1. Introduction
Cardiovascular diseases (CVDs) including coronary
heart disease, cerebrovascular disease and rheumatic heart disease
are the leading cause of death worldwide, claiming ~17.9 million
lives annually. The major cause of CVDs is atherosclerosis, an
inflammatory disease that occurs in vessel walls (1,2). The
development of arteriosclerosis is associated with various factors,
including increased shear stress due to hypertension (3,4),
inflammation induced by damage-associated molecular patterns
(DAMPs) from dead cells (5-7)
and augmentation of oxidation and glycation products resulting from
hyperlipidemia including hypercholesterolemia (8,9) and
hyperglycemia (10,11). These factors contribute to the
damage of vascular endothelial cells, which subsequently leads to
the accumulation of low-density lipoprotein (LDL) cholesterol and
migration of circulating monocytes into the blood vessel wall
(12,13). The infiltrated monocytes
differentiate into macrophages, which phagocytose oxidized LDL
(oxLDL) and become foam cells, leading to the formation of plaque
with a lipid core (14,15). Subsequently, the progression of
fibrosis and calcification of the plaque make the plaque unstable
and prone to rupture. Plaque rupture and subsequent thrombus
formation can trigger cerebral or myocardial infarction (16). Thus, proper reduction of these risk
factors can help prevent and/or slow both the onset and progression
of atherosclerosis.
Garlic has been previously reported to be effective
in inhibiting the pathogenesis of CVDs and to help prevent chronic
diseases such as diabetes mellitus, cranial nerve disease and
cancer (17). Aged garlic extract
(AGE), prepared by aging crushed raw garlic in water-soluble
ethanol for at least 10 months, contains a variety of compounds
produced through aging processes. These constituents include
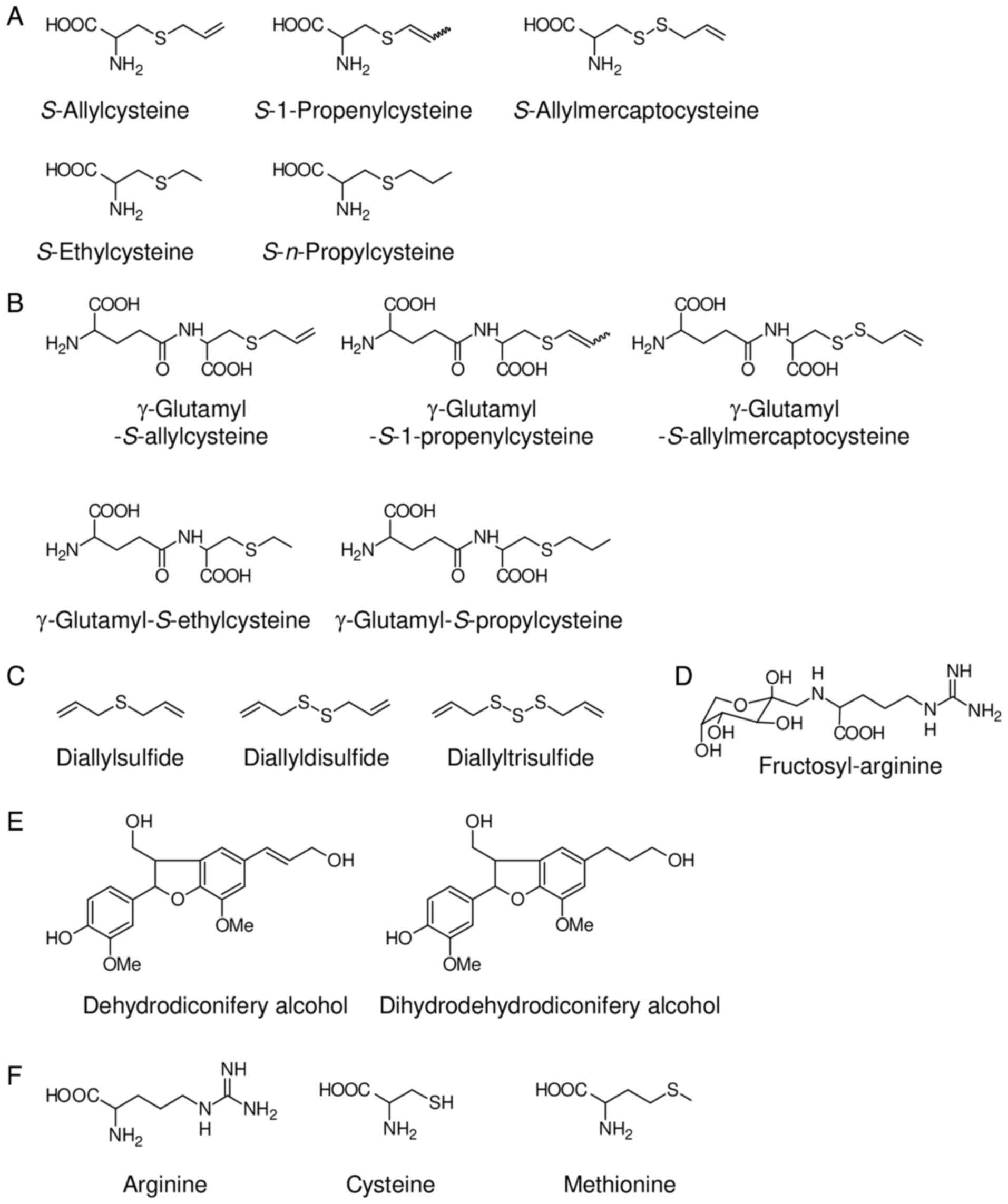
S-alk(en)ylcysteine compounds such as S-allylcysteine
(SAC), S-1-propenylcysteine (S1PC™; Wakunaga Holdings Co.,
Ltd.), S-allylmercaptocysteine (SAMC),
S-propylcysteine (SPC) and S-ethylcysteine (SEC);
diallyl polysulfide compounds such as diallyl sulfide (DAS),
diallyl disulfide (DADS) and diallyl trisulfide (DATS); Maillard
reaction-related compounds such as fructosyl-L-arginine (Fru-Arg);
phenolic compounds such as dehydrodiconiferyl alcohol (DDC) and
dihydrodehydrodiconiferyl alcohol (DDDC); and amino acids such as
L-arginine, L-cysteine and L-methionine (Fig. 1) (18,19).
However, AGE contains fewer irritating compounds, such as allicin,
when compared with raw garlic and causes little damage to the
gastric mucosa (20,21). Efendy et al (22) first reported in 1997 that AGE
inhibits the development of experimental atherosclerosis in
rabbits. Since then, the anti-atherosclerotic effects of AGE have
been demonstrated in various clinical (23-36)
and animal studies (19,22,37-40).
For example, clinical trials have shown that AGE can reduce
lipid-rich low attenuation plaque by ~30% (24,26)
and inhibit the progression of vascular calcification by ~65%
(27,29,30).
This review aimed to highlight the beneficial
effects and underlying mechanisms of action of AGE and its active
constituents in mitigating risk factors associated with the onset
of atherosclerosis and modulating key processes in its progression.
Additionally, the clinical relevance, availability and potential
applications of AGE in the prevention and treatment of
atherosclerosis was explored.
2. Effect of AGE and its constituents on
risk factors for atherosclerosis
Atherosclerosis is a complex multifactorial disease
triggered by several risk factors, such as chronic inflammation,
hypercholesteremia and hypertension. The prevention and treatment
of atherosclerosis requires the control of risk factors. In the
following section, the effect of AGE and its key constituents on
these risk factors will be reviewed (Fig. 2).
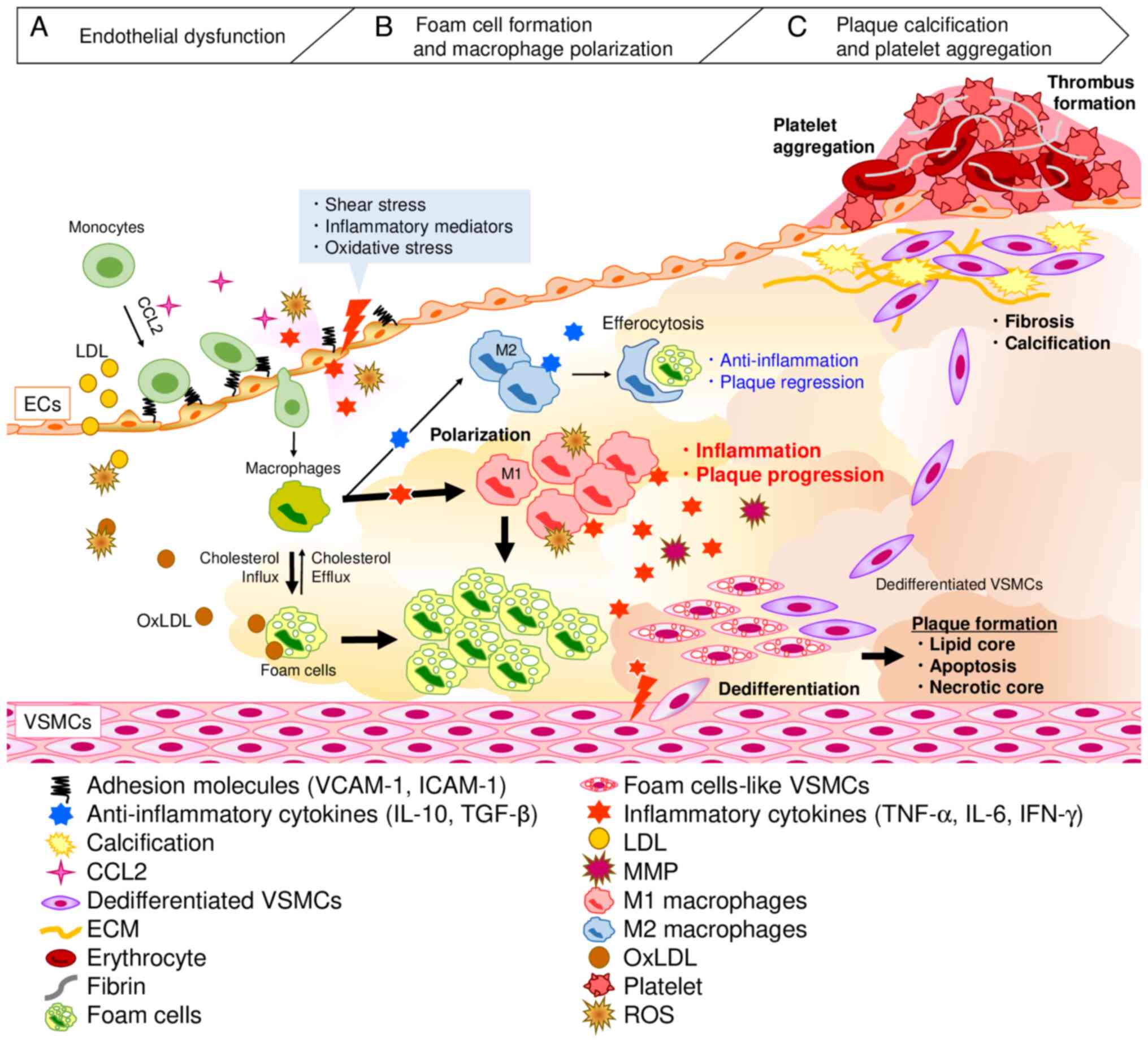 | Figure 2Process of plaque formation. (A)
Endothelial dysfunction. Damage to vascular endothelial cells by
inflammatory mediators and ROS reduces endothelial barrier function
and induces infiltration of circulating monocytes and lipids into
the vascular intima. (B) Foam cell formation and macrophage
polarization. Infiltrating monocytes differentiate into
macrophages, which take up oxLDL via scavenger receptors, including
CD36, to form foam cells. Macrophages polarize into inflammatory M1
macrophages and anti-inflammatory M2 macrophages depending on the
plaque microenvironment. M1 macrophages release inflammatory
factors to exacerbate inflammation, promoting plaque formation,
while M2 macrophages release anti-inflammatory cytokines and
efferocytose foam cells and apoptotic cells, contributing to plaque
regression. (C) Plaque calcification and platelet aggregation.
