Introduction
Endometrial carcinomas are the most common
gynecological malignancies and the fifth most common cancer in
women worldwide (1). Endometrial
carcinomas can be subdivided into two main categories: type I,
estrogen-dependent endometrioid carcinoma; and type II,
estrogen-independent non-endometrioid carcinoma (2,3).
Studies have shown that exposure to high levels of
estrogen is an important risk factor for endometrial carcinomas
(2). Endometrioid carcinoma
represents ~80–85% of all endometrial malignancies. Previous
studies have identified various genetic alterations in endometrioid
carcinoma, including silencing of the phosphatase and tensin
homolog deleted on chromosome 10 (PTEN) gene, microsatellite
instability (MSI), and mutations in K-ras and/or
PIK3CA (3–6).
While the majority of endometrial carcinomas are
diagnosed at an early stage, resulting in a favorable prognosis,
women diagnosed with advanced or recurrent disease have much lower
survival rates and limited adjuvant treatment options. Over the
past few decades, survival rates in patients with advanced disease
have not improved sufficiently.
Our recent genome-wide sequencing analyses of all
exons and transcriptomes in uterine serous carcinomas (USCs) showed
that approximately half of all USC cases harbor either somatic
mutations in F-box and WD repeat domain-containing 7 (FBXW7)
or gene amplification in CCNE1 (encoding cyclin E1)
(7). The cyclin E-FBXW7 pathway is
thought to be one of the most important pathways in USC development
(7).
FBXW7 is the substrate recognition component of the
Skp1-Cul1-F-box (SCF) ubiquitin-ligase and is located within 4q32,
a chromosomal region that is commonly deleted in cancers (8–10).
FBXW7 acts as a tumor suppressor by targeting several oncogenic
regulators of proliferation, growth and apoptosis for proteasomal
degradation. These include cyclin E, c-MYC, Notch and MCL1
(11–14). Furthermore, lower expression of
FBXW7 contributes to lymph node metastasis, tumor size and poor
prognosis in gastric cancer (15).
DNA copy number alterations, including
amplification, deletion, and aneuploidy in chromosomes, are the
hallmarks of neoplasia (16).
Amplification of chromosomal regions plays a critical role in tumor
development. Increases in the copy numbers of oncogenes promote
initiation and progression of a variety of solid tumors, whereas
amplification of genes that modify or detoxify chemotherapeutic
agents can lead to drug resistance and is associated with tumor
recurrence (17,18). Well-known amplified oncogenes
include c-myc, ERBB2, EGFR, AKT2, CCND1 and
CCNE1. Studies on these genes have not only provided new
insights into the mechanisms of cancer development but also
revealed significant translational implications. For example, ERBB2
and EGFR are molecular targets of the humanized antibodies
trastuzumab (Herceptin) and matuzumab, respectively, which are used
in the treatment of breast and lung cancer.
Although CCNE1 amplifications and FBXW7
mutations are frequently present in serous-type endometrial cancer,
the clinicopathological and prognostic roles of these modifications
in endometrioid-type endometrial cancer are still unclear.
Inactivating mutations in tumor suppressors could participate not
only in tumor initiation, but also in tumor progression and
response to therapy. In the present study, we examined the
prognostic and clinicopathological significance of CCNE1
amplification/expression and loss of FBXW7 expression in
endometrial carcinoma by investigating the relationship between
CCNE1 amplification/expression or FBXW7 expression and
various clinicopathological variables in endometrial carcinoma. In
addition, we compared phenotypes in cultured endometrial carcinoma
cells with variations in CCNE1 expression levels after transfection
with small interfering RNA (siRNA) targeting CCNE1.
Materials and methods
Tissue samples
Formalin-fixed, paraffin-embedded tissue samples
from 108 endometrioid-type endometrial carcinomas were used in this
study. Samples were obtained from the Department of Obstetrics and
Gynecology at the Shimane University Hospital. Diagnosis was based
on conventional morphological examination of hematoxylin and eosin
(H&E)-stained sections. Cancer patients had received
appropriate therapy at Shimane University Hospital between January
1998 and August 2010. Acquisition of tissue specimens and clinical
information was approved by the institutional review board of
Shimane University. Endometrial carcinomas were classified
according to the surgical staging system of the International
Federation of Gynecology and Obstetrics (FIGO). Invasive carcinomas
were divided by stage, with 76 patients having stage I disease, 9
patients having stage II disease, 17 patients having stage III
disease, and 6 patients having stage IV disease. All tumors were
classified histologically according to the World Health
Organization criteria. The median patient age was 60 years (range,
26–85 years). Primary surgeries included total hysterectomy in 39
patients, modified radical hysterectomy in 60 patients and radical
hysterectomy in 9 patients. Systemic retroperitoneal
lymphadenectomy was performed in ~90% of patients. Patients with
risk factors for recurrence (e.g., deep myometrial invasion,
cervical invasion, lymph node metastasis, lymphovascular space
invasion and positive peritoneal cytology) underwent postoperative
adjuvant external radiotherapy and/or 4–6 cycles of CAP therapy
(cisplatin 100 mg/m2, doxorubicin 40 mg/m2,
cyclophosphamide 500 mg/m2) or TC therapy (paclitaxel
175 mg/m2, carboplatin according to Chatelut's formula -
area under the curve = 5 mg/m2). All 108 patients were
included in the survival analysis. The follow-up period ranged from
5 to 139 months, with a median of 52 months.
