Introduction
Lung cancer is one of the most common malignant
tumors worldwide, with a 5-year survival rate of 14% (1). Thus far, the statistics for cancer
occurrence and outcome show that lung cancer remains a primary
cause of mortality from cancer (2,3). Since
the incidence and mortality of lung cancer increases significantly
every year, it represents a major economic burden to society.
Currently, the screening and early diagnosis of lung cancer in
clinics relies mainly on magnetic resonance imaging (MRI) and
computed tomography (CT) imaging, whereas the final diagnosis is
established on the basis of histopathological examination results.
The early clinical manifestations of lung cancer patients are
relatively mild and not typical, easily overlooked or confused with
benign inflammatory disease, such as lung infection. Therefore,
early identification and diagnosis of lung cancer is significant
since lung cancer may be curable in its early stages (1).
Thus far, increasing attention has focused on
searching for improved biomarkers that function not only to detect
lung cancer at an early stage but also to explore the molecular
mechanisms that underlie cancer development. Tumor markers,
including carcinoembryonic antigen (CEA), carbohydrate antigen 125
(CA125), neuron-specific enolase (NSE), squamous cell carcinoma
antigen (SCCAg) and cytokeratin-19 fragments (Cyfra21-1), are
clinically applied for the early detection of lung cancer (4,5).
However, the sensitivity and specificity of these biomarkers are
not adequate (6–8). Combined detection using a panel of
biomarkers can improve the sensitivity and accuracy in lung cancer
diagnosis, but consequently results in a lower specificity and
increased financial burden to patients. Determining the diagnosis
and establishing a more appropriate treatment in early stage
patients with lung cancer remains a challenge. Additional
biomarkers to benefit early diagnosis of lung cancer are required.
Since malignant pleural effusion is a common complication of lung
cancer patients, biochemical and pathogenic microorganisms analysis
in pleural fluid contribute to differential diagnosis between lung
cancer and benign inflammatory diseases.
Currently, several proteomic approaches have been
used for the identification of cancer biomarkers, including
two-dimensional electrophoresis (2-DE) and mass spectrometry (MS).
Monitoring the protein expression pattern by proteomic technologies
contributes to the early detection of potentially novel cancer
biomarkers. Proteomics has proved to be a powerful tool in clinical
diagnosis and biomarker discovery, particularly in the
identification of specific post-translational modifications
(9). Previously, those proteomic
approaches have been applied to screen biomarkers for early
diagnosis of lung cancer (10–13).
The transthyretin (TTR) monomer has been shown to be upregulated in
the sera of adenocarcinoma lung cancer patients using 2-DE coupled
to matrix-assisted laser desorption/ionization time-of-flight MS
(MALDI-TOF-MS) peptide mass fingerprinting (14). Previous studies have shown that TTR
may be a novel serum biomarker for distinguishing lung cancer
patients from normal control individuals using surface-enhanced
LDI-TOF-MS (15,16).
TTR is a normal serum protein synthesized primarily
in the liver, the choroid plexus and the retina (17). As a homotetramer in plasma, TTR
binds and transports the thyroid hormones and the retinol-binding
protein-retinal complex (18). The
decreased serum concentration of TTR has been used as a marker to
evaluate malnutritional/inflammatory status under a variety of
conditions (19–21). The implication of TTR in the
formation of amyloid deposits in familial amyloidosis and senile
systemic amyloidosis has been shown previously (22,23).
In addition, a number of studies have shown the potential value of
serum TTR in cancer diagnosis, including ovarian (21,24),
endometrial (25) and lung cancer
(14–16).
The aim of the present study was to investigate the
changes of the biochemical and protein composition between lung
cancer and lung infection patients in order to screen biomarkers
for the differential diagnosis of malignant pleural effusions. An
MS-based proteomic approach, MALDI-TOF-MS, was applied to
characterize TTR variants in serum and pleural effusion of patients
with lung cancer and lung infection.
