Introduction
Hepatocellular carcinoma (HCC) is one of the most
common malignant tumors and the fourth leading cause of
cancer-related death worldwide (1).
Although progress has been achieved in the diagnosis and therapy
over the last decades, the long-term survival rate is
unsatisfactory due to the high recurrence and metastasis rates
(2). Resection is the most widely
used curative treatment for patients with early HCC; however, a
number of patients diagnosed at an advanced stage are ineligible
for surgery (3). Therefore, it is
imperative to unravel the molecular mechanisms underlying the
occurrence and development of HCC to identify novel therapeutic
strategies for this malignancy.
As conserved single-stranded non-coding RNAs of ~22
nucleotides in length, microRNAs (miRNAs) serve pivotal regulatory
roles by protein-coding gene cleavage or translation repression via
interacting with the 3′-untranslated region (3′UTR) of target mRNAs
with imperfect complementarity (4,5).
Accumulating evidence has demonstrated that miRNAs capable of
controlling cell proliferation, metabolism, invasion and
angiogenesis serve crucial roles in the initiation, progression and
metastasis of various types of cancer, including HCC (6,7).
Recent findings have demonstrated that miRNA (miR)-1468,
miR-876-5p, miR-532-3p, miR-3194-3p and miR-519c-3p regulate the
growth and metastasis of HCC via different underlying molecular
mechanisms (8–12). Previous results have suggested that
miR-875-5p is dysregulated in various types of cancer, such as
colorectal carcinoma, lung and prostate cancer (13–16).
In those studies, miR-875-5p was demonstrated to function as a
tumor suppressor or as an oncogenic factor involved in the
proliferation, migration, invasion, apoptosis, chemotherapeutic
sensitivity and radiation response of cancer cells. However,
limited information is currently available about the role of
miR-875-5p and its underlying molecular mechanisms in HCC.
Eukaryotic translation initiation factor 3, which
serves a central role in translation initiation, comprises 13
subunits, among which eukaryotic translation initiation factor 3
subunit a (eIF3a) is the largest one (17). In humans, eIF3a appears to be
ubiquitously expressed and involved in cellular processes such as
translation initiation, cell cycle, and DNA synthesis and repair
(18). Previous findings have
revealed that eIF3a is abnormally expressed and involved in the
tumorigenesis of lung cancer, ameloblastoma, pancreatic cancer and
HCC (19–22). In HCC, eIF3a regulates the
translation of hypoxia-inducible factor 1α (HIF-1α), mediating
HIF-1α-dependent glycolytic metabolism in HCC (22). However, the exact functions of eIF3a
on tumor growth and metastasis and the mechanisms underlying the
aberrant expression of eIF3a in HCC remain unclear.
Current findings demonstrated that miR-875-5p was
downregulated in HCC cells and tissues, which significantly
correlated with short survival and progressed clinical features.
The results of the loss- and gain-of-function experiments confirmed
that miR-875-5p inhibited tumor growth and metastasis in
vitro and in vivo. Mechanistically, miR-875-5p
interacted with the 3′UTR of eIF3a mRNA, downregulating the
expression of eIF3a, which exhibited oncogenic activities in HCC.
Thus, the present study validated that miR-875-5p may inhibit tumor
growth and metastasis by targeting eIF3a in HCC and may represent a
potential target for HCC treatment.
Materials and methods
Tissue samples
HCC and corresponding adjacent non-tumor tissues
were obtained from 90 patients in The First Affiliated Hospital of
Xian Jiaotong University (Xian, China) after providing informed
consent. The patients did not receive any therapy including
radiotherapy, chemotherapy or radiofrequency ablation before the
surgery. All procedures involving human participants were in
accordance with the ethical standards of the Research Ethics
Committee of The First Affiliated Hospital of Xian Jiaotong
University and with the Declaration of Helsinki as revised in 2013.
The clinicopathological and demographic information of the patients
is presented in Table I.
 | Table I.Clinical correlation of miR-875-5p
expression in HCC (n=90). |
Table I.
Clinical correlation of miR-875-5p
expression in HCC (n=90).
|
|
| Expression
level |
|
|---|
|
|
|
|
|
|---|
| Clinical
parameters | Cases (n) |
miR-875-5phigh (n=45) |
miR-875-5plow (n=45) | P-value |
|---|
| Age (years) |
|
|
| 0.499 |
|
<50 | 29 | 16 | 13 |
|
|
≥50 | 61 | 29 | 32 |
|
| Sex |
|
|
| 0.561 |
|
Male | 76 | 39 | 37 |
|
|
Female | 14 | 6 | 8 |
|
| Tumor size
(cm) |
|
|
| 0.006a |
|
<5 | 47 | 30 | 17 |
|
| ≥5 | 43 | 15 | 28 |
|
| Tumor number |
|
|
| 0.267 |
|
Solitary | 59 | 33 | 26 |
|
|
Multiple | 31 | 12 | 19 |
|
| TNM stage |
|
|
| 0.016a |
|
I+II | 72 | 41 | 31 |
|
|
III+IV | 18 | 4 | 14 |
|
| Capsular
infiltration |
|
|
| 0.255 |
|
Present | 62 | 34 | 28 |
|
|
Absent | 28 | 11 | 17 |
|
| Venous
infiltration |
|
|
| 0.033a |
|
Present | 38 | 14 | 24 |
|
|
Absent | 52 | 31 | 21 |
|
| AFP (ng/ml) |
|
|
| 0.809 |
|
<20 | 23 | 12 | 11 |
|
|
≥20 | 67 | 33 | 34 |
|
| HBsAg |
|
|
| 0.788 |
|
Positive | 73 | 36 | 37 |
|
|
Negative | 17 | 9 | 8 |
|
Cell culture and transfection
The human HCC cell lines (Hep3B and HuH-7) and the
immortalized human non-cancerous hepatic cell line THLE-3 were
obtained from the Chinese Academy of Sciences (Shanghai, China).
