Introduction
Lung cancer is the most common type of malignant
cancer with the highest mortality rate worldwide (1). In China, approximately 300,000 new lung
cancer patients and more than 250,000 mortalities associated with
lung cancer are predicted annually (2). Lung adenocarcinoma (LAC) is the most
common type of lung cancer, accounting for ~40% of cases (3). Despite recent advances in diagnosis,
chemotherapy and targeted therapy, the overall survival rate of
patients with LAC remains low at an advanced stage (5-year overall
survival rate varies from 70% in stage IA to 25% in stage IIIA)
(4). Currently, the most effective
therapy for LA is complete surgical resection. However, a large
number of patients with LAC have advanced stage IIIB or IV disease
when first diagnosed (3). Thus, the
elucidation of the molecular mechanisms underlying the
tumorigenicity of LAC is essential for the development of novel
treatments for this disease.
MicroRNAs (miRNAs/miRs) are a class of endogenous
single-stranded short non-coding RNAs that silence target mRNAs by
base-pairing with the 3′-untranslated region (3′UTR) of target
genes in order to mediate translational repression and mRNA
degradation (5). It has been reported
that miRNAs may regulate >50% of all human protein-coding genes
expressions and serve an important role in various biological
processes, including cell differentiation, metabolism,
proliferation, apoptosis and tumorigenesis (6–8). It has
been demonstrated that dysregulation of specific miRNAs contributes
to the development and progression of cancer, including LAC cancer
(9). Furthermore, miRNAs are markedly
implicated in multiple steps of LAC occurrence and development,
including proliferation, recurrence and metastasis, and
miRNA-targeted treatment approaches have revealed marked potential
in controlling the advanced stage of LAC (10–12).
The aim of the present study was to investigate the
biological function of miR-383-5p in LAC. The expression profile of
miR-383-5p and its clinicopathological characteristics was analyzed
in patients with LAC. Subsequently, the effect of miR-383-5p on
proliferation, cell cycle and apoptosis in LAC cells was observed.
The results demonstrated that miR-383-5p suppresses the
proliferation by directly targeting cancerous inhibitor of protein
phosphatase 2A (CIP2A) in LAC.
Materials and methods
Patients and tissue samples
Paired human LAC samples and adjacent normal tissues
(≥3 cm away from the tumor) were obtained from 72 patients
(male=40; female=32; average age, 43.79±6.33; range 35–61 years)
who received surgical resection between January 2013 and May 2014
at the Department of Respiratory, The First Hospital Affiliated to
the Xinxiang Medical College (Weihui, China). All surgical
specimens were snap-frozen in liquid nitrogen and stored at −80°C
following resection until RNA extraction was performed. All
patients did not receive chemotherapy or radiotherapy prior to
surgery. The diagnoses of these tissue samples were confirmed by
pathologists in The First Hospital Affiliated to the Xinxiang
Medical College. Written informed consent was obtained from all
patients prior to enrolment in the present study the study protocol
was approved by the Ethics Committee of The First Hospital
Affiliated to the Xinxiang Medical College.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR) for miRNA
Total RNA from fresh tissues was isolated using an
RNA Extraction Kit (Qiagen, Inc., Valencia, CA, USA), according to
the manufacturer's protocol. Complementary DNA was obtained using
specific miRNA primers for miR-383-5p (reverse transcription
primer:
5′-GTCGTATCCAGTGCGTGTCGTGGAGTCGGCAATTGCACTGGATACGACAGCCAC-3′;
protocol: 30 min at 16°C, 30 min at 42°C, and 5 min at 85°C)
Applied Biosystems; Thermo Fisher Scientific, Inc., Waltham, MA,
USA) using the miScript Reverse Transcription Kit (Qiagen, Inc.).
The expression level of miR-383-5p was quantified using miRNA
specific TaqMan miRNA Assay kit (Applied Biosystems; Thermo Fisher
Scientific, Inc.). qPCR was performed using the SYBR PCR Master Mix
(Applied Biosystems; Thermo Fisher Scientific, Inc.) and ABI 7500
Fast (Applied Biosystems; Thermo Fisher Scientific, Inc.). PCR was
performed as follows: 25 cycles of 10 min at 98°C, 10 sec at 98°C,
10 sec at 55°C and 20 sec at 72°C, with a final extension at 72°C
for 5 min. The PCR primers used were as follows: miR-383-5p
forward, 5′-GGGAGATCAGAAGGTGATTGTGGCT-3′ and reverse,
5′-CAGTGCGTGTCGTGGAGT-3′; U6 forward, 5′-CTCGCTTCGGCAGCACA-3′ and
reverse, 5′-AACGCTTCACGAATTTGCGT-3′. The relative quantification of
miR-383-5p was determined using the 2−ΔΔCq method
(13), with U6 small nuclear (sn)RNA
used as the endogenous control to normalize the data.
