Introduction
Aging is a major risk factor for various diseases
and disorders (1) and leads to a
decline in the biological functions of multiple organs, including
the gastrointestinal (GI) tract (2). Aging-related GI disorders, including
esophageal reflux, dysphagia, chronic constipation, and fecal
incontinence (3), can also be
associated with sarcopenia and frailty leading to increased overall
mortality (4,5). Approximately half of people 65 years
or older experience at least one significant GI symptom (6). Dysphagia was found in approximately
one third of community-dwelling elderly, and more than half of
elderly nursing home residents (7). Almost one-third of adults 60 years or
older report at least occasional constipation (8). Also, GI disorders are more prevalent
in women than men, and the prevalence of several GI disorders
increases with age (9,10). GI disorders are more common in
females due to various factors including reproductive hormones
(11,12), gut sensation (10), and mental disorders (13-15).
Despite high prevalence, the treatment options for aging-related GI
disorders are limited, therefore novel therapeutic approaches are
warranted to improve the quality of life of elderly individuals.
The enteric nervous system (ENS) is an extensive network of neurons
and glial cells within the wall of the GI tract and plays a
critical role in regulating gut motility and other fundamental gut
functions (16,17). ENS abnormalities and GI dysfunction
including neuronal loss and slow transit, respectively have been
well described in older animals and humans and are believed to be
one of the etiologies of aging-related GI disorders (18-21).
Therefore, targeting the ENS may be a novel therapeutic approach
for these conditions (19).
Aged garlic extract (AGE) is a unique garlic product
produced by aging garlic in a water-ethanol process for more than
10 months. During this time, sulfur-containing amino acids
including S-allylcysteine (SAC), S-1-propenylcysteine (S1PC), and
S-allylmercaptocysteine (SAMC) are produced (22,23).
Several clinical trials have shown that AGE has beneficial effects
in patients with hypertension (24-27)
and gingivitis (28,29). Increasing evidence indicates that
SAC and S1PC possess diverse medical benefits, including
anti-hypertensive (30-34),
anti-oxidant (35-37),
anti-aging (38,39), and anti-inflammatory (40-44)
properties. Several GI studies have shown that AGE changes
microbiota composition (27,45,46),
attenuates inflammation in an animal model of colitis (44), and protects epithelial cells from
methotrexate-induced cytotoxicity (47). However, the effects of AGE on GI
motility and ENS in older subjects are not well understood. In this
study we investigated the effects of AGE on gut motility and ENS
cells using old mice and explored potential mechanisms of
action.
Materials and methods
Animals
All animal protocols were approved by the
Institutional Animal Care and Use Committee at Massachusetts
General Hospital (Protocols #2009N000239 an #2013N000115). All
methods were performed in accordance with relevant regulations.
Females of C57BL/6J (Stock #007914) mice, both 2-3-month-old
(Young) and 18-month-old (Old), were purchased from Jackson
Laboratory (Bar Harbor, ME). 18-month-old mice represent
approximately 56 years of age in humans (48,49).
For generation of dual reporter mice in which enteric neurons
express tdTomato and enteric glia/progenitors express GFP,
Plp1GFP mice (50) were kindly gifted by Dr. Wendy
Macklin, University of Colorado, Denver. To obtain
Plp1GFP;Actl6b::Cre; ROSA26-tdTomato (annotated
as Plp1GFP; Baf53b-tdT) mice, Actl6b::Cre
mice (Stock #027826) were crossed with Plp1GFP
mice, and their offspring were crossed with ROSA26-tdTomato
mice (Stock #007914) (51). In
order to isolate longitudinal muscle layer with myenteric plexus
(LMMP) and enteric neural cells animals were euthanized by carbon
dioxide overdose, which is displacement of chamber air with
compressed carbon dioxide at 30-70% per min. Death of animals was
confirmed by lack of chest movement and heartbeat, after which
cervical dislocation was performed. Both male and female mice were
used for the in vitro studies as the influence of factors
such as gut sensation and reproductive hormons can be excluded.
Chemicals
AGE was prepared from cloves of garlic (Allium
sativum L.) through a process of rinsing with purified water,
slicing, soaking in ethanol 20-50% (v/v), and extracting/aging for
more than 10 months (52). AGE
powder was obtained from Wakunaga Pharmaceutical Co. ltd and sent
to Bio-Serv (Flemington, NJ) to prepare 3% AGE-formulated diet
using standard mouse chow (AIN-93G, Bio-Serv, NJ). The AIN-93G the
purified rodent diet that containing 200 g Casein, 100 g Sucrose,
397.5 g Cornstarch, 132 g Dyetrose, 3 g L-cystine, 50 g Cellulose,
70 g Soybean oil, 0.014 g t-Butylhydroquinone, 35 g Mineral mix, 10
g Vitamin mix, and 2.5 g Choline bitartrate per kg of diet
(53). The 3% AGE-formulated diet
was given to female mice for 2 weeks after the mice were randomized
based on body weight. Two weeks after the initiation of feeding the
AGE-formulated diet, in vivo assays and analysis of
oxidative stress in myenteric plexus were performed. For in
vitro and ex vivo studies, AGE powder was dissolved in
sterile PBS and the solution was passed through with a 0.22 µM
filter.
Gastrointestinal transit time
Total gastrointestinal transit time was measured as
previously described (54). Mice
(n=6, each group) were acclimatized for 30 min individually in
cages without bedding, and 0.15 ml of 6% (w/v) carmine red dye
(Sigma, C1022) in 0.5% (w/v) methylcellulose was administered to
each mouse by oral gavage. The time from gavage to the appearance
of the first red pellet was recorded as total gastrointestinal
transit time. Maximum observation time was 6 h.
Bead expulsion test
Mice (n=6, each group) were fasted overnight before
the test was performed as described previously (55). Mice were acclimatized for 30 min,
then a 3-mm glass bead (Sigma, #1040150500) was inserted into the
rectum of each mouse using a silicone pusher under anesthesia by
isoflurane (Covetrus, #11695-6777-2). Isoflurane, at 3 and 2%, was
used for induction and maintenance, respectively. After bead
insertion, mice were placed in individual plastic cages. The time
to evacuate the bead started after the mice recovered from the
anesthesia.
Fecal pellet output
Fecal pellet output was measured as reported
previously (56) in individual
metabolic cages for 24 h (n=6, each group). The weight of food
consumed over 24 h was obtained. Pellet number was calculated using
the average weights of dry feces (n=25) per mouse.
Fecal water content
The previously described method was used (57). Each mouse (n=6, each group) was
placed in a plastic cage individually and wet fecal pellets were
collected for 2 h. The wet fecal pellets were dried in the oven at
60˚C for 48 h. Fecal water content was calculated according to the
following equation: (wet fecal weight-dry fecal weight)/wet fecal
weight x100.
Organ bath measurements of colonic
smooth muscle activity
The organ bath experiments with colon rings followed
well-established protocols. We conducted pilot studies to optimize
parameters such as intensity (40-50 volts), frequency (5 Hz), and
pulse duration (0.3 ms) to ensure reproducible and physiologically
relevant responses for assessing smooth muscle contractility. These
parameters have been validated in our laboratory, and experiments
were performed using sparameters previously described (58-61).
Freshly excised segments of distal colon were immediately placed in
oxygenated Krebs solution (118 mmol/l NaCl, 4.7 mmol/l KCl, 1.2
mmol/l MgSO4·7H2O, 1.2 mmol/l
KH2PO4, 25 mmol/l NaHCO3, 11.7
mmol/l glucose, and 1.25 mmol/l CaCl2) at 37˚C. Tissue
rings, approximately 5 mm in length, were mounted between two metal
hooks attached to force displacement transducers in a muscle strip
myograph bath (Model 820 MS; Danish Myo Technology, Aarhus,
Denmark) containing 7 ml of oxygenated Krebs solution. The rings
were gently stretched to establish a basal tension of 0.5 g and
allowed to equilibrate for 30-45 min, with Krebs solution being
replaced every 20 min. Spontaneous contractions were recorded in
both the absence and presence of AGE (1% w/v). Afterwards,
contractions were recorded again following the addition of the
nitric oxide synthase inhibitor L-NAME (100 mM; Sigma-Aldrich, St.
Louis, MO). Electrical field stimulation (EFS) was then applied to
the tissue using a pulse train of 40-50 V (15-sec duration, 300 µs
pulse width, 5 Hz frequency) via a CS4+ constant voltage stimulator
controlled by MyoPulse software (Danish Myo Technology). The
procedure was repeated after the addition of AGE (1% w/v). Force
contraction data from the circular smooth muscle were recorded and
analyzed using a Power Lab 16/35 data acquisition system and
LabChart Pro Software v8.1.16 (ADInstruments, NSW, Australia).
Tissue viability was confirmed by assessing the contraction
response to 60 mM KCl at the conclusion of the experiment. Baseline
spontaneous activity was quantified by measuring the area under the
curve (AUC), from 60 sec of data collected 5 min before the
addition of AGE or L-NAME This was compared to the response
following AGE or L-NAME, which was similarly quantified by
measuring the AUC, from 60 sec of data collected immediately after
the addition of AGE or L-NAME. Baseline maximum values were
determined by averaging 60 sec of data recorded 1 min before EFS
application. Changes in contraction were measured from the first 60
sec after stimulation onset and expressed as absolute differences
from baseline. EFS was applied three times at 5-min intervals, and
the maximum response was calculated as the mean of the three
trials.
Isolation and expansion of mouse
enteric neural cells
Enteric neural cells, including enteric neural stem
cells (ENSCs), were isolated from mice as previously reported
(62-64).
Briefly, LMMP was separated from large intestine of mice (young,
old, and Plp1GFP; Baf53b-tdT). Enzymatic dissociation
was achieved using dispase (250 µg/ml, STEMCELL Technologies,
Vancouver, Canada) and collagenase XI (1 mg/ml, Sigma Aldrich, St.
Louis, Missouri) at 37˚C for 45 min. Single cells were isolated by
filtration through a 40-µm filter and plated at 5x105
cells/ml in a 25-cm2 flask in a 1:1 mixture of DMEM
(Thermo Fisher Scientific) and NeuroCult Mouse Basal Medium
(StemCell Technologies) supplemented with 1%
penicillin/streptomycin (Gibco, #15140122), 20 ng/ml insulin growth
factor (StemCell Technologies), and 20 ng/ml basic fibroblast
growth factor (StemCell Technologies), 2% B27 supplement (gibco),
1% N2 supplement (gibco), 50 µM beta-mercaptoethanol (gibco), and
75 ng/ml retinoic acid (Sigma Aldrich). After 7 days in culture,
primary neurospheres were obtained.
Neurosphere assay and cell viability
assay
Primary neurospheres from wildtype C57BL/6J (n=2,
male and female) and PLP1GFP; BAF53b-tdT mice (n=2, male
and female) were dissociated by Accutase (StemCell technologies, #
7920). 5,000 cells/well were plated into 96-well plate (CORNING,
#3474) and secondary neurospheres treated with AGE at 0.25 to 1
mg/ml in culture media for 7 days (n=3, each group). The samples
were dissociated with dispase and collagenase XI to generate single
cell suspension and fixed with 4% PFA for 15 min. Random images of
secondary neurospheres from wildtype C57BL/6J were taken using a
Keyence BZX-700 All-In-One Microscope (Keyence America Itasca, IL)
and the number of neurospheres from wildtype C57BL/6J quantified by
ImageJ software (NIH). A cell viability assay was performed using
the CellTiter-Glo Luminescent Cell Viability Assay (Promega,
Madison, WI, USA, PAG7570). Secondary neurospheres from
Plp1GFP; Baf53b-tdT mice were dissociated to single
cells and tdT+ and GFP+ positive cell numbers were counted using
ImageJ software (NIH).