Patients and methods
Patients and samples
The serum and pleural effusion samples were obtained
from Tianjin Chest Hospital including 92 patients with lung cancer
and 38 patients with lung infection (bacterial pneumonia). The
serum and pleural effusion samples were collected prior to any
clinical treatment. Control serum samples were also obtained from
42 healthy adult volunteers. The diagnosis of lung cancer and lung
infection was based on the clinical outcome, MRI/CT imaging and
laboratory findings. Stage and histological classification were
performed according to the World Health Organization 1999 criteria
for lung cancer classification (26). The detailed clinical characteristics
of the participants are shown in Table
I.
 | Table IClinical characteristics of the
subjects in each group. |
Table I
Clinical characteristics of the
subjects in each group.
| Characteristics | Lung cancer
(n=92) | Lung infection
(n=38) | Healthy controls
(n=42) |
|---|
| Gender, n |
| Male | 60 | 23 | 24 |
| Female | 32 | 15 | 18 |
| Mean age, years
(range) | 65.8 (48–83) | 54.9 (18–78) | 58.6 (45–65) |
| Lung cancer
histology, n |
| Adenocarcinoma | 45 | | |
| Squamous cell lung
cancer | 30 | | |
| Small cell lung
cancer | 17 | | |
| Disease stages,
n |
| I | 10 | | |
| II | 36 | | |
| III | 28 | | |
| IV | 18 | | |
The Medical Ethics and Human Clinical Trial
Committee of Tianjin Medical University (Tianjin, China) approved
the study and informed consent was obtained from all the study
subjects.
Sample processing
The serum samples were collected and maintained at
4°C for 1 h for clotting, subsequently centrifuged at 1,700 × g for
15 min and immediately aliquoted and stored at −80°C. All the serum
samples were only allowed to thaw once. The pleural effusion
samples were collected from the patients with lung cancer-induced
malignant pleural effusion and pleural effusion induced by lung
infection. The effusions were collected in sterile tubes and
centrifuged immediately at 4°C. The cell-free supernatants were
collected and the aliquots were stored at −80°C until use.
Clinical and laboratory measurements
Simultaneous to the collection of the serum and
pleural effusion samples, the following clinical and laboratory
data were obtained: Age, gender, triglycerides (TG), cholesterol
(CHO), apolipoprotein A (ApoA), ApoB, glucose (GLU), TTR, total
protein (TP), albumin (ALB), adenosine deaminase (ADA) and lactate
dehydrogenase (LDH). Regarding the laboratory features, TG, CHO,
GLU, TP, ALB, ADA and LDH were measured using a Toshiba TBA-120
auto-analyzer (Toshiba Medical Systems Co., Ltd., Tokyo, Japan).
The concentration of TTR, ApoA and ApoB were measured by the
immunonephelometric method using an automatic clinical analyzer
(TBA-40, Toshiba Medical Systems Co., Ltd.).
MALDI-TOF-MS analysis of TTR
All the experiments were performed with a
MALDI-TOF-MS (Shimadzu/Kratos, Manchester, UK) operated at a
wavelength of 337 nm. The optimal spectra of TTR were obtained at
an ion-accelerating voltage of 27.5 kV and a reflectron voltage of
30 kV. The spectra were calculated by using external calibration
with [M+H] ions produced from horse cytochrome c (12,361.96
m/z) and horse myoglobin (16,952.27 m/z). The matrix was a
saturated solution of sinapinic acid in acetonitrile plus water
(1:2, v/v) containing 0.1% trifluoroacetic acid. The samples were
deposited onto the sample probe assembly. MALDI-TOF-MS data were
analyzed using Launchpad software version 2.4 (Kratos Analytical,
Manchester, UK) (27).
Statistical analysis
Data were expressed as mean ± standard deviation.
Data were processed with SPSS software 13.0. (SPSS, Inc., Chicago,
IL, USA). Statistical analysis was performed using the independent
samples t-test between the groups. P<0.05 was considered to
indicate a statistically significant difference.
