Introduction
With increasing incidence and mortality, cancer is
the leading cause of death in China and is a major public health
problem. Based on GLOBOCAN estimates, approximately 14.1 million
new cancer cases and 8.2 million deaths occurred in 2012 worldwide
(1). Moreover, based on the
estimates of National Central Cancer Registry (NCCR) of China,
4.292 million new cancer cases and 2.814 million cancer deaths
would occur in China in 2015, lung cancer is the leading cause of
cancer-related death worldwide (2),
and the 5-year survival rate of patients with advanced lung cancer
remains low.
miRNAs are a class of endogenous, single-stranded,
small noncoding RNA molecules (~22 nucleotides), which are crucial
for post-transcriptional regulators of gene expression (3,4). In
recent years, many studies have reported promising biomarkers for
differential diagnosis of lung cancer (5–8).
However, accurate biomarkers of lung cancer early diagnosis still
remain largely unexplored. The discovery of miRNAs, are frequently
dysregulated in lung cancer and are implicated in lung cancer
growth, recurrence, and metastasis (9,10).
Consequently, miRNAs are considered as valuable biomarkers for
diagnosis of lung cancer (11,12).
Evidence from recent studies indicates promise of miRNAs in risk
assessment, prevention, early diagnosis, and prognosis of lung
cancer (9,13–16).
miRNA expression profiling is becoming an increasingly important
tool to reveal gene activity in cancer.
In this study, microarray of the miRNAs and mRNAs on
the same samples was performed and the intersection taken with The
Cancer Genome Atlas (TCGA) lung cancer miRNA/RNAseq dataset. Then,
miRNA-mRNA regulatory network was constructed to identify miRNA
candidates associated with lung cancer through integrating gene
expression and miRNA-target prediction. Furthermore, the expression
levels of miRNA candidates were validated by stem-loop real-time
reverse transcription PCR (qRT-PCR) in larger lung cancer
population. The relationship between signature miRNAs and the risk
of lung cancer were assessed by conditional logistic regression
analysis. Diagnostic value of these miRNAs was determined by areas
under receiver operating characteristic curves (ROC). The aim of
our study was to identify potential miRNA biomarkers of lung cancer
for early screening of high-risk populations and early
diagnosis.
Materials and methods
Specimen collection
Three lung tumor tissues and their paired adjacent
non-tumor tissues samples were collected for miRNA and mRNA
microarray analysis. In addition, 81 patients, including 64 males
and 17 females aged between 36–76 years, were recruited for qRT-PCR
analysis from the Nanjing Chest Hospital, with their consent and
agreement. All patients were confirmed by pathology as lung tumor
without preoperative radiotherapy or chemotherapy. Lung tumor
tissues and adjacent non-tumor tissues were obtained from surgical
specimens immediately after resection from patients. The adjacent
non-tumor tissues was located at least 5 cm away from tumor edge.
Tissue samples (0.5 cm3) were immersed in RNA locker
(Tiandz, China) at 4°C overnight and stored at −20°C until use.
This study was approved by the ethics committee of Zhongda Hospital
Affiliated to Southeast University.
Total RNA extraction
Total RNA was extracted from both lung tumor tissues
and adjacent non-tumor tissues using TRIzol reagent (Invitrogen,
Carlsbad, CA, USA) according to the instructions. Concentration and
integrity of extracted RNA was assessed using a NanoDrop 1000
spectrophotometer (NanoDrop Technologies, Houston, TX, USA).
Finally, all RNA samples showing A260/280 ratios between 1.8 and
2.0 were further analyzed for RNA integrity using agarose gel
electrophoresis. The quality and integrity of RNA were satisfactory
for all samples.
miRNA and mRNA microarray
Affymetrix™ Genechip miRNA 4.0 Array (Affymetrix,
Inc., Santa Clara, CA, USA) and Affymetrix Genechip HTA 2.0 Array
(Affymetrix, Inc.) were used in this study. Samples of miRNAs and
mRNAs from 6 specimens were labeled and hybridized according to the
manufacturer's protocol. Signals were normalized using the median
center tool for genes.
TCGA analysis of differential
expression of miRNAs and mRNAs
To improve the reliability and accuracy of the
studies, we analyzed miRNAs and mRNAs expression profile dataset of
lung cancer from The Cancer Genome Atlas (TCGA) database. A total
of 521 lung cancer patient were obtained from TCGA database. After
criteria of exclusion, a total of 465 lung cancer patients were
included in our study. In addition, the lung cancer RNA expression
data (level 3) of the corresponding patients were downloaded from
the TCGA Data Portal (March 2016). The RNA sequencing raw reads
(mRNAs) were post-processed and normalized by TCGA RNASeqV2 system.
Level 3, normalized miRNA expression data were downloaded from the
TCGA Data Portal performed using Illumina HiSeq and Illumina GA
miRNAs sequencing platforms (Illumina Inc., Hayward, CA, USA) and
quantile normalized before performing analysis.
Construction of miRNA-mRNAs network
and bioinformatics analysis
To construct miRNAs-RNAs network, the miRNA-mRNA
interactions were download from the starBase v2.0 database
(http://starbase.sysu.edu.cn/) (17), then aberrantly expressed
intersection miRNAs and mRNAs with absolute value of fold change ≥2
and P<0.05 were retained. Targetscan (http://www.targetscan.org/) and miRTarBase (http://mirtarbase.mbc.nctu.edu.tw/) (18) were performed to predict miRNA target
genes. The results could be verified in starBase database.
