Introduction
The constant increase in cancer incidence and the
failure of conventional chemotherapy to protect against advanced
cancer warrants the development of novel agents to treat and
prevent the malignancy. The search for successful anticancer agents
began decades ago and is ongoing (1). For many years, the cytotoxic actions
of the chemotherapeutic drugs were ascribed solely to their ability
to induce genotoxic death. There is evidence that insufficient
apoptosis has been associated with the development and progression
of tumors (2). There is also
accumulating evidence that many agents exert their cytotoxic
effects mainly by inducing apoptosis in tumor cells (3,4).
Currently, induction of apoptosis has become a useful marker for
screening compounds for subsequent development as possible
anticancer agents (5–7).
Natural medicine provides a rich pool of novel and
efficacious agents for cancer prevention and treatment; previous
research has resulted in the identification of several bioactive
components from natural products such as resveratrol, curcumin,
isothiocyanates, quercetin and polyphenols that selectively inhibit
the growth of malignant cells in vitro by inducing apoptosis
(8,9), and which have been used as cancer
chemopreventive agents (10).
Therefore, intensive efforts have been made to identify new
bioactive compounds from natural products, through isolation of
apoptosis-inducing agents and elucidation of the apoptosis
mechanisms.
Reineckia carnea, also known as ‘guanyin
cao’, one of the most popular traditional herbs in China, has been
used to prevent cough, eliminate phlegm, as well as to treat
rheumatism disease and hepatitis, for at least one thousand years
(11,12). Previous studies showed that many
bioactivity components including spirostanol sapogenin and
spirostanol saponins were found in Reineckia carnea
(13–15). It remains unclear whether
Reineckia carnea contains any active chemical components
with cytotoxic effects on cancer cells. RCE-4, a spirostanol
saponin, was first isolated from Reineckia carnea ethyl
acetate fraction, although this compound was firstly isolated from
Rohdea japonica by Miyahara (16). However, it is still uncertain
whether this kind of saponin has cytotoxic effects on cancer cells.
Therefore, in this study we evaluated the cytotoxicity of RCE-4 on
different cancer cell lines and further elucidated one of the
possible mechanisms underlying its cytotoxic action.
Materials and methods
Reagent and antibodies
RCE-4 (Fig. 1A)
was isolated from the whole plant of Reineckia carnea and
purified at the Hubei Key Laboratory of Natural Products Research
and Development (China Three Gorges University); it was dissolved
in DMSO at a stock concentration of 10 mM, and diluted to the
indicated concentration with RPMI-1640 medium. Antibodies against
Bax, Bcl-2, cytochrome c and HRP-labeled secondary
antibodies were purchased from Santa Cruz Biotechnology, Inc.
(Santa Cruz, CA, USA). Annexin V-FITC/PI apoptosis assay kit, Cell
Cycle assay kit and JC-1 mitochondrial membrane potential (Δψm)
assay kit were obtained from KeyGen Biotech Co., Ltd. (Nanjing,
China). The NE-PER nuclear and cytoplasmic extraction kit were
obtained from Thermo Scientific Pierce.
Cell lines
The cancer cell lines CaSki, HT-29, CNE-2 and the
normal cell lines Marc-145 and MDCK, were obtained from the
Shanghai Institute of Cell Biology, Chinese Academy of Science,
Shanghai, China. All cells were maintained in RPMI-1640 medium
supplemented with 10% fetal calf serum, 25 mM HEPES buffer, 2
mmol/l L-glutamine, 100 U/ml penicillin and 100 μg/ml streptomycin.
Cultures were incubated in a humidified atmosphere of 5%
CO2 at 37°C.
Cell viability assay
Cells (1×104/well) were seeded in
supplemented culture medium (100 μl/well) in a 96-well plate and
incubated for 24 h. Then the medium was replaced with a
drug-containing medium, and the cells were further incubated for 48
h. All experiments were run in parallel with controls (0.1% DMSO)
and the cell viabilities were evaluated by MTT assays. The
absorbance of formazan formed was measured at 570 nm by a
microplate reader. Each experiment was repeated at least 3
times.
Transmission electron microscopy
CaSki cells treated with DMSO or RCE-4 were
collected by trypsinization, washed twice with PBS, and then fixed
with 0.5 ml of ice-cold glutaraldehyde (2.5% in 0.1 cacodylate
buffer, pH 7.4) at 4°C overnight. The subsequent steps were
performed according to standard procedures, including fixing,
incubation, rinsing, gradient dehydration, embedding and ultrathin
sections. Ultrathin sections were doubly stained with uranyl
acetate and lead citrate and analyzed by transmission electron
microscopy (Hitachi H-7500).
