Introduction
Bronchial asthma refers to a chronic airway
inflammatory disease, which is associated with multiple cells
(including eosinophils, mast cells, T-lymphocytes, neutrophils,
smooth muscle cells and airway epithelial cells) and cellular
components in the human body. It is usually accompanied by airway
hyperreactivity (AHR), widely variable and reversible airway
limitation and symptoms, such as recurrent wheezing, dyspnea, chest
distress or cough. This disease often occurs or is aggravated at
night and in the early hours of the morning, and the symptoms can
be relieved spontaneously or following treatment (1,2). At
present, asthma is one of the relatively commonly observed chronic
diseases globally, exhibiting a gradually increasing prevalence
rate, and markedly affects the quality of life and physical health
of patients. Thus far, inhaled corticosteroids are the most
effective treatment for bronchial asthma; however, this type of
treatment is associated with drug dependence and cannot achieve
ideal therapeutic efficacy in some children, since it cures the
symptoms and not the disease (2).
Traditional Chinese medicine (TCM) has long been applied in the
treatment of asthma (3); it is of
immense medical value to search for more effective TCM treatments
for allergic asthma.
Astragalus, a traditional Chinese herbal
medicine, has been extensively adopted for the treatment of asthma.
It is a component found in well-known TCM prescriptions, such as
YuPingFengSan, PingChuanGuBen Decoction and modified LiuJunZi
Decoction, while Astragalus polysaccharide (APS) is one of
the primary active ingredients in Astragalus (4,5). Gut
microbial imbalance is the new direction of research on the
occurrence and progress of asthma in children, and regulating the
gut microbial composition may become an effective method for the
prevention or treatment of asthma (6). Based on the TCM theories of ‘the lung
and the large intestine being interior-exteriorly related’ and
‘reinforcing earth to generate metal’, modern medicine has put
forth the concept of the ‘lung-intestinal’ axis. The present study
aimed to examine the effects of APS on the gut microbiota and
metabolites in asthmatic mice based on 16S rRNA and non-targeted
metabolomics.
Materials and methods
Drugs and reagents
APS (purity, >98%; Shanghai Macklin Biochemical
Co., Ltd.), ovalbumin (OVA) and aluminium hydroxide (Sigma-Aldrich;
Merck KGaA) were used in the experiments in the present study.
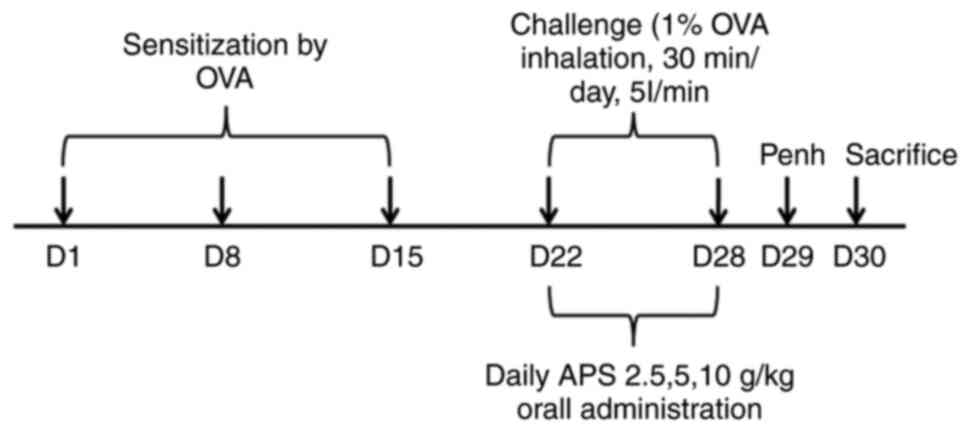
Animal and experimental protocols
A total of 36 healthy female BALB/c mice (weighing
18-22 g, 4-6 weeks old) were purchased from the Laboratory Animal
Center of Hangzhou Medical College. All experimental protocols
involving animals were approved by the Ethics Committee of the
Zhejiang Center of Laboratory Animals (Hangzhou, China; approval
no. ZJCLA-IACUC-20020187) and conducted conforming to the
guidelines of the China Animal Protection Commission. In addition,
all mice were raised in an environment with a temperature of 22±2˚C
and with a relative humidity of 50±1%.
In a random manner, the 36 mice were classified into
six groups, with 6 mice in each group, as follows: i) The normal
control group sensitized with phosphate-buffered saline (PBS;
HyClone; Cytiva); ii) the asthma group sensitized and challenged
with OVA (grade V, Sigma-Aldrich; Merck KGaA); iii) the OVA + APS
(2.5 g/kg) treatment group; iv) the OVA + APS (5 g/kg) treatment
group; v) the OVA + APS (5 g/kg) treatment group; and vi) the OVA +
dexamethasone (DXM, Sigma-Aldrich; Merck KGaA, 2 mg/kg) treatment
group used as a positive control.
The establishment of the mouse model of asthma was
as follows: i) The sensitization stage: The asthma model was
constructed by the injection of 20 µg emulsifying OVA in 200 µl PBS
on days 1, 8 and 15 of the experiment. ii) The excitation stage:
Following initial sensitization, the mice were subjected to the
aerosol inhalation of 1% OVA in a closed container for excitation
every day from day 22, and each aerosol inhalation lasted for ~30
min for 1 week consecutively. In the negative control group, PBS
was used instead of OVA during the sensitization and excitation
stages. All drugs (APS and DXM) were dissolved in an equivalent
amount of PBS and administered at a dose of 0.2 ml/mouse. APS was
administered via intragastric administration every day for 1 week.
DXM (2 mg/kg) served as the positive control and was administered
in the same manner. On day 29, the enhancement suspension (Penh)
was evaluated, and the mice were sacrificed on day 30. Serum, lung
tissue and splenic cells were harvested for further analysis
(Fig. 1). At the end of the animal
experimental period, the experimental animals were subjected to
cervical dislocation under anesthesia. The mice were anesthetized
with 40 mg/kg pentobarbital sodium solution by intraperitoneal
injection. All procedures were carried out following strict ethical
guidelines, and all efforts were made to minimize animal
suffering.
Methacholine (Mch) measurement by
AHR
At 24 h following the final excitation, AHR was
indirectly assessed using whole-body plethysmography. More
specifically, each conscious mouse was stimulated with Mch at
elevating doses (Mch aerosol containing 3.125, 6.25, 12.5, 25, 5
and 50 mg/ml saline) for 3 min. The 3-min Penh value following each
Mch excitation was calculated.
Hematoxylin and eosin (H&E)
staining of lung tissue for histological examination
Following collection, the lung tissue was fixed with
a concentration of 4% paraformaldehyde (20˚C,48 h),
paraffin-imbedded and subjected to histological staining. Briefly,
the 4-µm-thick left lung sections were stained with H&E
(Beijing Solarbio Science & Technology Co., Ltd.; 20˚C, 30 min)
to evaluate eosinophil infiltration and inflammatory cell
infiltration in the surrounding lung tissue.
16S rDNA sequencing analysis of gut
microbiota
DNA was extracted from the mouse feces, and cellular
DNA was amplified using PCR. In brief, the amplification region was
bacterial 16SV3+V4, and the primers used were 338F and 806R. The V3
upstream primer sequence was 5'-ACTCCTACGGGAGGCAGCA-3', and the V4
downstream primer sequence was 5'-GGACTACHVGGGTWTCTAAT-3'. The
DNBseq sequencing process and method were applied in sequencing. In
addition, the raw data were spliced with FLASH (version 1.2.11),
and the spliced Tags were clustered into operational taxonomic
units (OTUs) with the application of USEARCH software
(v7.0.1090).
