Introduction
Breast cancer (BC) is among the most frequent
cancers in women in worldwide, and is the second leading cause of
cancer-related mortality in women (1). Breast cancer may generally spread to
distant locations, which affects curability of the cancer (2). Therefore, accurate and early detection
of BC, particularly when pre-symptomatic, is a crucial factor in
attaining a higher survival rate and improved prognosis for
patients (3). Mammography is a
generally preferred method in the detection of BC; however, in
young women and women with dense breast tissue, mammographic
screening may be less sensitive (4).
In these cases, magnetic resonance imaging, an alternative
screening approach, may be more sensitive than mammography
(5). Blood-based screening tests
would be cost-effective and efficient as an application for
large-scale screenings. Studies on the specific molecular targets
(oncogenes, tumor suppressor genes, growth factors, tumor antigens
or other gene products) in BC make possible the application of
blood-based screening approaches for BC. Further improvements in
protein expression analysis and proteomics methods have led to the
development of serum based diagnostics and prognostics for many
types of cancer (6). One of these
methods, surface-enhanced laser desorption ionization (SELDI),
enables the identification of biomarkers in physiological fluid
samples to distinguish patients with patients from healthy
individuals (7). Although certain
biomarkers may be used to identify BC, few of them have been
validated for clinical use. Previous studies have reported a number
of potential serum biomarkers that may be used to diagnose BC; BC1
(4.3 kDa), BC2 (8.1 kDa) and BC3 (8.9 kDa) (8,9). The
structural definitions of these biomarkers are as follows: BC1,
inter-α-trypsin inhibitor heavy chain H4; BC2, C3a des-arginine-C
terminal truncated peptide; and BC3, C3a des-arginine (10). A number of other potential biomarkers
have been identified and demonstrated to have high diagnostic
accuracy (7,11,12).
However, validating these proteins is difficult due to different
protocols for sample handling, patient populations and tumor
characteristics.
The aim of the present study was to investigate and
develop an efficient biomarker panel to distinguish patients with
BC from healthy individuals and patients with benign breast disease
(BBD) using SELDI time-of-flight mass spectrometry
(SELDI-TOF-MS).
Materials and methods
Patient characteristics
This prospective study included 62 patients with
histopathologically confirmed BC with invasive ductal carcinoma and
47 non-cancerous individuals (NCIs). The study was approved by the
Ataturk Training and Research Hospital Ethics Committee of Katip
Celebi University (Izmir, Turkey) and performed in accordance with
the Declaration of Helsinki. Among the patients with BC, 27 were in
the early stages of disease (stages I and II) and 35 patients were
in the advanced stages (stages III and IV). All patients were
staged using the American Joint Committee on Cancer (AJCC) 6th
edition staging manual (13). In the
NCI group, 16 subjects were healthy controls (HCs) and 31 were
patients with BBD. Patients with carcinoma in situ were not
enrolled in the present study. Informed consent was obtained from
all patients and control subjects. Demographic and clinical
characteristics of the participants are presented in Table I. Patients did not receive
radiotherapy or chemotherapy prior to surgery. Patients with BC had
no evidence of chronic obstructive pulmonary disease, diabetes
mellitus or malignancy at other sites. For women in the HC group
who had breast examinations, the mammography and ultrasound results
were normal.
 | Table I.Demographics of patients and control
subjects enrolled in the present study. |
Table I.
Demographics of patients and control
subjects enrolled in the present study.
|
|
| Age (years) |
|
|---|
|
|
|
|
|
|---|
| Groups | n | Median | Range | Carbohydrate antigen
15-3 level (U/ml) |
|---|
| Breast cancer |
|
|
|
24.59±15.79 |
| Stages I
and II | 27 | 53.7 | 37–73 |
16.77±2.50 |
| Stages
III and IV | 35 | 53.5 | 41–61 |
30.62±18.89 |
| Non-cancerous
individuals |
|
|
|
|
| Healthy
controls | 16 | 38.7 | 21–52 |
15.92±4.8 |
| Benign
breast disease | 31 | 41.3 | 23–71 |
16.01±5.27 |
Laboratory analysis
A standardized protocol was used for sample
collection and processing. Blood samples were collected in a BD
Vacutainer P100 v1.1 Tube (8.5 ml; BD Biosciences, Franklin Lakes,
NJ, USA) and centrifuged at 3,000 × g and 4°C for 15 min. Samples
were subsequently aliquoted and stored at −80°C for further
analysis. An automatic electrochemistry luminescence immunoassay
system (ECLIA, Elecsys 2010 system; Roche Diagnostics GmbH, Basel,
Switzerland) was used to determine the serum carbohydrate antigen
15-3 (CA15-3 level), according to the manufacturer's instructions.
The cut-off value of CA15-3 was 25 U/ml, with values >25 U/ml
considered to be positive.
