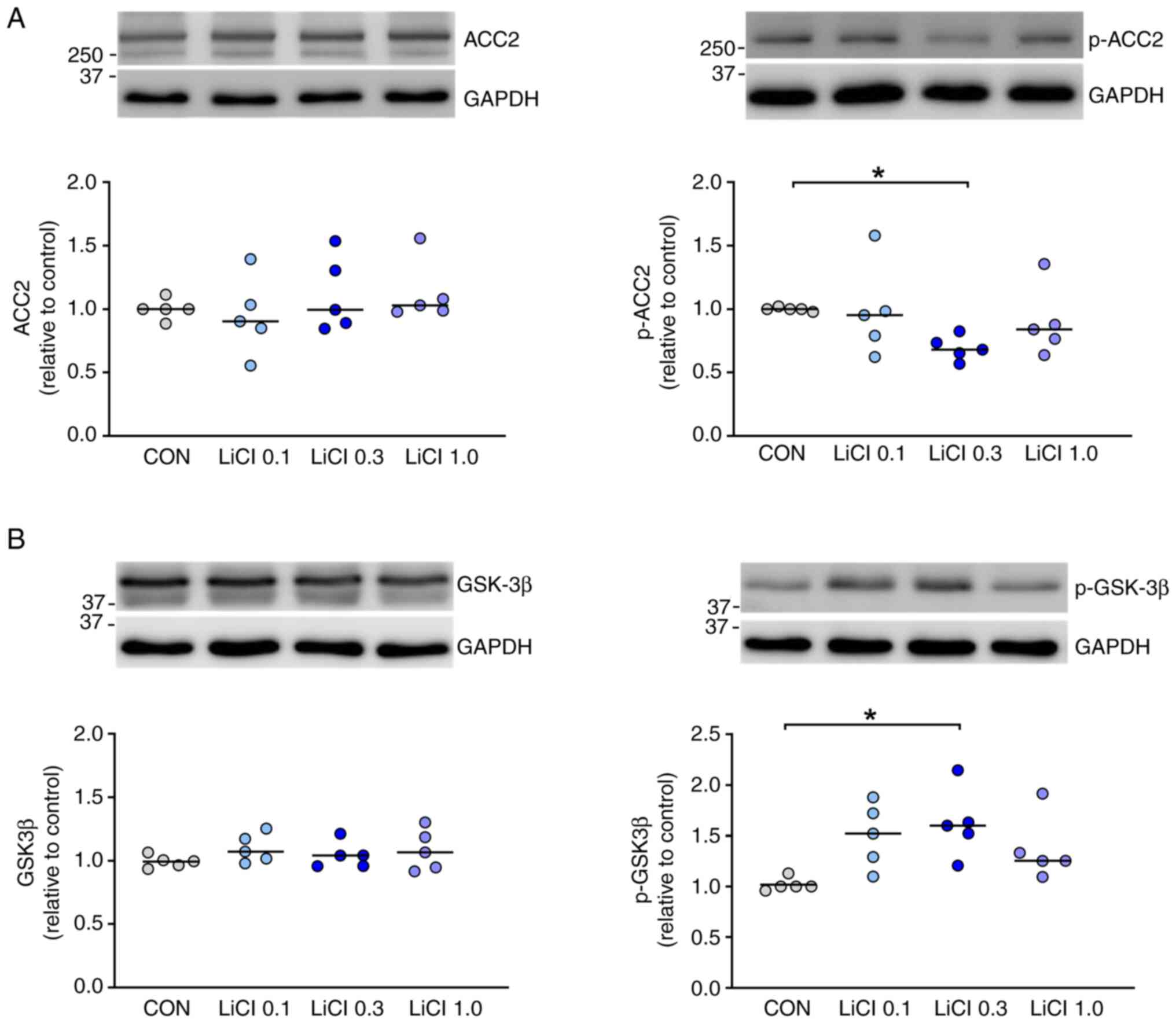
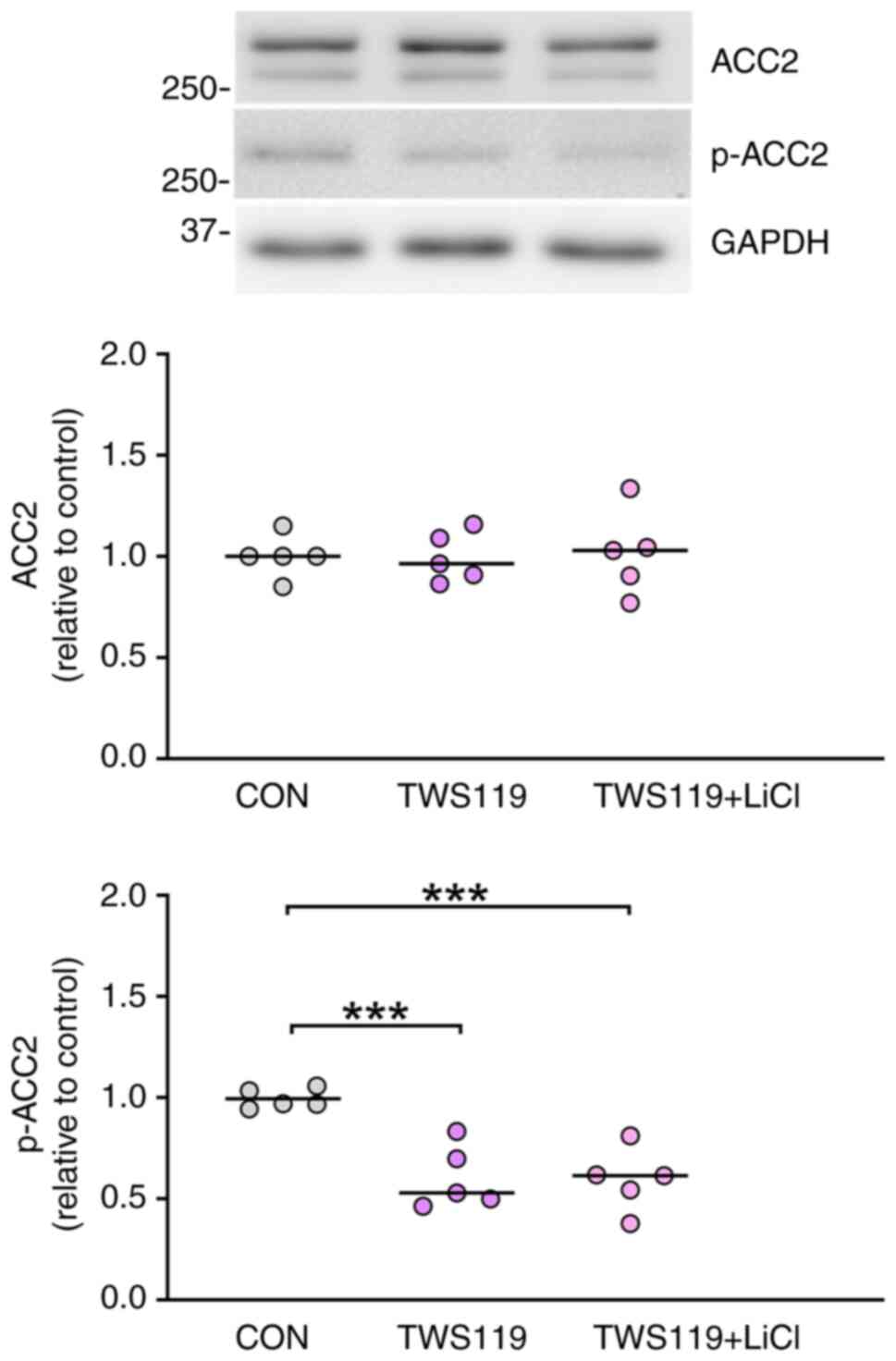