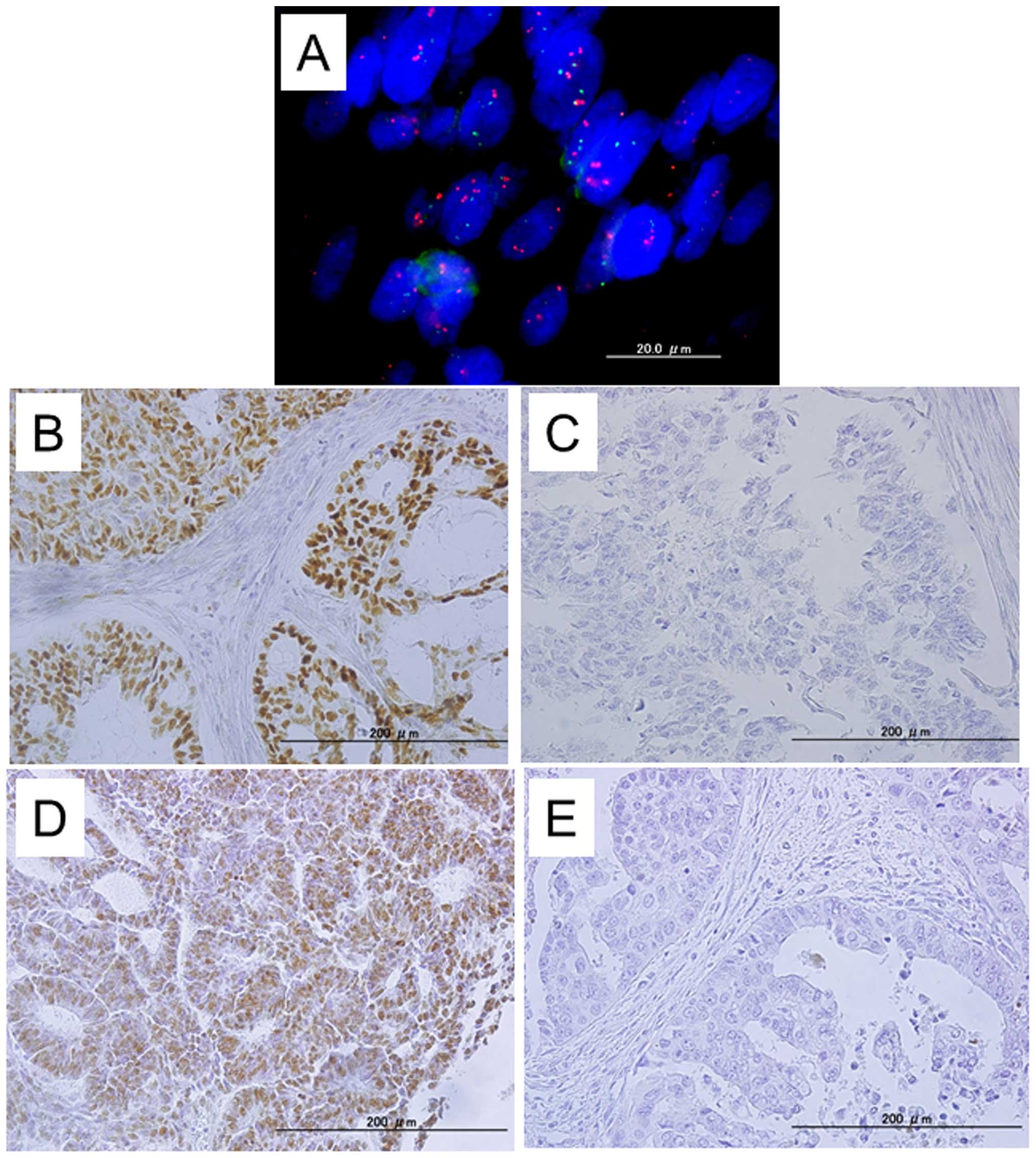
Fluorescence in situ hybridization
(FISH)
BAC clones (RP11-345J21 and CTD-3005A16) containing
the genomic sequences of the 19q12 amplicon at 15.00–15.25 Mb were
purchased from Bacpac Resources Center (Children's Hospital,
Oakland, CA, USA) and Invitrogen (Carlsbad, CA, USA). Bac clones
located at Chr2q11.2 (e.g., RP11-127K18 and RP11-629A22) or at
Ch19P12 (CTD-2518O18) were used to generate reference probes.
RP11-127K18, RP11-629A22 and CTD-2518O18 were
labeled by nick translation with biotin-dUTP; RP11-345J21 and
CTD-3005A16 were labeled similarly with digoxigenin-dUTP. To detect
biotin-labeled and digoxigenin-labeled signals, slides were first
incubated with FITC-avidin (Vector Laboratories, Burlingame, CA,
USA) and an anti-digoxigenin mouse antibody (Roche Molecular
Biochemicals, Mannheim, Germany). Slides were subsequently
incubated with a biotinylated anti-avidin antibody (Vector
Laboratories) and tetramethylrhodamine B isothiocyanate
(TRITC)-conjugated rabbit anti-mouse antibodies (Sigma, St. Louis,
MO, USA). The final incubation was with FITC-avidin and
TRITC-conjugated goat anti-rabbit antibodies (Sigma). Slides were
counterstained with 4′,6′-diamidino-2-phenylindole stain
(Sigma).
Fluorescence in situ hybridization (FISH)
signals were evaluated with an Olympus fluorescence microscope BX41
(Olympus, Tokyo, Japan) by two individuals who were blinded to the
treatment history of each patient. Separate narrow band pass
filters were used for detection of tetramethylrhodamine B
isothiocyanate, FITC, and 4′,6′-diamidino-2-phenylindole staining
signals. Using a x60 objective lens, ~100 tumor cells were examined
for each specimen, and the numbers of fluorescent signals within
tumor cells from the CCNE1 gene BAC probe and chromosome
2q11.2 or 19p12 reference BAC probe were recorded. Amplification of
CCNE1 was defined as the ratio of CCNE1 BAC probe
signals to chromosome 2 or chromosome 19 centromeric reference BAC
probe signals of 2:1 or more.
Immunohistochemistry
Paraffin-embedded tissues were organized into tissue
microarrays, which were made by removing tumor cores (3 mm in
diameter) from each block. Selection of the area to core was made
by a gynecologic oncologist (K.N.) and pathology technician (K.I.)
and was based on a review of the H&E slides.
Expression of CCNE1 and FBXW7 was assessed by
immunohistochemistry and/or western blot analysis. The antibodies
used in the present study were mouse monoclonal antibodies
targeting CCNE1 (Zymed Laboratories-Invitrogen, Carlsbad, CA, USA)
and mouse monoclonal antibodies targeting FBXW7 (Abcam, Cambridge,
MA, USA). Immunohistochemistry studies for CCNE1 and FBXW7 were
performed on tissue microarrays at a dilution of 1:250 or 1:50
followed by detection with the EnVision+ system using the
peroxidase method (Dako, Carpinteria, CA, USA). After antigen
retrieval in sodium citrate buffer, slides were incubated with
antibodies overnight at 4°C. Slides for all samples were evaluated
with a light microscope by two researchers; the researchers were
blinded to the clinicopathological factors. The antibody staining
intensity was then analyzed in glands using the HSCORE (19). The modified HSCORE was calculated
as follows: HSCORE = ∑Pi(i), where
i is the intensity of staining (0, undetectable, 1, weakly
positive, 2, moderately positive, 3, intensely positive) and
Pi is a score based on the percentage of stained
cells for each intensity, varying from 0 to 100%.
Immunohistochemistry methods for ARID1A, PTEN, MLH1,
p53, HER2 ER and progesterone receptor (PR), as well as evaluation
criteria, have been previously described (20,21).
Cell culture and cell lines
The human endometrial carcinoma cell lines HEC251
(endometrioid carcinoma), HEC50B (endometrioid carcinoma G3), and
HEC108 (endometrioid carcinoma) were obtained from the Japanese
Health Science Research Resources Bank (Osaka, Japan). HHUA
(endometrioid carcinoma) and JHUC-1 (endometrial carcinosarcoma)
cells were obtained from Riken Bioresource Center (Ibaragi,
Japan).
Western blot analysis
Cell lysates were prepared by dissolving cell
pellets in Laemmli sample buffer (Bio-Rad Laboratories, Hercules,
CA, USA) supplemented with 5% beta-mercap-toethanol (Sigma).