Serum protein profiling
Serum protein profiles were determined using
immobilized metal affinity capture arrays (IMAC30; Bio-Rad
Laboratories, Inc., Hercules, CA, USA). Samples were incubated with
50 µl of 100 mM CuSO4 solution for 5 min, rinsed with
distilled water and washed three times for 10 min with binding
solution (200 µl; comprising 500 mM NaCl and 100 mM
NaH2PO4/NaOH; pH 7.0). Samples were
subsequently diluted with dilution solution (9 M urea, 50 mM
Tris/HCl, pH 9.0, 2% w/v CHAPS (Sigma-Aldrich; Merck KGaA,
Darmstadt, Germany) at a ratio of 5:1 and vortexed at medium mixing
mode for 30 min at 4°C. A total of 100 µl binding buffer was added
into 10 µl of this mixture. From this, 100 µl was applied to each
spot on IMAC30 chips. The chips were incubated at room temperature
for 60 min on the horizontal shaker (900 rpm) and subsequently
washed 4 times with 200 µl binding solution for 10 min on the
horizontal shaker. Chips were washed with 1 mM HEPES, pH 7.0 and
stored at room temperature until dry. Matrix solution (1 µl)
including 50% saturated solution of sinapinic acid (Fluka Chemie
AG, Buchs, Switzerland) in 50% acetonitrile and 0.5%
trifluoroacetic acid (both from Sigma-Aldrich; Merck KGaA), was
added to each well and stored at room temperature until dry. Chips
were analyzed by using the Protein Biological System IIc SELDI-TOF
(Bio-Rad Laboratories, Inc., Hercules, CA, USA). The generation of
TOF spectra was achieved by 192 laser shots with laser intensity of
220 eV and a detector sensitivity of 9. Peaks between 3,000 and
50,000 Da were recorded, with a maximum peak of 200,000 Da. The
external calibration of the instrument was achieved using the
all-in-one peptide molecular mass standard (Bio-Rad Laboratories,
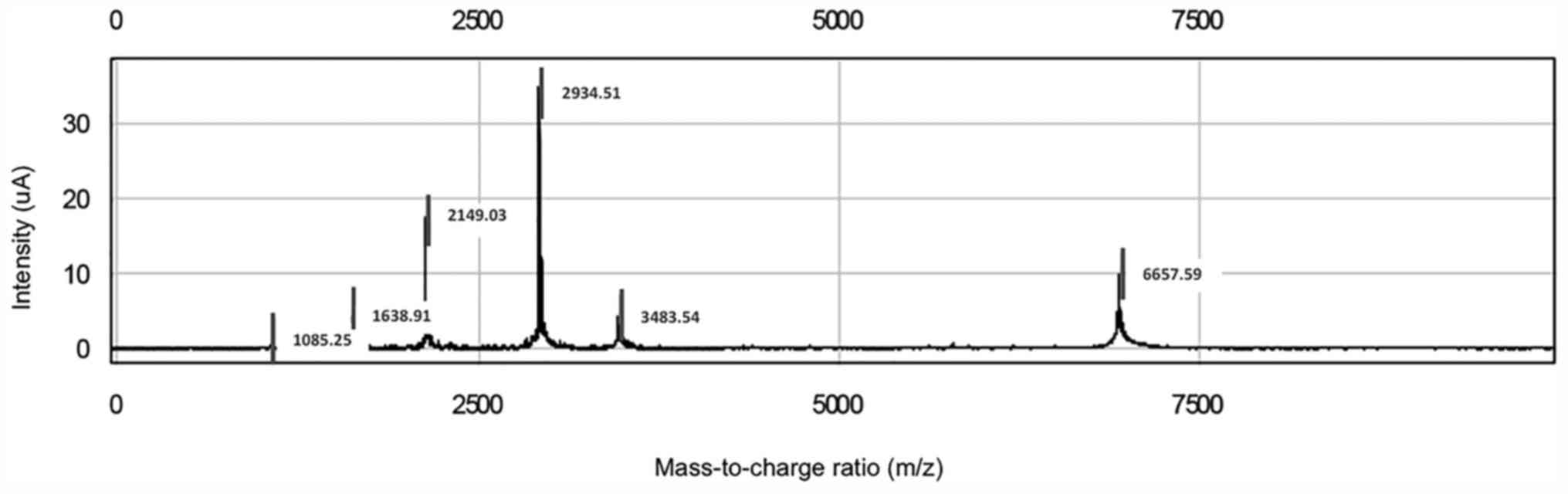
Inc.) as presented in Fig. 1. The
mass accuracy was 0.08% with this system. All mass spectra were
calibrated internally. Peak intensities were normalized to total
ion flow.