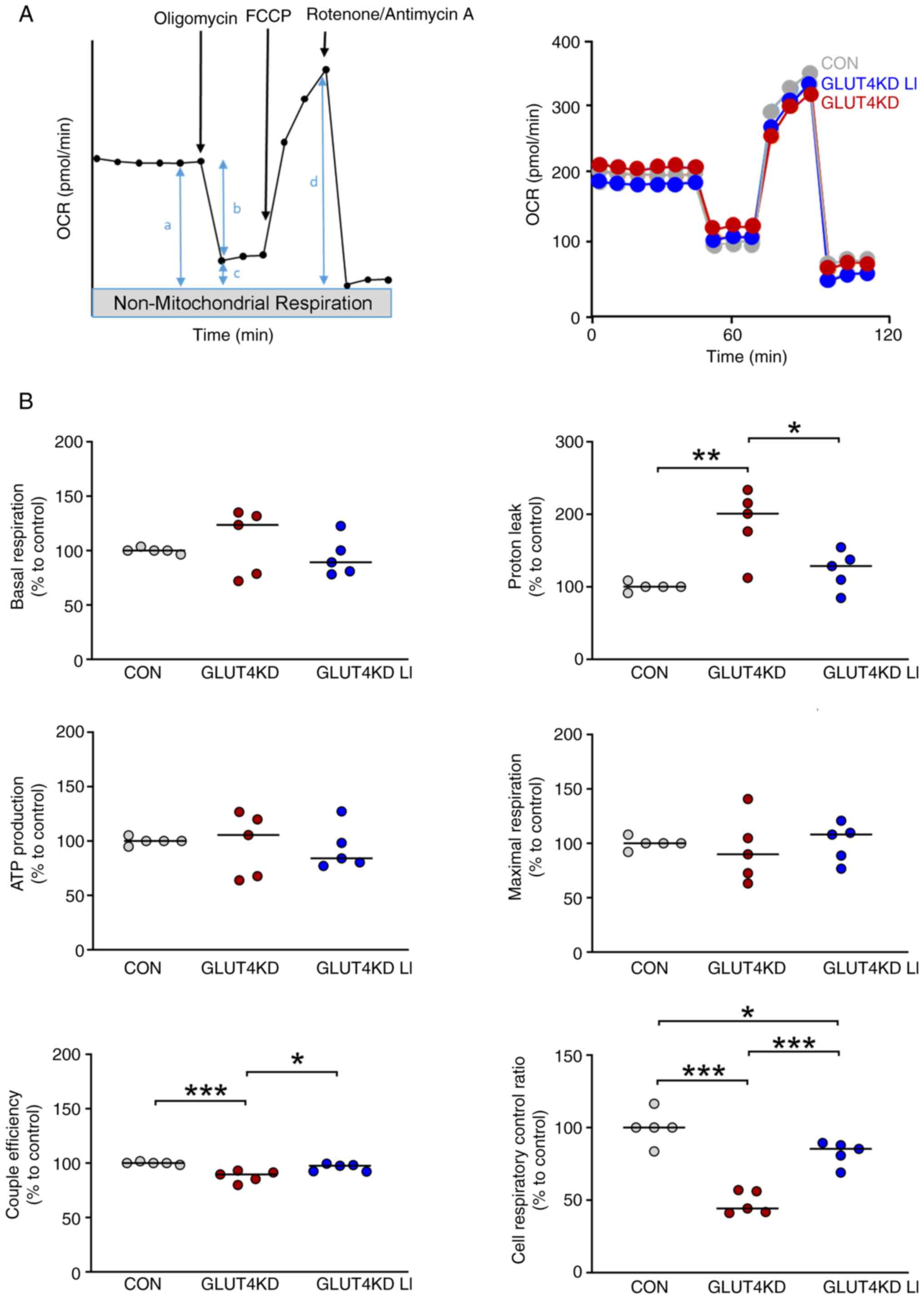
