Introduction
Gastric carcinoma (GC) is the second most common
human cancer type and a leading cause of cancer-associated
mortality worldwide. Although the incidence has significantly
declined due to recent advances in diagnostics and therapeutics,
the survival rate remains poor. Multistep processes, including
genetics, epigenetics and environmental factors, have pivotal roles
in tumorigenesis and progression (1). The identification of novel biomarkers
in the above processes for clinical applications is urgently
required.
The histone core proteins (two of each H2A, H2B, H3
and H4) together with 146 bp DNA are wrapped around each other and
form a nucleosome, which constitutes the basic units of chromatin
(2). In dynamic and reversible
processes, as the chromosomes are condensed or loosened, the
N-terminus of the histone proteins may be altered by multiple
covalent modifications, including acetylation, methylation,
phosphorylation and ubiquitination, at the post-transcriptional
level to regulate gene expression. Numerous studies on covalent
modifications have focused on exploring the roles of acetylation
and methylation (3,4). Dysregulations of these epigenetic
modifications and associated gene expression causes may drive
carcinogenesis. Downregulation of lysine acetyltransferase 5 (KAT5)
(5) and upregulation of enhancer of
zeste 2 polycomb repressive complex 2 subunit (EZH2) (6), protein arginine methyltransferase 1
(PRMT1) (7) and the lysine
demethylase 1A (KDM1A) (8) have been
reported to be significantly associated with poor
clinicopathological features and survival of patients with GC and
other cancer types. As one of the epigenetic mechanisms, histone
modifications participate in transcriptional regulation, DNA repair
and condensation (9). Histone
deacetylase 4 has been reported to facilitate GC progression by
inhibiting p21 (10). P300
acetylates transcription factor (TF) STAT3 in histone H3 on lysine
56 to regulate gene expression (11). Furthermore, EZH2 recruits DNA
methyltransferase to the promoter region of PcG target genes to
downregulate PcG targets (12). In
addition, lysine demethylase 6B interacts with NF-κB via
demethylation of histone H3 trimethylated at lysine 27 (H3K27me3)
at downstream gene promoters participating in wound healing
(13). In the DNA damage repair
process, KAT8 was also reported to be required (14). Alterations in histone modification
levels and the expression of numerous genes encoding histone
modification-associated enzymes have also been reported in various
cancer types as epigenetic changes. For instance, SUV420H2
(KMT5C)-mediated histone H4 trimethylation on lysine 20 is
important for epidermal homeostasis. SET domain bifurcated histone
lysine methyltransferase 1, which performs H3K9 trimethylation
(H3K9me3), is established as an oncogene in melanoma (15). Upregulation of JMJD3 in metastatic
prostate cancer indicates its potential oncogenic role (16). Similarly, KDM1A and JMJD1C are
upregulated in certain GC cells and tissues (17). Therefore, histone
modification-associated changes may serve as biomarkers for early
diagnosis, therapeutic targets and prognosis prediction of GC.
Although a large number of studies have indicated
that histone modifications and abnormal expression of associated
genes have important roles in oncogenesis, only few studies have
provided comprehensive analyses of the expression and prognostic
value of associated genes in carcinomas, particularly in GC. The
present study analyzed Oncomine, The Cancer Genome Atlas (TCGA) and
Gene Ontology (GEO) datasets to perform comprehensive analyses on
histone modifications and associated gene expression profiles, as
well as their prognostic role in GC.
Materials and methods
Oncomine database analysis
The Oncomine database (http://www.oncomine.org) incorporates 715 datasets
that include 35 cancer types and contain microarray data of 86,733
samples, supporting various methods of online statistical analysis
(18). The differential expression
of histone modification and associated genes (HMGs) was compared by
using the Student's t-test to generate a P-value. As cut-off
values, the |logFC| was defined as >1 and the P-value was set at
0.05, whereas the data type was restricted to mRNA.
TCGA and GEO datasets
The UALCAN database (http://ualcan.path.uab.edu/index.html) was used to
screen the HMGs (19). The mRNA
expression datasets from the TCGA stomach adenocarcinoma (STAD)
dataset, which contained 375 GC samples and 32 normal samples, were
downloaded for further analysis (http://cancergenome.nih.gov/). The clinical data of
443 patients with GC, including overall survival (OS), survival
state, age, sex, location, American Joint Committee on Cancer
(AJCC) Tumor-Nodes-Metastasis (TNM) stage and pathological T/N/M
stage were also obtained/estimated. The integrated above data were
used to assess the association between mRNA expression levels and
prognosis. The mRNA microarray expression profile dataset GSE79973
was obtained from GEO (https://www.ncbi.nlm.nih.gov/geo/) and contained 10 GC
samples and 10 paired normal samples.
Kaplan-Meier plotter database
analysis
Kaplan-Meier plotter is capable of assessing the
effect of 54,675 genes on survival using 10,461 cancer samples.
These include the data of 5,143 breast, 1,816 ovarian, 2,437 lung
and 1,065 GC patients. The association between the expression of
certain genes and survival of GC patients was analyzed using
Kaplan-Meier plotter (http://kmplot.com/analysis/) (20). The hazard ratios (HRs) with 95%
confidence intervals (CI) and log-rank P-values were also
computed.
Statistical analysis
The association between histone modifications and
the expression of relevant genes as well as clinicopathological
features was evaluated by the χ2 test. An unpaired
t-test was used to compare the differentially expressed genes
(DEGs) between the tumor and non-tumor group. Spearman's test was
used for correlation analysis. The Kaplan-Meier method was used to
determine the patients' survival and differences between groups
were assessed using a log-rank test. In TCGA dataset, statistically
significant variables in the univariate analysis were included into
the multivariate analysis in the Cox proportional hazards model and
results were expressed as the HR with 95% CI. Data analysis was
performed using SPSS software v.23.0 (IBM Corp.). The mRNA data
downloaded from TCGA and GEO were analyzed using the edgeR package
in R (v.3.5.1) to identify DEGs between GC and non-tumor tissues.
The median value of mRNA expression was applied as a cut-off to
stratify samples into high- or low-expression groups. The STRING
v.10.5 online tool (https://string-db.org/) was used to analyze the
interactions among differential proteins. All of the P-values
reported were two-sided and P<0.05 was considered to indicate
statistical significance.
Results
Screening and identification of DEGs
in the HMGs from Oncomine, GEO and TCGA datasets
The microarray expression profile dataset GSE79973
and mRNA expression data were respectively downloaded from the GEO
and TCGA databases. DEGs were screened out using the cutoffs of
P<0.05 and |logFC|>1 for the comparison between tumor and
non-tumor samples. A total of 1,311 upregulated and 384
downregulated genes screened from GSE79973 (Fig. S1) and 8,159 upregulated and 3,758
downregulated genes were screened from TCGA (Fig. 1A). The HMGs were then retrieved and
determined using the UALCAN website. A total of 30 genes involved
the acetyl modification of histones (12 acetyltransferases and
associated genes, 18 deacetylases and associated genes) and 61
genes participating in methyl modification (53 methyltransferases
and associated genes, 8 demethylases and associated genes) were
identified. Detailed information on the location and function of
the HMGs is provided in supplementary Table SI. The differences in expression
from HMGs between various carcinomas and paracancerous tissues were
also indicated in the Oncomine analysis (Fig. S2).
To screen the HMGs that were differentially
expressed in GC vs. non-cancer tissues among all Oncomine, GEO and
TCGA datasets, Venn diagram analysis was used to obtain the
intersection of the three datasets. As presented in Fig. 1B-E, the five genes KAT2A, nuclear
receptor coactivator 1 (NCOA1), SMYD family member 5 (SMYD5), PRMT1
and PR/SET domain 16 (PRDM16) were identified. Detailed information
on the location and function of these HMGs is provided in Table I.
 | Table I.Basic characteristics and function of
five histone modification enzymes and associated genes. |
Table I.