Bioinformatics analysis
The Epi Info-7 program was used to calculate sample
size Spectral data were collected using Protein Chip Data Manager
3.0 software (Bio-Rad Laboratories, Inc.), which was also used for
data processing and univariate statistical analysis. Internal mass
calibration and normalization of peak intensities were performed
based on total ion flow. Automatic baseline correction was used.
For peak selection, the very low mass region (0–1,500 Da)
overlapping with single-photon absorptiometry peaks was excluded.
The default average filter with a setting of ×0.2 expected peak
width was used for data filtering. Eligible mass peaks were with
signal/noise >5. Mass window for peak clustering was 0.3% of the
peak mass. Inter-group comparison of each peak cluster was
performed using the one-way Mann-Whitney U test. Areas under the
receiver operating characteristic (ROC) curve (AUC) were calculated
for each peak cluster. A total of 118 clusters were obtained; the
cluster mass was 0.5% and the signal-to-noise ratio for the second
pass was 1 for the settings used for cluster completion.
Statistical analysis
All continuous variables are presented as the mean ±
standard deviation. Comparisons between groups were achieved by
two-sample t-tests and Pearson's χ2 tests. Relative peak
intensity levels were compared using the Student's t-test.
Univariate analysis using the Mann-Whitney U test was used to
compare sera from patients with BC with the NCI group. ROC analysis
was achieved by the calculation of AUC. SPSS 21.0 (IBM Corp.,
Armonk, NY, USA) was used for all statistical analysis. P<0.05
was considered to indicate a statistically significant
difference.
Results
Reproducibility
Each serum sample was spotted on eight locations on
one chip, to attain a reliable analysis technique. The coefficient
variations of m/z values were 0.03–0.07 and the protein intensity
was 0.34–0.79 for randomly selected peaks. The reproducibility of
SELDI ProteinChip assays was assessed by running 16 pooled normal
human serum samples. The mean variance coefficient based on pooled
sera was lower <12%. Variation in day-to-day sampling and
instrumentation or chip variations were negligible.
Serum protein profiling of the BC
group compared with the HC group
For the study group, 118 peaks per spectrum were
obtained, with masses ranging from 2 to 20 kDa. Of these, no single
peak could be used to distinguish BC from HC serum. From the 118
peaks, 9 protein peaks at m/z 1,452, 2,670, 3,972, 5,354, 5,523,
6,850, 7,926, 8,143 and 8,115 (BC2) were automatically selected as
splitters. A total of 5 peaks were decreased (m/z 1,452, 2,670,
3,972, 5,354 and 5,523; P<0.001; Table II) and 4 were significantly increased
(m/z 6,850, 7,926, 8,115 and 8,143; P<0.001; Table II) in patients with BC compared with
the HC group.
 | Table II.Surface-enhanced laser desorption
ionization time-of-flight mass spectrometry results for BC and HC
groups. |
Table II.
Surface-enhanced laser desorption
ionization time-of-flight mass spectrometry results for BC and HC
groups.
|
|
| Peak intensity |
|
|
|---|
|
|
|
|
|
|
|---|
| Protein peaks
mass-to-change ratio | Trend | BC (n=62) | HC (n=16) | Area under curve | P-value |
|---|
| 1452 | Decreased |
154.74±88.02 |
262.18±124.99 | 0.775 | <0.001 |
| 2670 | Decreased |
19.88±17.01 |
42.80±20.85 | 0.796 | <0.001 |
| 3972 | Decreased |
62.29±36.27 |
104.06±45.17 | 0.781 | <0.001 |
| 5354 | Decreased |
20.69±18.48 |
50±28.64 | 0.805 | <0.001 |
| 5523 | Decreased |
8.94±0.68 |
17±0.76 | 0.806 | <0.001 |
| 6850 | Increased |
44.55±2.99 |
21.95±1.6 | 0.781 | <0.001 |
| 7926 | Increased |
70.86±36.19 |
37.56±17.54 | 0.796 | <0.001 |
| 8115 (BC2) | Increased |
97.65±10.15 |
22.76±
13.85 | 0.751 | <0.001 |
| 8143 | Increased |
138.37±80.33 |
62.19±35.95 | 0.781 | <0.001 |
Serum protein profiling of the BC
group compared with the BBD group
The SELDI-TOF-MS results for the BC and BBD groups
revealed that 3 protein peaks (m/z 3,972, 5,336, and 11,185) were
significantly decreased and 6 (m/z 4,062, 4,071, 4,609, 6,850,
8,115, and 8,133) were significantly increased in the BC group
compared with the BBD group (P<0.01; Table III). Of these, the protein peak at
8,115 Da (BC2) was one of the previously defined biomarkers of BC
(8,9).
 | Table III.Surface-enhanced laser desorption
ionization time-of-flight mass spectrometry results for BC and BBD
groups. |
Table III.