Mouse serum non-targeted metabolomics
detection
An appropriate amount of serum sample was
supplemented with 200 µl water for homogenization, followed by
vortexing for 60 sec. Thereafter, 800 µl methanol-acetonitrile
solution (1:1, Merck, V:V) was supplemented for vortexing for a
further 60 sec, followed by low-temperature ultrasonic treatment
twice (30 min each). After being allowed to stand at -20˚C for 1 h
to precipitate the protein, the sample was subject to
centrifugation at 14,000 x g and 4˚C for 20 min to collect the
supernatant. Subsequently, the sample was separated with the
ACQUITY UPLC BEH Amide chromatographic column (100x2.1 mm, 1.7 µm)
of the Agilent 1290 Infinity LC ultra-performance liquid
chromatography system (UPLC), and mass spectrometry was carried out
using the Agilent 6550 mass spectrometer (Agilent Technologies,
Inc.). In addition, the metabolites were detected with the AB
Triple TOF 6600 mass spectrometer, and the structural
identification of metabolites was completed by collecting the
first-order and second-order spectra of quality control (QC)
samples. Following data preprocessing with Pareto scaling,
unidimensional and multidimensional statistical analyses were
performed, and the volcano plot was drawn using R software.
Kyoto Encyclopedia of Genes and
Genomes (KEGG) metabolic pathway enrichment
Based on the KEGG compound database, the metabolites
were annotated. Furthermore, the annotated metabolites were then
matched based on the KEGG pathway database. Significantly regulated
metabolites were imported into the metabolite enrichment analysis
MetaboAnalyst 5.0 for their metabolic pathway enrichment. The
enrichment results were calculated for the significance (P-values)
by hypergeometric tests and presented as bubble plots.
Statistical analysis
Statistical analysis was conducted using SPSS
version 22.0 software (IBM Corp.). Measurement data are presented
as the mean ± standard deviation (SD) and were compared using
one-way analysis of variance for comparisons among multiple groups.
In pairwise comparisons, the least significant difference (LSD)
test was adopted in the case of homogeneity of variance. One-way
ANOVA was used for comparison between multiple groups and pairwise
comparisons were conducted using the Bonferroni method. Tamhane's
T2 test was used for multiple comparisons in the case of
heterogeneity of variance. A value of P<0.05 was considered to
indicate a statistically significant difference.
Results
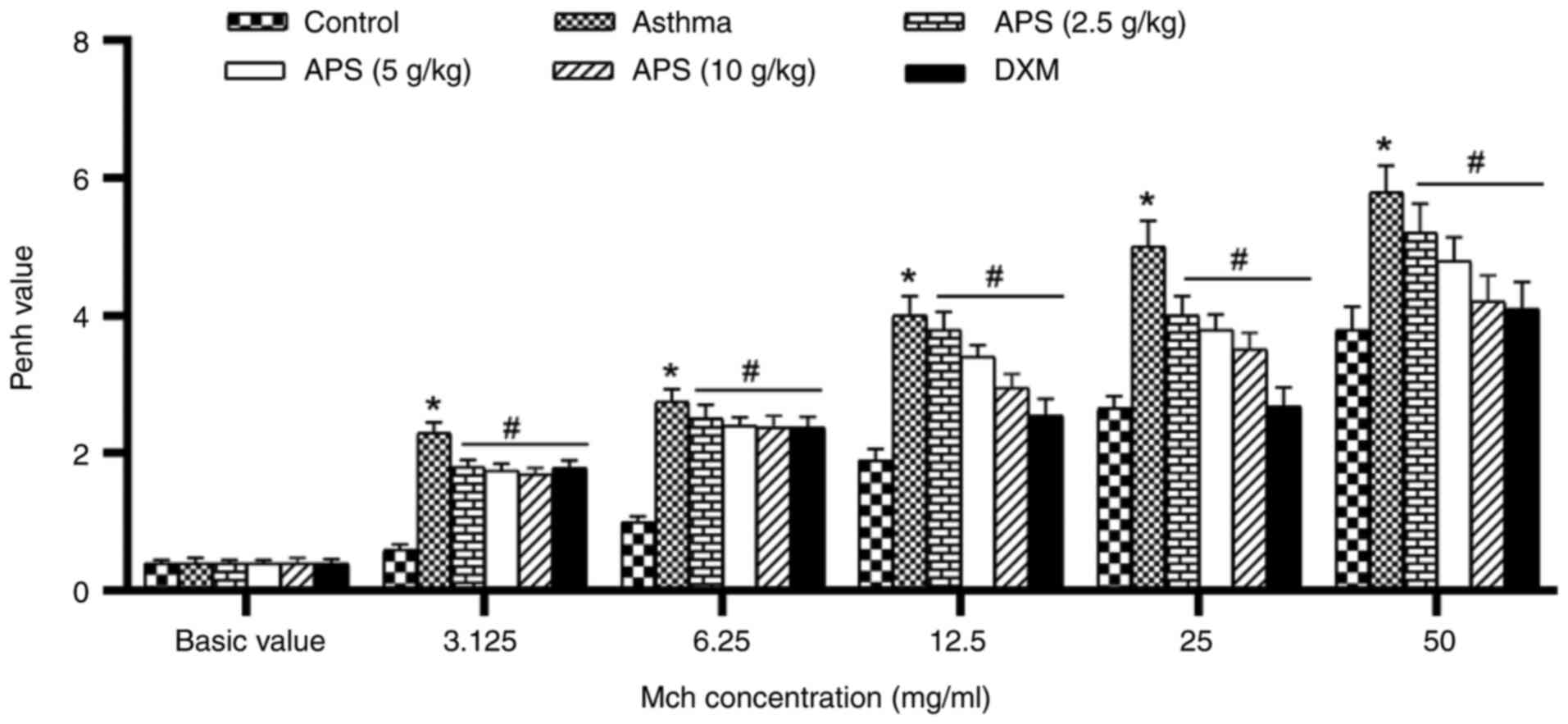
Effects of APS on alleviating AHR in
the bodies of mice
With the purpose of evaluating the effects of APS on
AHR, the Penh values induced with various doses of Mch (3.125,
6.25, 12.5, 25 and 50 mg/ml) were measured. The AHR level in each
group increased with the increase in the inhaled Mch concentration.
The AHR in the asthma group was markedly higher than that in the
normal control group following induction with Mch (P<0.05).
However, the Penh value decreased following treatment with APS and
DXM (P<0.05; Fig. 2).