Basic characteristics and function of
five histone modification enzymes and associated genes.
| Gene name | Location | Exon | Protein mass
(kDa) | Encoding protein
and biological function |
|---|
| KAT2A | 17q21.2 | 18 | 93.94 | Also known as GCN5,
KAT2A is a HAT that functions primarily as a transcriptional
activator. It also functions as a repressor of NF-κB by promoting
ubiquitination of the NF-κB subunit RELA in a HAT-independent
manner |
| NCOA1 | 2p23.3 | 24 | 156.8 | A transcriptional
coactivator for steroid and nuclear hormone receptors. A member of
the p160/steroid receptor coactivator family, which has histone
acetyltransferase activity and contains a nuclear localization
signal, as well as basic helix-loop-helix and PAS domains. NCOA1
also binds nuclear receptors directly and stimulates
transcriptional activities in a hormone-dependent fashion. |
| SMYD5 | 2p13.2 | 13 | 34.42 | – |
| PRMT1 | 19q13.33 | 13 | 39.61 | A member of the
PRMT family, which is involved in post-translational modification
of target proteins in numerous biological processes. As a type I
PRMT, it is responsible for the majority of cellular arginine
methylation activity. Increased expression of this gene may have a
role in numerous types of cancer. |
| PRDM16 | 1p36.32 | 18 | – | A zinc finger
transcription factor containing an N-terminal PR domain.
Translocation results in the overexpression of a truncated version
of this protein that lacks the PR domain, which may have an
important role in the pathogenesis of myelodysplastic syndrome and
acute myelocytic leukemia. |
Differential expression of KAT2A,
NCOA1, SMYD5, PRMT1 and PRDM16 in GC vs. normal tissues
KAT2A, SMYD5 and PRMT1 were upregulated in GC vs.
normal tissues in all three datasets (Oncomine, GSE79973 and TCGA).
In the GSE79973 and TCGA STAD datasets, PRDM16 was observed to be
downregulated. Furthermore, in the GSE79973 dataset and the dataset
DErrico Gastric (Oncomine dataset), NCOA1 was upregulated, and
these results were contrary to those obtained with the TCGA STAD
dataset (Table II). A 29% decline
of NCOA1 mRNA expression was observed in TCGA STAD dataset. In the
Wang Gastric and Cui Gastric datasets (Oncomine dataset), KAT2A was
1.756-fold (P=1.15×10−4) and 1.461-fold
(P=1.37×10−5) increased, respectively, in GC vs. normal
tissues. Furthermore, the FC of upregulated KAT2A in the GSE79973
dataset was 1.809 (P=1.89×10−5). In the Wang Gastric
dataset from Oncomine, SMYD5 and PRMT1, which in turn was also
determined to be 1.470-fold (P=1.00×10−3) increased in
the Cui Gastric dataset, were 2.154-fold (P=6.18×10−4)
and 1.884-fold (P=8.01×10−4) elevated, respectively.
Similar to the results of the Oncomine analysis, screening of the
GSE79973 dataset indicated that NCOA1, SMYD5 and PRMT1 were
1.347-fold (P=8.95×10−3), 1.708-fold
(P=7.39×10−6) and 1.724-fold (P=2.00×10−6)
increased. Furthermore, comparisons based on different pathological
classifications were performed to determine differential expression
of KAT2A, NCOA1, SMYD5 and PRMT1, and in the datasets DErrico
Gastric and Cho Gastric in the Oncomine analysis, the FC of KAT2A,
NCOA1, SMYD5 and PRMT1 in gastric intestinal-type adenocarcinoma
was higher than in gastric mixed and diffuse gastric adenocarcinoma
compared to normal tissues (respective FCs and P-values: 2.132,
1.61×10−11; 1.562, 7.00×10−3; 4.106,
1.08×10−10; 2.776, 2.62×10−11). The box plots
in Fig. 2A-I display the significant
alterations in expression among the five genes in subgroup
comparisons.
 | Figure 2.Box plots that represent the mRNA
expression levels of histone modification-associated genes in
different types of gastric cancer. (A-G) Datasets of Oncomine; (A)
KAT2A: 0, normal tissues; 1, diffuse gastric adenocarcinoma; 2,
gastric adenocarcinoma; 3, gastric intestinal type adenocarcinoma;
4, gastric mixed adenocarcinoma; (B) KAT2A: 1, gastric mucosa; 2,
gastric tissue; 3, gastric cancer; (C) NCOA1: 0, normal tissue; 1,
gastric mucosa; 2, diffuse gastric adenocarcinoma; 3, gastric
intestinal type adenocarcinoma; 4, gastric mixed adenocarcinoma;
(D) PRMT1: 0, normal tissues; 1, diffuse gastric adenocarcinoma; 2,
gastric adenocarcinoma; 3, gastric intestinal type adenocarcinoma;
4, gastric mix adenocarcinoma; (E) PRMT1: 1, gastric mucosa; 2,
gastric tissue; 3, gastric cancer; (F) SMYD5: 0, normal tissues; 1,
diffuse gastric adenocarcinoma; 2, gastric adenocarcinoma; 3,
gastric intestinal type adenocarcinoma; 4, gastric mix
adenocarcinoma; (G) SMYD5: 1, gastric mucosa; 2, gastric tissue; 3,
gastric cancer. (H) Datasets of GSE79973. (I) Dataset of TCGA.
*P<0.05; **P<0.01 and ***P<0.001 vs. non-tumor. TCGA, The
Cancer Genome Atlas; KAT2A, lysine acetyltransferase 2A; NCOA1,
nuclear receptor coactivator 1; PRMT1, protein arginine
methyltransferase 1; SMYD5, SMYD family member 5; PRDM16,
PRDF1-RIZ/Su(var)3-9, enhancer-of-zeste and trithorax domain
16. |
 | Table II.Differential expression of five
histone modification enzymes and associated genes between different
types of gastric cancer and normal tissues. |
Table II.
Differential expression of five
histone modification enzymes and associated genes between different
types of gastric cancer and normal tissues.
| Gene/comparison of
groups |
Up/downregulation | Fold change | t-test | P-value | Dataset |
|---|
| KAT2A |
|
|
|
|
|
| Gastric
cancer vs. Normal | ↑ | 1.756 | 4.356 |
1.15×10−4 | Wang Gastric |
|
| ↑ | 1.461 | 4.374 |
1.37×10−5 | Cui Gastric |
|
| ↑ | 1.809 | 6.698 |
1.89×10−5 | GSE79973 |
| Gastric
intestinal type adenocarcinoma vs. Normal | ↑ | 2.132 | 8.265 |
1.61×10−11 | DErrico
Gastric |
| Gastric
mixed adenocarcinoma vs. Normal | ↑ | 1.636 | 5.839 |
7.92×10−6 |
|
| Diffuse
gastric adenocarcinoma vs. Normal | ↑ | 1.848 | 4.568 |
7.12×10−4 |
|
| Gastric
adenocarcinoma vs. Normal | ↑ | 1.998 | – |
3.10×10−14 | TCGA STAD |
| NCOA1 |
|
|
|
|
|
| Gastric
cancer vs. Normal | ↑ | 1.347 | 3.283 |
8.95×10−3 | GSE79973 |
| Gastric
mixed adenocarcinoma vs. Normal | ↑ | 1.562 | 3.675 |
7.00×10−3 | DErrico
Gastric |
| Gastric
adenocarcinoma vs. Normal | ↓ | 1.509 | – |
2.65×10−12 | TCGA STAD |
| SMYD5 |
|
|
|
|
|
| Gastric
cancer vs. Normal | ↑ | 2.154 | 3.660 |
6.18×10−4 | Wang Gastric |
|
| ↑ | 1.708 | 7.350 |
7.39×10−6 | GSE79973 |
| Gastric
intestinal type adenocarcinoma vs. Normal | ↑ | 4.106 | 7.776 |
1.08×10−10 | DErrico
Gastric |
|
| ↑ | 1.404 | 6.754 |
1.58×10−9 | Chen Gastric |
|
| ↑ | 1.215 | 3.231 |
1.00×10−3 | Cho Gastric |
| Gastric
mixed adenocarcinoma vs. Normal | ↑ | 3.901 | 8.553 |
1.63×10−9 | DErrico
Gastric |
|
| ↑ | 1.359 | 2.863 |
9.00×10−3 | Chen Gastric |
|
| ↑ | 1.191 | 2.887 |
4.00×10−3 | Cho Gastric |
| Diffuse
gastric adenocarcinoma vs. Normal | ↑ | 2.563 | 2.207 |
3.40×10−2 | DErrico
Gastric |
|
| ↑ | 1.263 | 4.691 |
1.87×10−5 | Chen Gastric |
|
| ↑ | 1.268 | 4.349 |
3.94×10−5 | Cho Gastric |
| Gastric
adenocarcinoma vs. Normal | ↑ | 1.597 | – |
1.03×10−10 | TCGA STAD |
| PRMT1 |
|
|
