Introduction
Exosomes are small, lipid bilayer membrane vesicles
of endocytic origin (30–100 nm in diameter). In recent years, there
has been increasing interest in the relevance between biological
and pathophysiological processes and these extracellular vesicles
(EVs). Increasing evidence suggests that interactions between stem
cells and human tumor cells involve the exchange of biological
information via EVs, including exosomes (1). Stem cells secrete a large number of
exosomes, which act as communicators in the tumor microenvironment
and which play diverse roles in tumorigenesis, tumor angiogenesis
and tumor metastases. However, the role of stem cell-derived
exosomes in the pathophysiological processes of tumors has not been
clarified until now. In this review, the recent findings with
regard to the role of stem cell derived-exosomes in cancer are
briefly summarized.
Biogenesis, contents and secretion of
exosomes
Exosomes, which were first described in 1981, are
derived from the internal vesicles of multivesicular bodies (MVBs)
and consist of a lipid bilayer membrane surrounding a small amount
of cytosol (2). Exosomes are secreted
by all types of cells in culture and are observed in abundance in
body fluids, including saliva, urine, blood and breast milk
(3). In the present study, the
current knowledge with regard to the biogenesis, contents and
secretion of exosomes is summarized.
The formation of MVBs during exosome biogenesis is
similar to the formation of MVBs during lysosome formation. First,
the cell membrane is internalized to produce an endosome.
Subsequently, the endosome forms inside a large number of small
vesicles via invagination of portions of the endosome membrane.
Such endosomes are called MVBs. Finally, exosomes are produced and
packed with cytoplasmic contents, and the membrane of MVBs bulges
inward and pinches off to create small membranous vesicles within
the MVBs. The molecular mechanisms of exosome formation have been
studied extensively; however, the exact mechanism of exosome
packaging has not been fully clarified. Endosomal sorting complex
required for transport (ESCRT)-dependent and ESCRT-independent
signals have been suggested to be associated with the sorting of
exosomes (4). ESCRT consists of four
complexes and their associated proteins: ESCRT-0 identifies
ubiquitinated proteins in the endosomal membrane, ESCRT-I and
ESCRT-II direct endosomal membrane budding, and ESCRT-III
facilitates separation from the endosomal membrane (5,6). Numerous
studies have revealed that the ESCRT-0 protein hepatocyte growth
factor-regulated tyrosine kinase substrate (HRS) is necessary for
exosome formation (7). ESCRT-I
members, including HeLa-CIITA, MCF-7 and tumor susceptibility gene
101 (TSG101), are also associated with another ESCRT-independent
mechanism of exosome biogenesis. Research has demonstrated that
ALG-2-interacting protein X (Alix), an ESCRT-III-associated
protein, promotes exosome biogenesis and, thus, intraluminal
budding of vesicles in endosomes. Two proteins that are degradation
products of the ESCRT-III complex, namely vacuolar protein
sorting-associated protein 4 (VPS4) and charged multivesicular body
protein 4 (CHMP4), are also involved in exosome biogenesis
(6). However, certain studies have
demonstrated that ESCRT-independent signals were also involved in
exosome biogenesis. These pathways may involve lipids including
sphingosine-1-phosphate and four spin-enriched microdomains or heat
shock proteins (6).
After exosomes are formed, they contain >14,000
biomolecules, including proteins, RNAs and DNA (8). In the late 1990s, the first ‘proteomic’
analyses of the protein composition of dendritic cell-derived
exosomes were performed (9).
Biochemically, exosomes contain common marker proteins [e.g.,
tetraspanins, including cluster of differentiation (CD) 9, CD10,
CD26, CD53, CD63, CD81 and CD82], which are present in the exosomal
membrane. Exosomes also contain Alix and TSG101, which are involved
in the formation of MVBs. Two cytoplasmic heat shock proteins,
Hsc70 and Hsp90, have also been observed in exosomes (3,10). mRNAs
and microRNAs (miRNAs/miRs) were definitively identified in
exosomes for the first time by Valadi et al (11). Thakur et al observed that
exosomes from cancer cells contained double-stranded DNA that could
reflect the mutational status of the originated cells (12). It has also been demonstrated that RNA
carried in exosomes may be delivered to target cells and that the
expression of genes in target cells is influenced by miRNAs
contained in exosomes (13). In
addition, mRNAs and miRNAs from different cells may be cell-type
specific.
Exosomes may be secreted via the fusion of MVBs and
the cell membrane, followed by the release of the contents of the
MVBs (exosomes) into the extracellular environment. Alternatively,
the contents of MVBs are degraded through lysosomes. There are
numerous studies on the secretion of exosomes, and various proteins
associated with this process. Rab2b, Rab5a, Rab7, Rab9a, Rab11,
Rab27a, Rab27b and Rab35, members of the Rab family of small
guanosine triphosphatase (GTPase) proteins, have been demonstrated
to accurately regulate the secretion of exosomes (14). Soluble NSF-attachment protein receptor
complexes are associated with the fusion of exosomes and the lipid
bilayers (15). The accumulation of
intracellular Ca2+ and intercellular pH has been
observed to regulate the secretion of exosomes (16). In addition, heparanase overexpression
promotes the secretion of exosomes (17). When exosomes are secreted, some of
them are taken up by target cells localized near the cell of
origin, while other exosomes are delivered to more distant sites
through the blood or other biological fluids.
Uptake and functions of exosomes
In recent years, there has been increasing interest
in intercellular communication via exosomes. A number of studies
have attempted to determine the mechanism by which the cargo in
exosomes is exchanged between exosomes and target cells. After
exosomes are secreted, they may be taken up by the target cell via
direct fusion with the plasma membrane, a receptor-ligand
interaction, or endocytosis by phagocytosis (Fig. 1) (18,19). A
number of biological molecules have significant roles in this
process. Heat shock protein (HSP) 70, which is contained in
exosomes, mediates the communication of cardioprotective signals to
the heart and then activates a pathway downstream of toll-like
receptor 4 (20). T-cell
immunoglobulin- and mucin-domain-containing molecule, intercellular
adhesion molecule 1 and heparan sulfate proteoglycans also
influence the uptake of exosomes (16).