Surface-enhanced laser desorption
ionization time-of-flight mass spectrometry results for BC and BBD
groups.
|
|
| Peak intensity |
|
|
|---|
|
|
|
|
|
|
|---|
| Protein peaks
mass-to-change ratio | Trend | BC (n=62) | BBD (n=31) | Area under
curve | P-value |
|---|
| 3972 | Decreased |
62.29±36.27 |
123.57±73.94 | 0.783 | <0.001 |
| 4062 | Increased |
127.77±115.72 |
36.76±22.98 | 0.721 | <0.001 |
| 4071 | Increased |
118.95±79.82 |
59.03±23.58 | 0.740 | <0.001 |
| 4609 | Increased |
92.38±40.01 |
67.23±16.39 | 0.702 | 0.001 |
| 5336 | Decreased |
45.74±39.68 |
93.61±57.77 | 0.783 | <0.001 |
| 6850 | Increased |
44.55±2.99 |
25.75±11.68 | 0.721 | <0.001 |
| 8115 (BC2) | Increased |
97.65±10.15 |
31.53±23.77 | 0.698 | 0.002 |
| 8133 | Increased |
131.77±114.87 |
48.46±23.45 | 0.698 | 0.001 |
| 11185 | Decreased |
6.02±8.75 |
16.04±15.94 | 0.769 | <0.001 |
Serum protein profiling of the BC
group compared with the NCI group
SELDI-TOF-MS results revealed peaks at m/z 3,972,
6,850, 8,115 (BC2) and 8,949 Da (BC3) for patients with BC compared
with NCI (Table IV). ROC analysis of
each candidate tumor marker as a biomarker to distinguish between
the BC and NCI groups is presented in Fig. 2. The AUC values for peaks are as
follows: 0.799 for m/z 3,972, 0.752 for m/z 6,850, 0.732 for m/z
8,115 (BC2), 0.603 for m/z 8,949 (BC3), and 0.762 for CA15-3. The
ROC data revealed that the protein peaks at 3,972 and 6,850 in
patients with BC were novel biomarkers suitable for diagnosis. The
sensitivity rates of 3,972, 6,850, 8,115 (BC2) and 8,949 Da (BC3)
peaks were 63.0, 83.87, 66.13 and 77.0%, respectively (P<0.05).
Sensitivity and specificity values of CA15-3 were determined as
25.81 and 97.87%, respectively, whereas the sensitivity and
specificity of the serum protein profiling test (4 peak pattern
tests and CA15-3) were 98.39 and 100%, respectively (data not
shown).
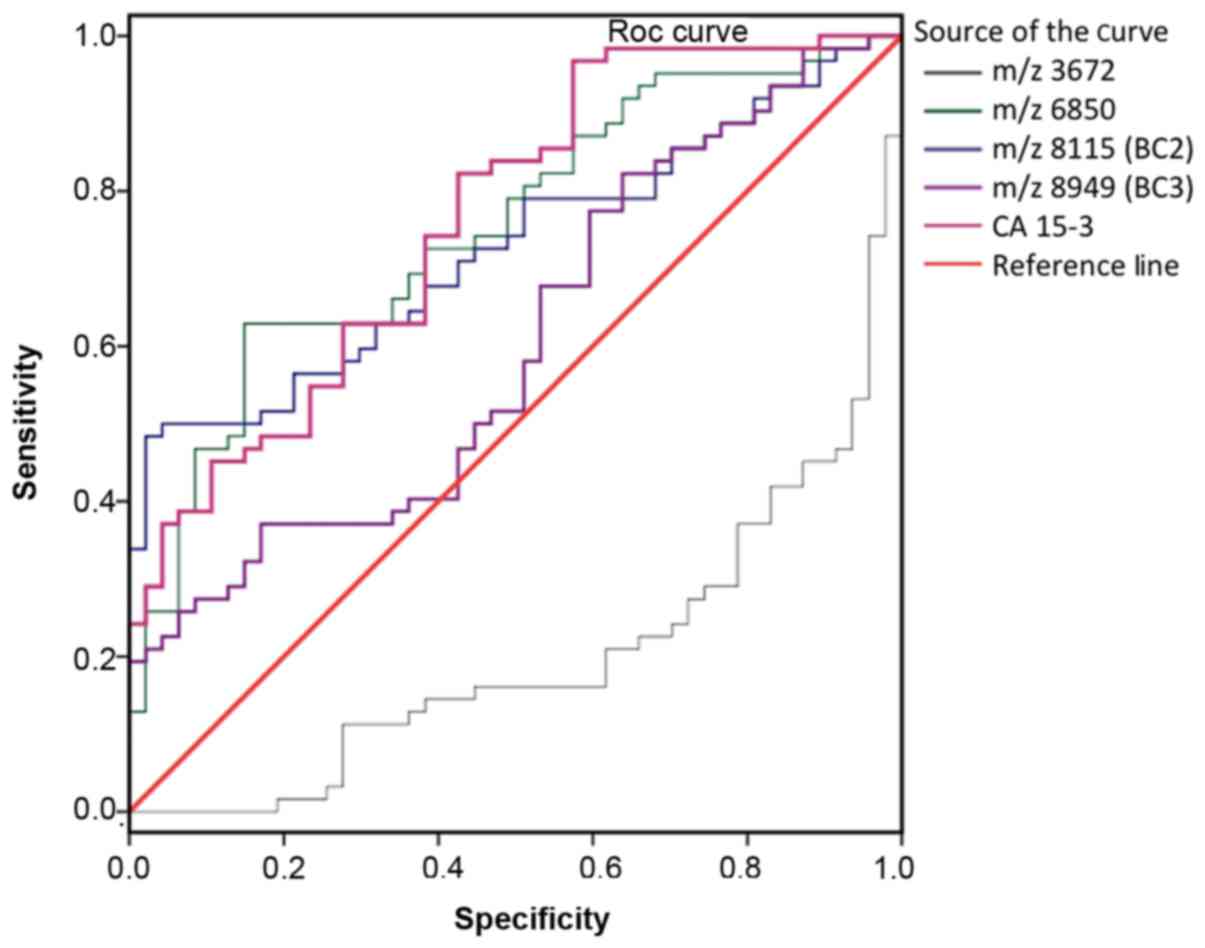 | Figure 2.ROC curve for each protein peak
identified using SELDI-TOF-MS in patients with breast cancer
compared with non-cancerous individuals. Area under curve values
were as follows: m/z 3,972, 0.700; m/z 6,850, 0.752; m/z 8,115,
0.732; m/z 8,949, 0.603; CA15-3, 0.762. ROC, receiver operating
