1. Introduction
Garlic (Allium sativum L.), a plant belonging to the Allium genus, has been reported to exert pharmacologically beneficial effects in a variety of diseases (1). Garlic products have been prepared using diverse processing and preparation methods to enhance their medicinal benefits (2). Aged garlic extract (AGE) is a unique preparation produced by aging raw garlic in an alcoholic solution for more than 10 months (3). During aging, the odorous and irritating constituents of raw garlic are transformed into nontoxic compounds (3). Thus, AGE is a useful garlic product with few side effects (4-7). As shown in Fig. 1, AGE is rich in sulfur-containing amino acids such as S-allylcysteine (SAC), S-1-propenylcysteine (S1PC™; Wakunaga Holdings Co., Ltd.), S-methylcysteine (SMC), and S-allylmercaptocysteine (SAMC), which are absent in raw garlic (3). These substances exhibit various pharmacological actions; for example, SAC has peripheral circulation-improving (4,8-10) and anti-inflammatory effects (11); S1PC has anti-inflammatory (12), anti-hypertensive (13-15) and anti-atherosclerotic effects (16); SMC has anti-cancer (17), anti-diabetic (18) and anti-parkinsonian effects (19); SAMC has anti-inflammatory (20), anti-cancer (21), hepatoprotective (22) and nephroprotective effects (23). Because these components have unique metabolic profiles, it is important to clarify their pharmacokinetic properties to understand whether their beneficial activities are affected by themselves or their metabolites. In other words, pharmacokinetic studies are essential to comprehensively understand the mechanisms underlying the pharmacological actions of AGE.
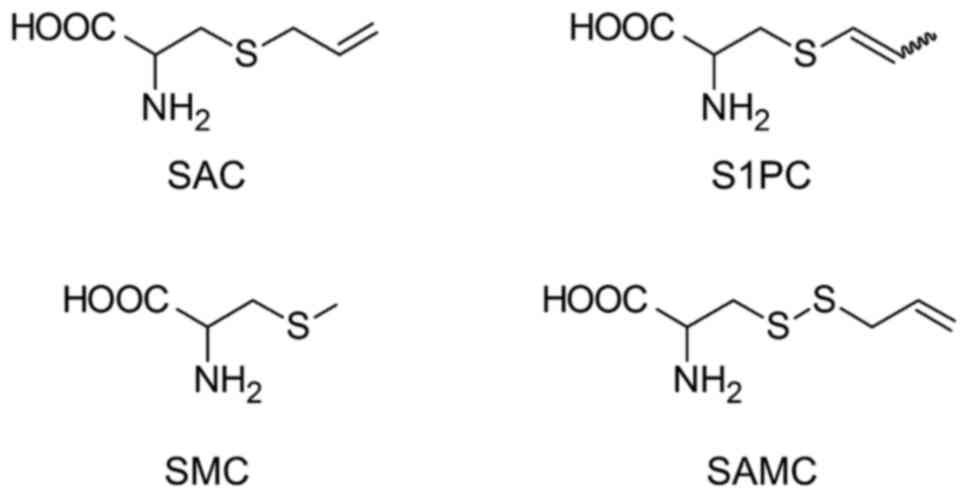 |
Figure 1
Chemical structures of major sulfur-containing amino acids in aged garlic extract. SAC, S-Allylcysteine; SAMC, S-Allylmercaptocysteine; SMC, S-Methylcysteine; S1PC, S-1-Propenylcysteine.
|
In this review, we will summarize and discuss the pharmacokinetics of representative sulfur-containing components of AGE and other garlic-derived products.
2. Pharmacokinetics of SAC
SAC is a cysteine derivative that contains an allyl SH group (Fig. 1). Nagae et al (24) reported the pharmacokinetics of SAC in mice. They have shown that SAC has a half-life (t1/2) of 0.77 h and bioavailability (BA) of 103% after oral administration, accounting for its high absorption efficacy. In contrast, the urinary excretion rate of N-acetyl (NAc)-SAC, the principal metabolite of SAC, was only 7.2%, suggesting the existence of unknown excretion routes via the bile duct, or metabolites other than the N-acetylated form of SAC.
The pharmacokinetics and metabolic pathways of SAC in rats have been previously reported by Amano et al (25) (Fig. 2A). In this study, the BA of SAC and the urinary excretion rate of NAc-SAC after oral administration were 92.1 and 83%, respectively. The t1/2 value of SAC after oral administration in rats was 1.2 h. The t1/2 value of SAC was similar to that reported Park et al (26); thus, the pharmacokinetic parameters after SAC administration were reproducible. The S-oxidized (alliin) and N-acetylated (NAc-alliin) forms of SAC were also excreted in the urine, although their excretion rates were lower than those of NAc-SAC. In other words, the majority of administered SAC was excreted in its N-acetylated form in urine. The t1/2 value and distribution volume at steady state (Vss) of SAC after intravenous administration to rats were 1.1 h and 1.0 l/kg, respectively. Vss is a value used to calculate the amount of a component in the body under equilibrium conditions, which allows for the estimation of its transferability to various tissues by comparing it with the total volume of water in the body. The Vss of SAC was higher than 0.62 l/kg of the total water volume, suggesting that SAC is distributed throughout the body. The total clearance (CL) of SAC was 0.91 l/h/kg, of which the renal clearance (CLr) was 0.0086 l/h/kg. The CLr value of SAC was only 0.9% of the CL value, indicating that the disappearance of SAC from the blood may depend on its metabolism rather than renal clearance. Furthermore, as evidenced by the smaller CLr value than the glomerular filtration rate (GFR) in rats (only 3.6% of GFR), the majority of the SAC filtered through the glomerulus may be reabsorbed in the renal tubules. In contrast, the CLr value of NAc-SAC administered intravenously to rats was higher than the GFR, suggesting that SAC is actively excreted in the urine after conversion to NAc-SAC.