EdU proliferation assay and
immunohistochemistry
Primary neurospheres from wildtype C57BL/6J mice
(n=2, male and female) were generated as described previously. AGE
(1 mg/ml) was added to the media for secondary neurospheres in the
presence of 10 µM of 5-ethynyl-2'-deoxyuridine (EdU) for 2 days
(n=3, each group). After removing the media, secondary neurospheres
were incubated in new AGE-containing media for 5 days. The samples
were dissociated with dispase and collagenase XI to generate single
cell suspension fixed with 4% PFA for 15 min and Click-iT EdU Cell
Proliferation Kit for Imaging (Fisher Scientific, C10340) was
performed. For immunohistochemical staining, 10% donkey serum and
1% Triton X-100 in phosphate-buffered saline (PBS) was used for
blocking. Primary antibodies, including human anti-HuC/D (Anna1,
1:20, kindly gifted by Lennon lab) and rabbit anti-P75 (1;400,
Promega, G3231), were incubated overnight at 4˚C, followed by
secondary antibodies for 1 h. Secondary antibodies included
anti-human IgG (1:200, Alexa Fluor 594, Jackson ImmunoResearch) and
anti-rabbit IgG (1:200; Alexa Fluor 488, Invitrogen). Random Images
were taken from each group using a Keyence BZX-700 All-In-One
Microscope (Keyence America Itasca, IL) and the number of
double-labelled Hu+EdU+ or P75+EdU+ cells counted using ImageJ
software (NIH).
Oxidative stress in myenteric
plexus
MitoSOX (Thermo Fisher Scientific, M36008) was used
to identify mitochondrial-derived production of superoxide in the
myenteric ganglia of the ENS (65). Fresh colonic LMMP preparations were
collected from the mice (n=4, each group). The samples were
incubated in Hanks' Balanced Salt Solution (HBSS, Thermo Fisher
Scientific) containing 5 µM of MitoSOX at 37˚C for 30 min. Tissues
were washed with PBS and fixed with 4% PFA overnight at 4˚C. Images
were captured, converted into binary format, and area of
fluorescence within ganglia was measured in arbitrary units using
ImageJ software (NIH).
ENS cell culture and evaluation of the
effects of AGE
LMMPs were separated from colons dissected from 2
months old Plp1GFP; Baf53b-tdT mice (n=2, male and
female) and dissociated enzymatically using dispase (250 µg/ml,
STEMCELL Technologies) and collagenase XI (1 mg/ml, Sigma-Aldrich)
at 37˚C for 45 min. Counter filtration was performed using a 20-µm
cell strainer (pluriSelect, #43-50020-01) as previously (66). Samples were centrifuged at 350 G
for 5 min and resuspended in NeuroCult Mouse Basal Medium (StemCell
Technologies) containing 10% FBS and 1% penicillin/streptomycin
(Gibco, #15140122). Isolated enteric ganglia in the media were
plated into fibronectin (Sigma-Aldrich, #F1141)-coated 48 well
plates and cultured for 24 h. Pre-treatment of AGE without hydrogen
peroxide (H2O2) was performed for 48 h. Then,
PBS treatment as control, 100 µM of H2O2
(Sigma-Aldrich, H1009) alone, and co-treatment of AGE and 100 µM of
H2O2 were carried out for 24 h (n=3, each
group). The samples were fixed with 4% PFA for 15 min at RT. Random
images were taken using a Keyence BZX-700 All-In-One Microscope
(Keyence America Itasca, IL). The number of neurons and glial
cells, and neurite length, were quantified using ImageJ software
(NIH).
Statistical analysis
Data analysis was performed using GraphPad Prism v10
(GraphPad Software, Inc., San Diego, CA). Two-tailed t-tests were
performed for pairwise comparisons. A one-way analysis of variance
(ANOVA) was performed with a post hoc Dunnett's test and Tukey's
test for multiple comparisons. For all analyses, P<0.05 was
considered significant. All data are presented as mean ± SEM,
unless otherwise stated.
Results
AGE improves colorectal dysfunction in
old mice
To characterize gastrointestinal (GI) motility in
old mice, we performed multiple in vivo functional assays in
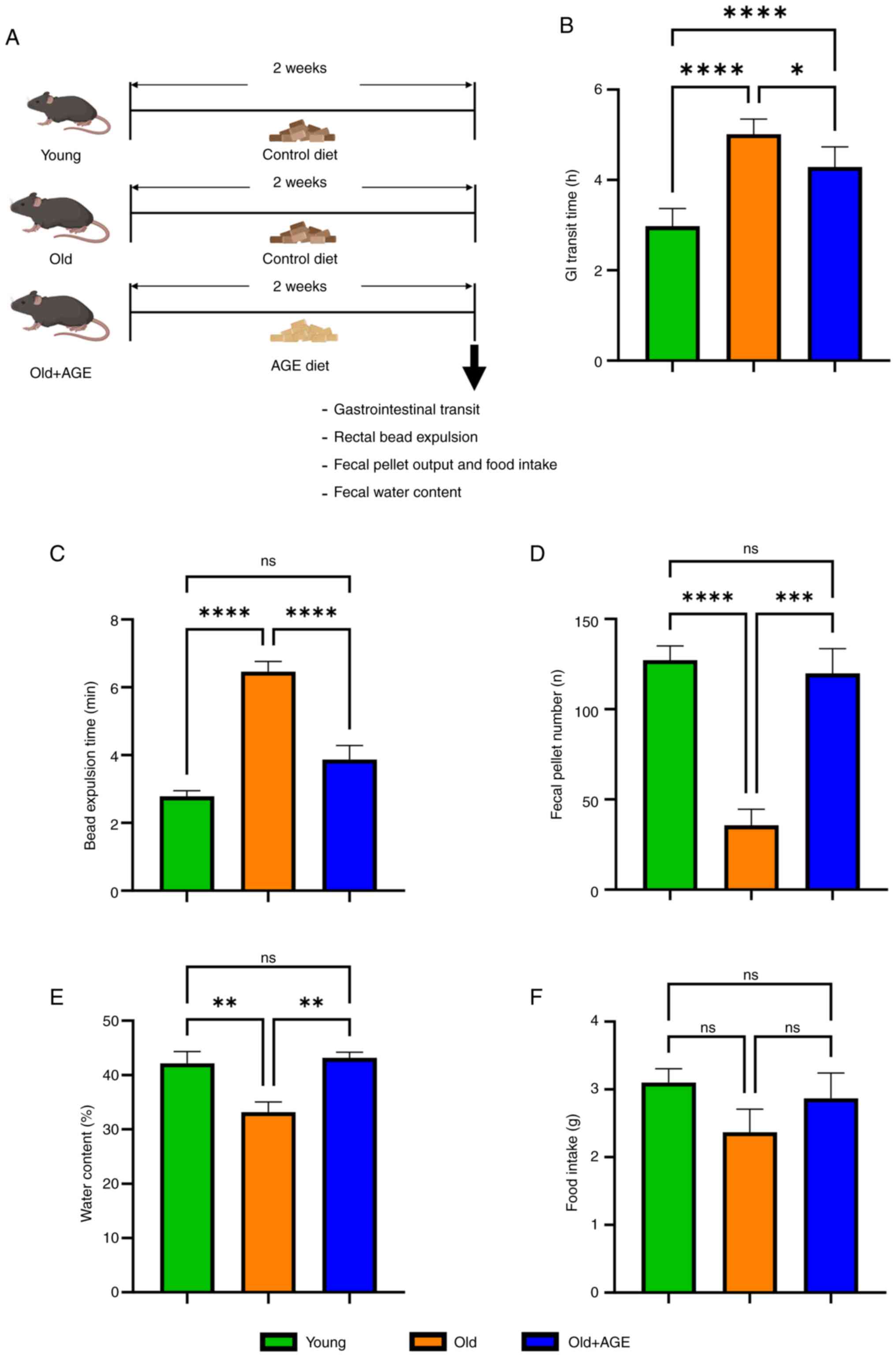
young (2-3-month-old) and old (18-month-old) mice (Fig. 1A). Both total GI transit time
(Fig. 1B, 2.98±0.2 h in young vs.
5.01±0.1 h in old, P<0.0001) and rectal bead expulsion time
(Fig. 1C, 2.78±0.2 min in young
vs. 6.47±0.3 min in old, P<0.0001) were significantly delayed in
old mice. Furthermore, significantly decreased fecal pellet output
(Fig. 1D, 127±7.8 in young vs.
35.7±8.9 in old, P<0.0001) and fecal water content (Fig. 1E, 42.7±2.2% in young vs. 33.2±1.9%
in old, P<0.01) were observed in old mice despite no significant
difference in food intake (Fig.
1F, 3.10±0.2 g in young vs. 2.37±0.3 g in old, ns).
AGE-formulated diet was given to old mice for 2
weeks and the same GI functional analyses were performed (Fig. 1A). Interestingly, all GI functional
parameters, including total GI transit time (Fig. 1B, 5.01±0.1 h in old vs. 4.29±0.2 h
in old+AGE, P<0.05), rectal bead expulsion time (Fig. 1C, 6.47±0.3 min in old vs. 3.87±0.4
min in old+AGE, P<0.0001), fecal pellet output (Fig. 1D, 35.7±8.9 in old vs. 120±14 in
old+AGE, P<0.001), and fecal water content (Fig. 1E, 33.2±1.9% in old vs. 43.2±1.0% in
old+AGE, P<0.01) were significantly improved by the AGE-diet.
Food intake was not significantly reduced by AGE-diet (Fig. 1F, 2.37±0.3 g in old vs. 2.87±0.4 g
in old+AGE, ns). These findings suggest that AGE has the potential
to restore aging-related GI dysmotility in mice.