characteristic; SELDI-TOF-MS, surface-enhanced laser desorption
ionization time-of-flight mass spectrometry; CA15-3, carbohydrate
antigen 15-3. |
 | Table IV.Biomarkers used to determine patients
with BC compared with NCI. |
Table IV.
Biomarkers used to determine patients
with BC compared with NCI.
|
|
| Protein peak |
|
|
|---|
|
|
|
|
|
|
|---|
| Peak intensity
mass-to-change ratio | Trend | BC (n=62) | NCI (n=47) | Area under
curve | P-value |
|---|
| 3972 | Decreased |
62.29±36.27 |
116.93±65.71 | 0.799 | <0.001 |
| 6850 | Increased |
44.55±2.99 |
24.45±13.52 | 0.752 | <0.001 |
| 8115 (BC2) | Increased |
97.65±10.15 |
28.54±21.19 | 0.732 | <0.001 |
| 8949 (BC3) | Increased |
185.28±95.47 |
148.26±62.16 | 0.603 | 0.016 |
| Carbohydrate
antigen 15-3 | Increased |
24.59
±15.79 |
15.72±4.72 | 0.762 | <0.001 |
Bioinformatics analysis
A logistic regression curve was calculated using the
equation [y = 2.438 + 0.020(m/z 397) + 0.033(m/z 6850) + 0.011(m/z
8115) + 0.154(CA15-3)]. As presented in Table V, the minimum increase was 0.98-fold
[odds ratio (OR) 0.982] at 3,972 Da, and the maximum increase was
1.17-fold (OR 1.167) at CA15-3. Considering the results of
multivariable logistic regression, a set of 4 protein peaks was
selected to diagnose BC. The 4 peaks were 3,972, 6,850, 8,115 (BC2)
and 8,949 (BC3), as well as CA15-3. Of these, 8,115 (BC2), 8,949
(BC3) and CA15-3 have been previously reported as biomarkers
(8,9).
The peaks at m/z 3,972 and 6,850 may therefore be novel candidate
markers for distinguishing patients with BC from NCI.
 | Table V.Results of multivariable logistic
regression of risk factors for breast cancer. |
Table V.
Results of multivariable logistic
regression of risk factors for breast cancer.
| Covariate
mass-to-change ratio | Odds ratio | 95% Confidence
limit | P-value | Diagnostic
cut-off |
|---|
| 3972 | 0.982 | 0.968–0.995 | 0.009 | 63.54 |
| 6850 | 1.051 | 1.008–1.096 | 0.019 | 59.27 |
| 8115 (BC2) | 1.021 | 1.000–1.042 | 0.047 | 25.33 |
| 8949 (BC3) | 0.991 | 0.981–1.002 | 0.115 | 140.94 |
| Carbohydrate
antigen 15-3 | 1.167 | 1.036–1.314 | 0.011 | 19.21 |
Discussion
Proteomics are widely used in BC studies to identify
diagnostic markers; however, the numbers of validated clinical
markers for BC is limited (13).