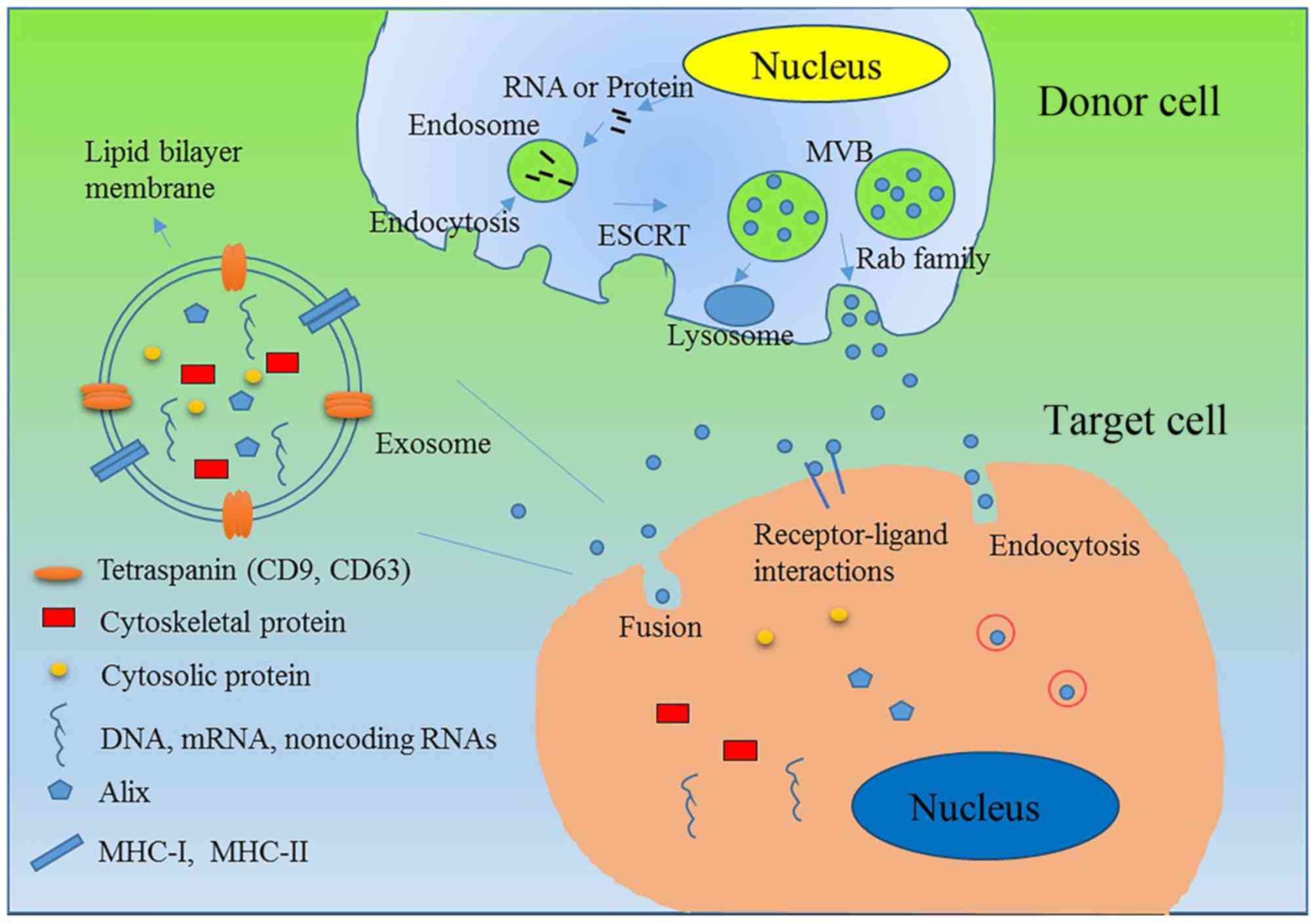 | Figure 1.Biogenesis, contents, secretion and
uptake of exosomes. Exosomes are small extracellular
membrane-enclosed vesicles that contain a variety of molecules,
including proteins, DNA, mRNA and non-coding RNA. Exosomes are
initially formed by endocytosis to produce MVBs, and small
membranous vesicles within the MVBs are created to produce
exosomes. Exosomes contain RNAs/proteins of interest, including
tetraspanins CD9 and CD63, cytosolic protein Rab family protein
transmembrane molecules MHC I and MHC II, RNA and microRNAs.
ESCRT-dependent and ESCRT-independent signals have been
demonstrated to regulate the sorting of exosomes. When MVBs are
produced, some of them fuse with the cell membrane and release
their vesicles into the extracellular space to produce exosomes.
Rab family members and soluble NSF-attachment protein receptor
complexes play a key role in the secretion of exosomes. After
exosomes are secreted, they may be taken up by target cells via
direct fusion with the plasma membrane, a receptor-ligand
interaction, or endocytosis by phagocytosis. MVB, multivesicular
bodies; CD, cluster of differentiation; MHC, major
histocompatibility complex; ESCRT, endosomal sorting complex
required for transport; Alix, ALG-2-interacting protein X. |
After exosomes are taken up by target cells, they
play a vital role in cells. The primary function of exosomes in
intercellular communication is the transfer of biologically active
proteins, lipids and RNAs (21).
Several studies have demonstrated that exosomes play crucial roles
under normal and pathophysiological conditions, including
lactation, the immune response, neuronal function and infectious
diseases, as well as in the development and progression of liver
disease, neurodegenerative diseases and cancer (22). Immune stimulation and tolerization are
noted to be associated with exosomes; a previous study has
suggested the potential use of exosomes in immunotherapy (23). Placental exosomes are involved in
suppressive immunity during normal pregnancy (24). In addition, exosomes from human breast
milk may contribute to the development of the infant immune system
(23). The generation and progression
of neurodegenerative diseases are also associated with exosomes.
Exosomes transport proteins; thus, they may serve as a novel
treatment approach or as new biomarkers in neurodegenerative
diseases (24). In addition, exosomes
have been promoted as specific therapeutic transporters for
cardiovascular diseases (24), and
are involved in the processes of infection biology, modulating the
immune response and functioning as new acellular vaccines or
infection biomarkers for infectious diseases (24). Additionally, exosomes are essential in
the pathogenesis, diagnostics and therapeutics of liver diseases
(24,25).
Increasing evidence has suggested that exosomes have
significant roles in tumor growth, progression, metastasis and drug
resistance (16). Tumor-derived
exosomes regulate the formation of new blood vessels, which support
tumor angiogenesis, and exosomes have crucial roles in tumor cell
proliferation (16). In addition,
exosomes induce the formation of the pre-metastatic niche, which
regulates tumor metastasis (26).
Exosome-mediated mechanisms also clearly contribute to tumor cell
drug resistance (26). Certain
exosomes have a significant influence on the ability of tumors to
evade immune surveillance; however, exosomes from different sources
may enhance the immune response (26). Except for the roles of exosomes in the
pathogenesis of cancer, there are numerous studies on the use of
exosomes as a new tool for cancer diagnosis and therapeutics
(Table I).
 | Table I.Functions of exosomes from different
sources. |
Table I.
Functions of exosomes from different
sources.
| Conditions | Outcome | Reference |
|---|
| Normal
conditions | Cell-cell
communication | 21 |
|
| Removal of
unnecessary protein from cell | 22 |
|
| Immune
function | 23,24,26 |
| Liver diseases | Pathogenesis,
diagnostics and therapeutics | 23,25 |
| Neurodegenerative
diseases | Transporting
proteins, a novel treatment approach or new biomarkers | 24 |
| Neurodegenerative
diseases | New acellular
vaccines or infection biomarkers for infectious diseases | 24 |
| Infectious
diseases | New acellular
vaccines or infection biomarkers for infectious diseases | 24 |
| Cancer | Growth | 16 |
|
| Angiogenesis | 16 |
|
| Progression | 26 |
|
| Metastasis | 26 |
|
| Drug
resistance | 26 |
|
| Manipulation of
their microenvironment | 26 |
|
| Immune
response | 26 |
|
| Diagnosis and
therapeutics biomarker | 16 |
In addition to the abovementioned studies, a number
studies have examined the functions of exosomes (16,23,24,26);
however, which specific class of molecules contained in exosomes
influences the target cells remains unclear.
Stem cells
Stem cells are a class of pluripotent cells that
self-renew and differentiate into a variety of cell types. In this
review, we focus on mesenchymal stem cells (MSCs) and cancer stem
cells (CSCs).
MSCs, which originate from almost all vascularized
organs and tissues, exhibit migratory capabilities and regenerative
potential (27). MSCs may be isolated
from various sources, including umbilical cord (UC), bone marrow
(BM), liver, adipose tissue, multiple dental tissues and induced
pluripotent stem cells (28,29). MSCs express CD44, CD73, CD90 and
CD105, but not CD45, CD34 and CD14; these cells are characterized
by their ability to adhere to plastics under standard cell culture
conditions (30). MSCs affect the
surrounding microenvironment by secreting factors, including growth
factors, in an autocrine and paracrine manner. These cells decrease
inflammation, promote angiogenesis, support tissue repair and
suppress immunity (31). Since MSCs
have the unique ability to home to damaged and cancerous tissue,
they are of great interest in regenerative medicine and cancer
therapy (32). MSCs also have a
significant role in tumors. Previous studies have demonstrated that
human MSCs (hMSCs) promote tumor growth and angiogenesis, in
addition to providing stromal support (33,34),
through autocrine and paracrine signaling (35,36).
Although there are numerous studies of MSCs, the underlying
mechanism of the correlation between MSCs and tumors remains
largely unexplored. In recent years, a number of studies have noted
that the interaction between MSCs and human tumor cells is
associated with the exchange of biological material via EVs,
including exosomes from MSCs (37–39). In
the present review, MSC-derived exosomes are highlighted, which
have a significant role in tumors, in order to realize the
mechanism of this interaction.
During the past decade, CSCs have been increasingly
identified in a number of malignancies. CSCs have stem-like
characteristics and exhibit numerous features of embryonic or
tissue stem cells (40). CSCs are
associated with tumor initiation, metastasis, progression,
invasion, recurrence and resistance to therapies. It is becoming
widely accepted that CSCs play a central role in cancer cell
biology; CSCs are essential for cancer initiation, formation and
relapse. Previous studies have suggested that CSC-derived exosomes
act as a vehicle to deliver genetic information and produce a
favorable microenvironment for cancer development (41–43). In
the present review, the role of CSC-derived exosomes in cancer is
summarized.