AGE increases smooth muscle relaxation
of aged colon via nNOS signaling
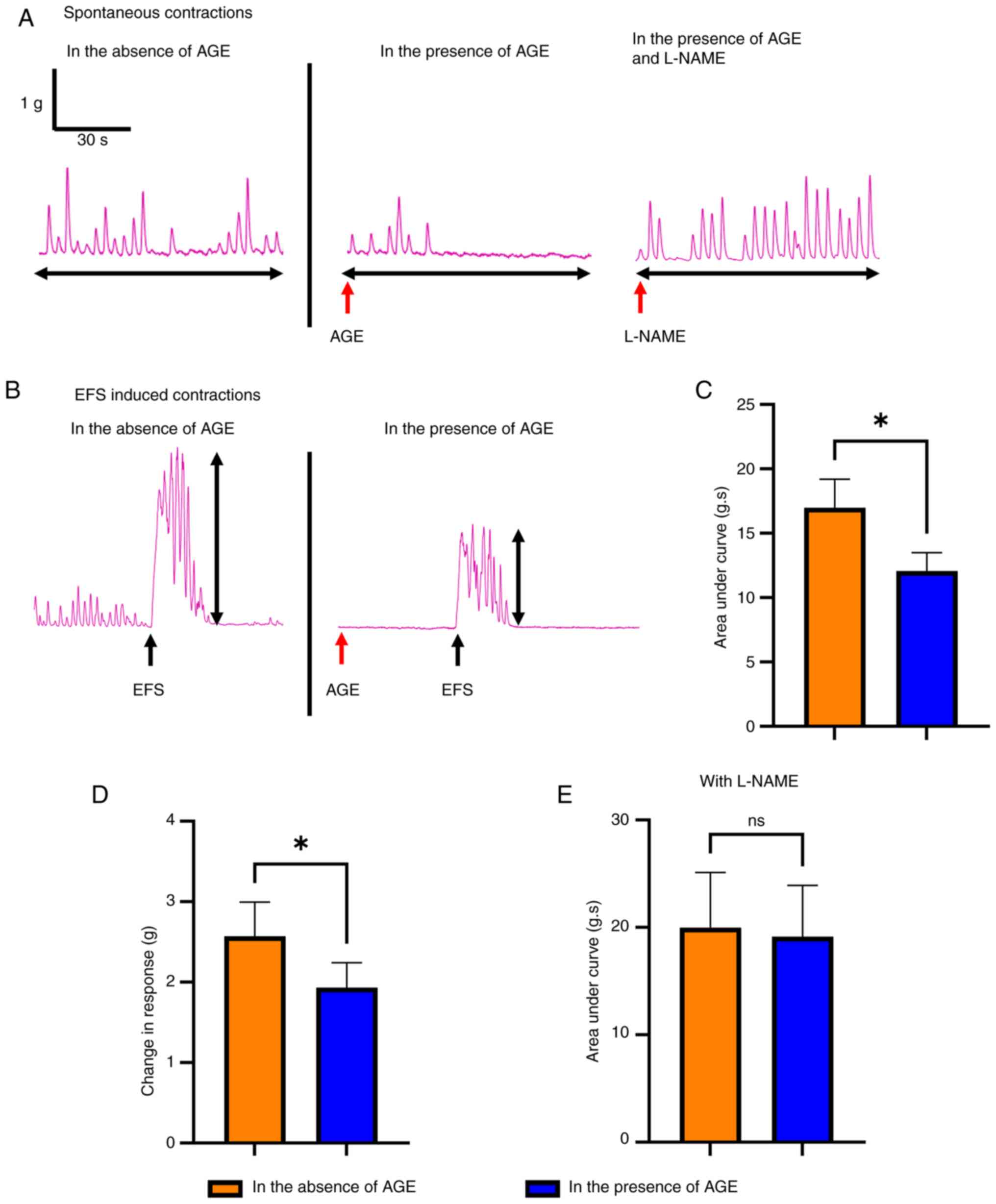
To evaluate the effect of AGE on colonic smooth
muscle contractility, we performed organ bath studies on colon from
18-month-old mice. The representative tracings of each group during
the baseline recording (Fig. 2A)
and in response to electrical field stimulation (EFS) (Fig. 2B) are shown. In the presence of
AGE, both baseline contractile activity (Fig. 2A) and EFS induced responses
(Fig. 2B) were reduced compared to
non-AGE-treated tissues. Quantitative analysis, determined by
measuring area under the curve (AUC) and the amplitude of EFS
responses, were significantly reduced in the presence of AGE
(Fig. 2C, 16.4±1.8 g.s in absence
of AGE vs. 13.4±1.7 g.s in presence of AGE, P<0.05; Fig. 2D, 2.41±0.4 g in absence of AGE vs.
1.75±0.3 g in presence of AGE, P<0.05). These changes in
response to AGE were attenuated in the presence of L-NAME
(N(ω)-nitro-L-arginine methyl ester), an inhibitor of neuronal
nitric oxide synthase (nNOS) (Fig.
2E, 20.0±5.1 g.s in absence of AGE vs. 19.1±4.8 g.s in presence
of AGE, ns), suggesting that the effect of AGE on colonic
contractility is mediated, at least in part, by nNOS.
AGE leads to expansion of cultured ENS
cells
Recent evidence has demonstrated active regeneration
and remodeling of the ENS postnatally (67). To evaluate the effect of AGE on ENS
homeostasis in old mice, we isolated enteric neuronal
stem/progenitor cells (ENSCs) from the colon of young and old
mice.
Interestingly, viability of ENSCs isolated from old
mice was significantly reduced in comparison to young-derived ENSCs
(Fig. 3A, 100±5.0% in young ENSC
vs. 14.8±0.3% in old ENSC, P<0.0001), and the number of
old-mouse-derived ENSCs was also significantly lower (Fig. S1A, 4.9±0.4 in young ENSC vs.
2.5±0.4 in old ENSC, P<0.05). Addition of AGE to the culture
media increased cell viability in and the number of both young- and
old-mouse-derived ENSCs (Figs. 3B
and S1B). We isolated ENSCs from
Plp1GFP; Baf53b-tdT mice in which enteric neurons
express tdTomato and enteric glia/progenitors express GFP (51), and cultured them in the presence or
absence of AGE (Fig. 3D-I).
Quantitative analysis demonstrated that AGE significantly increased
the Plp1-GFP population in a dose dependent manner (Fig. 3F and G, yellow arrows; Fig. 3J). AGE also expanded the neuronal
(Baf53b-tdT positive, Fig. 3D and
E, white arrows; Fig. 3K) population, but only at the 1
mg/ml concentration (Fig. 3K,
7.41±0.8 in control vs. 12.3±0.6 in AGE 1 mg/ml, P<0.01).
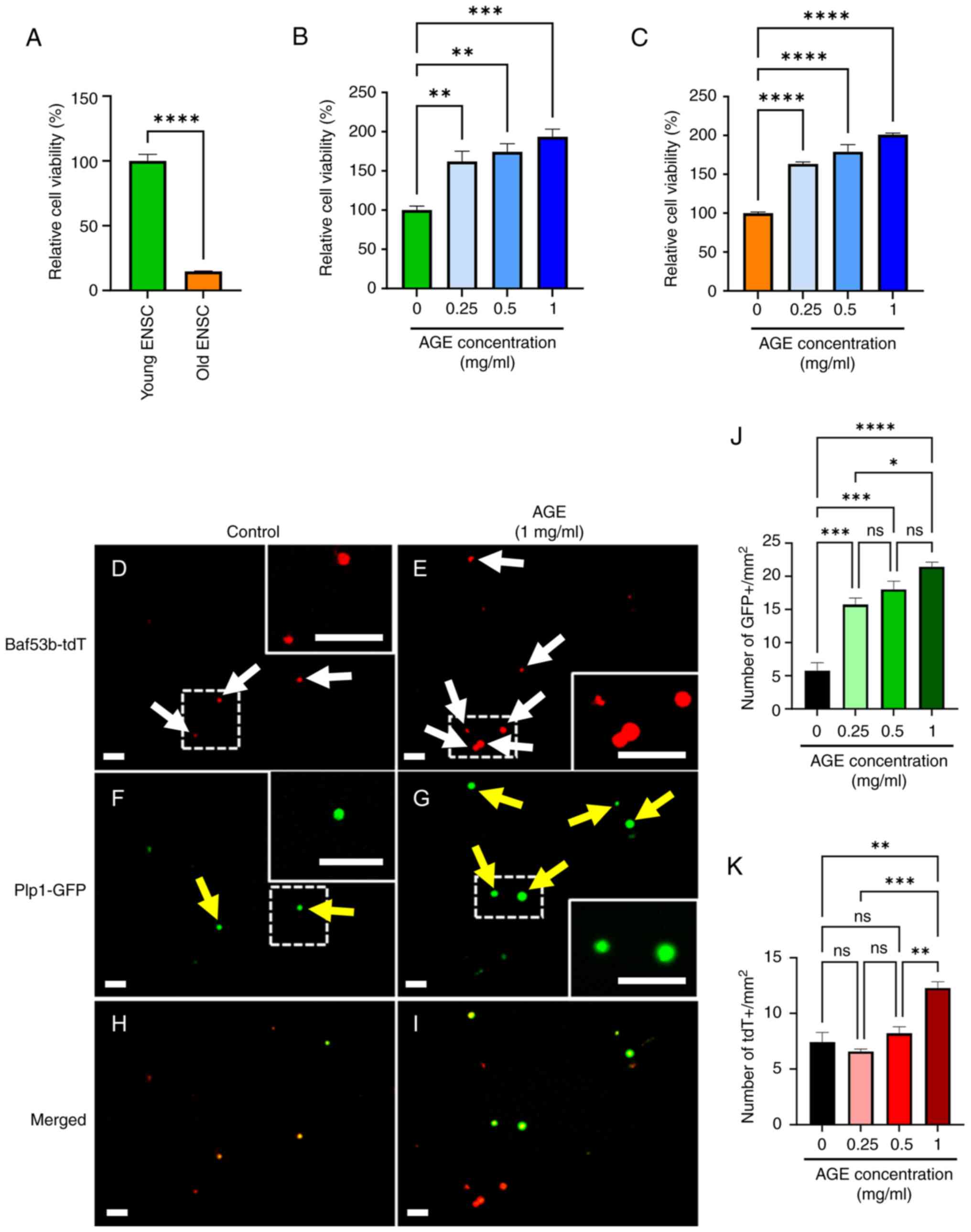 | Figure 3Effects of AGE on cultured ENS cells.
(A) Significant reduction in viability of cells isolated from ‘Old’
mice. AGE improved viability of (B) ‘Young’- or (C) ‘Old’-derived
ENSCs. (D-I) ENS cells were isolated from Plp1GFP;
Baf53b-tdT mice. (F, G and J) AGE increased the number of Plp1-GFP
positive glia/neural progenitors (yellow arrows) and (D, E and K)
Baf-tdT positive neurons (white arrows), respectively (dashed box
enlarged in inset). Scale bars, 50 µm; magnification, x20. Results
are shown as mean ± SEM, n=3/group. *P<0.05,
**P<0.01, ***P<0.001,
****P<0.0001. AGE, aged garlic extract; ENS, enteric
nervous system; ENSC, enteric neural stem cells. |
AGE promotes proliferation of enteric
neural cells
We tested whether AGE activates proliferation in ENS
cells using the thymidine analogue, EdU
(5-Ethynyl-2'-deoxyuridine). Isolated ENSCs were cultured in the
absence (Control) or presence of AGE (1 mg/ml) for 2 days in medium
containing EdU. We replaced the culture medium on day 3 and
maintained the culture for an additional 5 days. Immunofluorescent
staining was performed using anti-Hu and anti-P75 antibodies
(Fig. 4A-H) to label neurons and
glia/ENS progenitors, respectively. AGE promoted cell
proliferation, as shown by an increase in neurons that are double
positive for EdU+/Hu+ (Fig. 4I,
1.87±0.2 in control vs. 5.87±0.5 in AGE 1 mg/ml, P<0.01) and an
increase in EdU+/P75+ as glia/progenitors (Fig. 4J, 1.43±0.2 in control vs. 3.41±0.2
in AGE 1 mg/ml, P<0.01).