Dedifferentiated VSMCs, which have been dedifferentiated by
exacerbated inflammation, migrate and proliferate in the intima,
inducing uptake of oxLDL and fibrosis and calcification on the
plaque surface. As the disease progresses and the plaque becomes
unstable, platelets adhere and aggregate at the site of endothelial
cell loss, forming a thrombus that can occlude blood vessels and
induce cardiovascular disease. CCL2, C-C motif chemokine ligand 2;
ECM, extracellular matrix; ECs, endothelial cells; ICAM-1,
intercellular adhesion molecule-1; LDL, low-density lipoprotein;
MMP, matrix metalloproteinase; oxLDL, oxidized LDL; ROS, reactive
oxygen species; VCAM-1, vascular cell adhesion molecule-1; VSMCs,
vascular smooth muscle cells. |
Chronic inflammation
Chronic inflammation associated with obesity and
aging is characterized by persistent and prolonged low-grade
inflammation, often triggered by DAMPs released from dead cells
(41,42). This type of inflammation serves a
crucial role in the initiation and progression of atherosclerosis.
Furthermore, autoimmune diseases such as systemic lupus
erythematosus, antiphospholipid syndrome and rheumatoid arthritis,
which also induce chronic inflammation, are associated with an
increased risk for atherosclerosis. Patients with these autoimmune
diseases face a significantly increased risk of developing new
atherosclerotic plaques, 3.3-50.0 times higher compared with
healthy individuals (43,44). Notably, the production of DAMPs,
which mediate chronic inflammation, is influenced by various
factors, such as smoking, obesity and hyperlipidemia (6). The key DAMPs involved in this process
include the chromatin protein high mobility group box 1, S100
calcium-binding protein A (S100A) 8, S100A9, S100A12 and oxLDL
(5,6). These molecules further perpetuate the
inflammatory response, contributing to the progression of
atherosclerosis. In addition, these DAMPs serve a critical role in
inducing the release of pro-inflammatory cytokines, such as IL-6
and TNF-α. The recognition and binding of DAMPs by pattern
recognition receptors, particularly Toll-like receptor 4 (TLR4) and
receptor for advanced glycation end products, are central to this
inflammatory response. This pathway underscores the complex
interplay between chronic inflammation and the pathophysiological
processes driving atherosclerosis, DAMPs being key mediators in
both the initiation and progression of the disease (7,45,46).
Previous studies have demonstrated that AGE induced
a 20% decrease in IL-6 production in patients with coronary artery
calcium scores <5 and in healthy adults with obesity (47,48).
In addition, in vitro studies have reported that several
AGE-constituents, S1PC, DADS and DATS inhibit IL-6 production
induced by lipopolysaccharide (LPS), a TLR4 agonist (49-52).
Among these components, DADS and DATS suppress TLR4 signaling by
inhibiting NF-κB, whereas S1PC acts through a distinct mechanism.
Specifically, S1PC induces the degradation of the adapter protein
myeloid differentiation primary response 88 by activating autophagy
(51,52). Thus, S1PC has been reported to
exhibit anti-inflammatory effects through a novel mechanism
involving the suppression of TLR4 signaling by activating
autophagy. However, since the mechanism of autophagy activation by
S1PC is not yet fully understood, the anti-inflammatory effects
mediated by this mechanism require further investigation. In
addition, several AGE constituents have been reported to inhibit
autoimmune diseases. For example, DADS prevents cartilage
destruction, ameliorates arthritis and reduces inflammation by
decreasing the expression of pro-inflammatory cytokines in
arthritis rat models (53).
Furthermore, L-arginine improves arthritis and mitigates
inflammatory bone loss by reducing the number of osteoclasts
(54).
These findings suggest that AGE may suppress chronic
inflammation, a key risk factor for atherosclerosis by suppressing
TLR signaling, which is a primary trigger of inflammation,
potentially acting prophylactically to inhibit the progression of
atherosclerosis.
Hypercholesterolemia
Hypercholesterolemia contributes to the development
of atherosclerosis. The risk for atherosclerosis increases by 2-3%
for each 1% rise in the serum cholesterol level (55,56).
Conversely, lowering serum cholesterol by 10% can reduce the risk
for atherosclerosis by 50% in 40 year old men and by 25% in 60 year
old men over a 5 year period according to the results of an
epidemiological survey (55,56).
Hypercholesterolemia enhances the production of reactive oxygen
species (ROS), which promotes the secretion of several
pro-inflammatory cytokines, including IL-1, IL-2, IL-6, IL-8, TNF-α
and IFN-γ, by activating NF-κB (55,57,58).
Supplementation with AGE alone or in combination with B vitamins
has been shown to decrease total cholesterol (TC) level by ~7% in
clinical studies (36,59-61)
and ~15% in animal studies (40,59).
AGE has also been shown to inhibit cholesterol synthesis in rat
hepatocytes, thus it is suggested that the cooperative action of
several components of AGE, such as SAC, SPC, SEC, γ-glutamyl SAC
and γ-glutamyl SPC, may contribute to its effect (59). These studies suggest that AGE
decreases TC, which may contribute to the prevention of
atherosclerosis.
Hypertension
Hypertension has been shown to significantly elevate
the risk for developing atherosclerosis in clinical trials
(62). According to
epidemiological research, arterial hypertension was identified as
the most crucial cardiovascular risk factor, contributing to 48% of
all strokes and 18% of all coronary events (62). A randomized trial with 3,845
participants, averaging 83 years in age, demonstrated that reducing
blood pressure from 161/84 to 144/78 mmHg decreased the risk for
cerebral circulatory disorders by 30% and cardiovascular events by
23% (62,63). Additionally, in patients with
vascular disease or diabetes mellitus plus an additional
cardiovascular risk factor, treatment with ramipril, an
angiotensin-converting enzyme inhibitor, resulted in a 22%
reduction in the composite endpoint of cardiovascular death,
myocardial infarction and stroke (64). Therefore, antihypertensive therapy
is important for the prevention or improvement of atherosclerosis.
AGE has been reported to improve not only atherosclerosis but also
hypertension in several clinical trials. The AGE-treated group
demonstrated a decreased mean systolic blood pressure (SBP) by ~10
mm Hg after 12 weeks of administration (65-67).
In addition, it has been reported that active components of AGE,
including S1PC and SAC, reduce blood pressure. The repeated
administration of S1PC for 10 weeks significantly decreases SBP of
spontaneously hypertensive rats (68). Furthermore, it was reported that
the single administration of S1PC reduces SBP after 3 h via the
central histamine H3 receptor by altering histidine metabolism
(69,70). Moreover, administration of SAC has
been shown to decrease SBP in both ovariectomized and five-sixths
of a group of nephrectomized rats (71,72).
These results suggested that AGE may help prevent atherosclerosis
by mitigating hypertension, which is a risk factor for the
development of atherosclerosis.
Since AGE contains multiple bioactive components
that act on various targets through different mechanisms, they have
the potential to simultaneously improve major risk factors for
atherosclerosis, such as chronic inflammation, dyslipidemia and
hypertension. By acting on these factors, AGE may help prevent the
progression of atherosclerosis.
3. Effect of AGE and its components on
vascular endothelial function
Vascular endothelial cells, located on the innermost
layer of blood and lymphatic vessels, serve crucial roles in
delivering oxygen and nutrients, regulating blood flow, modulating
immune cell trafficking and maintaining tissue homeostasis
(73). C-reactive protein (CRP),
induced by inflammation, can cause endothelial dysfunction by
directly damaging endothelial cells and reducing the number and
function of endothelial progenitor cells (43,44,74).
Endothelial injury triggers ROS production and vascular
inflammation, increasing the expression levels of intercellular
adhesion molecule-1 (ICAM-1) and vascular cell adhesion molecule-1
(VCAM-1) on the cell surface and the secretion of C-C motif
chemokine ligand 2 (CCL2) (75).
This disrupts tight junctions, which normally restrict the entry of
circulating substances and immune cells from the bloodstream into
the vessel wall, allowing monocytes to infiltrate the vessel wall
(76). Non-inflammatory
Ly6Clow monocytes typically patrol the vasculature to
phagocytose and scavenge debris and maintain endothelial integrity.
By contrast, the number of inflammatory Ly6Chi monocytes
increases during chronic inflammation and hypercholesterolemia,
preferentially adhere to activated endothelium, infiltrate the
vessel wall and differentiate into lesional macrophages (77-80).