Human HCC cell lines (MHCC97-L, MHCC97-H and HCCLM3) were kindly
provided by Dr Qing-An Jia (Liver Cancer Institute, Zhongshan
Hospital, Fudan University, Shanghai, China). The cells were
cultured in Dulbecco's modified Eagle's medium (DMEM; HyClone;
Cytiva) supplemented with 10% fetal bovine serum (FBS; Gibco;
Thermo Fisher Scientific, Inc.) and 1% penicillin-streptomycin
(Sigma-Aldrich; Merck KGaA) in a humidified 5% CO2
incubator at 37°C.
Cell transfection was performed using
Lipofectamine® 2000 (Thermo Fisher Scientific, Inc.)
according to the manufacturer's instructions. miR-875-5p mimics
(HmiR-SN0806), miR-875-5p inhibitors (HmiR-AN0806-SN-10), miRNA
mimics negative control (CmiR-SN0001-SN), miRNA inhibitor negative
control (CmiR-AN0001-SN) and miR-875-5p clones in lentiviral
vectors (HmiR0504-MR04) were purchased from iGeneBio, Inc. Small
interfering RNA (siRNA) used for eIF3a silencing and negative
control (NC) siRNA were purchased from Biomics Biotechnologies Co.,
Ltd. Plasmids used for overexpressing eIF3a (M0274) and empty
vectors (EV) were purchased from GeneCopoeia, Inc.
RNA extraction and reverse
transcription-quantitative (RT-q) PCR
RNA was extracted from tissues and cells using
TRIzol® reagent (Invitrogen; Thermo Fisher Scientific,
Inc.) and Qiagen AllPrep DNA/RNA FFPE kit (cat. no. 80234)
according to the manufacturer's instructions. For the detection of
mRNA and miRNA expression, cDNA was synthesized using a cDNA
Synthesis kit (Thermo Fisher Scientific, Inc.). qPCR was performed
with SYBR®-Green Premix PCR Master Mix (Roche
Diagnostics GmbH). miR-875-5p primers (MQPS0002239-1-200) and eIF3a
primers were purchased from Guangzhou RiboBio Co., Ltd. U6 snRNA
and GAPDH were used as normalization controls. The primer sequences
for eIF3a were: 5′-ACAGGCAGTGTTTGGACCTTC-3′ (forward) and
5′-CTTACGCGTGTATTGGAGGCA-3′ (reverse). The primer sequences for
GAPDH were: 5′-GGTATGACAACGAATTTGGC-3′ (forward) and
5′-GAGCACAGGGTACTTTATTG-3′ (reverse).
Cell proliferation assays
Cell Counting Kit-8 (CCK-8; Dojindo Molecular
Technologies, Inc.) assay was used to assess cell viability. Hep3B
and HCCLM3 cells were seeded in a 96-well plate (3×103
cells/well); 10 µl CCK-8 solution was added to each well at 0, 24,
48 and 72 h and incubated at 37°C for 1 h. A microplate reader
(Bio-Rad Laboratories, Inc.) was used to read the absorbance at 450
nm.
An EdU kit (cat. no. C10310-1; Guangzhou RioBio Co.,
Ltd.) was used to detect cell proliferation. HCC cells were seeded
in a 24-well plate (5×104 cells/well). Following 4-h
incubation with EdU solution, the nuclei were stained with DAPI.
Images were captured under a Zeiss fluorescence photomicroscope
(Carl Zeiss AG) in at least five random fields for
quantification.
Transwell invasion and migration
assays
Serum-free DMEM containing cells pre-starved for 12
h were added into the upper chambers of the Transwell inserts
(5×104 cells/well) with or without pre-coating with
Matrigel, and DMEM with 10% FBS was added into the lower chambers.
Following 24-h incubation, the remaining cells in the upper chamber
were removed, and the invaded or migrated cells were fixed with
formalin and stained using crystal violet for 20 min. Cells from at
least five random fields were counted under a light microscope with
100-fold magnification.
In vivo experiments
Male BALB/c nude mice were housed under
pathogen-free conditions in the Centre of Laboratory Animals at The
Medical College of Xian Jiaotong University. Animal experiments
were performed according to the protocol approved by the Ethics
Review Committee of Xian Jiaotong University. Humane endpoints
included i) the body condition score was 1/5; ii) the body
condition score was 2/5 and the mouse was profoundly lethargic;
iii) the tumor affected the mouse's gait or normal posture, ability
to eat, urinate, or defecate. No mice met these criteria and were
euthanized before the end of the experiment.
Mice (4–6 weeks old) were randomly grouped for
animal experiments (n=4 mice per group) for animal experiments. The
mice were housed with filtered air, 12-h light/dark cycle, constant
temperature (25°C) and had free access to food and water. A
subcutaneous xenograft model was established for evaluating the
tumor growth of HCC cells. HCCLM3 cells (3×106)
transfected with miR-875-5p clones in lentiviral vectors or control
vectors were suspended in 100 µl PBS and inoculated subcutaneously
into the flank of the mice. Tumor volumes were determined every 3
days as length × width × width/2. The mice were sacrificed by
cervical dislocation under 10% ether inhalant anesthesia at 3 weeks
after implantation, and the xenograft tumor tissues were dissected
for further examination. The pulmonary metastatic model was
established to investigate the metastatic ability of HCC cells;
transfected HCCLM3 cells (1×106) were injected into the
tail vein. Mice were sacrificed at 10 weeks after injection, and
the lung tissues were examined microscopically following
hematoxylin and eosin (H&E) staining.
