Introduction
The unprecedented growth in the field of
telecommunications has raised concerns about health risks
associated with exposure to radiofrequency (RF) radiation. Frequent
mobile users constantly complain of headaches, a burning sensation
after an extended duration of communication (1), as well as sleep disturbances
(2). RF radiation has been
reported to have an impact on neuronal function, including the
regulation of synaptic plasticity, neurotransmitter release,
neuronal survival and learning and memory (3), since RF radiation possibly induces a
decrease in the neuronal cell number and affects brain activity
(3–5). Following exposure to RF radiation,
neuronal loss and damage have been observed in the cerebellum,
basal ganglia and hippocampus (4,5). A
decrease in the number of of pyramidal cells in the cornu ammonis
(CA) areas and the dentate gyrus has also been demonstrated after
three months of exposure to a specific absorption rate (SAR) of 1.6
W/kg at an RF of 835 MHz (6) and
28 days (1 h/day) of whole body exposure to SAR 0.016 and exposure
of the head to SAR 2 W/kg at 900 MHz electromagnetic field (EMF)
(7), providing clues as to the
considerable effects of exposure to RF radiation on living
organisms. In addition to neuronal cells, glial cells, particularly
astrocytes, are also activated by exposure to RF radiation in the
cortex, caudate putamen, striatum, hippocampus and the cerebellum
(8,9). A study employing low-level frequency
exposure for 90 days to 50 Hz revealed alterations in the activity
of the central nervous system (CNS), affecting the regulation of
Ca2+ and N-methyl-D-aspartate (NMDA) receptor activity,
suggesting perturbed neuronal functions due to exposure to EMF
(3). Exposure to pulsed RF
radiation (900 MHz at SAR 4.0 W/kg) has shown a selective
diminution of rat Purkinje cell processes immunostained by
γ-aminobutyric acid (GABA)-specific antibody (10). GABA immunoreactivity (IR) in the
three layers of the rat cerebellar cortex was also decreased by
continuous exposure to RF radiation (900 MHz at 32 W/kg) (10).
The interplay between neuronal function, excitation
and inhibitory inputs at the central level plays a major role in
auditory processing (11) since
the excitatory responses induced by sound stimulation on one side
are counteracted by the inhibition obtained from stimulation on the
other side (12). Inhibitory
interactions based on neuronal networks crucially depend on the
GABA or glycine, acting as transmitters within the auditory system
(11). Glycine, similar to GABA,
is a major inhibitory neurotransmitter, predominantly localized in
the brainstem (13). Glycinergic
inputs are involved in sound localization or lateral inhibition
(11) and play a major role in
affecting the development of post-synaptic properties (14), as well as in the refinement of
synaptic connections during the postnatal period (15). In addition, glycinergic inputs are
essential to auditory function in the auditory nuclei of the
brainstem (16) from the cochlear
nuclear complex (CNC) to the inferior colliculus (IC) (17,18).
Auditory information of the ascending and descending
signal is processed by various nuclei situated along the auditory
brainstem including, the CNC, the superior olivary complex (SOC),
nuclei of the lateral lemniscus (NLL) and the IC, each possessing
distinct properties as regards activation (19). The CNC is composed of the dorsal
cochlear nucleus (DCN) and the ventral cochlear nucleus with
further divisions of the ventral nuclei into anteroventral cochlear
nuclei (AVCN) and posteroventral cochlear nuclei (PVCN) (20). Fibers from the cells of the
cochlear nuclei comprise parallel ascending pathways to the SOC,
while it receives morphologically distinct axosomatic endings of
primary afferent fibers from the cochlea (21).
The SOC as a part of the auditory system is
comprised of the lateral superior olive (LSO), the superior
paraolivary nucleus (SPN) and the medial nucleus of the trapezoid
body (MNTB) (22). The LSO
receives bilateral innervation and comprises an important part of
hearing as it is at this level in the ascending auditory pathway
where binaural processing of sound localization cues first occurs
(23). The MNTB provides
inhibitory input to the LSO and imparts sensitivity to interaural
intensity differences in cells in the LSO (24).
The IC is an obligatory processing station in the
auditory pathway receiving inhibitory and excitatory afferents from
the majority of the brainstem nuclei (25,26). The ascending auditory fibers end
in an orderly tonotopic array of fibrodendritic layers of the
central nucleus of the IC (ICC) (27).
Glycinergic synapse is known to occur throughout the
auditory brainstem, exerting inhibitory control over the discharge
of auditory neurons through the action of the post-synaptic glycine
receptors (GlyRs) (13,28,29). Changes in receptor expression may
have an impact on synaptic strength (30). The nuclei of the SOC, mainly LSO,
medial superior olivary nucleus (MSO) and SPN along with cochlear
nuclei, provide the glycinergic input, while in the IC, these
glycinergic inputs are provided by projections from the ventral
nuclei of the lateral lemniscus (VNLL) and LSO. In addition, the
glycinergic input is strongly provided to the SPN by the MNTB
(31), whose neurons display
offset responses to pure tones. The release from MNTB-derived
glycinergic inhibition is critical to the formation of SPN offset
responses (32). Hence, any
disturbance in the auditory circuit region is likely to affect the
normal hearing process. Previous reports have shown that a decrease
in the number of GlyR immunoreactive cells in the auditory system
is associated with hearing loss (22,33).
Considering that the use of the cellular phone
entails its close proximity with hearing regions, exposure to RF
radiation may induce changes in the neurotransmitters of
hearing-related brain regions, including the central auditory
nucleus. However, few studies have focused on the auditory system;
furthermore, the majority of studies have concentrated on
otoacoustic emission in both humans (34) and animals (35,36). Moreover, to the best of our
knowledge, studies employing immunohistochemical methods as a
quantitative approach for the acquisition of crucial information as
to the effects of exposure to RF radiation in the auditory
brainstem region have not been conducted to date. Therefore, the
present study focused on alterations in GlyR IR in the central
auditory brainstem following exposure to RF radiation using
free-floating immunohistochemistry to assess the possible effects
of exposure to RF radiation on GlyR IR in the auditory region (IC,
NLL, SOC and CNC) of mice following exposure to RF radiation for
three months at SAR 4.0 W/kg.
Materials and methods
Animal experimentats
Six-week-old ICR male mice (n=20; weighing, 20–30 g)
obtained from Orientbio, Inc., Sungnam-si, Korea) were obtained and
kept in an animal room under controlled conditions (mean
temperature, 22.5±1°C; humidity, 55±10%; 12-h light/dark cycle).
Food (Samtako Bio Korea, Osan, Korea) and water were supplied ad
libitum. The experimental procedures were reviewed and approved
by the Dankook University Institutional Animal Care and Use
Committee (DUIAC), which adheres to the guidelines issued by the
Institution of Laboratory and Animal Resources (ILAR) and were
performed in compliance with NIH guidelines for animal
research.
Exposure system
The exposure system (Wave Exposer V20) was used in
this study (6). The Wave Exposer
V20, which emits 835 MHz (equivalent to the Korean CDMA mobile
phone frequency), was designed by the Division of Information
Technology Engineering, Soonchunhyang University (Asan, Korea)
(6). The SAR was adjusted to 1.6
and 4.0 W/kg, which is the same value as the electric field
intensity between 59.56 and 94.18 V/m for muscle (0.92, 57 and 1020
kg/m3) on the 835 MHz CDMA frequency. Waves were
generated and amplified in an electronic unit, and were eventually
radiated by a pyramidal rectangular horn antenna connected by a
waveguide to the coaxial transition. A standard mouse cage of 22
inches was used for the apparatus. The output powers of the horn
antenna from the exposure apparatus were 2.5 W for SAR 1.6 W/kg and
6.3 W for SAR 4.0 W/kg. Electric field intensities due to SAR
values were be calculated, and the power value was obtained by a
computer simulation with a high frequency structure simulator
(HFSS) manufactured by Ansoft, Co. (Pittsburgh, PA, USA). Five
three-dimensional cylindrical MEMS antennas were used for the
simulation. The simulation variable included both the location of
the mouse and the distance from the horn aperture for freely-moving
mice. The power was obtained by averaging the simulated peak
electric field intensities from each mouse body. The wave exposure
from the horn antenna to the mouse cage was provided by the wave
absorption material (TDK ceramic absorber) which mimics the
radiation exposure in an open environment and limits the influence
the number of mice may have on exposure. To eliminate potential
stress during exposure, the exposure apparatus used in the present
study had a cooling system that did not exceed a temperature of
26°C during the exposure period as reported in our previous study
(6). Likewise, the internal
temperature, which is always denoted by the digital number in front
of the instrument, was checked on a regular basis in order to
maintain the optimum temperature at 24°C. Additionally, the
exposure apparatus provides an automatic light system with a water
feeder and no restriction in movement. To elucidate ambient noise
levels in the present study, a sound-level meter NA-24 (Rion, Co.,
Ltd., Tokyo, Japan) was used to determine ambient noise levels both
within the exposure apparatus and in the animal room during RF
radiation. When steady-state noise levels were recorded on >5
consecutive trials, each noise level of the animal room and
exposure apparatus during RF radiation was confirmed to range from
42.0 to 44.6 dB and from 56.4 to 58.2 dB, respectively.