Western blot analysis was performed on endometrial carcinoma cell
lines, including HEC251, HEC50B, HHUA, JHUC-1 and HEC108 cells.
Similar amounts of total protein from each lysate were loaded and
separated on 10% Tris-Glycine-SDS polyacrylamide gels (Novex, San
Diego, CA, USA) and electroblotted to Millipore Immobilon-P
polyvinylidene difluoride membranes (Millipore, Bedford, MA, USA).
Membranes were probed with anti-CCNE1 antibodies (1:1,000 dilution;
Zymed Laboratories-Invitrogen) followed by peroxidase-conjugated
anti-mouse or anti-rabbit immunoglobulin (1:20,000 dilution). The
same membrane was probed with antibodies targeting GAPDH (1:10,000
dilution; Cell Signaling Technology, Beverly, MA, USA) as a loading
control. Western blots were developed by chemiluminescence (Pierce,
Rockford, IL, USA).
siRNA-mediated knockdown of CCNE1 gene
expression
Two siRNAs that targeted CCNE1 were designed
with the following sense sequences: UCAGUUGACAGUGUACAAUGCCUTT and
UGACUUACAUGAAGUGCUACUGCCG. Control siRNA (luciferase siRNA) was
purchased from Integrated DNA Technologies (Coralville, IA, USA).
Cells were seeded onto 96-well plates and transfected with siRNAs
using Oligo-fectamine (Invitrogen). Cell numbers were determined
indirectly using the
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-tetrazolium bromide (MTT)
assay 72 h after transfection (22).
Cell proliferation assay
Cells were seeded in 96-well plates at a density of
3,000 cells/well. Cell numbers were determined indirectly using MTT
assays (22). Data were expressed
as the mean ± standard deviation (SD) from triplicate
determinations.
Statistical methods for clinical
correlations
Overall survival was calculated from the date of
diagnosis to the date of death or last follow-up. Patients with and
without CCNE1 or FBXW7 expression or CCNE1-amplified tumors
had similar ages and performance status distributions. Statistical
significance was determined by the log-rank test. Data were
censored when patients were lost to follow-up.
Results
Relationships among CCNE1 amplification,
CCNE1 protein expression, FBXW7 protein expression and
clinicopathological factors
As shown in Table
I, the frequency of CCNE1 amplification in high-grade
carcinomas (G3; 20.6%: 6/29) was significantly higher than that in
low-grade tumors (G1, G2; 3.8%: 3/79; P=0.0087). CCNE1
amplification was also correlated with lymphovascular space
invasion (P=0.0258). There were no significant correlations between
CCNE1 amplification and FIGO stage (P=0.851), lymph node
metastasis (P=0.078), patient age (P=0.0817), body mass index
(P=0.265), deep myometrial invasion (P=0.256) or menopausal status
(P=0.289; Table I).
 | Table IAssociation between CCNE1 gene
amplification and clinocopathological factors in patients with
endometrial cancer. |
Table I
Association between CCNE1 gene
amplification and clinocopathological factors in patients with
endometrial cancer.
| | CCNE1gene
amplification | | CCNE1 protein
expression | | FBXW7 protein
expression | |
|---|
| |
| |
| |
| |
|---|
| Factors | Patients | Negative | Positive | P-value | Low | High | P-value | Negative | Positive | P-value |
|---|
| FIGO stage | | | | | | | | | | |
| I | 75 | 69 | 6 | 0.851 | 42 | 33 | 0.0601 | 41 | 34 | 0.2439 |
| II, III, IV | 33 | 30 | 3 | | 12 | 21 | | 22 | 11 | |
| Grade | | | | | | | | | | |
| G1, G2 | 79 | 76 | 3 | 0.0087 | 45 | 34 | 0.0169 | 45 | 34 | 0.228 |
| G3 | 29 | 23 | 6 | | 9 | 20 | | 18 | 11 | |
| Lymph node
metastasis | | | | | | | | | | |
| Negative | 93 | 87 | 6 | 0.078 | 47 | 46 | 0.7808 | 53 | 40 | 0.4805 |
| Positive | 15 | 12 | 3 | | 7 | 8 | | 10 | 5 | |
| Depth (myometrial
invasion) | | | | | | | | | | |
| a, b | 67 | 63 | 4 | 0.256 | 37 | 30 | 0.1651 | 33 | 34 | 0.0144 |
| c | 41 | 36 | 5 | | 17 | 24 | | 30 | 11 | |
| Lymphovascular
space invasion | | | | | | | | | | |
| Negative | 62 | 60 | 7 | 0.0258 | 37 | 25 | 0.0195 | 31 | 31 | 0.0414 |
| Positive | 46 | 39 | 7 | | 17 | 29 | | 32 | 14 | |
| Menopause | | | | | | | | | | |
| Peri, pre | 28 | 27 | 1 | 0.289 | 19 | 9 | 0.0281 | 16 | 12 | 0.882 |
| Post | 80 | 72 | 8 | | 35 | 45 | | 47 | 33 | |
| Body mass
index | | | | | | | | | | |
| <25 | 79 | 71 | 8 | 0.265 | 41 | 38 | 0.5148 | 47 | 32 | 0.162 |
| ≥25 | 29 | 28 | 1 | | 13 | 16 | | 16 | 13 | |
| Age (years) | | | | | | | | | | |
| <60 | 54 | 52 | 2 | 0.0817 | 33 | 21 | 0.0209 | 31 | 23 | 0.038 |
| ≥60 | 54 | 47 | 7 | | 21 | 33 | | 32 | 22 | |
Patients were stratified into one of two groups
depending on the median CCNE1 immunohistochemical HSCORE. The
relationships between CCNE1 protein expression and
clinicopathological factors are shown in Table I. CCNE1 protein expression was
significantly correlated with high-grade carcinoma (P=0.0169),
lymphovascular space invasion (P=0.0195), postmenopausal status
(P=0.0281), and patient age (P=0.0209). There was no significant
correlation between CCNE1 protein expression and FIGO stage
(P=0.0601), body mass index (P=0.5148), lymph node metastasis
(P=0.7808), or deep myometrial invasion (P=0.1651).
Negative expression of FBXW7 (FBXW7
immunohistochemical HSCORE = 0) was observed in 62% (63/101) of the
analyzed tumors. Patients were stratified into one of two groups
depending on the status of the negative FBXW7 immunohistochemical
HSCORE. The relationships between FBXW7 protein expression and
clinicopathological factors are shown in Table I. Negative FBXW7 protein expression
was significantly correlated with deep myometrial invasion
(P=0.0144), lymphovascular space invasion (P=0.0414) and patient
age (P=0.038). There was no significant correlation between
negative FBXW7 protein expression and FIGO stage (P=0.2439), tumor
grade (P=0.228), lymph node metastasis (P=0.4805), postmenopausal
status (P=0.882) or body mass index (P=0.162).