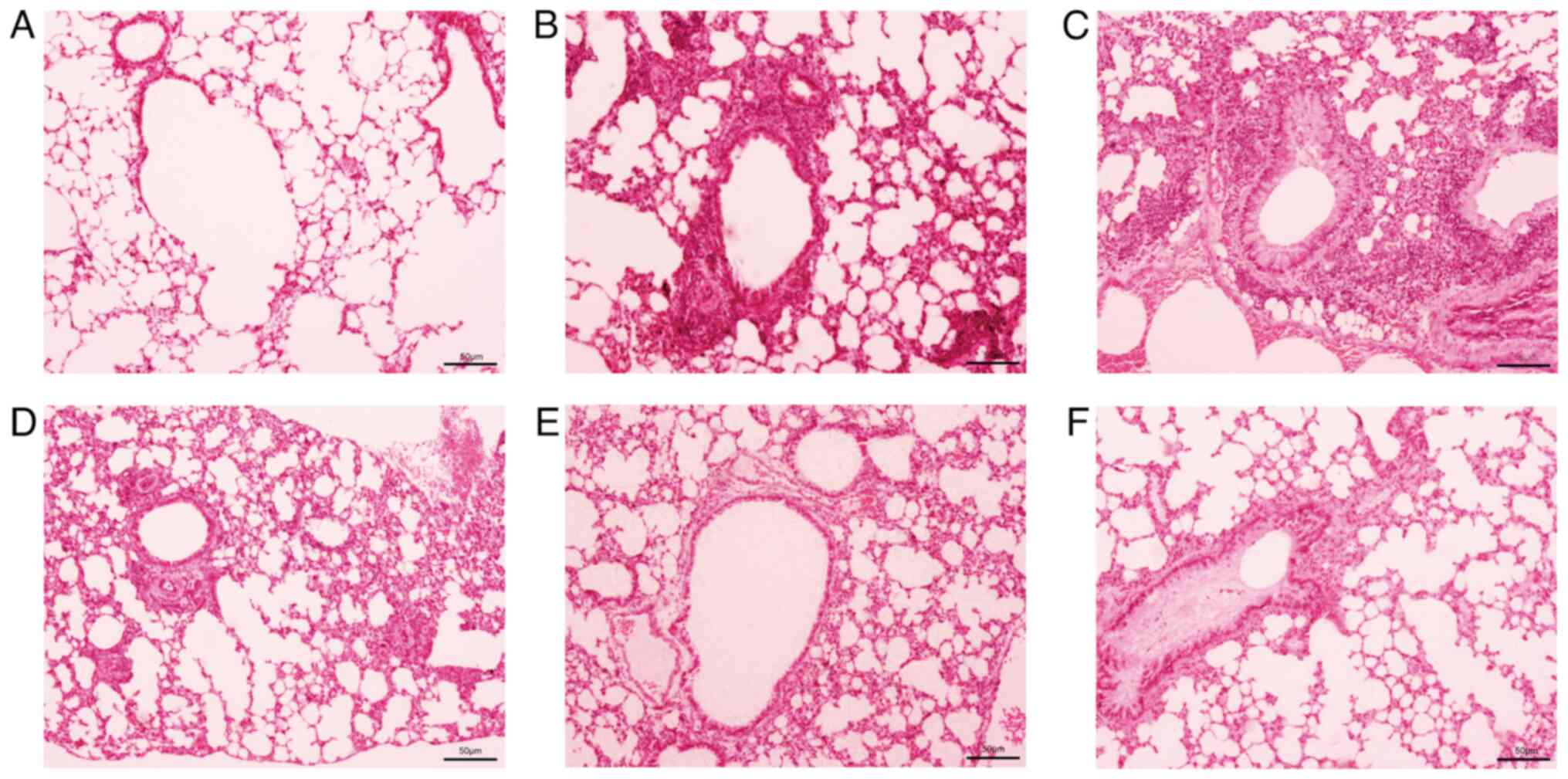
Ameliorative effects of APS on airway
inflammation and eosinophil infiltration in asthmatic mice
The H&E staining of mouse lung tissue revealed a
large amount of inflammatory cell infiltration in the peribronchial
area, such as eosinophils, neutrophils and lymphocytes (Fig. 3), with eosinophils being more
prominent. Following treatment with APS, inflammatory cell
infiltration markedly declined.
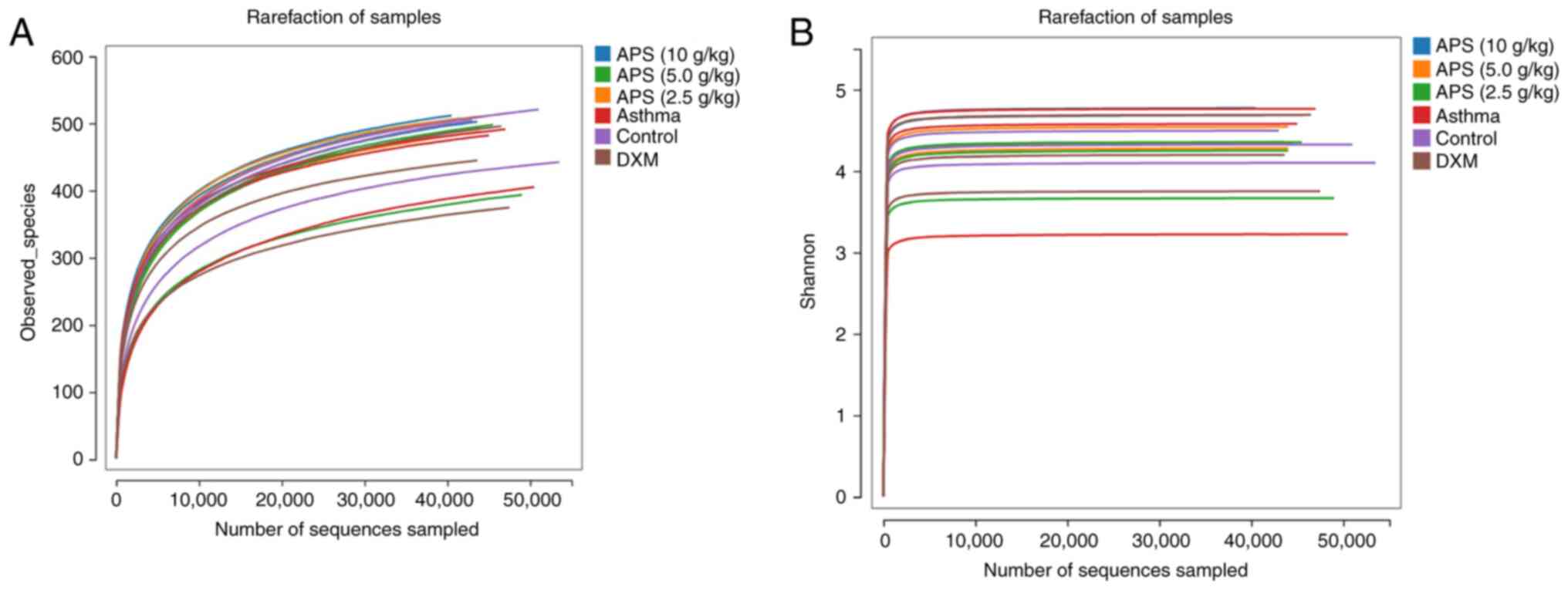
Effects of APS on gut microbiota in
asthmatic mice
In the present study, the rarefaction curve tended
to be flat, suggesting the reasonable sequencing data volume, which
indirectly reflected the species richness. The Shannon curve also
tended to be flat, suggesting a sufficiently large sequencing data
volume (Fig. 4).
Alpha diversity of the gut microbiota
in the different groups
In the present study, the sequencing depth index
coverage value of each sample was >0.996, suggesting that the
experimental data could truly reflect the microbial communities of
the experimental samples. Alpha diversity indicates the species
evenness and richness, mainly including the ACE, Chao, Shannon and
Simpson indexes. As shown in Table
I, compared with the control group, the OTU number of gut
microbiota in the asthma group was markedly decreased (P<0.05),
and the ACE, Chao and Shannon indexes exhibited a significant
decrease (P<0.05). Compared with the asthma group, the OTU
numbers in the APS (5.0 and 10 g/kg) groups were notably increased
(P<0.05), and the ACE, Chao and Shannon indexes were also
significantly elevated (P<0.05), which tended to be similar to
the levels in the control group. These results suggested a marked
difference in the gut microbiota between the asthmatic and normal
mice, following medium- and high-dose APS intervention. In
addition, the OTU numbers and alpha diversity indexes in the gut
microbiota of the asthmatic mice were increased, and the microbial
community abundances were recovered.
 | Table IAlpha diversity index comparison based
on the total number of OTUs. |
Table I
Alpha diversity index comparison based
on the total number of OTUs.