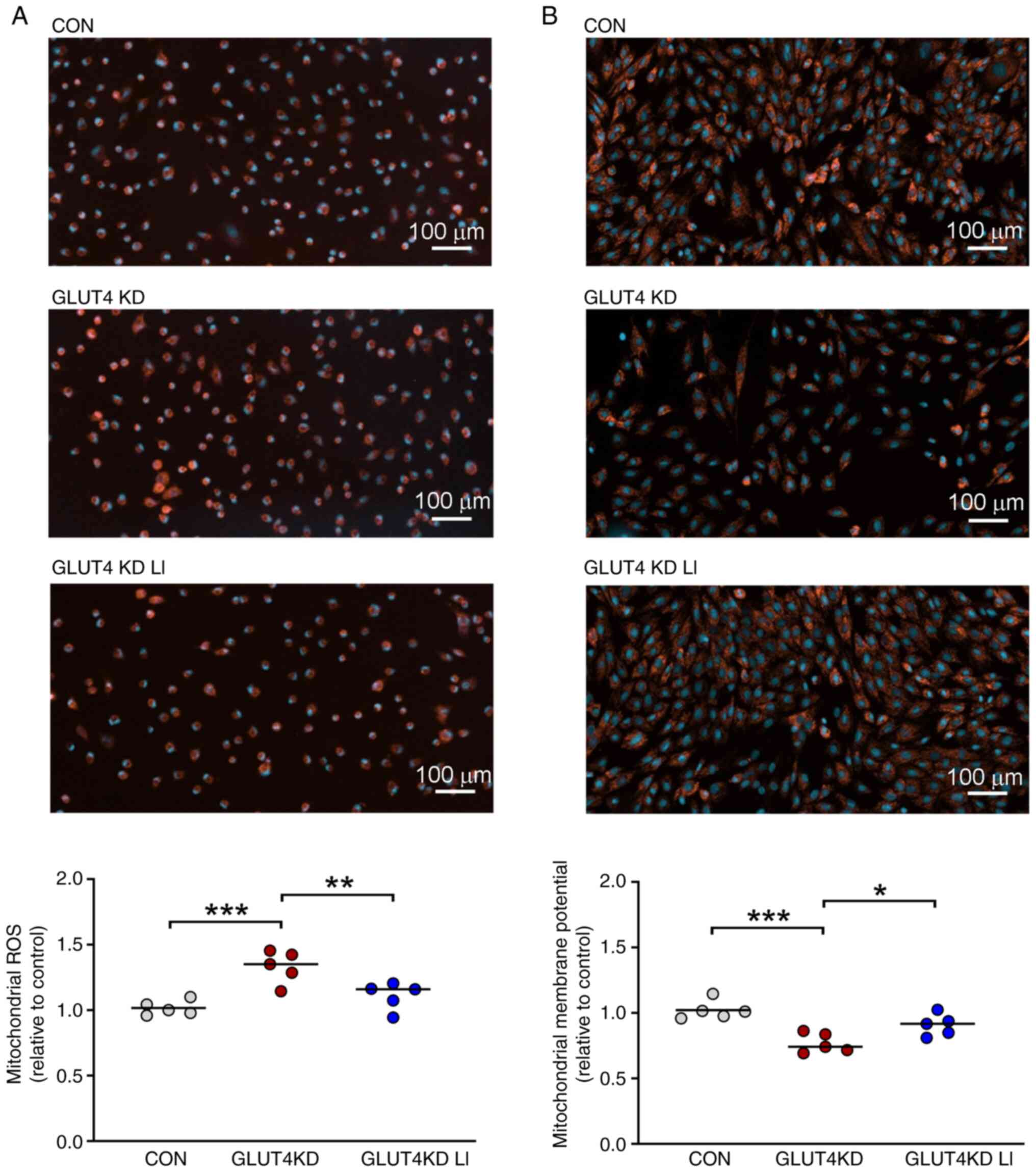
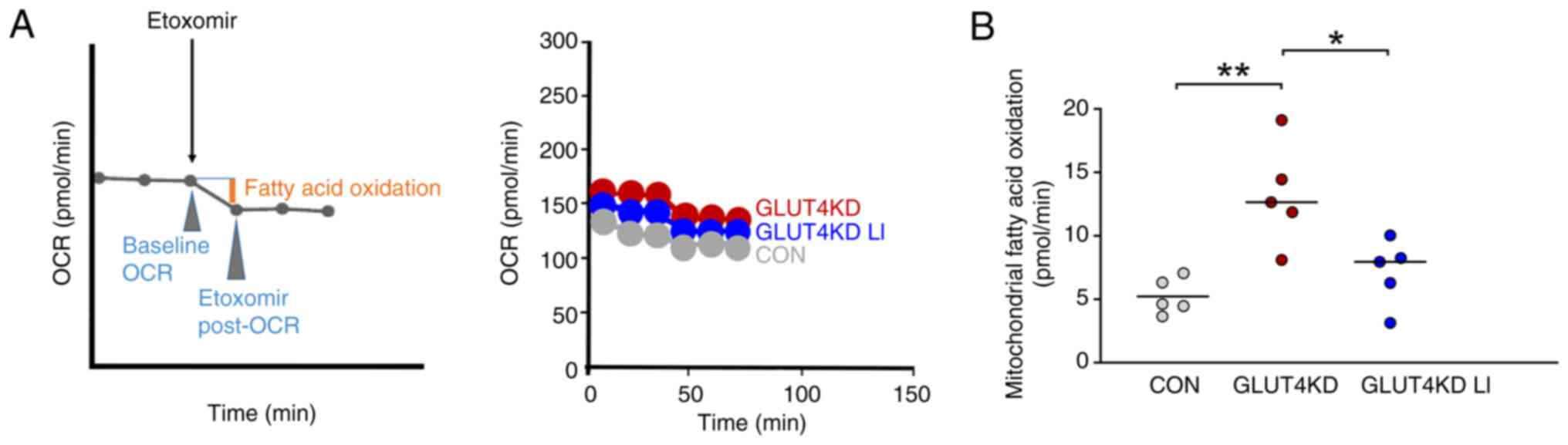