Effects of CCNE1 amplification or CCNE1
protein expression on progression-free survival
Next, we evaluated the prognostic relevance of CCNE1
amplification or CCNE1 protein expression in terms of
progression-free survival. Kaplan-Meier estimates of
progression-free/overall survival are plotted in Fig. 2. Amplification of CCNE1
correlated with shorter progression-free survival in patients with
endometrial endometrioid carcinomas. A total of 108 patients were
diagnosed at stages I–IV. Among them, the 9 patients with
CCNE1 amplifications had shorter progression-free survival
than those without gene amplification (P=0.0081; log-rank test;
Fig. 2A). Univariate analysis
demonstrated that deep myometrial invasion (P=0.0008; log-rank
test), high-grade histological subtype (P<0.0001; log-rank
test), lymph node metastasis (P<0.0001; log-rank test),
lymphovascular space invasion (P=0.001; log-rank test) and
CCNE1 amplification (P=0.0081; log-rank test) correlated
with shorter progression-free survival. CCNE1 HSCORE tended to
influence progression-free survival, but this relationship was not
statistically significant (P=0.3148; Fig. 2B). We excluded lymph node
metastasis and lymphovascular space invasion from multivariate
analysis because these two factors are similar events in the
clinical setting and to prevent any issues with statistical
analyses. Therefore, for multivariate analysis, we chose FIGO
stage, tumor grade, depth of myometrial invasion and CCNE1
amplification. When the data were stratified for multivariate
analysis, high-grade histological subtype and deep myometrial
invasion remained significantly associated with shorter
progression-free survival (P=0.0003 and P=0.0142, respectively;
Table II).
 | Figure 2CCNE1 amplification is
correlated with shorter progression-free/overall survival in
patients with endometrial endometrioid carcinoma. (A) Kaplan-Meier
survival analysis showed that CCNE1 amplification (solid
line, n=9) was associated with shorter progression-free survival
than absence of CCNE1 amplification (dashed line, n=99)
(P=0.0081, log-rank test). CCNE1 overexpression was not
significantly associated with progression-free/overall survival in
patients with endometrial endometrioid carcinoma. (B) Kaplan-Meier
survival analysis showed that a high CCNE1 expression (solid line,
n=44) was associated with shorter relapse-free survival than low
CCNE1 expression; however, the difference was not
statistically significant (dashed line, n=54; P=0.3148, log-rank
test). (C) Kaplan-Meier survival analysis showed that CCNE1
amplification (solid line, n=9) was associated with shorter overall
survival than absence of CCNE1 amplification (dashed line,
n=99; P=0.0073, log-rank test). (D) Kaplan-Meier survival analysis
showed that high CCNE1 expression (solid line, n=54) was
associated with shorter overall survival than low CCNE1
expression; however, this difference was not statistically
significant (dashed line, n=54; P=0.2205, log-rank test). Negative
FBXW7 expression was not significantly associated with
progression-free/overall survival in patients with endometrial
endometrioid carcinoma. (E) Kaplan-Meier survival analysis showed
that negative FBXW7 expression (solid line, n=63) was associated
with shorter progression-free survival than positive FBXW7
expression; however, this difference was not statistically
significant (dashed line, n=54; P=0.3148, log-rank test). (F)
Kaplan-Meier survival analysis showed that negative FBXW7
expression (solid line, n=63) was associated with shorter overall
survival than positive FBXW7 expression; however, this difference
was not statistically significant (dashed line, n=54; P=0.2307,
log-rank test). |
 | Table IIUnivariate analysis of
progression-free prognostic factors in patients with endometrial
carcinoma. |
Table II
Univariate analysis of
progression-free prognostic factors in patients with endometrial
carcinoma.
| Factors | Patients | Univariate hazard
ratio | 95% CI | P-value | Multivariate hazard
ratio | 95% CI | P-value |
|---|
| FIGO stage |
| I | 75 | | | | | | |
| II, III, IV | 33 | 2.8 | 1.0–7.7 | 0.0532 | NA | NA | NA |
| Grade |
| G1, G2 | 79 | | | | | | |
| G3 | 29 | 27.1 | 6.0–122.1 | <0.0001 | 18.2 | 3.8–86.8 | 0.0003 |
| Lymph node
metastasis |
| Negative | 93 | | | | | | |
| Positive | 15 | 9.7 | 3.4–27.7 | <0.0001 | NA | NA | NA |
| Depth (myometrial
invasion) |
| a, b | 67 | | | | | | |
| c | 41 | 8.824 | 2.5–31.4 | 0.0008 | 5.3 | 1.4–19.8 | 0.0142 |
| Lymphovascular
space invasion |
| Negative | 62 | | | | | | |
| Positive | 46 | 30.5 | 4.0–233.6 | 0.001 | NA | NA | NA |
| Menopause |
| Peri, pre | 28 | | | | | | |
| Post | 80 | 5.5 | 0.7–42.0 | 0.0991 | NA | NA | NA |
| Body mass
index |
| <25 | 79 | | | | | | |
| ≥25 | 29 | 1.1 | 0.3–3.5 | 0.8597 | NA | NA | NA |
| Age (years) |
| <60 | 54 | | | | | | |
| ≥60 | 54 | 2.9 | 0.9–9.1 | 0.069 | NA | NA | NA |
| CCNE1 FISH |
| Normal | 9 | | | | | | |
| Amp | 99 | 4.7 | 1.5–14.8 | 0.0081 | 2.2 | 0.6–7.4 | 0.2175 |
| CCNE1
immunostaing |
| Low | 54 | | | | | | |
| High | 54 | 1.7 | 0.6–4.8 | 0.3148 | NA | NA | NA |
| FBXW7
immunostaing |
| Positive | 45 | | | | | | |
| Negative | 63 | 1.9 | 0.6–5.5 | 0.2637 | NA | NA | NA |
Effects of CCNE1 amplification or CCNE1
protein expression on overall survival
Next, we examined the prognostic relevance of
CCNE1 amplification or CCNE1 protein expression in terms of
overall survival. Kaplan-Meier estimates of
progression-free/overall survival are plotted in Fig. 3. Amplification of CCNE1
correlated with shorter overall survival in patients with
endometrial endometrioid carcinomas treated. A total of 108
patients were diagnosed at stages I–IV. Among them, the 9 patients
with CCNE1 amplification had shorter overall survival than
those without CCNE1 amplification (P=0.0073; log-rank test;
Fig. 2C). Univariate analysis
demonstrated that FIGO stage III or IV (P=0.035; log-rank test),
high-grade histological subtype (P<0.0001; log-rank test), deep
myometrial invasion (P=0.0008; log-rank test), lymph node
metastasis (P<0.0001; log-rank test), lymphovascular space
invasion (P=0.0011; log-rank test), and CCNE1 amplification
(P=0.0073; log-rank test) were correlated with shorter overall
survival. High CCNE1 HSCOREs tended to be associated with poor
overall survival; however, this relationship was not significant
(P=0.2205; Fig. 2D). As described
in the previous section, lymph node metastasis and lymphovascular
space invasion were excluded from multivariate analysis. Therefore,
for multivariate analysis, we chose FIGO stage, tumor grade, depth
of myometrial invasion and CCNE1 amplification. When data
were stratified for multivariate analysis, high-grade subtype, deep
myometrial invasion and CCNE1 amplification remained
significantly associated with shorter overall survival (P=0.0006,
P=0.0252 and P=0.0454, respectively; Table III).