| Group | No. of mice | OTU | Chao | ACE | Shannon | Simpson |
|---|
| Control | 6 | 489.67±22.64 | 542.45±21.25 | 539.83±19.76 | 4.31±0.06 | 0.04±0.008 |
| Asthma | 6 |
460.33±12.59a |
518.34±15.05a |
510.94±18.07a |
4.19±0.087a | 0.05±0.019 |
| APS (2.5 g/kg) | 6 |
461.67±47.89b |
512.38±42.64b |
512.45±44.79b |
4.09±0.37b | 0.05±0.017 |
| APS (5.0 g/kg) | 6 |
503.33±10.84b |
554.75±14.85b |
547.89±9.91b |
4.39±0.11b | 0.04±0.006 |
| APS (10 g/kg) | 6 |
507.67±3.68b |
554.33±4.92b |
555.96±3.92b |
4.61±0.18b | 0.05±0.008 |
| DXM | 6 |
438.67±49.60b |
486.02±45.53b |
484.06±47.99b |
4.21±0.38b | 0.04±0.019 |
Beta diversity analysis of the gut
microbiota in each group
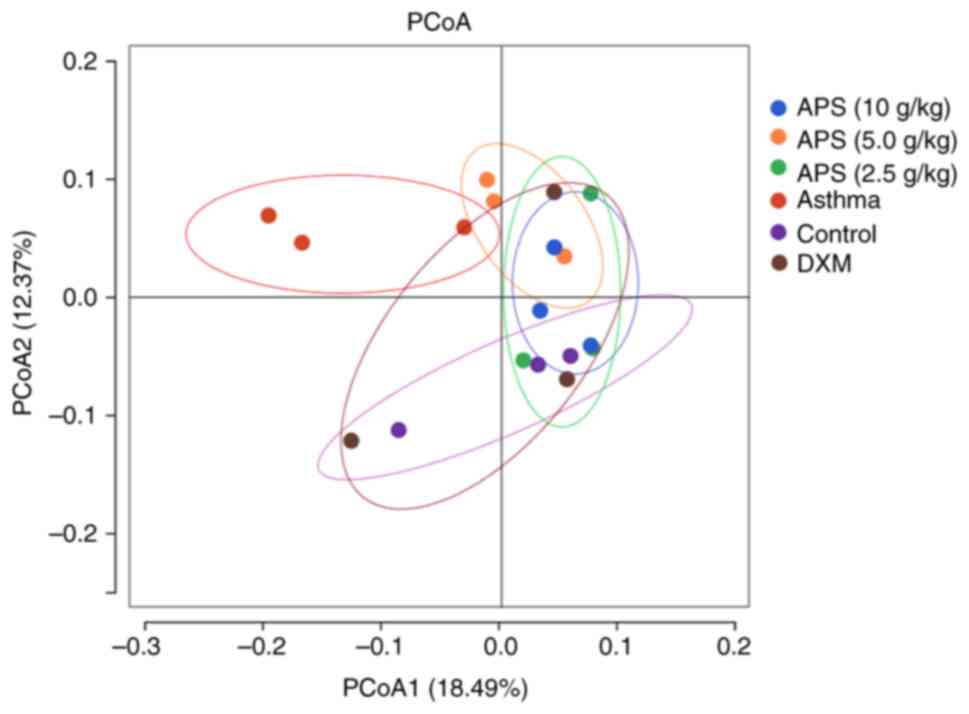
As indicated by principal coordinates analysis
(PCoA), the sample distance was the farthest between the model and
control group, and these two groups were well distinguished,
revealing the obvious difference in community structure between the
two groups (Fig. 5). In other
words, the gut microbial communities in the asthmatic mice were
markedly altered. By contrast, the sample distribution in the
high-dose APS group gradually approached the level in the normal
group, and exhibited intersection with the latter. In particular,
the high-dose APS group was the closest to the normal group,
suggesting that APS regulated the gut microbial communities in
asthmatic mice and alleviated asthma-induced dysbacteriosis.
Gut microbial species composition and
differential analysis in each group. Analysis of gut microbial
species structure at the phylum level
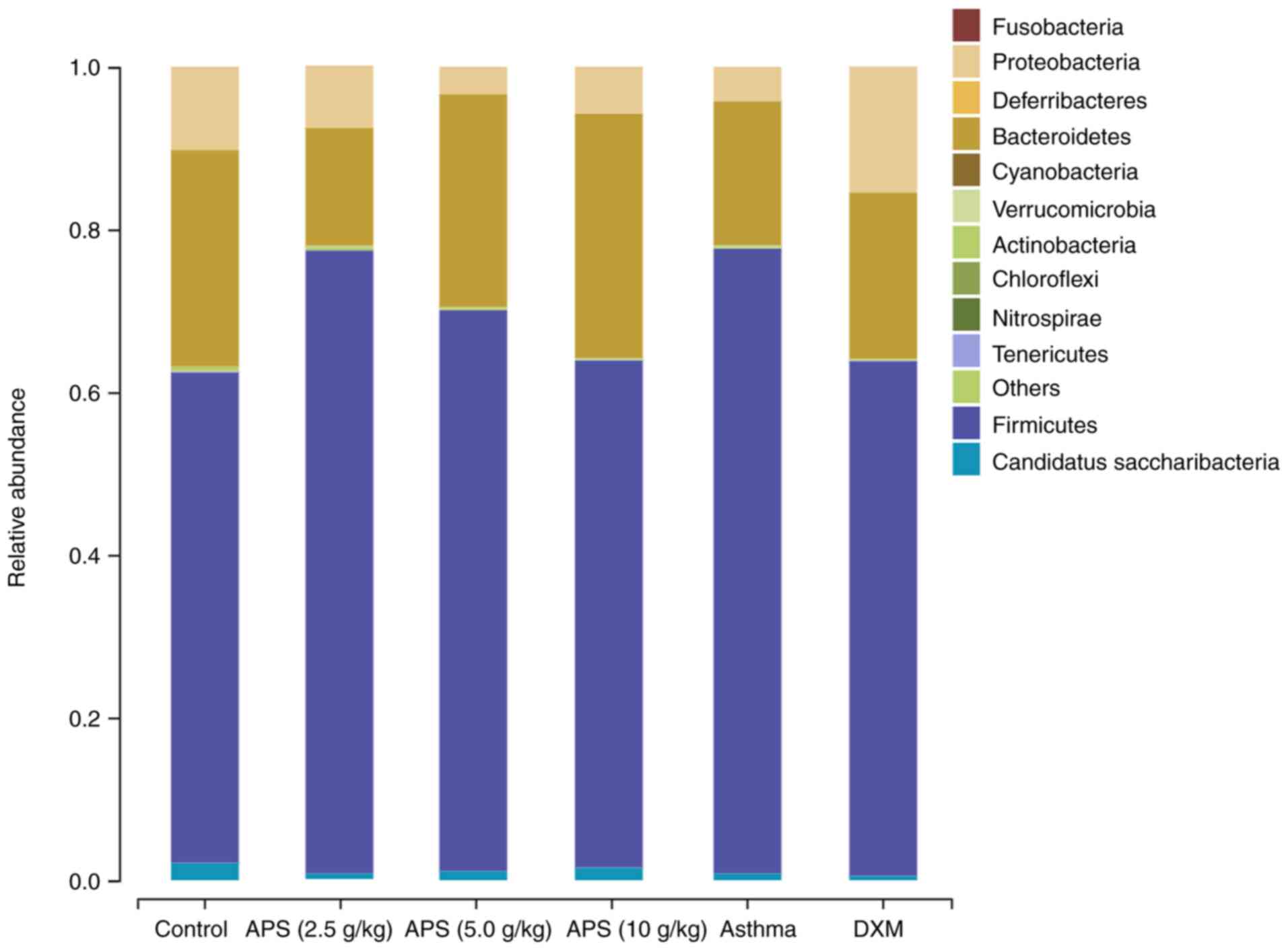
As illustrated in Fig.