Roles of stem cell-derived exosomes in
cancer
Previous studies have demonstrated that stem cells
generate a number of exosomes that may act as paracrine mediators
by exchanging genetic information (38,39). There
are certain differences between stem cell (MSC and CSC)-derived
exosomes and other sources of exosomes. CSC-derived exosomes
contain several proteins, including TSG101, Rab GTPases, annexins
and signal transduction molecules (e.g., 14-3-3, a heterotrimeric G
protein, and Alix), that are potentially associated with their
biogenesis, targeting and putative immunological function (44). In addition, MSC-derived exosomes are
amenable to immortalization without compromising exosome
production, and are not immunogenic. MSC-derived exosomes have
intrinsic therapeutic properties that reduce tissue injury.
Tumor growth
Stem cell-derived exosomes have been noted to
deliver gene regulatory information to target cells; this
information regulates cell growth and angiogenesis by modulating a
variety of cellular pathways. There have been a number studies into
the use of stem cell-derived exosomes to promote tumor cell
proliferation in order to analyze the effects of cellular
interactions between stem cells and various cancer cells. In recent
years, studies have demonstrated that MSCs have a significant role
in regulating tumor growth and metastasis (40,45). EVs,
including exosomes isolated from hMSCs, were first completely
biochemically and molecularly analyzed by Vallabhaneni et al
(46). These authors used
co-injection xenograft assays to demonstrate that the exosomes
secreted from hMSCs support breast cancer cell proliferation and
metastasis. They also observed that hMSC-derived EVs contain a
large amount of miR-21 and 34a, which are tumor-supportive miRNAs,
and ~150 different proteins, most of which are known tumor
supportive factors, including platelet-derived growth factor
receptor-β, tissue inhibitor of metalloproteinase (TIMP)-1 and
TIMP-2. The presence of bioactive lipids, including sphingomyelin,
was verified in EVs, including exosomes, through lipidomic assays.
Furthermore, metabolite assays identified the presence of lactic
acid and glutamic acid in the EVs. In addition, Zhu et al
revealed that MSC-derived exosomes enhanced vascular endothelial
growth factor (VEGF) expression in tumor cells by activating the
extracellular signal-regulated kinase 1/2 (ERK1/2) pathway, which
promotes tumor growth (47). These
authors were the first to demonstrate that exosomes from MSCs have
a role in promoting tumor growth that is similar to that of the
MSCs themselves, thus providing new insight into the actions of
MSCs in tumor development and progression in vivo. Hernanda
et al observed that exosomes (or microvesicles) secreted by
MSCs may also be associated with tumor promotion (48). Subsequently, Yang et al
demonstrated that the internalization of MSC-derived exosomes was
involved in the acquisition of new tumor cell properties by
altering cellular functionalities and providing the capability to
re-organize the tumor microenvironment, which improves tumor growth
(49). However, the effects of
exosomes from different types of stem cells on cell proliferation
may be completely different. Del Fattore et al demonstrated
that BM- and UC-MSC-EVs (including exosomes) suppressed cell
proliferation, while the opposite effect was observed with adipose
tissue MSC-EVs (including exosomes) (50). In addition, microvesicles from human
BM-derived MSCs inhibited tumor growth (51). Another experiment indicated that
intra-tumoral injection of exosomes derived from miR-146-expressing
MSCs significantly reduced glioma xenograft growth in a rat primary
brain tumor model (52). miR-146
suppressed epidermal growth factor receptor (EGFR) expression
through binding-targeting EGFR mRNA, and reduced in vitro
growth, migration and invasion of cancer (53). These studies revealed that miRNAs may
be packaged into MSC exosomes, delivered to target tumor cells in
culture, and reduce glioma cells, which suggested that the export
of specific therapeutic miRNAs into MSC exosomes represented a new
treatment strategy for malignant glioma. Additionally, although
exosomes derived from multiple myeloma BM-MSCs have been noted to
improve multiple myeloma progression, normal BM MSC-derived
exosomes reduce tumor promotion (54). Exosomes from human umbilical cord
Wharton's jelly MSCs reduced bladder tumor cell growth in
vitro and in vivo (55).
Thus, the effects of different sources of MSC-derived exosomes are
uncertain.
Based on the aforementioned studies, it is known
that exosomes secreted from MSCs may result in cell-to-cell
transfer of mRNA, miRNA and proteins (56). However, the exact roles and mechanisms
of MSC-produced exosomes in tumor biology remain largely
elusive.
Cancer stem cell-derived exosomes also have a
notable influence on tumor growth. It is known that human gliomas
have a population of stem cells with a tumor-supporting ability;
these cells are called glioma-associated stem cells (GASCs).
Bourkoula et al demonstrated that exosomes derived from
GASCs support tumor growth and have a tumor-supporting phenotype
(42). In addition, EVs, including
exosomes from glioblastoma-derived CSCs, regulate tumor growth
through chloride intracellular channel-1 (CLIC1) (43).
Thus, exosomes released from stem cells affect tumor
growth (Table II); however, further
studies of stem cell-derived exosomes are required.
 | Table II.Overview of functions of stem
cell-derived exosomes in cancer. |
Table II.
Overview of functions of stem
cell-derived exosomes in cancer.
| Cancer type | Donor cells | Exosomal cargo | Target cells | Pathway
involved | Application or
outcome | Reference |
|---|
| Breast cancer | hMSCs | miR-21, miR34a,
~150 different proteins, sphingomyelin | MCF-7 breast cancer
cells | Tumors co-injected
with EVs exhibited higher angiogenesis | Supported breast
cancer cell proliferation | 46 |
| Human gastric
carcinoma | MSC | Unclear | SGC-7901 cells | VEGF ERK1/2
pathway | Promoted tumor
growth | 47 |
| Breast cancer and
carcinoma of the ovary | MSC | MMP-2 protein | SCCOHT-1 cells,
MCF-7 breast cancer cells | Altered cellular
functionalities | Improved tumor
growth and provided capability to re-organize tumor
microenvironment | 49 |
| Malignant
glialtumors | Umbilical cord
MSC | Unclear | U87MG glioblastoma
cells | Cell-cell
communication | Decreased cell
proliferation | 50 |
| Malignant
glialtumors | Adipose tissue
MSC | Unclear | U87MG glioblastoma
cells | Cell
communication | Improved tumor
growth | 50 |
| Glioma | miR-146-expressing
MSCs | miR-146 | Glioma cells | miR-146 bound EGFR
mRNA and suppressed EGFR expression | Reduced glioma
xenograft growth | 52 |
| Multiple
myeloma | Multiple myeloma
BM-MSC | Lower miR-15a
levels and higher content levels of CCL2 | Multiple myeloma
cells | Lower miR-15a
expression in MM vs. normal BM-MSC-derived exosomes | MM BM-MSC-derived
exosomes promoted MM tumor growth | 54 |
| Multiple
myeloma | Normal BM-MSC | Suppressor miRNAs
and normal level of CCL2 | Multiple myeloma
cells | Transfer of tumor
suppressor miRNAs (including miR-15a) | Normal
BM-MSC-derived exosomes inhibited growth of MM cells | 54 |
| Bladder tumor | hWJMSCs | Unclear | T24 cells | Downregulated
phosphorylation of Akt protein kinase and upregulated cleaved
caspase 3 | Anti-proliferation
and pro-apoptosis | 55 |
| Glioma | GASC | Unclear | Glioblastoma cell
lines | Unclear | Supported tumor
growth | 42 |
| Glioblastoma | GBM-derived
CSCs | CLIC1 | GBM cell lines | Stimulated cell
growth through CLIC1 | Novel regulator of
GBM growth | 43 |
| Renal cancer | CD105-positive
cancer stem cells | Proangiogenic mRNAs
and miRNAs | HUVECs | mRNAs, including
VEGF, FGF, angiopoietin1, ephrin A3, MMP-2 and MMP-9, and