|
|
|
| Gastric
cancer vs. Normal | ↑ | 1.884 | 3.608 |
8.01×10−4 | Wang Gastric |
|
| ↑ | 1.470 | 3.114 |
1.00×10−3 | Cui Gastric |
|
| ↑ | 1.724 | 8.310 |
2.00×10−6 | GSE79973 |
| Gastric
intestinal type adenocarcinoma vs. Normal | ↑ | 2.776 | 8.515 |
2.62×10−11 | DErrico
Gastric |
|
| ↑ | 1.473 | 6.843 |
2.16×10−9 | Chen Gastric |
|
| ↑ | 1.623 | 3.641 |
4.16×10−4 | Cho Gastric |
| Gastric
mixed adenocarcinoma vs. Normal | ↑ | 2.215 | 8.197 |
3.00×10−6 | DErrico
Gastric |
|
| ↑ | 1.521 | 4.054 |
1.00×10−3 | Chen Gastric |
|
| ↑ | 1.687 | 4.156 |
1.83×10−4 | Cho Gastric |
| Diffuse
gastric adenocarcinoma vs. Normal | ↑ | 1.962 | 3.234 |
8.00×10−3 | DErrico
Gastric |
|
| ↑ | 1.353 | 4.563 |
4.39×10−5 | Chen Gastric |
|
| ↑ | 1.476 | 4.848 |
9.11×10−6 | Cho Gastric |
| Gastric
adenocarcinoma vs. Normal | ↑ | 1.218 | – |
2.27×10−2 | TCGA STAD |
| PRDM16 |
|
|
|
|
|
| Gastric
cancer vs. Normal | ↓ | 1.574 | −3.539 |
5.42×10−3 | GSE79973 |
| Gastric
adenocarcinoma vs. Normal | ↓ | 1.832 | – |
6.87×10−5 | TCGA STAD |
Association of KAT2A, NCOA1, SMYD5,
PRMT1 and PRDM16 expression with clinicopathological features
To explore the potential clinical significance of
the five DEGs in patients with GC, the clinical data of 443
patients were downloaded from TCGA and only 334 patients with
complete clinical and gene data were retained after elimination of
patients with missing data by processing in R. The cases were
divided into a low-expression group and a high-expression group
according to the median mRNA expression value of the five genes in
tumor or non-tumor tissues. The associations between the expression
of the five genes and clinicopathological characteristics are
presented in Table III. Factors
including age, sex, tumor size, AJCC stage and T/N/M stage were
evaluated. Unfortunately, no significant associations were observed
between the five aberrantly expressed genes and the above-mentioned
clinical features.
 | Table III.Association between five histone
modification enzymes and associated genes and clinicopathological
parameters of gastric cancer patients. |
Table III.
Association between five histone
modification enzymes and associated genes and clinicopathological
parameters of gastric cancer patients.
|
|
| KAT2A |
| NCOA1 |
| SMYD5 |
| PRMT1 |
| PRDM16 |
|
|---|
|
|
|
|
|
|
|
|
|
|
|
|
|
|---|
| Feature | N | Low | High | P-value | Low | High | P-value | Low | High | P-value | Low | High | P-value | Low | High | P-value |
|---|
| Sample type |
|
|
| <0.001 |
|
| <0.001 |
|
| <0.001 |
|
| 0.042 |
|
| 0.022 |
|
Tumor | 375 | 213 | 162 |
| 211 | 164 |
| 207 | 168 |
| 217 | 158 |
| 243 | 132 |
|
|
Non-tumor | 32 | 30 | 2 |
| 8 | 24 |
| 31 | 1 |
| 24 | 8 |
| 14 | 18 |
|
| Age (years) |
|
|
| 0.408 |
|
| 0.401 |
|
| 0.098 |
|
| 0.634 |
|
| 0.109 |
|
<60 | 105 | 63 | 42 |
| 62 | 43 |
| 64 | 41 |
| 63 | 42 |
| 75 | 30 |
|
|
≥60 | 229 | 126 | 103 |
| 131 | 98 |
| 116 | 113 |
| 130 | 99 |
| 142 | 87 |
|
| Sex |
|
|
| 0.730 |
|
| 0.937 |
|
| 1.000 |
|
| 0.489 |
|
| 0.096 |
|
Male | 214 | 123 | 91 |
| 124 | 90 |
| 118 | 96 |
| 127 | 87 |
| 132 | 82 |
|
|
Female | 120 | 66 | 54 |
| 69 | 51 |
| 62 | 58 |
| 66 | 54 |
| 85 | 35 |
|
| Location |
|
|
| 0.780 |
|
| 0.432 |
|
| 0.342 |
|
| 0.878 |
|
| 0.097 |
|
GEJ | 35 | 23 | 12 |
| 24 | 11 |
| 19 | 16 |
| 26 | 9 |
| 19 | 16 |
|
|
Cardia/proximal | 43 | 17 | 16 |
| 24 | 19 |
| 23 | 20 |
| 25 | 18 |
| 28 | 15 |
|
|
Fundus/body | 119 | 59 | 60 |
| 67 | 52 |
| 59 | 60 |
| 56 | 63 |
| 74 | 45 |
|
| Stomach
body | 5 | 3 | 2 |
| 3 | 2 |
| 2 | 3 |
| 4 | 1 |
| 3 | 2 |
|
|
Antrum/distal | 127 | 73 | 54 |
| 73 | 54 |
| 73 | 54 |
| 78 | 49 |
| 91 | 36 |
|
|
Other | 5 | 4 | 1 |
| 2 | 3 |
| 4 | 1 |
| 4 | 1 |
| 2 | 3 |
|
| AJCC stage |
|
|
| 0.390 |
|
| 0.772 |
|
| 0.836 |
|
| 0.676 |
|
| 0.941 |
| I | 46 | 26 | 20 |
| 26 | 20 |
| 25 | 21 |
| 26 | 20 |
| 27 | 19 |
|
| II | 112 | 66 | 46 |
| 67 | 45 |
| 57 | 55 |
| 62 | 50 |
| 78 | 34 |
|
|
III | 142 | 82 | 60 |
| 77 | 65 |
| 81 | 61 |
| 86 | 56 |
| 91 | 51 |
|
| IV | 34 | 14 | 18 |
| 23 | 11 |
| 17 | 17 |
| 19 | 15 |
| 21 | 13 |
|
| T-stage |
|
|
| 0.168 |
|
| 0.577 |
|
| 0.528 |
|
| 0.798 |
|
| 0.380 |
|
T1a/b | 15 | 6 | 9 |
| 10 | 5 |
| 9 | 6 |
| 11 | 4 |
| 8 | 7 |
|
| T2 | 68 | 46 | 22 |
| 37 | 31 |
| 37 | 31 |
| 38 | 30 |
| 44 | 24 |
|
| T3 | 158 | 92 | 66 |
| 96 | 62 |
| 77 | 81 |
| 85 | 73 |
| 102 | 56 |
|
|
T4a/b | 93 | 45 | 48 |
| 50 | 43 |
| 57 | 36 |
| 59 | 34 |
| 63 | 30 |
|
| N-stage |
|
|
| 0.910 |
|
| 0.112 |
|
| 0.492 |
|
| 0.142 |
|
| 0.440 |
| N0 | 105 | 57 | 48 |
| 62 | 43 |
| 56 | 49 |
| 55 | 50 |
| 71 | 34 |
|
| N1 | 89 | 52 | 37 |
| 57 | 32 |
| 51 | 38 |
| 51 | 38 |
| 59 | 30 |
|
| N2 | 71 | 43 | 28 |
| 42 | 29 |
| 41 | 30 |
| 44 | 27 |
| 43 | 28 |
|
|
N3a/b | 69 | 37 | 32 |
| 32 | 37 |
| 32 | 37 |
| 43 | 26 |
| 44 | 25 |
|
| M-stage |
|
|
| 0.163 |
|
| 0.319 |
|
| 0.434 |
|
| 0.843 |
|
| 0.680 |
| M0 | 306 | 177 | 129 |
| 174 | 132 |
| 167 | 139 |
| 176 | 130 |
| 200 | 106 |
|
| M1 | 28 | 12 | 16 |
| 19 | 9 |
| 13 | 15 |
| 17 | 11 |
| 17 | 11 |
|
Prognostic value of KAT2A, NCOA1,
SMYD5, PRMT1 and PRDM16 in GC
The prognostic value of the expression status of
KAT2A, NCOA1, SMYD5, PRMT1 and PRDM16 was first examined in the
TCGA dataset (Fig. 3A-E). Low mRNA
expression of NCOA1 mRNA was observed to be linked to significantly
better OS in GC (HR=0.667, 95% CI=0.473–0.943, P=0.022; Fig. 3B). However, the mRNA expression
status of KAT2A, SMYD5, PRMT1 and PRDM16 mRNA was not associated
with OS in GC (HR=1.146, 95% CI=0.811–1.620, P=0.440; HR=1.031, 95%
CI=0.733–1.452, P=0.860; HR=1.024, 95% CI=0.725–1.447, P=0.892;
HR=0.950, 95% CI=0.667–1.354, P=0.777; Fig. 3A and C-E, respectively). Kaplan-Meier
plotter analysis was also used to verify the associations between
the mRNA expression of five genes and the clinical survival outcome
(Fig. 3F-J). In order to reduce the
bias caused by the conversion of time (day or mouth), the time
units were used as presented in the respective datasets for the
Kaplan-Meier survival curves. As in the TCGA dataset, low NCOA1
expression in GC tissues was associated with better OS (HR=0.690,
95% CI=0.570–0.840, P<0.001), and the survival curve for the
group with high PRMT1 mRNA expression in GC tissues did not exhibit
any significant difference from that of the low expression group
(HR=0.840, 95% CI=0.710–1.010, P=0.061; Fig. 3I). Contrary to the results obtained
with the TCGA dataset, patients with high expression of KAT2A,
SMYT5 and PRDM16 in their GC tissue exhibited significantly poorer
OS than those with corresponding low expression according to
Kaplan-Meier plotter analysis (Fig. 3F,
H and J, respectively). The Cox proportional hazards model
indicated that NCOA1 expression, age, N stage, M stage were
independent predictors for survival in GC (HR=1.523, 95%
CI=1.072–2.163, P=0.019; HR=1.939, 95% CI=1.291–2.914, P=0.001;
HR=1.233, 95% CI=1.049–1.450, HR=2.419, 95% CI=1.423–4.115,
respectively; Table IV).