6, at the phylum level, the gut microbiota in the mice
primarily consisted of Firmicutes, Proteobacteria, Bacteroidetes
and Actinoidetes, among which, Firmicutes and Bacteroidetes were
the dominant bacterial communities. Compared with the control
group, the relative abundance of Firmicutes in the model group was
elevated by 16.5%; compared with the model group, the relative
abundances of Firmicutes in the three APS groups (2.5, 5.0 and 10
g/kg) decreased by 0.21, 7.9 and 14.5%, respectively. Compared with
the normal control group, the relative abundance of Bacteroidetes
was reduced by 8.9% in the model group. Compared with the model
group, the relative abundance of Bacteroidetes in the APS (2.5
g/kg) group decreased by 3.2%, while the abundance in the other APS
(5.0 and 10 g/kg) groups was elevated by 8.4 and 12.3%,
respectively. The Firmicutes/Bacteroidetes (F/B) ratio is usually
used as a marker to evaluate gut microbial disturbance (7). In asthmatic mice, the gut microbial
imbalance mainly manifested as a low abundance of Bacteroidetes and
a high abundance of Firmicutes, yielding an increased F/B ratio.
When compared with the control group, the F/B ratio in the model
group tended to significantly increase. Compared with the model
group, the F/B ratios in the APS (5.0 and 10 g/kg) groups exhibited
a decreasing trend. In the high-dose APS group, the abundance of
Firmicutes decreased, and that of Bacteroidetes increased, thereby
decreasing the F/B ratio and correcting the gut microbial imbalance
(Table SI).
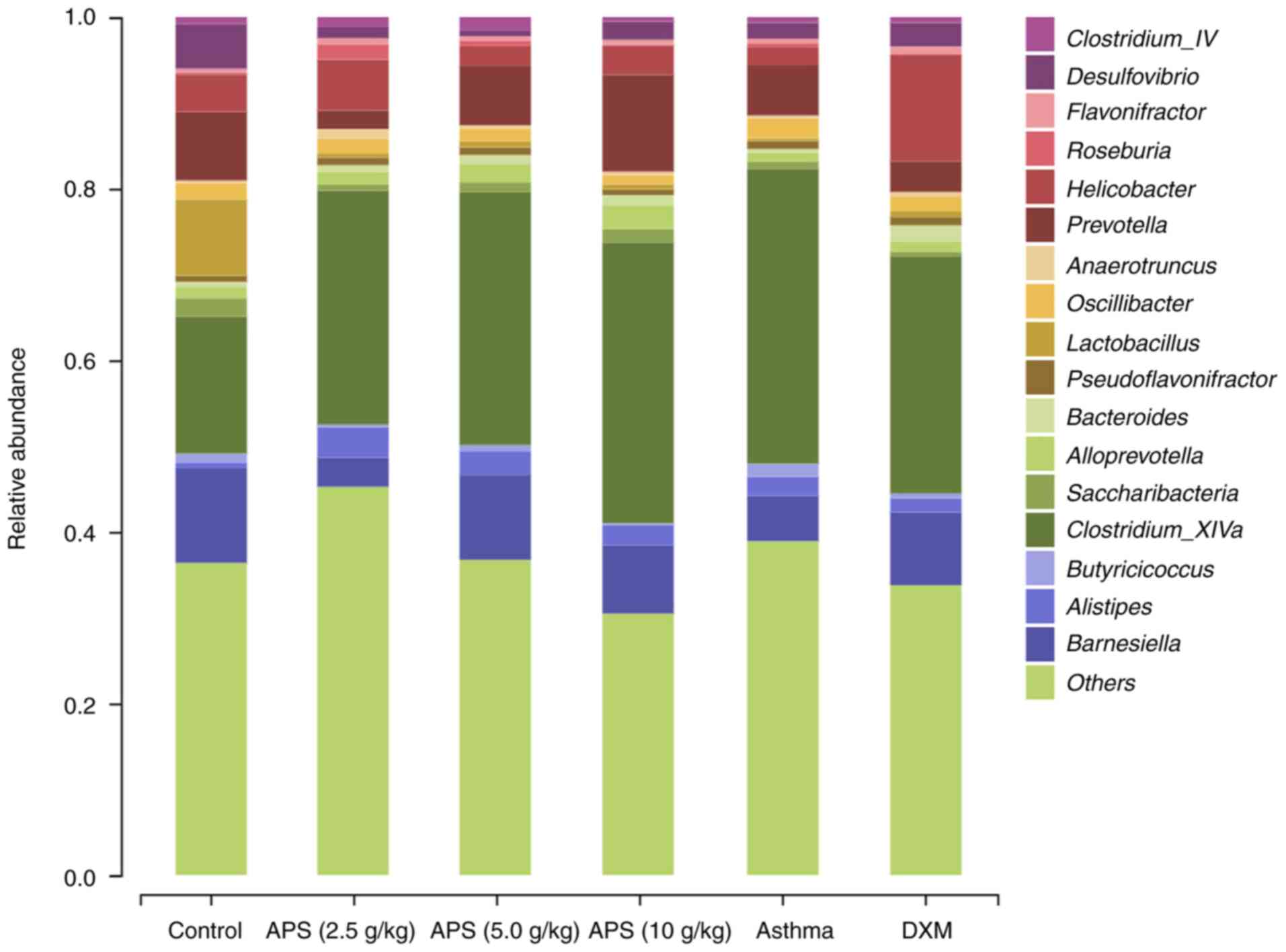
Analysis of gut microbial species structure at
the genus level. As demonstrated in Fig. 7, compared with the normal control
group, the relative abundances of Lactobacillus,
Prevotella, Helicobacter and Barnesiella were
decreased in the model group, while those of Alistipes and
Clostridium_XlVa were increased. Compared with the model
group, the abundances of Lactobacillus, Barnesiella
and Prevotella in the fecal samples of mice in the APS (5.0
and 10 g/kg) groups were increased, while the abundance of
Clostridium_XlVa was decreased (Table SII). These findings indicated the
positive regulatory effects of APS on gut microbial communities in
asthmatic mice at the genus level. More specifically, APS increased
the abundances of probiotics and suppressed the growth of
pernicious bacteria.