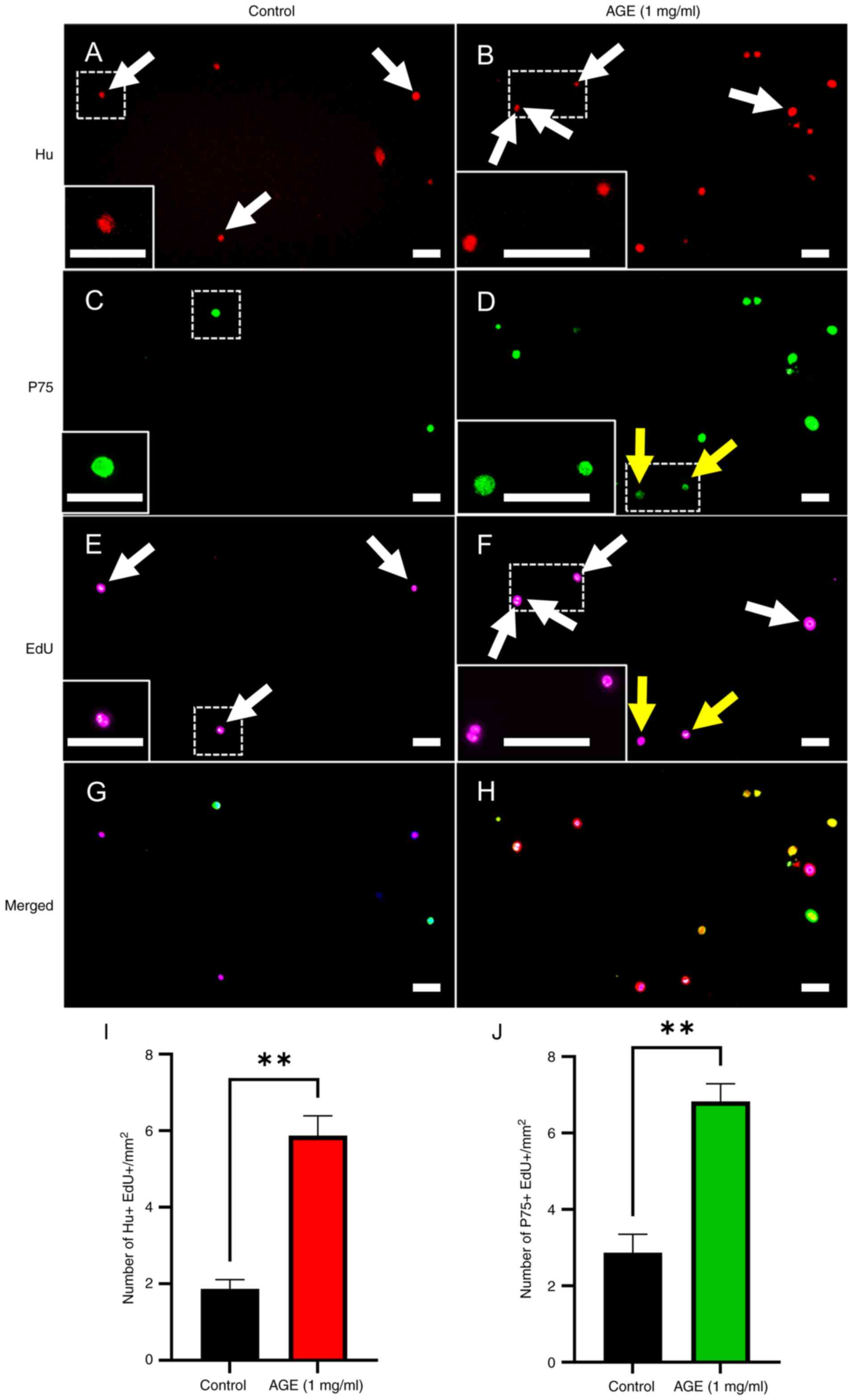 | Figure 4Effects of AGE on proliferation of
cultured enteric neural cells. Cultured enteric neural cells were
treated with PBS as vehicle control and immunostained for (A) Hu
(neuron), (C) P75 (glia/neural progenitor cell) and (E) EdU, with
the merged image shown in (G), (dashed box enlarged in inset).
Treatment of 1 mg/ml AGE for cultured enteric neural cells was
performed, followed by immunocytochemistry for (B) Hu, (D) P75 and
(F) EdU, with the merged image presented in (H) (dashed boxes
indicate enlarged insets). White arrows indcate Hu+/EdU+ neurons by
treatment of (A and E) vehicle control and (B and F) 1 mg/ml AGE,
respectively. (D and F) P75+/EdU+ cells were marked by yellow
arrows in presence of 1 mg/ml AGE. Both the number of (I) Hu+/EdU+
neurons and (J) P75+/EdU+ cells significantly increased in the
presence of 1 mg/ml AGE. Scale bars, 50 µm; magnification, x20,
Results are shown as mean ± SEM, n=3/group. **P<0.01.
EdU; 5-ethynyl-2'-deoxyuridine; AGE, aged garlic extract. |
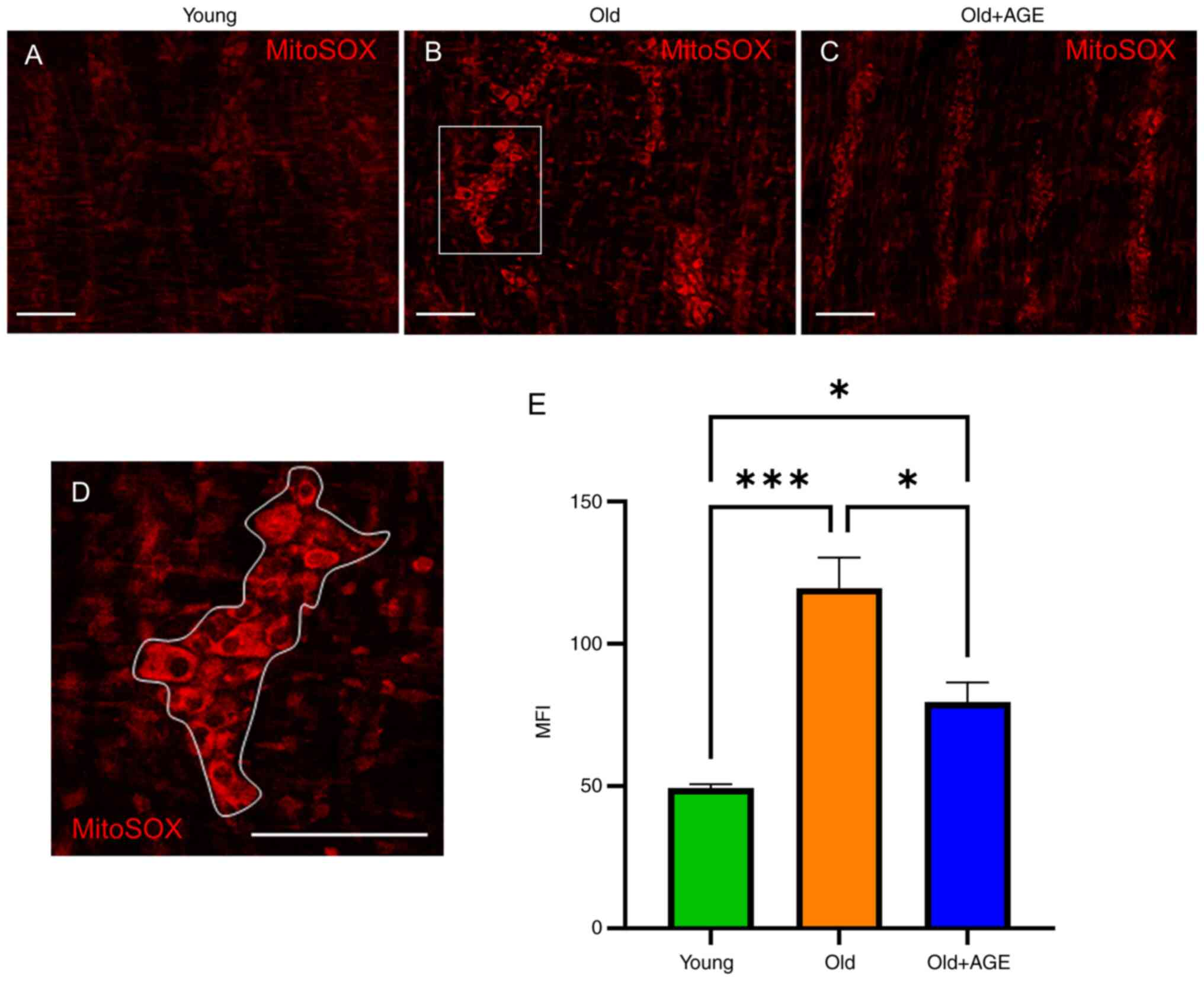
AGE reduces oxidative stress in ENS
from old mice
Neurons are susceptible to oxidative stress
(68), which plays a role in
aging-related enteric neuronal damage (69). Therefore, we evaluated reactive
oxygen species (ROS) within the myenteric plexus of the muscular
layer of mouse colon using MitoSOX labeling. The ROS visualized by
MitoSOX in old mice (Fig. 5B and
E) (Fig. 5E, 48.7±1.5 in young vs. 117±13 in
old, P<0.001) was significantly more prominent compared to that
in young mice (Fig. 5A and
E), supporting the idea that aging
increases oxidative stress in the ENS. Old mice fed an
AGE-formulated diet for 2 weeks (Fig.
1A) demonstrated significantly reduced ROS in the myenteric
plexus (Fig. 5C and E) (Fig.
5E, 117±13 in old vs. 74.7±6.1 in old+AGE, P<0.05). These
findings suggest that AGE ameliorates oxidative stress in the ENS
of old mice.
AGE protects against oxidative
stress-induced degeneration of cultured enteric neural cells
Several recent studies have shown that prevention of
oxidative stress could be a novel therapeutic strategy for
neurodegenerative disorders (70-72).
Here, we tested whether AGE has neuroprotective effects on cultured
ENS cells. Hydrogen peroxide (H2O2) is
commonly used to induce neuronal damage via oxidative stress
(73,74). We isolated ENS cells from
Plp1GFP; Baf53b-tdT mice and cultured them in the
absence (Fig. 6A-C) or presence of
H2O2 (Fig.
6D-F). In the presence of H2O2, there was
a significant reduction in the number of tdT+ neurons (Fig. 6M, 37.3±3.2 in control vs. 5.00±0.6
in H2O2, P<0.0001) and GFP+
glia/progenitors (Fig. 6N, 156±9.4
in control vs. 63.2±4.6 in H2O2,
P<0.0001). We also found a significant reduction in the length
of neurites (Fig. 6O, 50.0±2.7 in
control vs. 4.67±0.9 in H2O2, P<0.0001)
and decreased cell viability (Fig.
6P, 100±11 in control vs. 46.3±7.9 in
H2O2, P<0.01) in
H2O2-treated ENS cells, confirming that
oxidative stress elicits damage to ENS cells in culture. When AGE
was added to the H2O2-treated ENS cells at
0.5 mg/ml (Fig. 6G-I) and 1 mg/ml
(Fig. 6J-L), there was significant
improvement in the survival and health of neurons and glia. These
findings suggest that AGE possesses neuroprotective effects on ENS
cells against oxidative stress.
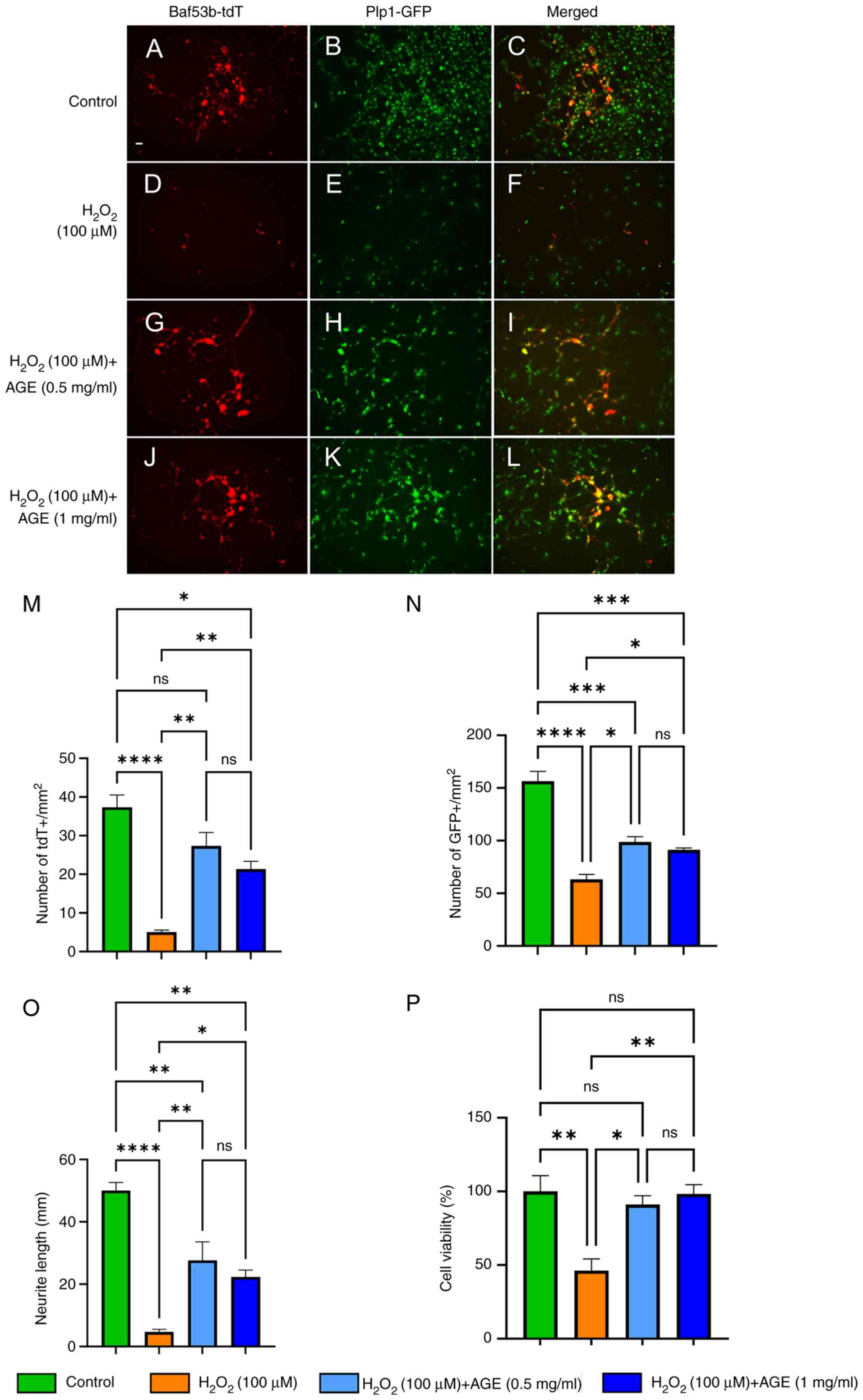 | Figure 6Neuroprotective effects of AGE on
oxidative stress-induced enteric neural cells in culture. (A and B)
Fluorescence images showed neurons and glial/neural progenitor
cells labeled with Baf53b-tdT and Plp1GFP, respectively in the
presence of PBS as a vehicle control, exhibiting normal morphology,
and (C) is the merged image of (A) and (B). (D) The tdT-labeled
neurons exposed to 100 µM hydroperoxide display neuronal damage.