Results
Concentrations of the biochemical
indicators in lung cancer and lung infection patients
The biochemical indicators in the serum and pleural
effusion of the two groups of patients were detected. The results
showed that the serum levels of CHO, ApoA and TTR in lung cancer
patients were higher than that of the lung infection patients
(P<0.05). The levels of TTR were higher, whereas the activity of
ADA was lower (P<0.05) in the pleural effusion of lung cancer
patients compared to lung infection patients (Table II).
 | Table IIConcentrations of the biochemical
indicators in the serum and pleural effusion. |
Table II
Concentrations of the biochemical
indicators in the serum and pleural effusion.
| Serum | Pleural effusion |
|---|
|
|
|
|---|
| Indicators | Lung cancer
(n=92) | Lung infection
(n=38) | Lung cancer
(n=92) | Lung infection
(n=38) |
|---|
| TG, mmol/l | 1.21±0.55 | 1.02±0.33 | 0.33±0.35 | 0.26±0.13 |
| CHO, mmol/l | 4.27±0.90a | 3.67±0.86 | 1.46±0.61 | 1.47±0.72 |
| ApoA, g/l | 1.05±0.17a | 0.94±0.18 | 0.43±0.12 | 0.47±0.28 |
| ApoB, g/l | 1.03±0.17 | 0.96±0.14 | 0.53±0.11 | 0.53±0.14 |
| GLU, mmol/l | 5.62±1.36 | 5.91±2.25 | 4.82±2.29 | 4.24±2.17 |
| TTR, mg/l | 180.12±50.16a | 150.08±50.18 | 100.45±40.22a | 68.36±35.29 |
| TP, g/l | 60.64±7.18 | 62.31±7.10 | 35.91±7.26 | 35.36±11.53 |
| ALB, g/l | 37.58±3.55 | 36.46±4.72 | 23.16±5.36 | 21.44±7.95 |
| ADA, U/l | 8.21±10.59 | 7.90±2.35 | 5.85±3.16a | 15.66±8.89 |
| LDH, U/l | 224.72±196 | 190.73±63.60 | 319.87±346.25 | 212.60±131.76 |
Pleural effusion/serum ratios of
biochemical indicators in lung cancer and lung infection
patients
To further compare the changes of the biochemical
indicators between the two groups of patients, the pleural
effusion/serum ratios were calculated and analyzed. The results
showed that the pleural effusion/serum TTR ratios were higher