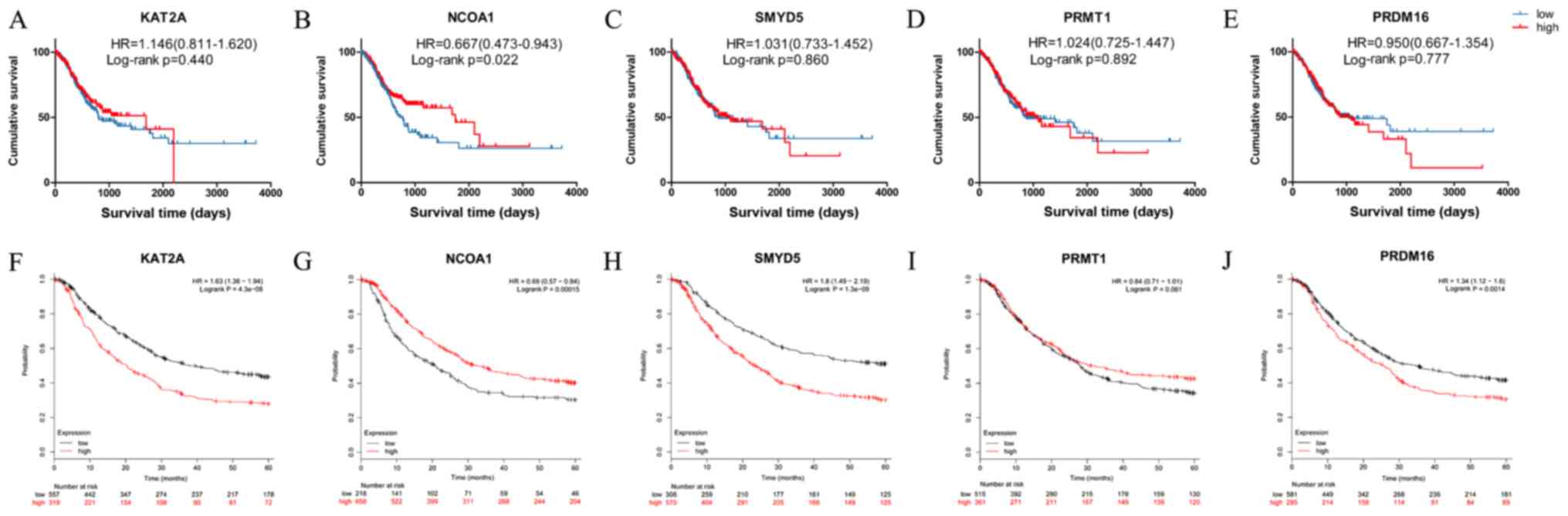 | Figure 3.Kaplan-Meier survival curves from
TCGA and Kaplan-Meier plotter analyses, depicting the survival of
gastric cancer patients according to the expression levels of
histone modification-associated genes. (A-E) Kaplan-Meier survival
curves from TCGA datasets; (A) KAT2A, (B) NCOA1; (C) SMYD5; (D)
PRMT1; (E) PRDM16; (F-J) Kaplan-Meier plotter analysis for five
genes (F) KAT2A, (G) NCOA1; (H) SMYD5; (I) PRMT1; (J) PRDM16. TCGA,
The Cancer Genome Atlas; KAT2A, lysine acetyltransferase 2A; NCOA1,
nuclear receptor coactivator 1; PRMT1, protein arginine
methyltransferase 1; SMYD5, SMYD family member 5; PRDM16,
PRDF1-RIZ/Su(var)3-9, enhancer-of-zeste and trithorax domain 16;
HR, hazard ratio; The values in brackets are the 95% CI. |
 | Table IV.Univariate and multivariate analysis
in the COX proportional hazard model. |
Table IV.
Univariate and multivariate analysis
in the COX proportional hazard model.
|
| Univariate
analysis | Multivariate
analysis |
|---|
|
|
|
|
|---|
| Features | HR | 95% CI | P-value | HR | 95% CI | P-value |
|---|
| KAT2A | 1.147 | 0.809–1.627 | 0.440 |
|
|
|
| NCOA1 | 1.487 | 1.057–2.093 | 0.023 | 1.523 |
1.072–2.163 | 0.019 |
| SMYD5 | 0.970 | 0.689–1.365 | 0.859 |
|
|
|
| PRMT1 | 0.976 | 0.691–1.380 | 0.892 |
|
|
|
| PRDM16 | 1.052 | 0.740–1.496 | 0.777 |
|
|
|
| Age | 1.633 | 1.096–2.433 | 0.016 | 1.939 |
1.291–2.914 | 0.001 |
| Sex | 0.820 | 0.568–1.183 | 0.288 |
|
|
|
| Location | 1.006 | 0.892–1.135 | 0.925 |
|
|
|
| AJCC stage |
|
|
|
|
|
|
|
T-stage | 1.307 | 1.052–1.625 | 0.016 | 1.185 | 0.942–1.492 | 0.147 |
|
N-stage | 1.339 | 1.149–1.560 |
<0.001 | 1.233 |
1.049–1.450 | 0.011 |
|
M-stage | 2.387 | 1.447–3.936 | 0.001 | 2.419 |
1.423–4.115 | 0.001 |
Bromodomain protein ATPase family AAA
domain- containing protein 2 (ATAD2) is associated with the
HMGs
ATAD2, a member of the AAA + ATPase family of
proteins, contains a bromodomain and its functions are linked to
genome regulation and histone modification. A previous study by our
group on hepatocellular carcinoma suggested that ATAD2 was
overexpressed in tumor tissue and associated with poor survival
(21). To explore the association
between ATAD2 and the five genes screened out from datasets in the
present study, the STRING v.10.5 online tool (https://string-db.org/) was used to display the
interactions among them. As presented in Fig. 4A, protein-protein interaction network
highlighted the association between ATAD2 and estrogen receptor 1
(ESR1), PRMT1, NCOA1 and KAT2A. Associations between
clinicopathological features and ATAD2 or ESR1 mRNA expression were
also assessed (Table SII) and no
statistical significances were observed. Spearman's correlation
analysis revealed that ATAD2 expression was positively correlated
with PRMT1 (R2=0.123, Spearman rho=0.356,
P=1.19×10−12; Fig. 4B)
and negatively correlated with ESR1 (R2=0.177, Spearman
rho=−0.449, P=5.30×10−20; Fig. 4C) and NCOA1 (R2=0.022,
Spearman rho=−0.160, P=0.002; Fig.
