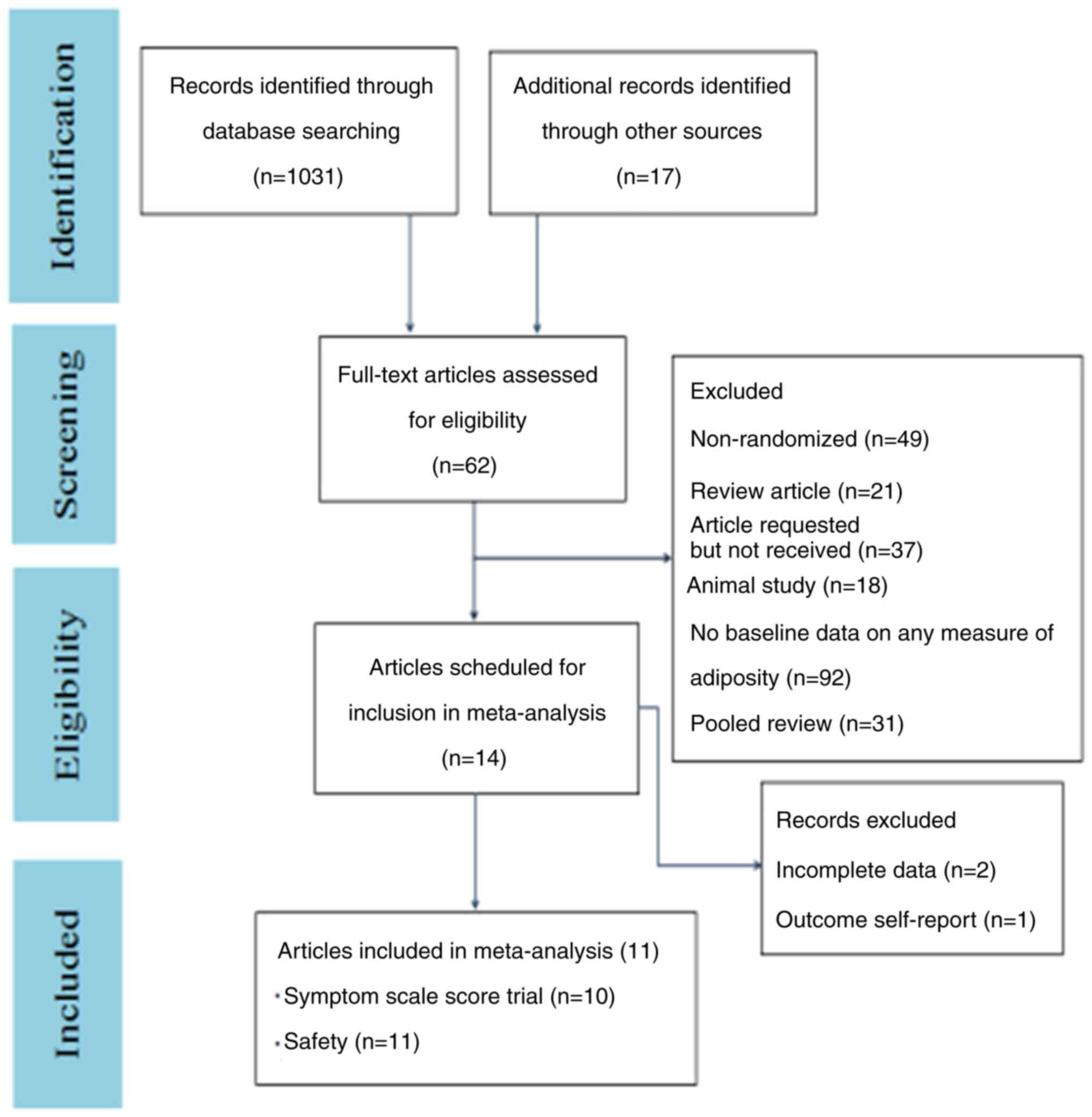
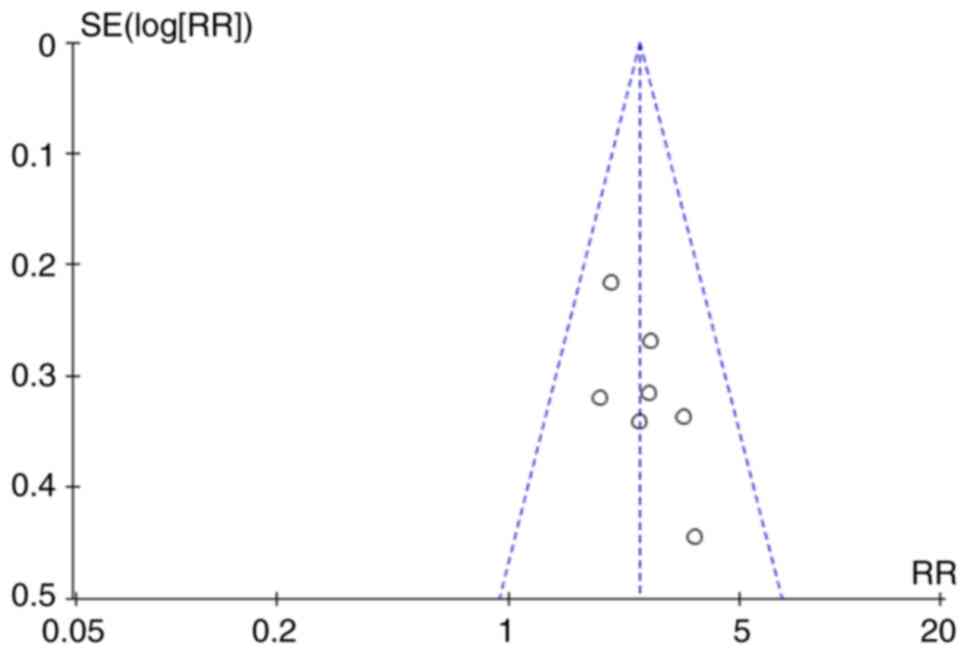
|
1
|
Hirschfeld RM: Differential diagnosis of
bipolar disorder and major depressive disorder. J Affect Disord.
169 (Suppl 1):S12–S16. 2014.PubMed/NCBI View Article : Google Scholar
|
|
2
|
DE Hert M, Correll CU, Bobes J,
Cetkovich-Bakmas M, Cohen D, Asai I, Detraux J, Gautam S, Möller
HJ, Ndetei DM, et al: Physical illness in patients with severe
mental Disorders. I. Prevalence, impact of medications and
disparities in health care. World Psychiatry. 10:52–77.
2011.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Grigoriadis S, VonderPorten EH,
Mamisashvili L, Eady A, Tomlinson G, Dennis CL, Koren G, Steiner M,
Mousmanis P, Cheung A, et al: The effect of prenatal antidepressant
exposure on neonatal adaptation: A systematic review and
meta-analysis. J Clin Psychiatry. 74:e309–e320. 2013.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Hair P, Cameron F and Garnock-Jones KP:
Levomilnacipran extended-release: First global approve. Drugs.
73:1639–1645. 2013.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Sartorius N: Comorbidity of mental and
physical disorders: A main challenge to medicine in the 21st
century. Psychiatr Danub. 25 (Suppl 1):S4–S5. 2013.PubMed/NCBI