Several genes have previously been reported to be upregulated in BC
compared with control tissues (14,15). In
addition, proteomic methods are easy to apply in the clinic to
diagnose patients (16,17). Serum SELDI-TOF-MS protein profiling is
a powerful method used for biomarker discovery and can be used to
distinguish patients with BC from NCI with high sensitivity and
specificity (18). It has previously
been reported that monitoring protein-based markers in blood and
urine samples may be used for cancer staging (19). In the past decade, a number of studies
have utilized SELDI-TOF to identify proteomic patterns in
biological fluids, including serum, urine and nipple aspirates
(20–22).
In the present study, serum samples of 62 patients
with BC and 47 NCI were analyzed using SELDI-TOF-MS. Among the 118
examined peaks, multivariate analysis revealed 9 peaks in
SELDI-TOF-MS results for patients with BC compared with those with
BBD, as well as 9 peaks for patients with BC compared with HC
subjects. By using the common peaks in both sets, 4 protein peaks
were predicted as biomarkers to distinguish patients with BC from
NCI. Several mutations are known to increase susceptibility to BC;
the majority of inherited cases of BC are associated with two
abnormal genes, BC1 and BC2 (23). It
has been identified that the protein expression of BC1/2 is
associated with increased susceptibility to BC by 40–80% (24). Although several biomarker panels have
been reported for various diseases using SELDI and ProteinChip
arrays (25), there are few validated
markers. The first validation study by Li et al (8) proposed the use of BC2 and BC3 as
biomarkers for BC. Decreases in BC1 and increased expression of BC2
and BC3 in patients with BC have been reported in several studies
(22,26,27). BC2
and BC3 have also been reported to possess independent diagnostic
merit to distinguish between malignant disease, benign disease and
control groups (9). BC3 was therefore
included in the protein panel for analysis.
CA15-3 is the most widely used serum marker in
patients with BC (22). Serum CA15-3
has been extensively studied, mainly to monitor the response of BC
to treatment or to detect early relapse in BC follow-up (28). A study by Gautam et al
(29) reported that elevated
concentrations of CA15-3 may be a useful and reliable diagnostic
and prognostic indicator, as well as an indicator of treatment
efficacy (29). The number of
metastatic sites has been demonstrated to affect levels of CA15-3
(30). As CA15-3 is the most widely
used serum tumor marker, it was included in the diagnostic panel in
the present study.
Two peaks, at m/z values 3,972 and 6,850 Da, were
identified for the first time as potential biomarkers of BC using
SELDI-TOF-MS. These peaks were demonstrated to be able to
distinguish patients with BC from HC subjects and patients with
BBD. The peak at 3,972 Da was significantly decreased and the peak
at 6,850 Da was significantly increased. Previous studies have
reported 4.3, 8.1 and 8.9 kDa proteins as biomarkers of BC;
however, these results differ to those of the present study
(8,11,22,26). A 3.8
kDa protein, close to m/z 3,972, was reported to be highly
sensitive for the diagnosis of BC by Chung et al (12). A number of serum-based candidate
biomarkers have been identified in different studies (8,10,22,31).
Studies may have some differences in terms of inclusion/exclusion
criteria, biologic samples, preparation protocols, arrays and
analytical settings, which will affect the reproducibility and
robustness of results. SELDI-TOF-MS proteomic profiling has been
proposed as a promising high-throughput technology with potential
applications in BC screening, detection and prognostication. In
some studies, detected ion peaks were identified as different due
to variations in population demographics, stage of disease,
collection and storage conditions of samples, and analytical
procedures (11,32,33). Aside
from the most common proteins (BC1, BC2, BC3 and CA15-3), many of
the studied peaks had poor sensitivities and specificities and
alone were insufficient to diagnose BC. Furthermore, discrepancies
in the peaks have been reported for several studies. For example,
peaks at m/z 4,276 and 4,292 were reported by Winden et al
(11) to be decreased in patients
with BC, which was consistent with other studies; however, the peak
at m/z 8,941 was reported to be decreased, whereas it was increased
in previous studies (8,22,26). A
review by Muthu et al (34),
which summarized the features of SELDI-TOF-MS and its application
in cancer biomarker discovery, reported that the technique has not
yet been sufficiently investigated in different types of cancer and
other diseases (34).
In present study, the resultant 4 peaks panel
together with CA15-3 was demonstrated to have good sensitivity and
specificity for the diagnosis of BC. These results indicate that
protein peaks at 3,972 and 6,850 Da may be the novel candidate
biomarkers for the diagnosis of BC. In conclusion, 4 peak clusters
which are significantly different in patients with BC compared with
NCI were identified using SELDI-TOF-MS, The identified peaks:
3,972, 6,850, 8,115 (BC2) and 8,949 Da (BC3) combined with CA15-3
expression may be used as a protein-profiling test to diagnose BC.