Experimental design
The mice were exposed to 835 MHz of radiation with
an average SAR of 4.0 W/kg using the Wave Exposer V20. The mice
were divided randomly into two groups (n=10): i) a sham control
(SC) group and ii) a group exposed to SAR of 4.0 W/kg for three
months [exposed (E4) group). The exposure duration was 8 h/day.
Both the SC and E4 groups were subjected to the same surgical and
anesthetic procedure for auditory brainstem response (ABR)
testing.
ABR
The ABR was recorded using a signal-processing
system (System III, Tucker Davis Technologies, Alachua, FL, USA).
The ABR recording was performed immediately after the three-month
period of exposure to RF radiation. For anesthesia, zolazepam
(Zoletil; Virbac, Carros Cedex, France) and xylazine (Rompun;
Bayer, Leverkusen, Germany) were mixed in a 4:1 ratio (0.1 ml/100
g). During ABR recording, the animals were placed on a warm pad,
the temperature of which was approximately 40°C. Acquired
auditory-evoked brainstem responses were filtered through a 10-kHz
high pass and a 3-kHz low pass filter. The sampling rate was 25 k
samples/sec. The presented stimulus was a rarefaction click of 0.1
msec duration.
The animals were placed in a soundproof booth and
three electrodes were inserted subcutaneously, one at the vertex
and the other two ventrolaterally to each ear, beneath the pinna
(active, reference and ground electrodes, respectively). The click
stimuli were delivered through a tube inserted into the ear canal
of the mouse. Hearing thresholds were determined by the assessment
of the lowest stimulus level required to elicit the ABR peaks III
or V at levels from 10 to 80 dB sound pressure level (SPL) in 5-dB
steps. When the ABR threshold was >80 dB SPL, it was defined as
80 for statistical analysis. One thousand and twenty-four tone
presentations were averaged. The ABR hearing thresholds were
confirmed by an independent observer. Statistical procedures were
implemented using Statistical Package for Social Sciences (SPSS)
version 17 software. Statistical differences in the ABR thresholds
between the SC and E4 groups were compared using the Mann-Whitney
test. The difference between the groups was considered
statistically significant at a value of P<0.05.
Immunohistochemical analysis
The animals were anesthetized with diethyl ether and
their brains were collected following transcardial perfusion with
phosphate-buffered saline (PBS) and a 4% paraformaldehyde (PFA)
solution. Anesthesia was also used to avoid animal stress and to
lower the augmentation of blood pressure during perfusion and
fixation. Following perfusion, the brains were immediately removed,
post-fixed overnight in 4% PFA and cryoprotected by infiltration
with a sucrose series (10, 20 and 30%) solution at 4°C. Serial
coronal sections of 40 μm thickness were obtained using a cryocut
microtome (CM3050S, Leica Biosystems Nussloch GmbH) and collected
in 6-well plates. Immunohistochemistry was performed using a
free-floating method, as previously described (6). Briefly, the coronal sections were
incubated for 48 h at 4°C in rabbit polyclonal antibodies to GlyR
α1+α2 (ab23809, dilution ratio of 1:2,500;
Abcam, Cambridge, UK) in blocking buffer containing 1% bovine serum
albumin, 0.3% Triton X-100, and 1% normal goat serum. To eliminate
peroxidase activity, the sections were treated with 1% hydrogen
peroxide in PBS. The sections were incubated with a biotinylated
secondary antibody at the dilution ratio of 1:250 for 1.5 h at room
temperature, followed by treatment with an avidin-biotin-peroxidase
complex (Vectastain ABC mouse Elite kit; Vector Laboratories,
Burlingame, CA, USA). Following three washes in PBS, the sections
were reacted with 3,3′-diaminobenzidine (DAB) and hydrogen peroxide
in a distilled water solution for 5 min. The sections from each
group were stained together to minimize variability. A sample of
sections was reacted without primary antiserum. The sections from
these samples did not exhibit any of the IR described in this
study. Following additional washes, dehydration in solutions of
increasing percentages of ethanol and clearing in xylene, the
sections were mounted on gelatin-coated slides with a cover slide
for analysis.
Image analysis
An Olympus BX51 microscope was used for analysis and
images were acquired using a digital camera system (DP50; Olympus,
Tokyo, Japan). The NIH image program (ImageJ, version 1.44) was
used to determine the staining densities and was also used for cell
counting using the manual cell counting and marking method. Only
sections with clearly differentiated layers of nuclei from the
brainstem auditory circuit were collected for densitometry
analysis. Immunolabeling in the SC and E4 groups was carried out as
previously described (6). Gray
values of the digitized micrographs were analyzed within the
outlines of all the identifiable nuclei within a section. The sum
of the gray values of all pixels in each corresponding region was
divided by the total number of pixels in the region to determine
the mean density of IR per unit area (mm2). Only the
clearly stained nuclei were included in the analysis. Outlined
regions of interest for corresponding nuclei were analyzed with
similar size while the out-of-focus structures and non-uniform
stained figures were excluded (total of 50–100 nuclei per antibody
and animal group). The background of each section was measured by
outlining an area in the nearby tissue in which the stain was very
weak. The analysis of the slides was performed by an investigator
blinded to the experimental procedures.
Statistical analysis
Differences between different nuclei from the
auditory brainstem regions were determined by one-way analysis of
variance (ANOVA) followed by post-hoc analysis with the Bonferroni
test (SigmaPlot version 10.0; Systat Software Inc., Chicago, IL,
USA). Student’s t-tests using SPSS software version 17 were used
when only two groups were compared. Values are expressed as the
means ± standard deviation (SD). A P-value <0.05 was considered
to indicate a statistically significant difference.
Results
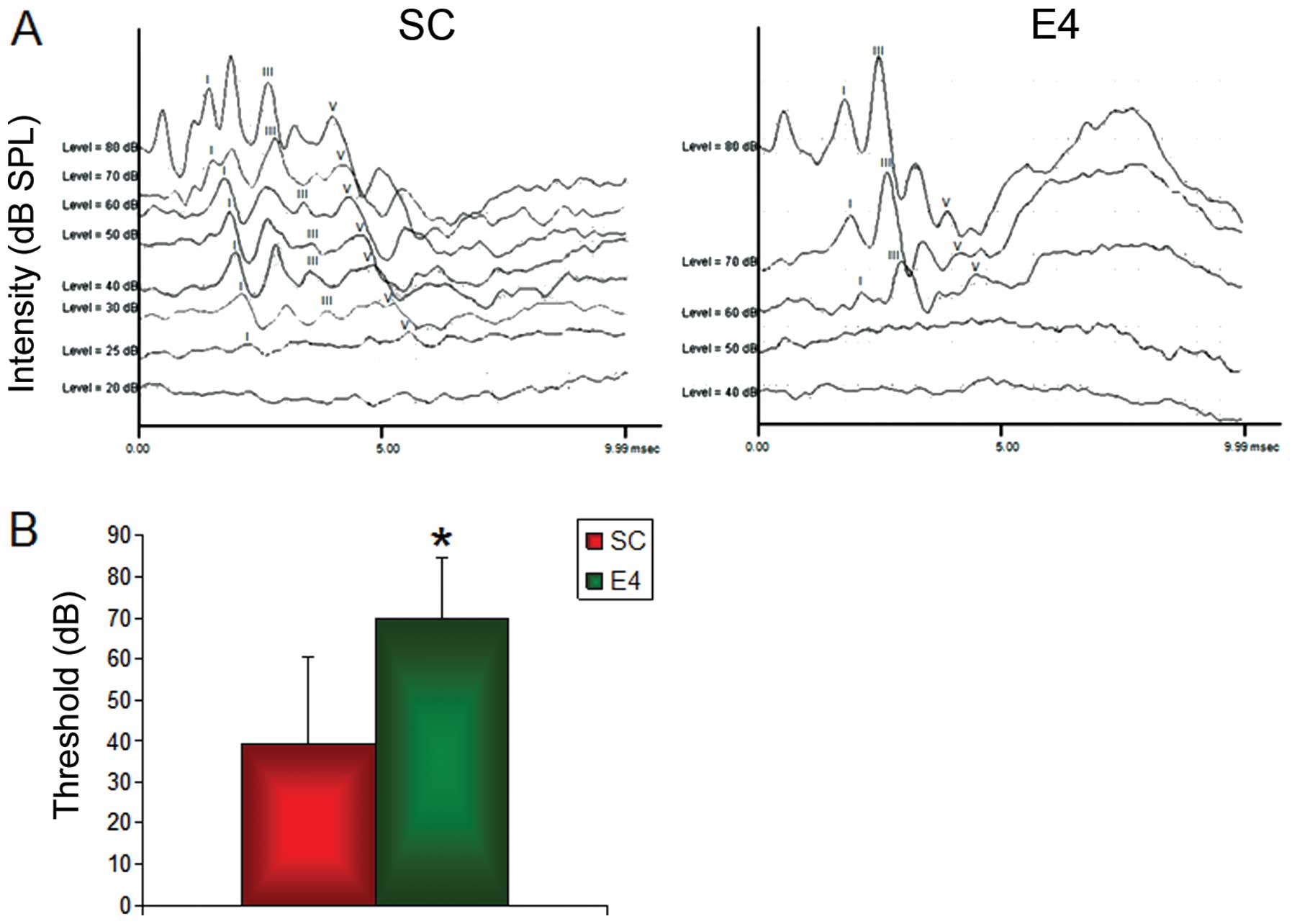
ABR
The ABR response was detectable even with a very
small stimulus intensity in the SC group, but was only detectable
with a large stimulus intensity in the E4 group (Fig. 1A). The SC group exhibited
characteristic ABR waveforms at an SPL as low as 25 dB, while the
E4 group ABR threshold was measured at 60 dB. The ABR threshold of
the SC group was 39.3±21.1 dB SPL, and that of the E4 group was
70.0±14.7 dB SPL (Fig. 1B). The
ABR threshold of the E4 group was significantly higher than that of
the SC group (P<0.001), signifying a decrease in hearing
intensity in the E4 group (Fig.