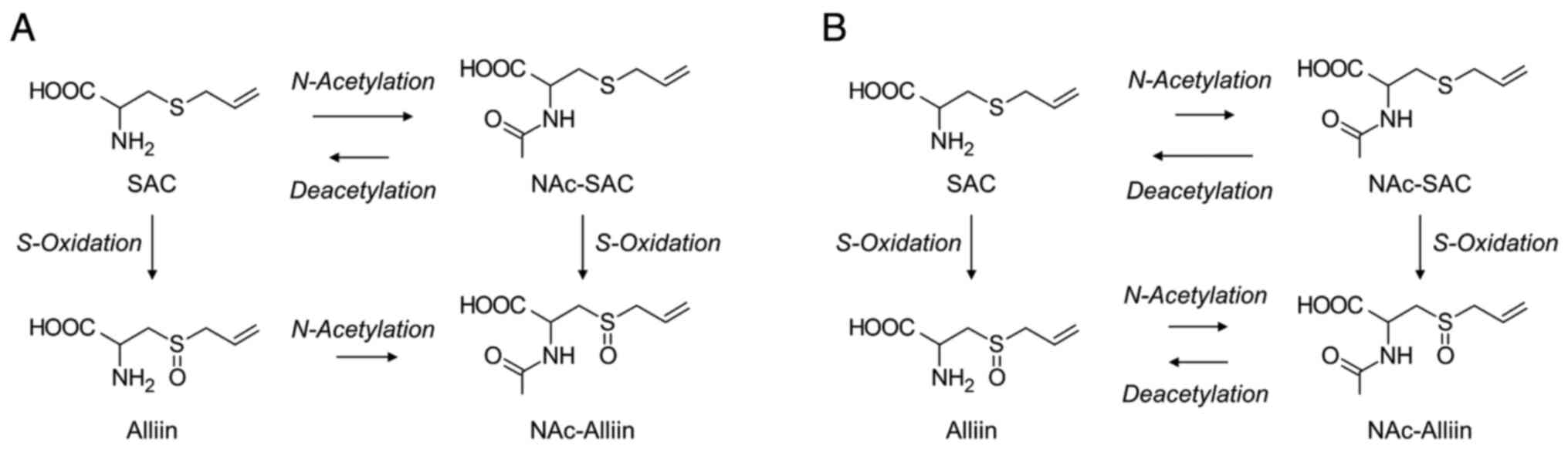 |
Figure 2
Metabolic pathway of SAC in (A) rats and (B) dogs. The length of the arrows for the N-acetylation and deacetylation processes indicates the intensity of the activity. NAc, N-acetyl; SAC, S-Allylcysteine.
|
The pharmacokinetics and metabolic pathways of SAC in dogs have been previously reported by Amano et al (25) (Fig. 2B). As observed in mice and rats, SAC was well-absorbed in dogs, with a BA of 92% after oral administration. In contrast, the t1/2 value of SAC in dogs was approximately 12 h. Nagae et al (24) also reported that oral administration of SAC resulted in high BA and a long t1/2 in dogs. One of the factors contributing to the large t1/2 value appears to be the substantially low CL of SAC in dogs (0.048 l/h/kg). In addition, Amano et al performed in vitro experiments using enzyme fractions from the kidney and demonstrated that the difference in N-acetylation and deacetylation enzyme activities against SAC was due to its long t1/2 in dogs. The renal intrinsic clearance (CLint) values for N-acetylation of SAC and for deacetylation of NAc-SAC calculated in the dog renal enzyme fraction were 5.9 and 460 µl/min/mg protein, respectively; while in the rat fraction they were 190 and 27 µl/min/mg protein, respectively; in other words, the N-acetylation activity against SAC relative to the deacetylation one against NAc-SAC was 80-fold lower in dog, but 7-fold higher in rat. These results suggest that a large proportion of NAc-SAC is deacetylated to SAC in the canine kidney and then reabsorbed in the renal tubules and reintroduced into the systemic circulation, resulting in the prolonged residence of SAC within the blood in dogs rather than in rats. Another characteristic of SAC pharmacokinetics in dogs compared to that in rats is the proportion of metabolites excreted in the urine. In rats, the amount of identified metabolites excreted in the urine corresponded to 95% of the SAC administered, whereas only 14% was excreted in dogs. This finding suggests that there are unidentified metabolites of SAC or that the majority of metabolites are eliminated via biliary excretion in dogs.
In humans, amino acids are actively absorbed from the intestinal tract and reabsorbed from the urine via several types of transporters in the kidneys. Thus, SAC may also be absorbed orally and may undergo renal reabsorption in humans. Amano et al (25) revealed that the deacetylation activity against NAc-SAC was more than 10 times higher than N-acetylation activity against SAC in experiments using hepatic and renal enzyme fractions from humans. These results suggest that SAC is retained in the blood for a long time because it is less susceptible to N-acetylation in humans, as observed in dogs. Kodera et al collected blood samples from volunteers after AGE ingestion and measured SAC concentrations in the blood; SAC was still detected 24 h later (27).
In summary, these findings show that the similar and high BA of SAC after oral administration is similar in mice, rats, and dogs. Since SAC circulates in the blood in its intact form, its pharmacological effects may be exerted by SAC itself rather than its metabolites. However, it can be speculated that their metabolism and excretion differ among species, whereas those in dogs and humans may be similar. Clarification of species-specific characteristics, such as metabolic enzyme activities and excretion routes, may explain why pharmacokinetics, such as t1/2 and CL, for SAC differ widely among these animals.
3. Pharmacokinetics of S1PC
S1PC is the stereoisomer of SAC (Fig. 1). Owing to the presence of a propenyl group, S1PC has cis and trans geometric isomers, with the trans form being the most abundant in nature. The pharmacokinetics and metabolic pathways of S1PC in rats have been previously reported by Amano et al (28) (Fig. 3A). After oral administration of S1PC to rats, BA and t1/2 values were 88% and 0.56 h, respectively, indicating that S1PC disappears faster than SAC in the blood. The difference in the t1/2 value between S1PC and SAC may attribute to the higher clearance rate of S1PC than SAC; CL value after the intravenous administration of S1PC was 1.4 l/h/kg, while that of SAC was 0.91 l/h/kg. Furthermore, as estimated from the higher CLr value of S1PC (0.03 l/h/kg) compared with that of SAC (0.0086 l/h/kg), the higher systemic clearance of S1PC than SAC is probably due to its fast elimination rate. The final metabolites of S1PC excreted in the urine were similar to those of SAC, with NAc-S1PC™ (Wakunaga Holdings Co., Ltd.) accounting for 69% of the S1PC administered. The S-oxidized (isoalliin) and N-acetylated (NAc-isoalliin) forms of S1PC were also excreted in the urine, although their excretion rates were lower than those of NAc-S1PC. The in vitro experiments using enzyme fractions from the liver and kidney showed that N-acetylation activity against S1PC in the liver and kidney was 5.5- to 5.7-fold higher than that against NAc-S1PC, suggesting that S1PC is excreted into urine after conversion to NAc-S1PC in rats.