Furthermore, in combination with microarray data and TCGA result,
miRNA-mRNA co-expression network was constructed by Cytoscape v3.0
(19). The potential function of
these miRNAs was analyzed by DIANA-mirPath v.3.
miRNA expression analysis in lung
cancer population by qRT-PCR
To confirm the reliability and validity of the
microarray data, we selected 7 (miR-205-5p, miR-3917, miR-27a-5p,
miR-30a-3p, miR-30a-5p, miR-30c-2-3p and miR-30d-5p) differentially
expressed key miRNAs, which were found in non-small cell lung
cancer pathway, and analyzed their actual expression levels in 81
pairs of diagnosed lung tumor and adjacent non-tumor tissues by
qRT-PCR on StepOnePlus™ PCR System (Applied Biosystems, Waltham,
MA, USA). Reverse transcription reactions were conducted in two
steps. First, 0.5 µg of RNA samples was incubated in a 0.2 ml Ep
tube for 5 min at 65°C and held at 4°C. Then, the 10 µl mixture
which comprised 2 µl RT product which obtained from the front, 2 µl
5X RT buffer (Promega, Madison, WI, USA), 0.5 µl RT Enzyme Mix, 0.5
µl miRNA-specific stem-loop RT primers (RiboBio, Guangzhou, China),
and 5 µl RNase-free water (Tiangen, China) was incubated in a 0.2
ml Ep tube for 15 min at 37°C, 5 min at 98°C and subsequently held
at 4°C. PCR was then performed using Thunderbird SYBR qPCR Mix
(Toyobo Corp., Osaka, Japan) according to the manufacturer's
protocol. The PCR reaction components were 1 µl cDNA, 5 µl
Thunderbird SYBR qPCR Mix, 0.3 µl PCR primers (RiboBio) and 3.4 µl
RNase-free water. The reaction was performed at 95°C for 1 min,
followed by 40 cycles of 95°C for 15 sec, 60°C for 30 sec and 72°C
for 30 sec. Dissociation curve was analyzed from 60 to 90°C. U6 was
chosen as the endogenous standard. All assays were performed in
triplicates.
Statistical analysis
The microarray data and TCGA data analyses were
performed using one-way ANOVA, and absolute value of fold change ≥2
and P<0.05 was considered to indicate statistically significant
differences. qRT-PCR results were expressed as mean ± SD. miRNA
expression was calculated using the 2−∆∆Ct method, which
∆Ct = CtmiRNAs - CtU6 and ∆∆Ct =
∆Cttumor tissues - ∆Ctadjacent non-tumor
tissues. The threshold cycle indicates the fractional cycle
number at which the amount of amplified target reaches a fixed
threshold and the Ct value is negatively correlated with copy
numbers. Thus, in the final calculation, the Ct values are
multiplied with −1 for logistic regression analysis. Statistical
analysis was carried out with the Student's t-test for comparison
of two groups in qRT-PCR analysis, and conditional logistic
regression for evaluate the association between differentially