This infiltration of Ly6Chi monocytes
corresponds to the early stages of atherosclerosis development,
with Ly6Chi monocytes eventually transforming into foam
cells that accumulate in blood vessels (Fig. 2A) (76-79).
The following mechanisms of actions discussed relate to the effects
of AGE and its components in protecting vascular endothelial
function.
Antioxidative effect on vascular
endothelial cells
AGE, SAC and Fru-Arg have been reported to inhibit
the production of H2O2 and lipid peroxides
induced by oxLDL through ROS. These compounds promote the nuclear
accumulation of nuclear factor erythroid 2-related factor 2 (Nrf2),
which is a transcription factor activated in response to oxidation,
and increase the gene and protein expression level of the
antioxidant enzymes heme oxygenase 1 (HO-1) and glutamate-cysteine
ligase modifier subunit (GCLM) in human umbilical vein endothelial
cells (HUVECs), thereby improving endothelial dysfunction. This
change is accompanied by an increase in the intracellular level of
the antioxidant glutathione, suggesting that AGE and its
constituents exert antioxidant activity to prevent oxLDL-induced
oxidation and cellular damage (81-86).
It has also been shown that S1PC enhances activation of the Nrf2
pathway in the presence of a nitric oxide (NO) donor by promoting
the degradation of broad complex, tramtrack and bric-a-brac domain
and cap'n'collar homology 1, a transcriptional repressor of Nrf2
(87,88). Additionally, SAC has been shown to
activate endothelial nitric oxide synthase (eNOS) in endothelial
cells and promote NO production (89,90).
These results suggest that AGE and its constituents may act
together to increase the cellular antioxidant capacity through
enhancement of the Nrf2 pathway and ameliorate the vascular
endothelial cell dysfunction and exacerbated inflammation caused by
oxidation (Fig. 3A).
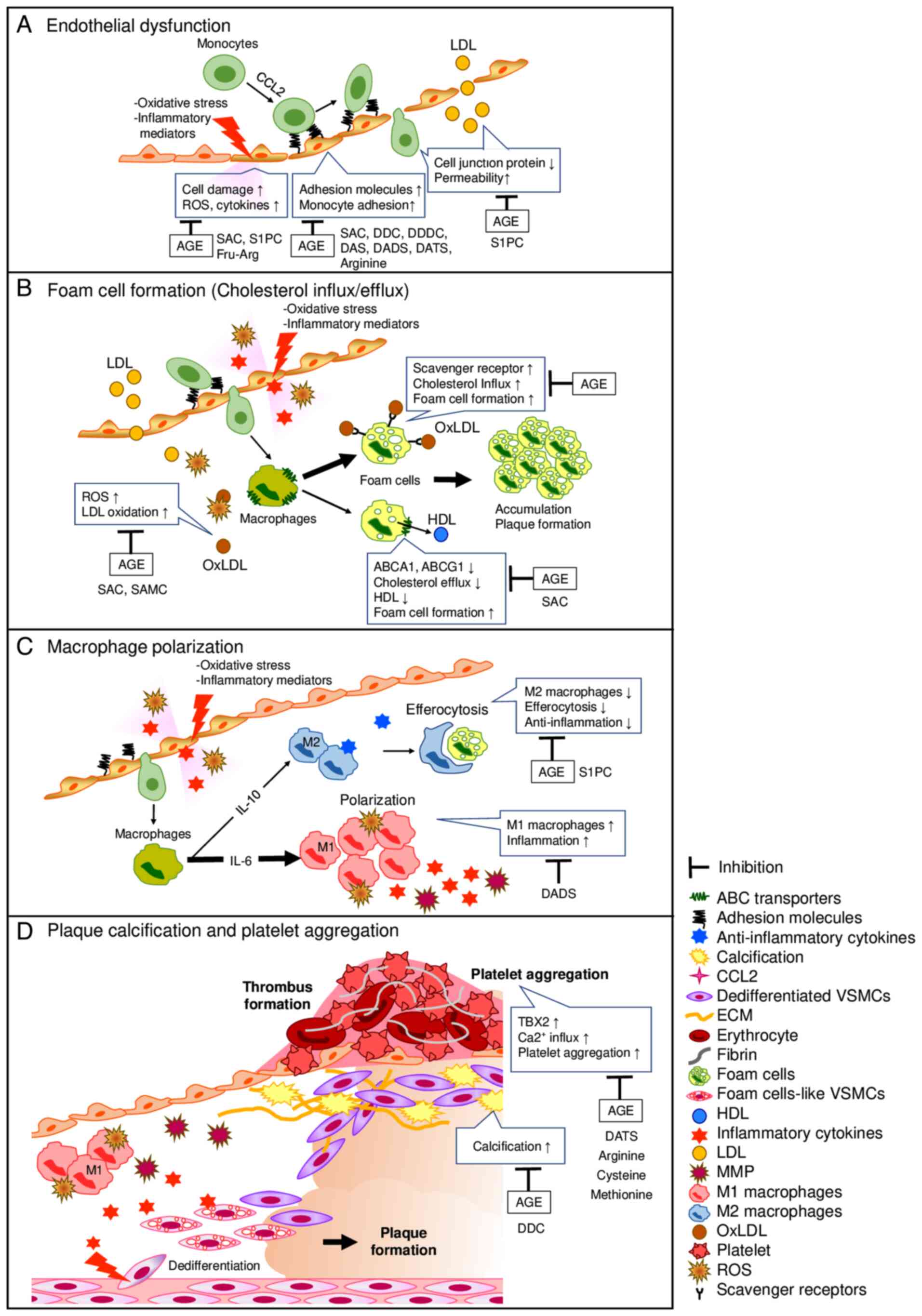 | Figure 3Effect of AGE and its constituents in
the processes of plaque formation. The inhibitory effects of AGE
and their constituents on the plaque formation processes. (A)
Endothelial dysfunction, (B) foam cell formation (macrophage
cholesterol influx/efflux), (C) macrophage polarization and (D)
plaque calcification and platelet aggregation. ABCA1, ATP-binding
cassette transporter A1; ABCG1, ATP-binding cassette transporter
G1; AGE, aged garlic extract; CCL2, C-C motif chemokine ligand 2;
DADS, diallyl disulfide; DAS, diallyl sulfide; DATS, diallyl
trisulfide; DDC, dehydrodiconiferyl alcohol; DDDC,
dihydrodehydrodiconiferyl alcohol; ECM, extracellular matrix;
Fru-Arg, fructosyl-L-arginine; HDL, high-density lipoprotein; IL,
interleukin; LDL, low-density lipoprotein; MMP, matrix
metalloproteinase; oxLDL, oxidized LDL; ROS, reactive oxygen
species; SAC, S-allylcysteine; SAMC,
S-allylmercaptocysteine; S1PC, S-1-propenylcysteine;
TBX2, thromboxane B2; VSMCs, vascular smooth muscle
cells. |
Monocyte adhesion
DDC and DDDC, identified as antioxidants in AGE,
have been shown to suppress VCAM-1 expression induced by LPS or
advanced glycation end products in HUVECs by inhibiting the
JNK/c-Jun pathway, but not the NF-κB pathway, thereby preventing
the adhesion of THP-1 monocytes to the surface of HUVECs (91). Similarly, DAS, DADS and DATS, which
are minor constituents of AGE (18), suppress oxLDL-induced VCAM-1 and
E-selectin expression levels on the cell surface, reducing the
adhesion of the human promyelocytic leukemia cell line HL-60 to
HUVECs (92). Notably, the
inhibitory potency of these sulfur-containing compounds increases
with the number of sulfur atoms, in the order of DATS > DADS
> DAS (92). Their mechanisms
involve activation of the PI3K/protein kinase (PK) B signaling
pathway to suppress E-selectin expression and dephosphorylation of
PKA and cAMP response element binding protein to reduce VCAM-1
expression levels, each of which is mediated through the PKB/PI3K
signaling pathway (92).
Additionally, AGE and S1PC inhibit the secretion of CCL2, a
chemokine that attracts monocytes (51,93).
In addition, L-arginine, a major amino acid in AGE, suppresses
IL-1β-induced VCAM-1 and ICAM-1 expression, inhibiting the adhesion
of human peripheral blood-derived monocytes to HUVECs (94). L-arginine also exhibits
anti-atherosclerotic effects, such as reducing lipid deposition in
the aorta, improving flow-dependent vasodilation and preventing
monocyte adhesion to the vascular surface in a hypercholesterolemic
rabbit atherosclerosis model (95,96).
These findings suggest that multiple constituents in
AGE suppress the adhesion of monocytes to vascular endothelial
cells by not only reducing the expression of adhesion molecules on
endothelial cells but also inhibiting the secretion of chemokines,
such as CCL2, that attract monocytes (Fig. 3A).
Endothelial barrier function
Vascular endothelial cells adhere to each other
through adherens junctions including vascular endothelial
(VE)-cadherin, and tight junctions including claudin, occludin and
zonula occludens-1 (ZO-1). These junctions restrict the entry of
circulating substances and immune cells from the blood into the
vessel wall (13). Disruption of
this barrier function by inflammation and oxidation leads to the
entry of lipids and circulating immune cells into the intima,
increasing lipid deposition and vascular inflammation in the aorta
(12,13). Kunimura et al (97) reported that AGE and S1PC, but not
SAC and SAMC, inhibited cell permeability by suppressing
TNF-α-induced downregulation of VE-cadherin, claudin-5 and ZO-1
through the suppression of the Rho guanine nucleotide exchange
factor-H1/RhoA/Rac pathway in HUVECs. These results suggest that
AGE prevents the disruption of tight junctions caused by
inflammation and maintains the integrity of intercellular adhesion
(Fig. 3A).
In summary, AGE and its components maintain vascular
endothelial barrier function and prevent monocyte adhesion and
infiltration, which may consequently inhibit atherosclerotic plaque
development (Fig. 3A).
4. Effect of AGE and its constituents on
foam cell formation
LDL in blood enters the vascular intima through the
gaps between endothelial cells, where it is oxidized by ROS derived
from vascular endothelial cells and macrophages, and is
subsequently deposited in the vascular intima as oxLDL (2). Macrophages take up oxLDL via
scavenger receptors (SRs) such as SR-AI, CD36 or lectin-like
oxidized low-density lipoprotein receptor 1 (LOX-1), and the
excessive uptake of oxLDL transforms macrophages into foam cells,
resulting in the development of an atherosclerotic plaque (Fig. 2B) (14-16).