functional miRNAs, including miR-200c, miR-92 and miR-141,
contribute to tumor angiogenesis | Contributed to
triggering angiogenic switch and coordinating metastatic
diffusion | 39 |
| Breast cancer
cells | MSCs | miR-16 | 4T1 cells | Downregulated VEGF
expression | Suppressed
angiogenesis | 59 |
| Gastric cancer | MSC | miR-221 | HGC-27 | Regulated miR-221
expression | Promoted
proliferation and migration | 65 |
| Breast cancer | Adipose MSC | Genes associated
with cell migration upregulated | Breast cancer cell
line MCF-7 | Wnt signaling
pathway | Promoted migration
and proliferation of breast cancer cell line MCF-7 | 66 |
| Breast cancer | DCIS stem-like
cells | miR-140 is
downregulated in exosomes | Breast cancer
cells | Downregulation of
miR-140 removed inhibition of Wnt, Sox2 and Sox9 stem cell
regulatory pathways | Lead to higher
cancer stem cell populations and breast cancer progression | 67 |
| Breast cancer | DCIS stem-like
cells | miR-21, miR-140 and
miR-29a | MDA-MB-231
cells | miRNAs signaled
through nearby immune cells via interaction with toll-like
receptors to upregulate secretion of TNF and IL-6 secretion | Enhanced migratory
capacity | 68 |
| Glioma | MSCs | miR-124 and
miR-145 | Glioma cells U87
and A172 | miR-124 and miR-145
mimics decreased luciferase activity of respected reporter target
genes, SCP-1 and Sox2 | Decreased migration
of glioma cells and self-renewal of GSCs | 69 |
| Breast cancer | BM-MSCs | miR-23b | BM2 cells | Increased miR-23b
and decreased MARCKS expression | Promoted breast
cancer cell dormancy in a metastatic niche | 70 |
| Osteosarcoma | BM-MSCs | miR-143 | Human osteosarcoma
cell line 143B | Delivered miR-143
to target cells | Reduced migration
of osteosarcoma cells | 71 |
Tumor angiogenesis
The formation of new blood vessels is necessary for
tumor growth and development, and plays a significant role in tumor
progression and metastasis. The process of angiogenesis is
extremely complex; it generally includes vascular endothelial
substrate degradation, vascular endothelial cell migration and
endothelial cell proliferation, as well as the formation of
vascular pipeline branches and a new basement membrane. Exosomes
contain abundant angiogenic factors that regulate tumor
angiogenesis. For example, MSC-derived exosomes promote tumor
angiogenesis by increasing VEGF expression in tumor cells and
activating ERK1/2 and p38 mitogen-activated protein kinase pathways
(47). Other studies have
demonstrated that placental MSC exosomes promote vascular network
formation and improve microvascular endothelial cell migration in a
concentration- and oxygen-dependent manner (57). In addition, EVs released by adipose
mesenchymal stem cells (ASCs) may contribute to ASC-induced
angiogenesis (58). In cancer stem
cells, exosomes derived only from CD105-positive cancer stem cells
conferred an activated angiogenic phenotype to normal human
endothelial cells, stimulating their growth and vessel formation
(39). The results defined a specific
source of cancer stem cell-derived MVs that contribute to
triggering the angiogenic switch and coordinating metastatic
diffusion during tumor progression (39). The effects of exosomes from different
types of stem cells on tumor angiogenesis are similar to the
effects of stem cell-derived exosomes on tumor growth. However,
their effect may be completely different. Lee et al observed
that MSC-derived exosomes suppressed angiogenesis by transferring
anti-angiogenic molecules and serving as a significant mediator of
cell-to-cell communication within the tumor microenvironment
(59). These authors noted that
MSC-derived exosomes inhibited tumor growth and angiogenesis in
breast cancer by downregulating the expression of VEGF, which is a
pro-angiogenic factor that is frequently overexpressed in cancer
(60). miR-16, which is contained in
MSC-derived exosomes, reduces the VEGF expression level in 4T1
cells. It has been demonstrated that miR-16 controls VEGF
expression (61–63). In summary, exosomes are significantly
associated with tumor angiogenesis.
Tumor metastases
Exosomes released from stem cells also contribute to
tumor metastasis. A number of the key steps in tumor invasion and
metastasis are associated with MSCs, including facilitating
epithelial-mesenchymal transition and the induction of stem-like
properties that allow cancer stem cells to increase their
survivability through the circulation (64). A number of studies have examined the
role of CSC- and MSC-derived exosomes in metastasis, tumor
reseeding (self-seeding) and the formation of a pre-metastatic
niche. Gastric cancer (GC) MSC-derived exosomes were observed to
deliver miR-221 to HGC-27 cells, which facilitated the
proliferation and migration of these cells (65). In addition, MSC-derived exosomes
promoted Wnt signaling pathway activation to facilitate the
migration and proliferation of the breast cancer cell line MCF-7
(66). The Wnt signaling pathway is
characterized by the nuclear accumulation of β-catenin, which is
involved in not only embryonic development but also tumor
development (66). As for CSCs,
Wolfson et al noted that exosomes derived from ductal
carcinoma in situ (DCIS) stem-like cells contained lower
levels of miR-140 compared with exosomes derived from a DCIS whole
cell population, which could improve tumor growth and metastases
(67). Dysregulation of miR-140 has
an important role in regulating the transition of DCIS to invasive
ductal carcinoma (IDC) (67). As the
tumor grade increases, miR-140 is progressively downregulated and
plays a significant role in the stem cell regulatory pathways.
Downregulation of miR-140 leads to higher CSC populations and
breast cancer progression by removing tumor suppressive pathways
(67). Other studies have
characterized the exosomal exchange of miRNAs between DCIS
stem-like cells and target cells; exosomes from DCIS stem-like
cells were observed to enhance the migratory capacity via several
miRNAs, including miR-140, miR-29a and miR-21, which are
differentially expressed in the exosomes (68). These findings suggest that CSC-derived
exosomes may improve tumor metastases.
However, there are certain contrary findings. Lee
et al demonstrated that MSC-derived exosomes transferred
specific miRNA mimics in a gap junction-dependent and
contact-independent manner. miR-124 and miR-145 mimics vitally
decrease the luciferase activity of their respective reporter
target genes, including small carboxy-terminal domain phosphatase 1
(SCP-1) and sex-determining region Y-box 2 (Sox2), and decrease the
migration of glioma cells and the self-renewal of glioma stem cells
(69). Additionally, Ono et al
noted that breast cancer BM-MSC-derived exosomes have more varied
miRNAs than adult fibroblast-derived exosomes. BM-MSC-derived
exosomes overexpress miR-23b, which induces a dormant phenotype
through suppressing a target gene, myristoylated alanine-rich
C-kinase substrate, which encodes a protein that promotes cell
cycling and motility (70). These
findings suggest that exosomal transfer of miRNAs from the BM may
promote breast cancer cell dormancy in a metastatic niche. Another
study has also presented the same result; these authors observed
that the delivery of miR-143 via MSC-derived exosomes significantly
reduced the migration of osteosarcoma cells (71).
Taken together, these findings reveal that stem
cell-derived exosome-mediated intercellular communication may be an
essential mechanism for tumor metastasis.
Tumor therapy
Personalized medicine is used to identify patient-
and tumor-specific factors that are useful for the identification
of therapeutic options and the prognostic stratification of
patients in order to maximize effectiveness and minimize
treatment-associated toxicity (72).