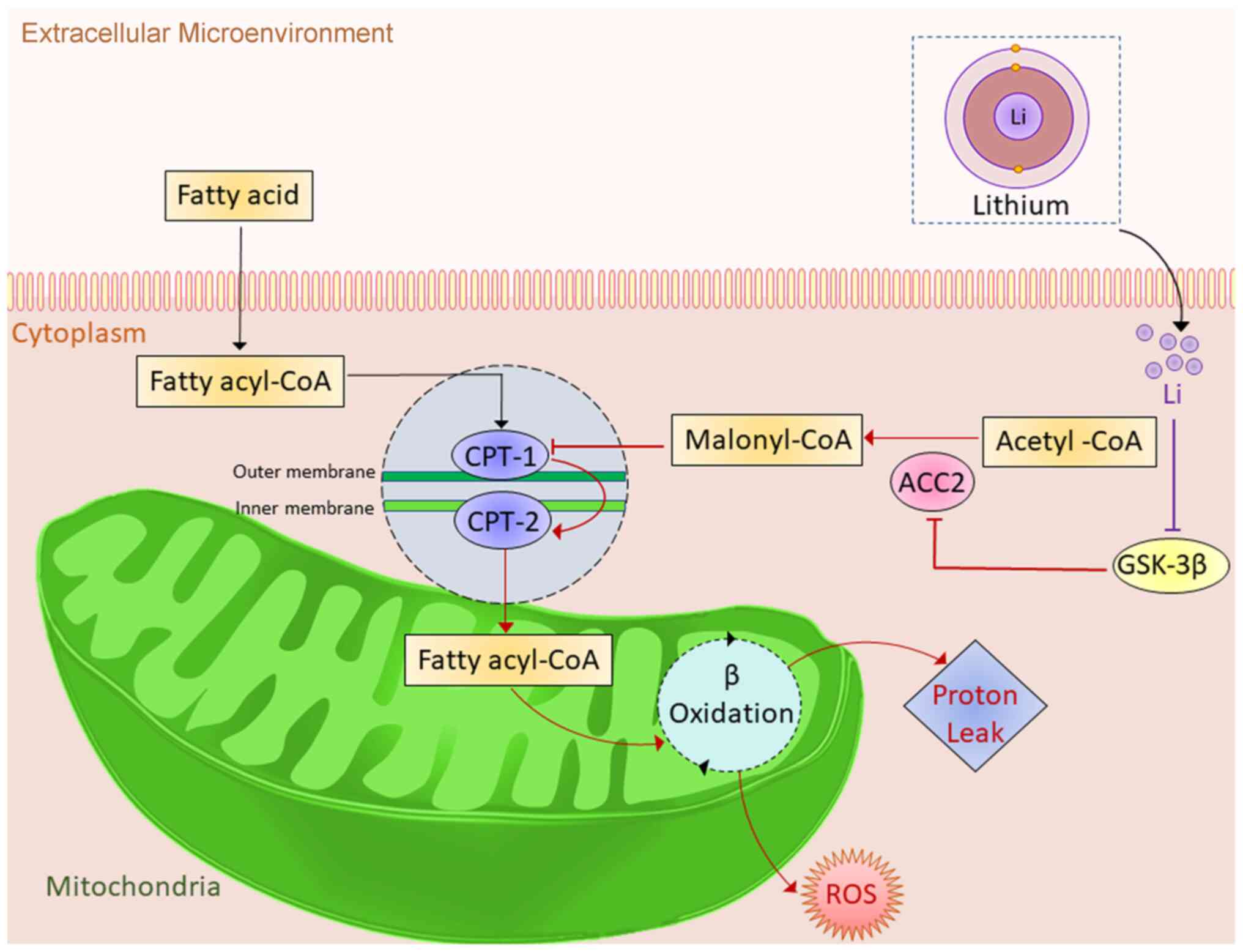
|
1
|
Bertero E and Maack C: Metabolic
remodelling in heart failure. Nat Rev Cardiol. 15:457–470.