Annexin V-FITC/PI cytometric
analysis
Early apoptosis was assessed by detecting surface
exposure of phosphatidylserine (PS) in cells using an Annexin
V-FITC/PI kit. Briefly, CaSki cells (2×105) were seeded
into a 100-ml culture flask and incubated for 12 h. Then, cells
were treated with RCE-4 of 2.5, 5 and 10 μM for 24 h. The cells
(both adherent and floating cells) were collected and treated
according to the manufacturer’s instructions. Finally, the cells
were analyzed with FITC/PI double-staining using a flow cytometer
(Beckman Coulter, USA) with the single beam at 488 nm
excitation.
Cell cycle analysis
To investigate the effect of RCE-4 on the cell cycle
distribution, CaSki cells (2×106) were subcultured into
culture flasks and treated with 2.5, 5 and 10 μM of RCE-4 for 24 h.
The cells were resuspended in 2 ml of 70% ice-cold ethanol solution
and fixed at 4°C overnight. After washing with PBS, the pellets
were resuspended in 100 mg/ml PI solution containing 100 mg/ml
RNase, and then incubated at 37°C for at least 30 min. The cellular
DNA content was then detected by flow cytometer.
Analysis of mitochondrial membrane
potential (JC-1 staining)
The change in mitochondrial membrane potential (Δψm)
was measured using a JC-1 fluorescent probe assay kit, according to
the kit’s instructions. Briefly, following RCE-4 treatment of 2.5,
5 and 10 μM for 24 h, the cells were washed with PBS and incubated
for 30 min with JC-1 at 37°C. After washing in PBS twice, the cells
were subjected to two-color analysis by a flow cytometer.
Real-time quantitative PCR (qPCR)
To examine the role of Bcl-2 family members in
RCE-4-induced apoptosis, we measured the gene expression of Bax and
Bcl-2 using qPCR. CaSki cells were treated with 10 μM of RCE-4 for
0, 6, 12 and 24 h in 6-well plates. Total cellular RNA was isolated
using the TRIzol Reagent. The qPCR reaction was carried out on the
LightCycler 2.0 instrument (software v4.0; Roche Applied Science)
using the double-stranded DNA dye SYBR-Green I strategy. The
oligonucleotide sequences of the PCR primers used herein were
designed based on the human mRNA encoding the respective genes.
Quantification was based on threshold cycle (Ct) difference
performed according to the ΔΔCt method, using the following
equation: expression ratio=2-ΔΔCt, where ΔΔCt = (Ct
target - Ct reference) time x - (Ct target - Ct reference) time 0.
The expression level of each target gene was normalized to that of
glyceraldehyde-3 phosphate dehydrogenase (GAPDH), which fulfils the
requirements for validation of reference genes.
Western blot analysis
CaSki cells (6×106) were treated with 10
μM RCE-4 for 0, 6, 12 and 24 h. The cells were harvested and washed
with cold PBS twice. Cell pellets were lysed in 40 μl lysis buffer
(20 mM HEPES/NaOH, pH 7.5, 250 mM sucrose, 10 mM KCl, 2 mM
MgCl2, 1 mM EDTA, 1 mM DTT, protease inhibitor cocktail)
for 20 min on ice. The lysis solution was centrifuged at 25,000 × g
for 10 min at 4°C and protein contents in the supernatant were
measured using a Bio-Rad DC protein assay kit (Bio-Rad, Hercules,
CA, USA). The protein expression of Bax, Bcl-2, and cytochrome
c were detected by western blot analysis. Briefly, equal
amounts of protein were electrophoresed on 12% SDS acrylamide.
Following electrophoresis, the proteins were transferred from the
gel to a PVDF membrane. Non-specific binding was blocked with 5%
skim milk in TBST buffer (5 mM Tris-HCl, pH 7.6, 136 mM NaCl, 0.05%
Tween-20) at 4°C overnight. Blots were incubated at 37°C for 4 h
each with primary and secondary antibody conjugated with peroxidase
(HRP)-labeled anti-rabbit/mouse IgG. Blots were developed with ECL
western blotting detection reagents (Multi Sciences). Experimental
values were normalized to β-actin reactivity.
Statistical analysis
Statistical analyses were performed using Prism
software (GraphPad, San Diego, CA, USA). Data are presented as the
means ± SEM. Data were analyzed by one-way ANOVA for multiple
comparisons. P<0.05 was considered to indicate statistically
significant differences.
