Introduction
In 1986, Schermer et al (1), proposed that corneal epithelium was
maintained by undifferentiated or poorly differentiated limbal stem
cells (LSCs). LSCs generally function as the reserve for cell
proliferation, and are keep in balance between cell production and
loss (2). LSCs have long been
regarded as the sole repository of corneal epithelial stem cells
(3,4). The limbus is also called the palisades
of Vogt due to its radial structure, which prevents invasion of the
conjunctival epithelium onto the cornea like a ‘barrier’. If the
‘barrier’ is damaged, then the corneal epithelium will be replaced
by a conjunctival-like epithelium. Accumulated evidence has
demonstrated that the limbal niche is efficient in restoring the
corneal surface. For example, in the in vivo model of
partial limbal stem cell deficiency, removal of the central corneal
epithelium resulted in conjunctivalization of the cornea (5). Another example is the impressive
clinical results obtained with the transplantation of limbal
holoclones (6).
However, the role of LSCs has recently been
challenged. Specifically, Majo et al (7) stated that LSCs do not necessarily
maintain normal corneal homeostasis, although LSCs are an important
source for treating corneal epithelial diseases involving stem cell
deficiency. Histopathologically, peripheral corneal thinning
diseases such as Terrien's marginal degeneration, Mooren's ulcer,
and immune peripheral ulcerative keratitis leave only the unhealthy
epithelium, extremely thinned and degenerated stroma, and
Descemet's membrane.
Corneal transplantation is now a well-established
treatment for these peripheral corneal diseases, and in peripheral
corneal pathologies the procedure usually has low rates of graft
rejection. However, part of the graft and limbus are covered with
conjunctival-shaped epithelium and vascularization during the
follow-up period. Moreover, previous data attribute the presence of
conjunctivalization to limbal stem cell deficiency (LSCD) (8,9). The
longstanding concept proposes that corneal epithelial stem cells
reside mainly in the limbus. The limbus is essential for
maintenance of corneal epithelium and damage to it results in
abnormal corneal epithelium (10,11).
In this study, we enrolled patients with peripheral
corneal disease who underwent corneal transplantation surgery, and
preserved intact corneal epithelium of graft and limbus to perform
a long-term observation. The purpose of this study was to
investigate whether LSCs could serve as a barrier to prevent
migration of the conjunctival epithelium over the long term.
Patients and methods
Patients
This prospective study enrolled patients with
peripheral corneal lesions who visited Department of Ophthalmology
in First Hospital of Jilin University and received corneal
transplantation surgery between January 2008 and January 2016.
Patients with infectious keratitis, trauma, or other ocular
diseases (i.e., cataract, glaucoma, or macular degeneration) were
excluded. Surgery informed written consent forms were obtained from
all enrolled patients. And the patients provided informed consent
for participation in the present study. The surgical procedures
performed in our study included deep anterior lamellar keratoplasty
(DALK) and penetrating keratoplasty (PKP). The research was
approved by the First Hospital of Jilin University Review Board,
and was performed in accordance with the tenets of the Declaration
of Helsinki.
Donor corneas
Donor corneas in this study came from two eye banks
of Jilin Red Cross Society, Jilin, Changchun, China. The preserved
donor corneas for DALK were stored at −80°C in pure sterile
glycerin. All glycerol-preserved tissues were rehydrated at room
temperature for 60 min before the operations were performed. Fresh
corneas of PKP were preserved in medium-term solution (Eusol-C;
Al.Chi.Mi.A. Srl, Padova, Italy) at 4°C.
Surgical procedures
All DALKs and PKPs were performed by the same
surgeon (HJ). The selection of surgical procedures was based on the
size and shape of the corneal lesion.
Crescent or ring-shaped DALK was performed if the
inner edge of lesion was in the peripheral cornea without involving
the pupil area and progressed circumferentially. D-shaped or total
DALK was selected if the inner edge was close to or involving the
pupil, and the lesion progressed both circumferentially and
centrally. The outer edge was 1–1.5 mm larger than the thinned
corneal limbus meaning that the scope of resection included the
clear arc of the limbus, in a way to avoid the recurrence of the
corneal lesion.