(P<0.05) in patients with lung cancer compared to lung infection
patients, whereas the ratios of ADA were lower (P<0.05) in lung
cancer patients. There were no significant differences with regards
to the other biochemical indicators (Table III).
 | Table IIIPleural effusion to serum
concentration ratios of the biochemical indicators. |
Table III
Pleural effusion to serum
concentration ratios of the biochemical indicators.
| Indicators | Lung cancer
(n=92) | Lung infection
(n=38) |
|---|
| TG, mmol/l | 0.29±0.25 | 0.28±0.14 |
| CHO, mmol/l | 0.35±0.17 | 0.42±0.23 |
| ApoA, g/l | 0.41±0.12 | 0.53±0.32 |
| ApoB, g/l | 0.54±0.18 | 0.55±0.14 |
| GLU, mmol/l | 0.83±0.35 | 0.74±0.27 |
| TTR, mg/l | 0.61±0.19a | 0.48±0.23 |
| TP, g/l | 0.59±0.12 | 0.56±0.17 |
| ALB, g/l | 0.65±0.14 | 0.57±0.19 |
| ADA, U/l | 0.96±0.49a | 1.99±1.08 |
| LDH, U/l | 1.56±1.84 | 1.30±0.81 |
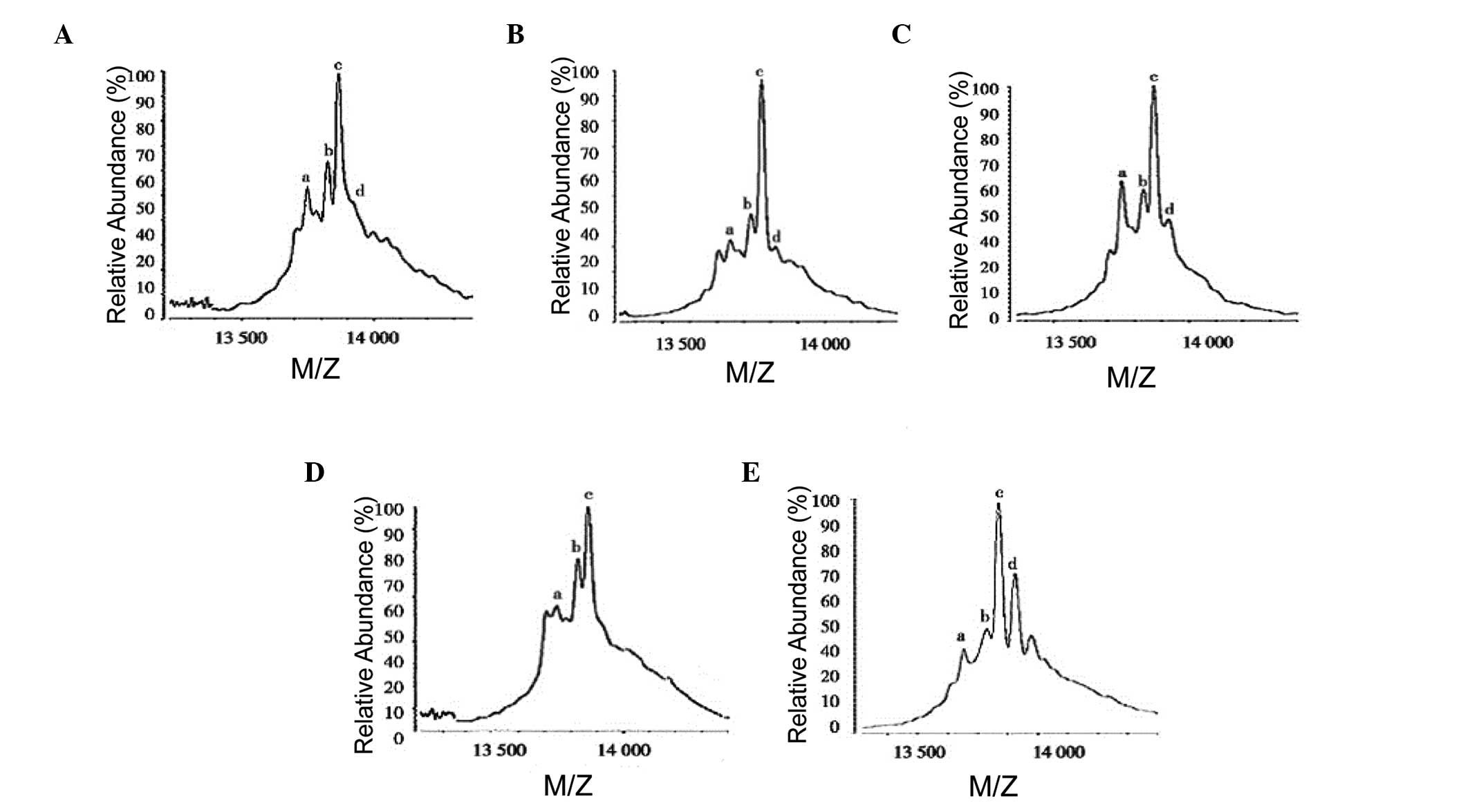
Four major TTR peaks were detected by
MALDI-TOF-MS
The modified TTR isoforms were detected by
MALDI-TOF-MS. The proportion of TTR isoforms in serum and pleural
effusion was further analyzed. As shown in Fig. 1, four major peaks, which were native
TTR (13,749.86±1.48 m/z), Sul-TTR (13,829.63±2.76 m/z), Cys-TTR
(13,870.70±2.70 m/z) and Cysgly-TTR (13,927±5.77 m/z), were
observed in the mass spectrum of the serum samples from the
patients with lung cancer, lung infection and the healthy
volunteers. In addition, the proportion of modified TTR isoforms
showed no significant differences among the three groups. The
proportion of Cysgly-TTR in the pleural effusion of the patients
with lung cancer significantly increased compared to the lung
infection patients. The results indicated that the proportion of
Cysgly-TTR varied in the pleural effusion between the lung cancer
and lung infection patients.