4D).
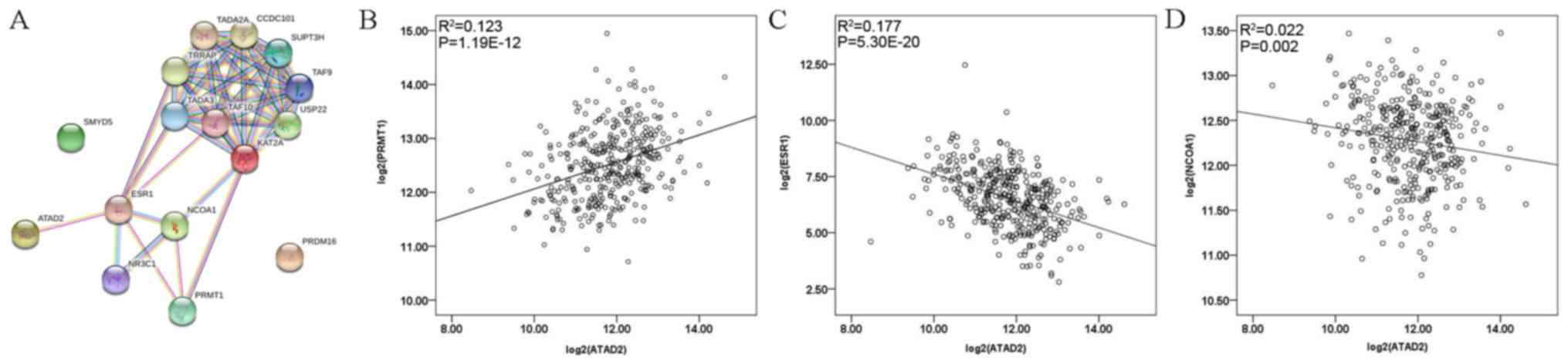 | Figure 4.(A) Protein-protein interaction
network displaying the interactions among ATAD2, ESR1, NCOA1,
PRMT1, PRDM16 and SMYD5, as determined with STRING v.10.5. (B-D)
Spearman analysis of the correlation between ATAD2 and (B) PRMT1,
(C) ESR1 and (D) NCOA1. KAT2A, lysine acetyltransferase 2A; NCOA1,
nuclear receptor coactivator 1; PRMT1, protein arginine
methyltransferase 1; SMYD5, SMYD family member 5; PRDM16,
PRDF1-RIZ/Su(var)3-9, enhancer-of-zeste and trithorax domain 16;
ESR1, estrogen receptor 1. |
Discussion
The maintenance of stable and ordered chromatin
during dynamic packaging is vital to normal cellular homeostasis.
Warped histones and DNA are subject to covalent post-translational
modifications in order to influence the number of
chromatin-associated cellular events, including transcription,
replication recombination and DNA repair (22). Dysregulation of the epigenetic
mechanisms that govern transcriptional regulation resembles steps
in the oncogenic process, causing inappropriate activation of
oncogenes or the inhibition of tumor suppressors and leading to
carcinogenesis. Accumulating evidence has demonstrated that
epigenetic alterations caused by histone modifications also have
important roles in gastric carcinogenesis, particularly the
well-studied acetylation and methylation modifications (23). The histone modifications-associated
enzymes and encoding genes are among the most frequent abnormal
targets in aberrant histone modifications. Genes that encode
histone modification enzymes, including the CREB binding protein
(CBP), p300, KAT5, KDM1A and JMJD1C, have been reported to be
aberrantly expressed in GC and are significantly correlated with
poor survival (5,6,17,24–26).
Possibly due to lack of data at the genomic level, comprehensive
molecular characterization of histone modification regulations in
GC has rarely been performed. To the best of our knowledge, the
present study was the first to elucidate histone modifications,
associated gene expression profiles and prognostic roles of key
genes by using Bioinformatics analysis of datasets to explore the
implication of deregulated histone modification in the initiation
and development of GC and the underlying mechanisms.
The present results suggested that KAT2A, NCOA1,
SMYD5, PRMT1 and PRDM16 were differentially expressed in GC vs.
non-cancer tissues. Among them, KAT2A, SMYD5 and PRMT1 were highly
upregulated in GC compared with normal tissues. The expression of
PRDM16 was observed to be downregulated in GC. However, the
opposite result was identified when detecting NCOA1 expression.
Analysis of the GEO and Oncomine datasets revealed that NCOA1 was
upregulated; these results were contrary to those of the TCGA STAD
dataset. As a member of the p160 SRC family, which also includes
NCOA2 and NCOA3, NCOA1 may interact with nuclear hormone receptors,
including ESR, and other TFs, including AP1, c-Jun/c-Fos and
HOXC11, as coactivators to facilitate the assembly of
transcriptional protein complexes for chromatin remodeling and the
activation of downstream target genes (27–31).
Earlier studies identified an association between the increased
expression of NCOA1 and enhanced angiogenesis, cell proliferation
and survival, disease recurrence, a higher tumor grade and poor
prognosis in a variety of cancer types. Analysis results from a low
NCOA1 expression by our analysis results in this article appeared
to contradict previous evidence; however, the GC dataset from Frycz
et al (32) indicated that
the mRNA expression of NCOA1 (P=0.00021) was significantly reduced
in the tumoral mucosa compared with that in the adjacent healthy
mucosa. Decreased levels of NCOA1 mRNA in GC tissue may be due to
upregulation of cytochrome P450 family 19 subfamily A member 1 mRNA
in the tumoral gastric mucosa, which causes dysregulation of two
17β-estradiol (E2) synthesis routes (the sulfatase and aromatase
signaling pathways), resulting in E2 deficiency and inhibition of
NCOA1 expression in E2-dependent methods (33). Furthermore, low expression of NCOA1
mRNA has been observed in bladder cancer urothelium samples
(34), and the results of the
present study were consistent with these results. NCOA1 not only
acts as a coactivator, but has also been indicated to possess
histone acetyltransferase activity. Sheppard et al (35) reported that the recruitment and
induction of NCOA1 in the H3 sequence are each critical for the
NCOA1-CBP interaction, which is necessary for ER function. Although
the FC of NCOA1 expression was <2 in the GEO, TCGA and Oncomine
datasets, the present results demonstrated that, no matter whether
the expression of NCOA1 was upregulated or downregulated, aberrant
expression of NCOA1 is significantly associated with poor prognosis
and is an independent predictor for GC from our analysis (TCGA
data: HR=0.639, 95% CI=0.437–0.933, P=0.020). This evidence
suggested that weakly DEGs may also have important functions and
roles in the occurrence and development of tumors. For instance, by
quantifying the adenomatosis polyposis coli (APC) gene, Yan et
al (36) determined that a weak
reduction of APC expression was closely associated with the
occurrence of hereditary colorectal tumors.
Unfortunately, no associations were observed between
the aberrant expression of five specific genes (KAT2A, NCOA1,
SMYD5, PRMT1 and PRDM16) and clinical features when their potential
clinical significance in patients with GC was explored in the TCGA
dataset. Although age may serve as an independent prognostic factor
according to the multivariate analysis, it was not a significant
predictor for survival of patients whose genes were differentially
expressed when stratified by age (data not presented). In addition,
in a report on 97 patients with stage III–IVa/b head and neck
squamous cell carcinoma, similar results were observed, in that
NCOA1 expression was not significantly associated with any
clinicopathological features, including sex, tumor site, T
classification or nodal status (37). However, the majority of previous
studies on certain malignant tumors indeed suggested that the
aberrant expression of KAT2A, NCOA1, SMYD5, PRMT1 and PRDM16 was
significantly associated with the patients' clinicopathological
features (38,39). It is known that the molecular
complexity and heterogeneity of GC made the elucidation of its
specific pathogenesis challenging. All external or internal factors
(e.g. poor dietary habits or genetic factors) and interwoven
pathways may facilitate the occurrence of GC (1). Therefore, the heterogeneity between
different studies on GC indicates a requirement for further
large-scale studies to clarify and verify the data.