Serum metabolomics analysis in
asthmatic mice treated with APS. Differential metabolites in mouse
serum samples
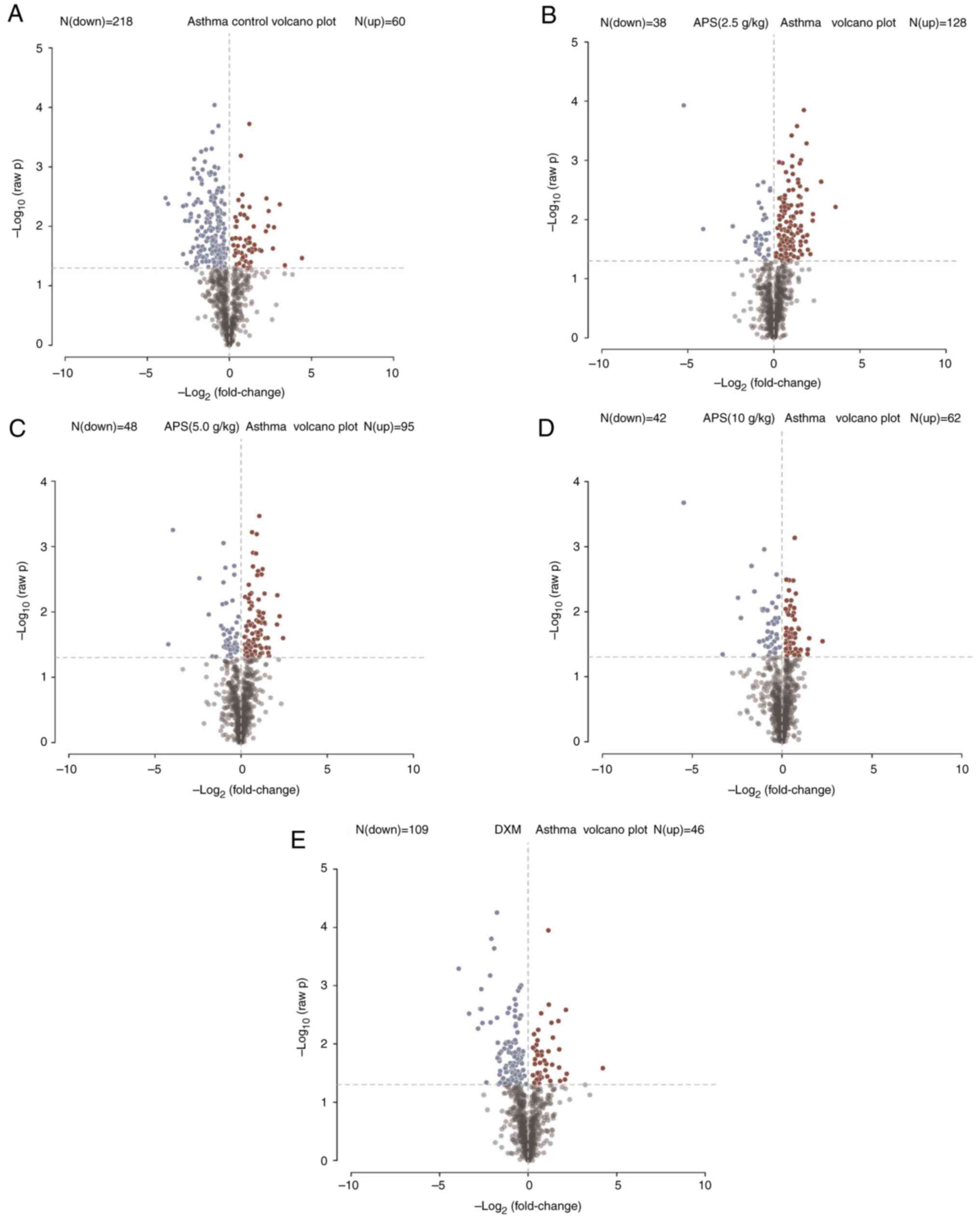
As shown in Fig. 8,
in the partial least squares-discriminant analysis (PLS-DA) model,
the model group was notably distinguished from the other groups,
indicating that the occurrence of asthma and the interventions of
low-, medium- and high-dose APS treatment markedly altered the
intestinal metabolite concentrations in the mice. Combined with
fold change (FC) analysis and the t-test, the significance in the
changes of metabolites between two samples was analyzed and
visualized in the form of a volcano plot (Fig. 8). In the volcano plot, red and blue
points indicated metabolites of FC>1.5 and P<0.05, namely,
the differential metabolites between two groups, with red points
indicating significantly upregulated, whereas blue points
represented significantly downregulated metabolites. In the present
study, significantly up- and downregulated differential metabolites
were found between the model and control groups (Fig. 8A), the model group and low-dose APS
group (Fig. 8B), the model group
and medium-dose APS group (Fig.
8C), the model group and high-dose APS group (Fig. 8D), as well as between the model
group and DXM group (Fig. 8E).
Further hierarchical clustering suggested that there were 145
differential metabolites between the model group and APS (5.0 g/kg)
group, including the significantly upregulated
3-hydroxyhexanoylcarnitine, N-palmitolenoyltaurine,
1-palmitoleoylglycero and 3-hydroxybutyrylcarnitine, as well as the
significantly downregulated glycylvaline, S-adenosylhomocysteine
and 5-methyl-2'-deoxycytidine (Fig.
9A).
Upon further hierarchical clustering, 105
significant differential metabolites were detected between the
model group and APS (10 g/kg) group, including the significantly
upregulated cholate, 7-ketodeoxycholate and ursocholate, together
with the significantly downregulated glycitein sulfate, pinitol,
glycylvaline, dihomo-linolenoyl-choline, catechol glucuronide,
3-(3-hydroxyphenyl)propionate sulfate and ursodeoxycholate. These
results revealed that APS could alter the blood metabolites in mice
(Fig. 9B).
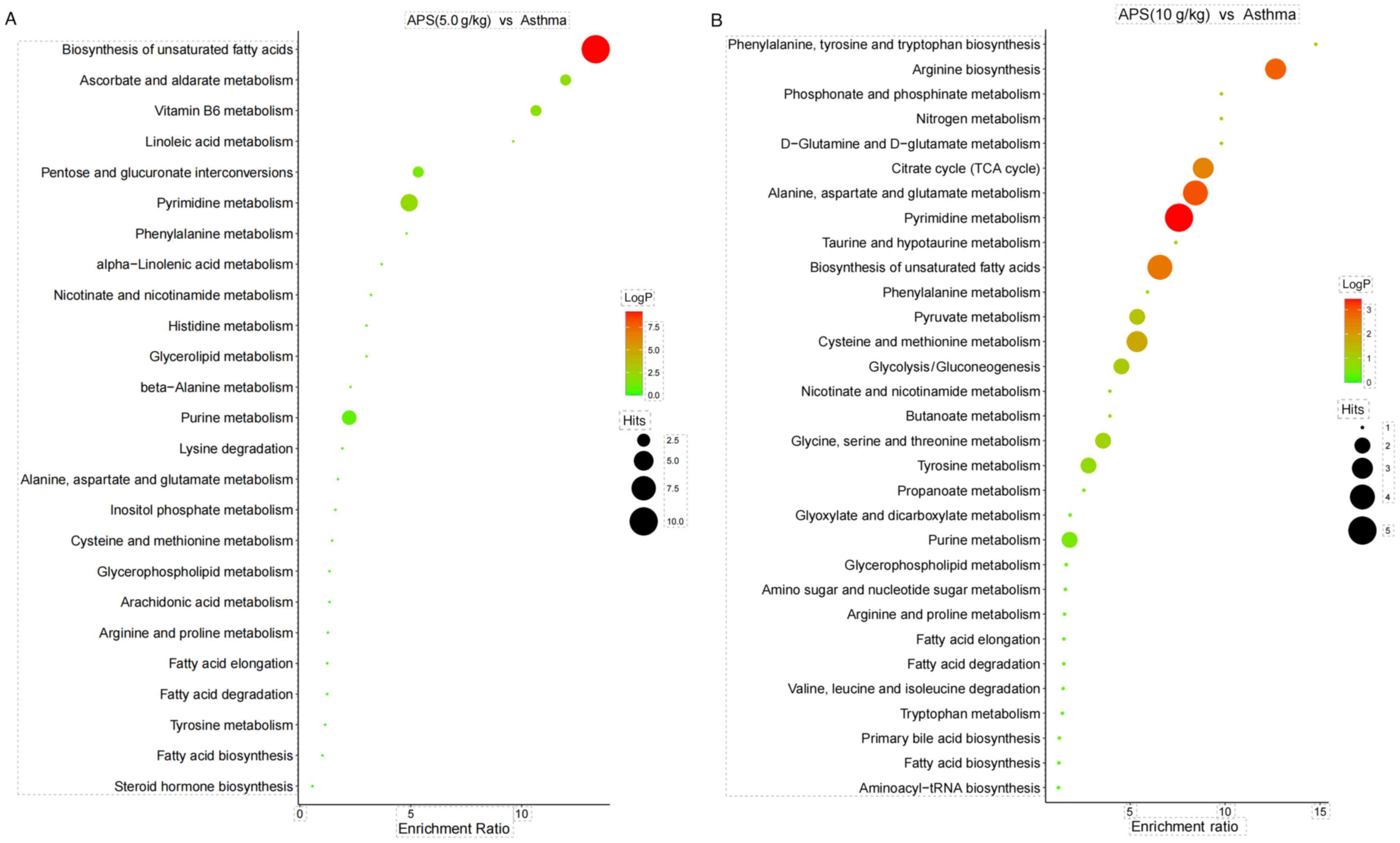
Analysis of related metabolic pathways. As
presented in Fig. 10, KEGG pathway
enrichment analysis suggested that the most significantly affected
metabolic pathways in the model group and APS (5.0 g/kg) group were
Biosynthesis of unsaturated fatty acids and Arginine biosynthesis
(Fig. 10A), while those in the
model group and APS (10 g/kg) group were Biosynthesis of
unsaturated fatty acids and Pyrimidine metabolism (Fig. 10B).