1B).
Histological observations
i) CNC
GlyR IR was observed in the cells and neuropils in
all the nuclei of the CNC (Fig.
2A–F). In the AVCN of the SC group, the GlyR IR was noted to be
localized in the cell bodies and the dendrites of the bushy cells
(BC) along with prominent GlyR immunoreactive puncta close to the
cell membrane (Fig. 3A and G).
Compared with the SC group, the GlyR IR of BCs was markedly
decreased in th E4 group, particularly in the puncta, and was
almost absent in some cells (Fig. 3B
and H). The DCN, irrespective of its subdivisions, revealed
prominent GlyR IR along with the primary dendrites and neuropils in
the SC group and darkly stained puncta were close to the cell
membrane (Fig. 3C and I).
However, in the E4 group, the overall decrease in the GlyR
immunoreactive cell number was accompanied by a reduction in cell
size and a marked decrease in GlyR IR was also observed in the DCN.
In addition, GlyR IR of puncta was also much decreased in the cells
as well too (Fig. 3D and J). GlyR
IR was identified in the fusiform (Fig. 3I and J) and cartwheel cells
(Fig. 3I and J) of the DCN and
GlyR IR of those cells was decreased in the E4 group compared with
the SC group. Similarly, the PVCN of the SC group stained with the
GlyR-specific antibody revealed prominent GlyR IR in the neuronal
somas and puncta closely lining the cells. In the E4 group,
compared to the SC group, GlyR IR of BCs in the PVCN was markedly
decreased. The decrease in cell number in the E4 group compared
with the SC group was clearly distinguishable and few GlyR
immunorective cells in the PVCN of the E4 group clearly showed a
decrease in GlyR IR and a decrease in staining intensity within the
puncta (Fig. 3E, F, K and L). A
decrease in the staining intensity of the neuropils in the E4 group
(Fig. 3H, J and L) compared with
the SC group (Fig. 3G, I and K)
was also observed in all three subdivisions of the CNC.
 | Figure 2Photomicrograph of glycine receptor
(GlyR) α1+α2 immunoreactivity (IR) in coronal
sections of the cochlear nuclear complex comprising of (A and B)
AVCN, (C and D) DCN and (E and F) PVCN of the (A, C and E) sham
control (SC) group and (B, D and F) group exposed to radiofrequency
(RF) radiation (E4 group) at SARs 0 and 4.0 W/kg, respectively,
after three months of exposure to RF radiation at 835 MHz. A
prominent decrease in GlyR IR of neuropils, as well as in the
number of GlyR immunoreactive neurons was observed in the E4 group
compared with the SC group. AVCN, anteroventral cochlear nucleus;
PVCN, posteroventral cochlear nucleus; DCN, dorsal cochlear
nucleus. Scale bar, 100 μm. |
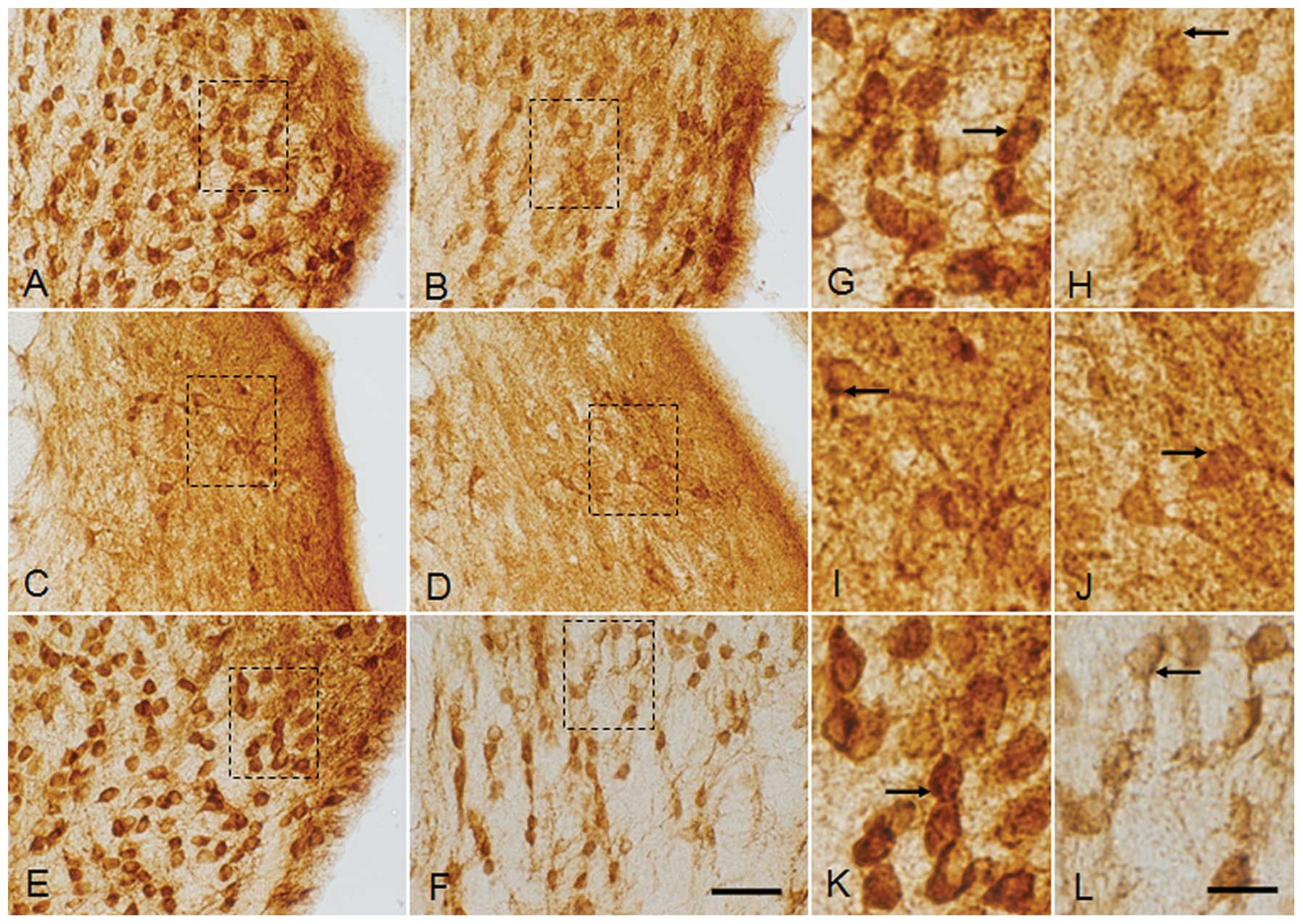 | Figure 3Magnified image of glycine receptor
(GlyR) α1+α2 immunoreactivity (IR) in coronal
sections through the cochlear nuclear complex, including the (A, B,
G and H) AVCN, (G, D, I and J) DCN and (E, F, K and L) PVCN of the
(A, C, E, G, I and K) sham control (SC) group and (B, D, F, H, J
and L) the group exposed to radiofrequency radiation (E4 group) at
SARs of 0 and 4.0 W/kg, respectively, after three months of
exposure to RF radiation at 835 MHz. Magnified images of the dotted
squares in (A, B, C, D, E and F) are represented in (G, H, I, J, K
and L, respectively). Note the loss of GlyR IR in the somas of the
E4 group in all the three regions of the cochlear nuclear complex,
which was found to be very severe in the PVCN. The number of GlyR
immunoreactive puncta (arrows) was also prominently reduced in the
E4 group compared with the SC group. AVCN and PVCN showed bushy
cells (BCs) (thick arrows) with lighter staining in the E4 group
compared with the SC group. A variety of cell types present in the
AVCN, DCN and PVCN showed decreased staining intensity in the E4
group as compared with SC group. AVCN, anteroventral cochlear
nucleus; PVCN, posteroventral cochlear nucleus; DCN, dorsal
cochlear nucleus. Scale bar: A–F, 50 μm; G–L, 10 μm. |
According to the relative density, GlyR IR was
significantly (P<0.05) decreased by 4.5% in the AVCN from
109.42±5.27 in the SC group to 104.41±5.82 in the E4 group
(Fig. 9A). GlyR IR in the DCN
region was 121.35±2.90 in the SC group which was significantly
decreased (P<0.0001) by 5.68% to 114.45±3.24 in the E4 group
(Fig. 9A). A significant
(P<0.0001) decrease in GlyR IR in the PVCN was also observed
from 103.96±4.62 in the SC group to 91.07±3.10 in the E4 group
which amounted to 12.40% (Fig.
9A).