To achieve this goal, exosomes from stem cells, possibly MSCs, may
provide a new approach to personalized medicine. Over the past few
years, the potential of using MSCs in regenerative medicine has
received increasing attention, and the use of MSCs in anticancer
therapy has been extensively studied. A previous study suggested
that MCS-derived exosomes may have a key role in not only
regenerative medicine to repair damaged tissue but also tumor
therapy (73). MSCs may communicate
with cancer cells via gap junctional intercellular communication
and via the secretion of exosomes (74,75). Chen
and Lim revealed that intercellular communication between MSCs and
tumor cells could be promoted by secreting microparticles,
including exosomes, through the exosomal transfer of miRNAs
(76). Additionally, in recent years,
miRNAs have emerged as potential anti-cancer agents. Although
miRNAs are an effective therapeutic method for cancer, the
effectiveness of this approach requires targeted delivery. Exosomes
work as delivery vehicles for nucleic acids or drugs; this novel
approach has gained increasing interest due to the effective
biocompatibility and biodistribution of exosomes. In addition, stem
cells have an infinite capacity for reproducible production of
exosomes as drug delivery vehicles, which may be essential for
tumor therapy. Lim et al observed that exosomes transmit
miRNAs from BM stroma to breast cancer cells in tumor cell
quiescence. This study revealed that the transfer of miRNAs from BM
stroma to breast cancer cells through exosomes may have a
significant role in the dormancy of BM metastases through exosomes
(77). Munoz et al revealed
that MSC-derived exosomes could deliver anti-miR-9, which blocks
miR-9, to glioblastoma multiforme (GBM) cells. Anti-miR-9 was
involved in the expression of the drug efflux transporter
P-glycoprotein, reversed the expression of the multidrug
transporter, and sensitized the GBM cells to temozolomide, which
increases cell death and caspase activity (78). These data demonstrated a potential
role for MSCs in the functional delivery of synthetic anti-miR-9 to
reverse the chemoresistance of GBM cells. Boelens et al
revealed that stromal cell-derived exosomes stimulated the pattern
recognition receptor retinoic acid-inducible gene I to activate
signal transducer and activator of transcription 1-dependent
antiviral signaling and activate NOTCH3 in breast cancer cells to
expand breast cancer cell subpopulations that are adept at
resisting therapy and reinitiating tumor growth. These findings
suggest a possible novel use of MSCs in the development of new
‘biotech drugs’ with increased efficacy and homing capacity
(79). In addition, miR-122-modified
adipose tissue-derived MSCs secrete exosomes to increase
chemosensitivity (80). MSC-derived
exosomes effectively silence the polo-like kinase 1 (PLK-1) gene by
transporting PLK-1 small interfering RNA to bladder cancer cells
(81). Pascucci et al were the
first to demonstrate that MSCs act as a factory to develop drugs
with a higher cell-target specificity through packaging and
delivering active drugs, and suggested the possibility of using
MSCs (82). These authors noted that
paclitaxel (PTX) is incorporated by MSCs and released in exosomes.
PTX is delivered to target cells and inhibits tumor growth through
a simple procedure of exposing the cells to an extremely high
concentration of PTX. Fuhrmann et al demonstrated that EVs,
including exosomes, loaded with hydrophilic porphyrins induced a
stronger phototoxic effect than free drugs in a cancer cell model;
this approach may significantly improve cellular uptake and the
therapeutic effect of phototoxic porphyrins in vitro
(83). Significantly, these methods
are simple and directly applicable to other drugs and vesicles,
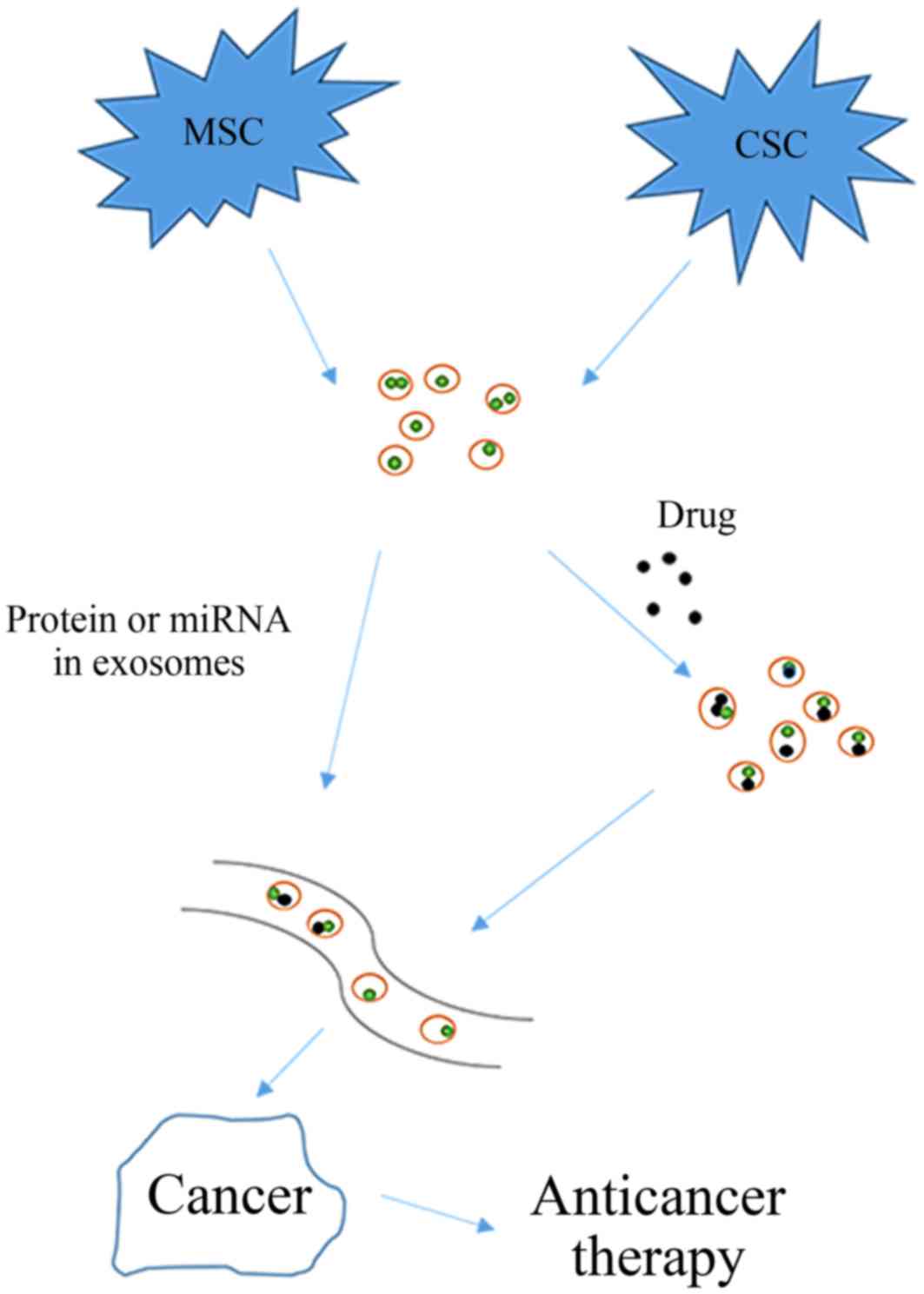
thus providing a new approach to cancer therapy.
EVs from other stem cells also have a potential role
in tumor therapy. Fonsato et al demonstrated that EVs
derived from human adult liver stem cells may inhibit HepG2
hepatoma and primary hepatocellular carcinoma cell growth and
survival (Table III) (84). In summary, stem cell-derived exosomes
have a significant role in anti-cancer treatment (Fig. 2).
 | Table III.Exosomes in tumor therapy. |
Table III.
Exosomes in tumor therapy.
| Cancer type | Donor cells | Exosomal cargo | Target cells | Application or
outcome | Reference |
|---|
| Glioblastoma | MSCs | Anti-miR-9 | Resistant GBM
cells | Increase tumor
death | 78 |
| Breast cancer | Stromal cells | Non-coding RNA and
proteins | Breast cancer cells
(1833) | Biotech drugs
regulate therapy resistance | 79 |
| Hepatocellular
carcinoma | miR-122-modified
adipose tissue-derived MSCs | miR-122 | HepG2 | Increase
chemosensitivity | 80 |
| Bladder cancer | MSCs | PLK-1 siRNA | Bladder cancer cell
lines Pancreatic cell | Selective gene
silencing of PLK-1 | 81 |
| Pancreatic
cancer | MSC with PTX | PTX | line sr4987 | Deliver drugs | 82 |
| Breast cancer | hMSCs | Hydrophilic
porphyrins | MDA-MB231 breast
cancer (MDA) cells | Deliver drugs | 83 |
| Hepatocellular
carcinoma | Liver stem
cells | Antitumor miRNAs,
including miR-451, miR-223 and miR-31 | HepG2 | Inhibit growth and
survival | 84 |
Tumor biomarkers
Stem cell-derived exosomes provide an enriched
source of biomarkers as they contain bioactive molecules that
reflect the pathological state of the origin cells. Recently, CSCs
have been used in cancer diagnostics and treatment. Exosomes
released from rotenone-treated prostate and breast CSCs have
specific biomolecular characteristics, including the expression of
several exosomal markers, including CD9, CD63, CD81, Alix and
TSG101. Thus, the release of exosomal markers may be highly
relevant for biological activity and may be used as potential
targets (44). In addition,
GC-MSC-derived exosomes contain miR-221, which is a potential new
biomarker for tumor diagnosis (65).