Cell culture
A549 and H1299 human lung adenocarcinoma cell lines
were obtained from the American Type Culture Collection (Manassas,
VA, USA) and grown in RPMI-1640 medium (Invitrogen; Thermo Fisher
Scientific, Inc.) supplemented with 10% fetal bovine serum (Thermo
Fisher Scientific, Inc.) and 100 U/ml penicillin/streptomycin
(Invitrogen; Thermo Fisher Scientific, Inc.) in a 37°C humidified
incubator containing 5% CO2.
RNA oligonucleotide and cell
transfection
The miR-383-5p mimic and control miRNA mimic were
purchased from Shanghai GenePharma Co., Ltd. (Shanghai, China). The
cells were treated with 50 nM GMR-miR™ mixed with the miRNA mimic
(30 nM) using Lipofectamine™ 2000 reagent (Invitrogen; Thermo
Fisher Scientific, Inc.), according to the manufacturer's protocol,
when the density of cells was <70%. All the assays were
performed 48 h after transfection.
[3H]thymidine incorporation
assay
A549 and H1299 cells were plated onto 24-well plates
at a density of ~1×104 cells/well. Cells were
serum-starved for 12 h at 37°C followed by addition of serum and
[3H]thymidine (2 Ci/mM) for 4 h. Subsequently, the cells
were fixed in 0.3 ml 10% trichloroacetic acid and lysed in 100 µl
0.2 M NaOH/0.2% SDS for 10 min at 25°C. The radioactivity was
detected using a liquid scintillation counting system (Beckman
Coulter, Inc., Brea, CA, USA).
Cell cycle analysis
A total of 1×106 A549 or H1299 cells were
harvested, washed with ice-cold PBS and fixed in 70% ice-cold
ethanol at 4°C overnight. The fixed cells were washed with PBS and
resuspended in 1 ml PBS supplemented with 100 µg/ml bovine
pancreatic RNase A (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany)
and 40 µg/ml propidium iodide (PI; Sigma-Aldrich; Merck KGaA) for
30 min at 4°C, cell cycle analysis was performed with a Becton
Dickinson FACSCalibur cytometer (BD Biosciences, Inc., Franklin
Lakes, NJ, USA). Cell cycle analysis was performed using ModFit
software (version 3.2.1, Verity Software House, Topsham, ME,
USA).
Apoptosis assay
Cell apoptosis were detected using the Annexin
V-Fluorescein Isothiocyanate (FITC) Apoptosis kit (Merck KGaA),
according to the manufacturer's protocol. Briefly, cells were
washed with ice-cold PBS and incubated in 500 µl ice-cold 1X
binding buffer containing 2.3 ml Annexin V-FITC for 10 min at 4°C,
followed by 10 min of incubation at room temperature in the dark.
Subsequently, all cells were resuspended in 500 ml ice-cold 1X
binding buffer supplemented with 5 ml PI at room temperature for 15
min. Annexin V-FITC and PI signals were detected using a flow
cytometer (FACSCalibur™;BD Biosciences, San Jose, CA, USA).
Western blot assays
Total protein lysates were extracted using
radioimmunoprecipitation assay lysis buffer (Sigma-Aldrich; Merck
KGaA) at 4°C for 30 min and detected using a bicinchoninic acid kit
(Pierce; Thermo Fisher Scientific, Inc.). Protein (30 µg) was
separated by 10% SDS-PAGE and transferred onto polyvinylidene
difluoride membranes. Subsequently the membranes were blocked by 5%
BSA (Sigma-Aldrich, Merck KGaA) at room temperature for 1 h, washed
with TBST 3 times and then probed with primary antibodies against
the following: CIP2A (cat no. NB110-59722; dilution, 1:1,000;
duration, 4°C overnight, Novus Biologicals, LLC, Littleton, CO,
USA) and GAPDH (cat. no. 5174; dilution, 1:2,000; duration, 4°C
overnight; Cell Signaling Technology, Inc., Danvers, MA, USA),
which was used as a control. The membranes were washed 3 times with
TBST and incubated with horseradish peroxidase-linked secondary
goat anti-rabbit antibody (cat. no. 1662408; dilution, 1:3,000;
duration, 37°C for 1 h; Bio-Rad Laboratories, Inc., Hercules, CA,
USA). The bands were visualized using an enhanced chemiluminescence
detection reagent by the ChemiDoc XRS system (Bio-Rad Laboratories,
Inc.).
Immunohistochemistry assay
For the immunohistochemistry assay, 10%
formalin-fixed paraffin-embedded tissue sections (5 µm thick) were
deparaffinized and rehydrated in graded alcohol (50, 65, 75, 85, 95
and 100%) at room temperature for 1 h. An endogenous
antigen-retrieval procedure was performed using 10 mM citrate
buffer, pH 6.0, at 95°C for 10 min. Subsequently, slides were
washed with PBS and incubated with primary antibodies at 4°C in a
humidified chamber overnight. The primary anti-CIP2A monoclonal
antibody (cat. no. NB110-59722; dilution, 1:400; Novus Biologicals,
LLC) were incubated at 4°C overnight. This was followed by
incubation with biotinylated goat anti-rabbit serum IgG (cat. no.