(E) GFP-labeled glial/neural progenitor cells appeared to a
reduction of cell number, and (F) is the merged image of (D) and
(E). The tdT-labeled neurons and GFP-labeled glial/neural
progenitor cells were co-treated with 100 µM hydroperoxide and (G
and H) 0.5 mg/ml or (J and K) 1 mg/ml AGE, which exhibited
neuroprotective effects. (I) Presents the merged image of (G) and
(H), and (L) presents the combined image of (J) and (K). The (M)
number of neurons and (N) glial/neural progenitors, (O) neurite
length and (P) cell viability were measured. Scale bars, 50 µm;
magnification, x10. Results are shown as mean ± SEM, n=3/group.
*P<0.05, **P<0.01,
***P<0.001, ****P<0.0001. AGE, aged
garlic extract. |
Discussion
In this study we examined the effects of AGE on
colonic smooth muscle and ENS cells in old mice and demonstrated
several positive effects on GI motility via colonic smooth muscle
relaxation, enhancement of proliferation in enteric neural cells,
and neuroprotective effects by reduction in oxidative stress. Our
findings provide new insights for the treatment of aging-related GI
dysmotility through use of a food supplement. The ENS is an
extensive network of neurons and glia within the wall of the GI
tract. The ENS regulates a variety of functions, including
intestinal motility, sensation, absorption, secretion, and immunity
(16). Abnormalities of the ENS,
which can result from a number of different conditions, including
age-associated neurodegeneration, lead to serious morbidity and
reduced quality of life. Aging-related GI motility disorders are
common, and include esophageal reflux, dysphagia, chronic
constipation, rectal prolapse, and fecal incontinence. These
contribute to the development of subsequent undernutrition,
immunosuppression, sarcopenia, and frailty. GI disorders including
irritable bowel syndrome and constipation are more prevalent in
women than men, and several GI disorders increase with age
(9,10). Therefore, we used aged female mice
in this study. The mechanisms underlying aging-associated GI
dysfunction are not fully understood, although a number of studies
have shown that neuronal loss and/or molecular changes in the ENS
may be involved (20,75,76).
In the current study, we used 18-month-old mice as ‘old’ mice to
evaluate i) gut motility using in vivo and ex vivo
assays, ii) neurodegenerative ENS phenotype based on the degree of
oxidative stress, and iii) properties of enteric neuronal stem
cells (ENSCs) in culture. Consistent with previous reports
(56,75), our study shows that both total gut
transit time and rectal bead expulsion time are significantly
delayed in old mice, and fecal pellet output and fecal water
content are significantly reduced. We also observed significantly
increased oxidative stress and a reduction in the proliferative
capacity of ENSCs from old mice grown in culture, consistent with
prior studies (69,77,78).
AGE is a nutritional supplement prepared by
prolonged extraction (normally over 10 months) of fresh garlic with
15-20% aqueous ethanol at room temperature (52). This product is odorless and appears
to be superior to normal garlic in its antioxidant properties
(79). It has been shown that AGE
reduces total serum cholesterol and systolic pressure in
hypercholesterolemic patients (27,80,81).
AGE was further shown to promote antioxidant protection in cells by
enhancing activity of the cellular antioxidant enzymes (35,79).
Furthermore, recent work has demonstrated AGE-related
neuroprotective effects against oxidative stress during
neuroinflammation (79), leading
us to hypothesize that AGE could have similar neuroprotective
effects on the ENS and thus ameliorate the symptoms of GI
dysmotility associated with aging.
Oxidative stress is characterized by an imbalance
between enhanced production of reactive oxygen species (ROS),
including hydrogen peroxide (H2O2) and
superoxide anion (O2-), and/or reduced antioxidant
defenses due to mitochondrial dysfunction and a decline in
antioxidant defenses with aging (82,83).
This imbalance leads to lipid peroxidation and oxidation of
proteins and DNA, leading to neurodegeneration (84,85).
Elevated ROS levels in myenteric neurons in aged mice are
associated with neuronal apoptosis (69), and dysmotility has been described
in aganglionic mouse models (58,86)
and old mice (56,75). Consistent with these previous
reports, we observed significant elevation of ROS in the muscular
layer of colon in old mice compared to young. Interestingly, these
changes were ameliorated by an AGE-formulated diet for 2 weeks. It
has been shown that AGE and its constituents, such as
S-allylcysteine (SAC) and S-allylmercaptocysteine (SAMC), contain
antioxidant properties (87). SAC
is known to activate Nuclear factor erythroid 2-related factor 2
(Nrf2) signaling pathway, resulting in enhancement of antioxidant
defense (22,88). Furthermore, some antioxidants,
including N-acetylcysteine (89),
Vitamin C (90), and Resveratrol
(91), have been shown to have
neuroprotective properties. The current study, using
H2O2-exposed neural cells to induce
neurodegeneration in vitro (89), demonstrated a significant reduction
in neuroglial damage in H2O2 treated cells in
the presence of AGE. Therefore, these neuroprotective effects via
reduction of ROS could be a mechanism explaining how AGE treatment
improves aging-related colorectal dysmotility. Further studies
investigating how AGE promotes ENS cell proliferation and reduces
oxidative stress could explore the effects of AGE and its
components on Nrf2 signaling including Nrf2, and its target genes
such as HO-1 and GCLC (92,93).
The ENS is embryologically derived from the neural
crest. During development, as undifferentiated NCCs migrate through
the wall of GI tract, they differentiate into neurons and glial
cells to form interconnecting enteric ganglia. A subpopulation of
those enteric neural crest-derived cells remains undifferentiated
and resides within enteric ganglia where they could act to
replenish damaged or lost neurons in response to various insults,
including inflammation, mechanical stretch, and aging (67,94,95).
These cells, so-called enteric neuronal stem/progenitor cells
(ENSCs), have been isolated from postnatal mice (60,62,96,97), swine, and humans and their
capabilities, such as proliferation and migration, have been
characterized following their culture in vitro (98-102).
Kruger et al showed that in mice, the number of ENSCs
declines more than 10-fold within the first 3 months of life.
Moreover, their self-renewal capacity and neuronal differentiation
potential decline by 50-60% (77).
In our current study, we observed that the viability and the number
of neurospheres generated by cultured ENSCs from old mice was
significantly reduced compared to those from young mice.
Interestingly, we found that the proliferative capacity of cultured
ENSCs was enhanced by the addition of AGE. Although the mechanisms
by which ENSC proliferation declines with age are not fully
understood, a major chemical component of AGE, promotes the Nrf2
signaling pathway (22,88), which plays a key role in driving
the cell cycle transition from G2 to M phase in hepatocytes
(103) and also plays a role in
regulating injury-induced neurogenesis in the brain (104). Based on our observed restoration
in colorectal dysmotility in old mice treated with AGE, we
hypothesize that improving an imbalance between contraction and
relaxation in the gut could be beneficial for treatment of
aging-related dysmotility. Since intestinal peristalsis consists of
coordinated movements involving both contractions and relaxations
(105), in support of this idea,
colorectal dysmotility was described in nNOS KO mice (64) and reduction in the number of nNOS
neurons was observed in old mice (76), suggesting that a reduction in
(inhibitory) nNOS neurons results in a change in the ratios of
excitatory and inhibitory enteric neurons, and that this imbalance
may contribute to aging-related dysmotility. Further, AGE has been
shown to have a relaxation effect in vascular smooth muscle by
increasing nitric oxide production (106). In this current study, we observed
that AGE significantly impacts colonic smooth muscle contractility,
as both baseline contractile activity and electrically evoked
responses in colonic muscle strips were reduced in the presence of
AGE, findings that together highlight its potent modulatory
influence on motility. The observed effects of AGE suggest that it
increases muscle relaxation, as reduced contraction and enhanced
relaxation are complementary aspects of muscle tone regulation
(107). Furthermore, these
effects of AGE were blocked by L-NAME, an inhibitor of nNOS,
suggesting that nitric oxide (NO) plays an important role in
mediating the muscle relaxant effects of AGE. NO is generated from
L-Arginine as a substrate by nNOS, which promotes smooth muscle
relaxation by activating guanylate cyclase and increasing cyclic
GMP levels (108,109). L-Arginine is one of the
components found in AGE, and as a main source of NO has a
relaxation effect in vascular smooth muscle (106). The role of NO in mediating smooth
muscle relaxation, particularly in the rodent colon, is
well-established, and NO released from sodium nitroprusside induces
relaxation of gastrointestinal smooth muscle (110-112).
The observed inhibition of AGE's effects by L-NAME reinforces the
involvement of the nNOS-NO pathway in mediating AGE's action
(91). Thus NO derived from
L-arginine contained in AGE may be inducing the relaxation effects
that we observed in colonic smooth muscle, which could lead to
improvement in aging-related dysmotility. This could be further
investigated in future studies using nitrate/nitrite colorimetric
assays as previously described (33,106), and molecular biological assays
(western blotting, quantitative PCR) and/or biochemical assays
could be used to evaluate the effects of AGE and its constituents
on downstream targets of nNOS signaling including guanylate
cyclase, PKA/PKG and myosin light chain kinase (113).
Although not directly tested in our studies,
another mechanism that could be involved in the observed effects of
AGE on gut contractility is smooth muscle relaxation via hydrogen
sulfide (H2S) (114),
and organic polysulfides as its potential source is contained in AGE
(115-117).