Taken together, these findings suggest that the
constituents of exosomes deliver information from cell to cell
(delivering proteins and nucleic acids), and may be used as a
biomarker for targeted cancer therapy. There are a number of
studies on the use of MSCs in regenerative medicine and anti-cancer
treatments (85–87). Over the past few years, these studies
have raised high expectations. However, safety concerns and tight
regulations hamper their practical use in clinical settings. The
use of stem cell-derived exosomes may have numerous advantages
compared with cell-based approaches, and may improve the safety of
MSCs in tumor therapy. However, further experiments are required to
demonstrate the safety and feasibility of stem cell-derived
exosomes.
Conclusion
In conclusion, studies have demonstrated that
exosomes derived from various cell types, including stem cells, may
act as mediators of cell-to-cell communication. The role of stem
cell-derived exosomes in tumor development has been intensively
studied due to the influence of exosomes on tumors and the
significant therapeutic potential of stem cells. MSC- and
CSC-derived exosomes contain different protein and RNA profiles
compared with their donor cells. Exosomes transfer their molecular
contents, including numerous types of special proteins and RNA,
between cells, thus influencing tumor behavior and proliferation.
In addition, exosomes carry a number of the desirable attributes of
a synthetic liposome vehicle, including the capacity to carry
hydrophobic drugs, and are effective and safe drug delivery
vehicles. In the present review, several aspects of stem
cell-derived exosome biology in cancer, which has the ability to
communicate with surrounding and distant cells, are discussed. MSC-
and CSC-derived exosomes have a significant role in synergistically
influencing cancer development, metastasis, progression and drug
resistance. Although a number of studies have examined the role of
stem cell-derived exosomes in cancer (46–49), the
exact mechanisms of the effects of stem cell-derived exosomes on
cancer have been largely unexplored and untested. The utility of
exosomes as a delivery vehicle is also unclear. Additional studies
of stem cell-derived exosomes are required to determine their roles
in the pathogenesis of cancer and to provide a new tool for cancer
diagnosis and therapeutics.
Acknowledgements
This study was supported by the National Basic
Research Program of China (973 Program, 2013CB834100), the National
Natural Science Foundation of China (no. 81572393, 81472820,
81172143, 11334004 and 81421091), the Key Project supported by
Medical Science and Technology Development Foundation, Nanjing
Municipality Health Bureau (no. ZKX15020 and ZKX 12011), the
Natural Science Foundation of Jiangsu Province (no. BK20141324),
the Jiangsu Province Innovation for Ph.D. Candidates (no.
KYLX_0058), the Scientific Research Foundation of the Graduate
School of Nanjing University (no. 2013CL14), and the Jiangsu
Special Program for Clinical Medical Science and Technology
(BL2014054).
Glossary
Abbreviations
Abbreviations:
|
MVBs
|
multivesicular bodies
|
|
MSCs
|
mesenchymal stroma/stem cells
|
|
CSCs
|
cancer stem cells
|
|
ESCRT
|
endosomal sorting complex required for
transport
|
|
GTPase
|
guanosine triphosphatase
|
|
EVs
|
extracellular vesicles
|
|
VEGF
|
vascular endothelial growth factor
|
|
CLIC1
|
chloride intracellular channel-1
|
|
ERK
|
extracellular signal-regulated
kinase
|
References
|
1
|
Lopatina T, Gai C, Deregibus MC, Kholia S
and Camussi G: Cross talk between cancer and mesenchymal stem cells
through extracellular vesicles carrying nucleic acids. Front Oncol.
6:1252016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Trams EG, Lauter CJ, Salem N Jr and Heine
U: Exfoliation of membrane ecto-enzymes in the form of
micro-vesicles. Biochim Biophys Acta. 645:63–70. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Vlassov AV, Magdaleno S, Setterquist R and
Conrad R: Exosomes: Current knowledge of their composition,
biological functions, and diagnostic and therapeutic potentials.
Biochim Biophys Acta. 1820:940–948. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Trajkovic K, Hsu C, Chiantia S, Rajendran
L, Wenzel D, Wieland F, Schwille P, Brügger B and Simons M:
Ceramide triggers budding of exosome vesicles into multivesicular
endosomes. Science. 319:1244–1247. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Brinton LT, Sloane HS, Kester M and Kelly
KA: Formation and role of exosomes in cancer. Cell Mol Life Sci.
72:659–671. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kowal J, Tkach M and Théry C: Biogenesis
and secretion of exosomes. Curr Opin Cell Biol. 29:116–125. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Colombo M, Moita C, van Niel G, Kowal J,
Vigneron J, Benaroch P, Manel N, Moita LF, Théry C and Raposo G:
Analysis of ESCRT functions in exosome biogenesis, composition and
secretion highlights the heterogeneity of extracellular vesicles. J
Cell Sci. 126:5553–5565. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Mathivanan S, Fahner CJ, Reid GE and
Simpson RJ: ExoCarta 2012: Database of exosomal proteins, RNA and
lipids. Nucleic Acids Res. 40:(Database Issue). D1241–D1244. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Théry C, Regnault A, Garin J, Wolfers J,
Zitvogel L, Ricciardi-Castagnoli P, Raposo G and Amigorena S:
Molecular characterization of dendritic cell-derived exosomes.
Selective accumulation of the heat shock protein hsc73. J Cell
Biol. 147:599–610. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Bang C and Thum T: Exosomes: New players
in cell-cell communication. Int J Biochem Cell Biol. 44:2060–2064.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Valadi H, Ekström K, Bossios A, Sjöstrand
M, Lee JJ and Lötvall JO: Exosome-mediated transfer of mRNAs and
microRNAs is a novel mechanism of genetic exchange between cells.
Nat Cell Biol. 9:654–659. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Thakur BK, Zhang H, Becker A, Matei I,
Huang Y, Costa-Silva B, Zheng Y, Hoshino A, Brazier H, Xiang J, et
al: Double-stranded DNA in exosomes: A novel biomarker in cancer
detection. Cell Res. 24:766–769. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ismail N, Wang Y, Dakhlallah D, Moldovan
L, Agarwal K, Batte K, Shah P, Wisler J, Eubank TD, Tridandapani S,
et al: Macrophage microvesicles induce macrophage differentiation
and miR-223 transfer. Blood. 121:984–995. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Tolmachova T, Anders R, Stinchcombe J,
Bossi G, Griffiths GM, Huxley C and Seabra MC: A general role for
Rab27a in secretory cells. Mol Biol Cell. 15:332–344. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zylbersztejn K and Galli T: Vesicular
traffic in cell navigation. FEBS J. 278:4497–4505. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang X, Yuan X, Shi H, Wu L, Qian H and
Xu W: Exosomes in cancer: Small particle, big player. J Hematol
Oncol. 8:832015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Thompson CA, Purushothaman A, Ramani VC,
Vlodavsky I and Sanderson RD: Heparanase regulates secretion,
composition, and function of tumor cell-derived exosomes. J Biol
Chem. 288:10093–10099. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Gruenberg J and van der Goot FG:
Mechanisms of pathogen entry through the endosomal compartments.
Nat Rev Mol Cell Biol. 7:495–504. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yu X, Harris SL and Levine AJ: The
regulation of exosome secretion: A novel function of the p53
protein. Cancer Res. 66:4795–4801. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Vicencio JM, Yellon DM, Sivaraman V, Das
D, Boi-Doku C, Arjun S, Zheng Y, Riquelme JA, Kearney J, Sharma V,
et al: Plasma exosomes protect the myocardium from
ischemia-reperfusion injury. J Am Coll Cardiol. 65:1525–1536. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Meckes DG Jr and Raab-Traub N:
Microvesicles and viral infection. J Virol. 85:12844–12854. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Admyre C, Johansson SM, Qazi KR, Filén JJ,
Lahesmaa R, Norman M, Neve EP, Scheynius A and Gabrielsson S:
Exosomes with immune modulatory features are present in human
breast milk. J Immunol. 179:1969–1978. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Robbins PD and Morelli AE: Regulation of
immune responses by extracellular vesicles. Nat Rev Immunol.