21537; dilution, 1:500; Novus Biologicals, LLC, Littleton, CO,
USA). Subsequently, the antigen-antibody reaction was visualized
using diaminobenzidine serving as the chromogen under an Olympus
CX41 microscope and counted in 5 high-power fields (magnification,
×200).
Plasmid construction
The 3′UTR region of CIP2A was amplified from human
genomic DNA and inserted into the pmirGLO vector (Promega
Corporation, Madison, WI, USA) with HindIII and EcoRI restriction
sites at the 3′ end of the luciferase gene in order to construct
the luciferase reporter plasmids. For sequence point mutation,
site-directed mutagenesis of potential target sites in the CIP2A
3′UTR were performed using a QuikChange Site-Directed Mutagenesis
kit (Promega). The CIP2A recombinant plasmid (lacking 3′UTR) was
amplified by PCR with the following primers: Forward,
5′-CTGCCATCATGCCGATGTTCAT-3′ and reverse, 5′-CGGCTCTTAGGCGAAGGTG-3′
and the PrimeSTAR GXL DNA Polymerase (Takara Biotechnology Co.,
Ltd., Dalian, China). PCR thermocycling conditions were as follows:
30 cycles of 30 sec at 98°C, 90 sec at 56°C and 45 sec at 72°C with
a final extension at 72°C for 5 min. A LightCycler®
instrument (Roche Diagnostics GmbH, Mannheim, Germany) was used for
the PCR. The resulting PCR amplicons of CIP2A were cloned into the
T vector (Promega). The correct clones were confirmed by
sequencing.
miRNA target prediction
The following online miRNA target prediction
algorithms were used to evaluate the potential target genes of
miR-383-5p: TargetScan 6.2 database (http://www.targetscan.org/vert_71/). The target
prediction runs were performed with a context percentile of 95% and
a conserved method (14). The list of
potential target gene and binding site was available by searching
its database.
Luciferase assays
For the luciferase reporter assay, A549 and H1299
cells were seeded into a 24-well plate at density of 105
and co-transfected with 50 nM miR-338-5p mimic or control mimic and
200 ng reporter recombinant plasmid using Lipofectamine™ 2000
reagent (Invitrogen; Thermo Fisher Scientific, Inc.), according to
the manufacturer's protocol. At 48 h after transfection, luciferase
activity was determined using a dual-luciferase system kit
(Promega). Firefly luciferase activity was normalized against
Renilla luciferase gene activity.
Statistical analysis
Data analyses were performed using SPSS software
(version 15.0; SPSS, Inc., Chicago, IL, USA). Results are presented
as the mean ± standard deviation. Differences between two groups
were tested by Student's t-test, and differences among three or
more groups were measured by one-way analysis of variance. Count
data were analyzed using Fisher's exact tests. Univariate survival
analysis was performed using the Kaplan-Meier estimator method and
the log-rank test. P<0.05 was considered to indicate a
statistically significant difference.
Results
Associations between miR-383-5p
expression level and clinicopathological features of lung
adenocarcinoma
The expression levels of miR-383-5p in 72 LAC
tissues and adjacent non-tumorous tissues were determined using
RT-qPCR. The associations of miR-383-5p expression level with
various clinicopathological parameters of patients with LAC are
summarized in Table I. These results
indicated that miR-383-5p was significantly downregulated in 58.3%
(42/72) of the LAC tissues examined in comparison with the matched
adjacent non-cancerous tissues, 31.9% (23/72), from the same
patients. The results demonstrated that there were significant
associations between miR-383-5p downregulation and unfavorable
variables, including tumor size (P=0.0309) and differentiation
(P=0.0299). Furthermore, patients with low miR-383-5p expression
levels had a significantly decreased overall survival and
disease-free survival rate compared with those with high miR-383-5p
expression levels (Fig. 1A and
B).
 | Table I.Clinical association of miR-383-5p
expression in lung adenocarcinoma. |
Table I.
Clinical association of miR-383-5p
expression in lung adenocarcinoma.