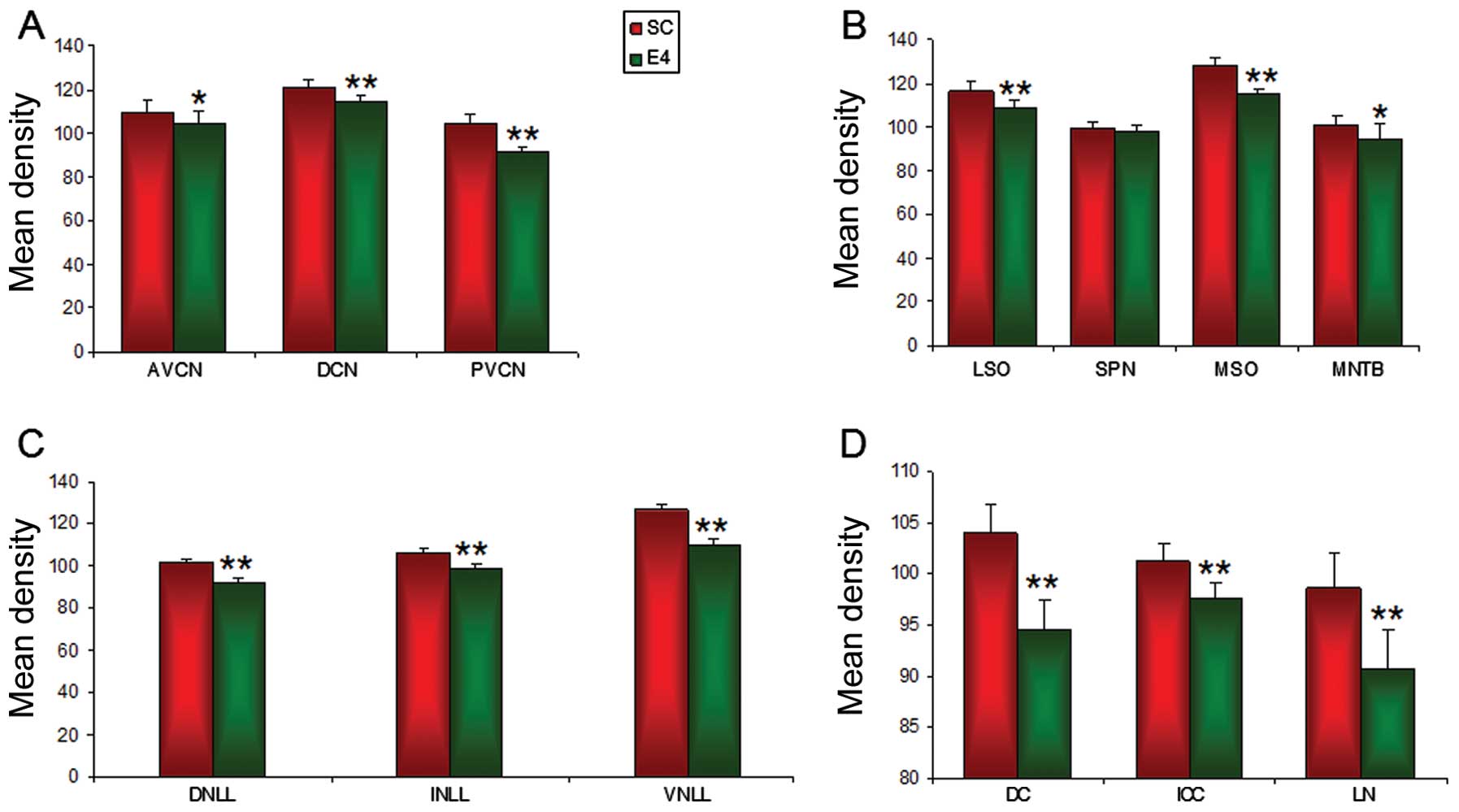 | Figure 9Image analysis of relative densities
of glycine receptor (GlyR) α1+α2
immunoreactivity (IR) in the (A) IC, (B) NLL, (C) SOC and (D) CNC
of the sham control (SC) group and the group exposed to
radiofrequency radiation (E4 group) at SARs 0 and 4.0 W/kg,
respectively after three months of exposure to RF radiation at 835
MHz. A decrease in IR was noted in the E4 group in the various
regions of the auditory brainstem as compared with the SC group.
The data shown are the means ± SD obtained from 5 different
experiments. CNC, cochlear nuclear complex; AVCN, anteroventral
cochlear nucleus; PVCN, posteroventral cochlear nucleus; DCN,
dorsal cochlear nucleus; SOC, superior olivary complex; LSO,
lateral superior olive; SPN, superior paraolivary nucleus; MSO,
medial superior olive; MNTB, medial nucleus of the trapezoid body;
DNLL, dorsal nucleus of lateral leminiscus; INLL, intermediate
nucleus of lateral leminiscus; VNLL, ventral nucleus of lateral
leminiscus; NLL, nucleus of lateral leminiscus; DC, dorsal cortex
of IC; ICC, central nucleus of IC; LN, lateral nucleus of IC; IC,
inferior colliculus (*P<0.05,
**P<0.0001). |
ii) SOC
GlyR IR was noted in all the major nuclei of the
SOC, most notably in the LSO, SPN, MSO and MNTB of the SC group.
GlyR IR was localized in both the somas and neuropils of the LSO,
SPN and MSO, while the somas were stained mainly in the MNTB region
(Fig. 4). Prominent GlyR
immunoreactive puncta were observed as close to the membrane of the
soma leaving the perikarya unstained. The GlyR immunoreactive
puncta were restricted to the soma and were not detected in the
dendrites or neuropils (Fig.
5K–T). In staining with GlyR-specific antibody, the bipolar
cells with the eccentric nucleus were mainly found in the LSO and
MSO, while the SPN comprised of GlyR immunoreactive bipolar, as
well as several multipolar neurons. The SPN and MSO contained
numerous GlyR immunoreactive fibers in between the GlyR
immunoreactive cells (Fig. 5O–R).
GlyR IR in the MNTB was specifically localized on the cellular
membrane with GlyR immunoreactive puncta. The somas on the lateral
aspect of the MNTB were prominently visible by staining with
GlyR-specific antibody and staining was observed in the cytoplasm
of the soma.
 | Figure 4Photomicrograph of glycine receptor
(GlyR) α1+α2 immunoreactivity (IR) in coronal
sections of the SOC comprising of (A and B) LSO; (C and D) SPN and
MSO; and (E and F) MNTB of (A, C and E) the sham control (SC) group
and (B, D and F) group exposed to radioafrequency (RF) radiation
(E4 group) at SARs 0 and 4.0 W/kg, respectively, after three months
of exposure to RF radiation at 835 MHz. GlyR IR was observed in the
neurons and neuropils of the (A and B) LSO; (C and D) SPN and MSO;
and (E and F) the MNTB of both the SC and E4 groups. Comparisons
with the SC group revealed a loss inf GlyR IR in all the nuclei as
well as the neuropils of the E4 group. SOC, superior olivary
complex; LSO, lateral superior olive; SPN, superior paraolivary
nucleus; MSO, medial superior olive; MNTB, medial nucleus of the
trapezoid body. Scale bar, 100 μm. |
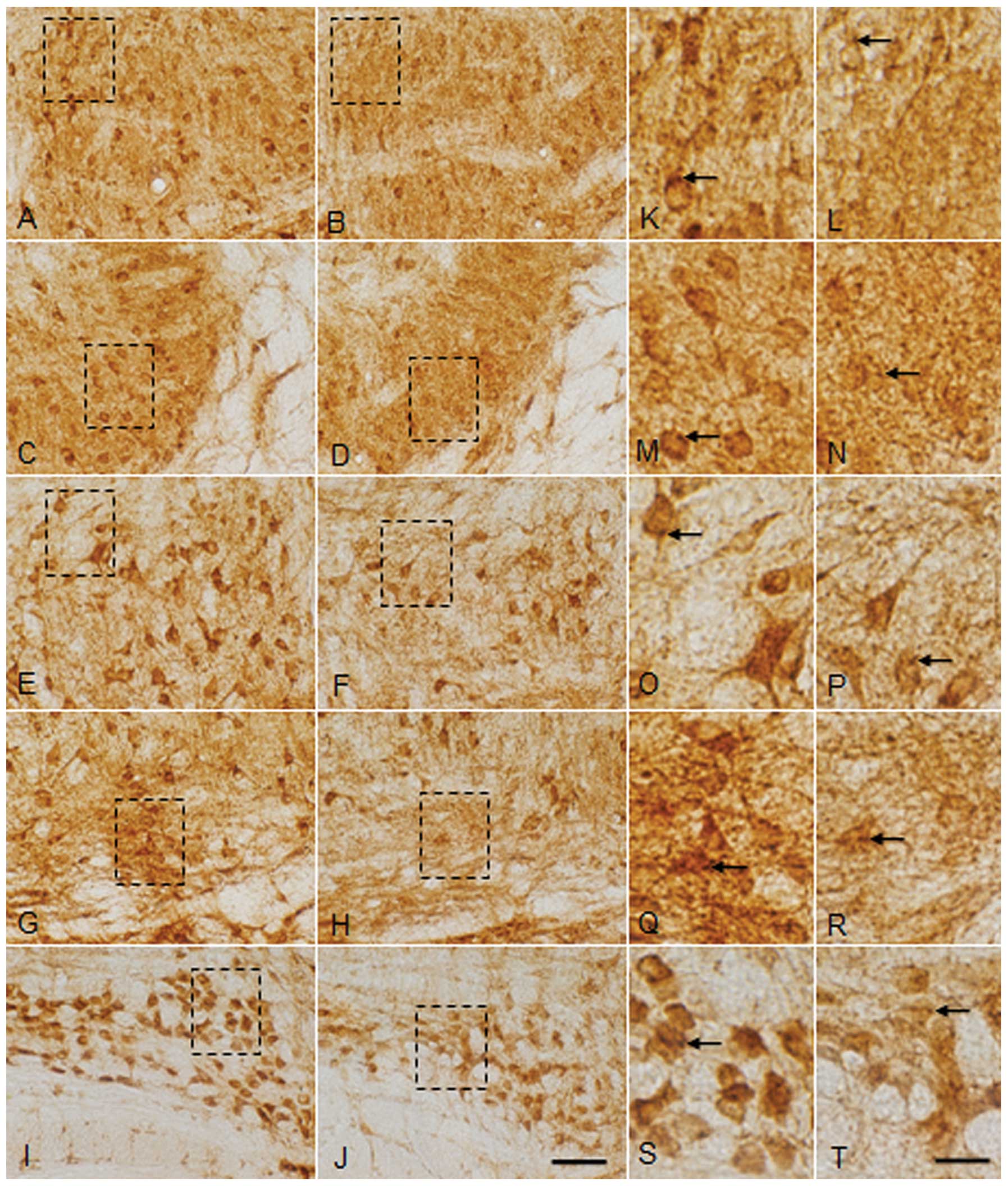 | Figure 5Magnified image of glycine receptor
(GlyR) α1+α2 immunoreactivity (IR) in coronal
sections through the SOC composed of the (A–D and K–L) LSO, (E, F,
O, P and L) SPN, (G, H, Q and R) MSO and (G, H, S and T) MNTB of
(A, C, E, G, H, K, M, O, Q and S) the sham control (SC) group and
(B, D, F, H, J, L, N, P, R and T) the group exposed to
radiofrequency radiation (E4 group) at SARs 0 and 4.0 W/kg,
respectively, after three months of exposure RF radiation at 835
MHz. Magnified images of the dotted squares in (A, B, C, D, E, F,
G, H, I and J) are represented in (K, L, M, N, O, P, Q, R, S and T,
respectively). Scattered highly GlyR immunoreactive cells were
noted in all the nuclei of the SOC. Puncta (arrows) representing
immunoreactive presynaptic terminals were also noted. Loss of the
GlyR immunoreactive cells was noted in the (B and L) medial and (D
and N) lateral area of the LSO, (F and P) SPN, (H and R) MSO and (J
and T) MNTB of the E4 group compared with the SC group. Also note
the decrease in staining intensity of the puncta (arrows) of the E4