expressed miRNAs and the risk of lung cancer. In both cases,
differences with P<0.05 were considered to indicate a
statistically significant result. Operating characteristic curve
(ROC) was used to determine the signature miRNAs as the sensitivity
and specificity of detection of lung cancer.
Results
miRNA and mRNA microarray
analysis
Microarray analysis identified 116 miRNAs and 502
mRNAs that could distinguish lung tumor tissues from adjacent
non-tumor tissues (fold change >2). A total of 70 miRNAs and 136
mRNAs were upregulated, while 46 miRNAs and 366 mRNAs were
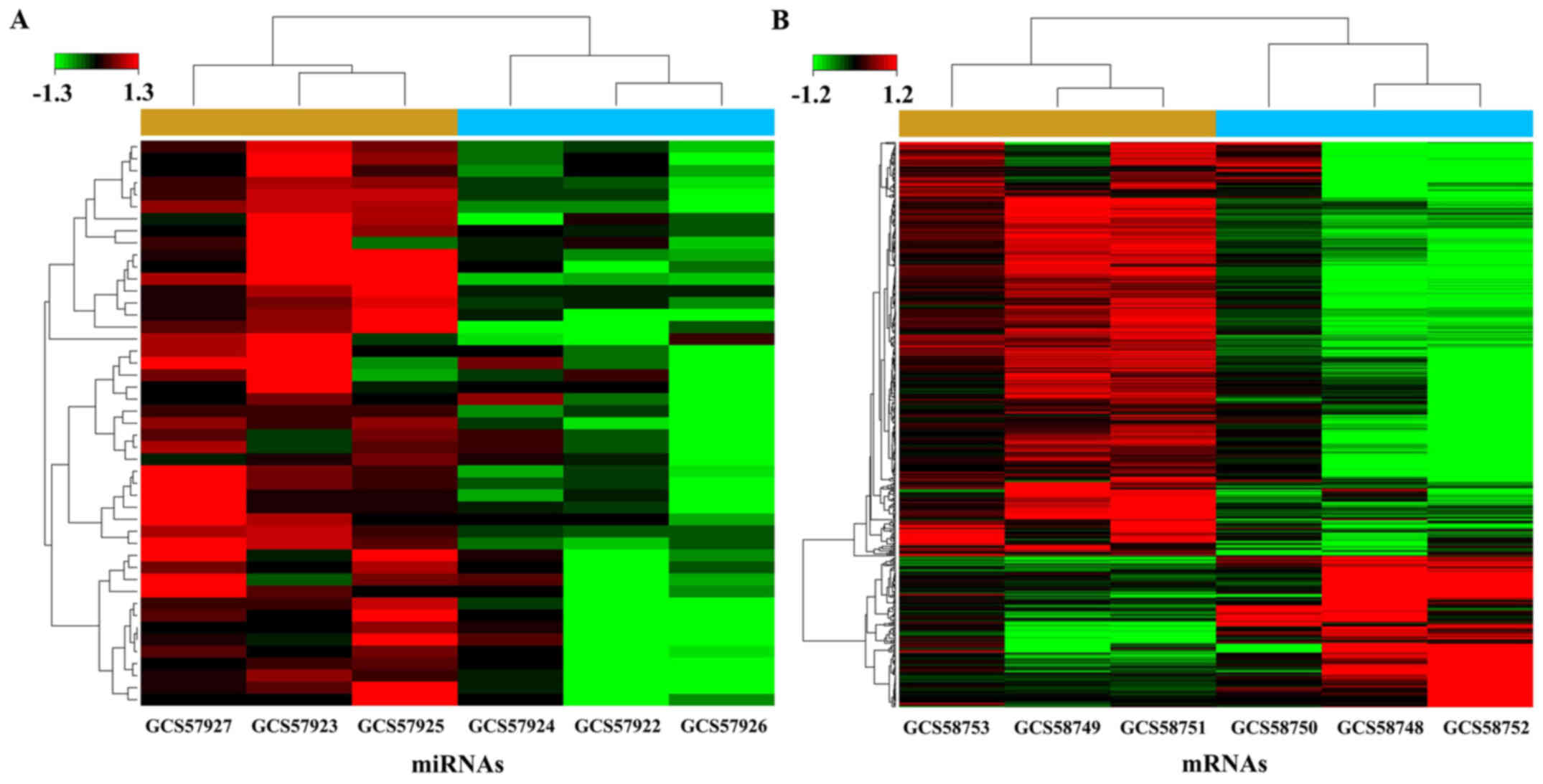
downregulated. Cluster analysis, based on the differentially
expressed miRNAs and mRNAs, successfully separated the tumor
tissues from the adjacent non-tumor tissues (Fig. 1). The analysis manifested the chips
in each case consistently. Top 40 differentially expressed miRNAs
and mRNAs are listed (Fig. 2).
miR-205-5p and miR-31-5p (fold change 10.825 and 10.083,
respectively) were the most upregulated miRNAs, and miR-4521 and
miR-486-5p (fold change −15.948 and −12.172) were the most
downregulated. IGHD3-3 and MMP1 (fold change 9.050 and 5.598,
respectively) were the most upregulated mRNAs, SLC6A4 and RTKN2
(fold change −11.842 and −11.063, respectively) were the most
downregulated.
TCGA analysis of differential
expression of miRNAs and mRNAs
We identified a total of 1030 miRNAs and 18633 mRNAs
from TCGA database, of these 118 and 3853 differentially expressed
miRNAs (absolute fold change >2, P<0.05) and mRNAs (absolute
fold change >2, P<0.05) were found between lung tumor tissues
and adjacent non-tumor lung tissues. Based on these data,
differentially expressed miRNAs and mRNAs were selected for further
analysis. Then 32 and 377 differentially expressed miRNAs and mRNAs
were obtained for further analysis after intersection between TCGA
and microarray data (Figs. 3 and
4).
miRNA target prediction and miRNA-mRNA
network construction
In order to establish miRNA-mRNA network, mRNAs
targeted by miRNAs was performed. Based on the miRNAs and mRNAs
described in Figs. 3 and 4, starBase v2.0 database, TargetScan and
miRTarbase were performed to predict miRNAs-targeted mRNAs. Then,
we identified 28 specific miRNAs related to 61 intersection mRNAs
(Table I). As described in Table I, the miRNA-mRNA network was
constructed via Cytoscape 3.0. Fig.
5, shows the 28 miRNAs and 61 mRNAs involved in the proposed
network.
 | Table I.Differential expression miRNAs and
their targeted mRNA prediction. |
Table I.
Differential expression miRNAs and
their targeted mRNA prediction.