|
|
| Relative miR-383-5p
expression level |
|
|---|
|
|
|
|
|
|---|
| Group | No. of patients | Low | High | P-value |
|---|
| Paraneoplastic
tissues | 72 | 23 | 49 | 0.0025 |
| Carcinoma tissue | 72 | 42 | 30 |
| Sex |
|
|
| 0.6356 |
| Male | 40 | 22 | 18 |
|
|
Female | 32 | 15 | 17 |
|
| Age, years |
|
|
| 1.0000 |
|
>65 | 39 | 18 | 21 |
|
|
≤65 | 33 | 15 | 18 |
|
| Size of carcinoma,
cm |
|
|
| 0.0309 |
|
>3 | 29 | 18 | 11 |
|
| ≤3 | 43 | 15 | 28 |
|
| TNM stage |
|
|
| 0.2381 |
|
I–II | 35 | 15 | 20 |
|
|
III–IV | 37 | 22 | 15 |
|
| Degree of
differentiation |
|
|
| 0.0299 |
| Well
and moderately | 43 | 16 | 27 |
|
|
Poorly | 29 | 19 | 10 |
|
| Lymph node
metastasis |
|
|
| 1.000 |
|
Negative | 31 | 17 | 14 |
|
|
Positive | 41 | 23 | 18 |
|
| Distant
metastasis |
|
|
| 0.6376 |
|
Negative | 37 | 17 | 20 |
|
|
Positive | 35 | 19 | 16 |
|
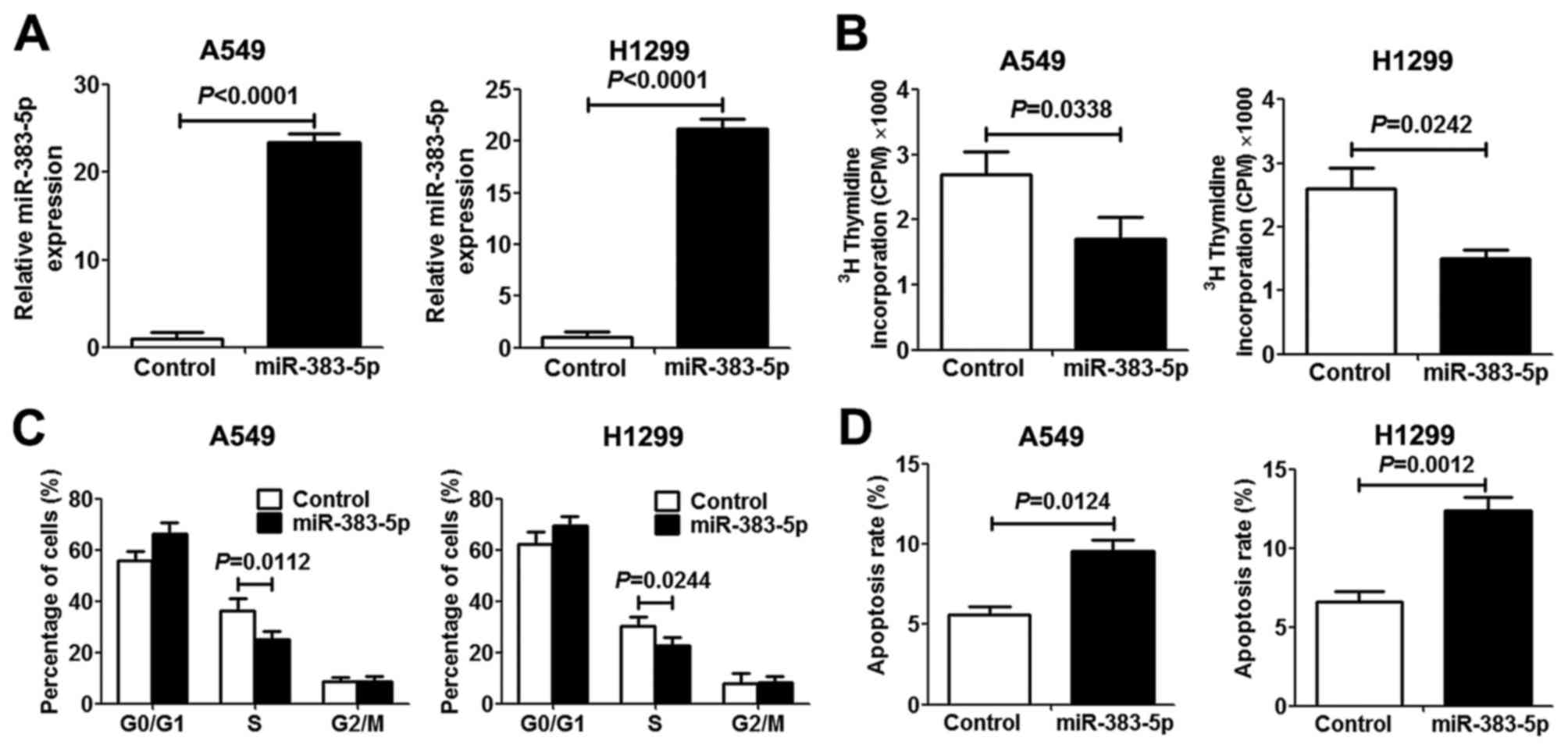
miR-383-5p inhibits proliferation and
induces apoptosis in LAC cells
In order to investigate the role of miR-383-5p in
human LAC proliferation and apoptosis, miR-383-5p was overexpressed
in human A549 and H1299 LAC cells by transfection with an
miR-383-5p mimic. The expression level of miR-383-5p in transfected
cells revealed a significant increase compared with transfected
control cells, which indicated that mir-383-5p was successfully
transfected into LAC cells (Fig. 2A).