Atherosclerotic lesions were reduced in an atherosclerosis model of
mice lacking theses SRs (98-100).
Thus, the suppressed expression of SRs in macrophages may
contribute to the inhibition of foam cell formation and plaque
development. It has been reported that AGE suppresses peroxisome
proliferator-activated receptor γ-mediated CD36 expression and
inhibits the intracellular uptake of oxLDL in macrophages derived
from the human monocyte THP-1 cell line (101-103).
On the other hands, macrophages also efflux free cholesterol (FC)
converted from oxLDL via ABC transporters, such as ABCA1 and ABCG1,
which are necessary for high-density lipoprotein (HDL) synthesis.
This FC is subsequently incorporated into HDL and transported back
to the liver (15,104). In clinical trials and animal
experiments, AGE increased plasma HDL-cholesterol concentration,
suggesting that it may improve cholesterol metabolism (36,40,59-61).
In addition, SAC increased ABCA1 gene and protein expression levels
in THP-1-derived macrophages, which may improve cholesterol
metabolism (105). It has been
reported that depletion of macrophage-specific ABCA1 and ABCG1
exacerbates plaque formation (106-108),
whereas overexpression of ABCA1 is protective against
atherosclerosis (109).
Thus, AGE may reduce cholesterol accumulation in
macrophages by suppressing the expression of SRs and increasing the
expression of ABC transporters. These results also suggest that AGE
inhibits the progression of atherosclerotic lesions by reducing
foam cell formation (Fig. 3B).
5. Effect of AGE and its constituents on
macrophage polarization
Macrophages polarize into two major phenotypes:
Inflammatory M1 macrophages and anti-inflammatory M2 macrophages,
depending on the arterial plaque microenvironment (110). In plaque lesions, M2 macrophages
are more dominant compared with M1 macrophages from the early
stages of plaque formation until plaque stabilization. They
contribute to plaque stabilization through the efflux of
cholesterol, production of anti-inflammatory cytokines,
phagocytosis of apoptotic cells and collagen production induced by
TGF-β, thereby regulating plaque progression (110). However, as plaque formation
progresses and a lipid and necrotic core is formed, M1 macrophages
become dominant. This shift leads to the production of inflammatory
cytokines, increased lipid accumulation due to decreased
cholesterol efflux and thinning of the cap caused by the production
of matrix metalloproteinases, resulting in an unstable plaque and
an increased risk of rupture (Fig.
2B) (111-113).
Crocin, an active ingredient of Crocus sativus L., and
pomegranate juice increased the number of M2 macrophages and
inhibited the progression of aortic plaque formation in
atherosclerotic mice (114,115). Therefore, it is important to
maintain the predominance of M2 macrophages over M1 macrophages to
inhibit plaque progression or to stabilize plaques (110,116).
AGE has been reported to decrease the expression
levels of M1 macrophage markers and increase the expression levels
of M2 macrophage markers in aortic and splenic lymphocytes. The
active component responsible for this effect of AGE is S1PC, which
has been shown in vitro to prolong IL-10-mediated STAT3
activation, thereby promoting polarization into M2c macrophages
with a high IL-10 production capacity (38). Additionally, DADS, another active
component of AGE, reduces the number of M1 macrophages by
suppressing LPS-induced NF-κB activation through the Nrf2 pathway,
thereby inhibiting polarization into M1 macrophages and reducing
inflammation (117).
However, since macrophage polarization is influenced
by the microenvironment, it is crucial to investigate whether AGE
or its components can induce M2 macrophage polarization within the
context of a chronic inflammatory environment, such as that found
in atherosclerotic plaques. Notably, clinical trials have
demonstrated that drugs such as pioglitazone and
thiazolidinediones, which promote polarization towards M2
macrophages, significantly suppressed atherosclerosis in patients
with type 2 diabetes (116,118-120).
Consequently, therapeutic strategies targeting M2 macrophage
polarization have emerged as promising avenues for atherosclerosis
treatment (116). These clinical
observations suggest that AGE and its components might contribute
to atheroprotection by promoting a shift towards the M2 macrophage
phenotype (Fig. 3C). However, the
polarization of macrophages within atherosclerotic plaques is a
complex process involving various subpopulations. Further research
is needed to pinpoint the specific macrophage subsets affected by
AGE and to elucidate the underlying molecular mechanisms that drive
these changes.
6. Effect of AGE and its constituents on
vascular calcification and platelet aggregation
Vascular calcification
Vascular smooth muscle cells (VSMCs) are typically
present in the vascular media and are involved in vascular
contraction and relaxation. However, they dedifferentiate and
migrate into the vascular intima in response to cytokines, growth
factors released from damaged vascular endothelial cells and
activated macrophages (121,122). Migrated VSMCs proliferate in the
vascular intima and change to various phenotypes, such as foam
cell-like and osteoblast-like cells, and are involved in the
development of plaque formation (121,122). In calcified plaque lesions, VSMCs
upregulate the expression of osteogenesis-related factors including
alkaline phosphatase (ALP), runt-related transcription factor 2,
osteopontin and bone morphogenetic protein 2, suggesting that the
proliferation of osteoblast-like VSMCs contributes to plaque
calcification (Fig. 2C) (122).
Several clinical trials have reported that AGE
administered for 1 year improved the coronary artery calcification
scores of patients with coronary artery disease (27,29,30,123), suggesting that AGE may regulate
the activation of osteoblast-like VSMCs. In addition, DDC has been
shown to significantly inhibit ALP activity induced by culturing
human coronary artery smooth muscle cells with dexamethasone and
the culture supernatant of THP-1 derived macrophages (124). These findings suggest that AGE
and DDC may inhibit plaque calcification by suppressing the
osteogenic differentiation of VSMCs (Fig. 3D) (27,29,30,123,124).
Platelet aggregation
Plaque rupture and subsequent thrombus formation
trigger arterial stenosis and disrupt blood flow in the late stage
of atherosclerosis. Platelets bind to von Willebrand factor (VWF)
present in the exposed subendothelial collagen layer due to plaque
rupture via the glycoprotein (GP) complexes. Platelets adhering to
subendothelial tissue release ADP, thrombin and thromboxane A2,
promoting platelet activation. Activated platelets bind to VWF and
fibrinogen via GP complex (GPIIb/IIIa) to form platelet aggregates,
which become thrombi (125,126). The thrombus is stabilized by
fibrin, which is produced from fibrinogen, and promotes the blood
clotting reaction on the platelet membrane (125-127).
This thrombus can cause stenosis, occlusion or dissection of blood
vessels (Fig. 2C).
Steiner and Li (128) reported that in the blood of
patients with moderate hypercholesterolemia taking AGE for 6 weeks,
collagen-, epinephrine- and ADP-induced platelet aggregation was
reduced, and platelet adhesion to collagen, VWF and fibrinogen was
inhibited. Additionally, ADP-induced platelet aggregation was
suppressed in the blood of normolipidemic subjects who took AGE for
13 weeks (129). Although AGE
inhibits platelet aggregation, no serious adverse events have been
reported when AGE is used in combination with the anticoagulant
warfarin, indicating it may be relatively safe to use (130). Furthermore, serum from
apolipoprotein E-knockout mice, whose atherosclerosis improved
after 12 weeks of AGE feeding, showed a significant decrease in the
concentration of thromboxane B2, a marker of platelet activation,
suggesting that AGE may inhibit platelet aggregation by suppressing
platelet activation (39).
Platelets are activated through MAPK kinase by several agonists,
such as ADP and collagen (131-133).
It has been reported that platelets from rats treated with AGE for
2 weeks exhibit inhibited collagen-induced platelet aggregation and
suppressed the phosphorylation of ERK, p38 and JNK (134). In addition, studies on human
platelets have suggested that AGE may inhibit platelet activation
by reducing ADP-induced Ca2+ influx into the cell,
thereby inhibiting GPIIb/IIIa activation (135-137).
It has been reported that L-arginine, L-cysteine and L-methionine,
which are components of AGE, inhibit ADP-induced platelet
aggregation, while DATS inhibits collagen- and thrombin-induced
platelet aggregation (19,138). Therefore, it is possible that AGE
may inhibit atherothrombosis by suppressing platelet aggregation
through the collaborative action of its multiple pharmacologically
active components (Fig. 3D).
These findings suggest that AGE may prevent plaque
rupture by inhibiting calcification of advanced atherosclerotic
lesions. Additionally, if plaques do rupture, AGE may inhibit
platelet aggregation, thereby reducing the risk of subsequent
myocardial infarction and angina pectoris (Fig. 3D).
7. Future directions and limitations of
current assessments
Establishing the causal relationship between AGE
intake and its diverse pharmacological effects in clinical trials
has inherent limitations due to the complexity of its mechanisms of
action and various confounding factors. Furthermore, the underlying
mechanisms driving AGE's broad biological activities remain largely
elusive, in part due to the presence of numerous bioactive
compounds. Developing robust targeting and screening systems is
essential for identifying these active constituents and their
specific roles. A comprehensive understanding of AGE's multifaceted
biological effects is therefore imperative, and OMICS-based
approaches, including proteomics and miRNA analysis using clinical,
animal and cell samples, could facilitate this process by
identifying key molecular targets and regulatory pathways (139-144).
In clinical studies conducted to date, the effect of
AGE has been evaluated for up to 1 year; however, its effects on
the onset and progression of atherosclerosis are expected to become
clearer with longer-term follow-up studies. Therefore, large,
randomized, double-blind clinical trials with long-term treatment
and follow-up periods are needed to assess the effects of AGE on
the development and progression of atherosclerosis, as well as
their impact on the clinical outcomes of patients with
atherosclerosis. Additionally, the dosage of AGE in clinical trials
for atherosclerosis treatment varies between 1,000-2,400 mg/day,
depending on the trial (27,29,30,123). Determining the optimal dosage for
each target disease through dose-response testing and similar
assessments remains a critical challenge.
To advance AGE research, it is essential to
comprehensively characterize its bioactive components, their
kinetics and their precise mechanisms of action. Future
investigations should also focus on identifying key active
constituents, elucidating their molecular targets and exploring
AGE's potential synergistic effects with existing drugs, its
applicability to clinical trials and its relevance beyond
atherosclerosis.