H2S facilitates membrane hyperpolarization in smooth
muscle cells, reducing their excitability and contractile
responses. This effect may explain the observed attenuation in both
spontaneous contractility and EFS-induced responses, but requires
further investigation. The ability of H2S to modulate
ion channel activity and intracellular calcium dynamics further
supports this hypothesis, as these processes are critical for
smooth muscle contraction and neuromuscular signaling (118,119). These mechanisms highlight AGE's
potential to counteract age-related decline in gut motility
(60,88,90)
by promoting smooth muscle relaxation. Studies on GI physiology
have shown that interventions reducing contractility effectively
promote muscle relaxation (107,120). Furthermore, it is unknown if the
effects of AGE persist in the long term. Several previous studies
have shown that gut microbiota can influence GI motility (121) and have a range of effects on the
ENS (122). A human study
demonstrated that garlic intake for one week improved gut microbial
diversity and increased the relative abundance of beneficial
bacteria including Faecalibacterium prausnitzii and Akkermansia spp
(123), supporting the idea that
changes induced in the short term could have long term
implications. However, we recognize that determination of the
prolonged impact of AGE on the gut is a limitation of our study.
Nevertheless, our results provide valuable insight into the
therapeutic potential of AGE for enhancing colonic motility through
NO-dependent pathways, supporting previous observations of its
effects on smooth muscle systems (124).
In conclusion, an AGE diet in mice resulted in
improvements in aging-related colorectal dysmotility involving
colonic smooth muscle relaxation via nNOS, enhancement of
proliferation in enteric neural cells, and neuroprotective effects.
Our findings provide both a characterization of motility in aged
mice and reveal potential beneficial effects for GI motility
disorders and ENS cells through AGE.
Supplementary Material
Effects of AGE on ENSC capacity to
form neurospheres. (A) Neurosphere assays were performed to
determine the effects of AGE on ENSCs isolated ‘Young’ or ‘Old’
mice. (B) ‘Old’ mice-derived ENSCs were grown in the presence of
AGE. Results are shown as mean ± SEM, n=3/group.
*P<0.05, **P<0.01,
***P<0.001. AGE, aged garlic extract; ENSC, enteric
neural stem cells.
Acknowledgements
Not applicable.
Funding
Funding: This work was supported by a research grant from
Wakunaga Pharmaceutical Co. Ltd. (grant no. 243138-MGB).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
KO and RH designed the experiments and are major
contributors in writing the manuscript. KO performed in
vitro study. KO, RH and TO preformed in vivo study. AAR
performed organ bath study. KO, RH and AAR confirmed the
authenticity of all the raw data. AJB and AMG supervised this
study, contributed to the conception and data interpretation, and
reviewed and edited the manuscript. All authors have read and
approved the final manuscript.
Ethics approval and consent to
participate
This study was conducted in accordance with the
protocols reviewed and approved by the Institutional Animal Care
and Use Committee at Massachusetts General Hospital (approval nos.
2009N000239 and 2013N000115). All methods were carried out in
accordance with relevant guidelines and regulations.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have competing
interests: the work was funded by Wakunaga Pharmaceutical Company
Ltd., where KO is an employee.
References
|
1
|
Kennedy BK, Berger SL, Brunet A, Campisi
J, Cuervo AM, Epel ES, Franceschi C, Lithgow GJ, Morimoto RI,
Pessin JE, et al: Geroscience: Linking aging to chronic disease.
Cell. 159:709–713. 2014.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Campisi J: Aging, cellular senescence, and
cancer. Annu Rev Physiol. 75:685–705. 2013.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Saffrey MJ: Aging of the mammalian
gastrointestinal tract: A complex organ system. Age (Dordr).
36(9603)2014.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Cox NJ, Ibrahim K, Sayer AA, Robinson SM
and Roberts HC: Assessment and treatment of the anorexia of aging:
A systematic review. Nutrients. 11(144)2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Parker BA and Chapman IM: Food intake and
ageing-the role of the gut. Mech Ageing Dev. 125:859–866.
2004.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Siddappa PK and Birk JW: Gastrointestinal
health and healthy aging. In: Healthy Aging: A Complete Guide to
Clinical Management. Coll PP (ed). Springer International
Publishing, Cham, pp67-79, 2019.
|
|
7
|
Doan TN, Ho WC, Wang LH, Chang FC, Nhu NT
and Chou LW: Prevalence and methods for assessment of oropharyngeal
dysphagia in older adults: A systematic review and meta-analysis. J
Clin Med. 11(2605)2022.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Mounsey A, Raleigh M and Wilson A:
Management of constipation in older adults. Am Fam Physician.
92:500–504. 2015.PubMed/NCBI
|
|
9
|
Chang L, Toner BB, Fukudo S, Guthrie E,
Locke GR, Norton NJ and Sperber AD: Gender, age, society, culture,
and the patient's perspective in the functional gastrointestinal
disorders. Gastroenterology. 130:1435–1446. 2006.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Kim YS and Kim N: Sex-gender differences
in irritable bowel syndrome. J Neurogastroenterol Motil.
24:544–558. 2018.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Alqudah M, Al-Shboul O, Al Dwairi A,
Al-U´Datt DG and Alqudah A: Progesterone inhibitory role on
gastrointestinal motility. Physiol Res. 71:193–198. 2022.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Oh JE, Kim YW, Park SY and Kim JY:
Estrogen rather than progesterone cause constipation in both female
and male mice. Korean J Physiol Pharmacol. 17:423–426.
2013.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Shah E, Rezaie A, Riddle M and Pimentel M:
Psychological disorders in gastrointestinal disease: Epiphenomenon,
cause or consequence? Ann Gastroenterol. 27:224–230.
2014.PubMed/NCBI
|
|
14
|
Haug TT, Mykletun A and Dahl AA: Are
anxiety and depression related to gastrointestinal symptoms in the
general population? Scand J Gastroenterol. 37:294–298.
2002.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Salk RH, Hyde JS and Abramson LY: Gender
differences in depression in representative national samples:
Meta-analyses of diagnoses and symptoms. Psychol Bull. 143:783–822.
2017.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Furness JB: The enteric nervous system and
neurogastroenterology. Nat Rev Gastroenterol Hepatol. 9:286–294.
2012.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Rao M and Gershon MD: Enteric nervous
system development: What could possibly go wrong? Nat Rev Neurosci.
19:552–565. 2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Camilleri M, Cowen T and Koch TR: Enteric
neurodegeneration in ageing. Neurogastroenterol Motil. 20:418–429.
2008.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Saffrey MJ: Cellular changes in the
enteric nervous system during ageing. Dev Biol. 382:344–355.
2013.PubMed/NCBI View Article : Google Scholar
|
|
20
|
El-Salhy M, Sandström O and Holmlund F:
Age-induced changes in the enteric nervous system in the mouse.
Mech Ageing Dev. 107:93–103. 1999.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Bernard CE, Gibbons SJ, Gomez-Pinilla PJ,
Lurken MS, Schmalz PF, Roeder JL, Linden D, Cima RR, Dozois EJ,
Larson DW, et al: Effect of age on the enteric nervous system of
the human colon. Neurogastroenterol Motil. 21:746–e46.
2009.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Colín-González AL, Santana RA, Silva-Islas
CA, Chánez-Cárdenas ME, Santamaría A and Maldonado PD: The
antioxidant mechanisms underlying the aged garlic extract- and
S-allylcysteine-induced protection. Oxid Med Cell Longev.
2012(907162)2012.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Matsutomo T and Kodera Y: Development of
an analytic method for sulfur compounds in aged garlic extract with
the use of a postcolumn high performance liquid chromatography
method with sulfur-specific detection. J Nutr. 146:450S–455S.
2016.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Ried K, Frank OR and Stocks NP: Aged
garlic extract lowers blood pressure in patients with treated but
uncontrolled hypertension: A randomised controlled trial.
Maturitas. 67:144–150. 2010.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Ried K, Frank OR and Stocks NP: Aged
garlic extract reduces blood pressure in hypertensives: A
dose-response trial. Eur J Clin Nutr. 67:64–70. 2013.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Ried K, Travica N and Sali A: The effect
of aged garlic extract on blood pressure and other cardiovascular
risk factors in uncontrolled hypertensives: The AGE at Heart trial.
Integr Blood Press Control. 9:9–21. 2016.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Ried K, Travica N and Sali A: The effect
of kyolic aged garlic extract on gut microbiota, inflammation, and
cardiovascular markers in hypertensives: The GarGIC trial. Front
Nutr. 5(122)2018.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Zini A, Mann J, Mazor S and Vered Y: The
efficacy of aged garlic extract on gingivitis-A randomized clinical
trial. J Clin Dent. 29:52–56. 2018.PubMed/NCBI
|
|
29
|
Takahashi K, Nango H, Ushijima M,
Takashima M, Nakamoto M, Matsutomo T, Jikihara H, Arakawa N, Maki
S, Yabuki A, et al: Therapeutic effect of aged garlic extract on
gingivitis in dogs. Front Vet Sci. 10(1277272)2023.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Harauma A and Moriguchi T: Aged garlic
extract improves blood pressure in spontaneously hypertensive rats
more safely than raw garlic. J Nutr. 136 (3 Suppl):769S–773S.
2006.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Matsutomo T, Ushijima M, Kodera Y,
Nakamoto M, Takashima M, Morihara N and Tamura K: Metabolomic study
on the antihypertensive effect of S-1-propenylcysteine in
spontaneously hypertensive rats using liquid chromatography coupled
with quadrupole-Orbitrap mass spectrometry. J Chromatogr B Analyt
Technol Biomed Life Sci. 1046:147–155. 2017.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Matsutomo T, Ushijima M, Kunimura K and
Ohtani M: Metabolomic study reveals the acute hypotensive effect of
S-1-propenylcysteine accompanied by alteration of the plasma
histidine level in spontaneously hypertensive rats. J Pharm Biomed
Anal. 168:148–154. 2019.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Ushijima M, Takashima M, Kunimura K,
Kodera Y, Morihara N and Tamura K: Effects of S-1-propenylcysteine,
a sulfur compound in aged garlic extract, on blood pressure and
peripheral circulation in spontaneously hypertensive rats. J Pharm
Pharmacol. 70:559–565. 2018.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Matsutomo T: Potential benefits of garlic
and other dietary supplements for the management of hypertension.
Exp Ther Med. 19:1479–1484. 2020.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Borek C: Antioxidant health effects of
aged garlic extract. J Nutr. 131 (3S):1010S–1015S. 2001.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Yamato O, Tsuneyoshi T, Ushijima M,
Jikihara H and Yabuki A: Safety and efficacy of aged garlic extract
in dogs: Upregulation of the nuclear factor erythroid 2-related
factor 2 (Nrf2) signaling pathway and Nrf2-regulated phase II
antioxidant enzymes. BMC Vet Res. 14(373)2018.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Tsuneyoshi T: BACH1 mediates the
antioxidant properties of aged garlic extract. Exp Ther Med.
19:1500–1503. 2020.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Moriguchi T, Saito H and Nishiyama N: Aged
garlic extract prolongs longevity and improves spatial memory
deficit in senescence-accelerated mouse. Biol Pharm Bull.
19:305–307. 1996.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Ogawa T, Kodera Y, Hirata D, Blackwell TK
and Mizunuma M: Natural thioallyl compounds increase oxidative
stress resistance and lifespan in Caenorhabditis elegans by
modulating SKN-1/Nrf. Sci Rep. 6(21611)2016.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Morihara N, Hino A, Miki S, Takashima M
and Suzuki JI: Aged garlic extract suppresses inflammation in
apolipoprotein E-knockout mice. Mol Nutr Food Res.
61:2017.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Suzuki JI, Kodera Y, Miki S, Ushijima M,
Takashima M, Matsutomo T and Morihara N: Anti-inflammatory action
of cysteine derivative S-1-propenylcysteine by inducing MyD88
degradation. Sci Rep. 8(14148)2018.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Ohtani M and Nishimura T:
Sulfur-containing amino acids in aged garlic extract inhibit
inflammation in human gingival epithelial cells by suppressing
intercellular adhesion molecule-1 expression and IL-6 secretion.