Results
Chemical structure of RCE-4
The structure of RCE-4 was identified as
(1β,3β,5β,25S)-spirostan-1,3-diol1-[α-L-rhamnopyranosyl-(1→2)-β-D-xylopyranoside]
(Fig. 1A).
Effect of RCE-4 on tumor cell
viability
MTT assays were performed to investigate the effects
of RCE-4 on the proliferation of tumor and normal cells. After
treatment for 48 h with different concentrations, RCE-4 exhibited
the greatest growth inhibitory effect against CaSki cells, followed
by HT-29, CNE2 cells and much less cytotoxicity to Marc 145 and
MDCK normal cells (~6-fold higher IC50) (Fig. 1B). The IC50 values were
3.37, 4.31, 4.81, 18.73 and 19.75 μM for CaSki, HT-29, CNE2,
Marc-145 and MDCK, respectively. These results indicated that RCE-4
had a good growth-inhibitory effect on CaSki cells in a
dose-dependent manner and relatively high selectivity.
Apoptotic morphological changes in CaSki
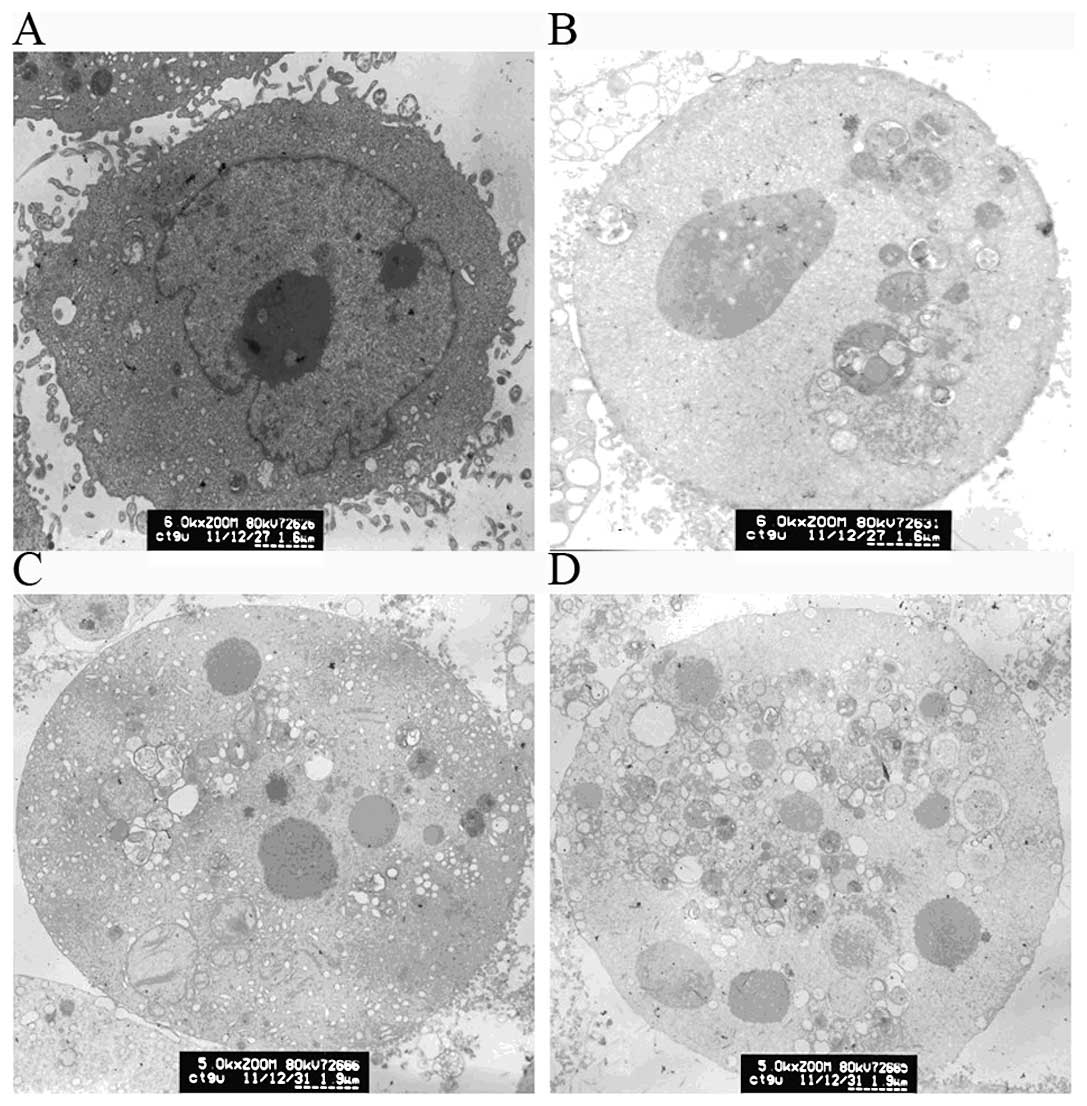
cells induced by RCE-4
To evaluate cellular ultrastructure for indications
of the mode of death, we compared RCE-4-treated and untreated CaSki
cells using transmission electron microscopy. Inspection of the
ultrastructural details revealed the presence of RCE-4-induced
apoptosis. CaSki control cells (Fig.