group compared with the SC group. SOC, superior olivary complex;
LSO, lateral superior olive; SPN, superior paraolivary nucleus;
MSO, medial superior olive; MNTB, medial nucleus of the trapezoid
body. Scale bar: A–H, 50 μm; I–P, 10 μm. |
As compared to the SC group, a marked decrease in
GlyR IR in the somas and neuropils was observed in the E4 group. In
the LSO of the E4 group, particularly in the lateral limb, a
decrease in GlyR IR in the cells of the E4 group and a loss of
neuropil staining was observed (Fig.
5K–N). Similarly, a marked decrease in GlyR IR was observed in
the SPN, MSO and MNTB of the E4 group (Fig. 5E–J). A significant decrease in the
size of the soma with a loss of GlyR immunoreactive puncta was
observed in the SPN of the E4 group (Fig. 5O and P). Furthermore, as compared
with the SC group, the loss of GlyR IR in the soma was observed in
the MSO of the E4 group (Fig. 5Q and
R) and a marked decrease in GlyR IR in the fibers of the SPN,
MSO and MNTB was also observed in the E4 group (Fig. 5I, J, S and T).
The relative mean density analysis revealed that
GlyR IR in the LSO was 116.34±4.45 in the SC group, which was
significantly decreased (P<0.0001) by 6.67% (108.57±3.39) in the
E4 group (Fig. 9B). Compared with
the SC group, measured as 99.22±2.94, a slight non-significant
decrease to 98.37±2.08 in GlyR IR was observed in the E4 group,
which amounted to a 0.85% decrease (Fig. 9B). A significant decrease
(P<0.0001) in 10.49% in the SC group from 128.15±3.65 to
114.70±2.66 in the E4 group was noted in the MSO. The MNTB also
exhibited a significant decrease in GlyR IR (P<0.05) of 6.23%
from 100.87±3.86 in the SC group to 94.59±7.13 in the E4 group
(Fig. 9B).
iii) NLL
In the SC group (Fig.
6), varying intensities of GlyR IR were observed in all three
subdivisions of the NLL [VNLL, intermediate NLL (INLL) and dorsal
NLL (DNLL)] localized in the somas and neuropils. Compared with the
DNLL and INLL in both groups (Fig.
7A–J), the number of GlyR immunoreative soma of the VNLL was
increased (Fig. 7E, F, K and L).
In the SC group, the VNLL exhibited a higher density of GlyR
immunoreactive soma than the soma in the DNLL and INLL (Fig. 7A–F). GlyR immunoreactive neuropils
were prominently observed in the VNLL (Fig. 7A–F). Prominent GlyR immunoreactive
puncta were also observed in the soma of the VNLL (Fig. 7K), while the number of GlyR
immunoreactive puncta in the DNLL and INLL was markedly decreased
(Fig. 7G and I). In the E4 group,
the staining intensity of the GlyR was markedly decreased in all
subdivisions, notably in the VNLL of the E4 group (Fig. 7F and L), compared with the SC
group.
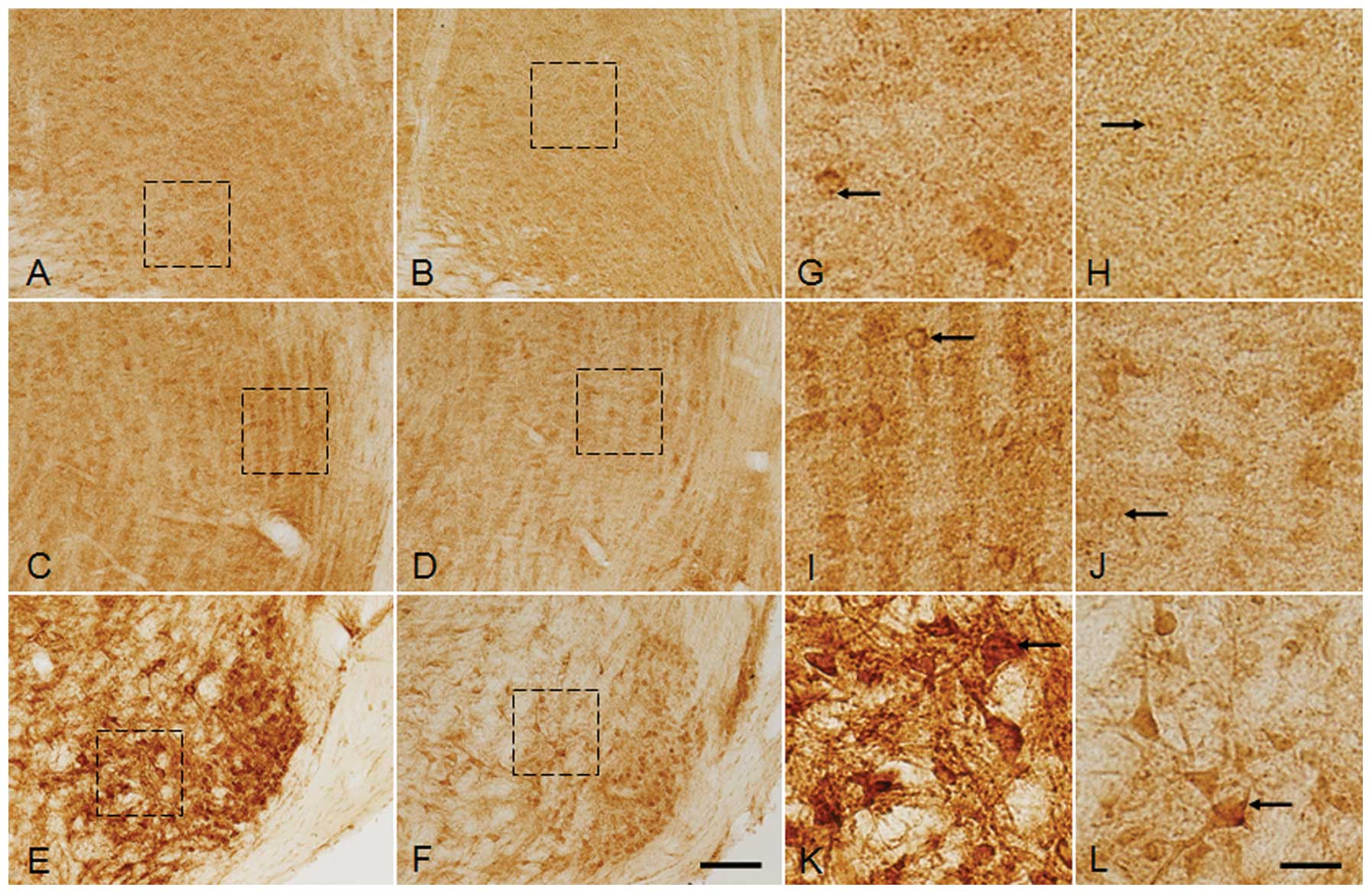 | Figure 7Magnified image of glycine receptor
(GlyR) α1+α2 immunoreactivity (IR) in coronal
sections through the (A, B, G and H) DNLL, (C, D, I and J) INLL and
(E, F, K and L) VNLL of the (A, C, E, G, I and K) sham control (SC)
group and (B, D, F, H, J and L) the group exposed to radiofrequency
radiation (E4 group) at SARs 0 and 4.0 W/kg, respectively, after
three months of exposure to RF radiation at 835 MHz. Magnified
images of the dotted squares in (A, B, C, D, E and F) are
represented in (G, H, I, J, K and L, respectively). The GlyR
immunoreactive soma were weakly stained in the subdivisions of the
NLL of the E4 group as compared with the SC group. Note the
decrease in the staining intensity of GlyR immunoreactive puncta
(arrows) in the E4 group as well. DNLL, dorsal nucleus of lateral
leminiscus; INLL, intermediate nucleus of lateral leminiscus; VNLL,
ventral nucleus of lateral leminiscus; NLL, nucleus of lateral
leminiscus. Scale bar: A–F, 50 μm; G–L, 10 μm. |
The relative mean density analysis revealed the
highest level of GlyR IR in the VNLL and the lowest level in the
DNLL in both groups. GlyR IR was markedly decreased in the E4 group
as compared with the SC group. GlyR IR in the DNLL was 101.72±1.86
in the SC group, but was significantly decreased (P<0.0001) by
9.38% (to 92.17±2.01) in the E4 group (Fig. 9C). A significant decrease
(P<0.0001) of 6.97% from 106.17±2.26 in the SC group to
98.76±1.96 in the E4 group was noted in the INLL (Fig. 9C). The VNLL also exhibited a
significant decrease in GlyR IR (P<0.0001) of 13.76% from
126.87±2.29 in the SC group to 109.41±3.33 in the E4 group
(Fig. 9C).