| miRNAs | miRNA feature | Gene symbol | Gene feature |
|---|
| miR-133a-3p | Down | COL1A1, THBS2 | Up |
| miR-133b | Down | MMP9 | Up |
| miR-184 | Down | SLC7A5 | Up |
| miR-30a-5p | Down | CDC20, ITGA2,
KRT6B, SERPINB5, SLC7A5 | Up |
| miR-30b-3p | Down | FAM83B, HIST1H2BD,
SLC7A5, TNS4 | Up |
| miR-30c-2-3p | Down | BUB1, FAM83B,
TNS4 | Up |
| miR-30d-5p | Down | SLC7A5 | Up |
| miR-338-5p | Down | CLDN1 | Up |
| miR-363-3p | Down | ARNTL2, HMGA2 | Up |
| miR-451a | Down | MMP9 | Up |
| miR-141-3p | Up | GATA6 | Down |
| miR-148a-3p | Up | S1PR1 | Down |
| miR-193b-3p | Up | TGFBR3, TMTC1,
RTKN2, TACC1 | Down |
| miR-19a-3p | Up | NR4A2, LDLR,
ARHGEF26, SOCS3, PTPRB, SFTPA1, TMTC1, HECW2 | Down |
| miR-200a-3p | Up | GATA6, DLC1 | Down |
| miR-200a-5p | Up | HHIP | Down |
| miR-205-5p | Up | LRRK2 | Down |
| miR-210-3p | Up | SERTM1 | Down |
| miR-21-5p | Up | TIMP3, BTG2, IL1B,
TCF21, EDIL3, CYBRD1, SASH1, FAXDC2, LIFR, SEMA5A, LIMCH1, OLR1,
KLF9, WWC2, HPGD | Down |
| miR-221-5p | Up | LRRTM2, CLIC5 | Down |
| miR-224-5p | Up | FOSB | Down |
| miR-301a-3p | Up | CYBRD1, LDLR,
ARHGEF26, EDN1, SFTPA1, DLC1, MYH11, TMTC1 | Down |
| miR-31-5p | Up | SELE | Down |
| miR-34b-3p | Up | ACADL, ADH1B,
PAQR5, PLCE1 | Down |
| miR-409-5p | Up | ABCA3 | Down |
| miR-411-5p | Up | ART4, ITM2A | Down |
| miR-708-5p | Up | CD274, PAQR5 | Down |
| miR-7-1-3p | Up | LDLR, LIFR | Down |
Function analysis of differentially
expressed miRNAs
We next used the DIANA-mirPath V.3, which the Kyoto
Encyclopedia of Genes and Genomes (KEGG) to classify and analyze
the potential functions of these 28 miRNAs in the pathways. Our
analysis indicated that 18 upregulated and 10 downregulated miRNAs
were involved in signaling pathways related to Environmental
Information Processing and Human Diseases, with -ln (P-value) >3
(Fig. 6). The former group included
the Hippo, TGF-β, Wnt, ErbB and MAPK signaling pathway corresponded
to up-/downregulated miRNAs, respectively. While the latter group
included proteoglycans in cancer, renal cell carcinoma, glioma,
pathways in cancer and non-small cell lung cancer corresponded to
up-/downregulated miRNAs, respectively. Moreover, 23 miRNAs and 29
gene involved in signaling pathways related to non-small cell lung
cancer pathway, respectively.
miRNA expression analysis in lung
cancer population by qRT-PCR
qRT-PCR results demonstrated that expression of
miR-205-5p and miR-3917 were significantly higher in tumor tissues
than adjacent non-tumor tissues (P<0.05), while expression of
miR-27a-5p, miR-30a-3p, miR-30a-5p, miR-30c-2-3p, and miR-30d-5p
were significantly lower in tumor tissues than adjacent non-tumor
tissues (P<0.05). Expression levels of these 7 miRNAs were
consistent with the microarray results and TCGA data (Table II; Figs. 7 and 8).
 | Table II.Expression level of miRNAs analyzed
by qRT-PCR in lung tumor tissues and non-tumor tissues. |
Table II.
Expression level of miRNAs analyzed
by qRT-PCR in lung tumor tissues and non-tumor tissues.
| miRNAs | N | Groupa | ∆Ctb (mean ± SD) | ∆∆Ctb (mean ± SD) |
2−∆∆Ct | Fold change | P-value | t-value |
|---|
| miR-205-5p | 81 | Ca | 5.757±4.287 | −3.423±4.962 | 10.729 | 10.729 | <0.001 | −6.210 |
|
|
| N | 9.181±2.873 |
|
|
|
|
|
| miR-3917 | 81 | Ca | 19.047±1.718 | −0.937±2.021 | 1.914 | 1.914 | <0.001 | −4.173 |
|
|
| N | 19.984±1.828 |
|
|
|
|
|
| miR-27a-5p | 78 | Ca | 12.954±2.724 | 1.206±1.060 | 0.433 | −2.309 | <0.001 | 5.172 |
|
|
| N | 11.748±2.778 |
|
|
|
|
|
| miR-30a-3p | 81 | Ca | 12.348±3.059 | 2.718±1.939 | 0.152 | −6.579 | <0.001 | 12.615 |
|
|
| N | 9.630±2.550 |
|
|
|
|
|
| miR-30a-5p | 81 | Ca | 7.535±2.168 | 2.019±1.756 | 0.247 | −4.049 | <0.001 | 10.348 |
|
|
| N | 5.516±1.849 |
|
|
|
|
|
| miR-30c-2-3p | 73 | Ca | 19.931±3.084 | 2.238±2.245 | 0.212 | −4.717 | <0.001 | 8.517 |
|
|
| N | 17.692±2.506 |
|
|
|
|
|
| miR-30d-5p | 81 | Ca | 6.712±1.846 | 1.640±1.936 | 0.321 | −3.115 | <0.001 | 7.625 |
|
|
| N | 5.072±1.369 |
|
|
|
|
|
Association between verified miRNAs
and lung cancer
Conditional logistic regression analysis was used to
evaluate the association between differentially expressed miRNAs
and the risk of lung cancer. As shown in Table III, significantly increased risk
for lung cancer was associated with increased expression of
miR-205-5p and miR-3917 (OR 1.287 and 1.360, respectively) and
reduced expression of miR-27a-5p, miR-30a-3p, miR-30a-5p,
miR-30c-2-3p and miR-30d-5p (OR 0.846, 0.700, 0.585, 0.745 and
0.479, respectively). This suggested that these miRNAs could impact
on the risk of lung cancer and play an important role during lung
cancer development.
 | Table III.Differentially expression of miRNAs
associated with a high risk of lung cancer. |
Table III.