[3H]thymidine incorporation assays and miR-383-5p
overexpression significantly inhibited A549 and H1299 cell
proliferation (Fig. 2B). To further
elucidate the anti-proliferative mechanism underlying miR-383-5p in
LAC cells, the cell cycle and apoptosis were analyzed. As presented
in Fig. 2C and D, miR-383-5p
overexpression in LAC cells significantly increased the proportion
of cells in the G0/G1 cell cycle phase and
decreased the proportion of cells in S phase compared with the
control group. Furthermore, promotion of cell apoptosis was
observed in LAC cells following transfection with miR-383-5p.
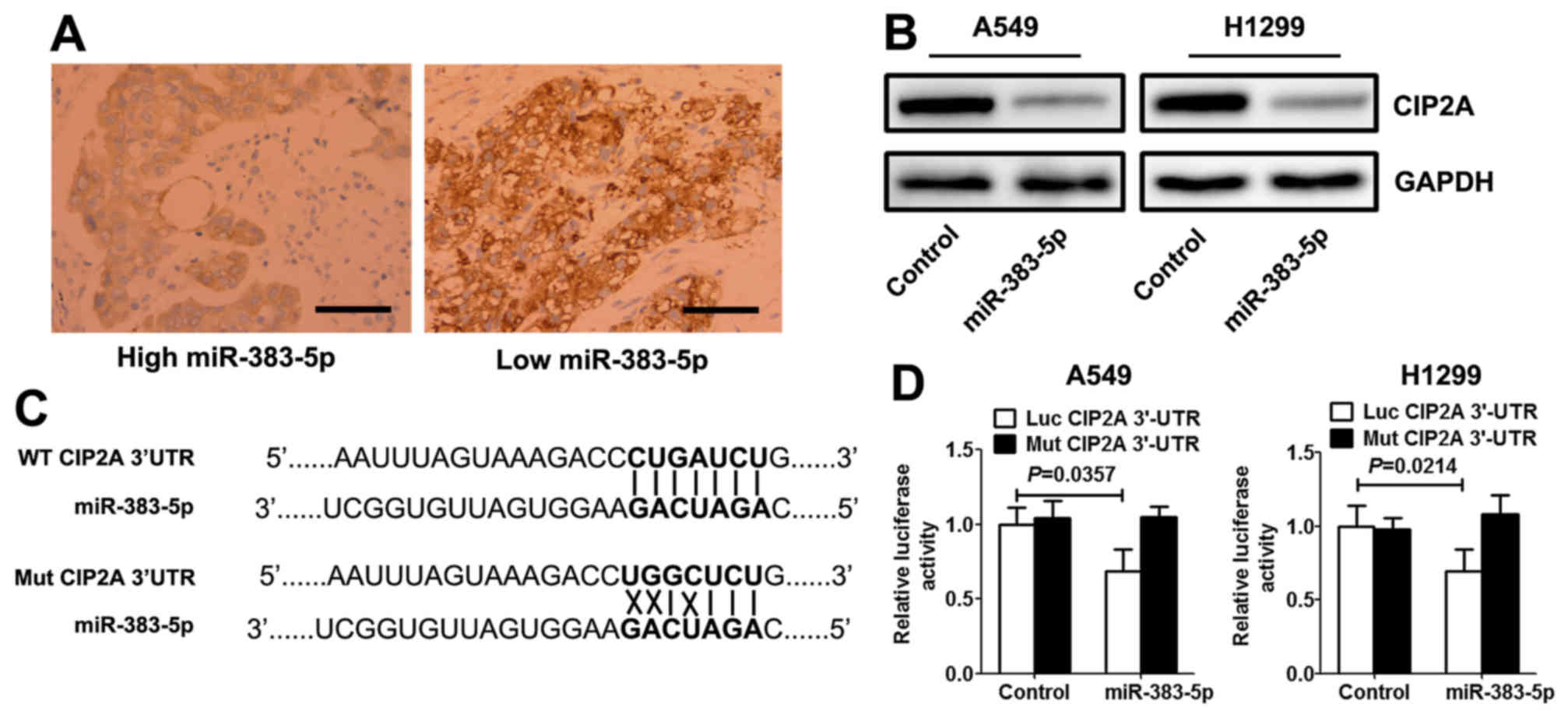
miR-383-5p directly targets CIP2A in
LAC cells
Using open access software TargetScan 6.2 database
(http://www.targetscan.org/vert_71/),
CIP2A was selected as a preferred candidate target gene of
miR-383-5p. Immunohistochemistry analysis demonstrated that the
expression levels of CIP2A in LAC with high miR-383-5p expression
levels were significantly decreased compared with those with low
miR-383-5p expression level (Fig.