|
|
6
|
He S, Yang XM and Li HF: Levomilnacipran:
A new drug for treatment of major depressive disorder. Chin J New
Drugs Clin Rem. 34:418–422. 2015.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Bang-Andersen B, Ruhland T, Jørgensen M,
Smith G, Frederiksen K, Jensen KG, Zhong H, Nielsen SM, Hogg S,
Mørk A and Stensbøl TB: Discovery of
1-[2-(2,4-dimethylphenylsulfanyl)phenyl]piperazine (Lu AA21004): A
novel multimodal compound for the treatment of major depressive
disorder. J Med Chem. 54:3206–3221. 2011.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Pehrson AL, Cremers T, Bétry C, van der
Hart MG, Jørgensen L, Madsen M, Haddjeri N, Ebert B and Sanchez C:
Lu AA21004, a novel multimodal antidepressant, produces regionally
selective increases of multiple neurotransmitters a rat
microdialysis and electrophysiology study. Eur
Neuropsychopharmacol. 23:133–145. 2013.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Gibb A and Deeks ED: Vortioxetine: First
global approval. Drugs. 74:135–145. 2014.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Boulenger JP, Loft H and Florea I: A
randomized clinical study of Lu AA21004 in theprevention of relapse
in patients with major depressive disorder. J Psychopharmacol.