2A) showed a clear nucleus, nuclear membrane and nucleoli,
irregular cell surface with cells very densely packed together.
Following treatment with different concentrations of RCE-4 (2.5, 5
and 10 μM), cells exhibited pronounced morphological changes and
typical apoptosis features (Fig.
2B-D), including cell shrinkage, nuclear fragmentation, and
chromatin condensation. The nucleus appeared to have broken down
and their volume decreased. The nucleoli were also absent and no
subcellular organelles were observed. Furthermore, irregular cell
surface disappeared and a large number of vacuoles in the cytoplasm
was present.
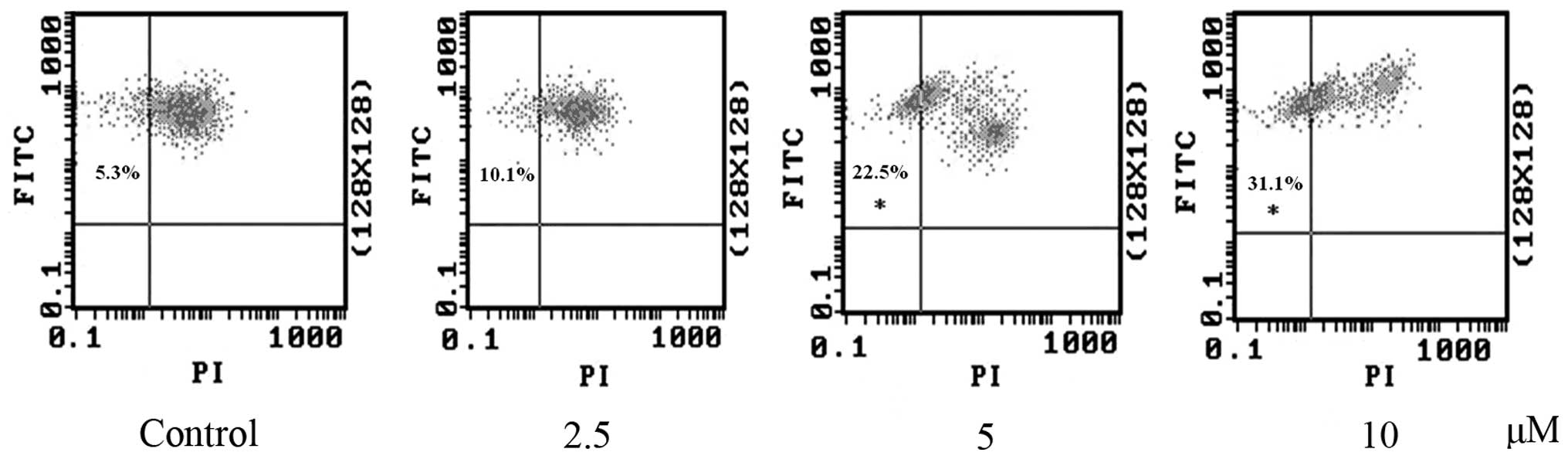
Effect of RCE-4 on apoptosis in CaSki
cells
To further differentiate between apoptosis or
necrosis, the cytotoxic effects of RCE-4 on CaSki cells were
evaluated using the early marker of apoptosis Annexin V, and the
dead cell marker PI. Numerous studies have reported that advanced
nuclear fragmentation is preceded by alteration in the plasma
membrane, such as PS externalization (17). Hence, cells treated with RCE-4 of
2.5, 5 and 10 μM for 24 h were double stained with Annexin
V-FITC/PI and analyzed by flow cytometry. Apoptotic cells were
determined by counting the percentage of early apoptosis in the
upper left quadrant (Annexin V+/PI-), and
late apoptosis in the upper right quadrant (Annexin
V+/PI+). Treatment with different doses of
RCE-4 (2.5, 5 and 10 μM) for 24 h resulted in separately 9.5, 23.3
and 26.3% early apoptosis compared with the control (0.8%)
(Fig. 3A). These results suggest
that RCE-4 can effectively induce apoptosis, and, particularly,
early apoptosis. The necrotic cell population (Annexin
V-/PI+) did not change apparently following
exposure to different concentrations of RCE-4, indicating that
apoptosis is the preferential cell death induced by RCE-4 in CaSki
cells.