Discussion
Satisfactory biomarkers for screening and early
diagnosis of lung cancer remain limited and require further
investigation. The identification of novel biomarkers with
potential diagnostic value is essential for the development of
novel therapeutic strategies in lung cancer. During previous years,
a few candidate cancer biomarkers, including CEA, CA125, NSE, SCCAg
and Cyfra21-1, have been widely used for the early detection of
lung cancer. However, the sensitivity and specificity of these
biomarkers are not adequate in establishing pathological diagnosis.
Therefore, identification of novel biomarkers that are specific for
lung cancer appears to be an important challenge for clinical
pathologists.
In the present study, ten biochemical indicators in
the serum and pleural effusion from patients with lung cancer and
lung infection were detected. The results showed that higher levels
of CHO, ApoA and TTR were found in the serum of patients with lung
cancer compared to lung infection patients. The levels of CHO and
ApoA are closely associated with dietary factors and are
nonspecific for the differential diagnosis of lung disease. The
changes in pleural effusion may contain information that directly
reflects the pathological status for pulmonary diseases. Therefore,
the levels of biochemical indicators were measured in the pleural
effusion. Higher levels of TTR were found in the pleural effusion
of lung cancer patients compared to lung infection patients,
whereas the activity of ADA was lower in lung cancer patients. The
pleural effusion/serum ratios of the biochemical indicators were
further analyzed, and the results showed higher pleural
effusion/serum TTR ratios in the lung cancer patients compared to
the lung infection patients, whereas the ratios of ADA in lung
cancer patients were lower. The results indicated that TTR and ADA
may directly reflect the pathological state of pulmonary
diseases.
TTR is a homotetrameric protein composed of four
127-amino acid residues subunits synthesized mainly in the liver.
The normal concentration of TTR in the blood ranges 20–40 mg/dl. As
a well-known negative acute-phase protein, a decreased serum
concentration of TTR has been reported in cases of severe liver
disease, malnutrition and acute inflammation. In addition, TTR was
found to decrease in the sera of patients with ovarian and
endometrial cancers (24,25), and the mechanisms it is involved in
remain unknown. Elevated levels of TTR were detected in the aqueous
humor of patients with primary open-angle glaucoma and were
considered to play a role in the onset of glaucoma (28). Previously, certain studies have
shown the potential value of serum TTR in lung cancer diagnosis
(14–16).
The results of the present study showed that the TTR
levels in the serum and pleural effusion of patients with lung
cancer were significantly higher compared to the TTR in lung
infection patients. The results were consistent with the study by
Liu et al (15), which
showed decreased levels of TTR in the sera of lung cancer and
benign lung disease patients compared to normal sera, and in
addition, the decreased level of TTR in benign lung diseases was
more evident compared to the patients with lung cancer. Recently,
Wang et al (29) found a
relatively increased level of TTR in the effusions and sera of lung
cancer patients compared to the benign inflammatory disease
samples. Collectively, the accumulated data indicated a potential
value of TTR in lung cancer diagnosis. There are several potential
mechanisms for the relatively higher level of TTR in lung cancer
patients compared to benign inflammatory disease, such as lung
infection. First, TTR is synthesized mainly in the liver and the
dysfunction of the liver may contribute to the reduced synthesis of
TTR. Second, as a negative acute-phase protein, TTR is
downregulated during inflammation, which may be the reason for the
relatively lower level of TTR in benign inflammatory disease.
Third, overexpression of TTR was detected in lung cancer tissue
cells and may be secreted into serum and pleural effusion to
supplement the decreasing TTR in the sera and pleural effusion of
lung cancer patients (15).