Histone modification, particularly acetylation and
methylation, constitutes a vital epigenetic mechanism involved in
the dynamic regulation of chromatin structure and gene expression
(3,4). However, heterogenous factors associated
with GC influence the onset and progression of histone modification
reactions, including post-transcriptional regulation, the
microenvironment, infection, immune responses and underlying
genetic alterations. The upstream and downstream effects affect
changes in the HMGs and associated enzymes that catalyze histone
modifications, resulting in malignant processes. In the present
study, five significant genes were identified: KAT2A, NCOA1, SMYD5,
PRMT1 and PRDM16, all of which have vital roles in histone
modification. In addition to histone substrates, these enzymes may
also directly catalyze non-histone substrates, including TFs, to
perform target gene transcriptional regulation. KAT2A, encoding a
histone lysine acetyltransferase, catalyzes the acetylation of
lysine residues on histones, including H2B, H3 or H4 and TFs
(40,41). PRDM16, a PR/SET domain family
members, contains an N-terminal positive regulatory (PR) domain and
zinc fingers, and functions as a histone 3 methyltransferase
(42). Contributing to 90% of
cellular PRMT activity and acting as a histone arginine
methyltransferase, PRMT1 methylates histones, RNA-binding proteins
and other TFs to epigenetically control the expression of
downstream genes (43,44). SMYD5, a member of the histone lysine
methyltransferase family, contains the catalytic SET domain and
trimethylates H4K20, which has a critical role in carcinogenesis.
However, due to the lack of a TPR domain in the structure,
substrates of SMYD5 are limited on anything besides histones
(45,46). Furthermore, it is worth mentioning
that these genes also have important roles in drug resistance
(47,48).
Underlying genetic alterations, including promoter
methylation, copy number variations (CNVs) and chromosomal
instability (CIN), have been reported to regulate histone
modification-associated gene expression. For instance, low PRDM16
expression levels in non-small cell lung cancer and esophageal
cancer were reported to be correlated with its promoter methylation
(49,50). A high frequency of CNVs at 1p36.32
harboring the PRDM16 gene was observed in GC, suggesting that
changes in gene CNVs also have vital roles in regulating gene
expression (51). In addition,
Burghel et al (52) indicated
that PRDM16 was highly expressed in gained focal minimal common
regions caused by CIN in colorectal cancer. Besides genetic
variations, post-transcriptional regulations also affected the
functioning of associated enzymes. For instance, NCOA1 was
confirmed as a target of miR-223-3p and demonstrated to have low
expression levels (53,54). Evidence has indicated that changes to
the microenvironment resulting from hypoxia, which is insufficient
to maintain cellular function, were associated with cancer
pathology. In glioma cells, hypoxia decreased the gene expression
of NCOA1 (55). It is known that
Helicobacter pylori is responsible for gastric inflammation
and gastric malignancy, which causes general inflammatory stress
within the gastric mucosa, activating multiple oncogenic pathways
and inducing epigenetic alterations, including histone
modifications (56). These results
suggested that factors upstream of histone modification-associated
genes are important for the regulation of the expression of these
genes.
Aberrant gene expression caused by upstream factors
initiates cascade reactions, resulting in normal cells transforming
into cancer cells and other types of malignant behavior. For
instance, deletion of the KAT2A gene induces apoptosis in acute
myeloid leukemia (57). Similarly,
an increased apoptotic rate was observed in prostate cancer cells
when PRDM16 expression was downregulated (58). Regarding cell regeneration and
differentiation, histone modification-associated genes were also
suggested to have important roles. PRMT1 methylates arginine on
substrates Six1 or Eya1, and has been indicated to regulate muscle
stem cell regeneration and differentiation (59). In embryonic stem cell
differentiation, SMYD5 increases H4K20me3 and H3K9me3 levels and
maintains chromosome integrity to ensure accurate differentiation
(60). The functional basis of
invasion and metastasis is the epithelial-to-mesenchymal transition
(EMT). Previous studies have demonstrated that PRMT1, as a
regulator, is closely associated with EMT. Katsuno et al
(61) reported that PRMT1 is an
essential mediator of transforming growth factor-r signaling,
regulating the EMT and epithelial cell stemness by methylating
SMAD7. Dysregulation of histone modification-associated genes not
only promotes the acquirement of malignant biological phenotypes of
cancer cells, but also has important effects on immunity. KAT2A,
which is recruited by nuclear factor of activated T cells during
the activation of T-cell receptor signaling pathways, methylates
H3K9 of the interleukin-2 gene promoter to regulate T-cell
activation and CD4+ T-cell differentiation into type 1
T-helper cells (Th1)/Th17 (62). In
addition, methylation catalyzed by PRMT1 has been indicated to be
required for pre-B-cell development and mature B-cell activation,
together with B-cell translocation gene (63).
The present analysis on GC also indicated that,
besides differential expression of NCOA1, our results on low PRDM16
and high PRMT1 levels were inconsistent with the corresponding
contents of previous studies (64,65).
Thee paradoxical results may be explained by the complexity of
heterogeneous factors in GC. Furthermore, the varying expression
profiles of KAT2A, NCOA1, SMYD5 and PRMT1 among different
pathological classifications of gastric adenocarcinoma also
demonstrated the presence of heterogeneity. A previous study by our
group and other studies suggested that aberrant ATAD2 expression is
associated with histone modification, hinting at a close
correlation between ATAD2 and histone modification-associated genes
(66). As speculated, the
correlation and STRING interaction analysis indicated that ATAD2
may interact with ESR1 to regulate NCOA1 and PRMT1 in GC. Previous
studies revealed that endogenous ATAD2 acts as a co-activator for
ESR1 to activate downstream target gene expression, together with
hormone-induced ESR1 recruiting to target genes at chromatin
(67). NCOA1 also interacts with
nuclear hormone receptors, including ESR1 (26). Of note, a negative correlation
between NCOA1 and PRMT1 expression was observed in the present
study, and the cross-talk among histone modification-associated
genes was also previously reported (68). Certain limitations of the present
study should be emphasized. Only a Bioinformatics analysis of
external data was performed, and the expression of the five genes
identified and interactions between NCOA1 and other genes,
including the protein levels of ATAD2, were not verified through
experimental methods as part of the present study. As a next step,
verification in clinical samples from our center and assessment of
potential interaction mechanism among ATAD2, ESR1 and NCOA1 will be
performed. Hence, the results of such future experiments are to be
anticipated.
In conclusion, the present study performed a
comprehensive Bioinformatics analysis of the expression of histone
modification-associated genes in GC and their association with
prognosis. KAT2A, NCOA1, SMYD5, PRMT1 and PRDM16 were screened out
and aberrant expression profiles were compared between GC and
non-cancer tissues. Low NCOA1 expression was a closely associated
with poor prognosis and was identified to be an independent
predictor for GC. ATAD2 may interact with ESR1 to regulate NCOA1
and PRMT1 in GC. Due to the heterogeneity in GC, well-designed
studies with larger sample sizes are required in the future.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
This study was supported by the Science Foundation
of Liaoning Province (grant no. 20170540567).
Availability of data and materials
All datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
XM, JL, LW, TZ, XG, ZD and YZ analyzed the data. XM,
ZZ and JL designed the study and prepared the manuscript. All
authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Nagini S: Carcinoma of the stomach: A
review of epidemiology, pathogenesis, molecular genetics and
chemoprevention. World J Gastrointest Oncol. 4:156–169. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Yang WY, Gu JL and Zhen TM: Recent
advances of histone modification in gastric cancer. J Cancer Res
Ther. 10 (Suppl):S240–S245. 2014. View Article : Google Scholar
|
|
3
|
Rice JC and Allis CD: Histone methylation
versus histone acetylation: New insights into epigenetic
regulation. Curr Opin Cell Biol. 13:263–273. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Schiza V, Molina-Serrano D, Kyriakou D,
Hadjiantoniou A and Kirmizis A: N-alpha-terminal acetylation of
histone H4 regulates arginine methylation and ribosomal DNA
silencing. PLoS Genet. 9:e10038052013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sakuraba K, Yokomizo K, Shirahata A, Goto
T, Saito M, Ishibashi K, Kigawa G, Nemoto H and Hibi K: TIP60 as a
potential marker for the malignancy of gastric cancer. Anticancer
Res. 31:77–79. 2011.PubMed/NCBI
|
|
6
|
He LJ, Cai MY, Xu GL, Li JJ, Weng ZJ, Xu
DZ, Luo GY, Zhu SL and Xie D: Prognostic significance of
overexpression of EZH2 and H3k27me3 proteins in gastric cancer.