8. Conclusion
The multifactorial mechanism involving endothelial
dysfunction, oxLDL accumulation, macrophage-induced inflammation
and other risk factors in the development of atherosclerosis makes
it difficult to prevent and treat the disease with a single target
or mechanism. In this context, AGE has potential therapeutic and
preventive applications, as it contains multiple active components
and causes few side effects. AGE and its components exhibit diverse
mechanisms of action that affect various aspects of disease
progression, such as reducing risk factors like chronic
inflammation, hyperlipidemia and hypertension, suppressing
endothelial dysfunction, reducing oxLDL formation and increasing in
HDL levels, reducing foam cell formation and promoting M2
macrophage polarization and suppressing platelet aggregation.
These effects of AGE can be attributed to its
pharmacologically active sulfur-containing components, which have
demonstrated inhibitory actions on various stages of
atherosclerosis progression. By acting on multiple pathways
simultaneously, AGE exhibits a unique multi-target approach to
atherosclerosis prevention and treatment, with its constituents
acting synergistically (Fig. 3).
Notably, AGE has also been shown to delay coronary artery
calcification in patients undergoing statin therapy without
affecting side effects, suggesting its potential to complement the
effects of existing atherosclerosis drugs.
Despite promising preclinical and clinical evidence,
the intricate mechanisms underlying the diverse biological
activities of AGE remain largely elusive. Contributing to this
complexity is the presence of a wide array of bioactive compounds
within AGE. Developing robust targeting and screening systems is
crucial for identifying these active constituents. To this end, a
comprehensive understanding of the multifaceted biological effects
of AGE is imperative. Future research should utilize chemical
proteomics and network pharmacology approaches to identify active
constituents, target molecules and mechanisms of action, thereby
elucidating the effects of AGE. A comprehensive characterization of
AGE is essential, and these findings should be leveraged to explore
its potential synergistic effects with existing drugs, its
applicability to clinical trials and its relevance beyond
atherosclerosis.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Author's contributions
SM and JIS conceived this review. SM, MT and JIS
analyzed the relevant literature. SM and JIS wrote the manuscript.
SM constructed the figures. MT and JIS critically revised the
manuscript. All authors read and approved the final version of the
manuscript. Data authentication is not available.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Mani V, Durmus C, Khushaim W, Ferreira DC,
Timur S, Arduini F and Salama KN: Multiplexed sensing techniques
for cardiovascular disease biomarkers-A review. Biosens
Bioelectron. 216(114680)2022.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Khatana C, Saini NK, Chakrabarti S, Saini
V, Sharma A, Saini RV and Saini AK: Mechanistic insights into the
oxidized low-density lipoprotein-induced atherosclerosis. Oxid Med
Cell Longev. 2020(5245308)2020.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Heo KS, Fujiwara K and Abe JI: Shear
stress and atherosclerosis. Mol Cells. 37:435–440. 2014.PubMed/NCBI View Article : Google Scholar
|
|
4
|
J M, L N, A V, E C, V L, K BS, L L, T G, M
P and P M: Wall shear stress alteration: A local risk factor of
atherosclerosis. Curr Atheroscler Rep. 24:143–151. 2022.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Singh P and Ali SA: Multifunctional role
of S100 protein family in the immune system: An update. Cells.
11(2274)2022.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Schiopu A and Cotoi OS: S100A8 and S100A9:
DAMPs at the crossroads between innate immunity, traditional risk
factors, and cardiovascular disease. Mediators Inflamm.
2013(828354)2013.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Averill MM, Kerkhoff C and Bornfeldt KE:
S100A8 and S100A9 in cardiovascular biology and disease.
Arterioscler Thromb Vasc Biol. 32:223–229. 2012.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Tietge UJF: Hyperlipidemia and
cardiovascular disease: Inflammation, dyslipidemia, and
atherosclerosis. Curr Opin Lipidol. 25:94–95. 2014.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Ross R and Harker L: Hyperlipidemia and
atherosclerosis. Science. 193:1094–1100. 1976.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Bornfeldt KE and Tabas I: Insulin
resistance, hyperglycemia, and atherosclerosis. Cell Metab.
14:575–585. 2011.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Aronson D and Rayfield EJ: How
hyperglycemia promotes atherosclerosis: Molecular mechanisms.
Cardiovasc Diabetol. 1(1)2002.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Zhang L and Lei S: Changes of junctions of
endothelial cells in coronary sclerosis: A review. Chronic Dis
Transl Med. 2:22–26. 2016.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Chistiakov DA, Orekhov AN and Bobryshev
YV: Endothelial barrier and its abnormalities in cardiovascular
disease. Front Physiol. 6(365)2015.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Chistiakov DA, Bobryshev YV and Orekhov
AN: Macrophage-mediated cholesterol handling in atherosclerosis. J
Cell Mol Med. 20:17–28. 2016.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Chistiakov DA, Melnichenko AA, Myasoedova
VA, Grechko AV and Orekhov AN: Mechanisms of foam cell formation in
atherosclerosis. J Mol Med (Berl). 95:1153–1165. 2017.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Falk E: Pathogenesis of atherosclerosis. J
Am Coll Cardiol. 47 (8 Suppl):C7–C12. 2006.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Rahman K: Garlic and aging: New insights
into an old remedy. Ageing Res Rev. 2:39–56. 2003.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Kodera Y, Kurita M, Nakamoto M and
Matsutomo T: Chemistry of aged garlic: Diversity of constituents in
aged garlic extract and their production mechanisms via the
combination of chemical and enzymatic reactions. Exp Ther Med.
19:1574–1584. 2020.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Allison GL, Lowe GM and Rahman K: Aged
garlic extract and its constituents inhibit platelet aggregation
through multiple mechanisms. J Nutr. 136 (3 Suppl):782S–788S.
2006.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Hoshino T, Kashimoto N and Kasuga S:
Effects of garlic preparations on the gastrointestinal mucosa. J
Nutr. 131 (3S):1109S–1113S. 2001.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Sumiyoshi H, Kanezawa A, Masamoto K,
Harada H, Nakagami S, Yokota A, Nishikawa M and Nakagawa S: Chronic
toxicity test of garlic extract in rats. J Toxicol Sci. 9:61–75.
1984.PubMed/NCBI View Article : Google Scholar : (In Japanese).
|
|
22
|
Efendy JL, Simmons DL, Campbell GR and
Campbell JH: The effect of the aged garlic extract, ‘Kyolic’, on
the development of experimental atherosclerosis. Atherosclerosis.
132:37–42. 1997.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Weiss N, Ide N, Abahji T, Nill L, Keller C
and Hoffmann U: Aged garlic extract improves homocysteine-induced
endothelial dysfunction in macro- and microcirculation. J Nutr. 136
(3 Suppl):750S–754S. 2006.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Shaikh K, Kinninger A, Cherukuri L,
Birudaraju D, Nakanishi R, Almeida S, Jayawardena E, Shekar C,
Flores F, Hamal S, et al: Aged garlic extract reduces low
attenuation plaque in coronary arteries of patients with diabetes:
A randomized, double-blind, placebo-controlled study. Exp Ther Med.
19:1457–1461. 2020.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Gómez-Arbeláez D, Lahera V, Oubiña P,
Valero-Muñoz M, de Las Heras N, Rodríguez Y, García RG, Camacho PA
and López-Jaramillo P: Aged garlic extract improves adiponectin
levels in subjects with metabolic syndrome: A double-blind,
placebo-controlled, randomized, crossover study. Mediators Inflamm.
2013(285795)2013.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Matsumoto S, Nakanishi R, Li D, Alani A,
Rezaeian P, Prabhu S, Abraham J, Fahmy MA, Dailing C, Flores F, et
al: Aged garlic extract reduces low attenuation plaque in coronary
arteries of patients with metabolic syndrome in a prospective
randomized double-blind study. J Nutr. 146:427S–432S.
2016.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Wlosinska M, Nilsson AC, Hlebowicz J,
Hauggaard A, Kjellin M, Fakhro M and Lindstedt S: The effect of
aged garlic extract on the atherosclerotic process-a randomized
double-blind placebo-controlled trial. BMC Complement Med Ther.
20(132)2020.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Williams MJA, Sutherland WHF, McCormick
MP, Yeoman DJ and de Jong SA: Aged garlic extract improves
endothelial function in men with coronary artery disease. Phytother
Res. 19:314–319. 2005.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Budoff M: Aged garlic extract retards
progression of coronary artery calcification. J Nutr. 136 (3
Suppl):741S–744S. 2006.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Hom C, Budoff M and Luo Y: The effects of
aged garlic extract on coronary artery calcification progression
and blood pressure. J Am Coll Cardiol. 65(A1472)2015.
|
|
31
|
Budoff MJ, Takasu J, Flores FR, Niihara Y,
Lu B, Lau BH, Rosen RT and Amagase H: Inhibiting progression of
coronary calcification using aged garlic extract in patients
receiving statin therapy: A preliminary study. Prev Med.
39:985–991. 2004.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Zeb I, Ahmadi N, Flores F and Budoff MJ:
Randomized trial evaluating the effect of aged garlic extract with
supplements versus placebo on adipose tissue surrogates for
coronary atherosclerosis progression. Coron Artery Dis. 29:325–328.
2018.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Ahmadi N, Nabavi V, Hajsadeghi F, Zeb I,
Flores F, Ebrahimi R and Budoff M: Aged garlic extract with
supplement is associated with increase in brown adipose, decrease
in white adipose tissue and predict lack of progression in coronary
atherosclerosis. Int J Cardiol. 168:2310–2314. 2013.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Larijani VN, Ahmadi N, Zeb I, Khan F,
Flores F and Budoff M: Beneficial effects of aged garlic extract
and coenzyme Q10 on vascular elasticity and endothelial function:
The FAITH randomized clinical trial. Nutrition. 29:71–75.
2013.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Zeb I, Ahmadi N, Nasir K, Kadakia J,
Larijani VN, Flores F, Li D and Budoff MJ: Aged garlic extract and
coenzyme Q10 have favorable effect on inflammatory markers and
coronary atherosclerosis progression: A randomized clinical trial.