2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Fillmore N, Mori J and Lopaschuk GD:
Mitochondrial fatty acid oxidation alterations in heart failure,
ischaemic heart disease and diabetic cardiomyopathy. Br J
Pharmacol. 171:2080–2090. 2014.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Lesnefsky EJ, Chen Q and Hoppel CL:
Mitochondrial metabolism in aging heart. Circ Res. 118:1593–611.
2016.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Lopaschuk GD, Karwi QG, Tian R, Wende AR
and Abel ED: Cardiac energy metabolism in heart failure. Circ Res.
128:1487–1513. 2021.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Marin W, Marin D, Ao X and Liu Y:
Mitochondria as a therapeutic target for cardiac
ischemia-reperfusion injury (Review). Int J Mol Med. 47:485–499.
2021.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Fukushima A and Lopaschuk GD: Cardiac
fatty acid oxidation in heart failure associated with obesity and
diabetes. Biochim Biophys Acta. 1861:1525–1534. 2016.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Lkhagva B, Lee TW, Lin YK, Chen YC, Chung
CC, Higa S and Chen YJ: Disturbed cardiac metabolism triggers
atrial arrhythmogenesis in diabetes mellitus: Energy substrate
alternate as a potential therapeutic intervention. Cells.
11(2915)2022.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Zuurbier CJ, Bertrand L, Beauloye CR,
Andreadou I, Ruiz-Meana M, Jespersen NR, Kula-Alwar D, Prag HA,
Eric Botker H, Dambrova M, et al: Cardiac metabolism as a driver
and therapeutic target of myocardial infarction. J Cell Mol Med.
24:5937–5954. 2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Tong L: Structure and function of
biotin-dependent carboxylases. Cell Mol Life Sci. 70:863–891.
2013.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Brownsey RW, Boone AN, Elliott JE, Kulpa
JE and Lee WM: Regulation of acetyl-CoA carboxylase. Biochem Soc
Trans. 34(Pt 2):223–227. 2006.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Malhi GS, Gessler D and Outhred T: The use
of lithium for the treatment of bipolar disorder: Recommendations
from clinical practice guidelines. J Affect Disord. 217:266–280.
2017.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Machado-Vieira R: Lithium, stress and
resilience in bipolar disorder: Deciphering this key homeostatic
synaptic plasticity regulator. J Affect Disord. 233:92–99.
2018.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Ochoa ELM: Lithium as a neuroprotective
agent for bipolar disorder: An overview. Cell Mol Neurobiol.
42:85–97. 2022.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Singulani MP, De Paula VJR and Forlenza
OV: Mitochondrial dysfunction in Alzheimer's disease: Therapeutic
implications of lithium. Neurosci Lett. 760(136078)2021.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Castillo-Quan JI, Li L, Kinghorn KJ,
Ivanov DK, Tain LS, Slack C, Kerr F, Nespital T, Thornton J, Hardy
J, et al: Lithium promotes longevity through GSK3/NRF2-dependent
hormesis. Cell Rep. 15:638–650. 2016.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Zarse K, Terao T, Tian J, Iwata N, Ishii N
and Ristow M: Low-dose lithium uptake promotes longevity in humans
and metazoans. Eur J Nutr. 50:387–389. 2011.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Chen PH, Hsiao CY, Chiang SJ, Shen RS, Lin
YK, Chung KH and Tsai SY: Cardioprotective potential of lithium and
role of fractalkine in euthymic patients with bipolar disorder.
Aust N Z J Psychiatry. 57:104–114. 2023.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Chen PH, Hsiao CY, Chiang SJ and Tsai SY:
Association of lithium treatment with reduced left ventricular
concentricity in patients with bipolar disorder. Psychiatry Clin
Neurosci. 77:59–61. 2023.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Snitow ME, Bhansali RS and Klein PS:
Lithium and therapeutic targeting of GSK-3. Cells.
10(255)2011.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Domoto T, Pyko IV, Furuta T, Miyashita K,
Uehara M, Shimasaki T, Nakada M and Minamoto T: Glycogen synthase
kinase-3β is a pivotal mediator of cancer invasion and resistance
to therapy. Cancer Sci. 170:1363–1372. 2016.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Terrand J, Bruban V, Zhou L, Gong W, El
Asmar Z, May P, Zurhove K, Haffner P, Philippe C, Woldt E, et al:
LRP1 controls intracellular cholesterol storage and fatty acid
synthesis through modulation of Wnt signaling. J Biol Chem.