Effect of RCE-4 on cell cycle
distribution
An experiment was performed to evaluate the effect
on the cell cycle phase distribution of CaSki cells after treatment
with 2.5, 5 and 10 μM of RCE-4. Results shown in Fig. 3B indicate that there was a
significant increase in the Sub-G1 DNA fraction (4.9, 7.5, 25.5 and
27.1%) at a concentration of 0-10 μM in a dose-depended manner,
which responded to apoptotic cells. Results of this experiment
demonstrated that RCE-4 arrested the cell cycle progression of
CaSki cells at S phase. Compared with the control (5.8%), RCE-4
(2.5, 5 and 10 μM) led to an S phase increase of 8.4, 11.3 and
12.5%, respectively.
RCE-4 decreases the Δψm in CaSki
cells
Apart form PS externalization, dissipation of Δψm
has also been reported to be an early apoptosis event in several
different systems. It is commonly used to detect mitochondrial
depolarization that occurs in early apoptosis (18). To signal the loss of Δψm, JC-1
probe was applied to test the occurrence of apoptosis. As shown in
Fig. 4, the majority of the
untreated cells were identified in the upper right quadrant
(FITC+/PI+). This corresponded to
mitochondria with a polarized Δψm. However, following treatment
with different concentrations of RCE-4 for 24 h, cells moved
towards the upper/left region (FITC+/PI-) and
Δψm began to decrease, suggesting disruption of mitochondrial
function. As can be seen in Fig.
4, RCE-4 treatment significantly decreased the Δψm in CaSki
cells, compared with the control. These results were
dose-dependent. These findings demonstrated that the RCE-4-induced
apoptosis in CaSki cells involved mitochondria dysfunction
associated with dissipation of the Δψm.
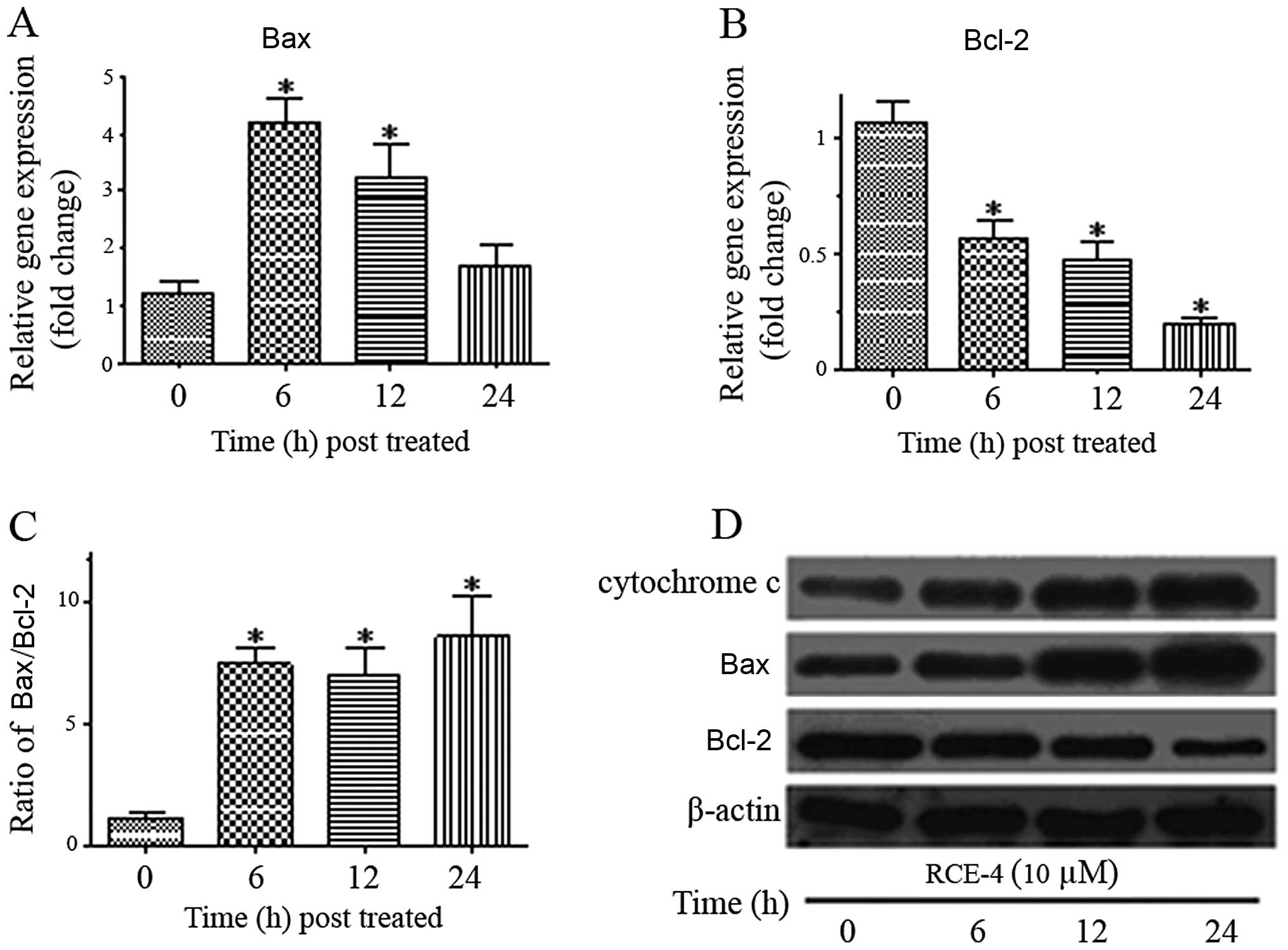
mRNA expression of Bax and Bcl-2
The Bcl-2 family plays an important regulatory role
in apoptosis, either as an activator (Bax) or inhibitor (Bcl-2).
Since RCE-4 showed the ability to interfere with the Δψm, we
hypothesized that Bax and Bcl-2 are involved in the RCE-4-induced
apoptosis. Therefore, we investigated the gene expression level of
Bax and Bcl-2 using qPCR. As depicted in Fig. 5A, Bax gene expression increased
following treatment with 10 μM RCE-4 and reached its peak at 6 h
(~4.5-fold higher compared to the untreated). In addition, Bcl-2
gene expression was clearly suppressed as it was found to decrease
2-fold at 6 h and remained lower during the treatment hours
(Fig. 5B). The increase in Bax
and the decrease in Bcl-2 expression significantly elevated the