Asian Pac J Cancer Prev. 13:3173–3178. 2013. View Article : Google Scholar
|
|
7
|
Goulet I, Gauvin G, Boisvenue S and Cote
J: Alternative splicing yields protein arginine methyltransferase 1
isoforms with distinct activity, substrate specificity, and
subcellular localization. J Biol Chem. 282:33009–33021. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kahl P, Gullotti L, Heukamp LC, Wolf S,
Friedrichs N, Vorreuther R, Solleder G, Bastian PJ, Ellinger J,
Metzger E, et al: Androgen receptor coactivators lysine-specific
histone demethylase 1 and four and a half LIM domain protein 2
predict risk of prostate cancer recurrence. Cancer Res.
66:11341–11347. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hirst M and Marra MA: Epigenetics and
human disease. Int J Biochem Cell Biol. 41:136–146. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kang ZH, Wang CY, Zhang WL, Zhang JT, Yuan
CH, Zhao PW, Lin YY, Hong S, Li CY and Wang L: Histone deacetylase
HDAC4 promotes gastric cancer SGC-7901 cells progression via p21
repression. PLoS One. 9:e988942014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Das C, Lucia MS, Hansen KC and Tyler JK:
CBP/p300-mediated acetylation of histone H3 on lysine 56. Nature.
459:113–117. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Vire E, Brenner C, Deplus R, Blanchon L,
Fraga M, Didelot C, Morey L, Van Eynde A, Bernard D, Vanderwinden
JM, et al: The polycomb group protein EZH2 directly controls DNA
methylation. Nature. 439:871–874. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Na J, Lee K, Na W, Shin JY, Lee MJ, Yune
TY, Lee HK, Jung HS, Kim WS and Ju BG: Histone H3K27 Demethylase
JMJD3 in cooperation with NF-κB regulates keratinocyte wound
healing. J Invest Dermatol. 136:847–858. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Li X, Corsa CA, Pan PW, Wu L, Ferguson D,
Yu X, Min J and Dou Y: MOF and H4 K16 acetylation play important
roles in DNA damage repair by modulating recruitment of DNA damage
repair protein Mdc1. Mol Cell Biol. 30:5335–5347. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ceol CJ, Houvras Y, Jane-Valbuena J,
Bilodeau S, Orlando DA, Battisti V, Fritsch L, Lin WM, Hollmann TJ,
Ferré F, et al: The histone methyltransferase SETDB1 is recurrently
amplified in melanoma and accelerates its onset. Nature.
471:513–517. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Xiang Y, Zhu Z, Han G, Lin H, Xu L and
Chen CD: JMJD3 is a histone H3K27 demethylase. Cell Res.
17:850–857. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zheng YC, Duan YC, Ma JL, Xu RM, Zi X, Lv
WL, Wang MM, Ye XW, Zhu S, Mobley D, et al:
Triazole-dithiocarbamate based selective lysine specific
demethylase 1 (LSD1) inactivators inhibit gastric cancer cell
growth, invasion, and migration. J Med Chem. 56:8543–8560. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Rhodes DR, Kalyana-Sundaram S, Mahavisno
V, Varambally R, Yu J, Briggs BB, Barrette TR, Anstet MJ,
Kincead-Beal C, Kulkarni P, et al: Oncomine 3.0: Genes, pathways,
and networks in a collection of 18,000 cancer gene expression
profiles. Neoplasia. 9:166–180. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chandrashekar DS, Bashel B, Balasubramanya
SAH, Creighton CJ, Ponce-Rodriguez I, Chakravarthi BVSK and
Varambally S: UALCAN: A portal for facilitating tumor subgroup gene
expression and survival analyses. Neoplasia. 19:649–658. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gyorffy B, Surowiak P, Budczies J and
Lanczky A: Online survival analysis software to assess the
prognostic value of biomarkers using transcriptomic data in
non-small-cell lung cancer. PLoS One. 8:e822412013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wu G, Liu H, He H, Wang Y, Lu X, Yu Y, Xia
S, Meng X and Liu Y: miR-372 down-regulates the oncogene ATAD2 to
influence hepatocellular carcinoma proliferation and metastasis.
BMC Cancer. 14:1072014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Venkatesh S and Workman JL: Histone
exchange, chromatin structure and the regulation of transcription.
Nat Rev Mol Cell Biol. 16:178–189. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Audia JE and Campbell RM: Histone
modifications and cancer. Cold Spring Harb Perspect Biol.
8:a0195212016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chan EM, Chan RJ, Comer EM, Goulet RJ III,
Crean CD, Brown ZD, Fruehwald AM, Yang Z, Boswell HS, Nakshatri H
and Gabig TG: MOZ and MOZ-CBP cooperate with NF-kappaB to activate
transcription from NF-kappaB-dependent promoters. Exp Hematol.
35:1782–1792. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yang XJ: The diverse superfamily of lysine
acetyltransferases and their roles in leukemia and other diseases.
Nucleic Acids Res. 32:959–976. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Rasti M, Grand RJ, Mymryk JS, Gallimore PH
and Turnell AS: Recruitment of CBP/p300, TATA-binding protein, and
S8 to distinct regions at the N terminus of adenovirus E1A. J
Virol. 79:5594–5605. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Xu J, Wu RC and O'Malley BW: Normal and
cancer-related functions of the p160 steroid receptor co-activator
(SRC) family. Nat Rev Cancer. 9:615–630. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Fleming FJ, Hill AD, McDermott EW,
O'Higgins NJ and Young LS: Differential recruitment of coregulator
proteins steroid receptor coactivator-1 and silencing mediator for
retinoid and thyroid receptors to the estrogen receptor-estrogen
response element by beta-estradiol and 4-hydroxytamoxifen in human
breast cancer. J Clin Endocrinol Metab. 89:375–383. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Qin L, Chen X, Wu Y, Feng Z, He T, Wang L,
Liao L and Xu J: Steroid receptor coactivator-1 upregulates
integrin α5 expression to promote breast cancer cell
adhesion and migration. Cancer Res. 71:1742–1751. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Qin L, Liu Z, Chen H and Xu J: The steroid
receptor coactivator-1 regulates twist expression and promotes
breast cancer metastasis. Cancer Res. 69:3819–3827. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Gouon-Evans V, Rothenberg ME and Pollard
JW: Postnatal mammary gland development requires macrophages and
eosinophils. Development. 127:2269–2282. 2000.PubMed/NCBI
|
|
32
|
Frycz BA, Murawa D, Borejsza-Wysocki M,
Wichtowski M, Spychala A, Marciniak R, Murawa P, Drews M and
Jagodziński PP: mRNA expression of steroidogenic enzymes, steroid
hormone receptors and their coregulators in gastric cancer. Oncol
Lett. 13:3369–3378. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Tai H, Kubota N and Kato S: Involvement of
nuclear receptor coactivator SRC-1 in estrogen-dependent cell
growth of MCF-7 cells. Biochem Biophys Res Commun. 267:311–316.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Boorjian SA, Heemers HV, Frank I, Farmer
SA, Schmidt LJ, Sebo TJ and Tindall DJ: Expression and significance
of androgen receptor coactivators in urothelial carcinoma of the
bladder. Endocr Relat Cancer. 16:123–137. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Sheppard HM, Harries JC, Hussain S, Bevan
C and Heery DM: Analysis of the steroid receptor coactivator 1
(SRC1)-CREB binding protein interaction interface and its
importance for the function of SRC1. Mol Cell Biol. 21:39–50. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Yan H, Dobbie Z, Gruber SB, Markowitz S,
Romans K, Giardiello FM, Kinzler KW and Vogelstein B: Small changes
in expression affect predisposition to tumorigenesis. Nat Genet.
30:25–26. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
37
|
Pavón MA, Parreño M, Téllez-Gabriel M,
León X, Arroyo-Solera I, López M, Céspedes MV, Casanova I, Gallardo
A, López-Pousa A, et al: CKMT1 and NCOA1 expression as a predictor
of clinical outcome in patients with advanced-stage head and neck
squamous cell carcinoma. Head Neck. 38 (Suppl 1):E1392–E1403. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Qin L, Wu YL, Toneff MJ, Li D, Liao L, Gao
X, Bane FT, Tien JC, Xu Y, Feng Z, et al: NCOA1 directly targets
M-CSF1 expression to promote breast cancer metastasis. Cancer Res.