284:381–388. 2009.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Lkhagva B, Kao YH, Lee TI, Lee TW, Cheng
WL and Chen YJ: Activation of Class I histone deacetylases
contributes to mitochondrial dysfunction in cardiomyocytes with
altered complex activities. Epigenetics. 13:376–385.
2018.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Brand MD and Nicholls DG: Assessing
mitochondrial dysfunction in cells. Biochem J. 435:297–312.
2011.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Nisr RB and Affourtit C: Insulin acutely
improves mitochondrial function of rat and human skeletal muscle by
increasing coupling efficiency of oxidative phosphorylation.
Biochim Biophys Acta. 1837:270–276. 2014.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Chen PH, Chung CC, Lin YF, Kao YH and Chen
YJ: Lithium reduces migration and collagen synthesis activity in
human cardiac fibroblasts by inhibiting store-operated
Ca2+ entry. Int J Mol Sci. 22(842)2021.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Eaton S: Control of mitochondrial
beta-oxidation flux. Prog Lipid Res. 41:197–239. 2022.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Schlaepfer IR and Joshi M: CPT1A-mediated
fat oxidation, mechanisms and therapeutic potential. Endocrinology.
161(bqz046)2020.PubMed/NCBI View Article : Google Scholar
|
|
28
|
McCarthy CP, Mullins KV and Kerins DM: The
role of trimetazidine in cardiovascular disease: Beyond an
anti-anginal agent. Eur Heart J Cardiovasc Pharmacother. 2:266–272.
2016.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Zhao C, Jin C, He X and Xiang M: The
efficacy of trimetazidine in non-ischemic heart failure patients: A
meta-analysis of randomized controlled trials. Rev Cardiovasc Med.
22:1451–1459. 2021.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Karwi QG, Sun Q and Lopaschuk GD: The
contribution of cardiac fatty acid oxidation to diabetic
cardiomyopathy severity. Cells. 10(3259)2021.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Shu H, Peng Y, Hang W, Zhou N and Wang DW:
Trimetazidine in heart failure. Front Pharmacol.
11(569132)2021.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Sung MM, Hamza SM and Dyck JR: Myocardial
metabolism in diabetic cardiomyopathy: Potential therapeutic
targets. Antioxid Redox Signal. 22:1606–1630. 2015.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Chen PH, Chung CC, Liu SH, Kao YH and Chen
YJ: Lithium treatment improves cardiac dysfunction in rats deprived
of rapid eye movement sleep. Int J Mol Sci.
23(11226)2022.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Jia G, DeMarco VG and Sowers JR: Insulin
resistance and hyperinsulinaemia in diabetic cardiomyopathy. Nat
Rev Endocrinol. 12:144–153. 2016.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Riehle C and Abel ED: Insulin signaling
and heart failure. Circ Res. 118:1151–1169. 2016.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Saotome M, Ikoma T, Hasan P and Maekawa Y:
Cardiac insulin resistance in heart failure: The role of
mitochondrial dynamics. Int J Mol Sci. 20(3552)2019.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Chen PH, Chao TF, Kao YH and Chen YJ:
Lithium interacts with cardiac remodeling: The fundamental value in
the pharmacotherapy of bipolar disorder. Prog Neuropsychopharmacol
Biol Psychiatry. 88:208–214. 2019.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Cheng J, Nanayakkara G, Shao Y, Cueto R,
Wang L, Yang WY, Tian Y, Wang H and Yang X: Mitochondrial proton
leak plays a critical role in pathogenesis of cardiovascular
diseases. Adv Exp Med Biol. 982:359–370. 2017.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Nadtochiy SM, Tompkins AJ and Brookes PS:
Different mechanisms of mitochondrial proton leak in
ischaemia/reperfusion injury and preconditioning: Implications for
pathology and cardioprotection. Biochem J. 395:611–618.
2006.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Carrer A, Laquatra C, Tommasin L and
Carraro M: Modulation and pharmacology of the mitochondrial
permeability transition: A journey from F-ATP synthase to ANT.