Bioinformatics analysis
The microRNA.org
website (2010 version; http://www.microrna.org/microrna/home.do) and
TargetScan Human 5.1 (http://www.targetscan.org/vert_72/) were used by
entering ‘miR-875-5p’ into the search box to predict potential
miRNA target genes and the binding sites.
Immunohistochemical (IHC)
staining
IHC was performed as previously described (23). Briefly, formalin-fixed
paraffin-embedded sections were dewaxed, dehydrated and rehydrated.
Antibodies against Ki-67 (cat. no. ab92742; Abcam), which is the
proliferation marker, were added to the sections and incubated at
4°C overnight, followed by the addition of the streptavidin
peroxidase-conjugated secondary antibody (cat. no. SP-9001; OriGene
Technologies, Inc.). The slides were counterstained with
hematoxylin and inspected under a microscope.
Luciferase reporter assay
The 3′UTR sequence of eIF3a mRNA and the
corresponding sequence with mutations in the predicted miR-875-5p
target sites were synthesized and inserted into pGL3 vectors
(Promega Corporation) to obtain eIF3a 3′UTR wild-type (wt) and
eIF3a 3′UTR mutant (mt), respectively. Subsequently, Hep3B or
HCCLM3 cells were seeded into a 24-well plate and transfected with
different combinations of miR-875-5p mimics or inhibitors and eIF3a
3′UTR wt or mt plasmids followed by 48-h cultivation. The relative
luciferase activities were quantified using the Dual-Luciferase
Reporter Assay system (Promega Corporation) and normalized to
Renilla luciferase activity.
Western blotting
Proteins were lysed from HCC cells using RIPA buffer
(Beyotime Institute of Biotechnology) supplemented with PMSF and
protease inhibitors (Beyotime Institute of Biotechnology). The
protein concentration was determined by the BCA Protein assay kit
(Beyotime Institute of Biotechnology) according to the
manufacturer's instructions. Cell lysates with 15 µg protein per
lane were separated by SDS-polyacrylamide gel electrophoresis and
transferred onto polyvinylidene difluoride membranes. Following
blocking with 10% non-fat milk, the membranes were probed with
primary antibodies overnight at 4°C and the corresponding secondary
antibodies for 1 h at room temperature. Finally, the ECL reagent
(Beyotime Institute of Biotechnology) was used for signal
detection. The antibodies against eIF3a (cat. no. ab128996) were
purchased from Abcam, Inc. The antibodies against E-cadherin (cat.
no. 3195S), N-cadherin (cat. no. 13116S) and vimentin (cat. no.
5741S) were purchased from Cell Signaling Technology, Inc. The
antibody against β-actin (cat. no. sc-47778) was purchased from
Santa Cruz Biotechnology, Inc.
Statistical analysis
Data are presented as the mean ± SD. GraphPad Prism
software version 8.0 (GraphPad Software, Inc.) was used for
statistical analysis. Data were compared using a two-tailed
Student's t-test and ANOVA with Dunnett-t test and Newman-Keuls
test. The paired t-test was used to compare tumor and adjacent
non-tumor samples of the same individuals. The overall survival
(OS) between two groups was analyzed using Kaplan-Meier curves and
log-rank analysis. The association between miR-875-5p expression
and clinicopathological features was analyzed using the Chi-squared
test and Fisher's exact test. Pearson's correlation analysis was
performed to determine the correlation between miR-875-5p and eIF3a
mRNA expression. P<0.05 was considered to indicate a
statistically significant difference.
Results
miR-875-5p is downregulated in HCC and
associated with the progression and survival of HCC
To determine whether miR-875-5p was dysregulated in
HCC, RT-qPCR was performed in 90 pairs of tumor and adjacent
non-tumor tissues. The results revealed that miR-875-5p expression
was significantly downregulated in HCC tissues compared with that
in the adjacent non-tumor tissues (P<0.001; Fig. 1A). Subsequently, the patients were
divided into two groups according to the median value of miR-875-5p
expression in HCC tissues, the high miR-875-5p group (n=45) and the
low miR-875-5p group (n=45). The Chi-square test results verified
that low expression of miR-875-5p was associated with a larger
tumor size (P=0.006), venous infiltration (P=0.033) and advanced
TNM stage (P=0.016) (Table I).
Kaplan-Meier and log-rank analysis further revealed that patients
with HCC with low miR-875-5p expression presented with unfavorable
OS (P<0.05, Fig. 1B). Consistent
with the results from the HCC tissue samples, RT-qPCR results
demonstrated that miR-875-5p expression levels were downregulated
in HCC cell lines compared with those in the immortalized hepatic
cell line THLE-3 (P<0.05 or P<0.01, respectively; Fig. 1C). Thus, the above results suggested
that miR-875-5p was downregulated in HCC tissues and cell lines,
and that low miR-875-5p expression was associated with tumor
progression and poor OS of patients with HCC.
miR-875-5p suppresses HCC cell
proliferation
To determine the effects of miR-875-5p on HCC cell
proliferation, we transfected miR-875-5p inhibitors in Hep3B cells
which exhibited relatively high endogenous miR-875-5p level and
overexpressed miR-875-5p in HCCLM3 cells which exhibited relatively
low endogenous miR-875-5p to obtain satisfactory transfection
efficiency and obvious biological effects. As determined by
RT-qPCR, the expression of miR-875-5p was decreased in Hep3B cells
(P<0.01; Fig. 2A) transfected
with the miR-875-5p inhibitors compared with that in the negative
control group and upregulated in HCCLM3 cells (P<0.01; Fig. 2B) transfected with the miR-875-5p
mimics compared with that in the control group. CCK-8 assay results
revealed that the miR-875-5p inhibitors enhanced the viability of
Hep3B cells (P<0.05; Fig. 2C),
whereas the viability of HCCLM3 cells transfected with the
miR-875-5p mimics was significantly reduced (P<0.05; Fig. 2D). In addition, an EdU assay was
performed, and the results demonstrated that the miR-875-5p
inhibitors accelerated the proliferation of Hep3B cells (P<0.05;
Fig. 2E), whereas the miR-875-5p
mimics inhibited the proliferation of HCCLM3 cells (P<0.05;
Fig. 2F). Thus, these results
suggested that miR-875-5p suppressed HCC cell proliferation.