Differentially expression of miRNAs
associated with a high risk of lung cancer.
| miRNAs | N | Groupa | β | SE | Wald | P-value | OR | 95% CI |
|---|
| miR-205-5p | 81 | Ca | 0.252 | 0.051 | 24.887 | <0.001 | 1.287 | 1.166–1.421 |
|
|
| N |
|
|
|
| 1.000 |
|
| miR-3917 | 81 | Ca | 0.308 | 0.098 | 9.916 |
0.002 | 1.360 | 1.123–1.647 |
|
|
| N |
|
|
|
| 1.000 |
|
| miR-27a-5p | 78 | Ca | 0.167 | 0.064 | 6.756 |
0.009 | 0.846 | 0.746–0.960 |
|
|
| N |
|
|
|
| 1.000 |
|
| miR-30a-3p | 81 | Ca | −0.357 | 0.070 | 25.815 | <0.001 | 0.700 | 0.610–0.803 |
|
|
| N |
|
|
|
| 1.000 |
|
| miR-30a-5p | 81 | Ca | −0.536 | 0.103 | 26.963 | <0.001 | 0.585 | 0.478–0.716 |
|
|
| N |
|
|
|
| 1.000 |
|
| miR-30c-2-3p | 73 | Ca | −0.294 | 0.070 | 11.475 | <0.001 | 0.745 | 0.649–0.855 |
|
|
| N |
|
|
|
| 1.000 |
|
| miR-30d-5p | 81 | Ca | −0.736 | 0.137 | 28.712 | <0.001 | 0.479 | 0.366–0.627 |
|
|
| N |
|
|
|
| 1.000 |
|
ROC curve analysis of signature
miRNAs
ROC curve analysis demonstrated the area under curve
(AUC) 0.728, 0.758, 0.772, 0.734, 0.776 for miR-205-5p, miR-30a-3p,
miR-30a-5p, miR-30c-2-3p and miR-30d-5p (P-value <0.01, 95%
confidence interval: 0.650–0.806, 0.683–0.832, 0.699–0.845,
0.650–0.817 and 0.704–0.848, respectively, Table IV and Fig. 9A, D-G), show the higher cutoff (0.7)
and could be considerable biomarker for early diagnosis of lung
cancer. ROC analysis measured an AUC 0.661, 0.637 for miR-3917 and
miR-27a-5p (P-value <0.01, 95% confidence interval: 0.578–0.744
and 0.549–0.725, respectively, Table
IV and Fig. 9B and C), show the
score very close to the cutoff (0.7) needed for a considerable
biomarker for early diagnosis of lung cancer. Furthermore, as total
7 miRNAs ROC curve conjoint analysis result, AUC=0.919 (P-value
<0.01, 95% confidence interval: 0.874–0.963, Table IV and Fig. 9H), which is the score higher the
cutoff (0.7) and could be considerable biomarkers for early
diagnosis of lung cancer.
 | Table IV.ROC curve analysis of signature
miRNAs. |
Table IV.
ROC curve analysis of signature
miRNAs.
| miRNAs | Area | SE | P-value | 95% CI |
|---|
| miR-205-5p | 0.728 | 0.040 | <0.001 | 0.650–0.806 |
| miR-3917 | 0.661 | 0.042 | <0.001 | 0.578–0.744 |
| miR-27a-5p | 0.637 | 0.045 |
0.003 | 0.549–0.725 |
| miR-30a-3p | 0.758 | 0.038 | <0.001 | 0.683–0.832 |
| miR-30a-5p | 0.772 | 0.037 | <0.001 | 0.699–0.845 |
| miR-30c-2-3p | 0.734 | 0.042 | <0.001 | 0.650–0.817 |
| miR-30d-5p | 0.776 | 0.037 | <0.001 | 0.704–0.848 |
| Total 7 miRNAs | 0.919 | 0.023 | <0.001 | 0.874–0.963 |
Discussion
A growing body of evidence indicates that miRNAs are
valuable source of markers for diagnosis in cancer (7,12,20–23).
Chen et al (24) identified
10-serum miRNAs as potential markers for early diagnosis of lung
cancer in 400 lung cancer cases and 220 controls. Yin et al
found that increased expression of miR-150 is associated with poor
prognosis in lung cancer by qRT-PCR (25). The above studies provide sound
evidence that the alteration in the miRNA profiles could be
reliable biomarkers for the early diagnosis and prognosis
prediction of cancer.
There already exist plenty of published data on
miRNA profiles in lung cancer. In a previous study, Wu et al
(26) revealed that 38 miRNAs were
significantly dysregulated in five pairs of lung cancer and
paracancerous tissues using miRNA microarray techniques and miR-19b
and miR-146a are potential biomarkers for the prediction of
survival and response to chemotherapy in lung cancer using qRT-PCR
in 94 healthy subjects and 94 advanced lung cancer patients.
