|
1
|
Vincze M and Danko K: Idiopathic
inflammatory myopathies. Best Pract Res Clin Rheumatol. 26:25–45.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Dimachkie MM and Barohn RJ: Idiopathic
inflammatory myopathies. Semin Neurol. 32:227–236. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Cox S, Limaye V, Hill C, Blumbergs P and
Roberts-Thomson P: Idiopathic inflammatory myopathies: Diagnostic
criteria, classification and epidemiological features. Int J Rheum
Dis. 13:117–124. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Szodoray P, Alex P, Knowlton N, Centola M,
Dozmorov I, Csipo I, Nagy AT, Constantin T, Ponyi A, Nakken B and
Danko K: Idiopathic inflammatory myopathies, signified by
distinctive peripheral cytokines, chemokines and the TNF family
members B-cell activating factor and a proliferation inducing
ligand. Rheumatology (Oxford). 49:1867–1877. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Gazeley DJ and Cronin ME: Diagnosis and
treatment of the idiopathic inflammatory myopathies. Ther Adv
Musculoskelet Dis. 3:315–324. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
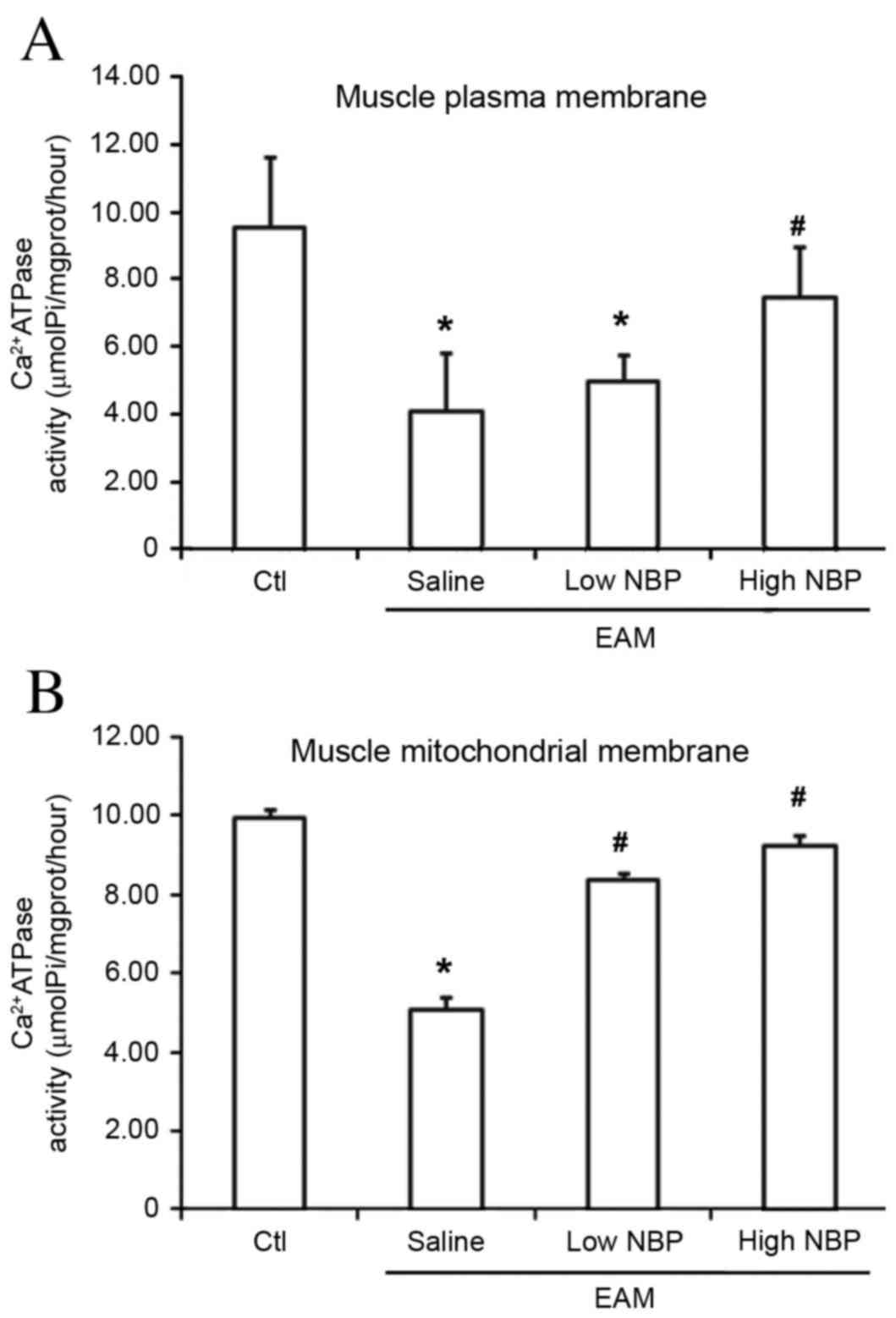
|
Carstens PO and Schmidt J: Diagnosis,
pathogenesis and treatment of myositis: Recent advances. Clin Exp
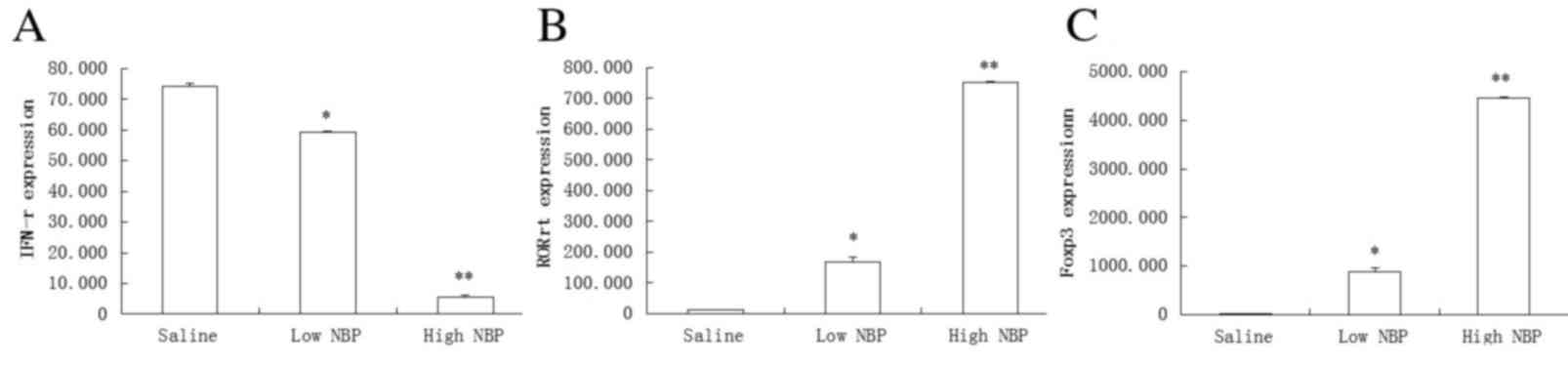