miR-875-5p inhibits HCC cell migration
and invasion
To determine whether miR-875-5p served a role in the
motility of HCC cells, Transwell assays were performed. The results
demonstrated that Hep3B cells transfected with the miR-875-5p
inhibitors exhibited increased migratory and invasive abilities
(P<0.05; Fig. 3A), whereas the
miR-875-5p mimics reduced the number of migrated and invasive
HCCLM3 cells (P<0.05; Fig. 3B).
As epithelial-mesenchymal transition (EMT) is a classical
phenomenon of morphology change in HCC cells and serves a pivotal
role on HCC cell migration and invasion, the present study further
investigated whether miR-875-5p inhibited HCC cell motility via
suppressing the EMT progression. Western blot analysis revealed
that the miR-875-5p inhibitors decreased the expression levels of
E-cadherin and increased those of vimentin and N-cadherin in Hep3B
cells (P<0.05; Fig. 3C). By
contrast, the miR-875-5p mimics increased the expression levels of
E-cadherin and decreased those of vimentin and N-cadherin in HCCLM3
cells (P<0.05; Fig. 3D).
Therefore, these results indicated that miR-875-5p inhibited HCC
cell migration and invasion by suppressing the EMT.
miR-875-5p inhibits HCC tumor growth
and metastasis in vivo
To further confirm the inhibitory effects of
miR-875-5p on HCC in vivo, HCCLM3 cells stably
overexpressing miR-875-5p were established and injected
subcutaneously into nude mice. We found that the miR-875-5p had no
detectable effect on the body weight in mice for xenograft model
(Fig. S1A). The tumor growth
curves revealed that miR-875-5p overexpression induced HCC growth
restriction in mice (P<0.05; Fig.
4A). The weight of the tumors formed by
miR-875-5p-overexpressing HCCLM3 cells was decreased (P<0.05;
Fig. 4B). RT-qPCR results confirmed
higher miR-875-5p expression in tumor tissues harvested from the
miR-875-5p-overexpressing group compared with those from the
control group (P<0.05; Fig. 4C).
Immunohistochemistry results demonstrated that overexpression of
miR-875-5p decreased the positive rate of the proliferation marker
Ki-67 staining (P<0.05; Fig.
4D). Additionally, to determine the metastatic potential in
vivo, a lung metastasis model was established by tail vein
injection with HCCLM3 cells overexpressing miR-875-5p. The weight
loss was lower in the mice injected with miR-875-5p-overexpressing
cells compared with that with control, while the difference was not
significant (Fig. S1B). RT-qPCR
results validated that miR-875-5p expression level in HCCLM3 cells
to be injected into the tail vein and in the metastatic nodules was
higher in miR-875-5p-overexpressing group compared with that in
control (P<0.05; Fig. S1C and
D). The results revealed that the miR-875-5p overexpression
group exhibited fewer and smaller foci in the lungs of nude mice
compared with those in the control group (P<0.05; Fig. 4E). Together, these results suggested
that miR-875-5p inhibited HCC growth and metastasis in mice.
eIF3a is the downstream target of
miR-875-5p in HCC
To investigate the exact mechanism underlying the
inhibitory function of miR-875-5p in HCC, the candidate downstream
targets of miR-875-5p were identified using bioinformatics tools
(microRNA.org, TargetScan). Comprehensive analysis
of a previous study identified eIF3a as an oncogenic molecule in
HCC (22), and bioinformatics
analysis predicted miR-875-5p binding sites in the eIF3a mRNA 3′UTR
(Fig. 5A); thus, eIF3a was selected
as the potential target of miR-875-5p. A luciferase reporter assay
was performed, and the results demonstrated that the miR-875-5p
inhibitors enhanced, whereas the mimics reduced the luciferase
activities of plasmids carrying the wt, but not the mt eIF3a 3′UTR
(Fig. 5B). In addition, RT-qPCR and
western blotting results demonstrated that the miR-875-5p
inhibitors increased the expression of eIF3a at the mRNA and
protein level in Hep3B cells (P<0.05; Fig. 5C and D). By contrast, the miR-875-5p
mimics decreased eIF3a mRNA and protein expression in HCCLM3 cells
(P<0.05; Fig. 5E and F).
Consistently, RT-qPCR results revealed that eIF3a mRNA expression
was significantly upregulated in HCC tissues compared with that in
adjacent non-tumor tissues (P<0.001; Fig. 5G), and eIF3a mRNA expression was
significantly negatively correlated with miR-875-5p expression in
HCC tissues (r=−0.4105, P<0.0001; Fig. 5H). Western blotting results
demonstrated that eIF3a expression was lower in HCC tissues with
high miR-875-5p expression compared with those with low miR-875-5p
expression (P<0.05; Fig. 5I).