Molina-Pinelo et al (27)
analyzed the expression of 381 miRNAs in bronchoalveolar lavage
fluid samples from 87 patients classified into four groups: COPD,
adenocarcinoma, adenocarcinoma with COPD, and control (neither COPD
nor adenocarcinoma), and found that four miRNA clusters (the
miR-17-92 cluster and its paralogues, miR-106a-363 and miR-106b-25,
miR-192-194 and miR-132-212 cluster) were involved in the
pathogenesis of adenocarcinoma and COPD, as biomarkers for
diagnosis and/or therapeutic purposes. These reports indicate that
specific miRNA profiles of lung cancer may bring breakthroughs to
the diagnosis and treatment of lung cancer. However, the problem is
that the results of these reports are not consistent because of the
small number of samples. Studies with large scale samples and
microarray detection are rare, and the relationship between
specific miRNAs and lung cancer still need further investigation.
TCGA database is a public platform, in which large scale sequencing
data of miRNA and mRNA for lung cancer can be downloaded. To
improve the reliability and accuracy of the studies, we integrated
the data from our miRNA microarray and TCGA database.
In the present study, we focused primary on whether
promising miRNAs could act as biomarkers to discriminate lung
cancer from normal cases by taking advantage of miRNA microarrays
in lung tumor tissues and adjacent non-tumor tissues. A total of
116 miRNAs and 502 mRNAs were found dysregulated in tumor tissues.
After comprehensive analysis between TCGA and microarray data, 32
and 377 differentially expressed miRNAs and mRNAs, which closely
associated with lung cancer, were obtained. Furthermore, after
miRNA-mRNA network construction, we identified 28 (miR-133a-3p,
miR-133b, miR-184, miR-30a-5p, miR-30b-3p, miR-30c-2-3p,
miR-30d-5p, miR-338-5p, miR-363-3p, miR-451a, miR-141-3p,
miR-148a-3p, miR-193b-3p, miR-19a-3p, miR-200a-3p, miR-200a-5p,
miR-205-5p, miR-210-3p, miR-21-5p, miR-221-5p, miR-224-5p,
miR-301a-3p, miR-31-5p, miR-34b-3p, miR-409-5p, miR-411-5p,
miR-708-5p, miR-7-1-3p) specific miRNAs, as a combination of
miRNAs, has more accurate predicted value in distinguishing cancer
cases from control. Pathway analysis results was indicative of
dysregulated miRNAs related to Environmental Information Processing
and Human Diseases, involved in non-small cell lung cancer. Studies
have reported that these miRNAs are involved in the regulation of
the development of lung cancer. For example, overexpression of
miR-30a in lung adenocarcinoma A549 cells inhibited cell migration
and invasion, which was partially through decrease of EYA2
expression (28). Chen et al
(29) reported that miR-30d-5p was
downregulated in lung cancer tissues, which significantly inhibited
growth, cell cycle distribution, and motility of lung cancer cells.
miR-30d-5p inhibited tumor cell proliferation and motility by
directly targeting CCNE2 in NSCLC. miR-21 were reported to target
MMP9 (30) responsible for cell
proliferation and migration. Overall, these finding suggested that
alterations of these miRNAs represent meaningful risk factors in
lung cancer.
To validate the miRNA expression results from
microarray and further confirm the relationship between
differentially expressed miRNAs and lung cancer, we selected 7
miRNAs (miR-205-5p, miR-3917, miR-27a-5p, miR-30a-3p, miR-30a-5p,
miR-30c-2-3p and miR-30d-5p) which were found in non-small cell
lung cancer pathway and analyzed their actual expression levels in
81 pairs of diagnosed lung tumor and adjacent non-tumor tissues by
qRT-PCR. The results indicated that miR-205-5p and miR-3917 were
upregulated and miR-27a-5p, miR-30a-3p, miR-30a-5p, miR-30c-2-3p,
miR-30d-5p were downregulated, which was highly consistent with the
results of microarrays and TCGA analysis. miR-205-5p and miR-3917
has previously been described as upregulated in many types of
cancer and tissues, including lung cancer, nasopharyngeal carcinoma
and gingival tissues (31–33). miR-30a-3p and miR-30c-2-3p has been
reported as downregulated in hepatocellular carcinoma and clear
cell renal cell carcinomas (34,35).
Then, we used conditional logistic regression analysis to further
find the association between these signature miRNAs and lung
cancer. The results showed that the increased risk of lung cancer
was significantly associated with increased expression of
miR-205-5p and miR-3917 and reduced expression of miR-27a-5p,
miR-30a-3p, miR-30a-5p, miR-30c-2-3p, and miR-30d-5p, respectively.
ROC curve analysis demonstrated that miR-205-5p, miR-30a-3p,
miR-30a-5p, miR-30c-2-3p and miR-30d-5p could be considerable
biomarkers for early diagnosis of lung cancer, also ROC analysis of
all 7 miRNAs have more effective diagnosis for lung cancer. The
results suggested that miR-205, miR-3917, miR-27a-5p, miR-30a-3p,
miR-30a-5p, miR-30c-2-3p, and miR-30d-5 may play an important role
during lung cancer development and may be effective diagnostic
biomarkers for lung cancer. Besides miR-27a, the function of the
other six miRNAs in cancer has rarely been reported. It is reported
that miR-30a-5p promoted an epithelial phenotype and suppressed
invasion by specifically targeting β3 integrin subunit to
subsequently interdict the β3 integrin/Erk/Ets-1 network in breast
cancer cells (36). miR-30a-5p
might function as a tumor suppressing miRNA by directly targeting
MTDH in hepatocellular carcinoma (37). Based on the above, the roles of
these signature miRNAs in lung cancer still need further
investigation in the future.