Immunol. 175:349–358. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Bodoki L, Vincze M, Griger Z and Dankó K:
Intravenous immunoglobulin treatment in idiopathic inflammatory
myopathy. Orv Hetil. 154:723–728. 2013.(In Hungarian). View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Mathur T, Manadan AM, Thiagarajan S, Hota
B and Block JA: Corticosteroid monotherapy is usually insufficient
treatment for idiopathic inflammatory myopathy. Am J Ther.
22:350–354. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wilson CW III: Relative recovery and
identification of carbonyl compounds from celery essential oil. J
Food Sci. 35:766–768. 1970. View Article : Google Scholar
|
|
10
|
Zhang T, Yan W, Li Q, Fu J, Liu K, Jia W,
Sun X and Liu X: 3-n-Butylphthalide (NBP) attenuated neuronal
autophagy and amyloid-β expression in diabetic mice subjected to
brain ischemia. Nerol Res. 33:396–404. 2011. View Article : Google Scholar
|
|
11
|
Feng X, Peng Y, Liu M and Cui L:
DL-3-n-butylphthalide extends survival by attenuating glial
activation in a mouse model of amyotrophic lateral sclerosis.
Neuropharmacology. 62:1004–1010. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Xu HL and Feng YP: Inhibitory effects of
chiral 3-n-butylphthalide on inflammation following focal ischemic
brain injury in rats. Acta Pharmacol Sin. 21:433–438.