J Cardiovasc Dis Res. 3:185–190. 2012.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Budoff MJ, Ahmadi N, Gul KM, Liu ST,
Flores FR, Tiano J, Takasu J, Miller E and Tsimikas S: Aged garlic
extract supplemented with B vitamins, folic acid and L-arginine
retards the progression of subclinical atherosclerosis: A
randomized clinical trial. Prev Med. 49:101–107. 2009.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Yeh YY and Yeh SM: Homocysteine-lowering
action is another potential cardiovascular protective factor of
aged garlic extract. J Nutr. 136 (3 Suppl):745S–749S.
2006.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Miki S, Suzuki JI, Takashima M, Ishida M,
Kokubo H and Yoshizumi M: S-1-Propenylcysteine promotes
IL-10-induced M2c macrophage polarization through prolonged
activation of IL-10R/STAT3 signaling. Sci Rep.
11(22469)2021.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Morihara N, Hino A, Miki S, Takashima M
and Suzuki JI: Aged garlic extract suppresses inflammation in
apolipoprotein E-knockout mice. Mol Nutr Food Res.
61(1700308)2017.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Morihara N, Hino A, Yamaguchi T and Suzuki
JI: Aged garlic extract suppresses the development of
atherosclerosis in apolipoprotein E-knockout mice. J Nutr.
146:460S–463S. 2016.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Li X, Li C, Zhang W, Wang Y, Qian P and
Huang H: Inflammation and aging: Signaling pathways and
intervention therapies. Signal Transduct Target Ther.
8(239)2023.PubMed/NCBI View Article : Google Scholar
|
|
42
|
He Y, Chen Y, Yao L, Wang J, Sha X and
Wang Y: The inflamm-aging model identifies key risk factors in
atherosclerosis. Front Genet. 13(865827)2022.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Raj R, Thomas S and Gorantla V:
Accelerated atherosclerosis in rheumatoid arthritis: A systematic
review. F1000Res. 11(466)2022.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Adawi M, Firas S and Blum A: Rheumatoid
Arthritis and Atherosclerosis. IMAJ. 21:460–463. 2019.PubMed/NCBI
|
|
45
|
Sreejit G, Abdel Latif A, Murphy AJ and
Nagareddy PR: Emerging roles of neutrophil-borne S100A8/A9 in
cardiovascular inflammation. Pharmacol Res.
161(105212)2020.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Roh JS and Sohn DH: Damage-associated
molecular patterns in inflammatory diseases. Immune Netw.
18(e27)2018.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Wlosinska M, Nilsson AC, Hlebowicz J,
Fakhro M, Malmsjö M and Lindstedt S: Aged garlic extract reduces
IL-6: A double-blind placebo-controlled trial in females with a low
risk of cardiovascular disease. Evid Based Complement Alternat Med.
2021(6636875)2021.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Xu C, Mathews AE, Rodrigues C, Eudy BJ,
Rowe CA, O'Donoughue A and Percival SS: Aged garlic extract
supplementation modifies inflammation and immunity of adults with
obesity: A randomized, double-blind, placebo-controlled clinical
trial. Clin Nutr ESPEN. 24:148–155. 2018.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Lee HH, Han MH, Hwang HJ, Kim GY, Moon SK,
Hyun JW, Kim WJ and Choi YH: Diallyl trisulfide exerts
anti-inflammatory effects in lipopolysaccharide-stimulated RAW
264.7 macrophages by suppressing the Toll-like receptor 4/nuclear
factor-κB pathway. Int J Mol Med. 35:487–495. 2015.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Lee HH, Jeong JW, Hong SH, Park C, Kim BW
and Choi YH: Diallyl trisulfide suppresses the production of
lipopolysaccharide-induced inflammatory mediators in BV2 microglia
by decreasing the NF-κB pathway activity associated with toll-like
Receptor 4 and CXCL12/CXCR4 pathway blockade. J Cancer Prev.
23:134–140. 2018.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Suzuki JI, Kodera Y, Miki S, Ushijima M,
Takashima M, Matsutomo T and Morihara N: Anti-inflammatory action
of cysteine derivative S-1-propenylcysteine by inducing MyD88
degradation. Sci Rep. 8(14148)2018.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Suzuki J, Miki S, Ushijima M and Kodera Y:
Regulation of immune response by S-1-propenylcysteine through
autophagy-mediated protein degradation. Exp Ther Med. 19:1570–1573.
2020.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Chen Y, Xue R, Jin X and Tan X:
Antiarthritic activity of diallyl disulfide against freund's
adjuvant-induced arthritic rat model. J Environ Pathol Toxicol
Oncol. 37:291–303. 2018.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Cao S, Li Y, Song R, Meng X, Fuchs M,
Liang C, Kachler K, Meng X, Wen J, Schlötzer-Schrehardt U, et al:
L-arginine metabolism inhibits arthritis and inflammatory bone
loss. Ann Rheum Dis. 83:72–87. 2024.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Prasad K and Mishra M: Mechanism of
hypercholesterolemia-induced atherosclerosis. Rev Cardiovasc Med.
23(212)2022.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Davis CE, Rifkind BM, Brenner H and Gordon
DJ: A single cholesterol measurement underestimates the risk of
coronary heart disease. An empirical example from the lipid
research clinics mortality follow-up study. JAMA. 264:3044–3046.
1990.PubMed/NCBI
|
|
57
|
Minatel IO, Francisqueti FV, Corrêa CR and
Pereira Lima GPP: Antioxidant activity of Ƴ-oryzanol: A complex
network of interactions. Int J Mol Sci. 17(1107)2016.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Ranneh Y, Ali F, Akim AM, Hamid HA,
Khazaai H and Fadel A: Crosstalk between reactive oxygen species
and pro-inflammatory markers in developing various chronic
diseases: A review. Appl Biol Chem. 60:327–338. 2017.
|
|
59
|
Yeh YY and Liu L: Cholesterol-lowering
effect of garlic extracts and organosulfur compounds: Human and
animal studies. J Nutr. 131 (3S):989S–993S. 2001.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Steiner M, Khan AH, Holbert D and Lin RIS:
A double-blind crossover study in moderately hypercholesterolemic
men that compared the effect of aged garlic extract and placebo
administration on blood lipids. Am J Clin Nutr. 64:866–870.
1996.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Lau BHS: Suppression of LDL oxidation by
garlic compounds is a possible mechanism of cardiovascular health
benefit. J Nutr. 136 (3 Suppl):765S–768S. 2006.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Poznyak AV, Sadykhov NK, Kartuesov AG,
Borisov EE, Melnichenko AA, Grechko AV and Orekhov AN: Hypertension
as a risk factor for atherosclerosis: Cardiovascular risk
assessment. Front Cardiovasc Med. 9(959285)2022.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Visseren F, Mach F, Smulders YM, Carballo
D, Koskinas KC, Bäck M, Benetos A, Biffi A, Boavida JM, Capodanno
D, et al: 2021 ESC guidelines on cardiovascular disease prevention
in clinical practice. Eur Heart J. 42:3227–3337. 2021.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Maraj I, Makaryus JN, Ashkar A, McFarlane
SI and Makaryus AN: Hypertension management in the high
cardiovascular risk population. Int J Hypertens.
2013(382802)2013.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Ried K, Frank OR and Stocks NP: Aged
garlic extract reduces blood pressure in hypertensives: A
dose-response trial. Eur J Clin Nutr. 67:64–70. 2013.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Ried K, Travica N and Sali A: The effect
of aged garlic extract on blood pressure and other cardiovascular
risk factors in uncontrolled hypertensives: The AGE at heart trial.
Integr Blood Press Control. 9:9–21. 2016.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Ried K, Travica N and Sali A: The effect
of kyolic aged garlic extract on gut microbiota, inflammation, and
cardiovascular markers in hypertensives: The GarGIC trial. Front
Nutr. 5(122)2018.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Matsutomo T, Ushijima M, Kodera Y,
Nakamoto M, Takashima M, Morihara N and Tamura K: Metabolomic study
on the antihypertensive effect of S-1-propenylcysteine in
spontaneously hypertensive rats using liquid chromatography coupled
with quadrupole-orbitrap mass spectrometry. J Chromatogr B Analyt
Technol Biomed Life Sci. 1046:147–155. 2017.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Matsutomo T, Ushijima M, Kunimura K and
Ohtani M: Metabolomic study reveals the acute hypotensive effect of
S-1-propenylcysteine accompanied by alteration of the plasma
histidine level in spontaneously hypertensive rats. J Pharm Biomed
Anal. 168:148–154. 2019.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Ushijima M, Takashima M, Kunimura K,
Kodera Y, Morihara N and Tamura K: Effects of S-1-propenylcysteine,
a sulfur compound in aged garlic extract, on blood pressure and
peripheral circulation in spontaneously hypertensive rats. J Pharm
Pharmacol. 70:559–565. 2018.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Cruz C, Correa-Rotter R, Sánchez-González
DJ, Hernández-Pando R, Maldonado PD, Martínez-Martínez CM,
Medina-Campos ON, Tapia E, Aguilar D, Chirino YI and
Pedraza-Chaverri J: Renoprotective and antihypertensive effects of
S-allylcysteine in 5/6 nephrectomized rats. Am J Physiol Renal
Physiol. 293:F1691–F1698. 2007.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Halim RM, Kamisah Y, Aziz NF, Sudirman UM,
Ahmad NAN, Kok-Yong C and Zainalabidin S: Pakistan veterinary
journal S-allylcysteine supplementation effects on vascular and
bone health in ovariectomized wistar rats. Pakistan Vet J.
41(507)2021.
|
|
73
|
Trimm E and Red-Horse K: Vascular
endothelial cell development and diversity. Nat Rev Cardiol.
20:197–210. 2023.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Badimon L, Peña E, Arderiu G, Padró T,
Slevin M, Vilahur G and Chiva-Blanch G: C-reactive protein in
atherothrombosis and angiogenesis. Front Immunol.
9(430)2018.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Čejková S, Králová-Lesná I and Poledne R:
Monocyte adhesion to the endothelium is an initial stage of
atherosclerosis development. Cor Vasa. 58:e419–e425. 2016.
|
|
76
|
Glass CK and Witztum JL: Atherosclerosis.
The road ahead. Cell. 104:503–516. 2001.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Schiopu A, Nadig SN, Cotoi OS, Hester J,
Van Rooijen N and Wood KJ: Inflammatory Ly-6C(hi) monocytes play an
important role in the development of severe transplant
arteriosclerosis in hyperlipidemic recipients. Atherosclerosis.