14:195–208. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
De Toro J, Herschlik L, Waldner C and
Mongini C: Emerging roles of exosomes in normal and pathological
conditions: New insights for diagnosis and therapeutic
applications. Front Immunol. 6:2032015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Masyuk AI, Masyuk TV and Larusso NF:
Exosomes in the pathogenesis, diagnostics and therapeutics of liver
diseases. J Hepatol. 59:621–625. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Miller IV and Grunewald TG: Tumour-derived
exosomes: Tiny envelopes for big stories. Biol Cell. 107:287–305.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Caplan AI: Mesenchymal stem cells. J
Orthop Res. 9:641–650. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Bieback K and Klüter H: Mesenchymal
stromal cells from umbilical cord blood. Curr Stem Cell Res Ther.
2:310–323. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Lu T, Hu P, Su X, Li C, Ma Y and Guan W:
Isolation and characterization of mesenchymal stem cells derived
from fetal bovine liver. Cell Tissue Bank. 15:439–450. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Natunen S, Lampinen M, Suila H, Ritamo I,
Pitkänen V, Nairn AV, Räbinä J, Laitinen S, Moremen KW, Reutter W
and Valmu L: Metabolic glycoengineering of mesenchymal stromal
cells with N-propanoylmannosamine. Glycobiology. 23:1004–1012.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Park JS, Suryaprakash S, Lao YH and Leong
KW: Engineering mesenchymal stem cells for regenerative medicine
and drug delivery. Methods. 84:3–16. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Baglio SR, Pegtel DM and Baldini N:
Mesenchymal stem cell secreted vesicles provide novel opportunities
in (stem) cell-free therapy. Front Physiol. 3:3592012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Orimo A, Gupta PB, Sgroi DC,
Arenzana-Seisdedos F, Delaunay T, Naeem R, Carey VJ, Richardson AL
and Weinberg RA: Stromal fibroblasts present in invasive human
breast carcinomas promote tumor growth and angiogenesis through
elevated SDF-1/CXCL12 secretion. Cell. 121:335–348. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Huang WH, Chang MC, Tsai KS, Hung MC, Chen
HL and Hung SC: Mesenchymal stem cells promote growth and
angiogenesis of tumors in mice. Oncogene. 32:4343–4354. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Karnoub AE, Dash AB, Vo AP, Sullivan A,
Brooks MW, Bell GW, Richardson AL, Polyak K, Tubo R and Weinberg
RA: Mesenchymal stem cells within tumour stroma promote breast
cancer metastasis. Nature. 449:557–563. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Ljujic B, Milovanovic M, Volarevic V,
Murray B, Bugarski D, Przyborski S, Arsenijevic N, Lukic ML and
Stojkovic M: Human mesenchymal stem cells creating an
immunosuppressive environment and promote breast cancer in mice.
Sci Rep. 3:22982013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Akyurekli C, Le Y, Richardson RB,
Fergusson D, Tay J and Allan DS: A systematic review of preclinical
studies on the therapeutic potential of mesenchymal stromal
cell-derived microvesicles. Stem Cell Rev. 11:150–160. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Ratajczak J, Miekus K, Kucia M, Zhang J,
Reca R, Dvorak P and Ratajczak MZ: Embryonic stem cell-derived
microvesicles reprogram hematopoietic progenitors: Evidence for
horizontal transfer of mRNA and protein delivery. Leukemia.
20:847–856. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Grange C, Tapparo M, Collino F, Vitillo L,
Damasco C, Deregibus MC, Tetta C, Bussolati B and Camussi G:
Microvesicles released from human renal cancer stem cells stimulate
angiogenesis and formation of lung premetastatic niche. Cancer Res.
71:5346–5356. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Takebe N, Miele L, Harris PJ, Jeong W,
Bando H, Kahn M, Yang SX and Ivy SP: Targeting notch, hedgehog, and
Wnt pathways in cancer stem cells: Clinical update. Nat Rev Clin
Oncol. 12:445–464. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Hannafon BN and Ding WQ: Cancer stem cells
and exosome signaling. Stem Cell Investig. 2:112015.PubMed/NCBI
|
|
42
|
Bourkoula E, Mangoni D, Ius T, Pucer A,
Isola M, Musiello D, Marzinotto S, Toffoletto B, Sorrentino M,
Palma A, et al: Glioma-associated stem cells: A novel class of
tumor-supporting cells able to predict prognosis of human low-grade
gliomas. Stem Cells. 32:1239–1253. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Setti M, Osti D, Richichi C, Ortensi B,
Del Bene M, Fornasari L, Beznoussenko G, Mironov A, Rappa G, Cuomo
A, et al: Extracellular vesicle-mediated transfer of CLIC1 protein
is a novel mechanism for the regulation of glioblastoma growth.
Oncotarget. 6:31413–31427. 2015.PubMed/NCBI
|
|
44
|
Kumar D, Gupta D, Shankar S and Srivastava
RK: Biomolecular characterization of exosomes released from cancer
stem cells: Possible implications for biomarker and treatment of
cancer. Oncotarget. 6:3280–3291. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Kahlert C and Kalluri R: Exosomes in tumor
microenvironment influence cancer progression and metastasis. J Mol
Med (Berl). 91:431–437. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Vallabhaneni KC, Penfornis P, Dhule S,
Guillonneau F, Adams KV, Mo YY, Xu R, Liu Y, Watabe K, Vemuri MC
and Pochampally R: Extracellular vesicles from bone marrow
mesenchymal stem/stromal cells transport tumor regulatory microRNA,
proteins, and metabolites. Oncotarget. 6:4953–4967. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Zhu W, Huang L, Li Y, Zhang X, Gu J, Yan
Y, Xu X, Wang M, Qian H and Xu W: Exosomes derived from human bone
marrow mesenchymal stem cells promote tumor growth in vivo. Cancer
Lett. 315:28–37. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Hernanda PY, Pedroza-Gonzalez A, van der
Laan LJ, Bröker ME, Hoogduijn MJ, Ijzermans JN, Bruno MJ, Janssen
HL, Peppelenbosch MP and Pan Q: Tumor promotion through the
mesenchymal stem cell compartment in human hepatocellular
carcinoma. Carcinogenesis. 34:2330–2340. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Yang Y, Bucan V, Baehre H, von der Ohe J,
Otte A and Hass R: Acquisition of new tumor cell properties by
MSC-derived exosomes. Int J Oncol. 47:244–252. 2015.PubMed/NCBI
|
|
50
|
Del Fattore A, Luciano R, Saracino R,
Battafarano G, Rizzo C, Pascucci L, Alessandri G, Pessina A,
Perrotta A, Fierabracci A and Muraca M: Differential effects of
extracellular vesicles secreted by mesenchymal stem cells from
different sources on glioblastoma cells. Expert Opin Biol Ther.
15:495–504. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Bruno S, Collino F, Deregibus MC, Grange
C, Tetta C and Camussi G: Microvesicles derived from human bone
marrow mesenchymal stem cells inhibit tumor growth. Stem Cells Dev.
22:758–771. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Katakowski M, Buller B, Zheng X, Lu Y,
Rogers T, Osobamiro O, Shu W, Jiang F and Chopp M: Exosomes from
marrow stromal cells expressing miR-146b inhibit glioma growth.
Cancer Lett. 335:201–204. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Katakowski M, Zheng X, Jiang F, Rogers T,
Szalad A and Chopp M: MiR-146b-5p suppresses EGFR expression and
reduces in vitro migration and invasion of glioma. Cancer Invest.
28:1024–1030. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Roccaro AM, Sacco A, Maiso P, Azab AK, Tai
YT, Reagan M, Azab F, Flores LM, Campigotto F, Weller E, et al: BM
mesenchymal stromal cell-derived exosomes facilitate multiple
myeloma progression. J Clin Invest. 123:1542–1555. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Wu S, Ju GQ, Du T, Zhu YJ and Liu GH:
Microvesicles derived from human umbilical cord Wharton's jelly
mesenchymal stem cells attenuate bladder tumor cell growth in vitro
and in vivo. PLoS One. 8:e613662013. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Biancone L, Bruno S, Deregibus MC, Tetta C
and Camussi G: Therapeutic potential of mesenchymal stem
cell-derived microvesicles. Nephrol Dial Transplant. 27:3037–3042.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Salomon C, Ryan J, Sobrevia L, Kobayashi
M, Ashman K, Mitchell M and Rice GE: Exosomal signaling during
hypoxia mediates microvascular endothelial cell migration and
vasculogenesis. PLoS One. 8:e684512013. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Lopatina T, Bruno S, Tetta C, Kalinina N,
Porta M and Camussi G: Platelet-derived growth factor regulates the
secretion of extracellular vesicles by adipose mesenchymal stem
cells and enhances their angiogenic potential. Cell Commun Signal.