Bax/Bcl-2 expression ratio (Fig.
5C).
Effects of RCE-4 on the expression of
apoptosis-related proteins in CaSki cells
In order to further prove that the
anti-proliferative effect of RCE-4 was due to mitochondria-mediated
apoptosis, CaSki cells were treated with RCE-4 at 10 μM for 6, 12
and 24 h and proteins implicated in apoptosis were evaluated using
western blot analysis. The release of cytochrome c from
mitochondria into cytosol induces the mitochondrial-dependent
apoptotic pathway. Cytochrome c gradually increased in
cytosol with a time-dependent increase as exposed to RCE-4
(Fig. 5D). We further
investigated the involvement of Bcl-2 and Bax, the key regulatory
factor, when cytochrome c was released into the cytosol
during apoptosis induction. RCE-4 treatment resulted in the
upregulation of Bax and downregulation of Bcl-2, leading to an
increase in the Bax/Bcl-2 ratio (Fig.
5D). These results strongly indicate that apoptosis induced by
RCE-4 in CaSki cells occurs via the mitochondria-dependent signal
pathway.
Discussion
In the present study, we first demonstrated that
RCE-4 possesses a sound antitumor activity against three cancer
cell lines and limited toxicity against two normal cell lines.
Furthermore, the involvement of antitumor mechanisms in this
cytotoxic effect was clarified showing that apoptotic cell death of
human cervical cancer CaSki cells was induced by the
mitochondria-dependent activation of caspase cascade.
An important parameter in the evaluation of
chemotherapeutic agents is their ability to inhibit cancer cell
growth and induce cancer cell death. Apoptosis is an important
means to maintain cellular homeostasis between cell division and
cell death (19,20). Apoptosis and its related signaling
pathways have a profound effect on the progression of cancer and so
induction of apoptosis is a highly desirable goal of preventive
strategies for cancer control (21). RCE-4 was found to induce a
significant loss of cell viability in a time-dependent manner when
CaSki cells were treated with RCE-4 for 48 h; a significant
reduction of cell viability was induced with the IC50
value 3.37 μM. In agreement with the cytotoxic effects of RCE-4 on
CaSki cells, marked morphological changes indicative of cell
apoptosis were clearly observed under transmission electron
microscopy, including cell shrinkage, nuclear fragmentation, loss
of cell-cell adhesion, membrane blebbing, and chromatin
condensation, alterations commonly associated with apoptosis
(22).