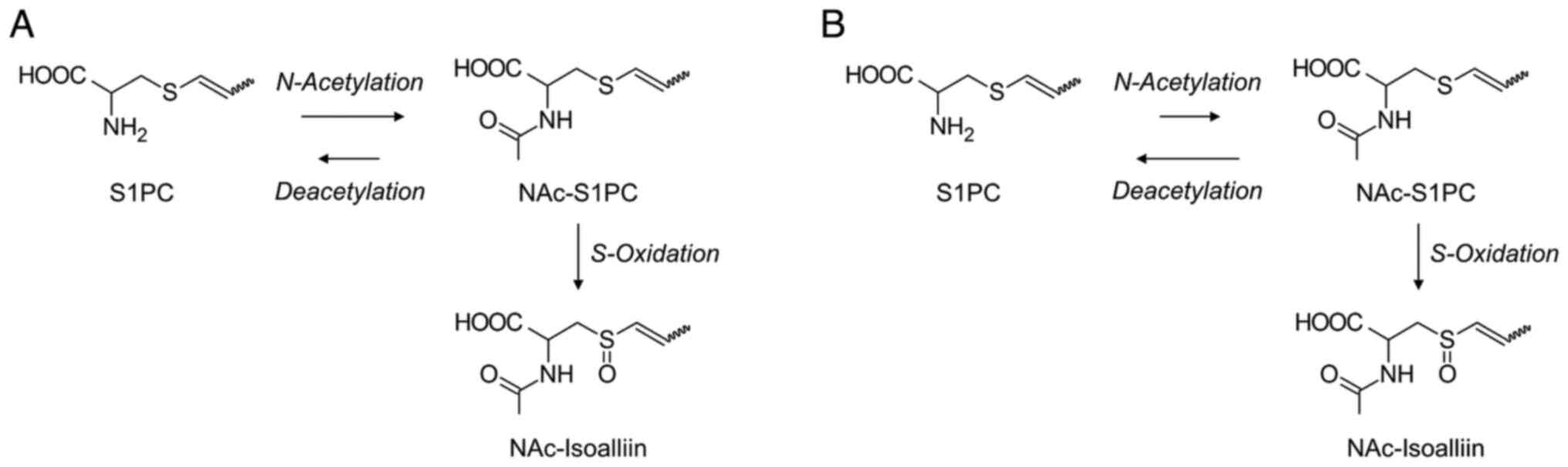 |
Figure 3
Metabolic pathway of S1PC in (A) rats and (B) dogs. The length of the arrows for the N-acetylation and deacetylation processes indicates the intensity of the activity. NAc, N-acetyl; S1PC, S-1-Propenylcysteine.
|
Amano et al (28) reported the pharmacokinetics and metabolic pathways of S1PC in dogs (Fig. 3B). Similar to rats, oral administration of S1PC resulted in a high BA in dogs (approximately 100%). However, the excretion routes of S1PC metabolites appeared to be different between rats and dogs, since the proportion of metabolites, including NAc-S1PC, excreted in rat urine was 70% of the S1PC administered, whereas that in dogs was only 3.4%. The t1/2 of S1PC was 5.3 h in dogs. As observed in the in vitro experiments with SAC, renal Clint value for deacetylation of NAc-S1PC (1,337 µl/min/mg protein) was about 40 times higher than that for N-acetylation of S1PC (33 µl/min/mg protein) in the renal enzyme fractions, suggesting that S1PC is reabsorbed in the kidney similarly to SAC, thereby sustaining its blood concentration for long time after the administration in dogs. Additional findings regarding the enzyme activity against SAC and S1PC may also explain the differences in t1/2 values in dogs. The deacetylation activity of NAc-SAC was 80-fold higher than N-acetylation activity against SAC in the kidneys; however, the deacetylation activity of N-acetylation activity was lower than SAC in the case of S1PC (40-fold). Thus, the low deacetylation activity of S1PC may contribute to lower renal reabsorption and a shorter t1/2 value of S1PC than that of SAC in dogs.
Amano et al (28) examined the enzymatic activity of S1PC and NAc-S1PC in human hepatic and renal enzyme fractions. The renal CLint values for N-acetylation of S1PC and for deacetylation of NAc-S1PC calculated in the human renal enzyme fraction were 61 and 172 µl/min/mg protein, respectively, and with the human hepatic enzyme fraction they were 8.3 and 70 µl/min/mg protein, respectively; in other words, deacetylation activity against NAc-S1PC was higher than N-acetylation activity against S1PC in human. This indicates that S1PC ingested by humans may be retained in the blood for a long time, as in the case of SAC.
In summary, orally administered S1PC was well absorbed by both rats and dogs, although its metabolism and excretion routes seemed to differ between the two species. This difference may depend on the enzymatic activities involved in the metabolism of S1PC, especially in the kidneys, as observed in the case of SAC. It is also conceivable that S1PC itself, rather than its metabolites, is responsible for its pharmacological effects, as it is retained in the blood in its intact form with high BA.
4. Pharmacokinetics of SMC
SMC is a cysteine derivative with a methyl group in its SH group (Fig. 1). Barnsley and Sklan and Barnsley (29) reported that approximately 50% of the administered SMC was excreted into the urine as inorganic sulfate when stable 35S-labeled SMC was subcutaneously injected into rats. In addition, four sulfoxide derivatives, NAc-Methiin, 2-hydroxy-3-methylsulphinylpropionic acid, methylsulfinylacetic acid and, unidentified compounds were found to be metabolites of SMC, and the proportion of metabolites excreted in the urine corresponded to 19% of the administered SMC. Amano et al (28). investigated the pharmacokinetics and metabolic pathways of SMC in rats (Fig. 4A). When SMC was orally administered to rats, BA and t1/2 values were 95.8% and 2.6 h, respectively, indicating that SMC disappeared from the blood circulation more slowly than SAC and S1PC. Since CL value of SMC after intravenous administration was lower (0.32 l/h/kg) than those of SAC and S1PC, it is assumed that the relatively slow clearance of SMC may contribute to its longer t1/2 value in rats than those of SAC and S1PC. The proportions of NAc-SMC and NAc-methiin excreted in urine corresponded to 13 and 11% of the SMC administered, respectively, indicating that the amounts of N-acetylated forms were smaller among the SMC metabolites excreted in the urine, in contrast to those of SAC and S1PC. In addition, N-acetylation activity against SMC was weaker (hepatic Clint in liver enzyme fractions, not detected; renal Clint in renal enzyme fractions, 36 µl/min/mg) as compared with those of SAC and S1PC in rats, leading to the low proportion of NAc-SMC excreted in the urine.