Additionally, the protein expression of eIF3a was downregulated in
miR-875-5p-overexpressing xenograft tumor tissues compared with
that in the control group (P<0.05; Fig. 5J). Collectively, these results
confirmed that miR-875-5p downregulated eIF3a expression by
directly targeting the 3′UTR of eIF3a mRNA in HCC.
eIF3a-knockdown inhibits HCC cell
proliferation, migration and invasion
To determine the role of eIF3a in the proliferation
and motility of HCC cells, HCCLM3 cells were used to establish an
eIF3a-knockdown cell line. Western blotting was performed to
validate the transfection efficiency (P<0.05; Fig. 6A). CCK-8 assay results revealed that
eIF3a-knockdown significantly decreased the viability of HCCLM3
cells (P<0.05; Fig. 6B). In
addition, eIF3a-knockdown reduced the number of active
proliferating HCC cells as determined by the EdU assay (P<0.05;
Fig. 6C). To explore the effects of
eIF3a on the migratory and invasive abilities of HCC cells,
Transwell assays were performed, and the results demonstrated that
knockdown of eIF3a decreased the migration and invasion of HCCLM3
cells (P<0.05; Fig. 6D). In
addition, eIF3a-knockdown inhibited the EMT progression in HCCLM3
cells (P<0.05; Fig. 6E). Taken
together, these results suggested that eIF3a promoted the
proliferation and motility of HCC cells.
eIF3a mediates the inhibitory effects
of miR-875-5p on HCC cell proliferation and mobility
Based on the functions of miR-875-5p and eIF3a, and
the correlation between them, the present study further examined
whether eIF3a may mediate the inhibitory functions of miR-875-5p in
HCC. RT-qPCR results revealed that miR-875-5p expression was
significantly upregulated by miR-875-5p mimics but not affected by
eIF3a overexpression plasmid in HCCLM3 cells (P<0.05; Fig. S1E). Western blot analysis validated
that eIF3a expression was significantly restored by an eIF3a
overexpression plasmid in miR-875-5p-overexpressing HCCLM3 cells
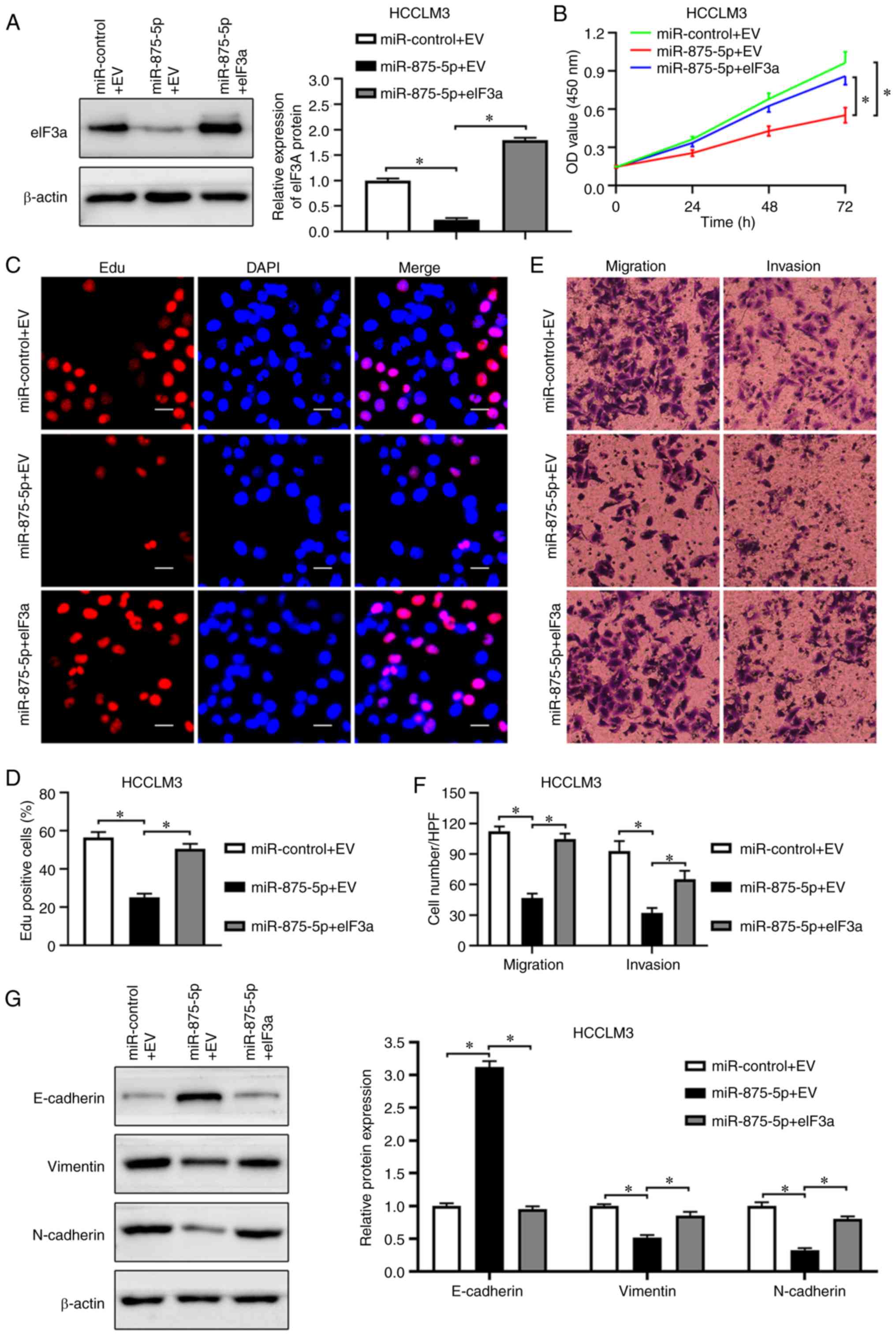
(P<0.05; Fig. 7A). Consistent
with the promoting effects of eIF3a on HCC cell proliferation,
eIF3a accelerated the proliferation of HCCLM3 cells inhibited by
miR-875-5p, determined by the CCK-8 and EdU assays (P<0.05;
Fig. 7B-D). The Transwell assay
results demonstrated that eIF3a overexpression reversed the
inhibitory effects of miR-875-5p on HCCLM3 cell migration and
invasion (P<0.05; Fig. 7E and
F). In addition, eIF3a overexpression rescued the miR-875-5p
mimic-inhibited EMT progression of HCCLM3 cells (P<0.05;
Fig. 7G). In summary, these results
supported the role of eIF3a in mediating the tumor suppressor
function of miR-875-5p in HCC cells.