However, further investigation using a larger sample size should be
performed to verify these results.
References
|
1
|
Sun YS, Zhao Z, Yang ZN, Xu F, Lu HJ, Zhu
ZY, Shi W, Jiang J, Yao PP and Zhu HP: Risk factors and preventions
of breast cancer. Int J Biol Sci. 13:1387–1397. 2017. View Article : Google Scholar
|
|
2
|
Rahman M and Mohammed S: Breast cancer
metastasis and the lymphatic system. Oncol Lett. 10:1233–1239.
2015. View Article : Google Scholar
|
|
3
|
Niell BL, Freer PE, Weinfurtner RJ, Arleo
EK and Drukteinis JS: Screening for breast cancer. Radiol Clin
North Am. 55:1145–1162. 2017. View Article : Google Scholar
|
|
4
|
Nothacker M, Duda V, Hahn M, Warm M,
Degenhardt F, Madjar H, Weinbrenner S and Albert US: Early
detection of breast cancer: Benefits and risks of supplemental
breast ultrasound in asymptomatic women with mammographically dense
breast tissue. A systematic review. BMC Cancer. 9:3352009.
View Article : Google Scholar
|
|
5
|
Drukteinis JS, Mooney BP, Flowers CI and
Gatenby RA: Beyond mammography: New frontiers in breast cancer
screening. Am J Med. 126:472–479. 2013. View Article : Google Scholar
|
|
6
|
Baskın Y and Yiğitbaşı T: Clinical
proteomics of breast cancer. Curr Genomics. 11:528–536. 2010.
View Article : Google Scholar
|
|
7
|
Liu C: The application of SELDI-TOF-MS in
clinical diagnosis of cancers. J Biomed Biotechnol.
2011:2458212011. View Article : Google Scholar
|
|
8
|
Li J, Orlandi R, White CN, Rosenzweig J,
Zhao J, Seregni E, Morelli D, Yu Y, Meng XY, Zhang Z, et al:
Independent validation of candidate breast cancer serum biomarkers
identified by mass spectrometry. Clin Chem. 51:2229–2235. 2005.
View Article : Google Scholar
|
|
9
|
Atahan K, Küpeli H, Gür S, Yiğitbaşı T,
Baskın Y, Yiğit S, Deniz M, Cökmez A and Tarcan E: The value of
serum biomarkers (Bc1, Bc2, Bc3) in the diagnosis of early breast
cancer. Int J Med Sci. 8:148–155. 2011. View Article : Google Scholar
|
|
10
|
Belluco C, Petricoin EF, Mammano E,
Facchiano F, Ross-Rucker S, Nitti D, Di Maggio C, Liu C, Lise M,
Liotta LA, et al: Serum proteomic analysis identifies a highly
sensitive and specific discriminatory pattern in stage 1 breast
cancer. Ann Surg Oncol. 14:2470–2476. 2007. View Article : Google Scholar
|
|
11
|
van Winden AW, Gast MC, Beijnen JH,
Rutgers EJT, Grobbee DE, Peeters PH and van Gils CH: Validation of
previously identified serum biomarkers for breast cancer with
SELDI-TOF MS: A case control study. BMC Med Genomics. 2:42009.
View Article : Google Scholar
|
|
12
|
Chung L, Moore K, Phillips L, Boyle FM,
Marsh DJ and Baxter RC: Novel serum protein biomarker panel
revealed by mass spectrometry and its prognostic value in breast
cancer. Breast Cancer Res. 16:R632014. View
Article : Google Scholar
|
|
13
|
Singletary SE and Connolly JL: Breast
cancer staging: working with the sixth edition of the AJCC Cancer
Staging Manual. CA Cancer J Clin. 56:37–47. 2006. View Article : Google Scholar
|
|
14
|
Harris L, Fritsche H, Mennel R, Norton L,
Ravdin P, Taube S, Somerfield MR, Hayes DF and Bast RC Jr; American
Society of Clinical Oncology, : American Society of Clinical
Oncology 2007 update of recommendations for the use of tumor
markers in breast cancer. J Clin Oncol. 25:5287–5312. 2007.
View Article : Google Scholar
|
|
15
|
Paweletz CP, Gillespie JW, Ornstein DK,
Simone NL, Brown MR, Cole KA, Wang QH, Huang J, Hu N, Yip TT, et
al: Rapid protein display profiling of cancer progression directly
from human tissue using a protein biochip. Drug Dev Res. 49:34–42.
2000. View Article : Google Scholar
|
|
16
|
Nakanishi T, Shimizu A, Okamoto N,
Ingendoh A and Kanai M: Analysis of serum protein precipitated with
antiserum by matrix-assisted laser desorption
ionization/time-of-flight and electrospray ionization mass
spectrometry as a clinical laboratory test. J Am Soc Mass Spectrom.