iv) IC
In both the SC and E4 groups, the overall intensity
of GlyR IR throughout the IC was observed to be lower than that in
the SOC and CNC (Fig. 8A–F). In
the SC group, all three subdivisions of the IC, the dorsal cortex
(DC), ICC and lateral nucleus of the IC (LN) consisted of numerous
GlyR immunoreactive neurons and dense GlyR immunoreactive puncta,
which were mainly observed in the somas (Fig. 8A, C and E). In particular, GlyR
immunoreactive bipolar neurons were observed in the SC group with
specific dendritic distribution. When the E4 group was compared
with the SC group, it was difficult to find the GlyR immunoreactive
cells throughout the IC. Along with decreased number of GlyR
immunoreactive neurons in all subdivisions of the IC, a marked
decrease in GlyR immunoreactive soma was observed in the E4 group
as well (Fig. 8G–L). In addition,
GlyR IR intensity of the puncta was markedly decreased in the somas
(Fig. 8G–L).
 | Figure 8Photomicrograph of glycine receptor
(GlyR) α1+α2 immunoreactivity (IR) in the
coronal sections of the inferior colliculus (IC) composed of the
(A, B, G and H) DC, (C, D, I and J) ICC and the (E, F, K and L) LN
of the (A, C, E, G, I and K) sham control (SC) group and (B, D, F,
H, J and L) the group exposed to radiofrequency radiation (E4
group) at SARs 0 and 4.0 W/kg, respectively, after three months of
exposure to (RF) radiation at 835 MHz. Magnified images of the
dotted squares in (A, B, C, D, E and F) are represented in (G, H,
I, J, K and L, respectively). Note the prominent loss of GlyR IR in
the immunoreactive soma (arrows) in all the regions of the IC of
the E4 groups as compared with the SC group. DC, dorsal cortex of
IC; ICC, central nucleus of IC; LN, lateral nucleus of IC. Scale
bar: A–F, 100 μm; G–L, 10 μm. |
GlyR IR in the IC was analyzed in three different
regions namely, the DN, ICC and LN. According to the relative
density, GlyR IR was markedly decreased in the IC of the E4 group
compared with the SC group. GlyR IR was significantly (P<0.0001)
decreased by 9.10% in the DC, from 104.01±2.71 in the SC group to
94.55±2.88 in the E4 group (Fig.
9D). GlyR IR in the ICC was also decreased significantly
(P<0.0001) by 3.53%, from 101.18±1.71 in the SC group to
97.60±1.57 in the E4 group (Fig.
9D). Similarly, a 7.97% decrease was noted in the LN from
98.56±3.54 in the SC group to 90.70±3.75 in the E4 group, which was
shown to be statistically significant (P<0.0001) (Fig. 9D).
v) GlyR immunoreactive cell count
In the comparison of the GlyR immunoreactive cells
in the SOC and CNC regions between the SC and E4 groups, the number
of the GlyR immunoreactive cells in the E4 group was found to be
significantly decreased. A 31.09% decrease (p<0.0001) in GlyR IR
was observed in the AVCN from 250.2±18.29 in the SC group to
172.4±16.68 in the E4 group (Fig.
10A). Although a 14.08% decrease was observed in the DCN from
42.6±9.28 in the SC groiup to 36.6±4.27 in the E4 group, the
difference was not statistically significant (Fig. 10A). The PVCN also displayed a
significant difference (P<0.0001) with a 32.79% decrease in the
number of GlyR immunoreactive cells from 285.4±3.36 in the SC group
to 191.8±8.78 in the E4 group (Fig.
10A).
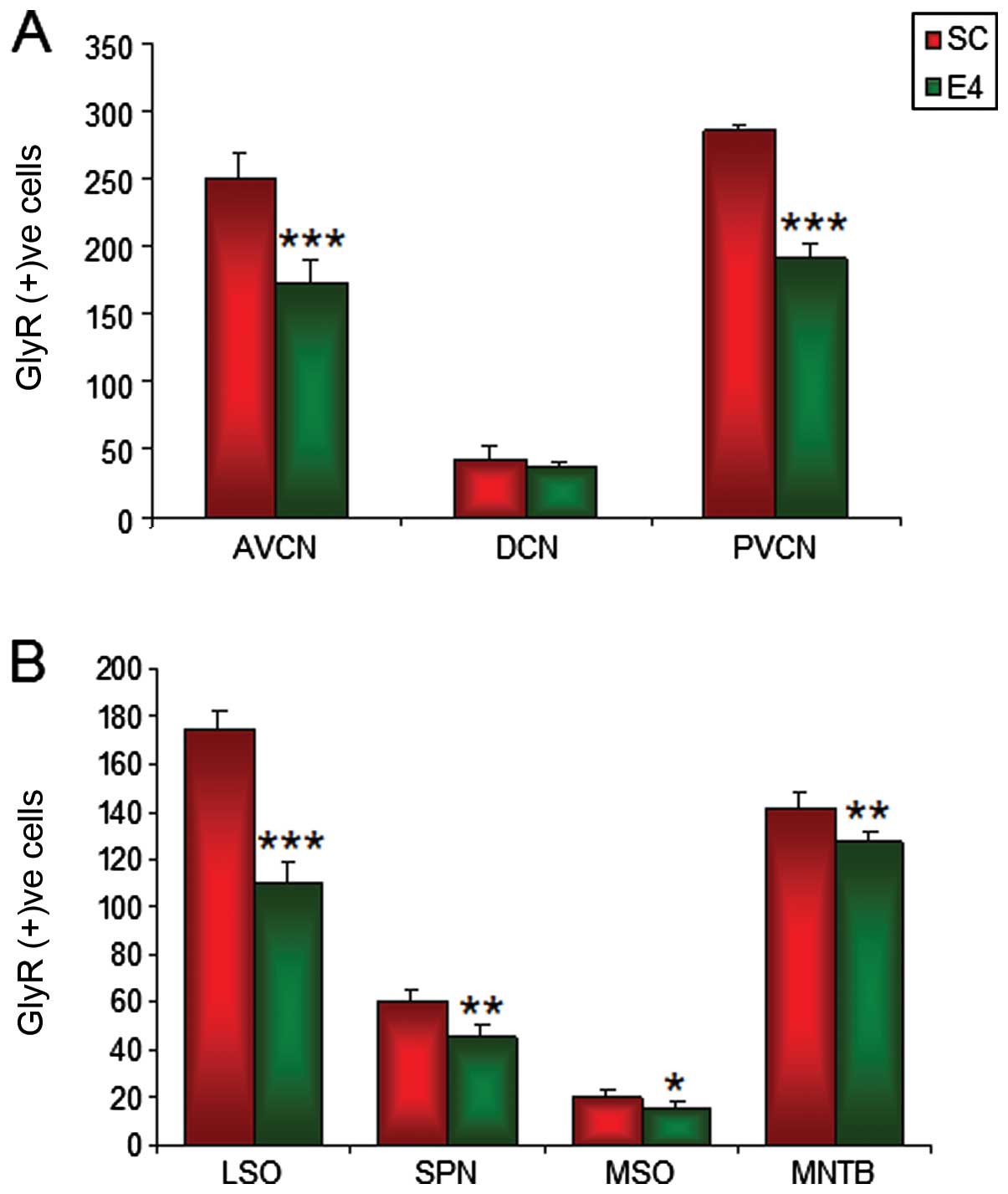 | Figure 10Graph demonstrating glycine receptor
(GlyR) α1+α2 immunoreactive cell numbers in
the CNC and SOC of sham control (SC) group (SAR 0 W/kg) and the
group exposed to radiofrequency radiation (E4 group) (SAR 4.0W/kg)
groups after three months of exposure to RF radiation at 835 MHz.