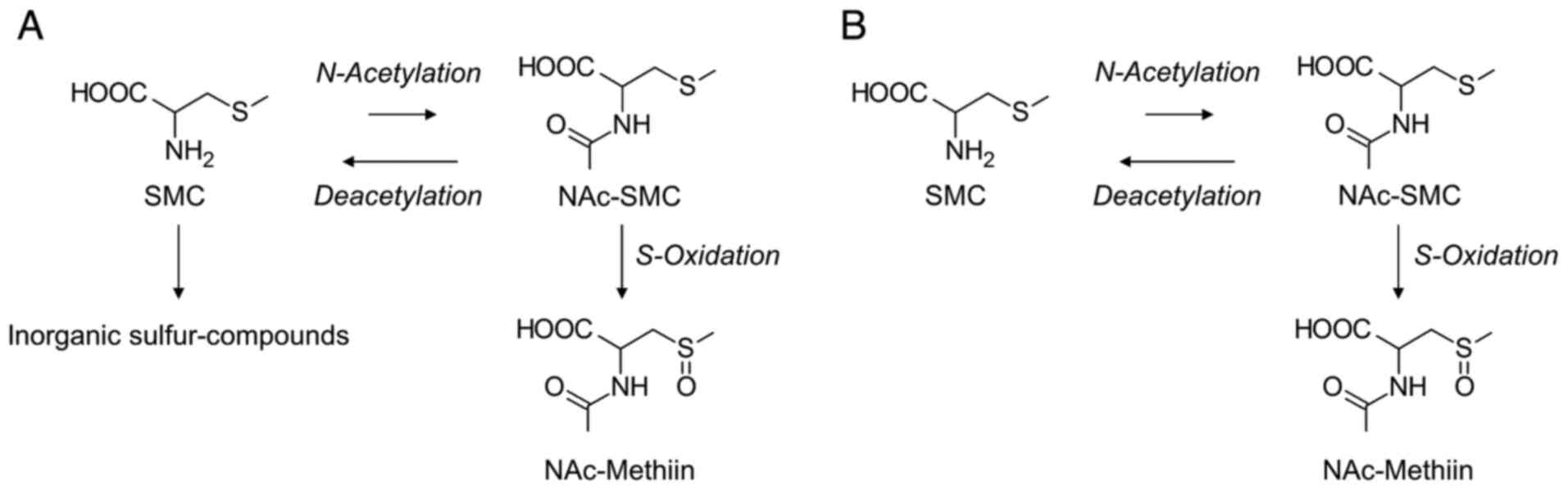 |
Figure 4
Metabolic pathway of SMC in (A) rats and (B) dogs. The length of the arrows for the N-acetylation and deacetylation processes indicates the intensity of the activity. NAc, N-acetyl; SMC, S-Methylcysteine.
|
Amano et al (28) reported the pharmacokinetics and metabolic pathways of SMC in dogs (Fig. 4B). The BA of the SMC was 95.5% after oral administration. Interestingly, although the t1/2 of SMC was shorter (8.0 h) than that of SAC (12 h), the CL of intravenously administered SMC was comparable to that of SAC. As deduced from the small Vss value of SMC, the differences in the t1/2 values between SMC and SAC may depend on their transferability to tissues, but not on their clearance. As observed in rats, the N-acetylation activity against SMC was similarly weak in dogs (hepatic Clint in liver enzyme fractions, not detected; renal Clint in renal enzyme fractions, 5.6 µl/min/mg). This result may account for the low urinary excretion of SMC metabolites in N-acetylated forms, with NAc-SMC being below the detection threshold and NAc-SMCS corresponding to 0.7% of the SMC administered.
Mitchell et al (30) studied the metabolism of SMC in humans and reported its high oral absorption; the mean total recovery of 35S radioactivity in urine and 14C radioactivity in exhaled air was 95.9 and 77.2% of the orally administered 35S-laveled and 14C-labeled SMC, respectively. They also detected that several metabolites of SMC underwent S-oxidation, N-acetylation, and deamination and identified the majority of them as inorganic sulfate and CO2. Amano et al (28) evaluated the enzymatic activities of SMC and NAc-SMCs using human hepatic and renal enzyme fractions. As observed in the case of dogs, deacetylation activity against NAc-SMC was higher to N-acetylation activity against SMC in both enzyme fractions from liver and kidney of human (CLint values for N-acetylation of SMC were 1.1 and 0.74 µl/min/mg protein in renal and hepatic enzyme fractions, respectively; CLint values for deacetylation of NAc-SMC were 104 and 77 µl/min/mg protein, respectively). These findings suggested that the circulation of SMC may be maintained for a relatively long time after ingestion by humans.
In summary, the proportions of the major metabolites of SMC secreted in the urine are different from those of SAC and S1PC, as nearly 50% of SMC is excreted in the urine as inorganic sulfate after administration. The reason why the proportion of SMC excreted in urine as N-acetylated form is low compared to that of SAC and S1PC may be due to the relatively weak N-acetylation activity against SMC in major metabolic organs, such as the liver and kidney. However, similar to SAC and S1PC, SMC circulates in the blood in an intact form with a high BA; thus, SMC itself is likely responsible for its pharmacological effects.