6:854–859. 1995. View Article : Google Scholar
|
|
17
|
Lacey JM, Bergen HR, Magera MJ, Naylor S
and O'Brien JF: Rapid determination of transferrin isoforms by
immunoaffinity liquid chromatography and electrospray mass
spectrometry. Clin Chem. 47:513–518. 2001.
|
|
18
|
Vlahou A, Laronga C, Wilson L, Gregory B,
Fournier K, McGaughey D, Perry RR, Wright GL Jr and Semmes OJ: A
novel approach toward development of a rapid blood test for breast
cancer. Clin Breast Cancer. 4:203–209. 2003. View Article : Google Scholar
|
|
19
|
Yao F, Zhang C, Du W, Liu C and Xu Y:
Identification of gene-expression signatures and protein markers
for breast cancer grading and staging. PLoS One. 10:e01382132015.
View Article : Google Scholar
|
|
20
|
Diamandis EP: Point: Proteomic patterns in
biological fluids: Do they represent the future of cancer
diagnostics? Clin Chem. 49:1272–1275. 2003. View Article : Google Scholar
|
|
21
|
Higgins SA, Matloff ET, Rimm DL, Dziura J,
Haffty BG and King BL: Patterns of reduced nipple aspirate fluid
production and ductal lavage cellularity in women at high risk for
breast cancer. Breast Cancer Res. 7:R1017–R1022. 2005. View Article : Google Scholar
|
|
22
|
Li J, Zhang Z, Rosenzweig J, Wang YY and
Chan DW: Proteomics and bioinformatics approaches for
identification of serum biomarkers to detect breast cancer. Clin
Chem. 48:1296–1304. 2002.
|
|
23
|
Ford D, Easton DF, Stratton M, Narod S,
Goldgar D, Devilee P, Bishop DT, Weber B, Lenoir G, Chang-Claude J,
et al The Breast Cancer Linkage Consortium, : Genetic heterogeneity
and penetrance analysis of the BRCA1 and BRCA2 genes in breast
cancer families. Am J Hum Genet. 62:676–689. 1998. View Article : Google Scholar
|
|
24
|
Vos S, Moelans CB and van Diest PJ: BRCA
promoter methylation in sporadic versus BRCA germline
mutation-related breast cancers. Breast Cancer Res. 19:642017.
View Article : Google Scholar
|
|
25
|
Neagu M, Constantin C, Tanase C and Boda
D: Patented biomarker panels in early detection of cancer. Recent
Pat Biomark. 1:10–24. 2011. View Article : Google Scholar
|
|
26
|
Mathelin C, Cromer A, Wendling C,
Tomasetto C and Rio MC: Serum biomarkers for detection of breast
cancers: A prospective study. Breast Cancer Res Treat. 96:83–90.
2006. View Article : Google Scholar
|
|
27
|
Atalay C and Kubilay D: The results of
screening with SELDI-TOF-MS method in breast cancer diagnosis. J
Breast Health. 7:106–108. 2011.
|
|
28
|
Stearns V, Yamauchi H and Hayes DF:
Circulating tumor markers in breast cancer: Accepted utilities and
novel prospects. Breast Cancer Res Treat. 52:239–259. 1998.
View Article : Google Scholar
|
|
29
|
Gautam A and Verma S, Pantola C and Verma
S: Utility of CA 15-3 as diagnostic and prognostic marker in breast
cancer. IOSR-JDMS. 14:17–20. 2015.
|
|
30
|
Geng B, Liang MM, Ye XB and Zhao WY:
Association of CA 15-3 and CEA with clinicopathological parameters
in patients with metastatic breast cancer. Mol Clin Oncol.
3:232–236. 2015. View Article : Google Scholar
|
|
31
|
Zeidan BA, Cutress RI, Hastie C, Mirnezami
AH, Packham G and Townsend PA: SELDI-TOF-MS proteomics in breast
cancer. Clin Proteomics. 5:133–147. 2009. View Article : Google Scholar
|
|
32
|
Yin P, Lehmann R and Xu G: Effects of
pre-analytical processes on blood samples used in metabolomics
studies. Anal Bioanal Chem. 407:4879–4892. 2015. View Article : Google Scholar
|
|
33
|
Chandramouli K and Qian PY: Proteomics:
Challenges, techniques and possibilities to overcome biological
sample complexity. Hum Genomics Proteomics. 2009:2392042009.
View Article : Google Scholar
|
|
34
|
Muthu M, Vimala A, Mendoza OH and Gopal J:
Tracing the voyage of SELDI-TOF-MS in cancer biomarker discovery
and its current depreciation trend-need for resurrection? Trends
Analyt Chem. 76:95–101. 2016. View Article : Google Scholar
|