74:3477–3488. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Gao H, Chakraborty G, Lee-Lim AP, Mavrakis
KJ, Wendel HG and Giancotti FG: Forward genetic screens in mice
uncover mediators and suppressors of metastatic reactivation. Proc
Natl Acad Sci USA. 111:16532–16537. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Krebs AR, Karmodiya K, Lindahl-Allen M,
Struhl K and Tora L: SAGA and ATAC histone acetyl transferase
complexes regulate distinct sets of genes and ATAC defines a class
of p300-independent enhancers. Mol Cell. 44:410–423. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Xu W, Edmondson DG, Evrard YA, Wakamiya M,
Behringer RR and Roth SY: Loss of Gcn5l2 leads to increased
apoptosis and mesodermal defects during mouse development. Nat
Genet. 26:229–232. 2000. View
Article : Google Scholar : PubMed/NCBI
|
|
42
|
Sonnet M, Claus R, Becker N, Zucknick M,
Petersen J, Lipka DB, Oakes CC, Andrulis M, Lier A, Milsom MD, et
al: Early aberrant DNA methylation events in a mouse model of acute
myeloid leukemia. Genome Med. 6:342014. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Li Y, Zhu R, Wang W, Fu D, Hou J, Ji S,
Chen B, Hu Z, Shao X, Yu X, et al: Arginine Methyltransferase 1 in
the nucleus accumbens regulates behavioral effects of cocaine. J
Neurosci. 35:12890–12902. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Boisvert FM, Rhie A, Richard S and Doherty
AJ: The GAR motif of 53BP1 is arginine methylated by PRMT1 and is
necessary for 53BP1 DNA binding activity. Cell Cycle. 4:1834–1841.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Qian C and Zhou MM: SET domain protein
lysine methyltransferases: Structure, specificity and catalysis.
Cell Mol Life Sci. 63:2755–2763. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Abu-Farha M, Lanouette S, Elisma F,
Tremblay V, Butson J, Figeys D and Couture JF: Proteomic analyses
of the SMYD family interactomes identify HSP90 as a novel target
for SMYD2. J Mol Cell Biol. 3:301–308. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Wang Y, Qu Y, Zhang XL, Xing J, Niu XL,
Chen X and Li ZM: Autocrine production of interleukin-6 confers
ovarian cancer cells resistance to tamoxifen via ER isoforms and
SRC-1. Mol Cell Endocrinol. 382:791–803. 2012. View Article : Google Scholar
|
|
48
|
Liao HW, Hsu JM, Xia W, Wang HL, Wang YN,
Chang WC, Arold ST, Chou CK, Tsou PH, Yamaguchi H, et al:
PRMT1-mediated methylation of the EGF receptor regulates signaling
and cetuximab response. J Clin Invest. 125:4529–4543. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Tan SX, Hu RC, Xia Q, Tan YL, Liu JJ, Gan
GX and Wang LL: The methylation profiles of PRDM promoters in
non-small cell lung cancer. Onco Targets Ther. 11:2991–3002. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Peng X, Xue H, Lü L, Shi P and Wang J and
Wang J: Accumulated promoter methylation as a potential biomarker
for esophageal cancer. Oncotarget. 8:679–691. 2017.PubMed/NCBI
|
|
51
|
Bibi F, Ali I, Naseer MI, Ali Mohamoud HS,
Yasir M, Alvi SA, Jiman-Fatani AA, Sawan A and Azhar EI: Detection
of genetic alterations in gastric cancer patients from Saudi Arabia
using comparative genomic hybridization (CGH). PLoS One.
13:e02025762018. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Burghel GJ, Lin WY, Whitehouse H, Brock I,
Hammond D, Bury J, Stephenson Y, George R and Cox A: Identification
of candidate driver genes in common focal chromosomal aberrations
of microsatellite stable colorectal cancer. PLoS One. 8:e838592013.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Ma YS, Wu TM, Lv ZW, Lu GX, Cong XL, Xie
RT, Yang HQ, Chang ZY, Sun R, Chai L, et al: High expression of
miR-105-1 positively correlates with clinical prognosis of
hepatocellular carcinoma by targeting oncogene NCOA1. Oncotarget.
8:11896–11905. 2017.PubMed/NCBI
|
|
54
|
Guo J, Cao R, Yu X, Xiao Z and Chen Z:
MicroRNA-223-3p inhibits human bladder cancer cell migration and
invasion. Tumour Biol. 39:10104283176916782017. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Minchenko OH, Garmash IA, Minchenko DO,
Kuznetsova AY and Ratushna OO: Inhibition of IRE1 modifies hypoxic
regulation of G6PD, GPI, TKT, TALDO1, PGLS and RPIA genes
expression in U87 glioma cells. Ukr Biochem J. 89:38–49. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Ding SZ, Goldberg JB and Hatakeyama M:
Helicobacter pylori infection, oncogenic pathways and
epigenetic mechanisms in gastric carcinogenesis. Future Oncol.
6:851–862. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Tzelepis K, Koike-Yusa H, De Braekeleer E,
Li Y, Metzakopian E, Dovey OM, Mupo A, Grinkevich V, Li M, Mazan M,
et al: A CRISPR dropout screen identifies genetic vulnerabilities
and therapeutic targets in acute myeloid leukemia. Cell Rep.
17:1193–1205. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Zhu S, Xu Y, Song M, Chen G, Wang H, Zhao
Y, Wang Z and Li F: PRDM16 is associated with evasion of apoptosis
by prostatic cancer cells according to RNA interference screening.
Mol Med Rep. 14:3357–3361. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Blanc RS, Vogel G, Li X, Yu Z, Li S and
Richard S: Arginine methylation by PRMT1 regulates muscle stem cell
fate. Mol Cell Biol. 37(pii): e00457–16. 2017.PubMed/NCBI
|
|
60
|
Kidder BL, He R, Wangsa D, Padilla-Nash
HM, Bernardo MM, Sheng S, Ried T and Zhao K: SMYD5 controls
heterochromatin and chromosome integrity during embryonic stem cell
differentiation. Cancer Res. 77:6729–6745. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Katsuno Y, Qin J, Oses-Prieto J, Wang H,
Jackson-Weaver O, Zhang T, Lamouille S, Wu J, Burlingame A, Xu J
and Derynck R: Arginine methylation of SMAD7 by PRMT1 in
TGF-β-induced epithelial-mesenchymal transition and epithelial
stem-cell generation. J Biol Chem. 293:13059–13072. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Gao B, Kong Q, Zhang Y, Yun C, Dent SYR,
Song J, Zhang DD, Wang Y, Li X and Fang D: The histone
acetyltransferase Gcn5 positively regulates T Cell activation. J
Immunol. 198:3927–3938. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Wu GS and Bassing CH: Flip the switch:
BTG2-PRMT1 protein complexes antagonize pre-B-cell proliferation to
promote B-cell development. Cell Mol Immunol. 15:808–811. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Lei Q, Liu X, Fu H, Sun Y, Wang L, Xu G,
Wang W, Yu Z, Liu C, Li P, et al: miR-101 reverses hypomethylation
of the PRDM16 promoter to disrupt mitochondrial function in
astrocytoma cells. Oncotarget. 7:5007–5022. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Altan B, Yokobori T, Ide M, Mochiki E,
Toyomasu Y, Kogure N, Kimura A, Hara K, Bai T, Bao P, et al:
Nuclear PRMT1 expression is associated with poor prognosis and
chemosensitivity in gastric cancer patients. Gastric Cancer.
19:789–797. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Wu G, Lu X, Wang Y, He H, Meng X, Xia S,
Zhen K and Liu Y: Epigenetic high regulation of ATAD2 regulates the
Hh pathway in human hepatocellular carcinoma. Int J Oncol.
45:351–361. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Zou JX, Revenko AS, Li LB, Gemo AT and
Chen HW: ANCCA, an estrogen-regulated AAA+ ATPase coactivator for
ERalpha, is required for coregulator occupancy and chromatin
modification. Proc Natl Acad Sci USA. 104:18067–18072. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Jin Q, Wang C, Kuang X, Feng X, Sartorelli
V, Ying H, Ge K and Dent SY: Gcn5 and PCAF regulate PPARγ and
Prdm16 expression to facilitate brown adipogenesis. Mol Cell Biol.
34:3746–3753. 2014. View Article : Google Scholar : PubMed/NCBI
|


















