiency
sensitizes endometrioid endometrial cancer to compound PARP-PI3K
inhibition but not PARP inhibition as monotherapy. Oncogene.
37:341–351. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shepherd JH: Cervical cancer. Best Pract
Res Clin Obstet Gynaecol. 26:293–309. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kastan MB and Bartek J: Cell-cycle
checkpoints and cancer. Nature. 432:316–323. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wang D, Wang M, Jiang N, Zhang Y, Bian X,
Wang X, Roberts TM, Zhao JJ, Liu P and Cheng H: Effective use of
PI3K inhibitor BKM120 and PARP inhibitor Olaparib to treat PIK3CA
mutant ovarian cancer. Oncotarget. 7:13153–13166. 2016.PubMed/NCBI
|
|
29
|
Ibrahim YH, Garcia-Garcia C, Serra V, He
L, Torres-Lockhart K, Prat A, Anton P, Cozar P, Guzmán M, Grueso J,
et al: PI3K inhibition impairs BRCA1/2 expression and sensitizes
BRCA-proficient triple-negative breast cancer to PARP inhibition.
Cancer Discov. 2:1036–1047. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
van Gent DC, Hoeijmakers JH and Kanaar R:
Chromosomal stability and the DNA double-stranded break connection.
Nat Rev Genet. 2:196–206. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Fong PC, Boss DS, Yap TA, Tutt A, Wu P,
Mergui-Roelvink M, Mortimer P, Swaisland H, Lau A, O'Connor MJ, et
al: Inhibition of poly(ADP-ribose) polymerase in tumors from BRCA
mutation carriers. N Engl J Med. 361:123–134. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
McCabe N, Turner NC, Lord CJ, Kluzek K,
Bialkowska A, Swift S, Giavara S, O'Connor MJ, Tutt AN, Zdzienicka
MZ, et al: Deficiency in the repair of DNA damage by homologous
recombination and sensitivity to poly(ADP-ribose) polymerase
inhibition. Cancer Res. 66:8109–8115. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Konstantinopoulos PA, Ceccaldi R, Shapiro
GI and D'Andrea AD: Homologous recombination deficiency: Exploiting
the fundamental vulnerability of ovarian cancer. Cancer Discov.
5:1137–1154. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Cardillo TM, Sharkey RM, Rossi DL, Arrojo
R, Mostafa AA and Goldenberg DM: Synthetic lethality exploitation
by an anti-trop-2-SN-38 antibody-drug conjugate, IMMU-132, plus
PARP inhibitors in BRCA1/2-wild-type triple-negative breast cancer.
Clin Cancer Res. 23:3405–3415. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Johnson N, Li YC, Walton ZE, Cheng KA, Li
D, Rodig SJ, Moreau LA, Unitt C, Bronson RT, Thomas HD, et al:
Compromised CDK1 activity sensitizes BRCA-proficient cancers to
PARP inhibition. Nat Med. 17:875–882. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Mo W, Liu Q, Lin CC, Dai H, Peng Y, Liang
Y, Peng G, Meric-Bernstam F, Mills GB, Li K and Lin SY: mTOR
inhibitors suppress homologous recombination repair and synergize
with PARP inhibitors via regulating SUV39H1 in BRCA-proficient
triple-negative breast cancer. Clin Cancer Res. 22:1699–1712. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
González-Billalabeitia E, Seitzer N, Song
SJ, Song MS, Patnaik A, Liu XS, Epping MT, Papa A, Hobbs RM, Chen
M, et al: Vulnerabilities of PTEN-TP53-deficient prostate cancers
to compound PARP-PI3K inhibition. Cancer Discov. 4:896–904. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Cardnell RJ, Feng Y, Mukherjee S, Diao L,
Tong P, Stewart CA, Masrorpour F, Fan Y, Nilsson M, Shen Y, et al:
Activation of the PI3K/mTOR pathway following PARP inhibition in
small cell lung cancer. PLoS One. 11:e01525842016. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Philip CA, Laskov I, Beauchamp MC, Marques
M, Amin O, Bitharas J, Kessous R, Kogan L, Baloch T, Gotlieb WH and
Yasmeen A: Inhibition of PI3K-AKT-mTOR pathway sensitizes
endometrial cancer cell lines to PARP inhibitors. BMC Cancer.
17:6382017. View Article : Google Scholar : PubMed/NCBI
|