26:1408–1416. 2012.PubMed/NCBI View Article : Google Scholar
|
|
11
|
FDA. Vortioxetine(EB/OL). http://www.accessdata.fda.gov/drugsatfda_docs/label/2013/204447s000lbl.pdf,
2013-09-30.
|
|
12
|
Li G, Wang X and Ma D: Vortioxetine versus
duloxetine in the treatment of patients with major depressive
disorder: A Meta-analysis of randomized controlled trials. Clin
Drug Investig. 36:509–517. 2016.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Pae CU, Wang SM, Han C, Lee SJ, Patkar AA,
Masand PS and Serretti A: Vortioxetine: A meta-analysis of 12
short-term, randomized, placebo-controlled clinical trials for the
treatment of major depressive disorder. J Psychiatry Neurosci.
40:174–186. 2015.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Baldwin DS, Chrones L, Florea I, Nielsen
R, Nomikos GG, Palo W and Reines E: The safety and tolerability of
vortioxetine: Analysis of data from randomized
Placebo-controlledtrials and open-label extension studies. J
Psychopharmacol. 30:242–252. 2016.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Page MJ and Moher D: Evaluations of the
uptake and impact of the preferred reporting items for systematic
reviews and Meta-analysis (PRISMA) statement and extensions: A
scoping review. Syst Rev. 6(263)2017.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Sandini M, Mattavelli I, Nespoli L, Uggeri
F and Gianotti L: Systematic review and meta-analysis of sutures
coated with triclosan for the prevention of surgical site infection
after elective colorectal surgery according to the PRISMA
statement. Medicine (Baltimore). 95(e4057)2016.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Panic N, Leoncini E, de Belvis G,
Ricciardi W and Boccia S: Evaluation of the endorsement of the
preferred reporting items for systematic reviews and meta-analysis
(PRISMA) statement on the quality of published systematic review
and meta-analyses. PLoS One. 8(e83138)2013.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Durak S, Ercan ES, Ardic UA, Yuce D, Ercan
E and Ipci M: Effect of methylphenidate on neurocognitive test
battery: An evaluation according to the diagnostic and statistical
manual of mental disorders, fourth edition, subtypes. J Clin
Psychopharmacol. 34:467–474. 2014.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Shabsigh R and Rowland D: The Diagnostic
and statistical manual of mental distal disorders, Fouth edition,
text revision as an appropriate diagnostic for premature
ejaculation. J Sex Med. 4:1468–1478. 2007.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Faiad Y, Khoury B, Daouk S, Maj M, Keeley
J, Gureje O and Reed G: Frequency of use of the international
classification of diseases ICD-10 diagnostic categories for mental
and behavioural disorders across world regions. Epidemiol Psychiatr
Sci. 11:1–9. 2017.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Janca A, Ustün TB, Early TS and Sartorius
N: The ICD-10 symptom checklist: A companion to the ICD-10
classification of mental and behavioural disorders. Soc Psychiatry
Psychiatr Epidemiol. 28:239–242. 1993.PubMed/NCBI View Article : Google Scholar
|
|
22
|
International Advisory Group for the
Revision of ICD-10 Mental and Behavioural Disorders. A conceptual
framework for the revision of the ICD-10 classification of mental
and behavioural disorders. World Psychiatry. 10:86–92.