Biomed Rep. 12:99–108. 2020.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Miki S, Suzuki JI, Kunimura K and Morihara
N: Mechanisms underlying the attenuation of chronic inflammatory
diseases by aged garlic extract: Involvement of the activation of
AMP-activated protein kinase. Exp Ther Med. 19:1462–1467.
2020.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Liu J: Aged garlic therapeutic
intervention targeting inflammatory pathways in pathogenesis of
bowel disorders. Heliyon. 10(e33986)2024.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Maeda T, Miki S, Morihara N and Kagawa Y:
Aged garlic extract ameliorates fatty liver and insulin resistance
and improves the gut microbiota profile in a mouse model of insulin
resistance. Exp Ther Med. 18:857–866. 2019.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Ried K: Garlic lowers blood pressure in
hypertensive subjects, improves arterial stiffness and gut
microbiota: A review and meta-analysis. Exp Ther Med. 19:1472–1478.
2020.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Horie T, Li T, Ito K, Sumi S and Fuwa T:
Aged garlic extract protects against methotrexate-induced apoptotic
cell injury of IEC-6 cells. J Nutr. 136 (3 Suppl):861S–863S.
2006.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Kang YK, Min B, Eom J and Park JS:
Different phases of aging in mouse old skeletal muscle. Aging
(Albany NY). 14:143–160. 2022.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Jackson SJ, Andrews N, Ball D, Bellantuono
I, Gray J, Hachoumi L, Holmes A, Latcham J, Petrie A, Potter P, et
al: Does age matter? The impact of rodent age on study outcomes.
Lab Anim. 51:160–169. 2017.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Mallon BS and Macklin WB: Overexpression
of the 3'-untranslated region of myelin proteolipid protein mRNA
leads to reduced expression of endogenous proteolipid mRNA.
Neurochem Res. 27:1349–1360. 2002.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Guyer RA, Stavely R, Robertson K, Bhave S,
Mueller JL, Picard NM, Hotta R, Kaltschmidt JA and Goldstein AM:
Single-cell multiome sequencing clarifies enteric glial diversity
and identifies an intraganglionic population poised for
neurogenesis. Cell Rep. 42(112194)2023.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Kodera Y, Kurita M, Nakamoto M and
Matsutomo T: Chemistry of aged garlic: Diversity of constituents in
aged garlic extract and their production mechanisms via the
combination of chemical and enzymatic reactions. Exp Ther Med.
19:1574–1584. 2020.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Bradham KD, Scheckel KG, Nelson CM, Seales
PE, Lee GE, Hughes MF, Miller BW, Yeow A, Gilmore T, Serda SM, et
al: Relative bioavailability and bioaccessibility and speciation of
arsenic in contaminated soils. Environ Health Perspect.
119:1629–1634. 2011.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Sasselli V, Boesmans W, Vanden Berghe P,
Tissir F, Goffinet AM and Pachnis V: Planar cell polarity genes
control the connectivity of enteric neurons. J Clin Invest.
123:1763–1772. 2013.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Natale G, Kastsiushenka O, Fulceri F,
Ruggieri S, Paparelli A and Fornai F: MPTP-induced parkinsonism
extends to a subclass of TH-positive neurons in the gut. Brain Res.
1355:195–206. 2010.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Patel BA, Patel N, Fidalgo S, Wang C,
Ranson RN, Saffrey MJ and Yeoman MS: Impaired colonic motility and
reduction in tachykinin signalling in the aged mouse. Exp Gerontol.
53:24–30. 2014.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Jeong D, Kim DH, Kang IB, Kim H, Song KY,
Kim HS and Seo KH: Modulation of gut microbiota and increase in
fecal water content in mice induced by administration of
Lactobacillus kefiranofaciens DN1. Food Funct. 8:680–686.
2017.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Rahman AA, Ohkura T, Bhave S, Pan W,
Ohishi K, Ott L, Han C, Leavitt A, Stavely R, Burns AJ, et al:
Enteric neural stem cell transplant restores gut motility in mice
with Hirschsprung disease. JCI Insight. 9(e179755)2024.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Rahman AA, Stavely R, Pan W, Ott L, Ohishi
K, Ohkura T, Han C, Hotta R and Goldstein AM: Optogenetic
activation of cholinergic enteric neurons reduces inflammation in
experimental colitis. Cell Mol Gastroenterol Hepatol. 17:907–921.
2024.PubMed/NCBI View Article : Google Scholar
|
|
60
|
McCann CJ, Cooper JE, Natarajan D, Jevans
B, Burnett LE, Burns AJ and Thapar N: Transplantation of enteric
nervous system stem cells rescues nitric oxide synthase deficient
mouse colon. Nat Commun. 8(15937)2017.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Fried DE, Watson RE, Robson SC and
Gulbransen BD: Ammonia modifies enteric neuromuscular transmission
through glial γ-aminobutyric acid signaling. Am J Physiol
Gastrointest Liver Physiol. 313:G570–G580. 2017.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Stavely R, Bhave S, Ho WLN, Ahmed M, Pan
W, Rahman AA, Ulloa J, Bousquet N, Omer M, Guyer R, et al: Enteric
mesenchymal cells support the growth of postnatal enteric neural
stem cells. Stem Cells. 39:1236–1252. 2021.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Bhave S, Arciero E, Baker C, Ho WL,
Stavely R, Goldstein AM and Hotta R: Enteric neuronal cell therapy
reverses architectural changes in a novel diphtheria toxin-mediated
model of colonic aganglionosis. Sci Rep. 9(18756)2019.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Hotta R, Rahman A, Bhave S, Stavely R, Pan
W, Srinivasan S, de Couto G, Rodriguez-Borlado L, Myers R, Burns AJ
and Goldstein AM: Transplanted ENSCs form functional connections
with intestinal smooth muscle and restore colonic motility in
nNOS-deficient mice. Stem Cell Res Ther. 14(232)2023.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Sahakian L, Filippone RT, Stavely R,
Robinson AM, Yan XS, Abalo R, Eri R, Bornstein JC, Kelley MR and
Nurgali K: Inhibition of APE1/Ref-1 redox signaling alleviates
intestinal dysfunction and damage to myenteric neurons in a mouse
model of spontaneous chronic colitis. Inflamm Bowel Dis.
27:388–406. 2021.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Mueller JL, Leavitt AR, Rahman AA, Han CY,
Ott LC, Mahdavian NS, Carbone SE, King SK, Burns AJ, Poole DP, et
al: Highly neurogenic glia from human and mouse myenteric ganglia
generate functional neurons following culture and transplantation
into the gut. Cell Rep. 43(114919)2024.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Jonscher R and Belkind-Gerson J: Concise
review: Cellular and molecular mechanisms of postnatal
injury-induced enteric neurogenesis. Stem Cells. 37:1136–1143.
2019.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Sohal RS and Weindruch R: Oxidative
stress, caloric restriction, and aging. Science. 273:59–63.
1996.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Thrasivoulou C, Soubeyre V, Ridha H,
Giuliani D, Giaroni C, Michael GJ, Saffrey MJ and Cowen T: Reactive
oxygen species, dietary restriction and neurotrophic factors in
age-related loss of myenteric neurons. Aging Cell. 5:247–257.
2006.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Li J, O W, Li W, Jiang ZG and Ghanbari HA:
Oxidative stress and neurodegenerative disorders. Int J Mol Sci.
14:24438–24475. 2013.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Liu Z, Zhou T, Ziegler AC, Dimitrion P and
Zuo L: Oxidative stress in neurodegenerative diseases: From
molecular mechanisms to clinical applications. Oxid Med Cell
Longev. 2017(2525967)2017.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Morén C, deSouza RM, Giraldo DM and Uff C:
Antioxidant therapeutic strategies in neurodegenerative diseases.
Int J Mol Sci. 23(9328)2022.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Fukui K: Reactive oxygen species induce
neurite degeneration before induction of cell death. J Clin Biochem
Nutr. 59:155–159. 2016.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Zhao X, Fang J, Li S, Gaur U, Xing X, Wang
H and Zheng W: Artemisinin attenuated hydrogen peroxide
(H2O2)-induced oxidative injury in sh-sy5y
and hippocampal neurons via the activation of AMPK pathway. Int J
Mol Sci. 20(2680)2019.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Becker L, Nguyen L, Gill J, Kulkarni S,
Pasricha PJ and Habtezion A: Age-dependent shift in macrophage
polarisation causes inflammation-mediated degeneration of enteric
nervous system. Gut. 67:827–836. 2018.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Sun T, Li D, Hu S, Huang L, Sun H, Yang S,
Wu B, Ji F and Zhou D: Aging-dependent decrease in the numbers of
enteric neurons, interstitial cells of Cajal and expression of
connexin43 in various regions of gastrointestinal tract. Aging
(Albany NY). 10:3851–3865. 2018.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Kruger GM, Mosher JT, Bixby S, Joseph N,
Iwashita T and Morrison SJ: Neural crest stem cells persist in the
adult gut but undergo changes in self-renewal, neuronal subtype
potential, and factor responsiveness. Neuron. 35:657–669.
2002.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Laszczyk AM, Fox-Quick S, Vo HT, Nettles
D, Pugh PC, Overstreet-Wadiche L and King GD: Klotho regulates
postnatal neurogenesis and protects against age-related spatial
memory loss. Neurobiol Aging. 59:41–54. 2017.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Song H, Lu Y, Qu Z, Mossine VV, Martin MB,
Hou J, Cui J, Peculis BA, Mawhinney TP, Cheng J, et al: Effects of
aged garlic extract and FruArg on gene expression and signaling
pathways in lipopolysaccharide-activated microglial cells. Sci Rep.
6(35323)2016.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Wlosinska M, Nilsson AC, Hlebowicz J,
Hauggaard A, Kjellin M, Fakhro M and Lindstedt S: The effect of
aged garlic extract on the atherosclerotic process-a randomized
double-blind placebo-controlled trial. BMC Complement Med Ther.
20(132)2020.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Lindstedt S, Wlosinska M, Nilsson AC,
Hlebowicz J, Fakhro M and Sheikh R: Successful improved peripheral
tissue perfusion was seen in patients with atherosclerosis after 12
months of treatment with aged garlic extract. Int Wound J.
18:681–691. 2021.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Yang J, Luo J, Tian X, Zhao Y, Li Y and Wu
X: Progress in understanding oxidative stress, aging, and
aging-related diseases. Antioxidants (Basel).
13(394)2024.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Chen X, Guo C and Kong J: Oxidative stress
in neurodegenerative diseases. Neural Regen Res. 7:376–385.
2012.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Sienes Bailo P, Llorente Martín E,
Calmarza P, Montolio Breva S, Bravo Gómez A, Pozo Giráldez A,
Sánchez-Pascuala Callau JJ, Vaquer Santamaría JM, Dayaldasani
Khialani A, Cerdá Micó C, et al: The role of oxidative stress in
neurodegenerative diseases and potential antioxidant therapies. Adv
Lab Med. 3:342–360. 2022.PubMed/NCBI View Article : Google Scholar : (In English,
Spanish).
|
|
85
|
Smith AN, Shaughness M, Collier S, Hopkins
D and Byrnes KR: Therapeutic targeting of microglia mediated
oxidative stress after neurotrauma. Front Med (Lausanne).
9(1034692)2022.PubMed/NCBI View Article : Google Scholar
|
|
86
|
Pan W, Rahman AA, Ohkura T, Stavely R,
Ohishi K, Han CY, Leavitt A, Kashiwagi A, Burns AJ, Goldstein AM
and Hotta R: Autologous cell transplantation for treatment of
colorectal aganglionosis in mice. Nat Commun.
15(2479)2024.PubMed/NCBI View Article : Google Scholar
|
|
87
|
Imai J, Ide N, Nagae S, Moriguchi T,
Matsuura H and Itakura Y: Antioxidant and radical scavenging
effects of aged garlic extract and its constituents. Planta Med.