Currently, pathologists have sought to utilize
proteomic technologies, such as MS, for the identification of
useful biomarkers and therapeutic targets in lung cancer. MS could
represent a powerful and sensitive tool for screening protein
profiling, as well as providing high-dimensional information
regarding proteins (including post-translational proteins). These
MS-based proteomics technologies offer novel approaches in
identifying the potential biomarkers for lung cancer diagnosis and
clinical management of this disease. The detection of TTR isoforms
by MS would aid in the analysis of lung cancer pathogenesis and the
investigation of biomarker panels for clinical practice.
In order to identify TTR isoforms and further
explore their role in lung cancer diagnosis, MALDI-TOF-MS was used
in the present study to identify the relative abundance, types and
proportion of TTR modification in serum and pleural effusion of
patients with lung cancer and lung infection. The present results
showed that four major peaks were observed in the mass spectrum of
serum samples from patients with lung cancer, lung infection and
healthy volunteers, including native TTR, Sul-TTR, Cys-TTR and
Cysgly-TTR. In addition, the proportion of modified TTR isoforms
showed no significant differences among the three groups. Notably,
the proportion of Cysgly-TTR in the pleural effusion of patients
with lung cancer significantly increased compared to the lung
infection patients. The pleural effusion samples obtained from
local lesions reflect the pathological state more accurately.
Therefore, an increased proportion of Cysgly-TTR in the pleural
effusion of patients with lung cancer may have a potential
diagnostic value. The role for TTR post-translational modification
involved in the pathogenesis of lung cancer requires further
investigation.
In conclusion, higher pleural effusion/serum TTR
ratios were demonstrated in lung cancer patients compared to lung
infection patients. Furthermore, four modified TTRs were identified
in lung cancer by MALDI-TOF-MS and the proportion of Cysgly-TTR was
significantly increased in the pleural effusion of patients with
lung cancer. The results indicated that a combination of pleural
effusion/serum TTR ratios and modified TTR may contribute to the
differential diagnosis between lung cancer and lung infection.
Acknowledgements
The authors would like to thank all the subjects for
their participation in the present study. The study was supported
by the National Natural Science Foundation of China (grant no.
30973157).
References
|
1
|
Spira A and Ettinger DS: Multidisciplinary
management of lung cancer. N Engl J Med. 350:379–392. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ferlay J, Steliarova-Foucher E,
Lortet-Tieulent J, et al: Cancer incidence and mortality patterns
in Europe: estimates for 40 countries in 2012. Eur J Cancer.
49:1374–1403. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ferlay J, Parkin DM and Steliarova-Foucher
E: Estimates of cancer incidence and mortality in Europe in 2008.
Eur J Cancer. 46:765–781. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Schneider J: Tumor markers in detection of
lung cancer. Adv Clin Chem. 42:1–41. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Harmsma M, Schutte B and Ramaekers FC:
Serum markers in small cell lung cancer: opportunities for
improvement. Biochim Biophys Acta. 1836.255–272. 2013.
|
|
6
|
Chu XY, Hou XB, Song WA, Xue ZQ, Wang B
and Zhang LB: Diagnostic values of SCC, CEA, Cyfra21-1 and NSE for
lung cancer in patients with suspicious pulmonary masses: a single
center analysis. Cancer Biol Ther. 11:995–1000. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kley K and Oehr P: Usefulness of combined
FDG-PET with CT or tumour markers in lung cancer diagnosis.
Anticancer Res. 30:1741–1745. 2010.PubMed/NCBI
|
|
8
|
Grunnet M and Sorensen JB:
Carcinoembryonic antigen (CEA) as tumor marker in lung cancer. Lung
Cancer. 76:138–143. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Witze ES, Old WM, Resing KA and Ahn NG:
Mapping protein post-translational modifications with mass
spectrometry. Nat Methods. 4:798–806. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhukov TA, Johanson RA, Cantor AB, Clark
RA and Tockman MS: Discovery of distinct protein profiles specific
for lung tumors and pre-malignant lung lesions by SELDI mass
spectrometry. Lung Cancer. 40:267–279. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Howard BA, Wang MZ, Campa MJ, Corro C,
Fitzgerald MC and Patz EF Jr: Identification and validation of a
potential lung cancer serum biomarker detected by matrix-assisted
laser desorption/ionization-time of flight spectra analysis.