2000.PubMed/NCBI
|
|
13
|
Wang HM, Zhang T, Huang JK and Sun XJ:
3-N-butylphthalide (NBP) attenuates the amyloid-β-induced
inflammatory responses in cultured astrocytes via the nuclear
factor-κB signaling pathway. Cell Physiol Biochem. 32:235–242.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Allenbach Y, Solly S, Grégoire S, Dubourg
O, Salomon B, Butler-Browne G, Musset L, Herson S, Klatzmann D and
Benveniste O: Role of regulatory T cells in a new mouse model of
experimental autoimmune myositis. Am J Pathol. 174:989–998. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang YG, Li Y, Wang CY, Ai JW, Dong XY,
Huang HY, Feng ZY, Pan YM, Lin Y, Wang BX and Yao LL:
L-3-n-Butylphthalide protects rats' cardiomyocytes from
ischaemia/reperfusion-induced apoptosis by affecting the
mitochondrial apoptosis pathway. Acta Physiol (Oxf). 210:524–533.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Xin DL, Harris MY, Wade CK, Amin M, Barr
AE and Barbe MF: Aging enhances serum cytokine response but not
task-induced grip strength declines in a rat model of
work-relatedmusculoskeletal disorders. BMC Musculoskelet Disord.
12:632011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mitzelfelt JD, Dupree JP, Seo DO, Carter
CS and Morgan D: Effects of chronic fentanyl administration on
physical performance of aged rats. Exp Gerontol. 46:65–72. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kietrys DM, Barr AE and Barbe MF: Exposure
to repetitive tasks induces motor changes related to skill
acquisition and inflammation in rats. J Mot Behav. 43:465–476.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Michael G, Kane KA and Coker SJ:
Adrenaline reveals the torsadogenic effect of combined blockade of
potassium channels in anaesthetized guinea pigs. Br J Pharmacol.
154:1414–1426. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Mengistu M, Abebe Y, Mekonnen Y and
Tolessa T: In vivo and in vitro hypotensive effect of aqueous
extract of Moringa stenopetala. Afr Health Sci. 12:545–551.
2012.PubMed/NCBI
|
|
21
|
Johnson DM, Geys R, Lissens J and Guns PJ:
Drug-induced effects on cardiovascular function in pentobarbital
anesthetized guinea-pigs: Invasive LVP measurements versus the QA
interval. J Pharmacol Toxicological Methods. 66:152–159. 2012.
View Article : Google Scholar
|
|
22
|
Guns PJ, Johnson DM, Weltens E and Lissens
J: Negative electro-mechanical windows are required for
drug-induced Torsades de Pointes in the anesthetized guinea pig. J
Pharmacol Toxicol Methods. 66:125–134. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Mooney L, Marks L, Philp KL, Skinner M,
Coker SJ and Currie S: Optimising conditions for studying the acute
effects of drugs on indices of cardiac contractility and on
haemodynamics in anaesthetized guinea pigs. J Pharmacol Toxicol
Methods. 66:43–51. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kågström J, Laumola EL, Poijes N,
Johansson M and Ericson AC: Assessment of the effects of changes in
body temperature on cardiac electrophysiology in anaesthetised
guinea pigs. J Pharmacol Toxicol Methods. 65:1–7. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Michael G, Kane KA and Coker SJ:
Adrenaline reveals the torsadogenic effect of combined blockade of
potassium channels in anaesthetized guinea pigs. Br J Pharmacol.
154:1414–1426. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lopes FDTQS, Alvarenga GS, Quiles R, Dorna
MB, Vieira JE, Dolhnikoff M and Martins MA: Pulmonary responses to
tracheal or esophageal acidification in guinea pigs with airway
inflammation. J Appl Physiol. 93:842–847. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kojima T, Tanuma N, Aikawa Y, Shin T,
Sasaki A and Matsumoto Y: Myosin-induced autoimmune polymyositis in
the rat. J Neurol Sci. 151:141–148. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ito T, Kumamoto T, Horinouchi H, Yukishige
K, Sugihara R, Fujimoto S and Tsuda T: Adhesion molecule expression
in experimental myositis. Muscle Nerve. 25:409–418. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-tie quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Currie S: Experimental myositis: The
in-vivo and in-vitro activity of lymph-node cells. J Pathol.