3A). Western blotting revealed that miR-383-5p mimic
significantly decreased the expression level of CIP2A protein in
LAC cells (Fig. 3B). A target
prediction program (TargetScan) was used to identify putative
miRNA-binding sites in the 3′UTR of CIP2A. The potential wild-type
and mutant CIP2A 3′UTR fragment were cloned into a luciferase
reporter gene system (Fig. 3C). LAC
cells were co-transfected with a vector containing wild-type/mutant
3′UTR of CIP2A and miR-383-5p mimic (or control mimic).
Overexpression of miR-383-5p in the two LAC cell lines induced a
significantly decreased luciferase activity for wild-type, whereas
no alteration in luciferase activity was detected with the mutant
CIP2A 3′UTR luciferase reporter plasmid (Fig. 3D).
Upregulation of CIP2A reverses the
effects of miR-383-5p on proliferation
To further investigate miR-383-5p repression of LAC
cell proliferation mediated by CIP2A, A549 and H1299 cells were
co-transfected with miR-383-5p mimic (or control mimic) with CIP2A
constructs lacking the respective 3′UTR or empty vector. Western
blotting was performed to evaluate the expression levels of CIP2A
protein. As presented in Fig. 4A, the
co-transfection rescued the decreased expression level of CIP2A
protein in LAC cells that was induced by miR-383-5p. Additionally,
restoration of CIP2A expression level reversed the inhibitory
effects of exogenous miR-383-5p on proliferation, resulting in a
significant increase in DNA synthesis (Fig. 4B). Similarly, re-expression of CIP2A
exhibited an apparent rescued S cell cycle phase and decreased the
apoptosis rate in LAC cells (Fig. 4C and
D).
Discussion
According to previous studies, miRNAs may offer a
novel regulatory mechanism of gene expression, and miRNAs may act
as either oncogenes or tumor suppressors in light of the biological
function of their target genes (15–19). It
has been suggested that miRNA expression levels are associated with
specific clinical characteristics of cancer, thus they can be used
to classify normal and cancerous tissues, as well as to predict
prognosis (20). Recent studies have
revealed that dysregulation of miR-383 is associated with various
malignancies, including hepatocellular carcinoma (21), pancreatic cancer (22), glioma (23), testicular embryonal carcinoma
(24) and medulloblastoma (25).
Our results demonstrated that the expression level
of miR-383-5p was decreased in LAC, and the expression level of
miR-383-5p was associated with tumor size and differentiation,
suggesting that miR-383-5p may be associated with tumorigenesis of
LAC. Furthermore, patients with low expression levels of miR-383-5p
had decreased overall and disease-free survival rates. The results
of the present study identified that miR-383-5p was an independent
prognostic marker for predicting overall and disease-free survival
of patients with LAC. The results of the present study suggested
that the status of miR-383-5p was critical for progression of LAC.
Overexpression of miR-383-5p in LAC cells suppressed proliferation
by blocking G1-S transition and inducing apoptosis,
suggesting that miR-383-5p acted as a tumor suppressor in LAC.
Numerous studies have suggested that miR-383-5p inhibits tumor cell
growth and functions as a negative regulator of proliferation
(24,26), and upregulation of miR-383 induces a
inhibition of the transition from the G0/G1
phase to S cell cycle phase (27).
The results of the present study were consistent with those of
previous studies demonstrating that miR-383-5p inhibits cell growth
by blocking the G1-S cell cycle transition and inducing
apoptosis.
miRNAs are known to control diverse biological
processes via the regulation of target genes. Furthermore, the
present study demonstrated that an inverse association between
miR-383-5p and CIP2A expression levels was observed in LAC tissue
samples, and revealed that the increased miR-383-5p expression
level in LAC cells resulted in downregulation of the CIP2A protein
expression level. Additionally, the present study searched the
TargetScan database and demonstrated that CIP2A has a predicted
binding site of miR-383-5p within its 3′UTR. To verify whether
CIP2A is a direct target of miR-383-5p in LAC cells, the luciferase
reporter assay was performed and the data indicated that miR-383-5p
was able to bind efficiently to the predicted miR-383-5p-binding
site within the CIP2A 3′UTR. This phenomenon provided important
evidence indicating a direct interaction between miR-383-5p and
CIP2A.
CIP2A, also known as KIAA1524 and p90, is implicated
as a human oncoprotein that promotes the stability of c-Myc protein
and inhibits the degradation of c-Myc by inhibiting the protein
phosphatase 2A-mediated dephosphorylation of Myc at
Ser62 (28). A number of
studies have documented that CIP2A serves an important role in cell
proliferation (29), transformation
(30), drug resistance (31) and maintenance of a malignant cellular
phenotype (32). Furthermore, CIP2A
status was a significant prognostic factor for patients with
non-small cell carcinoma, and CIP2A protein expression levels were
revealed to be overexpressed in human lung cancer samples and
associated with poor survival rates (33). Previous studies have provided direct
evidence that CIP2A may promote cell proliferation via the protein
kinase B signaling pathway and protect the non-small cell lung
cancer cells from apoptosis (34,35). These
results imply an oncogenic role for CIP2A. The present study
verified that restoration of the CIP2A expression level abrogated
the inhibitory effect of miR-383-5p on LAC cell proliferation,
supporting evidence that CIP2A overexpression is a feature and may
be a critical event that occurs in LAC carcinogenesis. Taken
together, the results of the present study indicated that
miR-383-5p exerts an inhibitory effect on LAC, at least in part, by
inhibiting CIP2A.