60:417–420. 1994.PubMed/NCBI View Article : Google Scholar
|
|
88
|
Shi H, Jing X, Wei X, Perez RG, Ren M,
Zhang X and Lou H: S-allyl cysteine activates the Nrf2-dependent
antioxidant response and protects neurons against ischemic injury
in vitro and in vivo. J Neurochem. 133:298–308. 2015.PubMed/NCBI View Article : Google Scholar
|
|
89
|
Wu W, Liu BH, Xie CL, Xia XD and Zhang YM:
Neuroprotective effects of N-acetyl cysteine on primary hippocampus
neurons against hydrogen peroxide-induced injury are mediated via
inhibition of mitogen-activated protein kinases signal transduction
and antioxidative action. Mol Med Rep. 17:6647–6654.
2018.PubMed/NCBI View Article : Google Scholar
|
|
90
|
Shah SA, Yoon GH, Kim HO and Kim MO:
Vitamin C neuroprotection against dose-dependent glutamate-induced
neurodegeneration in the postnatal brain. Neurochem Res.
40:875–884. 2015.PubMed/NCBI View Article : Google Scholar
|
|
91
|
Hui Y, Chengyong T, Cheng L, Haixia H,
Yuanda Z and Weihua Y: Resveratrol attenuates the cytotoxicity
induced by amyloid-β1-42 in PC12 cells by upregulating
heme oxygenase-1 via the PI3K/Akt/Nrf2 pathway. Neurochem Res.
43:297–305. 2018.PubMed/NCBI View Article : Google Scholar
|
|
92
|
Tonelli C, Chio IIC and Tuveson DA:
Transcriptional regulation by Nrf2. Antioxid Redox Signal.
29:1727–1745. 2018.PubMed/NCBI View Article : Google Scholar
|
|
93
|
Moratilla-Rivera I, Sánchez M,
Valdés-González JA and Gómez-Serranillos MP: Natural products as
modulators of Nrf2 signaling pathway in neuroprotection. Int J Mol
Sci. 24(3748)2023.PubMed/NCBI View Article : Google Scholar
|
|
94
|
Nagy N and Goldstein AM: Enteric nervous
system development: A crest cell's journey from neural tube to
colon. Semin Cell Dev Biol. 66:94–106. 2017.PubMed/NCBI View Article : Google Scholar
|
|
95
|
Belkind-Gerson J, Graham HK, Reynolds J,
Hotta R, Nagy N, Cheng L, Kamionek M, Shi HN, Aherne CM and
Goldstein AM: Colitis promotes neuronal differentiation of Sox2+
and PLP1+ enteric cells. Sci Rep. 7(2525)2017.PubMed/NCBI View Article : Google Scholar
|
|
96
|
Hotta R, Stamp LA, Foong JP, McConnell SN,
Bergner AJ, Anderson RB, Enomoto H, Newgreen DF, Obermayr F,
Furness JB and Young HM: Transplanted progenitors generate
functional enteric neurons in the postnatal colon. J Clin Invest.
123:1182–1191. 2013.PubMed/NCBI View Article : Google Scholar
|
|
97
|
Stamp LA, Gwynne RM, Foong JPP, Lomax AE,
Hao MM, Kaplan DI, Reid CA, Petrou S, Allen AM, Bornstein JC and
Young HM: Optogenetic demonstration of functional innervation of
mouse colon by neurons derived from transplanted neural cells.
Gastroenterology. 152:1407–1418. 2017.PubMed/NCBI View Article : Google Scholar
|
|
98
|
Hotta R, Pan W, Bhave S, Nagy N, Stavely
R, Ohkura T, Krishnan K, de Couto G, Myers R, Rodriguez-Borlado L,
et al: Isolation, expansion, and endoscopic delivery of autologous
enteric neuronal stem cells in swine. Cell Transplant.
32(9636897231215233)2023.PubMed/NCBI View Article : Google Scholar
|
|
99
|
Lindley RM, Hawcutt DB, Connell MG, Almond
SL, Vannucchi MG, Faussone-Pellegrini MS, Edgar DH and Kenny SE:
Human and mouse enteric nervous system neurosphere transplants
regulate the function of aganglionic embryonic distal colon.
Gastroenterology. 135:205–216.e6. 2008.PubMed/NCBI View Article : Google Scholar
|
|
100
|
Cheng LS, Hotta R, Graham HK,
Belkind-Gerson J, Nagy N and Goldstein AM: Postnatal human enteric
neuronal progenitors can migrate, differentiate, and proliferate in
embryonic and postnatal aganglionic gut environments. Pediatr Res.
81:838–846. 2017.PubMed/NCBI View Article : Google Scholar
|
|
101
|
Metzger M, Bareiss PM, Danker T, Wagner S,
Hennenlotter J, Guenther E, Obermayr F, Stenzl A, Koenigsrainer A,
Skutella T and Just L: Expansion and differentiation of neural
progenitors derived from the human adult enteric nervous system.
Gastroenterology. 137:2063–2073.e4. 2009.PubMed/NCBI View Article : Google Scholar
|
|
102
|
Rollo BN, Zhang D, Stamp LA, Menheniott
TR, Stathopoulos L, Denham M, Dottori M, King SK, Hutson JM and
Newgreen DF: Enteric neural cells from hirschsprung disease
patients form ganglia in autologous aneuronal colon. Cell Mol
Gastroenterol Hepatol. 2:92–109. 2015.PubMed/NCBI View Article : Google Scholar
|
|
103
|
Lin L, Wu Q, Lu F, Lei J, Zhou Y, Liu Y,
Zhu N, Yu Y, Ning Z, She T and Hu M: Nrf2 signaling pathway:
Current status and potential therapeutic targetable role in human
cancers. Front Oncol. 13(1184079)2023.PubMed/NCBI View Article : Google Scholar
|
|
104
|
Kärkkäinen V, Pomeshchik Y, Savchenko E,
Dhungana H, Kurronen A, Lehtonen S, Naumenko N, Tavi P, Levonen AL,
Yamamoto M, et al: Nrf2 regulates neurogenesis and protects neural
progenitor cells against Aβ toxicity. Stem Cells. 32:1904–1916.
2014.PubMed/NCBI View Article : Google Scholar
|
|
105
|
Mittal RK: Integrated Systems Physiology:
From Molecule to Function to Disease. In: Motor Function of the
Pharynx, Esophagus, and its Sphincters. Morgan & Claypool Life
Sciences. Copyright © 2011 by Morgan & Claypool Life Sciences,
San Rafael (CA), 2011.
|
|
106
|
Takashima M, Kanamori Y, Kodera Y,
Morihara N and Tamura K: Aged garlic extract exerts
endothelium-dependent vasorelaxant effect on rat aorta by
increasing nitric oxide production. Phytomedicine. 24:56–61.
2017.PubMed/NCBI View Article : Google Scholar
|
|
107
|
Sanders KM: Regulation of smooth muscle
excitation and contraction. Neurogastroenterol Motil. 20 (Suppl
1):S39–S53. 2008.PubMed/NCBI View Article : Google Scholar
|
|
108
|
Sarna SK: Integrated Systems Physiology:
From Molecule to Function to Disease. In: Colonic Motility: From
Bench Side to Bedside. Morgan & Claypool Life Sciences.
Copyright © 2010 by Morgan & Claypool Life Sciences, San Rafael
(CA), 2010.
|
|
109
|
Mori M and Gotoh T: Regulation of nitric
oxide production by arginine metabolic enzymes. Biochem Biophys Res
Commun. 275:715–719. 2000.PubMed/NCBI View Article : Google Scholar
|
|
110
|
Bayer S, Jellali A, Crenner F, Aunis D and
Angel F: Functional evidence for a role of GABA receptors in
modulating nerve activities of circular smooth muscle from rat
colon in vitro. Life Sci. 72:1481–1493. 2003.PubMed/NCBI View Article : Google Scholar
|
|
111
|
Ghia JE, Pradaud I, Crenner F,
Metz-Boutigue MH, Aunis D and Angel F: Effect of acetic acid or
trypsin application on rat colonic motility in vitro and modulation
by two synthetic fragments of chromogranin A. Regul Pept.
124:27–35. 2005.PubMed/NCBI View Article : Google Scholar
|
|
112
|
Tepavcević SN, Isenović ER, Varagić VM and
Milovanović SR: Sodium nitroprusside regulates the relaxation of
the longitudinal muscle in the gut. Pharmazie. 63:151–155.
2008.PubMed/NCBI
|
|
113
|
Shah V, Lyford G, Gores G and Farrugia G:
Nitric oxide in gastrointestinal health and disease.
Gastroenterology. 126:903–913. 2004.PubMed/NCBI View Article : Google Scholar
|
|
114
|
Dunn WR, Alexander SP, Ralevic V and
Roberts RE: Effects of hydrogen sulphide in smooth muscle.
Pharmacol Ther. 158:101–113. 2016.PubMed/NCBI View Article : Google Scholar
|
|
115
|
Rose P, Moore PK and Zhu YZ: Garlic and
gaseous mediators. Trends Pharmacol Sci. 39:624–634.
2018.PubMed/NCBI View Article : Google Scholar
|
|
116
|
Benavides GA, Squadrito GL, Mills RW,
Patel HD, Isbell TS, Patel RP, Darley-Usmar VM, Doeller JE and
Kraus DW: Hydrogen sulfide mediates the vasoactivity of garlic.
Proc Natl Acad Sci USA. 104:17977–17982. 2007.PubMed/NCBI View Article : Google Scholar
|
|
117
|
Abe K, Hori Y and Myoda T: Volatile
compounds of fresh and processed garlic. Exp Ther Med.
19:1585–1593. 2020.PubMed/NCBI View Article : Google Scholar
|
|
118
|
Gallego D, Clavé P, Donovan J, Rahmati R,
Grundy D, Jiménez M and Beyak MJ: The gaseous mediator, hydrogen
sulphide, inhibits in vitro motor patterns in the human, rat and
mouse colon and jejunum. Neurogastroenterol Motil. 20:1306–1316.
2008.PubMed/NCBI View Article : Google Scholar
|
|
119
|
Gil V, Parsons S, Gallego D, Huizinga J
and Jimenez M: Effects of hydrogen sulphide on motility patterns in
the rat colon. Br J Pharmacol. 169:34–50. 2013.PubMed/NCBI View Article : Google Scholar
|
|
120
|
Kerfoot WW, Park HY, Schwartz LB, Hagen PO
and Carson CC III: Characterization of calcium channel blocker
induced smooth muscle relaxation using a model of isolated corpus
cavernosum. J Urol. 150:249–252. 1993.PubMed/NCBI View Article : Google Scholar
|
|
121
|
Zheng Z, Tang J, Hu Y and Zhang W: Role of
gut microbiota-derived signals in the regulation of
gastrointestinal motility. Front Med (Lausanne).
9(961703)2022.PubMed/NCBI View Article : Google Scholar
|
|
122
|
Carabotti M, Scirocco A, Maselli MA and
Severi C: The gut-brain axis: Interactions between enteric
microbiota, central and enteric nervous systems. Ann Gastroenterol.
28:203–209. 2015.PubMed/NCBI
|
|
123
|
Panyod S, Wu WK, Chen PC, Chong KV, Yang
YT, Chuang HL, Chen CC, Chen RA, Liu PY, Chung CH, et al:
Atherosclerosis amelioration by allicin in raw garlic through gut
microbiota and trimethylamine-N-oxide modulation. NPJ Biofilms
Microbiomes. 8(4)2022.PubMed/NCBI View Article : Google Scholar
|
|
124
|
Li M, Yun W, Wang G, Li A, Gao J and He Q:
Roles and mechanisms of garlic and its extracts on atherosclerosis:
A review. Front Pharmacol. 13(954938)2022.PubMed/NCBI View Article : Google Scholar
|