Discussion
miRNAs are small non-coding RNAs that serve pivotal
roles in the majority of types of cancer, affecting numerous
cancer-associated processes, such as cell proliferation, cell
cycle, apoptosis, differentiation, migration and metabolism
(24–25). Previous studies have observed the
dysregulation of miR-875-5p in various types of cancer (13–15).
For instance, miR-875-5p acts as a tumor suppressor by curbing the
epidermal growth factor receptor (EGFR)-ZEB1 axis, repressing the
EMT and increasing radiation response in prostate cancer (14). In addition, miR-875-5p promotes the
proliferation and motility of non-small cell lung cancer cell lines
by targeting SATB homeobox 2 (15).
By contrast, miR-875-5p exerts a tumor suppressor role by
inhibiting cell proliferation and metastasis and accelerating
apoptosis via targeting EGFR in colorectal carcinoma (13). To determine the role of miR-875-5p
in HCC, which remains largely elusive, the present study
demonstrated that miR-875-5p was downregulated in HCC, and low
expression of miR-875-5p was significantly associated with an
unfavorable prognosis and clinical features including large tumor
size, venous infiltration and an advanced TNM stage. Consistent
with the clinical analysis, the results of the loss- and
gain-of-function experiments further revealed that miR-875-5p
suppressed the proliferation, migration and invasion of HCC cells.
Additionally, in vivo experiments demonstrated that
miR-875-5p overexpression inhibited tumor growth and metastasis. In
a previous study, a hypoxic microenvironment was demonstrated to
modulate the expression levels of miR-187-3p, miR-204, miR-1296,
miR-671-5p (26–29) and long non-coding RNA AGAP2
antisense RNA 1 (30), which
sponges miR-16-5p and promotes cell proliferation and metastasis in
HCC. Therefore, whether hypoxia is responsive for the
downregulation of miR-875-5p in HCC requires further
investigation.
As the crucial component for translation initiation,
eIF3a serves a vital role in various physiological and pathological
processes, such as the cell cycle and DNA synthesis (31–33).
Regarding its role in cancer, eIF3a has been reported to be
involved in decreasing the expression of DNA repair proteins,
resulting in enhanced chemotherapeutic sensitivity in lung cancer
(16). Consistently, eIF3a
negatively regulates the resistance to cisplatin via suppressing
the cellular synthesis and activity of nucleotide excision repair
proteins in nasopharyngeal carcinoma (34). In addition, eIF3a exerts an
oncogenic role by accelerating cell proliferation and inhibiting
apoptosis in ameloblastoma (21).
Accumulating evidence has demonstrated that eIF3a participates in
the development of ovarian, urinary bladder and pancreatic cancer,
as well as HCC (20,22,35,36).
eIF3a is upregulated in HCC and facilitates the translation of
HIF-1α, which, in turn, regulates glycolytic metabolism (22). Additionally, a serum anti-eIF3a
autoantibody has been identified as a potential diagnostic
biomarker for HCC, further supporting the role of eIF3a in HCC
(37). However, the mechanism
underlying the upregulation of eIF3a and the functions of eIF3a on
the proliferation and metastasis in HCC remains unclear. In the
present study, eIF3a expression levels were upregulated and
negatively correlated with those of miR-875-5p in HCC tissues.
Knockdown of eIF3a inhibited the proliferation, migration and
invasion of HCC cells. Additionally, the results of the present
study demonstrated that eIF3a was a downstream target of miR-875-5p
in HCC. Firstly, miR-875-5p negatively modulated the luciferase
activity of reporter vectors carrying wild-type, but not mutant
3′UTR of eIF3a. Secondly, altering miR-875-5p expression negatively
regulated the mRNA and protein expression of eIF3a in HCC cells.
Lastly, overexpression of eIF3a reversed the suppressive effects of
miR-875-5p on HCC cell proliferation and mobility. Therefore, the
present study confirmed that miR-875-5p served a tumor suppressor
role by downregulating eIF3a in HCC. The majority of HCC cases
arise from liver fibrosis or cirrhosis (38). Of note, eIF3a has been reported to
be involved in the fibrosis of various organs, including the lung,
kidney, heart, skin and liver (39–43).
The aforementioned studies demonstrated that eIF3a, the expression
of which is upregulated by the transforming growth factor β1
(TGF-β1)/smad3 signaling pathway, mediated the TGF-β1-induced
fibrosis. Thus, further studies are required to determine whether
liver fibrosis participates in the eIF3a-induced tumorigenesis in
HCC.
In summary, the downregulation of miR-875-5p serves
a crucial role in tumor growth and metastasis in HCC and may be a
valuable prognostic marker and potential therapeutic target for
HCC.
Supplementary Material
Supporting Data
Acknowledgements
We would like to thank Dr Qing-An Jia (Liver Cancer
Institute, Zhongshan Hospital, Fudan University, Shanghai, P.R.
China) for kindly providing Human HCC cell lines (MHCC-97L,
MHCC-97H, HCCLM3).
Funding
This study was supported by grants from the National
Natural Science Foundation of China (grant no. 81874069).
Availability of data and materials
The analyzed data sets generated during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
TC contributed to writing the manuscript, study
conception and design, and collection and analysis of data. LS, BY,
and LW collected and interpreted data. YW, YN, RL, HM contributed
to data analysis and interpretation and drafting the manuscript. ZL
and KT contributed to study conception and revised the manuscript.
QL contributed to study conception and design as well as revising
and approving the final version of the manuscript. All authors have
read and approved the final version of this manuscript.