In conclusion, the results of the present study
indicated that downregulation of miR-383-5p is significantly
associated with larger tumor size, lower differentiation degree and
poor survival in patients with LAC. The results of the present
study revealed novel insights into the molecular mechanisms by
which miR-383-5p exerts its negative effects on cell proliferation
in LAC cells by inhibition of CIP2A. This newly identified target
of miR-383-5p may provide a novel therapeutic target and strategy
for the treatment of patients with LAC.
Acknowledgements
The present study was supported by the Scientific
and Technical Project of Henan Health Department (grant no.
200804056).
References
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
She J, Yang P, Hong Q and Bai C: Lung
cancer in China: Challenges and interventions. Chest.
143:1117–1126. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wang H, Zhu LJ, Yang YC, Wang ZX and Wang
R: MiR-224 promotes the chemoresistance of human lung
adenocarcinoma cells to cisplatin via regulating G1/S
transition and apoptosis by targeting p21(WAF1/CIP1). Br J Cancer.
111:339–354. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Yan G, Yao R, Tang D, Qiu T, Shen Y, Jiao
W, Ge N, Xuan Y and Wang Y: Prognostic significance of microRNA
expression in completely resected lung adenocarcinoma and the
associated response to erlotinib. Med Oncol. 31:2032014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wang K, Liang Q, Wei L, Zhang W and Zhu P:
MicroRNA-608 acts as a prognostic marker and inhibits the cell
proliferation in hepatocellular carcinoma by macrophage migration
inhibitory factor. Tumour Biol. 37:3823–3830. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Hu J, Qiu M, Jiang F, Zhang S, Yang X,
Wang J, Xu L and Yin R: MiR-145 regulates cancer stem-like
properties and epithelial-to-mesenchymal transition in lung
adenocarcinoma-initiating cells. Tumour Biol. 35:8953–8961. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Johnnidis JB, Harris MH, Wheeler RT,
Stehling-Sun S, Lam MH, Kirak O, Brummelkamp TR, Fleming MD and
Camargo FD: Regulation of progenitor cell proliferation and
granulocyte function by microRNA-223. Nature. 451:1125–1129. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Xing F, Wu K and Watabe K: MicroRNAs in
cancer stem cells: New regulators of stemness. Curr Pharm Des.
20:5319–5327. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Qi J and Mu D: MicroRNAs and lung cancers:
From pathogenesis to clinical implications. Front Med. 6:134–155.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Gu Y, Wang XD, Lu JJ, Lei YY, Zou JY and
Luo HH: Effect of mir-16 on proliferation and apoptosis in human
A549 lung adenocarcinoma cells. Int J Clin Exp Med. 8:3227–3233.
2015.PubMed/NCBI
|
|
11
|
Arima C, Kajino T, Tamada Y, Imoto S,
Shimada Y, Nakatochi M, Suzuki M, Isomura H, Yatabe Y, Yamaguchi T,
et al: Lung adenocarcinoma subtypes definable by lung
development-related miRNA expression profiles in association with
clinicopathologic features. Carcinogenesis. 35:2224–2231. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chen DQ, Pan BZ, Huang JY, Zhang K, Cui
SY, De W, Wang R and Chen LB: HDAC 1/4-mediated silencing of
microRNA-200b promotes chemoresistance in human lung adenocarcinoma
cells. Oncotarget. 5:3333–3349. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Riffo-Campos ÁL, Riquelme I and
Brebi-Mieville P: Tools for Sequence-Based miRNA Target Prediction:
What to Choose? Int J Mol Sci. 17:pii: E1987. 2016. View Article : Google Scholar
|
|
15
|
Wang S, Zhao X, Wang J, Wen Y, Zhang L,
Wang D, Chen H, Chen Q and Xiang W: Upregulation of microRNA-203 is
associated with advanced tumor progression and poor prognosis in
epithelial ovarian cancer. Med Oncol. 30:6812013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhu K, Ding H, Wang W, Liao Z, Fu Z, Hong
Y, Zhou Y, Zhang CY and Chen X: Tumor-suppressive miR-218-5p
inhibits cancer cell proliferation and migration via EGFR in
non-small cell lung cancer. Oncotarget. 7:28075–28085. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Xiao P and Liu WL: MiR-142-3p functions as
a potential tumor suppressor directly targeting HMGB1 in
non-small-cell lung carcinoma. Int J Clin Exp Pathol.