Proteomics. 3:1720–1724. 2003. View Article : Google Scholar
|
|
12
|
Yang SY, Xiao XY, Zhang WG, et al:
Application of serum SELDI proteomic patterns in diagnosis of lung
cancer. BMC Cancer. 5:832005. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Indovina P, Marcelli E, Pentimalli F,
Tanganelli P, Tarro G and Giordano A: Mass spectrometry-based
proteomics: the road to lung cancer biomarker discovery. Mass
Spectrom Rev. 32:129–142. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Maciel CM, Junqueira M, Paschoal ME,
Kawamura MT, Duarte RL, Carvalho Mda G and Domont GB: Differential
proteomic serum pattern of low molecular weight proteins expressed
by adenocarcinoma lung cancer patients. J Exp Ther Oncol. 5:31–38.
2005.PubMed/NCBI
|
|
15
|
Liu L, Liu J, Dai S, et al: Reduced
transthyretin expression in sera of lung cancer. Cancer Sci.
98:1617–1624. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Liu L, Liu J, Wang Y, et al: A combined
biomarker pattern improves the discrimination of lung cancer.
Biomarkers. 16:20–30. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lim A, Sengupta S, McComb ME, Théberge R,
Wilson WG, Costello CE and Jacobsen DW: In vitro and in vivo
interactions of homocysteine with human plasma transthyretin. J
Biol Chem. 278:49707–49713. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Connors LH, Lim A, Prokaeva T, Roskens VA
and Costello CE: Tabulation of human transthyretin (TTR) variants,
2003. Amyloid. 10:160–184. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ingenbleek Y and Young VR: Significance of
transthyretin in protein metabolism. Clin Chem Lab Med.
40:1281–1291. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Potter MA and Luxton G: Transthyretin
measurement as a screening tool for protein calorie malnutrition in
emergency hospital admissions. Clin Chem Lab Med. 40:1349–1354.
2002. View Article : Google Scholar
|
|
21
|
Schweigert FJ and Sehouli J:
Transthyretin, a biomarker for nutritional status and ovarian
cancer. Cancer Res. 65:11142005.PubMed/NCBI
|
|
22
|
Buxbaum JN: The systemic amyloidoses. Curr
Opin Rheumatol. 16:67–75. 2004. View Article : Google Scholar
|
|
23
|
Buxbaum JN and Reixach N: Transthyretin:
the servant of many masters. Cell Mol Life Sci. 66:3095–3101. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kozak KR, Su F, Whitelegge JP, Faull K,
Reddy S and Farias-Eisner R: Characterization of serum biomarkers
for detection of early stage ovarian cancer. Proteomics.
5:4589–4596. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Farias-Eisner G, Su F, Robbins T,
Kotlerman J, Reddy S and Farias-Eisner R: Validation of serum
biomarkers for detection of early- and late-stage endometrial
cancer. Am J Obstet Gynecol. 202:73.e1–73.e5. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Travis WD, Colby TV, Corrin B, et al:
Histological Typing of Lung and Pleural Tumours. 3rd edition.
Springer; Berlin: 1999, View Article : Google Scholar
|
|
27
|
Heinold A, Kuehl B, Brenner-Weiss G, Opelz
G and Tran TH: Sequential analysis by
immunoprecipitation-MALDI-TOF: a novel method for detection and
identification of alloantibody specificities. Hum Immunol.
71:462–467. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Grus FH, Joachim SC, Sandmann S, Thiel U,
Bruns K, Lackner KJ and Pfeiffer N: Transthyretin and complex
protein pattern in aqueous humor of patients with primary
open-angle glaucoma. Mol Vis. 14:1437–1445. 2008.PubMed/NCBI
|
|
29
|
Wang Z, Wang C, Huang X, Shen Y, Shen J
and Ying K: Differential proteome profiling of pleural effusions
from lung cancer and benign inflammatory disease patients. Biochim
Biophys Acta. 1824.692–700. 2012.PubMed/NCBI
|