Ethics approval and consent to
participate
All procedures involving human participants were in
accordance with the ethical standards of the Research Ethics
Committee of The First Affiliated Hospital of Xian Jiaotong
University and with the Declaration of Helsinki as revised in 2013.
Written informed consent to participate in the study was obtained
from patients with HCC prior to sample collection. Animal
experiments were performed according to the protocol approved by
the Ethics Review Committee of Xian Jiaotong University.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Forner A, Reig M and Bruix J:
Hepatocellular carcinoma. Lancet. 391:1301–1314. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chan AWH, Zhong J, Berhane S, Toyoda H,
Cucchetti A, Shi K, Tada T, Chong CCN, Xiang BD, Li LQ, et al:
Development of pre and post-operative models to predict early
recurrence of hepatocellular carcinoma after surgical resection. J
Hepatol. 69:1284–1293. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Di Leva G, Garofalo M and Croce CM:
MicroRNAs in cancer. Annu Rev Pathol. 9:287–314. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bartel DP: MicroRNAs: Genomics,
biogenesis, mechanism, and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lin S and Gregory RI: MicroRNA biogenesis
pathways in cancer. Nat Rev Cancer. 15:321–333. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Giordano S and Columbano A: MicroRNAs: New
tools for diagnosis, prognosis, and therapy in hepatocellular
carcinoma? Hepatology. 57:840–847. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang L, Mo H, Jiang Y, Wang Y, Sun L, Yao
B, Chen T, Liu R, Li Q, Liu Q and Yin G: MicroRNA-519c-3p promotes
tumor growth and metastasis of hepatocellular carcinoma by
targeting BTG3. Biomed Pharmacother. 118:1092672019. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Xu Q, Zhu Q, Zhou Z, Wang Y, Liu X, Yin G,
Tong X and Tu K: MicroRNA-876-5p inhibits epithelial-mesenchymal
transition and metastasis of hepatocellular carcinoma by targeting
BCL6 corepressor like 1. Biomed Pharmacother. 103:645–652. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang Y, Yang Z, Wang L, Sun L, Liu Z, Li
Q, Yao B, Chen T, Wang C, Yang W, et al: MiR-532-3p promotes
hepatocellular carcinoma progression by targeting PTPRT. Biomed
Pharmacother. 109:991–999. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Liu Z, Wang Y, Dou C, Sun L, Li Q, Wang L,
Xu Q, Yang W, Liu Q and Tu K: MicroRNA-1468 promotes tumor
progression by activating PPAR-ү-mediated AKT signaling in human
hepatocellular carcinoma. J Exp Clin Cancer Res. 37:492018.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yao B, Li Y, Wang L, Chen T, Niu Y, Liu Q
and Liu Z: MicroRNA-3194-3p inhibits metastasis and
epithelial-mesenchymal transition of hepatocellular carcinoma by
decreasing Wnt/β-catenin signaling through targeting BCL9. Artif
Cells Nanomed Biotechnol. 47:3885–3895. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhang T, Cai X, Li Q, Xue P, Chen Z, Dong
X and Xue Y: Hsa-miR-875-5p exerts tumor suppressor function
through down-regulation of EGFR in colorectal carcinoma (CRC).
Oncotarget. 7:42225–42240. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
El Bezawy R, Cominetti D, Fenderico N,
Zuco V, Beretta GL, Dugo M, Arrighetti N, Stucchi C, Rancati T,
Valdagni R, et al: MiR-875-5p counteracts epithelial-to-mesenchymal
transition and enhances radiation response in prostate cancer
through repression of the EGFR-ZEB1 axis. Cancer Lett. 395:53–62.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang J, Lu Y, Ding H, Gu T, Gong C, Sun J,
Zhang Z, Zhao Y and Ma C: The miR-875-5p inhibits SATB2 to promote
the invasion of lung cancer cells. Gene. 644:13–19. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yin JY, Shen J, Dong ZZ, Huang Q, Zhong
MZ, Feng DY, Zhou HH, Zhang JT and Liu ZQ: Effect of eIF3a on
response of lung cancer patients to platinum-based chemotherapy by
regulating DNA repair. Clin Cancer Res. 17:4600–4609. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Dong Z and Zhang JT: Initiation factor
eIF3 and regulation of mRNA translation, cell growth, and cancer.
Crit Rev Oncol Hematol. 59:169–180. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yin JY, Zhang JT, Zhang W, Zhou HH and Liu
ZQ: eIF3a: A new anticancer drug target in the eIF family. Cancer
Lett. 412:81–87. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Fang C, Chen YX, Wu NY, Yin JY, Li XP,
Huang HS, Zhang W, Zhou HH and Liu ZQ: MiR-488 inhibits
proliferation and cisplatin sensibility in non-small-cell lung
cancer (NSCLC) cells by activating the eIF3a-mediated NER signaling
pathway. Sci Rep. 7:403842017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wang SQ, Liu Y, Yao MY and Jin J:
Eukaryotic translation initiation factor 3a (eIF3a) promotes cell
proliferation and motility in pancreatic cancer. J Korean Med Sci.
31:1586–1594. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ding Z, Liu J, Wang J, Huang B and Zhong
M: Upregulation of eukaryotic translation initiation factor 3
subunit a promotes cell survival in ameloblastoma. Oral Surg Oral
Med Oral Pathol Oral Radiol. 128:146–153. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Miao B, Wei C, Qiao Z, Han W, Chai X, Lu
J, Gao C, Dong R, Gao D, Huang C, et al: eIF3a mediates
HIF1α-dependent glycolytic metabolism in hepatocellular carcinoma
cells through translational regulation. Am J Cancer Res.