223:291–298. 2012.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Woollard KJ and Geissmann F: Monocytes in
atherosclerosis: Subsets and functions. Nat Rev Cardiol. 7:77–86.
2010.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Hilgendorf I, Swirski FK and Robbins CS:
Monocyte fate in atherosclerosis. Arterioscler Thromb Vasc Biol.
35:272–279. 2015.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Pang J, Maienschein-Cline M and Koh TJ:
Enhanced proliferation of Ly6C+ monocytes/macrophages
contributes to chronic inflammation in skin wounds of diabetic
mice. J Immunol. 206:621–630. 2021.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Ho SE, Ide N and Lau BH: S-allyl cysteine
reduces oxidant load in cells involved in the atherogenic process.
Phytomedicine. 8:39–46. 2001.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Ide N and Lau BH: S-allylcysteine
attenuates oxidative stress in endothelial cells. Drug Dev Ind
Pharm. 25:619–624. 1999.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Ide N and Lau BH: Garlic compounds
minimize intracellular oxidative stress and inhibit nuclear
factor-kappa b activation. J Nutr. 131 (3S):1020S–1026S.
2001.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Ide N and Lau BH: Garlic compounds protect
vascular endothelial cells from oxidized low density
lipoprotein-induced injury. J Pharm Pharmacol. 49:908–911.
1997.PubMed/NCBI View Article : Google Scholar
|
|
85
|
Ide N, Lau BH, Ryu K, Matsuura H and
Itakura Y: Antioxidant effects of fructosyl arginine, a Maillard
reaction product in aged garlic extract. J Nutr Biochem.
10:372–376. 1999.PubMed/NCBI View Article : Google Scholar
|
|
86
|
Hiramatsu K, Tsuneyoshi T, Ogawa T and
Morihara N: Aged garlic extract enhances heme oxygenase-1 and
glutamate-cysteine ligase modifier subunit expression via the
nuclear factor erythroid 2-related factor 2-antioxidant response
element signaling pathway in human endothelial cells. Nutr Res.
36:143–149. 2016.PubMed/NCBI View Article : Google Scholar
|
|
87
|
Tsuneyoshi T, Kunimura K and Morihara N:
S-1-Propenylcysteine augments BACH1 degradation and heme oxygenase
1 expression in a nitric oxide-dependent manner in endothelial
cells. Nitric Oxide. 84:22–29. 2019.PubMed/NCBI View Article : Google Scholar
|
|
88
|
Tsuneyoshi T: BACH1 mediates the
antioxidant properties of aged garlic extract. Exp Ther Med.
19:1500–1503. 2020.PubMed/NCBI View Article : Google Scholar
|
|
89
|
Kim KM, Chun SB, Koo MS, Choi WJ, Kim TW,
Kwon YG, Chung HT, Billiar TR and Kim YM: Differential regulation
of NO availability from macrophages and endothelial cells by the
garlic component S-allyl cysteine. Free Radic Biol Med. 30:747–756.
2001.PubMed/NCBI View Article : Google Scholar
|
|
90
|
Geddo F, Querio G, Asteggiano A,
Antoniotti S, Porcu A, Occhipinti A, Medana C and Gallo MP:
Improving endothelial health with food-derived H2S
donors: An in vitro study with S-allyl cysteine and with a
black-garlic extract enriched in sulfur-containing compounds. Food
Funct. 14:4163–4172. 2023.PubMed/NCBI View Article : Google Scholar
|
|
91
|
Tsuneyoshi T, Kanamori Y, Matsutomo T and
Morihara N: Dehydrodiconiferyl alcohol suppresses monocyte adhesion
to endothelial cells by attenuation of JNK signaling pathway.
Biochem Biophys Res Commun. 465:408–413. 2015.PubMed/NCBI View Article : Google Scholar
|
|
92
|
Lei YP, Chen HW, Sheen LY and Lii CK:
Diallyl disulfide and diallyl trisulfide suppress oxidized
LDL-induced vascular cell adhesion molecule and E-selectin
expression through protein kinase A- and B-dependent signaling
pathways. J Nutr. 138:996–1003. 2008.PubMed/NCBI View Article : Google Scholar
|
|
93
|
Miki S, Inokuma KI, Takashima M, Nishida
M, Sasaki Y, Ushijima M, Suzuki JI and Morihara N: Aged garlic
extract suppresses the increase of plasma glycated albumin level
and enhances the AMP-activated protein kinase in adipose tissue in
TSOD mice. Mol Nutr Food Res. 61(1600797)2017.PubMed/NCBI View Article : Google Scholar
|
|
94
|
Adams MR, Jessup W, Hailstones D and
Celermajer DS: L-arginine reduces human monocyte adhesion to
vascular endothelium and endothelial expression of cell adhesion
molecules. Circulation. 95:662–668. 1997.PubMed/NCBI View Article : Google Scholar
|
|
95
|
Cooke JP, Singer AH, Tsao P, Zera P, Rowan
RA and Billingham ME: Antiatherogenic effects of L-arginine in the
hypercholesterolemic rabbit. J Clin Invest. 90:1168–1172.
1992.PubMed/NCBI View Article : Google Scholar
|
|
96
|
Tsao PS, McEvoy LM, Drexler H, Butcher EC
and Cooke JP: Enhanced endothelial adhesiveness in
hypercholesterolemia is attenuated by L-arginine. Circulation.
89:2176–2182. 1994.PubMed/NCBI View Article : Google Scholar
|
|
97
|
Kunimura K, Miki S, Takashima M and Suzuki
JI: S-1-propenylcysteine improves TNF-α-induced vascular
endothelial barrier dysfunction by suppressing the GEF-H1/RhoA/Rac
pathway. Cell Commun Signal. 19(17)2021.PubMed/NCBI View Article : Google Scholar
|
|
98
|
Mehta JL, Sanada N, Hu CP, Chen J,
Dandapat A, Sugawara F, Satoh H, Inoue K, Kawase Y, Jishage K, et
al: Deletion of LOX-1 reduces atherogenesis in LDLR knockout mice
fed high cholesterol diet. Circ Res. 100:1634–1642. 2007.PubMed/NCBI View Article : Google Scholar
|
|
99
|
Febbraio M, Podrez EA, Smith JD, Hajjar
DP, Hazen SL, Hoff HF, Sharma K and Silverstein RL: Targeted
disruption of the class B scavenger receptor CD36 protects against
atherosclerotic lesion development in mice. J Clin Invest.
105:1049–1056. 2000.PubMed/NCBI View Article : Google Scholar
|
|
100
|
Kunjathoor VV, Febbraio M, Podrez EA,
Moore KJ, Andersson L, Koehn S, Rhee JS, Silverstein R, Hoff HF and
Freeman MW: Scavenger receptors class A-I/II and CD36 are the
principal receptors responsible for the uptake of modified low
density lipoprotein leading to lipid loading in macrophages. J Biol
Chem. 277:49982–49988. 2002.PubMed/NCBI View Article : Google Scholar
|
|
101
|
Morihara N, Ide N and Weiss N: Aged garlic
extract inhibits CD36 expression in human macrophages via
modulation of the PPARgamma pathway. Phytother Res. 24:602–608.
2010.PubMed/NCBI View Article : Google Scholar
|
|
102
|
Ide N, Keller C and Weiss N: Aged garlic
extract inhibits homocysteine-induced CD36 expression and foam cell
formation in human macrophages. J Nutr. 136 (3 Suppl):755S–758S.
2006.PubMed/NCBI View Article : Google Scholar
|
|
103
|
Morihara N, Ide N and Weiss N: Aged garlic
extract inhibits homocysteine-induced scavenger receptor CD36
expression and oxidized low-density lipoprotein cholesterol uptake
in human macrophages in vitro. J Ethnopharmacol. 134:711–716.
2011.PubMed/NCBI View Article : Google Scholar
|
|
104
|
Matsuo M: ABCA1 and ABCG1 as potential
therapeutic targets for the prevention of atherosclerosis. J
Pharmacol Sci. 148:197–203. 2022.PubMed/NCBI View Article : Google Scholar
|
|
105
|
Malekpour-Dehkordi Z, Javadi E, Doosti M,
Paknejad M, Nourbakhsh M, Yassa N, Gerayesh-Nejad S and Heshmat R:
S-Allylcysteine, a garlic compound, increases ABCA1 expression in
human THP-1 macrophages. Phytother Res. 27:357–361. 2013.PubMed/NCBI View Article : Google Scholar
|
|
106
|
Lammers B, Zhao Y, Foks AC, Hildebrand RB,
Kuiper J, van Berkel TJC and van Eck M: Leukocyte ABCA1 remains
atheroprotective in splenectomized LDL receptor knockout mice. PLoS
One. 7(e48080)2012.PubMed/NCBI View Article : Google Scholar
|
|
107
|
Yvan-Charvet L, Ranalletta M, Wang N, Han
S, Terasaka N, Li R, Welch C and Tall AR: Combined deficiency of
ABCA1 and ABCG1 promotes foam cell accumulation and accelerates
atherosclerosis in mice. J Clin Invest. 117:3900–3908.
2007.PubMed/NCBI View Article : Google Scholar
|
|
108
|
Wang X, Collins HL, Ranalletta M, Fuki IV,
Billheimer JT, Rothblat GH, Tall AR and Rader DJ: Macrophage ABCA1
and ABCG1, but not SR-BI, promote macrophage reverse cholesterol
transport in vivo. J Clin Invest. 117:2216–2224. 2007.PubMed/NCBI View Article : Google Scholar
|
|
109
|
Van Eck M, Singaraja RR, Ye D, Hildebrand
RB, James ER, Hayden MR and Van Berkel TJ: Macrophage ATP-binding
cassette transporter A1 overexpression inhibits atherosclerotic
lesion progression in low-density lipoprotein receptor knockout
mice. Arterioscler Thromb Vasc Biol. 26:929–934. 2006.PubMed/NCBI View Article : Google Scholar
|
|
110
|
Wu J, He S, Song Z, Chen S, Lin X, Sun H,
Zhou P, Peng Q, Du S, Zheng S and Liu X: Macrophage polarization
states in atherosclerosis. Front Immunol.