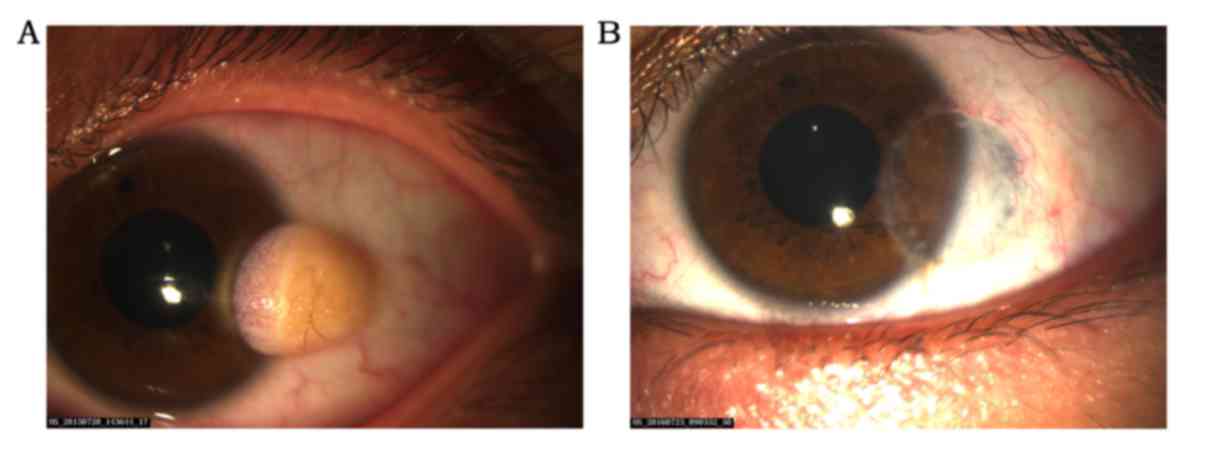
In cases of corneal limbal dermoid, the diameter of
recipient trephine was chosen according to the size of the tumor.
Generally, the diameter of the trephine was 0.25 mm larger than the
excised dermoid bed. In cases of the limbal squamous neoplasia,
incision was made 2 mm from the edge of mass. The outer edge was
3–4 mm larger than the limbus of corneal tumor. Epithelium could
desquamate for edema with rehydration.
After epithelium and endothelium were wiped away,
the full-thickness graft was sutured in place using an interrupted
10-0 nylon suture. If the range of perforation was less than 2 mm,
then DALK could be ended by anterior chamber injection of sterile
air. Otherwise, the procedure was converted into PKP
intraoperatively. The epithelium of graft was removed clearly as
well.
Postoperative medication and
follow-up
0.1% tacrolimus (Santen Pharmaceutical Co., Ltd.,
Osaka, Japan), 0.5% levofloxacin (Santen Pharmaceutical Co., Ltd.),
and 0.1% dexamethasone eyedrops (Alcon-Couvreur, S.A., Puurs,
Belgium) were routinely administered to the patients.
All patients were followed up on a weekly basis for
1 month and monthly for 6 months, and thereafter at different
intervals. Ocular examinations during each follow-up included best
corrected visual acuity (BCVA), corneal reconstruction accessed by
slit-lamp biomicroscopy, and anterior segment optical coherence
tomography (AS-OCT, Visante; Carl Zeiss Meditec AG, Dublin, CA,
USA), as well as laser scanning in vivo confocal microscopy
(IVCM, Heidelberg Retina Tomograph II with Rostock Cornea Module;
Heidelberg Engineering GmbH, Dossenheim, Germany).
Statistical analysis
All statistical analyses were performed with
Statistical Package for the Social Sciences v19 software (SPSS
Inc., Chicago, IL, USA). All tests were 2-tailed, and P<0.05 was
considered to indicate a statistically significant difference.