5. Pharmacokinetics of SAMC
SAMC is an SAC derivative with two sulfur atoms (Fig. 1), and its pharmacokinetics have been reported in rats by Yang et al (31) and Nakamoto and Kunimura (32). Yang et al showed that the t1/2 of SAMC administered intravenously was 3.8 min, which was much faster than that of other sulfur-containing amino acids such as SAC and S1PC. It has also been demonstrated by Nakamoto et al that SAMC rapidly disappeared from the blood within 15 min of intravenous administration. Moreover, SAMC was not detected in the blood after oral administration. These results suggested that the various pharmacological actions of SAMC observed in vivo could be derived from its metabolites. Nakamoto et al proposed the metabolic pathways of SAMC by performing ex vivo experiments using rat plasma, in which SAMC immediately reacted with components in the blood and was converted into diallyl polysulfides (DASn). Additionally, the experiments using hepatic and renal enzyme fractions revealed the following metabolic pathways: DASn are converted to S-3-oxopropylglutathione (S3OPG) thorough reacting with glutathione and then undergo the oxidation/reduction reactions to form glutathione conjugates, S-2-carboxyethylglutathione (S2CEG) and S-3-hydroxypropylglutathione (S3HPG); the glutamic acid and glycine are desorbed from these metabolites in the kidney to form the cysteine conjugates, and they are ultimately excreted as N-acetylated form into the urine (Fig. 5). Although two N-acetylcysteine conjugates, NAc-S-3-hydroxypropylcysteine (NAc-S3HPC) and NAc-S-2-carboxyethylcysteine (NAc-S2CEC), were identified as metabolites excreted in the urine after the intravenous administration of SAMC, the proportion of excreted metabolites was only 8.1 and 2.6% of the total amount of SAMC administered, respectively. Nakamoto et al suggested that these low excretion rates in urine may be due to one or a combination of the following reasons: 1. The majority of DASn produced from SAMC is released through exhaled breath and skin; 2. SAMC and its metabolites readily bind to blood proteins; and 3. Finally, SAMC is converted to inorganic sulfate in a pattern similar to that of SMC.
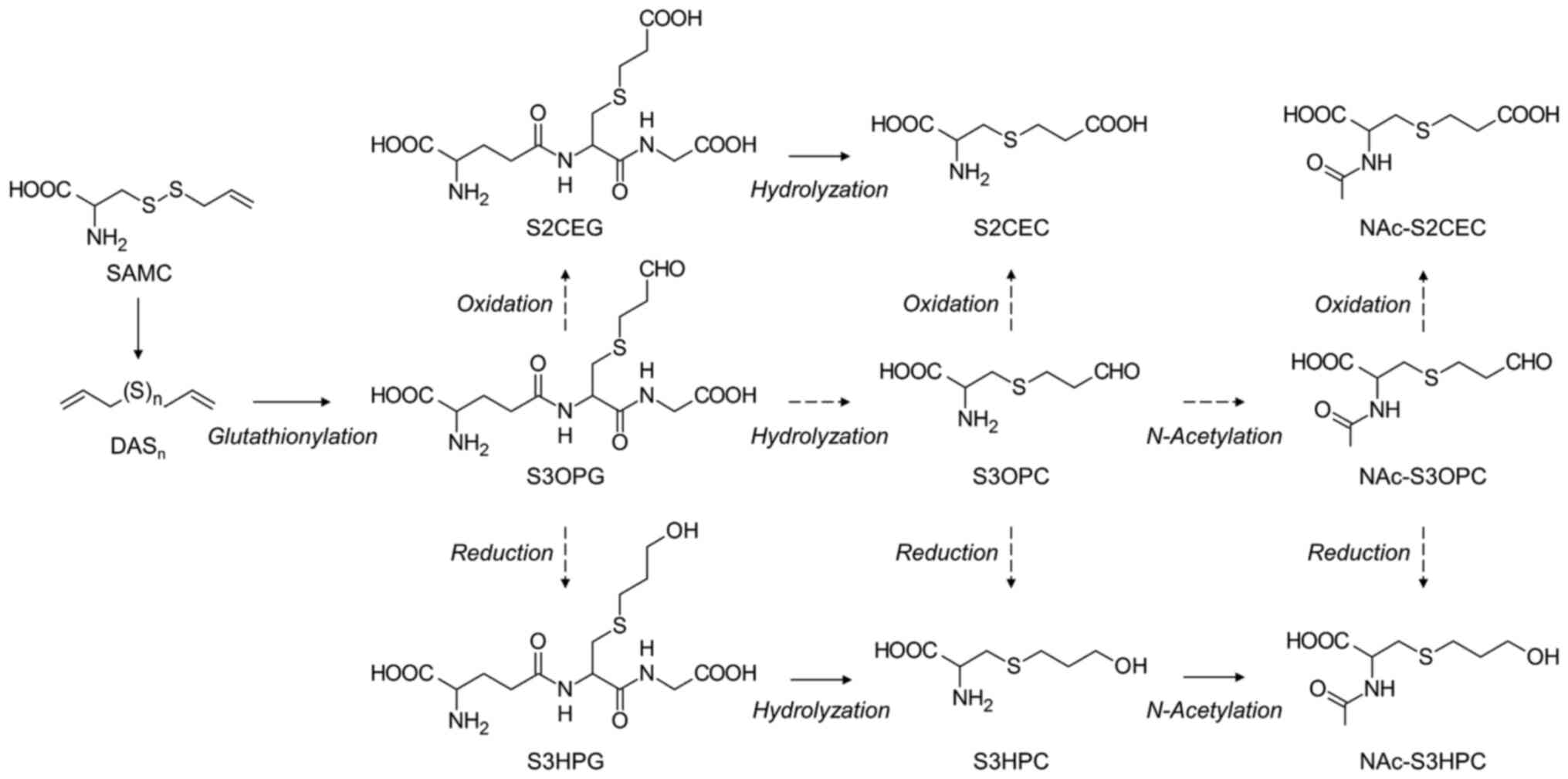 |
Figure 5
Metabolic pathway of SAMC in rats. Dashed arrows indicate putative metabolic pathways. DASn, Diallylpolysulfides; NAc, N-acetyl; SAMC, S-Allylmercaptocysteine; S2CEC, S-2-Carboxyethylcysteine; S2CEG, S-2-Carboxyethylglutathione; S3HPC, S-3-Hydroxypropylcysteine; S3HPG, S-3-Hydroxypropylglutathione; S3OPC, S-3-Oxopropylcysteine; S3OPG, S-3-Oxopropylglutathione.
|
In summary, after administration, SAMC immediately reacts with components in the blood and is converted to volatile sulfur compounds, resulting in a shorter t1/2 value than that of other sulfur-containing amino acids, such as SAC and S1PC. This suggests that the pharmacological effects of SAMC are derived from its metabolites rather than from SAMC itself. The proportion of final SAMC metabolites identified after intravenous administration was only approximately 11%; thus, further investigations are required to clarify how SAMC is metabolized and excreted from the body.