14(1185587)2023.PubMed/NCBI View Article : Google Scholar
|
|
111
|
Bisgaard LS, Mogensen CK, Rosendahl A,
Cucak H, Nielsen LB, Rasmussen SE and Pedersen TX: Bone
marrow-derived and peritoneal macrophages have different
inflammatory response to oxLDL and M1/M2 marker
expression-implications for atherosclerosis research. Sci Rep.
6(35234)2016.PubMed/NCBI View Article : Google Scholar
|
|
112
|
Khallou-Laschet J, Varthaman A, Fornasa G,
Compain C, Gaston AT, Clement M, Dussiot M, Levillain O,
Graff-Dubois S, Nicoletti A and Caligiuri G: Macrophage plasticity
in experimental atherosclerosis. PLoS One. 5(e8852)2010.PubMed/NCBI View Article : Google Scholar
|
|
113
|
Seifert R, Kuhlmann MT, Eligehausen S,
Kiefer F, Hermann S and Schäfers M: Molecular imaging of MMP
activity discriminates unstable from stable plaque phenotypes in
shear-stress induced murine atherosclerosis. PLoS One.
13(e0204305)2018.PubMed/NCBI View Article : Google Scholar
|
|
114
|
Li J, Lei HT, Cao L, Mi YN, Li S and Cao
YX: Crocin alleviates coronary atherosclerosis via inhibiting lipid
synthesis and inducing M2 macrophage polarization. Int
Immunopharmacol. 55:120–127. 2018.PubMed/NCBI View Article : Google Scholar
|
|
115
|
Aharoni S, Lati Y, Aviram M and Fuhrman B:
Pomegranate juice polyphenols induce a phenotypic switch in
macrophage polarization favoring a M2 anti-inflammatory state.
Biofactors. 41:44–51. 2015.PubMed/NCBI View Article : Google Scholar
|
|
116
|
Bi Y, Chen J, Hu F, Liu J, Li M and Zhao
L: M2 macrophages as a potential target for antiatherosclerosis
treatment. Neural Plast. 2019(6724903)2019.PubMed/NCBI View Article : Google Scholar
|
|
117
|
Zhang XN, Zhao N, Guo FF, Wang YR, Liu SX
and Zeng T: Diallyl disulfide suppresses the
lipopolysaccharide-driven inflammatory response of macrophages by
activating the Nrf2 pathway. Food Chem Toxicol.
159(112760)2022.PubMed/NCBI View Article : Google Scholar
|
|
118
|
Xiang AH, Peters RK, Kjos SL, Ochoa C,
Marroquin A, Goico J, Tan S, Wang C, Azen SP, Liu CR, et al: Effect
of thiazolidinedione treatment on progression of subclinical
atherosclerosis in premenopausal women at high risk for type 2
diabetes. J Clin Endocrinol Metab. 90:1986–1991. 2005.PubMed/NCBI View Article : Google Scholar
|
|
119
|
Pioglitazone inhibits progression of
atherosclerosis. Nat Clin Pract Cardiovasc Med. 5(512)2008.
|
|
120
|
Nakayama T, Komiyama N, Yokoyama M,
Namikawa S, Kuroda N, Kobayashi Y and Komuro I: Pioglitazone
induces regression of coronary atherosclerotic plaques in patients
with type 2 diabetes mellitus or impaired glucose tolerance: A
randomized prospective study using intravascular ultrasound. Int J
Cardiol. 138:157–165. 2010.PubMed/NCBI View Article : Google Scholar
|
|
121
|
Chen R, McVey DG, Shen D, Huang X and Ye
S: Phenotypic switching of vascular smooth muscle cells in
atherosclerosis. J Am Heart Assoc. 12(e031121)2023.PubMed/NCBI View Article : Google Scholar
|
|
122
|
Durham AL, Speer MY, Scatena M, Giachelli
CM and Shanahan CM: Role of smooth muscle cells in vascular
calcification: Implications in atherosclerosis and arterial
stiffness. Cardiovasc Res. 114:590–600. 2018.PubMed/NCBI View Article : Google Scholar
|
|
123
|
Hom C, Luo Y and Budoff MJ: The effects of
aged garlic extract on coronary artery calcification progression. J
Nutr Food Sci. S5(005)2015.
|
|
124
|
Yamakawa T, Matsutomo T, Hofmann T and
Kodera Y: Aged garlic extract and one of the constituent,
(+)-(2S,3R)-dehydrodiconiferyl alcohol, inhibits alkaline
phosphatase activity induced by inflammation factors in human
vascular smooth muscle cells. Food Nutr Sci. 5:177–184. 2014.
|
|
125
|
Jackson SP: The growing complexity of
platelet aggregation. Blood. 109:5087–5095. 2007.PubMed/NCBI View Article : Google Scholar
|
|
126
|
Bye AP, Unsworth AJ and Gibbins JM:
Platelet signaling: A complex interplay between inhibitory and
activatory networks. J Thromb Haemost. 14:918–930. 2016.PubMed/NCBI View Article : Google Scholar
|
|
127
|
Asada Y, Yamashita A, Sato Y and
Hatakeyama K: Pathophysiology of atherothrombosis: Mechanisms of
thrombus formation on disrupted atherosclerotic plaques. Pathol
Int. 70:309–322. 2020.PubMed/NCBI View Article : Google Scholar
|
|
128
|
Steiner M and Li W: Aged garlic extract, a
modulator of cardiovascular risk factors: A dose-finding study on
the effects of AGE on platelet functions. J Nutr. 131
(3S):980S–984S. 2001.PubMed/NCBI View Article : Google Scholar
|
|
129
|
Rahman K and Billington D: Dietary
supplementation with aged garlic extract inhibits ADP-induced
platelet aggregation in humans. J Nutr. 130:2662–2665.
2000.PubMed/NCBI View Article : Google Scholar
|
|
130
|
Macan H, Uykimpang R, Alconcel M, Takasu
J, Razon R, Amagase H and Niihara Y: Aged garlic extract may be
safe for patients on warfarin therapy. J Nutr. 136 (3
Suppl):793S–795S. 2006.PubMed/NCBI View Article : Google Scholar
|
|
131
|
Li Z, Delaney MK, O'Brien KA and Du X:
Signaling during platelet adhesion and activation. Arterioscler
Thromb Vasc Biol. 30:2341–2349. 2010.PubMed/NCBI View Article : Google Scholar
|
|
132
|
Yacoub D, Théorêt JF, Villeneuve L,
Abou-Saleh H, Mourad W, Allen BG and Merhi Y: Essential role of
protein kinase C delta in platelet signaling, alpha IIb beta 3
activation, and thromboxane A2 release. J Biol Chem.
281:30024–30035. 2006.PubMed/NCBI View Article : Google Scholar
|
|
133
|
Lannan KL, Sahler J, Kim N, Spinelli SL,
Maggirwar SB, Garraud O, Cognasse F, Blumberg N and Phipps RP:
Breaking the mold: Transcription factors in the anucleate platelet
and platelet-derived microparticles. Front Immunol.
6(48)2015.PubMed/NCBI View Article : Google Scholar
|
|
134
|
Morihara N and Hino A: Aged garlic extract
suppresses platelet aggregation by changing the functional property
of platelets. J Nat Med. 71:249–256. 2017.PubMed/NCBI View Article : Google Scholar
|
|
135
|
Allison GL, Lowe GM and Rahman K: Aged
garlic extract may inhibit aggregation in human platelets by
suppressing calcium mobilization. J Nutr. 136 (3 Suppl):789S–792S.
2006.PubMed/NCBI View Article : Google Scholar
|
|
136
|
Rahman K, Lowe GM and Smith S: Aged garlic
extract inhibits human platelet aggregation by altering
intracellular signaling and platelet shape change. J Nutr.
146:410S–415S. 2016.PubMed/NCBI View Article : Google Scholar
|
|
137
|
Allison GL, Lowe GM and Rahman K: Aged
garlic extract inhibits platelet activation by increasing
intracellular cAMP and reducing the interaction of GPIIb/IIIa
receptor with fibrinogen. Life Sci. 91:1275–1280. 2012.PubMed/NCBI View Article : Google Scholar
|
|
138
|
Qi R, Liao F, Inoue K, Yatomi Y, Sato K
and Ozaki Y: Inhibition by diallyl trisulfide, a garlic component,
of intracellular Ca(2+) mobilization without affecting
inositol-1,4, 5-trisphosphate [IP(3)] formation in activated
platelets. Biochem Pharmacol. 60:1475–1483. 2000.
|
|
139
|
Tian KJ, Yang Y, Chen GS, Deng NH, Tian Z,
Bai R, Zhang F and Jiang ZS: Omics research in atherosclerosis. Mol
Cell Biochem: Oct 24, 2024 (Epub ahead of print).
|
|
140
|
Duan M, Zhao WL, Zhou L, Novák P, Zhu X
and Yin K: Omics research in vascular calcification. Clin Chim
Acta. 511:319–328. 2020.PubMed/NCBI View Article : Google Scholar
|
|
141
|
Wu X and Zhang H: Omics approaches
unveiling the biology of human atherosclerotic plaques. Am J
Pathol. 194:482–498. 2024.PubMed/NCBI View Article : Google Scholar
|
|
142
|
Das V, Zhang X, Djordjevic D, Bergman Q,
Narayanan S, Shungin D, Chemaly M, Karadimou G, Vuckovic S, Prasad
I, et al: Multi-omics integration from a large human biobank
identifies key molecular mechanisms and signatures of
atherosclerotic plaque instability. Atherosclerosis. 395 (Suppl
1)(S117757)2024.
|
|
143
|
Sánchez-Cabo F, Fuster V, Silla-Castro JC,
González G, Lorenzo-Vivas E, Alvarez R, Callejas S, Benguría A, Gil
E, Núñez E, et al: Subclinical atherosclerosis and accelerated
epigenetic age mediated by inflammation: A multi-omics study. Eur
Heart J. 44:2698–2709. 2023.PubMed/NCBI View Article : Google Scholar
|
|
144
|
Kardassis D, Vindis C, Stancu CS, Toma L,
Gafencu AV, Georgescu A, Alexandru-Moise N, Molica F, Kwak BR,
Burlacu A, et al: Unravelling molecular mechanisms in
atherosclerosis using cellular models and omics technologies.
Vascul Pharmacol. 158(107452)2024.PubMed/NCBI View Article : Google Scholar : (Epub ahead of
print).
|