Results
The 85 patients enrolled in this study included 26
Terrien's marginal degeneration patients (26 eyes), 16 Mooren's
ulcer patients (16 eyes), 6 immune peripheral ulcerative keratitis
patients (6 eyes), 24 limbal dermoids patients (24 eyes), and 13
limbal squamous neoplasia patients (13 eyes); These 13 limbal
squamous neoplasia patients included 8 squamous papilloma patients
(8 eyes), 2 intraepithelial neoplasia patients (2 eyes), and 3
corneal squamous cell carcinoma patients (3 eyes). The mean age of
patients at presentation was 42.80±24.06 years (range, 1–83 years)
and 45 of the 85 patients were males. The mean postoperative
follow-up time was 3.5±2.4 years (range, 1–8 years).
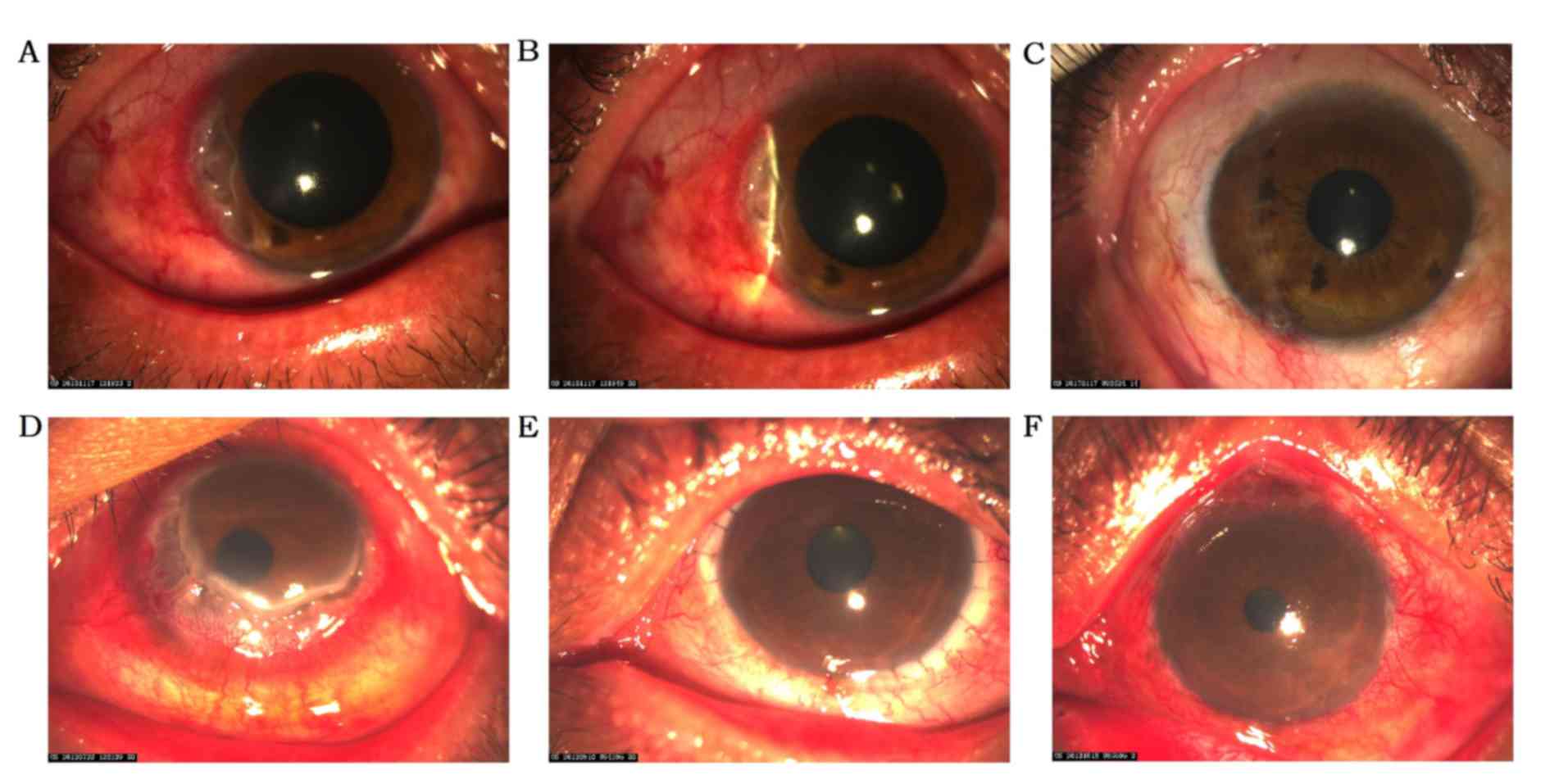
Crescent, ring-shaped, D-shaped, and circular grafts
were respectively used in 25, 4, 18, and 30 eyes with limbal tumor,
and total DALK was performed in 8 eyes (Table I) (Figs.