 | Table IIIUnivariate analysis of overall
prognostic factors in patients with endometrial carcinoma. |
Table III
Univariate analysis of overall
prognostic factors in patients with endometrial carcinoma.
| Factors | Patients | Univariate hazard
ratio | 95% CI | P-value | Multivariate hazard
ratio | 95% CI | P-value |
|---|
| FIGO stage |
| I | 75 | | | | | | |
| II, III, IV | 33 | 3 | 1.1–8.4 | 0.035 | 1.4 | 0.4–4.2 | 0.5957 |
| Grade |
| G1, G2 | 79 | | | | | | |
| G3 | 29 | 26.7 | 5.9–120.3 | <0.0001 | 15.7 | 3.2–76.1 | 0.0006 |
| Lymph-node
metastasis |
| Negative | 93 | | | | | | |
| Positive | 15 | 11.7 | 4.0–33.8 | <0.0001 | NA | NA | NA |
| Depth (myometrial
invasion) |
| a, b | 67 | | | | | | |
| c | 41 | 8.8 | 2.5–31.4 | 0.0008 | 4.6 | 1.2–17.4 | 0.0252 |
| Lymphovascular
space invasion |
| Negative | 62 | | | | | | |
| Positive | 46 | 29.4 | 3.9–224.3 | 0.0011 | NA | NA | NA |
| Menopause |
| Peri, pre | 28 | | | | | | |
| Post | 80 | 5.3 | 0.7–40.2 | 0.1081 | NA | NA | NA |
| Body mass
index |
| <25 | 79 | | | | | | |
| ≥25 | 29 | 1.1 | 0.4–3.6 | 0.883 | NA | NA | NA |
| Age (years) |
| <60 | 54 | | | | | | |
| ≥60 | 54 | 2.9 | 0.9–9.1 | 0.067 | NA | NA | NA |
| CCNE1 FISH |
| Normal | 9 | | | | | | |
| Amp | 99 | 4.8 | 1.5–15.2 | 0.0073 | 3.8 | 1.0–14.0 | 0.0454 |
| CCNE1
immunostaing |
| Low | 54 | | | | | | |
| High | 54 | 1.9 | 0.7–5.4 | 0.2205 | NA | NA | NA |
| FBXW7
immunostaing |
| Positive | 45 | | | | | | |
| Negative | 63 | 1.9 | 0.7–5.7 | 0.2307 | NA | NA | NA |
Effects of FBXW7 protein expression on
disease-free/overall survival
Because FBXW7 is the substrate recognition component
of the Skp1-Cul1-F-box (SCF) ubiquitin-ligase, which targets CCNE1,
we next analyzed whether loss of FBXW7 immunostaining was related
to poor survival and whether there was a positive relationship
between loss of FBXW7 protein expression and CCNE1 protein
expression. Immunoreactivity for FBXW7 was detected in tumor cell
nuclei (Fig. 1). The mean HSCORE
of FBXW7 was 46 (range, 0–300). The CCNE1 immunohistochemical
HSCORE was not correlated with the FBXW7 immunohistochemical HSCORE
(data not shown). Negative expression of FBXW7 (FBXW7
immunohistochemical HSCORE = 0) was observed in 58.3% (63/108) of
the analyzed tumors. Patients were stratified into one of two
groups depending on the status of negative FBXW7
immunohistochemical HSCORE. Kaplan-Meier estimates of
progression-free/overall survival are plotted in Fig. 2. Negative FBXW7 expression tended
to correlate with shorter overall/progression-free survival in
patients with endometrial endometrioid carcinoma; however, this
relationship was not statistically significant (Fig. 2E and F).
Relationship between CCNE1 amplification
and PTEN, p53, HER2, MLH1 or ARID1A expression in endometrial
endometrioid carcinoma
We previously performed immunohistochemical analysis
to evaluate the status of PTEN, p53, HER2, MLH1 and ARID1A
expression among analyzed tumor samples (20). We then investigated the
correlations between the expression of these molecules and
CCNE1 amplification (Table
IV). No significant correlations were observed between
CCNE1 amplification and the expression of these proteins,
except for positive PTEN expression (P=0.041).
 | Table IVAssociation between CCNE1 gene
amplification and status of p53, hMLH1, Her2, PTEN and ARID1A in
patients with endometrial cancer. |
Table IV
Association between CCNE1 gene
amplification and status of p53, hMLH1, Her2, PTEN and ARID1A in
patients with endometrial cancer.
| | CCNE1 gene
amplification | |
|---|
| |
| |
|---|
| Factors | Patients | Normal | Amplification | P-value |
|---|
| p53 |
| Negative | 58 | 55 | 3 | 0.2005 |
| Positive | 50 | 44 | 6 | |
| hMLH1 |
| Negative | 53 | 48 | 5 | 0.6846 |
| Positive | 55 | 51 | 4 | |
| Her2 |
| Low | 40 | 36 | 4 | 0.6308 |
| High | 68 | 63 | 5 | |
| PTEN |
| Negative | 27 | 25 | 2 | 0.041 |
| Positive | 81 | 74 | 7 | |
| ARID1A |
| Negative | 27 | 26 | 1 | 0.3149 |
| Positive | 81 | 73 | 8 | |
Effects of CCNE1 knockdown on endometrial
endometrioid carcinoma in vitro
A panel of endometrial carcinoma cell lines was
analyzed for CCNE1 amplification and protein expression
status (Fig. 3A). However, there
were no CCNE1 amplification cell lines in the tested cells,
as measured by FISH analysis (data not shown). Transfection with
CCNE1 siRNA significantly reduced CCNE1 protein expression compared
with control siRNA transfection (Fig.
3B). However, CCNE1 expression status correlated with the
growth inhibition induced by CCNE1 siRNA (Fig. 3C). Reduction of CCNE1 expression
significantly inhibited cell growth in CCNE1-overexpressing cells,
including JHUC-1 and HEC108 cells. In contrast, transfection with
CCNE1 siRNA did not affect cell growth in HEC251, HEC50B and HHUA
cells, which exhibit moderate or minimal CCNE1 expression,
suggesting that endometrial carcinomas with CCNE1 overexpression
are more dependent on activation of the CCNE1-related pathway for
cell proliferation and survival than those without CCNE1
expression.