Furthermore, to confirm that RCE-4 induces apoptosis
in CaSki cells, an Annexin V binding assay, which measures another
feature of apoptosis, was conducted by flow cytometric analysis.
Consequently, the population of early apoptotic cells increased
with increasing RCE-4 concentrations, but the late apoptotic cells
and necrotic cells did not change notably. These results
demonstrated that RCE-4 induces significant apoptosis instead of
necrosis in a dose-dependent manner. At the same time,
RCE-4-induced apoptosis was again identified by cell cycle
analysis. The visible sub-G1 peak, which represents apoptotic
cells, appeared to increase in a dose-dependent manner following
RCE-4 treatment, suggesting that the cytotoxic effect of RCE-4 on
CaSki cells is attributable to induced apoptotic cell death.
There are two main signaling pathways involved in
apoptosis, the extrinsic and the intrinsic pathway (23). The extrinsic pathway is activated
by ligand-bound death receptors of the tumor-necrosis factor
receptor (TNFR) super-family (22). The intrinsic pathway, also called
the mitochondria mediated apoptosis pathway, is a signal
transduction pathway involving the mitochondria and the Bcl-2
family. In the present study, we examined whether RCE-4-induced
apoptosis was mediated by the mitochondrial pathways. Since loss of
mitochondrial membrane potential (Δψm) is the primary target for
the majority of intrinsic apoptotic signals, we investigated the
integrity of mitochondrial membrane using JC-1 metachromatic dye.
The results clearly demonstrated that RCE-4 drastically reduced the
depolarization of the Δψm in CaSki cells and thus proceeded through
apoptosis. In the mitochondria-mediated apoptosis pathway, an
apoptotic stimulus is followed by the release of cytochrome
c from mitochondria into the cytosol. Following the release,
cytochrome c forms a complex in the cytoplasm with adenosine
triphosphate (ATP) and others and then triggers the apoptosis
(24). The present study found
that RCE-4 promoted the release of cytochrome c into the
cytosol with its protein expression upregulation in CaSki cells.
These experimental results suggested that the intrinsic pathway was
involved in RCE-4 induced apoptosis.
Of the Bcl-2 family members, the Bcl-2 and Bax
protein ratio has been recognized as a key factor in regulation of
the apoptotic process (25).
Other studies have suggested that Bcl-2 maintains the mitochondrial
integrity, while Bax destroys the mitochondrial integrity and
causes loss of Δψm (26).
Consequently, the ratio between Bcl-2 and Bax determines the
susceptibility to apoptosis and thus dictates the life and death of
a cell (27,28). In this study, RCE-4 treatment
altered the gene expression level of Bcl-2 and Bax proteins,
showing that Bcl-2 was decreased, while Bax was increased.
Furthermore, western blotting showed that RCE-4 was able to inhibit
the protein expression of Bcl-2 and stimulate the protein
expression of Bax and so significantly increase the Bax/Bcl-2
ratio. These results are consistent with those from the gene
expression studies.
In conclusion, the present study is the first to
report a molecular pathway of apoptosis induced by RCE-4 from
Reineckia carnea. This study also suggests that RCE-4 may be
a natural potential apoptosis-inducing agent for human cervical
cancer and possibly to treat other types of cancer.
Acknowledgements
This study was financially supported by the National
Natural Science Foundation of China (Grant no. 30870254,
31070313).
References
|
1
|
Novotny L, Rauko P, Kombian SB and
Edafiogho IO: Selenium as a chemoprotective anti-cancer agent:
reality or wishful thinking? Neoplasma. 57:383–391. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Fabregat I, Roncero C and Fernandez M:
Survival and apoptosis: a dysregulated balance in liver cancer.
Liver Int. 27:155–162. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Brown JM and Attardi LD: The role of
apoptosis in cancer development and treatment response. Nat Rev
Cancer. 5:231–237. 2005.PubMed/NCBI
|
|
4
|
Fesik SW: Promoting apoptosis as a
strategy for cancer drug discovery. Nat Rev Cancer. 5:876–885.
2005. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
von Schwarzenberg K and Vollmar AM:
Targeting apoptosis pathways by natural compounds in cancer: marine
compounds as lead structures and chemical tools for cancer therapy.
Cancer Lett. Jul 29–2010.(Epub ahead of print).
|
|
6
|
Ghobrial IM, Witzig TE and Adjei AA:
Targeting apoptosis pathways in cancer therapy. CA Cancer J Clin.