9:1079–1090. 2019.PubMed/NCBI
|
|
23
|
Liu Z, Dou C, Jia Y, Li Q, Zheng X, Yao Y,
Liu Q and Song T: RIG-I suppresses the migration and invasion of
hepatocellular carcinoma cells by regulating MMP9. Int J Oncol.
46:1710–1720. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Jansson MD and Lund AH: MicroRNA and
cancer. Mol Oncol. 6:590–610. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Farazi TA, Hoell JI, Morozov P and Tuschl
T: MicroRNAs in human cancer. Adv Exp Med Biol. 774:1–20. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Dou C, Zhou Z, Xu Q, Liu Z, Zeng Y, Wang
Y, Li Q, Wang L, Yang W, Liu Q and Tu K: Hypoxia-induced TUFT1
promotes the growth and metastasis of hepatocellular carcinoma by
activating the Ca2+/PI3K/AKT pathway. Oncogene.
38:1239–1255. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Liu Z, Wang Y, Dou C, Xu M, Sun L, Wang L,
Yao B, Li Q, Yang W, Tu K and Liu Q: Hypoxia-induced up-regulation
of VASP promotes invasiveness and metastasis of hepatocellular
carcinoma. Theranostics. 8:4649–4663. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Xu Q, Liu X, Liu Z, Zhou Z, Wang Y, Tu J,
Li L, Bao H, Yang L and Tu K: MicroRNA-1296 inhibits metastasis and
epithelial-mesenchymal transition of hepatocellular carcinoma by
targeting SRPK1-mediated PI3K/AKT pathway. Mol Cancer. 16:1032017.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Dou C, Liu Z, Xu M, Jia Y, Wang Y, Li Q,
Yang W, Zheng X, Tu K and Liu Q: MiR-187-3p inhibits the metastasis
and epithelial-mesenchymal transition of hepatocellular carcinoma
by targeting S100A4. Cancer Lett. 381:380–390. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Liu Z, Wang Y, Wang L, Yao B, Sun L, Liu
R, Chen T, Niu Y, Tu K and Liu Q: Long non-coding RNA AGAP2-AS1,
functioning as a competitive endogenous RNA, upregulates ANXA11
expression by sponging miR-16-5p and promotes proliferation and
metastasis in hepatocellular carcinoma. J Exp Clin Cancer Res.
38:1942019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Dong Z and Zhang JT: EIF3 p170, a mediator
of mimosine effect on protein synthesis and cell cycle progression.
Mol Biol Cell. 14:3942–3951. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Xu TR, Lu RF, Romano D, Pitt A, Houslay
MD, Milligan G and Kolch W: Eukaryotic translation initiation
factor 3, subunit a, regulates the extracellular signal-regulated
kinase pathway. Mol Cell Biol. 32:88–95. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Dong Z, Liu LH, Han B, Pincheira R and
Zhang JT: Role of eIF3 p170 in controlling synthesis of
ribonucleotide reductase M2 and cell growth. Oncogene.
23:3790–3801. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Liu RY, Dong Z, Liu J, Yin JY, Zhou L, Wu
X, Yang Y, Mo W, Huang W, Khoo SK, et al: Role of eIF3a in
regulating cisplatin sensitivity and in translational control of
nucleotide excision repair of nasopharyngeal carcinoma. Oncogene.
30:4814–4823. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhang Y, Yu JJ, Tian Y, Li ZZ, Zhang CY,
Zhang SF, Cao LQ, Zhang Y, Qian CY, Zhang W, et al: eIF3a improve
cisplatin sensitivity in ovarian cancer by regulating XPC and
p27Kip1 translation. Oncotarget. 6:25441–25451. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Spilka R, Ernst C, Bergler H, Rainer J,
Flechsig S, Vogetseder A, Lederer E, Benesch M, Brunner A, Geley S,
et al: eIF3a is over-expressed in urinary bladder cancer and
influences its phenotype independent of translation initiation.
Cell Oncol (Dordr). 37:253–267. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Heo CK, Hwang HM, Lee HJ, Kwak SS, Yoo JS,
Yu DY, Lim KJ, Lee S and Cho EW: Serum anti-EIF3A autoantibody as a
potential diagnostic marker for hepatocellular carcinoma. Sci Rep.
9:110592019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Fattovich G, Stroffolini T, Zagni I and
Donato F: Hepatocellular carcinoma in cirrhosis: Incidence and risk
factors. Gastroenterology. 127 (5 Suppl 1):S35–S50. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
He P, Yu ZJ, Sun CY, Jiao SJ and Jiang HQ:
Knockdown of eIF3a attenuates the pro-fibrogenic response of
hepatic stellate cells induced by TGF-β1. Cell Mol Biol
(Noisy-le-grand). 62:107–111. 2016.PubMed/NCBI
|
|
40
|
Zhang YF, Wang Q, Luo J, Yang S, Wang JL
and Li HY: Knockdown of elF3a inhibits collagen synthesis in renal
fibroblasts via Inhibition of transforming growth factor-β1/Smad
signaling pathway. Int J Clin Exp Pathol. 8:8983–8989.
2015.PubMed/NCBI
|
|
41
|
Li T and Zhao J: Knockdown of elF3a
inhibits TGF-β1-induced extracellular matrix protein expression in
keloid fibroblasts. Mol Med Rep. 17:4057–4061. 2018.PubMed/NCBI
|
|
42
|
Li XW, Wu YH, Li XH, Li D, Du J, Hu CP and
Li YJ: Role of eukaryotic translation initiation factor 3a in
bleomycin-induced pulmonary fibrosis. Eur J Pharmacol. 749:89–97.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Li WQ, Li XH, Wu YH, Du J, Wang AP, Li D
and Li YJ: Role of eukaryotic translation initiation factors 3a in
hypoxia-induced right ventricular remodeling of rats. Life Sci.
144:61–68. 2016. View Article : Google Scholar : PubMed/NCBI
|