The number of GlyR immunoreactive cells was significantly decreased
in both the SOC and CN regions of the E4 group as compared with the
SC group. The data shown are the means ± SD obtained from five
different experiments. CNC, cochlear nuclear complex; AVCN,
anteroventral cochlear nucleus; PVCN, posteroventral cochlear
nucleus; DCN, dorsal cochlear nucleus; SOC, superior olivary
complex; LSO, lateral superior olive; SPN, superior paraolivary
nucleus; MSO, medial superior olive; MNTB, medial nucleus of the
trapezoid body (*P<0.05, **P<0.005,
***P<0.0001). |
In the LSO region, there was a significant
(P<0.0001) 36.85% decrease in GlyR IR from 174.2±7.49 in the SC
group to 110±8.57 in the E4 group (Fig. 10B). A significant (P<0.005)
24.33% decrease in the number of GlyR immunoreactive cells was also
observed in the SPN region, decreasing from 60±5.24 in the SC group
to 45.48±4.72 in the E4 group (Fig.
10B). The MSO displayed a 23.23% decrease (P<0.05), from
19.8±2.86 in the SC group to 15.2±2.68 in the E4 group. The MNTB
had the lowest decrease by far (10.15%; P<0.005), 141.8±6.18 in
the SC group and 127.4±4.39 in the E4 group, although the IR
decrease was more severe in the MNTB compared with the other SOC
regions (Fig. 10B).
Discussion
The structural and functional integrity of the
neurons in the auditory brainstem nuclei is maintained by auditory
input (37). Sensorineural
differentiation and hair cell damage due to noise or chemical
ototoxicity lead to changes in GlyR expression in the auditory
brainstem nuclei (38–40).
The CNC is the major site through which binaural
information converges in the CNS (19). Specific cell types within the
cochlear nuclei receive morphologically distinct axosomatic endings
of primary afferent fibers from the cochlea (21) and are highly vulnerable to
pathological alterations from peripheral organs. The impairment of
cochlear integrity brings about various morphological, biochemical
and metabolic changes throughout the auditory system (41). Cochlear damage leads to a
significant reduction in the number of Gly immunoreactive cells
(42) and long-term deficiencies
in glycinergic synaptic inhibition with the downregulation of
post-synaptic GlyR activity in the VCN and LSO by unilateral
cochlear ablation (38). All
changes in the release, uptake and binding of Gly accompanying a
significant shrinkage of the PVCN, AVCN and LSO on the ipsilateral
side have been reported in the auditory brainstem associated with
unilateral cochlear ablation (43). In accordance with the data
presented in this study, prolonged monaural conductive hearing loss
has been reported to lead to the downregulation of the GlyR
α1 subunit (44).
Furthermore, as previously demonstrated, by using in situ
hybridization of the four GlyR subunits (α1,
α2, α3 and β) in Fischer-344 rats, the mRNA
expression of the α1 and β subunits in the AVCN
decreased in the older age groups, which may contribute to central
presbycusis (45).
The decrease in the number of GlyR immunoreactive
neurons and puncta is also associated with hearing loss (33). In fact, a decrement in the number
of GlyR immunoreactive cells and GlyR IR may be related to
abnormalities in glycinergic inhibition by exposure to RF radiation
as indicated by the present data. In the present study, a
statistically significant decrease in GlyR IR in the cells, as well
as in the cell number of the CNC (31.09% decrease in AVCN; 32.79%
decrease in the PVCN) and the SOC (36.85% decrease in the LSO;
23.23% in the MSO) was also noted in the brainstems of the E4
group. Although no apoptosis was observed in the present study
(data not shown), the ABR test demonstrated an elevation of the
threshold, which was possibly the result of the induction of
cochlear damage due to exposure to RF radiation.
A decrease in GlyR IR and the loss of GlyR
immunoreactive neurons observed in the present study may result in
cochlear damage by the downregulation of glycine release and
post-synaptic GlyRs activity, which may weaken transmission at
synapses made in the cochlear nucleus (CN) (43). Consecutively, the fibers from
cells of the CN provide parallel ascending pathways to the SOC for
processing different features of sound (19).
Improper functioning, particularly in the brainstem
region, in the SOC nuclei has been reported to decrease glycine
levels, which may lead to hearing impairment (22). SOC as a convergence site of the
binaural input is likely to be involved in sound localization
(46). In the SOC of the E4
group, a significant decrease in the number of GlyR immunoreactive
cells and puncta on the somas of the principal cells, which could
be the possible source of inhibitory input of SOC, may contribute
to altered receptor activity and auditory functions, which is often
an adjunct to hearing loss (43).
Given the role of SOC in the processing of interaural phase
disparity (47) and interaural
time differences (28), a
disturbance in the GlyR expression in the auditory brainstem may be
related to hearing deficits and auditory dysfunction in the E4
group in this study. In addition, hearing impairment may also
disrupt the afferent projections from the DNLL to the ICC (48,49). According to our data, the decrease
in GlyR IR in the soma of DNLL was also statistically significant
(p<0.0001).
GlyR expression, which plays a major role in
mediating the inhibitory input of hearing processes, is
considerably decreased under the experimental conditions that were
used in this study (the conditions the mice were subjected to). The
decrease in GlyR IR and the number of GlyR immunoreactive cells in
the auditory brainstem, as observed in the E4 group, certainly
points towards the vulnerability of the auditory brainstem region
to exposure to RF radiation. Hence, prolonged exposure to RF
radiation could affect the auditory brainstem circuit, leading to
hearing impairment by the decrease of an inhibitory source, such as
Gly through intriguing signaling cascades.
The scientific investigation of the possible health
consequences of RF radiation has become an issue of international
interest and public debate, since all populations are exposed to
varying degrees of RF radiation. However, the identification of the
potential health effects of exposure to RF radiation is a difficult
task due to rapid advances in the application of RF fields in our
environment, about which anxiety and assumption are spreading.
However, according to the present study, the controversial
association between exposure to RF radiation and disease has been
addressed, although, to date, no adverse health effects have been
established as being caused by the use of commercial mobile phones.
Further research is required in order to identify the potential
effects of RF radiation and the exact molecular signal cascades
involved and obtain a better assessment of the pathology of hearing
impairment.
Acknowledgements
This study was funded by the Institute of Medical
Science Research of Dankook University Medical Center in 2011. And
This research was also supported by the Basic Science Research
Program through the National Research Foundation of Korea (NRF)
funded by the Ministry of Education, Science and Technology
(2011-0011885).
References
|
1
|
Frey AH: Headaches from cellular
telephones: are they real and what are the implications. Environ
Health Perspect. 106:101–103. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Borbély AA, Huber R, Graf T, Fuchs B,
Gallmann E and Achermann P: Pulsed high-frequency electromagnetic
field affects human sleep and sleep electroencephalogram. Neurosci
Lett. 275:207–210. 1999.PubMed/NCBI
|
|
3
|
Manikonda PK, Rajendra P, Devendranath D,
Gunasekaran B, Channakeshava, Aradhya RS, Sashidhar RB and
Subramanyam C: Influence of extremely low frequency magnetic fields
on Ca2+signaling and NMDA receptor functions in rat
hippocampus. Neurosci Lett. 413:145–149. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Mausset AL, de Seze R, Montpeyroux F and
Privat A: Effects of radiofrequency exposure on the GABAergic
system in the rat cerebellum: clues from semiquantitative
immunohistochemistry. Brain Res. 912:33–46. 2001. View Article : Google Scholar
|
|
5
|
Salford LG, Brun AE, Eberhardt JL,
Malmgren L and Persson BR: Nerve cell damage in mammalian brain
after exposure to microwaves from GSM mobile phones. Environ Health
Perspect. 111:881–883. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
6
|
Maskey D, Pradhan J, Aryal B, Lee CM, Choi
IY, Park KS, Kim SB, Kim HG and Kim MJ: Chronic 835-MHz
radiofrequency exposure to mice hippocampus alters the distribution
of calbindin and GFAP immunoreactivity. Brain Res. 1346:237–246.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Bas O, Odaci E, Kaplan S, Acer N, Ucok K
and Colakoglu S: 900 MHz electromagnetic field exposure affects
qualitative and quantitative features of hippocampal pyramidal
cells in the adult female rat. Brain Res. 1265:178–185. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ammari M, Brillaud E, Gamez C, Lecomte A,
Sakly M, Abdelmelek H and de Seze R: Effect of a chronic GSM 900
MHz exposure on glia in the rat brain. Biomed Pharmacother.
62:273–281. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Maskey D, Kim HJ, Kim HG and Kim MJ:
Calcium-binding proteins and GFAP immunoreactivity alterations in
murine hippocampus after 1 month of exposure to 835 MHz
radiofrequency at SAR values of 1.6 and 4.0 W/kg. Neurosci Lett.