6. Others
The chemical structures and amounts of sulfur-containing compounds in processed garlic products vary markedly, depending on the processing method used. Although less present in AGE, raw garlic and other processed garlic, such as garlic oil, contain other sulfur-containing compounds, such as hydrophobic sulfur-containing compounds (Fig. 6). In this section, we discuss pharmacokinetics of these compounds.
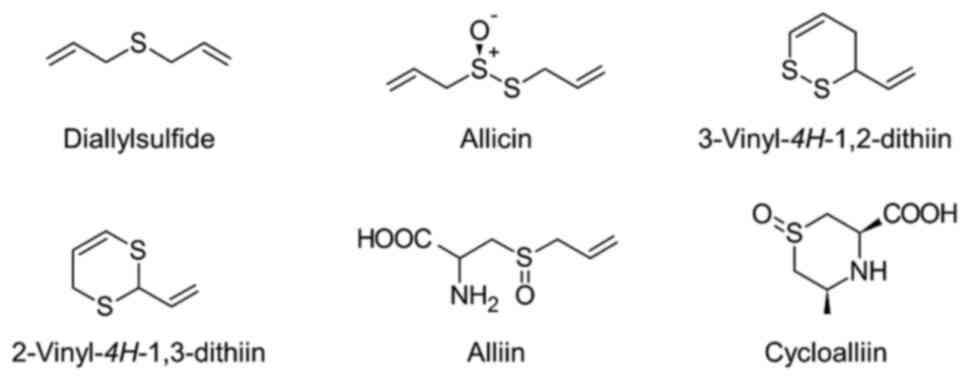 |
Figure 6
Chemical structures of other sulfur-containing compounds in garlic.
|
Diallyl sulfide
Diallyl sulfide is a sulfur-containing hydrophobic compound with one sulfur atom and two allyl groups. Diallyl sulfide has antimicrobial, antimetastatic, and antioxidant effects (33-35). The pharmacokinetics of diallyl sulfide have been reported by Jin and Baillie (36), who identified 10 species of glutathione conjugates as metabolites of diallyl sulfide in rats. The two identified metabolites of diallyl sulfide, S2CEG and S3HPG, are consistent with those of SAMC reported by Nakamoto and Kunimura (32). The metabolic pathway of diallyl sulfide appeared to be partially similar to that of SAMC.
Allicin
Allicin has a structure in which one sulfur atom of diallyl disulfide is oxidized. Allicin exerts antifibrotic, anti-inflammatory, and tissue-protective effects on the lung (37-39). The pharmacokinetics of allicin have been previously reported by Lachmann et al (40). They administered 35S-labeled allicin (35S-allicin) to rats and examined their blood profiles. The time required to reach the maximum blood concentration (Cmax) of 35S-allicin administered was 30-60 min and it was still detectable in the blood at 72 h. The total proportion of 35S-allicin excreted in both urine and feces for up to 72 h was 85.5% of the administered compound. However, Freeman and Kodera (41) reported that allicin is highly unstable, since it reacts immediately with some components in the blood.
Vinyldithiin
Vinyldithiins (3-vinyl-4H-1,2-dithiin and 2-vinyl-4H-1,3-dithiin) are produced via the conversion of allicin molecules. Vinyldithiins have anti-obesity effect (42). Lachmann et al (40) reported on the pharmacokinetics of vinyldithiins. They orally administered 35S-labeled vinyldithiins (35S-vinyldithiins) to rats and evaluated their blood profiles. It took 120 min for 35S-vinyldithiins to reach the Cmax, and the compound was still detectable in the blood after 72 h, similar to allicin. The total proportion of 35S-vinyldithiins excreted in both urine and feces for up to 72 h was 92.3% of the administered compounds.
Alliin
Alliin is the S-oxidized form of SAC. Alliin has renal protective, antihyperlipidemic, and mitophagy-activating effects (43-45). Lachmann et al (40) reported on the pharmacokinetics of alliin. They orally administered 35S-labeled alliin (35S-alliin) to rats and evaluated their blood profiles. The blood concentration of 35S-alliin reached Cmax within 10 min and was below the detection threshold after 6 h, demonstrating that the pharmacokinetics of alliin in blood were similar to those of SAC and S1PC.
Cycloalliin
Cycloalliin is a cyclic structure of the sulfur-containing amino acid. Cycloalliin has fibrinolytic activity, anti-obesity, and serum triglyceride-lowering effects (46-48). The pharmacokinetics of cycloalliin has been reported by Ichikawa et al (49), who showed several differences in its metabolism and excretion patterns when administered intravenously or orally to rats. After intravenous administration, 97.8% of cycloalliin was excreted in the urine within 48 h without being metabolized. However, after oral administration, cycloalliin was metabolized by intestinal bacteria and subsequently excreted in the feces as (3R, 5S)-5-methyl-1,4-thiazane-3-carboxylic acid. Thus, cycloalliin is poorly absorbed after oral administration, as evidenced by its low BA (only 3.73%) when administered intravenously.
7. Conclusion
AGE contains a variety of organosulfur compounds, such as SAC, S1PC, SMC, and SAMC, and numerous studies have revealed that these compounds exert a wide range of pharmacological actions. Previous studies have examined the pharmacokinetic properties of SAC, S1PC, and SMC in mammals. Particularly, in dogs, they have high oral absorption, and presumably undergo renal reabsorption, and circulate in the blood in an intact form, suggesting that the pharmacological effects of these compounds are caused by themselves and not by their metabolites. Although the pharmacokinetic properties of SAMC have not been fully clarified, it has been suggested that after administration, its metabolites may exert pharmacological actions because of the instability of SAMC in the blood. It should be noted that the pharmacokinetics of these sulfur compounds in rodents and dogs, which are becoming clearer, might be different from those in humans due to their varied physiological characteristics. Further pharmacokinetic research especially in humans is required to precisely identify the active components responsible for the beneficial effects of AGE.
Acknowledgements
The authors thank Dr Takami Oka of Wakunaga Pharmaceutical Co., Ltd. (Akitakata, Japan) for his helpful advice, encouragement and critical reading of the manuscript.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
MN and KK conceived this review. MN and KK analyzed the relevant literature. MN wrote the first draft of the manuscript and produced the figures. KK and MO critically revised the manuscript. All authors have reviewed and approved the final manuscript. Data authentication is not applicable.
Ethics approval and consent to participate
Not applicable.