In conclusion, 28 miRNAs associated with lung cancer
were identified by microarray, TCGA and miRNA-mRNA network
analysis. After population analysis, miRNAs in miR-205, miR-3917,
miR-27a-5p, miR-30a-3p, miR-30a-5p, miR-30c-2-3p, and miR-30d-5
were found to impact the risk of lung cancer and could be
considerable biomarkers for early diagnosis of lung cancer. The
results suggested a set of signature miRNAs may play a more
important role during lung cancer development and promise
biomarkers for the early screening of high-risk population.
Integrating analysis including gene expression profile, TCGA
database and population validation are effective methods to screen
cancer related miRNAs. Future studies will investigate the roles of
these miRNAs during the development of lung cancer.
Acknowledgements
The present study was financially supported by the
National Natural Science Foundation of China (81472939, 81673132,
and 81182618), the Liu Da Ren Cai Gao Feng Project of Jiangsu
Province (no. 2013-WSW-053) and the Fundamental Research Funds for
the Central Universities.
References
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chen W, Zheng R, Baade PD, Zhang S, Zeng
H, Bray F, Jemal A, Yu XQ and He J: Cancer statistics in China,
2015. CA Cancer J Clin. 66:115–132. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Vrijens K, Bollati V and Nawrot TS:
MicroRNAs as potential signatures of environmental exposure or
effect: A systematic review. Environ Health Perspect. 123:399–411.
2015.PubMed/NCBI
|
|
4
|
Zhang H, Li W, Nan F, Ren F, Wang H, Xu Y
and Zhang F: MicroRNA expression profile of colon cancer stem-like
cells in HT29 adenocarcinoma cell line. Biochem Biophys Res Commun.
404:273–278. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Xia S, Huang CC, Le M, Dittmar R, Du M,
Yuan T, Guo Y, Wang Y, Wang X, Tsai S, et al: Genomic variations in
plasma cell free DNA differentiate early stage lung cancers from
normal controls. Lung Cancer. 90:78–84. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Pérez-Ramírez C, Cañadas-Garre M,
Jiménez-Varo E, Faus-Dáder MJ and Calleja-Hernández MA: MET: A new
promising biomarker in non-small-cell lung carcinoma.
Pharmacogenomics. 16:631–647. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Fan L, Qi H, Teng J, Su B, Chen H, Wang C
and Xia Q: Identification of serum miRNAs by nano-quantum dots
microarray as diagnostic biomarkers for early detection of
non-small cell lung cancer. Tumour Biol. 37:7777–7784. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Geng Q, Fan T, Zhang B, Wang W, Xu Y and
Hu H: Five microRNAs in plasma as novel biomarkers for screening of
early-stage non-small cell lung cancer. Respir Res. 15:1492014.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Inamura K and Ishikawa Y: MicroRNA in lung
cancer: Novel biomarkers and potential tools for treatment. J Clin
Med. 5:52016. View Article : Google Scholar :
|
|
10
|
Kim JO, Gazala S, Razzak R, Guo L, Ghosh
S, Roa WH and Bédard EL: Non-small cell lung cancer detection using
microRNA expression profiling of bronchoalveolar lavage fluid and
sputum. Anticancer Res. 35:1873–1880. 2015.PubMed/NCBI
|
|
11
|
Wang Y, Zhang X, Liu L, Li H, Yu J, Wang C
and Ren X: Clinical implication of microRNA for lung cancer. Cancer
Biother Radiopharm. 28:261–267. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Solomides CC, Evans BJ, Navenot JM,
Vadigepalli R, Peiper SC and Wang ZX: MicroRNA profiling in lung
cancer reveals new molecular markers for diagnosis. Acta Cytol.
56:645–654. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Singh DK, Bose S and Kumar S: Role of
microRNA in regulating cell signaling pathways, cell cycle, and
apoptosis in non-small cell lung cancer. Curr Mol Med. 16:474–486.
2016. View Article : Google Scholar
|
|
14
|
Razzak R, Bédard EL, Kim JO, Gazala S, Guo
L, Ghosh S, Joy A, Nijjar T, Wong E and Roa WH: MicroRNA expression
profiling of sputum for the detection of early and locally advanced
non-small-cell lung cancer: A prospective case-control study. Curr
Oncol. 23:e86–e94. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Pu Q, Huang Y, Lu Y, Peng Y, Zhang J, Feng
G, Wang C, Liu L and Dai Y: Tissue-specific and plasma microRNA
profiles could be promising biomarkers of histological
classification and TNM stage in non-small cell lung cancer. Thorac
Cancer. 7:348–354. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Krutakova M, Sarlinova M, Matakova T,
Dzian A, Hamzik J, Pec M, Javorkova S and Halasova E: The role of
dysregulated microRNA expression in lung cancer. Adv Exp Med Biol.
911:1–8. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Li JH, Liu S, Zhou H, Qu LH and Yang JH:
starBase v2.0: Decoding miRNA-ceRNA, miRNA-ncRNA and protein-RNA
interaction networks from large-scale CLIP-Seq data. Nucleic Acids
Res. 42:D92–D97. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hsu SD, Tseng YT, Shrestha S, Lin YL,
Khaleel A, Chou CH, Chu CF, Huang HY, Lin CM, Ho SY, et al:
miRTarBase update 2014: An information resource for experimentally
validated miRNA-target interactions. Nucleic Acids Res. 42:D78–D85.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li CY, Liang GY, Yao WZ, Sui J, Shen X,
Zhang YQ, Peng H, Hong WW, Ye YC, Zhang ZY, et al: Integrated
analysis of long non-coding RNA competing interactions reveals the
potential role in progression of human gastric cancer. Int J Oncol.