1–4). Seventy-six patients
finally received DALK and 9 patients received PKP. The
intraoperative complications such as iris prolapse, limbal
perforation, and scleral perforation did not occur in our research.
The BCVA fluctuated in the early period, and was stable after 3–6
months postoperatively. The mean BCVA was increased from 20/400 to
20/40 preoperatively to 20/200 to 20/20 postoperatively (P<0.01)
(Table II).
 | Table I.Shapes of corneal grafts. |
Table I.
Shapes of corneal grafts.
| Shapes of grafts | Eyes, n (%) |
|---|
| Crescent | 25 (29.4) |
| Ring-shaped | 4 (4.7) |
| D-shaped | 18 (21.2) |
| Circular | 30 (35.3) |
| Total DALK | 8 (9.4) |
| Total | 85 |
 | Table II.Comparison of the BCVA of pre and post
surgery. |
Table II.
Comparison of the BCVA of pre and post
surgery.
|
| n (%) |
|---|
|
|
|
|---|
| BCVA | Preoperative | Postoperative |
|---|
| ≥20/400 |
|
|
| <20/200 | 17 (20) | 11 (12.9) |
| ≥20/200 |
|
|
| <20/40 | 28 (32.9) | 20 (23.5) |
| ≥20/40 |
|
|
| <20/25 | 26 (30.6) | 28 (33) |
| ≥20/25 |
|
|
| <20/20 | 14 (16.5) | 26 (30.6) |
| Total | 85 | 85 |
The day after surgery, a slit-lamp microscopy
examination showed that all patients had no corneal interface
fluid. The graft was comparatively edematous in the early
postoperative period and then gradually clarified. Donor grafts
achieved complete corneal epithelialization in 7 days. The corneal
fluorescein staining showed that the corneal epithelial cell sheet
was smooth. All operation eyes received successful reconstruction
of corneal structure. During each follow-up, slit-lamp microscopy
and AS-OCT examinations showed that all grafts had intact epithelia
and were well attached to the recipient beds, as well as all cases
preserved anatomic integrity. Neither vascularization nor
conjunctivalization of the graft occurred. Rejection occurred in
one patient with Mooren's ulcer.
All operation eyes presented clear corneal
characteristics by observing central and paracentral epithelial
morphology using IVCM. Polygonal and flat cells with
hyper-reflective nuclei in the superficial layer progressively
decreased in size in the intermediate layers and small cells
without detectable nuclei were in the basal layer with reflective
borders (Fig. 5A-C). Corneal frames
of the grafts demonstrated some cellular architecture similar to
normal appearing epithelium. No conjunctival epithelial cells or
goblet cells were detected in the epithelium of the grafts. The
superficial layer showed groups of polygonal cells with bright
cytoplasm and bright nuclei. The wing cells seemed smaller than the
superficial cells, with clear bright borders. The basal layer was
represented by a honeycomb pattern of cells with smaller diameters.
No nuclei were observed in the wing cells and the basal cells
(Fig. 5D-G). IVCM showed irregular
limbal architecture with the absence of the palisades of Vogt in
the limbal area of the grafts.