Discussion
Notably, we found that there was a significantly
higher frequency of CCNE1 amplification in high-grade
endometrial endometrioid carcinomas compared with that in low-grade
endometrial endometrioid carcinomas. This suggested that high-grade
and low-grade tumors may be distinguished based on characteristic
genetic alterations. Additionally, this observation further
supports the hypothesis that endometrial carcinoma arises from
multiple pathways (7,23). In this model, high-grade and
low-grade carcinomas develop independently from one another and are
characterized by different molecular genetic changes and gene
expression profiles (7,23).
Our findings were consistent with those of a
previous report showing a positive correlation between CCNE1
immunostaining and tumor grade (24). Taken together, these data,
including our current FISH and immunostaining analyses, suggested
that alterations in the CCNE1 gene could be a specific event
in high-grade endometrial endometrioid carcinoma.
Based on our combined FISH and immunohistochemical
analysis, we found that CCNE1 was consistently overexpressed
in all tumors exhibiting CCNE1 gene amplification (data not
shown). However, some tumors overexpressed CCNE1 protein in the
absence of CCNE1 amplification. Therefore, we concluded that
overexpression did not require gene amplification. Our findings
suggested that the mechanism of CCNE1 overexpression may be
dualistic, owing in part to gene amplification and in part to
activation at the transcriptional level.
Bio markers that can predict clinical prognosis,
including treatment response and overall survival, have substantial
clinical impact on the management of patients with cancer (25). To further explore the clinical
relevance of CCNE1 alterations in endometrial endometrioid
carcinomas, we evaluated the correlation between CCNE1
amplification or protein expression and length of
progression-free/overall survival in our population of patients
with endometrial endometrioid carcinoma. Interestingly, we found a
strong correlation between poor prognosis and CCNE1
amplification in patients with endometrial endometrioid carcinoma.
However, CCNE1 protein overexpression was associated with an
insignificant trend toward poor overall survival. CCNE1 functions
as a regulatory subunit of CDK2, which is essential for both the G1
to S-phase transition and DNA replication. Moreover, CCNE1 plays a
role in apoptosis and chromosomal instability in tumor cells
(26). Cyclin E-CDK2 acts through
phosphorylation of substrates involved in G1
progression, S-phase entry and centrosome duplication. This complex
also has kinase-independent functions involving the loading of
mini-chromosome maintenance proteins onto origins of replication as
quiescent cells enter the cell cycle (27).
CCNE1 amplification has been identified as a
mechanism for overexpression in endometrial carcinomas (24,28).
However, in the present study, we found a direct correlation
between CCNE1 amplification and poor outcomes, consistent
with the results of another report (29). To date, CCNE1 amplification
has not been shown to be explicitly associated with primary
treatment resistance. The mechanism underlying the association
between CCNE1 amplification and shorter survival is not
known; however, because mortality in patients with endometrial
cancer is directly related to recurrence of disease after
chemotherapy, it is conceivable that CCNE1 amplification may
confer resistance to chemotherapy and/or enhance cell proliferation
in chemoresistant recurrent tumors. Because the etiology of tumor
recurrence is multifactorial, it is likely that CCNE1
expression promotes the growth of tumor cells by providing them
with higher proliferative capacity and lower apoptotic activity, as
was shown in our previous study (30). Because the present study identified
a previously undescribed pattern of CCNE1
amplification/expression in endometrial endometrioid carcinoma
cells, further studies are required to elucidate the relationship
between CCNE1 gene amplification/ expression and tumor
sensitivity to chemotherapy
In the present study, the absence of FBXW7
expression in endometrial endometrioid carcinoma tissue was
associated with deep myometrial invasion and lymphovascular space
invasion, thereby potentially affecting overall survival and
progression-free survival, with marginal statistical significance.
Studies have demonstrated that FBXW7 regulates a wide range of
cancer-associated substrates, and FBXW7 mutation leads to high
expression of CCNE1, c-MYC, Notch and MCL1. c-MYC and CCNE1 are
important components of the cell cycle machinery that are
frequently deregulated in cancer and possess strong oncogenic
activity. FBXW7 loss and c-MYC upregulation may play roles in the
aggressive biological behaviors of cancer, which may in turn induce
cell proliferation (31). Cells
with increased c-MYC and reduced FBXW7 expression have been shown
to be more invasive and to be associated with poor prognosis due to
the high levels of matrix metalloproteinases (MMP2 and MMP9)
(32,33). Indeed, the tendency to undergo
lymph node and intrahepatic metastases may be correlated with these
substrates in intrahepatic cholangiocarcinoma. Furthermore,
mammalian target of rapamycin (mTOR) has been shown to function as
an oncoprotein, controlling many cellular processes, such as cell
proliferation and division. Interestingly, mTOR was also found to
be degraded through an FBXW7-dependent mechanism (34). As a major regulator of a set of
oncoproteins, FBXW7 has been shown to be lost and mutated in
various human cancers, such as human hepatocellular carcinoma and
breast carcinoma (35,36). Recently, Mitsuhashi et al
(37) reported that the
Notch1-JAG1 axis is related to invasive properties and a poor
prognosis in patients with endometrial cancer. Peiró et al
(38) reported that mTOR
expression is also related to poor outcomes in endometrial cancer.
Taken together, the above findings provided evidence for the
mechanisms through which loss of FBXW7 expression was related to
poor outcomes in endometrial endometrioid carcinoma in the present
study.
Molecular genetic evidence indicates that
endometrial carcinomas are likely to develop as a result of a
multistep process of oncogenic activation and tumor suppressor
inactivation (39,40). In this study, we focused on some
important genetic events of endometrial endometrioid carcinoma,
i.e., inactivation of the tumor-suppressor genes PTEN,
p53 and ARID1A; overexpression of the oncogene HER2;
and MSI (5,40–42).
In addition, we also analyzed the association between PTEN, HER2,
p53, MLH1 and ARID1A expression and CCNE1 amplification. No
significant relationships were observed between the expression of
these proteins and CCNE1 amplification, except for positive
PTEN expression; these data indicated that CCNE1
amplification was independent of p53, HER2, MLH1 and ARID1A
expression in endometrial endometrioid carcinoma. Notably,
CCNE1 amplification and PTEN loss may occur in a mutually
exclusive manner. However, assessment of mutational status via
direct sequencing was not performed in this study; only
immunohistochemical analysis was carried out to compare different
molecular events with CCNE1 amplification status. Thus,
further studies are required to fully explore the relationship
between CCNE1 amplification and other genetic events
occurring in endometrial endometrioid carcinomas.
Cancer cells contain multiple genetic and epigenetic
abnormalities. Despite this complexity, their growth and survival
can often be impaired by inactivation of a single oncogene. This
phenomenon, called ‘oncogene addiction’, provides a rationale for
molecular targeted therapy (43).
Based on this theory and our in vivo and in vitro
findings, we propose that patients with endometrial endometrioid
carcinoma having CCNE1 amplification may benefit from CDK
inhibitor therapy if the disease recurs after conventional platinum
and taxane or doxorubicin chemotherapy or from CDK inhibitor
therapy as the primary therapy with the conventional combination of
platinum and taxane or doxorubicin. To date, the CDK inhibitor
E7070 has fared poorly in clinical trials for melanoma and lung
cancer (44,45). However, its favorable therapeutic
index and high selectivity may outweigh its shortcomings in
CCNE1-amplified endometrial endometrioid carcinoma.