8:10800–10807. 2015.PubMed/NCBI
|
|
18
|
Li YQ, Lu JH, Bao XM, Wang XF, Wu JH and
Hong WQ: MiR-24 functions as a tumor suppressor in nasopharyngeal
carcinoma through targeting FSCN1. J Exp Clin Cancer Res.
34:1302015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yu T, Liu L, Li J, Yan M, Lin H, Liu Y,
Chu D, Tu H, Gu A and Yao M: MiRNA-10a is upregulated in NSCLC and
may promote cancer by targeting PTEN. Oncotarget. 6:30239–30250.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Xu F, Zhang H, Su Y, Kong J, Yu H and Qian
B: Up-regulation of microRNA-183-3p is a potent prognostic marker
for lung adenocarcinoma of female non-smokers. Clin Transl Oncol.
16:980–985. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chen L, Guan H, Gu C, Cao Y, Shao J and
Wang F: miR-383 inhibits hepatocellular carcinoma cell
proliferation via targeting APRIL. Tumour Biol. 37:2497–2507. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Han S, Cao C, Tang T, Lu C, Xu J, Wang S,
Xue L, Zhang X and Li M: ROBO3 promotes growth and metastasis of
pancreatic carcinoma. Cancer Lett. 366:61–70. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
He Z, Cen D, Luo X, Li D, Li P, Liang L
and Meng Z: Downregulation of miR-383 promotes glioma cell invasion
by targeting insulin-like growth factor 1 receptor. Med Oncol.
30:5572013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lian J, Tian H, Liu L, Zhang XS, Li WQ,
Deng YM, Yao GD, Yin MM and Sun F: Downregulation of microRNA-383
is associated with male infertility and promotes testicular
embryonal carcinoma cell proliferation by targeting IRF1. Cell
Death Dis. 1:e942010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Li KK, Pang JC, Lau KM, Zhou L, Mao Y,
Wang Y, Poon WS and Ng HK: MiR-383 is downregulated in
medulloblastoma and targets peroxiredoxin 3 (PRDX3). Brain Pathol.
23:413–425. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lü M, Tian H, Cao YX, He X, Chen L, Song
X, Ping P, Huang H and Sun F: Downregulation of
miR-320a/383-sponge-like long non-coding RNA NLC1-C (narcolepsy
candidate-region 1 genes) is associated with male infertility and
promotes testicular embryonal carcinoma cell proliferation. Cell
Death Dis. 6:e19602015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Xu Z, Zeng X, Tian D, Xu H, Cai Q, Wang J
and Chen Q: MicroRNA-383 inhibits anchorage-independent growth and
induces cell cycle arrest of glioma cells by targeting CCND1.
Biochem Biophys Res Commun. 453:833–838. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Liu N, He QM, Chen JW, Li YQ, Xu YF, Ren
XY, Sun Y, Mai HQ, Shao JY, Jia WH, et al: Overexpression of CIP2A
is an independent prognostic indicator in nasopharyngeal carcinoma
and its depletion suppresses cell proliferation and tumor growth.
Mol Cancer. 13:1112014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zheng Z, Qiao Z, Chen W, Gong R, Wang Y,
Xu L, Ma Y, Zhang L, Lu Y, Jiang B, et al: CIP2A regulates
proliferation and apoptosis of multiple myeloma cells. Mol Med Rep.
14:2705–2709. 2016.PubMed/NCBI
|
|
30
|
Liu Z, Ma L, Wen ZS, Cheng YX and Zhou GB:
Ethoxysanguinarine induces inhibitory effects and downregulates
CIP2A in lung cancer cells. ACS Med Chem Lett. 5:113–118. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Liu J, Wang M, Zhang X, Wang Q, Qi M, Hu
J, Zhou Z, Zhang C, Zhang W, Zhao W and Wang X: CIP2A is associated
with multidrug resistance in cervical adenocarcinoma by a
P-glycoprotein pathway. Tumour Biol. 37:2673–2682. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Junttila MR, Puustinen P, Niemelä M, Ahola
R, Arnold H, Böttzauw T, Ala-aho R, Nielsen C, Ivaska J, Taya Y, et
al: CIP2A inhibits PP2A in human malignancies. Cell. 130:51–62.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Dong QZ, Wang Y, Dong XJ, Li ZX, Tang ZP,
Cui QZ and Wang EH: CIP2A is overexpressed in non-small cell lung
cancer and correlates with poor prognosis. Ann Surg Oncol.
18:857–865. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Chao TT, Wang CY, Lai CC, Chen YL, Tsai
YT, Chen PT, Lin HI, Huang YC, Shiau CW, Yu CJ and Chen KF: TD-19,
an erlotinib derivative, induces epidermal growth factor receptor
wild-type nonsmall-cell lung cancer apoptosis through
CIP2A-mediated pathway. J Pharmacol Exp Ther. 351:352–358. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Lei N, Peng B and Zhang JY: CIP2A
regulates cell proliferation via the AKT signaling pathway in human
lung cancer. Oncol Rep. 32:1689–1694. 2014.PubMed/NCBI
|