Discussion
Based on the TCM theories of ‘the lung and the large
intestine being interior-exteriorly related and reinforcing earth
to generate metal’, modern medicine has put forth the concept of
the ‘lung-intestine’ axis. This term refers to the microbial
communities that colonize these two organs and serve as the link to
form a bidirectional axis that connects the lung and the intestine.
The gut microbiota influences the occurrence and development of
lung diseases. In addition, microbial disturbance induced by lung
diseases, particularly allergic disease, affects the
gastrointestinal tract through immunoregulation (8). The gut microbiota is considered the
largest immune organ in the human body, which involves ~1,000
resident bacteria, a figure that is 10-fold that in normal human
cells. Moreover, it contains genes in numbers that are 150-fold
higher than those in the human body; the total microorganism amount
in the gut microbiota reaches 1014, and is thereby known
as the second largest human genome. Gut microbial diversity is the
foundation for the promotion of nutrient absorption, and
maintaining body immunity and metabolism. Moreover, the changes in
microbial community structure, species and functions, and the
produced metabolites play a crucial role in improving asthma
(9,10).
The gut microbiota participates in the occurrence
and progression of allergic disease. A previous study collected the
urine and feces samples from 319 infants at the ages of 3 months
and 1 year, and recorded their health condition at the ages of 1, 3
and 5 years. In addition, that study detected the gut microbial
levels in the samples through high-throughput gene sequencing and
discovered that infants lacking four gut microbial communities
exhibited early asthma symptoms when they were 1 year old (11,12).
Regulating the gut microbial composition may become an effective
method for the prevention or treatment of asthma. The application
of antibiotics induces alterations in the gut microbiota, finally
increasing Th2 cell-induced asthma. Consequently, maintaining a
certain amount or certain species of gut microbial communities
plays an indispensable role in the effects of regulatory T-cells on
preventing hypersensitivity (13).
The substantial role of probiotics in the treatment of allergic
inflammatory disease has been verified. Probiotics can colonize the
intestine and affect lung immune function by affecting the lung
microbial communities via the lung-intestine axis; therefore, this
has a certain therapeutic effect on asthma (14,15).
However, some studies have demonstrated that probiotics are
ineffective (14,15). Thus, it may not be practical to
adjust the gut microbiota composition using probiotic preparations
alone. Research has indicated that increasing the Bacteroidetes
abundance can decompose the carbohydrates in plants into prebiotics
to alleviate inflammation. The decrease in the F/B ratio directly
affects the metabolism of dietary fiber by gut microbiota, and
enhances the concentration of short-chain fatty acids (SCFAs).
SCFAs can induce the production and differentiation of regulatory
T-cells in the intestine; as a result, they stimulate the
generation of the anti-inflammatory IL-10 level and exert a
critical regulatory effect on the asthma-related metabolic
disturbance via anti-inflammatory effects (16-18).
In the present study, the gut microbial abundance, evenness and
diversity in asthmatic mice was markedly decreased, verifying that
the occurrence and development of asthma in mice were accompanied
by gut microbial alteration. Medium- and high-dose APS intervention
increased the OTU number and alpha diversity indexes of the gut
microbiota in mice, and restored the microbial communities,
suggesting that APS improved the gut microbial imbalance in
asthmatic mice. At the phylum level, medium- and high-dose APS
reduced the relative abundance of Firmicutes, increased that of
Bacteroidetes, reduced the F/B ratio, and corrected the alterations
in the gut microbiota. Moreover, APS increased the probiotics
quantities (such as Lactobacillus, Barnesiella and
Prevotella), suppressed the growth of pernicious bacteria
(such as Clostridium_XlVa) and exerted certain protective
effects on the intestinal mucosal barrier. Thus, it can by
hypothesized that APS plays a key regulatory role in the
composition of the gut microbiota, which can increase the growth of
probiotics, suppress pernicious bacterial growth, and thereby
restrain the chronic inflammation. APS can regulate the gut
microbiota, thereby increasing the content of SCFA contents, which
can be absorbed directly into the blood through the intestine; APS
can also regulate the changes in serum metabolic pathways caused by
asthma, including biosynthesis of unsaturated fatty acids.
The concept of metabolomics was first proposed by
English scholars in 1999, which is another emerging and rapidly
developing bioscience after genomics and proteomics. It has a
tremendous application potential in illustrating the pathogenic
mechanism in the body and identifying biomarkers for diseases
(19,20). Metabolomics can analyze the changes
in endogenous small molecular compounds (relative molecular weight
<1,000), which exist in body fluids (blood and urine), cells and
tissue, at the cellular level within a short period of time
(21,22). Compared with genomics and
proteomics, the species of metabolites are far less than genes and
proteins, and thus less data need to be processed. Metabolites are
the final downstream products of transcription and translation,
which are considered as the very terminal of bioinformation
transfer. It is considered that ‘genomics and proteomics tell you
what may occur, while metabolomics tell you what actually happens’
(23). According to the findings of
the present study, the regulatory effects of APS on the metabolites
of asthmatic mice mainly focused on the Biosynthesis of unsaturated
fatty acids, the Biosynthesis of arginine and Pyrimidine
metabolism. However, certain limitations should be noted in the
present study. The experimental sample size was small, and thus,
larger-scale experimental samples are required in the future to
verify and explain the association between APS and gut microbiota,
as well as metabolites. In addition, the precise mechanism of APS
in affecting the gut microbiota and whether APS induces
pathophysiological changes in other systems remain to be further
explored, which is a future research direction.
APS, which has long been applied as a critical
ingredient derived from Astragalus and a nutraceutical,
exerts different pharmacological effects on the respiratory system.
Its anti-inflammatory effects are mediated through numerous
signaling pathways (24). A
previous study demonstrated that treatment with APS decreased the
sneezing and rubbing times of guinea pigs with allergic rhinitis
(AR) and hindered the OVA-sIgE, OVA-sIgG1, TNF-α and IL-6 levels in
guinea pig serum; it simultaneously elevated the
CD25+Foxp3+Treg cell proportion, whereas it
decreased the proportion of CD4+IL17+Th17
cells in serum or tissues of guinea pigs with AR, in a
dose-dependent manner (25). NF-κB
was highly expressed in guinea pigs with AR and its expression was
decreased following treatment with APS. NF-κB overexpression
stimulated inflammatory responses and Treg/Th17 imbalance in AR
guinea pigs. APS decreased the Treg/Th17 imbalance by hindering
NF-κB expression, which thus ameliorated the inflammatory responses
in guinea pigs with AR (25).
In conclusion, the present study explored the
mechanisms of action of APS in improving the immunity of asthmatic
mice on the basis of the gut microbiota combined with metabolomics.
According to the findings of the present study, APS not only
improved the gut microbial imbalance in asthmatic mice, but also
exerted its effect by regulating the biosynthesis of unsaturated
fatty acids, the biosynthesis of arginine and the
pyrimidine-related metabolites. Moreover, the obtained results
provide the theoretical foundation for the reasonable application
of APS in the clinical treatment of asthma.
Supplementary Material
Firmicutes/Bacteroidetes ratio.
Analysis of gut microbial species
structure at the genus level.
Acknowledgements
Not applicable.