Therefore, we recommend that patients participating in future
clinical trials evaluating the efficacy of CDK inhibitors for
endometrial endometrioid carcinoma should be stratified based on
CCNE1 amplification status.
In summary, we have demonstrated that phenotypic
changes in endometrial endometrioid carcinomas in response to
CCNE1 inactivation depend on the expression status of
CCNE1. The findings in the present study provide important
insights into the biological roles of the CCNE1 signaling pathway
in endometrial endometrioid carcinomas. Additionally, our
observations have important therapeutic implications for patients
with endometrial endometrioid carcinoma having CCNE1
amplification. Endometrial endometrioid carcinomas with
CCNE1 amplification are clinically high-grade carcinomas
with aggressive behavior (46,47).
Therefore, detection of CCNE1 amplification in endometrial
endometrioid carcinomas may identify patients who will benefit from
a CCNE1-specific inhibitor.
References
|
1
|
Pisani P, Bray F and Parkin DM: Estimates
of the world-wide prevalence of cancer for 25 sites in the adult
population. Int J Cancer. 97:72–81. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Akhmedkhanov A, Zeleniuch-Jacquotte A and
Toniolo P: Role of exogenous and endogenous hormones in endometrial
cancer: Review of the evidence and research perspectives. Ann NY
Acad Sci. 943:296–315. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Matias-Guiu X, Catasus L, Bussaglia E,
Lagarda H, Garcia A, Pons C, Muñoz J, Argüelles R, Machin P and
Prat J: Molecular pathology of endometrial hyperplasia and
carcinoma. Hum Pathol. 32:569–577. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Shiozawa T and Konishi I: Early
endometrial carcinoma: Clinicopathology, hormonal aspects,
molecular genetics, diagnosis, and treatment. Int J Clin Oncol.
11:13–21. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tashiro H, Blazes MS, Wu R, Cho KR, Bose
S, Wang SI, Li J, Parsons R and Ellenson LH: Mutations in PTEN are
frequent in endometrial carcinoma but rare in other common
gynecological malignancies. Cancer Res. 57:3935–3940.
1997.PubMed/NCBI
|
|
6
|
Oda K, Stokoe D, Taketani Y and McCormick
F: High frequency of coexistent mutations of PIK3CA and PTEN genes
in endometrial carcinoma. Cancer Res. 65:10669–10673. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kuhn E, Wu RC, Guan B, Wu G, Zhang J, Wang
Y, Song L, Yuan X, Wei L, Roden RB, et al: Identification of
molecular pathway aberrations in uterine serous carcinoma by
genome-wide analyses. J Natl Cancer Inst. 104:1503–1513. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Welcker M and Clurman BE: FBW7 ubiquitin
ligase: A tumour suppressor at the crossroads of cell division,
growth and differentiation. Nat Rev Cancer. 8:83–93. 2008.
View Article : Google Scholar
|
|
9
|
Spruck CH, Strohmaier H, Sangfelt O,
Müller HM, Hubalek M, Müller-Holzner E, Marth C, Widschwendter M
and Reed SI: hCDC4 gene mutations in endometrial cancer. Cancer
Res. 62:4535–4539. 2002.PubMed/NCBI
|
|
10
|
Knuutila S, Aalto Y, Autio K, Björkqvist
AM, El-Rifai W, Hemmer S, Huhta T, Kettunen E, Kiuru-Kuhlefelt S,
Larramendy ML, et al: DNA copy number losses in human neoplasms. Am
J Pathol. 155:683–694. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Strohmaier H, Spruck CH, Kaiser P, Won KA,
Sangfelt O and Reed SI: Human F-box protein hCdc4 targets cyclin E
for proteolysis and is mutated in a breast cancer cell line.
Nature. 413:316–322. 2001. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yada M, Hatakeyama S, Kamura T, Nishiyama
M, Tsunematsu R, Imaki H, Ishida N, Okumura F, Nakayama K and
Nakayama KI: Phosphorylation-dependent degradation of c-Myc is
mediated by the F-box protein Fbw7. EMBO J. 23:2116–2125. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Izumi N, Helker C, Ehling M, Behrens A,
Herzog W and Adams RH: Fbxw7 controls angiogenesis by regulating
endothelial Notch activity. PLoS One. 7:e411162012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Inuzuka H, Shaik S, Onoyama I, Gao D,
Tseng A, Maser RS, Zhai B, Wan L, Gutierrez A, Lau AW, et al:
SCF(FBW7) regulates cellular apoptosis by targeting MCL1 for
ubiquitylation and destruction. Nature. 471:104–109. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yokobori T, Mimori K, Iwatsuki M, Ishii H,
Onoyama I, Fukagawa T, Kuwano H, Nakayama KI and Mori M:
p53-Altered FBXW7 expression determines poor prognosis in gastric
cancer cases. Cancer Res. 69:3788–3794. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Debies MT and Welch DR: Genetic basis of
human breast cancer metastasis. J Mammary Gland Biol Neoplasia.
6:441–451. 2001. View Article : Google Scholar
|
|
17
|
Albertson DG, Collins C, McCormick F and
Gray JW: Chromosome aberrations in solid tumors. Nat Genet.
34:369–376. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wang TL, Diaz LA Jr, Romans K, Bardelli A,
Saha S, Galizia G, Choti M, Donehower R, Parmigiani G, Shih IeM, et
al: Digital karyotyping identifies thymidylate synthase
amplification as a mechanism of resistance to 5-fluorouracil in
metastatic colorectal cancer patients. Proc Natl Acad Sci USA.
101:3089–3094. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Mao TL, Seidman JD, Kurman RJ and Shih
IeM: Cyclin E and p16 immunoreactivity in epithelioid trophoblastic
tumor--an aid in differential diagnosis. Am J Surg Pathol.
30:1105–1110. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Rahman M, Nakayama K, Rahman MT, Katagiri
H, Katagiri A, Ishibashi T, Ishikawa M, Iida K and Miyazaki K:
Clinicopathologic analysis of loss of AT-rich interactive domain 1A
expression in endometrial cancer. Hum Pathol. 44:103–109. 2013.
View Article : Google Scholar
|
|
21
|
Rahman MT, Nakayama K, Ishikawa M, Rahman
M, Katagiri H, Katagiri A, Ishibashi T, Iida K and Miyazaki K:
Fatty acid synthase is a potential therapeutic target in estrogen
receptor-/ progesterone receptor-positive endometrioid endometrial
cancer. Oncology. 84:166–173. 2013. View Article : Google Scholar
|
|
22
|
Nakayama K, Miyazaki K, Kanzaki A,
Fukumoto M and Takebayashi Y: Expression and cisplatin sensitivity
of copper-transporting P-type adenosine triphosphatase (ATP7B) in
human solid carcinoma cell lines. Oncol Rep. 8:1285–1287.