Patient consent for publicationa
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
|
1
|
Block E: The chemistry of garlic and onions. Sci Am. 252:114–119. 1985.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Ahmed T and Wang CK: Black garlic and its bioactive compounds on human health diseases: A review. Molecules. 26(5028)2021.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Kodera Y, Kurita M, Nakamoto M and Matsutomo T: Chemistry of aged garlic: Diversity of constituents in aged garlic extract and their production mechanisms via the combination of chemical and enzymatic reactions. Exp Ther Med. 19:1574–1584. 2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Takemura S, Ihara H, Nakagawa K and Minamiyama Y: S-allyl cysteine blood flow in NO-dependent and -independent manners. Glycative Stress Research. 9:146–157. 2022.
|
|
5
|
Nakagawa S, Masamoto K, Sumiyoshi H and Harada H: Acute toxicity test of garlic extract. J Toxicol Sci. 9:57–60. 1984.PubMed/NCBI View Article : Google Scholar : (In Japanese).
|
|
6
|
Sumiyoshi H, Kanezawa A, Masamoto K, Harada H, Nakagami S, Yokota A, Nishikawa M and Nakagawa S: Chronic toxicity test of garlic extract in rat. J Toxicol Sci. 9:61–75. 1984.PubMed/NCBI View Article : Google Scholar : (In Japanese).
|
|
7
|
Yoshida S, Hirano Y and Nakagawa S: Mutagenicity and cytotoxicity tests of garlic. J Toxicol Sci. 9:77–86. 1984.PubMed/NCBI View Article : Google Scholar : (In Japanese).
|
|
8
|
Syu JN, Yang MD, Tsai SY, Chiang EI, Chiu SC, Chao CY, Rodriguez RL and Tang FY: S-allylcysteine improves blood flow recovery and prevents ischemic injury by augmenting neovasculogenesis. Cell Transplant. 26:1636–1647. 2017.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Chao WW, Chen YK, Chao HW, Pan WH and Chao HM: Fortified S-Allyl L-Cysteine: Animal safety, effect on retinal ischemia, and role of wnt in the underlying therapeutic mechanism. Evid Based Complement Alternat Med. 2020(3025946)2020.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Chao WW, Chao HW, Lee HF and Chao HM: The effect of S-Allyl L-Cysteine on retinal ischemia: The contributions of MCP-1 and PKM2 in the underlying medicinal properties. Int J Mol Sci. 25(1349)2024.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Shin NR, Kwon HJ, Ko JW, Kim JS, Lee IC, Kim JC, Kim SH and Skin IS: S-Allyl cysteine reduces eosinophilic airway inflammation and mucus overproduction on ovalbumin-induced allergic asthma model. Int Immunopharmacol. 68:124–130. 2019.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Nango H and Ohtani M: S-1-propenyl-L-cysteine suppresses lipopolysaccharide-induced expression of matrix metalloproteinase-1 through inhibition of tumor necrosis factor-α converting enzyme-epidermal growth factor receptor axis in human gingival fibroblasts. PLoS One. 18(e0284713)2023.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Ushijima M, Takashima M, Kunimura K, Kodera Y, Morihara N and Tamura K: Effects of S-1-propenylcysteine, a sulfur compound in aged garlic extract, on blood pressure and peripheral circulation in spontaneously hypertensive rats. J Pharm Pharmacol. 70:559–565. 2018.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Matsutomo T, Ushijima M, Kodera Y, Nakamoto M, Takashima M, Morihara N and Tamura K: Metabolomic study on the antihypertensive effect of S-1-propenylcysteine in spontaneously hypertensive rats using liquid chromatography coupled with quadrupole-Orbitrap mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci. 1046:147–155. 2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Matsutomo T, Ushijima M, Kunimura K and Ohtani M: Metabolomic study reveals the acute hypotensive effect of S-1-propenylcysteine accompanied by alteration of the plasma histidine level in spontaneously hypertensive rats. J Pharm Biomed Anal. 168:148–154. 2019.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Miki S, Suzuki J, Takashima M, Ishida M, Kokubo H and Yoshizumi M: S-1-Propenylcysteine promotes IL-10-induced M2c macrophage polarization through prolonged activation of IL-10R/STAT3 signaling. Sci Rep. 11(22469)2021.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Nishikawa-Ogawa M, Wanibuchi H, Morimura K, Kinoshita A, Nishikawa T, Hyashi S, Yano Y and Fukushima S: N-acetylcysteine and S-methylcysteine inhibit MeIQx rat hepatocarcinogenesis in the post-initiation stage. Carcinogenesis. 27:982–988. 2006.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Hsu CC, Yen HF, Yin MC, Tsai CM and Hsieh CH: Five cysteine-containing compounds delay diabetic deterioration in Balb/cA mice. J Nutr. 134:3245–3249. 2004.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Wassef R, Haenold R, Hansel A, Brot N, Heinemann SH and Hoshi T: Methionine sulfoxide reductase A and a dietary supplement S-methyl-L-cysteine prevent Parkinson's-like symptoms. J Neurosci. 27:12808–12816. 2007.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Yang G, Li S, Li B, Cheng L, Jiang P, Tian Z and Sun S: Protective effects of garlic-derived S-Allylmercaptocysteine on IL-1 β-stimulated chondrocytes by regulation of MMPs/TIMP-1 ratio and type II collagen expression via suppression of NF-κB pathway. Biomed Res Int. 2017(8686207)2017.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Zhang Y, Li HY, Zhang ZH, Bian HL and Lin G: Garlic-derived compound S-allylmercaptocysteine inhibits cell growth and induces apoptosis via the JNK and p38 pathways in human colorectal carcinoma cells. Oncol Lett. 8:2591–2596. 2014.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Sumioka I, Matsura T, Kasuga S, Itakura Y and Yamada K: Mechanisms of protection by S-allylmercaptocysteine against acetaminophen-induced liver injury in mice. Jpn J Pharmacol. 78:199–207. 1998.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Zhu X, Jiang X, Li A, Zhao Z and Li S: S-Allylmercaptocysteine attenuates cisplatin-induced nephrotoxicity through suppression of apoptosis, oxidative stress, and inflammation. Nutrients. 9(166)2017.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Nagae S, Ushijima M, Hatono S, Imai J, Kasuga S, Matsuura H, Itakura Y and Higashi Y: Pharmacokinetics of the garlic compound S-allylcysteine. Plant Med. 60:214–217. 1994.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Amano H, Kazamori D, Itoh K and Kodera Y: Metabolism, excretion, and pharmacokinetics of S-allyl-L-cysteine in rats and dogs. Drug Metab Dispos. 43:749–755. 2015.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Park T, Oh JH, Park SC, Jang YP and Lee YJ: Oral administration of (S)-Allyl-L-Cysteine and aged garlic extract to rats: Determination of metabolites and their pharmacokinetics. Plant Med. 83:1351–1360. 2017.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Kodera Y, Suzuki A, Imada O, Kasuga S, Sumioka I, Kanezawa A, Taru N, Fujikawa M, Nagae S, Masamoto K, et al: Physical, chemical, and biological properties of S-Allylcysteine, an amino acid derived from garlic. J Agric Food Chem. 50:622–632. 2002.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Amano H, Kazamori D and Itoh K: Pharmacokinetics and N-acetylation metabolism of S-methyl-l-cysteine and trans-S-1-propenyl-l-cysteine in rats and dogs. Xenobiotica. 46:1017–1025. 2016.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Sklan NM and Barnsley EA: The metabolism of S-methyl-L-cysteine. Biochem J. 107:217–223. 1968.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Mitchell SC, Smith RL, Waring RH and Aldington GF: The metabolism of S-methyl-L-cysteine in man. Xenobiotica. 14:767–779. 1984.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Yang M, Dong Z, Jiang X, Zhao Z, Zhang J, Cao X and Zhang D: Determination of S-Allylmercaptocysteine in Rat Plasma by LC-MS/MS and its application to a pharmacokinetics study. J Chromatogr Sci. 56:396–402. 2018.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Nakamoto M and Kunimura K: Study of the metabolism of S-allylmercaptocysteine to elucidate its metabolites and metabolic pathway in rats. Eur Food Res Technol. 249:1377–1389. 2023.