Discussion
In the present study, we performed a long-term
observation of limbus in the grafts in 85 peripheral corneal lesion
patients who underwent corneal transplantation. All patients
received successful corneal reconstructions. Neither
vascularization nor conjunctivalization of the graft occurred. The
grafts were covered by normal corneal epithelium without evidence
of limbus in any operation eyes. Our findings indicate that LSCs
may not play a critical role during normal corneal epithelial
turnover in patients with peripheral corneal disease following
corneal transplantation surgery.
Majo et al (7), proposed a hypothesis whereby the stem
cells residing throughout the basal layer of the corneal epithelium
maintain the homeostasis of the corneal epithelium. The authors
believed that the central epithelial explants could successfully
regenerate the entire denuded corneal epithelial surface, and
oligopotent epithelial stem cells in the central cornea contributed
to the repair of epithelium. Nevertheless, the findings from Majo
et al (7), are based on a
mouse model and interspecies differences need to be taken into
account when extrapolating results from animal models to humans.
Nevertheless, the findings reported by Majo et al (7), have provoked controversy and have
prompted re-evaluation of the conventional LSCs hypothesis.
Some authors have supported this new hypothesis. For
instance, Chang et al (12),
separated human corneal epithelial cells from the central cornea
and the limbus for sphere-forming assay and found that both human
limbal and central epithelial cells were capable of forming spheres
in cultures with properties of stem cells properties. This finding
indicates that human central corneal epithelium contains some cells
with progenitor potential.
In another study by Chang et al (13), the corneal healing was assessed by a
human corneal organotypic culture model. In this in vitro
model, the regenerative capability of central epithelial cells with
or without limbus was evaluated by a paracentral corneal epithelial
ring or ‘donut’ ablation. The authors' results demonstrated that
the central corneal epithelium could self-regenerate and
self-repair but LSCs had no immediate response to an acute wound.
However, we think that their observation of only 12 h was too short
to properly evaluate the real capability of corneal epithelial
regeneration.
Dua et al (14), in a clinical observation with a mean
follow-up of 60 months, reported that some patients with total LSCD
(with conjunctivalization) retain central islands of normal corneal
epithelium during the entire follow-up period. It is possible that
there are some cells that have characteristics closer to the stem
cell than to the transient amplifying cell. The authors termed
these cells ‘transient cells’. Thus it seems that the role of LSCs
in maintaining normal homeostasis of the corneal epithelium is
limited due to existence of ‘transient cells’.
Our findings challenge the traditional hypostasis
and are also consistent with the observation by Majo et al
(7). The stroma of lesion was
excised to the extent that only Descemet's membrane remained, which
indicated that the corneal epithelium and matrix tissue had been
removed completely. Meanwhile, the scope of resection was
sufficiently large enough to avoid the recurrence of the corneal
lesion, with the limbus structure unreserved. Therefore, the limbus
of recipient may have a potential role in the physiologic
homeostasis of corneal epithelium postoperatively impossibly.
Our study also showed that long-term
glycerin-cryopreserved lamellar grafts were just a stromal collagen
matrix for keratoplasty containing no living cells and only dead
keratocyte cells. In the 8 cases of PKP, the corneal epithelial
layer was completely wiped off. What is maintaining the intact
corneal epithelium sheet in these cases? The more likely scenario
is that: (1) the amount of LSCs
surrounding the limbus of corneal lesion is not clinically
detectable but they are enough to maintain the homeostasis of
grafts; (2) transient amplifying
cells (TACs) in the basal cell layer of the central epithelium may
have an unexpected capacity of maintaining the normal epithelial
turnover for a long time, even longer than the previously thought
proliferative potential; and (3) the
self-renewing capacity of precursor cells in the central cornea
suggests that the limbus may not have a critical role in
physiologic corneal epithelial homeostasis. All this evidence
indicates that the regeneration of corneal epithelium is not
different from other squamous epithelia and limbus is not the only
niche for corneal stem cells.