2011.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Khan A, Lewis C and Lindenmayer JP: Use of
non-parametric item response theory to develop a shortened version
of the Positive and Negative Syndrome Scale (PANSS). BMC
Psychiatry. 11(178)2011.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Garcia-Caballero A, Torrens-Lluch M,
Ramírez-Gendrau I, Garrido G, Vallès V and Aragay N: The efficacy
of motivational intervention and cognitive-behavioral therapy for
pathological gambling. Adicciones. 30:219–224. 2018.PubMed/NCBI View Article : Google Scholar : (In English,
Spanish).
|
|
25
|
Health Quality Ontario. Psychotherapy for
Major depressive disorder and generalized anxiety disorder: A
health technology assessment. Ont Health Technol Assess Ser.
17:1–167. 2017.PubMed/NCBI
|
|
26
|
Hedges DW, Brown BL and Shwalb DA: A
direct comparison of effect sizes from the clinical global
impression-improvement scale to effect sizes from other rating
scales in controlled trials of adult social anxiety disorder. Hum
Psychopharmacol. 24:35–40. 2009.PubMed/NCBI View
Article : Google Scholar
|
|
27
|
Higgins JPT and Green S, (eds.): Cochrane
Handbook for Systematic Reviews of Interventions Version 5.1.0. The
Cochrane Collaboration, Available from: http://handbook.cochrane.org, (Updated March
2011).
|
|
28
|
Berhan A and Barker A: Vortioxetine in the
treatment of adult patients with major depressive disorder: A
meta-analysis of randomized double-blind controlled trials. BMC
Psychiatry. 14:276–283. 2014.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Braga VL, Rocha LPDS, Bernardo DD, Cruz CO
and Riera R: What do Cochrane systematic reviews say about
probiotics as preventive interventions? Sao Paulo Med J.
135:578–586. 2017.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Henigsberg N, Mahableshwarkar AR, Jacobsen
P, Chen Y and Thase ME: A randomized, double-blind,
placebo-controlled 8-week trial of the efficacy and tolerability of
multiple doses of Lu AA21004 in adults with major depressive
disorder. J Clin Psychiatry. 73:953–959. 2012.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Nishimura A, Aritomi Y, Sasai K, Kitagawa
T and Mahableshwarkar AR: Randomized, double-blind,
placebo-controlled 8-week trial of the efficacy, safety, and
tolerability of 5, 10, and 20 mg/day vortioxetine in adults with
major depressive disorder. Psychiatry Clin Neurosci. 72:64–72.
2018.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Jain R, Mahableshwarkar AR, Jacobsen PL,
Chen Y and Thase ME: A randomized, double-blind, placebo-controlled
6-wk trial of the efficacy and tolerability of 5 mg vortioxetine in
adults with major depressive disorder. Int J Neuropsychopharmacol.
16:313–321. 2013.PubMed/NCBI View Article : Google Scholar
|
|
33
|
McIntyre RS, Lophaven S and Olsen CK: A
randomized, double-blind, placebo-controlled study of vortioxetine
on cognitive function in depressed adults. Int J
Neuropsychopharmacol. 17:1557–1567. 2014.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Mahableshwarkar AR, Jacobsen PL and Chen
Y: A randomized, double-blind trial of 2.5 mg and 5 mg vortioxetine
(Lu AA21004) versus placebo for 8 weeks in adults with major
depressive disorder. Curr Med Res Opin. 29:217–226. 2013.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Baldwin DS, Loft H and Dragheim M: A
randomised, double-blind, placebo controlled duloxetine-referenced,
fixed-dose study of three dosages of Lu AA21004 in acute treatment
of major depressive disorder (MDD). Eur Neuropsychopharmacol.
22:482–491. 2012.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Katona C, Hansen T and Olsen CK: A
randomized, double-blind, placebo-controlled,
duloxetine-referenced, fixed-dose study comparing the efficacy and
safety of Lu AA21004 in elderly patients with major depressive
disorder. Int Clin Psychopharmacol. 27:215–223. 2012.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Mahableshwarkar AR, Zajecka J, Jacobson W,
Chen Y and Keefe RS: A randomized, placebo-controlled,
active-reference, double-blind, flexible-dose study of efficacy of
vortioxetine on cognitive function in major depressive disorder.