Funding
Funding: The present study was funded by grants from the Major
Project of Hangzhou Medical Health Science and Technology (grant
no. Z20220105) and the Medical Science and Technology Program of
Zhejiang Province (grant nos. 2023RC246,2022KY1009 and 2022RC221),
Hangzhou Biomedical and Health Industry Development Support Science
and Technology Special Project (no. 2023WJC200), the Science and
Technology Development Plan Project of Hangzhou (grant no.
20201203B205).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request. All sequences used in the present study are publicly
available at the NCBI Sequence Read Archive (https://www.ncbi.nlm.nih.gov/bioproject/PRJNA1162806)
under the Accession ID PRJNA1162806. The metabolomics data reported
in the present study have been deposited in the OMIX, China
National Center for Bioinformation/Beijing Institute of Genomics,
Chinese Academy of Sciences (https://ngdc.cncb.ac.cn/omix/release/OMIX007407).
Authors' contributions
JZ and MG designed the experiments. JZ, MG, WS and
SW performed the experiments. MG and JZ analyzed the data. JZ and
MG wrote the manuscript. All authors have read and approved the
final manuscript, JZ and WS confirm the authenticity of all the raw
data.
Ethics approval and consent to
participate
All experimental protocols involving animals were
approved by the Ethics Committee of the Zhejiang Center of
Laboratory Animals (Hangzhou, China; approval no.
ZJCLA-IACUC-20020187) and conducted conforming to the guidelines of
the China Animal Protection Commission.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Sun J, Bai S, Zhao J, Li D, Ma X, Ma L and
Su X: Mapping knowledge structure and research of the biologic
treatment of asthma: A bibliometric study. Front Immunol.
14(1034755)2023.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Agusti A, Fabbri L, Lahousse L, Singh D
and Papi A: Single inhaler triple therapy (SITT) in asthma:
Systematic review and practice implications. Allergy. 77:1105–1113.
2022.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Chan HL and Ng T: Traditional Chinese
Medicine (TCM) and allergic diseases. Curr Allergy Asthma Rep.
20(67)2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Yang N and Shang YX: Epigallocatechin
gallate ameliorates airway inflammation by regulating Treg/Th17
imbalance in an asthmatic mouse model. Int Immunopharmacol.
72:422–428. 2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Zheng Y, Ren W, Zhang L, Zhang Y, Liu D
and Liu Y: A review of the pharmacological action of
Astragalus polysaccharide. Front Pharmacol.
11(349)2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Zhao X, Hu M, Zhou H, Yang Y, Shen S, You
Y and Xue Z: The role of gut microbiome in the complex relationship
between respiratory tract infection and asthma. Front Microbiol.
14(1219942)2023.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Chen J, Wang A and Wang Q: Dysbiosis of
the gut microbiome is a risk factor for osteoarthritis in older
female adults: A case control study. BMC bioinformatics.
22(299)2021.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Zhang D, Li S, Wang N, Tan HY, Zhang Z and
Feng Y: The Cross-Talk Between Gut microbiota and lungs in common
lung diseases. Front Microbiol. 11(301)2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Hansen AW and Venkatachalam KV:
Sulfur-Element containing metabolic pathways in human health and
crosstalk with the microbiome. Biochem Biophys Rep.
35(101529)2023.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Brown HN, Barber T, Renshaw D, Farnaud S,
Oduro-Donkor D and Turner MC: Associations between the gut
microbiome and metabolic, inflammatory, and appetitive effects of
sleeve gastrectomy. Obes Rev. 24(e13600)2023.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Arrieta MC, Stiemsma LT, Dimitriu PA,
Thorson L, Russell S, Yurist-Doutsch S, Kuzeljevic B, Gold MJ,
Britton HM, Lefebvre DL, et al: Early infancy microbial and
metabolic alterations affect risk of childhood asthma. Sci Transl
Med. 7(307ra152)2015.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Li R, Guo Q, Zhao J, Kang W, Lu R, Long Z,
Huang L, Chen Y, Zhao A, Wu J, et al: Assessing causal
relationships between gut microbiota and asthma: evidence from two
sample Mendelian randomization analysis. Front Immunol.
14(1148684)2023.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Luo X, Pan Z, Luo S, Liu Q, Huang S, Yang
G, Nong F, Fu Y, Deng X and Zhou L: Effects of ceftriaxone-induced
intestinal dysbacteriosis on regulatory T cells validated by
anaphylactic mice. Int Immunopharmacol. 60:221–227. 2018.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Loo EX, Llanora GV, Lu Q, Aw MM, Lee BW
and Shek LP: Supplementation with probiotics in the first 6 months
of life did not protect against eczema and allergy in at-risk Asian
infants: a 5-year follow-up. Int Arch Allergy Immunol. 163:25–28.
2014.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Wang F, Wu F, Chen H and Tang B: The
effect of probiotics in the prevention of atopic dermatitis in
children: A systematic review and meta-analysis. Transl Pediatr.
12:731–748. 2023.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Cavalcanti RFP, Gadelha FAAF, Paiva
Ferreira LKD, Paiva Ferreira LAM, Chaves Júnior JV, de Araújo
Batista RS, Melo TBL, de Souza FS, Alves AF, Maria Batista L and
Piuvezam MR: Limosilactobacillus fermentum modulates the gut-airway
axis by improving the immune response through FOXP3 activation on
combined allergic rhinitis and asthma syndrome (CARAS).
Immunobiology. 228(152721)2023.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Dou M, Chu Y, Zhou X, Wang M, Li X, Ma R,
Fan Z, Zhao X, Wang W, Li S, et al: Matrine mediated immune
protection in MS by regulating gut microbiota and production of
SCFAs. Mol Neurobiol. 61:74–90. 2024.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Hou Q, Huang J, Zhao L, Pan X, Liao C,
Jiang Q, Lei J, Guo F, Cui J, Guo Y and Zhang B: Dietary genistein
increases microbiota-derived short chain fatty acid levels,
modulates homeostasis of the aging gut, and extends healthspan and
lifespan. Pharmacol Res. 188(106676)2023.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Tweeddale H, Notley-McRobb L and Ferenci
T: Assessing the effect of reactive oxygen species on Escherichia
coli using a metabolome approach. Redox Rep. 4:237–241.
1999.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Shah SH, Kraus WE and Newgard CB:
Metabolomic profiling for the identification of novel biomarkers
and mechanisms related to common cardiovascular diseases: Form and
function. Circulation. 126:1110–1120. 2012.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Robinson O and Lau CE: How do metabolic
processes age: Evidence from human metabolomic studies. Curr Opin
Chem Biol. 76(102360)2023.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Yao Y, Wang X, Guan J, Xie C, Zhang H,
Yang J, Luo Y, Chen L, Zhao M, Huo B, et al: Metabolomic
differentiation of benign vs malignant pulmonary nodules with high
specificity via high-resolution mass spectrometry analysis of
patient sera. Nat Commun. 14(2339)2023.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Bjerrum JT, Nielsen OH, Wang YL and Olsen
J: Technology insight: metabonomics in gastroenterology-basic
principles and potential clinical applications. Nat Clin Pract
Gastroenterol Hepatol. 5:332–343. 2008.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Xu J, Zhang Q, Li Z, Gao Y, Pang Z, Wu Y,
Li G, Lu D, Zhang L and Li D: Astragalus polysaccharides
attenuate ovalbumin-induced allergic rhinitis in rats by inhibiting
NLRP3 inflammasome activation and NOD2-Mediated NF-κB activation. J
Med Food. 24:1–9. 2021.PubMed/NCBI View Article : Google Scholar
|
|
25
|
He X, Liu L, Luo X, Zhu J, Yang H, Wang J,
Chen L and Zhong L: Astragalus polysaccharide relieves
inflammatory responses in guinea pigs with allergic rhinitis via
ameliorating NF-kB-Mediated Treg/Th17 Imbalance. Am J Rhinol
Allergy. 36:638–648. 2022.PubMed/NCBI View Article : Google Scholar
|