In conclusion, the existence of long-term healthy
corneal epithelium in patients with peripheral corneal disease
following corneal transplantation surgery indicates that LSCs may
not play a critical role during normal corneal epithelial turnover.
Further research efforts are required to clarify these
possibilities and to explore the conundrum.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Author's contributions
HJ designed the study and reviewed the draft. YZ
conducted the analyses and wrote the original draft. YH completed
the methodology of the study, XL performed the data curation, XS
collected the data and FX performed the statistics.
Ethics approval and consent to
participate
Surgery informed written consent forms were obtained
from all enrolled patients. Patients provided informed consent for
participation in the present study.
The research was approved by the First Hospital of
Jilin University Review Board, and was performed in accordance with
the tenets of the Declaration of Helsinki.
Consent for publication
All identifying information is removed in the
manuscript. The patients have provided informed consent for
participation in this study and gave their consent for
publication.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Schermer A, Galvin S and Sun TT:
Differentiation-related expression of a major 64k corneal keratin
in vivo and in culture suggests limbal location of corneal
epithelial stem cells. J Cell Biol. 103:49–62. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Cotsarelis G, Cheng SZ, Dong G, Sun TT and
Lavker RM: Existence of slow-cycling limbal epithelial basal cells
that can be preferentially stimulated to proliferate: Implications
on epithelial stem cells. Cell. 57:201–209. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Li W, Hayashida Y, Chen YT and Tseng SCG:
Niche regulation of corneal epithelial stem cells at the limbus.
Cell Res. 17:26–36. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Castro-Muñozledo F: Review: Corneal
epithelial stem cells, their niche and wound healing. Mol Vis.
19:1600–1613. 2013.PubMed/NCBI
|
|
5
|
Kawakita T, Higa K, Shimmura S, Tomita M,
Tsubota K and Shimazaki J: Fate of corneal epithelial cells
separated from limbus in vivo. Invest Ophthalmol Vis Sci.
52:8132–8137. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Pathak M, Olstad OK, Drolsum L, Moe MC,
Smorodinova N, Kalasova S, Jirsova K, Nicolaissen B and Noer A: The
effect of culture medium and carrier on explant culture of human
limbal epithelium: A comparison of ultrastructure, keratin profile
and gene expression. Exp Eye Res. 153:122–132. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Majo F, Rochat A, Nicolas M, Jaoudé GA and
Barrandon Y: Oligopotent stem cells are distributed throughout the
mammalian ocular surface. Nature. 456:250–254. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Baradaran-Rafii A, Ebrahimi M, Kanavi MR,
Taghi-Abadi E, Aghdami N, Eslani M, Bakhtiari P, Einollahi B,
Baharvand H and Javadi MA: Midterm outcomes of autologous
cultivated limbal stem cell transplantation with or without
penetrating keratoplasty. Cornea. 29:502–509. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Tseng SC, He H, Zhang S and Chen SY: Niche
regulation of limbal epithelial stem cells: Relationship between
inflammation and regeneration. Ocul Surf. 14:100–112. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Shen C, Chan CC and Holland EJ: Limbal
stem cell transplantation for soft contact lens wear-related limbal
stem cell deficiency. Am J Ophthalmol. 160:1142–1149.e1. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Chan EH, Chen L, Yu F and Deng SX:
Epithelial thinning in limbal stem cell deficiency. Am J
Ophthalmol. 160:669–77.e4. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chang CY, Green CR, McGhee CN and Sherwin
T: Acute wound healing in the human central corneal epithelium
appears to be independent of limbal stem cell influence. Invest
Ophthalmol Vis Sci. 49:5279–5286. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chang CY, McGhee JJ, Green CR and Sherwin
T: Comparison of stem cell properties in cell populations isolated
from human central and limbal corneal epithelium. Cornea.
30:1155–1162. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Dua HS, Miri A, Alomar T, Yeung AM and
Said DG: The role of limbal stem cells in corneal epithelial
maintenance: Testing the dogma. Ophthalmology. 116:856–863. 2009.
View Article : Google Scholar : PubMed/NCBI
|