48:1965–1976. 2016.PubMed/NCBI
|
|
20
|
Yang YL, Xu LP, Zhuo FL and Wang TY:
Prognostic value of microRNA-10b overexpression in peripheral blood
mononuclear cells of nonsmall-cell lung cancer patients. Tumour
Biol. 36:7069–7075. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Xu C, Zheng Y, Lian D, Ye S, Yang J and
Zeng Z: Analysis of microRNA expression profile identifies novel
biomarkers for non-small cell lung cancer. Tumori. 101:104–110.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wiwanitkit V: MicroRNA assays for
diagnosis lung cancer biopsy. J Thorac Oncol. 10:e52–53. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wang P, Yang D, Zhang H, Wei X, Ma T,
Cheng Z, Hong Q, Hu J, Zhuo H, Song Y, et al: Early detection of
lung cancer in serum by a panel of microRNA biomarkers. Clin Lung
Cancer. 16:313–319 e1. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chen X, Hu Z, Wang W, Ba Y, Ma L, Zhang C,
Wang C, Ren Z, Zhao Y, Wu S, et al: Identification of ten serum
microRNAs from a genome-wide serum microRNA expression profile as
novel noninvasive biomarkers for nonsmall cell lung cancer
diagnosis. Int J Cancer. 130:1620–1628. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yin QW, Sun XF, Yang GT, Li XB, Wu MS and
Zhao J: Increased expression of microRNA-150 is associated with
poor prognosis in non-small cell lung cancer. Int J Clin Exp
Pathol. 8:842–846. 2015.PubMed/NCBI
|
|
26
|
Wu C, Cao Y, He Z, He J, Hu C, Duan H and
Jiang J: Serum levels of miR-19b and miR-146a as prognostic
biomarkers for non-small cell lung cancer. Tohoku J Exp Med.
232:85–95. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Molina-Pinelo S, Pastor MD, Suarez R,
Romero-Romero B, González De la Peña M, Salinas A, García-Carbonero
R, De Miguel MJ, Rodríguez-Panadero F, Carnero A, et al: MicroRNA
clusters: Dysregulation in lung adenocarcinoma and COPD. Eur Respir
J. 43:1740–1749. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yuan Y, Zheng S, Li Q, Xiang X, Gao T, Ran
P, Sun L, Huang Q, Xie F, Du J, et al: Overexpression of miR-30a in
lung adenocarcinoma A549 cell line inhibits migration and invasion
via targeting EYA2. Acta Biochim Biophys Sin (Shanghai).
48:220–228. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Chen D, Guo W, Qiu Z, Wang Q, Li Y, Liang
L, Liu L, Huang S, Zhao Y and He X: MicroRNA-30d-5p inhibits tumour
cell proliferation and motility by directly targeting CCNE2 in
non-small cell lung cancer. Cancer Lett. 362:208–217. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hu J, Ni S, Cao Y, Zhang T, Wu T, Yin X,
Lang Y and Lu H: The angiogenic effect of microRNA-21 targeting
TIMP3 through the regulation of MMP2 and MMP9. PLoS One.
11:e01495372016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Jiang M, Zhang P, Hu G, Xiao Z, Xu F,
Zhong T, Huang F, Kuang H and Zhang W: Relative expressions of
miR-205-5p, miR-205-3p, and miR-21 in tissues and serum of
non-small cell lung cancer patients. Mol Cell Biochem. 383:67–75.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wang F, Lu J, Peng X, Wang J, Liu X, Chen
X, Jiang Y, Li X and Zhang B: Integrated analysis of microRNA
regulatory network in nasopharyngeal carcinoma with deep
sequencing. J Exp Clin Cancer Res. 35:172016.doi:
10.1186/s13046-016-0292-4. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Stoecklin-Wasmer C, Guarnieri P, Celenti
R, Demmer RT, Kebschull M and Papapanou PN: MicroRNAs and their
target genes in gingival tissues. J Dent Res. 91:934–940. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Wang W, Lin H, Zhou L, Zhu Q, Gao S, Xie
H, Liu Z, Xu Z, Wei J, Huang X, et al: MicroRNA-30a-3p inhibits
tumor proliferation, invasiveness and metastasis and is
downregulated in hepatocellular carcinoma. Eur J Surg Oncol.
40:1586–1594. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Moch H and Lukamowicz-Rajska M:
miR-30c-2-3p and miR-30a-3p: New pieces of the jigsaw puzzle in
HIF2α regulation. Cancer Discov. 4:22–24. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Li W, Liu C, Zhao C, Zhai L and Lv S:
Downregulation of β3 integrin by miR-30a-5p modulates cell adhesion
and invasion by interrupting Erk/Ets-1 network in triple-negative
breast cancer. Int J Oncol. 48:1155–1164. 2016.PubMed/NCBI
|
|
37
|
Li WF, Dai H, Ou Q, Zuo GQ and Liu CA:
Overexpression of microRNA-30a-5p inhibits liver cancer cell
proliferation and induces apoptosis by targeting MTDH/PTEN/AKT
pathway. Tumour Biol. 37:5885–5895. 2016. View Article : Google Scholar : PubMed/NCBI
|