Neuropsychopharmacology. 40:2025–2037. 2015.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Boulenger JP, Loft H and Olsen CK:
Efficacy and safety of vortioxetine (Lu AA21004), 15 and 20 mg/day:
A randomized, double-blind, placebo-controlled,
duloxetine-referenced study in the acute treatment of adult
patients with major depressive disorder. Int Clin Psychopharmacol.
29:138–149. 2014.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Mahableshwarkar AR, Jacobsen PL, Chen Y,
Serenko M and Trivedi MH: A randomized, double-blind,
duloxetine-referenced study comparing efficacy and tolerability of
2 fixed doses of vortioxetine in the acute treatment of adults with
MDD. Psychopharmacology (Berl). 232:2061–2070. 2015.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Alvarez E, Perez V, Dragheim M, Loft H and
Artigas F: A double-blind, randomized, placebo-controlled, active
reference study of Lu AA21004 in patients with major depressive
disorder. Int J Neuropsychopharmacol. 15:589–600. 2012.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Ma XR, Hou CL and Xia FJ: Present
situation and challenges of somatic syndromes in patients with
serious mental illness. Chin J Behav Med Brain Sci. 23:181–183.
2014.
|
|
42
|
Shen HN, Lu CL and Yang HH: Increased
risks of acute organ dysfunction and mortality in intensive care
unit patients with schizophrenia: A nationwide population-based
study. Psychosom Med. 73:620–626. 2011.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Lawrence D and Kisely D: Inequalities in
healthcare provision for people with severe mental iuness. J
Psychopharmacol. 24 (4 Suppl):S6l–S68. 2010.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Ritchie S and Muldoon L: Cardiovascular
preventive care for patients with serious mental illness. Can Fam
Physician. 63:e483–e487. 2017.PubMed/NCBI
|
|
45
|
Kilbourne AM, Welsh D, McCarthy JF, Post
EP and Blow FC: Quality of care for cardiovascular disease-related
conditions in patients with and without mental disorders. J Gen
Intern Med. 23:1628–1633. 2008.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Zhao G, Ford ES, Dhingra S, Li C, Strine
TW and Mokdad AH: Depression and anxiety among US adults:
Associations with body mass index. Int J Obes (Lond). 33:257–266.
2009.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Waller JA, Chen F and Sanchez C:
Vortioxetine promotes maturation of dendritic spines in vitro: A
comparative study in hippocampal cultures. Neuropharmacology.
103:143–154. 2016.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Chen F, du Jardin KG, Waller JA, Sanchez
C, Nyengaard JR and Wegener G: Vortioxetine promotes early changes
in dendritic morphology compared to fluoxetine in rat hippocampus.
Eur Neuropsychopharmacol. 26:234–245. 2016.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Smagin GN, Song D, Budac DP, Waller JA, Li
Y, Pehrson AL and Sánchez C: Histamine may contribute
Tovortioxetine's procognitive effects; possibly through an
orexigenic mechanism. Prog Neuropsychopharmacol Biol Psychiatry.
68:25–30. 2016.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Meeker AS, Herink MC, Haxby DG and Hartung
DM: The safety and efficacy of vortioxetine for acute treatment of
major depressive disorder: A systematic review and meta-analysis.
Syst Rev. 4(21)2015.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Zhang X, Cai Y, Hu X, Lu CY, Nie X and Shi
L: Systematic review and meta-analysis of vortioxetine for the
treatment of major depressive disorder in adults. Front Psychiatry.
13(922648)2022.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Thase ME, Mahableshwarkar AR, Dragheim M,
Loft H and Vieta E: A meta-analysis of randomized,
placebo-controlled trials of vortioxetine for the treatment of
major depressive disorder in adults. Eur Neuropsychopharmacol.
26:979–993. 2016.PubMed/NCBI View Article : Google Scholar
|