105:169–185. 1971. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Dawkins RL: Experimental myositis
associated with hypersensitivity to muscle. J Pathol Bacteriol.
90:619–625. 1965. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kalden JR, Williamson WG and Irvine WJ:
Experimental myasthenia gravis, myositis and myocarditis in
guinea-pigs immunized with subcellular fractions of calf thymus or
calf skeletal muscle in Freund's complete adjuvant. Clin Exp
Immunol. 13:79–88. 1973.PubMed/NCBI
|
|
33
|
Wen-Jing L, Chuan-Qiang P, Hong-Hua L,
Xiang-Hui L and Jie-Xiao L: A new modified animal model of
myosin-induced experimental autoimmune myositis enhanced by
defibrase. Arch Med Sci. 11:1272–1278. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Nemoto H, Bhopale MK, Constantinescu CS,
Schotland D and Rostami A: Skeletal muscle myosin is the
autoantigen for experimental autoimmune myositis. Exp Mol Pathol.
74:238–243. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Nakano J, Yoshimura T, Okita M, Motomura
M, Kamei S, Matsuo H and Eguchi K: Laminin-induced autoimmune
myositis in rats. J Neuropathol Exp Neurol. 64:790–796. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Matsubara S and Takamori M: Experimental
allergic myositis: Strain 13 guinea pig immunised with rabbit
myosin B fraction. Acta Neuropathol. 74:158–162. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zhao Q, Zhang C, Wang X, Chen L, Ji H and
Zhang Y: (S)-ZJM-289, a nitric oxide-releasing derivative of
3-n-butylphthalide, protects against ischemic neuronal injury by
attenuating mitochondrial dysfunction and associated cell death.
Neurochem Int. 60:134–144. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Xu J, Wang Y, Li N, Xu L, Yang H and Yang
Z: L-3-n-butylphthalide improves cognitive deficits in rats with
chronic cerebral ischemia. Neuropharmacology. 62:2424–2429. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Ma S, Xu S, Liu B, Li J, Feng N, Wang L
and Wang X: Long-term treatment of l-3-n-butylphthalide attenuated
neurodegenerative changes in aged rats. Naunyn Schmiedebergs Arch
Pharmacol. 379:565–574. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Cartwright EJ, Oceandy D, Austin C and
Neyses L: Ca2+ signalling in cardiovascular disease: The
role of the plasma membrane calcium pumps. Sci China Life Sci.
54:691–698. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Despa S and Bers DM: Na+ transport in the
normal and failing heart-remember the balance. J Mol Cell Cardiol.
61:2–10. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Tajiri K, Shimojo N, Sakai S,
Machino-Ohtsuka T, Imanaka-Yoshida K, Hiroe M, Tsujimura Y, Kimura
T, Sato A, Yasutomi Y and Aonuma K: Pitavastatin regulates helper
T-cell differentiation and ameliorates autoimmune myocarditis in
mice. Cardiovasc Drugs Ther. 27:413–424. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Pot C, Apetoh L and Kuchroo VK: Type 1
regulatory T cells (Tr1) in autoimmunity. Semin Immunol.
23:202–208. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Carvalho MI, Pires I, Prada J, Gregório H,
Lobo L and Queiroga FL: Intratumoral FoxP3 expression is associated
with angiogenesis and prognosis in malignant canine mammary tumors.
Vet Immunol Immunopathol. 178:1–9. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Pasztoi M, Bonifacius A, Pezoldt J,
Kulkarni D, Niemz J, Yang J, Teich R, Hajek J, Pisano F, Rohde M,
et al: Yersinia pseudo-tuberculosis supports Th17 differentiation
and limits de novo regulatory T cell induction by directly
interfering with T cell receptor signaling. Cell Mol Life Sci.
74:2839–2850. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Du R, Zhao H, Yan F and Li H: IL-17+Foxp3+
T cells: An intermediate differentiation stage between Th17 cells
and regulatory T cells. J Leukoc Biol. 96:39–48. 2014. View Article : Google Scholar : PubMed/NCBI
|