|
|
33
|
Tsao SM and Yin M: In-vitro antimicrobial activity of four diallyl sulphides occurring naturally in garlic and Chinese leek oils. J Med Microbiol. 50:646–649. 2001.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Lai KC, Hsu SC, Kuo CL, Yang JS, Ma CY, Lu HF, Tang NY, Hsia TC, Ho HC and Chung JG: Diallyl sulfide, diallyl disulfide, and diallyl trisulfide inhibit migration and invasion in human colon cancer colo 205 cells through the inhibition of matrix metalloproteinase-2, -7, and -9 expressions. Environ Toxicol. 28:479–488. 2013.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Yu T, Wang Q, Li XJ, Li M, Liu ND and Xie KQ: Antioxidant mechanism of diallyl sulfide in inhibiting leucopenia in peripheral blood induced by benzene in rats. Zhonghua Lao Dong Wei Sheng Zhi Ye Bing Za Zhi. 37:737–745. 2019.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
36
|
Jin L and Baillie TA: Metabolism of the chemoprotective agent diallyl sulfide to glutathione conjugates in rats. Chem Res Toxicol. 10:318–327. 1997.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Zheng Y, Li G, Shi A, Guo J, Xu Y and Cai W: Role of miR-455-3p in the alleviation of LPS-induced acute lung injury by allicin. Heliyon. 10(e39338)2024.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Gan L, Geng L, Li Q, Zhang L, Huang Y, Lin J and Ou S: Allicin ameliorated high-glucose peritoneal dialysis solution-induced peritoneal fibrosis in rats via the JAK2/STAT3 signaling pathway. Cell Biochem Biophys: Oct 25, 2024 (Epub ahead of print).
|
|
39
|
Liu S, He Y, Shi J, Liu L, Ma H, He L and Guo Y: Allicin attenuates myocardial ischmia reperfusion injury in rats by inhibition of inflammation and oxidative stress. Transplant Proc. 51:2060–2065. 2019.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Lachmann G, Lorenz D, Radeck W and Steiper M: The pharmacokinetics of the S35 labeled labeled garlic constituents alliin, allicin and vinyldithiine. Arzneimittelforschung. 44:734–743. 1994.PubMed/NCBI(In German).
|
|
41
|
Freeman F and Kodera Y: Garlic chemistry: Stability of S-(2-propyl)-2-propen-1-sulfinothioate (allicin) in blood, solvents, and stimulated physiological fluids. J Agric Food Chem. 43:2332–2338. 1995.
|
|
42
|
Keophiphath M, Priem F, Jacquemond-Collet I, Clément K and Lacasa D: 1,2-vinyldithiin from garlic inhibits differentiation and inflammation of human preadipocytes. J Nur. 139:2055–2060. 2009.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Jiang C, Huang H, Zhong C, Feng S, Wang C, Xue H and Zhang J: Alliin mitigates the acute kidney injury by suppressing ferroptosis via regulating the Nrf2/GPX4 axis. Naunyn Schmiedebergs Arch Pharmacol. 398:1521–1533. 2025.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Zhang M, Zou X, Du Y, Pan Z, He F, Sun Y and Li M: Integrated transcriptomics and metabolomics reveal the mecanism of alliin in improving hyperlipidemia. Foods Sep. 12(3407)2023.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Liu M, Lu J, Yang S, Chen Y, Yu J and Guan S: Alliin alleviates LPS-induced pyroptosis via promoting mitophagy in THP-1 macrophages and mice. Food Chem Toxicol. 160(112811)2022.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Agarwal RK, Dewar HA, Newell DJ and Das B: Controlled trial of the effect of cycloalliin on the fibrinolytic activity of venous blood. Atherosclerosis. 27:347–351. 1977.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Yoshinari O, Shiojima Y and Igarashi K: Anti-obesity effects of onion extract in Zucker diabetic fatty rats. Nutrients. 4:1518–1526. 2012.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Yanagita T, Han SY, Wang YM, Tsuruta Y and Anno T: Cycloalliin, a cyclic sulfur imino acid, reduces serum triacylglycerol in rats. Nutrition. 19:140–143. 2003.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Ichikawa M, Mizuno I, Yoshida J, Ide N, Ushijima M, Kodera Y, Hayama M and Ono K: Pharmacokinetics of cycloalliin, an organosulfur compound found in garlic and onion, in rats. J Agric Food Chem. 54:9811–9819. 2006.PubMed/NCBI View Article : Google Scholar
|




