55:178–194. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hoye AT, Davoren JE, Wipf P, Fink MP and
Kagan VE: Targeting mitochondria. Acc Chem Res. 41:87–97. 2008.
View Article : Google Scholar
|
|
8
|
Guclu-Ustundag O and Mazza G: Saponins:
properties, applications and processing. Crit Rev Food Sci Nutr.
47:231–258. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Nomura T, Uehara Y, Kawajiri H, Ryoyama K,
Yamori T and Fuke Y: Alkyl isothiocyanates suppress epidermal
growth factor receptor kinase activity but augment tyrosine kinase
activity. Cancer Epidemiol. 33:288–292. 2009. View Article : Google Scholar
|
|
10
|
McChesney J, Venkataraman S and Henri J:
Plant natural products: Back to the future or into extinction?
Phytochemistry. 68:2015–2022. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Xu H and Du J: The use and application of
Reineckia carnea in folk medicine. J Med Phar Chin Minor.
12:43–44. 2006.(In Chinese).
|
|
12
|
Zhang Y, Du J and Qiu DW: Overview of the
research of Miao drug Reineckia carnea. J Guiyang Coll
Tradit Chin Med. 11:2052003.(In Chinese).
|
|
13
|
Kanmoto T, Mimaki Y, Sashida Y, Nikaido T,
Koike K and Ohmoto T: Steroidal constituents from the underground
parts of Reineckea carnea and their inhibitory activity on
cAMP phosphodiesterase. Chem Pharm Bull (Tokyo). 42:926–931. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wang Q, Hou Q, Guo Z, et al: Three new
steroidal glycosides from roots of Reineckia carnea. Nat
Prod Res. Feb 3–2012.(Epub ahead of print).
|
|
15
|
Zhang ZQ, Chen JC, Yan J and Qiu MH: Three
steroids with unique structural feature of
5beta-Spirostan-1beta,3beta,17alpha-trihydroxyl from Reineckia
carnea. Chem Pharm Bull (Tokyo). 59:53–56. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Miyahara K: Co-occurrence and
high-performance liquid chromatographic separation of the
glycosides of rhodeasapogenin and its analogs which differ in the
F-ring structure. Chem Pharm Bull (Tokyo). 31:348–351. 1983.
View Article : Google Scholar
|
|
17
|
Jakubikova J and Sedlak J: Garlic-derived
organosulfides induce cytotoxicity, apoptosis, cell cycle arrest
and oxidative stress in human colon carcinoma cell lines.
Neoplasma. 53:191–199. 2006.PubMed/NCBI
|
|
18
|
Salvioli S, Ardizzoni A, Franceschi C and
Cossarizza A: JC-1, but not DiOC6(3) or rhodamine 123, is a
reliable fluorescent probe to assess delta psi changes in intact
cells: implications for studies on mitochondrial functionality
during apoptosis. FEBS Lett. 411:77–82. 1997. View Article : Google Scholar
|
|
19
|
Hengartner MO: The biochemistry of
apoptosis. Nature. 407:770–776. 2000. View
Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kaufmann SH and Hengartner MO: Programmed
cell death: alive and well in the new millennium. Trends Cell Biol.
11:526–534. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lowe SW and Lin AW: Apoptosis in cancer.
Carcinogenesis. 21:485–495. 2000. View Article : Google Scholar
|
|
22
|
Elmore S: Apoptosis: a review of
programmed cell death. Toxicol Pathol. 35:495–516. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Edinger AL and Thompson CB: Death by
design: apoptosis, necrosis and autophagy. Curr Opin Cell Biol.
16:663–669. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chipuk JE, Bouchier-Hayes L and Green DR:
Mitochondrial outer membrane permeabilization during apoptosis: the
innocent bystander scenario. Cell Death Differ. 13:1396–1402. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Suen DF, Norris KL and Youle RJ:
Mitochondrial dynamics and apoptosis. Genes Dev. 22:1577–1590.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sharpe JC, Arnoult D and Youle RJ: Control
of mitochondrial permeability by Bcl-2 family members. Biochim
Biophys Acta. 1644:107–113. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chang J, Hsu Y, Kuo P, Kuo Y, Chiang L and
Lin C: Increase of Bax/Bcl-XL ratio and arrest of cell cycle by
luteolin in immortalized human hepatoma cell line. Life Sci.
76:1883–1893. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Adams JM and Cory S: The Bcl-2 apoptotic
switch in cancer development and therapy. Oncogene. 26:1324–1337.
2007. View Article : Google Scholar : PubMed/NCBI
|