506:292–296. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Mausset-Bonnefont AL, Hirbec H, Bonnefont
X, Privat A, Vignon J and de Seze R: Acute exposure to GSM 900-MHz
electromagnetic fields induces glial reactivity and biochemical
modifications in the rat brain. Neurobiol Dis. 17:445–454. 2001.
View Article : Google Scholar
|
|
11
|
Vater M, Habbicht H, Kössl M and Grothe B:
The functional role of GABA and glycine in monaural and binaural
processing in the inferior colliculus of horseshoe bats. J Comp
Physiol A. 171:541–553. 1992. View Article : Google Scholar
|
|
12
|
Caird DM, Palmer AR and Rees A: Binaural
masking level difference effects in single units of the guinea pig
inferior colliculus. Hear Res. 57:91–106. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wu SH and Kelly JB: Physiological evidence
for ipsilateral inhibition in the lateral superior olive: synaptic
responses in mouse brain slice. Hear Res. 73:57–64. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kotak VC and Sanes DH: Developmental
influence of glycinergic transmission: regulation of
NMDAreceptor-me diated EPSPs. J Neurosci. 16:1836–1843.
1996.PubMed/NCBI
|
|
15
|
Sanes DH and Takács C: Activity-dependent
refinement of inhibitory connections. Eur J Neurosci. 5:570–574.
1993. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wenthold RJ, Huie D, Altschuler RA and
Reeks KA: Glycine immunoreactivity localized in the cochlear
nucleus and superior olivary complex. Neuroscience. 22:897–912.
1987. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Moore MJ and Caspary DM: Strychnine blocks
binaural inhibition in lateral superior olivary neurons. J
Neurosci. 3:237–242. 1983.PubMed/NCBI
|
|
18
|
Wenthold RJ: Neurotransmitters of
brainstem auditory nuclei. Neurobiology of Hearing: The Central
Auditory System. Altschuler RA, Bobbin RP, Clopton BM and Hoffman
DW: Raven Press; New York: pp. 121–139. 1991
|
|
19
|
Cant NB and Benson CG: Parallel auditory
pathways: projection patterns of the different neuronal populations
in the dorsal and ventral cochlear nuclei. Brain Res Bull.
60:457–474. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Webster DB and Trune DR: Cochlear nuclear
complex of mice. Am J Anat. 163:103–130. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ryugo DK and Parks TN: Primary innervation
of the avian and mammalian cochlear nucleus. Brain Res Bull.
60:435–456. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Buras ED, Holt AG, Griffith RD, Asako M
and Altschuler RA: Changes in glycine immunoreactivity in the rat
superior olivary complex following deafness. J Comp Neurol.
494:179–189. 2006. View Article : Google Scholar
|
|
23
|
Kavanagh GL and Kelly JB: Midline and
lateral field sound localization in the ferret (Mustela
putorius): contribution of the superior olivary complex. J
Neurophysiol. 67:1643–1658. 1992.PubMed/NCBI
|
|
24
|
O’Neill WE, Zettel ML, Whittemore KR and
Frisina RD: Calbindin D-28k immunoreactivity in the medial nucleus
of the trapezoid body declines with age in C57BL/6, but not
CBA/CaJ, mice. Hear Res. 112:158–166. 1997.PubMed/NCBI
|
|
25
|
Irvine DRF: Physiology of the auditory
brainstem. The Mammalian Auditory Pathway: Neurophysiology. Popper
AN and Fay RR: Springer; New York: pp. 153–231. 1992, View Article : Google Scholar
|
|
26
|
Oliver DL and Huerta MF: Inferior and
superior colliculi. The Mammalian Auditory Pathway: Neuroanatomy.
Webster DB, Popper AN and Fay RR: Springer; New York: pp. 168–221.
1992, View Article : Google Scholar
|
|
27
|
Shneiderman A and Henkel CK: Banding of
lateral superior olivary nucleus afferents in the inferior
colliculus: a possible substrate for sensory integration. J Comp
Neurol. 266:519–534. 1987. View Article : Google Scholar
|
|
28
|
Grothe B and Sanes DH: Bilateral
inhibition by glycinergic afferents in the medial superior olive. J
Neurophysiol. 69:1192–1196. 1993.PubMed/NCBI
|
|
29
|
Wu SH and Kelly JB: Inhibition in the
superior olivary complex: pharmacological evidence from mouse brain
slice. J Neurophysiol. 73:256–269. 1995.PubMed/NCBI
|
|
30
|
Savtchouk I and Liu SJ: Remodeling of
synaptic AMPA receptor subtype alters the probability and pattern
of action potential firing. J Neurosci. 31:501–511. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Banks MI and Smith PH: Intracellular
recordings from neurobiotin-labeled cells in brain slices of the
rat medial nucleus of the trapezoid body. J Neurosci. 12:2819–2837.
1992.PubMed/NCBI
|
|
32
|
Kulesza RJ Jr and Berrebi AS: The superior
paraolivary nucleus of the rat is a GABAergic nucleus. J Assoc Res
Otolaryngol. 1:255–269. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Willott JF and Turner JG: Neural
plasticity in the mouse inferior colliculus: relationship to
hearing loss, augmented acoustic stimulation, and prepulse
inhibition. Hear Res. 147:275–281. 2000. View Article : Google Scholar
|
|
34
|
Ozturan O, Erdem T, Miman MC, Kalcioglu MT
and Oncel S: Effects of the electromagnetic field of mobile
telephones on hearing. Acta Otolaryngol. 122:289–293. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kizilay A, Ozturan O, Erdem T, Kalcioglu T
and Miman MC: Effects of chronic exposure of electromagnetic fields
from mobile phones on hearing in rats. Auris Nasus Larynx.
30:239–245. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Aran JM, Carrere N, Dulou PE, Larrieu S,
Letenneur L, Veyret B and Dulon D: Effects of exposure of the ear
to GSM microwaves: in vivo and in vitro experimental studies. Int J
Audiol. 43:454. 2004.PubMed/NCBI
|
|
37
|
Rubel EW, Hyson RL and Durham D: Afferent
regulation of neurons in the brain stem auditory system. J
Neurobiol. 21:169–196. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Potashner SJ, Suneja SK and Benson CG:
Altered glycinergic synaptic activities in guinea pig brain stem
auditory nuclei after unilateral cochlear ablation. Hear Res.
147:125–136. 2000. View Article : Google Scholar
|
|
39
|
Vale C and Sanes DH: The effect of
bilateral deafness on excitatory and inhibitory synaptic strength
in the inferior colliculus. Eur J Neurosci. 16:2394–2404. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Whiting B, Moiseff A and Rubio ME:
Cochlear nucleus neurons redistribute synaptic AMPA and glycine
receptors in response to monaural conductive hearing loss.
Neuroscience. 163:1264–1276. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Syka J: Plastic changes in the central
auditory system after hearing loss, restoration of function, and
during learning. Physiol Rev. 82:601–636. 2002.PubMed/NCBI
|
|
42
|
Potashner SJ, Suneja SK and Benson CG:
Regulation of D-aspartate release and uptake in adult brain stem
auditory nuclei after unilateral middle ear ossicle removal and
cochlear ablation. Exptl Neurol. 148:222–235. 1997. View Article : Google Scholar
|
|
43
|
Suneja SK, Benson CG and Potashner SJ:
Glycine receptors in adult guinea pig brain stem auditory nuclei:
regulation after unilateral cochlear ablation. Exp Neurol.
154:473–488. 1998. View Article : Google Scholar
|
|
44
|
Wang H, Yin G, Rogers K, Miralles C, De
Blas AL and Rubio ME: Monaural conductive hearing loss alters the
expression of the GluA3 AMPA and glycine receptor α1 subunits in
bushy and fusiform cells of the cochlear nucleus. Neuroscience.
199:438–451. 2011.PubMed/NCBI
|
|
45
|
Krenning J, Hughes LF, Caspary DM and
Helfert RH: Age-related glycine receptor subunit changes in the
cochlear nucleus of Fischer-344 rats. Laryngoscope. 108:26–31.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Masterton B, Jane JA and Diamond IT: Role
of brainstem auditory structures in sound localization. I.
Trapezoid body, superior olive, and lateral lemniscus. J
Neurophysiol. 30:341–359. 1967.PubMed/NCBI
|
|
47
|
Spitzer MW and Semple MN: Neurons
sensitive to interaural phase disparity in gerbil superior olive:
diverse monaural and temporal response properties. J Neurophysiol.
73:1668–1690. 1995.PubMed/NCBI
|
|
48
|
Franklin SR, Brunso-Bechtold JK and Henkel
CK: Unilateral cochlear ablation before hearing onset disrupts the
maintenance of dorsal nucleus of the lateral lemniscus projection
patterns in the rat inferior colliculus. Neuroscience. 143:105–115.
2006. View Article : Google Scholar
|
|
49
|
Franklin SR, Brunso-Bechtold JK and Henkel
CK: Bilateral cochlear ablation in postnatal rat disrupts
development of banded pattern of projections from the dorsal
nucleus of the lateral lemniscus to the inferior colliculus.
Neuroscience. 154:346–354. 2008. View Article : Google Scholar : PubMed/NCBI
|