12:262014. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Lee JK, Park SR, Jung BK, Jeon YK, Lee YS,
Kim MK, Kim YG, Jang JY and Kim CW: Exosomes derived from
mesenchymal stem cells suppress angiogenesis by down-regulating
VEGF expression in breast cancer cells. PLoS One. 8:e842562013.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Borgström P, Hillan KJ, Sriramarao P and
Ferrara N: Complete inhibition of angiogenesis and growth of
microtumors by anti-vascular endothelial growth factor neutralizing
antibody: Novel concepts of angiostatic therapy from intravital
videomicroscopy. Cancer Res. 56:4032–4039. 1996.PubMed/NCBI
|
|
61
|
Chamorro-Jorganes A, Araldi E, Penalva LO,
Sandhu D, Fernández-Hernando C and Suárez Y: MicroRNA-16 and
microRNA-424 regulate cell-autonomous angiogenic functions in
endothelial cells via targeting vascular endothelial growth factor
receptor-2 and fibroblast growth factor receptor-1. Arterioscler
Thromb Vasc Biol. 31:2595–2606. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Dejean E, Renalier MH, Foisseau M, Agirre
X, Joseph N, de Paiva GR, Al Saati T, Soulier J, Desjobert C,
Lamant L, et al: Hypoxia-microRNA-16 downregulation induces VEGF
expression in anaplastic lymphoma kinase (ALK)-positive anaplastic
large-cell lymphomas. Leukemia. 25:1882–1890. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Hua Z, Lv Q, Ye W, Wong CK, Cai G, Gu D,
Ji Y, Zhao C, Wang J, Yang BB and Zhang Y: MiRNA-directed
regulation of VEGF and other angiogenic factors under hypoxia. PLoS
One. 1:e1162006. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Chang AI, Schwertschkow AH, Nolta JA and
Wu J: Involvement of mesenchymal stem cells in cancer progression
and metastases. Curr Cancer Drug Targets. 15:88–98. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Wang M, Zhao C, Shi H, Zhang B, Zhang L,
Zhang X, Wang S, Wu X, Yang T, Huang F, et al: Deregulated
microRNAs in gastric cancer tissue-derived mesenchymal stem cells:
Novel biomarkers and a mechanism for gastric cancer. Br J Cancer.
110:1199–1210. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Lin R, Wang S and Zhao RC: Exosomes from
human adipose-derived mesenchymal stem cells promote migration
through Wnt signaling pathway in a breast cancer cell model. Mol
Cell Biochem. 383:13–20. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Wolfson B, Eades G and Zhou Q: Roles of
microRNA-140 in stem cell-associated early stage breast cancer.
World J Stem Cells. 6:591–597. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Li Q, Eades G, Yao Y, Zhang Y and Zhou Q:
Characterization of a stem-like subpopulation in basal-like ductal
carcinoma in situ (DCIS) lesions. J Biol Chem. 289:1303–1312. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Lee HK, Finniss S, Cazacu S, Bucris E,
Ziv-Av A, Xiang C, Bobbitt K, Rempel SA, Hasselbach L, Mikkelsen T,
et al: Mesenchymal stem cells deliver synthetic microRNA mimics to
glioma cells and glioma stem cells and inhibit their cell migration
and self-renewal. Oncotarget. 4:346–361. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Ono M, Kosaka N, Tominaga N, Yoshioka Y,
Takeshita F, Takahashi RU, Yoshida M, Tsuda H, Tamura K and Ochiya
T: Exosomes from bone marrow mesenchymal stem cells contain a
microRNA that promotes dormancy in metastatic breast cancer cells.
Sci Signal. 7:ra632014. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Shimbo K, Miyaki S, Ishitobi H, Kato Y,
Kubo T, Shimose S and Ochi M: Exosome-formed synthetic microRNA-143
is transferred to osteosarcoma cells and inhibits their migration.
Biochem Biophys Res Commun. 445:381–387. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Hamburg MA and Collins FS: The path to
personalized medicine. N Engl J Med. 363:301–304. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Dai S, Wei D, Wu Z, Zhou X, Wei X, Huang H
and Li G: Phase I clinical trial of autologous ascites-derived
exosomes combined with GM-CSF for colorectal cancer. Mol Ther.
16:782–790. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Greco SJ and Rameshwar P: Mesenchymal stem
cells in drug/gene delivery: Implications for cell therapy. Ther
Deliv. 3:997–1004. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Matuskova M, Hlubinova K, Pastorakova A,
Hunakova L, Altanerova V, Altaner C and Kucerova L: HSV-tk
expressing mesenchymal stem cells exert bystander effect on human
glioblastoma cells. Cancer Lett. 290:58–67. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Chen TS and Lim SK: Measurement of
precursor miRNA in exosomes from human ESC-derived mesenchymal stem
cells. Methods Mol Biol. 1024:69–86. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Lim PK, Bliss SA, Patel SA, Taborga M,
Dave MA, Gregory LA, Greco SJ, Bryan M, Patel PS and Rameshwar P:
Gap junction-mediated import of microRNA from bone marrow stromal
cells can elicit cell cycle quiescence in breast cancer cells.
Cancer Res. 71:1550–1560. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Munoz JL, Bliss SA, Greco SJ, Ramkissoon
SH, Ligon KL and Rameshwar P: Delivery of functional anti-miR-9 by
mesenchymal stem cell-derived exosomes to glioblastoma multiforme
cells conferred chemosensitivity. Mol Ther Nucleic Acids.
2:e1262013. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Boelens MC, Wu TJ, Nabet BY, Xu B, Qiu Y,
Yoon T, Azzam DJ, Twyman-Saint Victor C, Wiemann BZ, Ishwaran H, et
al: Exosome transfer from stromal to breast cancer cells regulates
therapy resistance pathways. Cell. 159:499–513. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Lou G, Song X, Yang F, Wu S, Wang J, Chen
Z and Liu Y: Exosomes derived from miR-122-modified adipose
tissue-derived MSCs increase chemosensitivity of hepatocellular
carcinoma. J Hematol Oncol. 8:1222015. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Greco KA, Franzen CA, Foreman KE, Flanigan
RC, Kuo PC and Gupta GN: PLK-1 silencing in bladder cancer by siRNA
delivered with exosomes. Urology. 91:241.e1–e7. 2016. View Article : Google Scholar
|
|
82
|
Pascucci L, Coccè V, Bonomi A, Ami D,
Ceccarelli P, Ciusani E, Viganò L, Locatelli A, Sisto F, Doglia SM,
et al: Paclitaxel is incorporated by mesenchymal stromal cells and
released in exosomes that inhibit in vitro tumor growth: A new
approach for drug delivery. J Control Release. 192:262–270. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Fuhrmann G, Serio A, Mazo M, Nair R and
Stevens MM: Active loading into extracellular vesicles
significantly improves the cellular uptake and photodynamic effect
of porphyrins. J Control Release. 205:35–44. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Fonsato V, Collino F, Herrera MB,
Cavallari C, Deregibus MC, Cisterna B, Bruno S, Romagnoli R,
Salizzoni M, Tetta C and Camussi G: Human liver stem cell-derived
microvesicles inhibit hepatoma growth in SCID mice by delivering
antitumor microRNAs. Stem Cells. 30:1985–1998. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Gjorgieva D, Zaidman N and Bosnakovski D:
Mesenchymal stem cells for anti-cancer drug delivery. Recent Pat
Anticancer Drug Discov. 8:310–318. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Toh WS, Lai RC, Hui JH and Lim SK: MSC
exosome as a cell-free MSC therapy for cartilage regeneration:
Implications for osteoarthritis treatment. Semin Cell Dev Biol. Nov
18–2016.(Epub ahead of print). View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Sherman LS, Shaker M, Mariotti V and
Rameshwar P: Mesenchymal stromal/stem cells in drug therapy: New
perspective. Cytotherapy. 19:19–27. 2017. View Article : Google Scholar : PubMed/NCBI
|