2001.PubMed/NCBI
|
|
23
|
Kandoth C, Schultz N, Cherniack AD, Akbani
R, Liu Y, Shen H, Robertson AG, Pashtan I, Shen R, Benz CC, et al;
Cancer Genome Atlas Research Network. Integrated genomic
characterization of endometrial carcinoma. Nature. 497:67–73. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kato N, Watanabe J, Jobo T, Nishimura Y,
Fujisawa T, Kamata Y and Kuramoto H: Immunohistochemical expression
of cyclin E in endometrial adenocarcinoma (endometrioid type) and
its clinicopathological significance. J Cancer Res Clin Oncol.
129:222–226. 2003.PubMed/NCBI
|
|
25
|
Scott M and Hall PA: Prognostic and
predictive factors. Methods Mol Med. 97:1–11. 2004.PubMed/NCBI
|
|
26
|
Hwang HC and Clurman BE: Cyclin E in
normal and neoplastic cell cycles. Oncogene. 24:2776–2786. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Geng Y, Lee YM, Welcker M, Swanger J,
Zagozdzon A, Winer JD, Roberts JM, Kaldis P, Clurman BE and
Sicinski P: Kinase-independent function of cyclin E. Mol Cell.
25:127–139. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Cassia R, Moreno-Bueno G,
Rodríguez-Perales S, Hardisson D, Cigudosa JC and Palacios J:
Cyclin E gene (CCNE) amplification and hCDC4 mutations in
endometrial carcinoma. J Pathol. 201:589–595. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Santala S, Talvensaari-Mattila A, Soini Y
and Santala M: Cyclin E expression correlates with cancer-specific
survival in endometrial endometrioid adenocarcinoma. Anticancer
Res. 35:3393–3397. 2015.PubMed/NCBI
|
|
30
|
Nakayama N, Nakayama K, Shamima Y,
Ishikawa M, Katagiri A, Iida K and Miyazaki K: Gene amplification
CCNE1 is related to poor survival and potential therapeutic target
in ovarian cancer. Cancer. 116:2621–2634. 2010.PubMed/NCBI
|
|
31
|
Pelengaris S and Khan M: The many faces of
c-MYC. Arch Biochem Biophys. 416:129–136. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kubben FJ, Sier CF, van Duijn W, Griffioen
G, Hanemaaijer R, van de Velde CJ, van Krieken JH, Lamers CB and
Verspaget HW: Matrix metalloproteinase-2 is a consistent prognostic
factor in gastric cancer. Br J Cancer. 94:1035–1040. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Calcagno DQ, Freitas VM, Leal MF, de Souza
CR, Demachki S, Montenegro R, Assumpção PP, Khayat AS, Smith MA,
dos Santos AK, et al: MYC, FBXW7 and TP53 copy number variation and
expression in gastric cancer. BMC Gastroenterol. 13:1412013.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Mao JH, Kim IJ, Wu D, Climent J, Kang HC,
DelRosario R and Balmain A: FBXW7 targets mTOR for degradation and
cooperates with PTEN in tumor suppression. Science. 321:1499–1502.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Tu K, Zheng X, Zan X, Han S, Yao Y and Liu
Q: Evaluation of Fbxw7 expression and its correlation with the
expression of c-Myc, cyclin E and p53 in human hepatocellular
carcinoma. Hepatol Res. 42:904–910. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Ibusuki M, Yamamoto Y, Shinriki S, Ando Y
and Iwase H: Reduced expression of ubiquitin ligase FBXW7 mRNA is
associated with poor prognosis in breast cancer patients. Cancer
Sci. 102:439–445. 2011. View Article : Google Scholar
|
|
37
|
Mitsuhashi Y, Horiuchi A, Miyamoto T,
Kashima H, Suzuki A and Shiozawa T: Prognostic significance of
Notch signalling molecules and their involvement in the
invasiveness of endometrial carcinoma cells. Histopathology.
60:826–837. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Peiró G, Peiró FM, Ortiz-Martínez F,
Planelles M, Sánchez-Tejada L, Alenda C, Ceballos S, Sánchez-Payá J
and Laforga JB: Association of mammalian target of rapamycin with
aggressive type II endometrial carcinomas and poor outcome: A
potential target treatment. Hum Pathol. 44:218–225. 2013.
View Article : Google Scholar
|
|
39
|
Okuda T, Sekizawa A, Purwosunu Y,
Nagatsuka M, Morioka M, Hayashi M and Okai T: Genetics of
endometrial cancers. Obstet Gynecol Int. 2010:9840132010.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Llobet D, Pallares J, Yeramian A,
Santacana M, Eritja N, Velasco A, Dolcet X and Matias-Guiu X:
Molecular pathology of endometrial carcinoma: Practical aspects
from the diagnostic and therapeutic viewpoints. J Clin Pathol.
62:777–785. 2009. View Article : Google Scholar
|
|
41
|
Ioffe OB, Papadimitriou JC and Drachenberg
CB: Correlation of proliferation indices, apoptosis, and related
oncogene expression (bcl-2 and c-erbB-2) and p53 in proliferative,
hyperplastic, and malignant endometrium. Hum Pathol. 29:1150–1159.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Sherman ME, Bur ME and Kurman RJ: p53 in
endometrial cancer and its putative precursors: Evidence for
diverse pathways of tumorigenesis. Hum Pathol. 26:1268–1274. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Weinstein IB, Joe A and Felsher D:
Oncogene addiction. Cancer Res. 68:3077–3080; discussion 3080.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Smyth JF, Aamdal S, Awada A, Dittrich C,
Caponigro F, Schöffski P, Gore M, Lesimple T, Djurasinovic N, Baron
B, et al: Phase II study of E7070 in patients with metastatic
melanoma. Ann Oncol. 16:158–161. 2005. View Article : Google Scholar
|
|
45
|
Talbot DC, von Pawel J, Cattell E, Yule
SM, Johnston C, Zandvliet AS, Huitema AD, Norbury CJ, Ellis P,
Bosquee L, et al: A randomized phase II pharmacokinetic and
pharmacodynamic study of indisulam as second-line therapy in
patients with advanced non-small cell lung cancer. Clin Cancer Res.
13:1816–1822. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Nakayama K, Nakayama N, Ishikawa M and
Miyazaki K: Endometrial serous carcinoma: Its molecular
characteristics and histology-specific treatment strategies.
Cancers (Basel). 4:799–807. 2012. View Article : Google Scholar
|
|
47
|
Birkeland E, Wik E, Mjøs S, Hoivik EA,
Trovik J, Werner HM, Kusonmano K, Petersen K, Raeder MB, Holst F,
et al: KRAS gene amplification and overexpression but not mutation
associates with aggressive and metastatic endometrial cancer. Br J
Cancer. 107:1997–2004. 2012. View Article : Google Scholar : PubMed/NCBI
|