Molecules. 26(6463)2021.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Bonora M, Giorgi C and Pinton P: Molecular
mechanisms and consequences of mitochondrial permeability
transition. Nat Rev Mol Cell Biol. 23:266–285. 2022.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Juhaszova M, Zorov DB, Kim SH, Pepe S, Fu
Q, Fishbein KW, Ziman BD, Wang S, Ytrehus K, Antos CL, et al:
Glycogen synthase kinase-3beta mediates convergence of protection
signaling to inhibit the mitochondrial permeability transition
pore. J Clin Invest. 113:1535–1549. 2004.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Juhaszova M, Zorov DB, Yaniv Y, Nuss HB,
Wang S and Sollott SJ: Role of glycogen synthase kinase-3beta in
cardioprotection. Circ Res. 104:1240–1252. 2009.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Yang K, Chen Z, Gao J, Shi W, Li L, Jiang
S, Hu H, Liu Z, Xu D and Wu L: The key roles of GSK-3β in
regulating mitochondrial activity. Cell Physiol Biochem.
44:1445–1459. 2017.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Hirotani S, Zhai P, Tomita H, Galeotti J,
Marquez JP, Gao S, Hong C, Yatani A, Avila J and Sadoshima J:
Inhibition of glycogen synthase kinase 3beta during heart failure
is protective. Circ Res. 101:1164–1174. 2007.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Murphy E and Steenbergen C: Inhibition of
GSK-3beta as a target for cardioprotection: The importance of
timing, location, duration and degree of inhibition. Expert Opin
Ther Targets. 9:447–456. 2005.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Sharma AK, Bhatia S, Al-Harrasi A, Nandave
M and Hagar H: Crosstalk between GSK-3β-actuated molecular cascades
and myocardial physiology. Heart Fail Rev. 26:1495–1504.
2021.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Sharma AK, Thanikachalam PV and Bhatia S:
The signaling interplay of GSK-3β in myocardial disorders. Drug
Discov Today. 25:633–641. 2020.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Bao H, Zhang Q, Liu X, Song Y, Li X, Wang
Z, Li C, Peng A and Gong R: Lithium targeting of AMPK protects
against cisplatin-induced acute kidney injury by enhancing
autophagy in renal proximal tubular epithelial cells. FASEB J.
33:14370–14381. 2019.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Lee Y, Kim SM, Jung EH, Park J, Lee JW and
Han IO: Lithium chloride promotes lipid accumulation through
increased reactive oxygen species generation. Biochim Biophys Acta
Mol Cell Biol Lipids. 1865(158552)2020.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Liu P, Zhang Z, Wang Q, Guo R and Mei W:
Lithium chloride facilitates autophagy following spinal cord injury
via ERK-dependent pathway. Neurotox Res. 32:535–543.
2017.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Qu Z, Sun D and Young W: Lithium promotes
neural precursor cell proliferation: Evidence for the involvement
of the non-canonical GSK-3β-NF-AT signaling. Cell Biosci.
1(18)2011.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Jakobsson E, Argüello-Miranda O, Chiu SW,
Fazal Z, Kruczek J, Nunez-Corrales S, Pandit S and Pritchet L:
Towards a unified understanding of lithium action in basic biology
and its significance for applied biology. J Membr Biol.
250:587–604. 2017.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Parris GE: A hypothesis concerning the
biphasic dose-response of tumors to angiostatin and endostatin.
Dose Response 13: dose-response. 14-020. Parris, 2015.
|
|
55
|
Ge W and Jakobsson E: Systems biology
understanding of the effects of lithium on cancer. Front Oncol.
9(296)2019.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Erguven M, Oktem G, Kara AN and Bilir A:
Lithium chloride has a biphasic effect on prostate cancer stem
cells and a proportional effect on midkine levels. Oncol Lett.
12:2948–2955. 2016.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Suganthi M, Sangeetha G, Gayathri G and
Ravi Sankar B: Biphasic dose-dependent effect of lithium chloride
on survival of human hormone-dependent breast cancer cells (MCF-7).
Biol Trace Elem Res. 150:477–486. 2012.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Gao XM, Fukamauchi F and Chuang DM:
Long-term biphasic effects of lithium treatment on phospholipase
C-coupled M3-muscarinic acetylcholine receptors in cultured
cerebellar granule cells. Neurochem Int. 22:395–403.
1993.PubMed/NCBI View Article : Google Scholar
|