Introduction
Prion diseases, also called transmissible spongiform
encephalopathies (TSEs), are a group of fatal neurodegenerative
diseases characterized by neuronal loss and spongiform degeneration
in the central nervous system (CNS). These diseases may affect many
mammalian hosts, such as Creutzfeldt-Jakob disease (CJD), fatal
familial insomnia (FFI), Gerstmann-Straussler-Scheinker (GSS)
syndrome and Kuru in human, scrapie in sheep and goat, bovine
spongiform encephalopathy (BSE) in cattle, and chronic wasting
disease (CWD) in deer and elk (1,2).
It is commonly accepted that these type of diseases are caused by
the conversion of a cellular protein PrPC in the CNS to
an abnormally folded isoform, PrPSc, which is insoluble,
proteinase K-resistant and infectious (3,4).
The accumulation of PrPSc may be linked to the
pathogenesis of prion diseases (5).
Microtubules and their associated proteins (MAPs)
provide a dynamic network that is critical for cell mitosis, the
establishment of cellular polarity and intracellular transport.
These processes require a dynamic microtubule array and are
regulated by motor proteins and structural MAPs, such as tau,
MAP1B, MAP2 and MAP4. The binding of tau and other MAPs to
microtubules are regulated instantaneously by phosphorylation on
their tubulin binding repeats (6,7).
Our previous studies identified the profiles of tau and relevant
phosphorylating kinases that are abnormally changed (8) and the levels of tubulin that are
decreased in the brain tissues of experimental scrapie rodents and
human CJD. Meanwhile, destruction of microtubule structures have
been repeatedly observed in cells receiving genetic CJD
(gCJD)-associated PrP mutants (9,10).
These data highlight that damage of microtubules is an essential
process in the pathogenesis of TSEs.
Microtubule affinity-regulating kinase 4 (MARK4)
belongs to a family of kinases that phosphorylate tau (p-tau) at
the serine motif (11). It has
two different splicing isoforms, MARK4-S, which is predominantly
expressed in the brain, and MARK4-L, which is ubiquitously
expressed in all tissues with high expression levels in testis,
neural progenitors and glial tumors (12–14). In contrast to the other three MARK
paralogs that all exhibit uniform cytoplasmic localization, MARK4
colocalizes with the centrosome and with microtubules in cultured
cells. In differentiated neuroblastoma cells, MARK4 localizes
prominently at the tips of the neurite-like processes (11). It has been reported that serine
262 in tau, which is specifically phosphorylated by MARK, is in a
hyperphosphorylated form in the neurofibrillary deposits found in
Alzheimer’s disease brains (15).
In addition, the microtubule-dependent transport is regulated
through MARK. In primary retinal ganglion cells, transfection with
tau leads to the inhibition of axonal transport of mitochondria,
amyloid precursor protein (APP) vesicles and other cell components.
This leads to the starvation of axons and their vulnerability
against stress. This transport inhibition can be rescued by
phosphorylating tau with MARK (16). However, the alteration of the MARK
family in TSEs remains unknown.
Our study of screening the transcriptional diversity
in the brain tissues of human prion diseases with a commercial mRNA
microarray shows that the expression of MARK4 is obviously
decreased (unpublished data). In the present study, we present
evidence that the MARK4 protein in the brain tissues of
scrapie-agent 263K and 139A-infected rodents is almost undetectable
at the terminal stages, and the decline of MARK4 in the brain
tissues of agent 263K-infected hamsters was closely related to the
prolonged incubation time. MARK4 was extremely decreased in the
brain regions with a mass of PrPSc in a G114V gCJD
patient, but was clearly observable in the regions with a minimum
amount of PrPSc or without detectable PrPSc
in a D178N FFI patient. Accordingly, the ratio of p-tau at Ser262
to total tau was also decreased in the brain tissues of agent
263K-infected hamsters. Those results could be reproduced in the
cell lines exposed to the synthetic peptide PrP106-126, which
possesses similar physicochemical and pathological properties to
PrPSc. It indicates that the decrease of MARK4 in brain
tissues may be involved in TSEs and may be correlated to the
deposits of PrPSc.
Materials and methods
Ethics statement
Usage of the stored human and animal samples in this
study was approved by the Ethics Committee of National Institute
for Viral Disease Prevention and Control, China CDC. All signed
informed consents were collected and stored by the China CJD
Surveillance Center Housing. Experimental protocols were in
accordance with the Chinese Regulations for the Administration of
Affairs Concerning Experimental Animals.
Specimens
Four Chinese golden hamsters inoculated
intracerebrally with hamster-adapted scrapie agent 263K and 4 mice
inoculated intracerebrally with mouse-adapted scrapie agent 139A
were examined in this study. The incubation time of 263K-infected
hamsters was 79.1±8.6 days (17),
while that of 139A-infected mice was 153±4 days (18). Brain samples of the hamsters
infected with 263K agent at the 20, 40, 60 and 80 days after
inoculation were collected. Samples of frontal lobe, parietal lobe,
occipital lobe, temporal lobe, thalamus and callosal gyrus from a
G114V gCJD patient (19,20) and a D178N FFI patient (21) that were reported previously were
also enrolled. All brains were removed surgically and stored at
−80°C until use. Brains from 4 normal hamsters and 4 normal mice
were collected as controls.
Preparations of brain homogenates
The brain tissues (10% w/v) were homogenized in
lysis buffer (100 mM NaCl, 10 mM ethylenediaminetetraacetic acid,
0.5% Nonidet P-40, 0.5% Na deoxycholate in 10 mM Tris-HCl, pH 7.4).
The homogenates were centrifuged at 2,000 × g for 10 min, and then
the supernatants were collected and frozen at −80°C for the further
experiments.
Immunohistochemical (IHC) assays
The brain tissues were subjected to formalin
fixation and paraffin embedding for conventional methodology
(20). The slices were subjected
to conventional immunostaining of MARK4, PrPSc and GFAP.
PrPSc and GFAP stainings were performed with 1:250
diluted PrP specific monoclonal antibody (mAb; Dako, Denmark) 3F4
and 1:200 diluted GFAP polyclonal antibody (pAb; Santa Cruz
Biotechnology, Inc., USA) according to the protocols described
previously (17). For MARK4
staining, the slices were digested by enzymes for 30 sec and 3%
hydrogen peroxide-methanol for 10 min. The slices were blocked with
normal goat serum for 10 min, then incubated with 1:100 diluted
anti-MARK4 pAb (Abcam, UK) at 4°C overnight, and subsequently with
horseradish peroxidase (HRP)-conjugated anti-rabbit IgG (Vector,
USA) at 37°C for 1 h. For visualization of immunostaining, a
commercial DAB kit (Vector) was used, and the slices were
counterstained with hematoxylin.
Western blot analyses
The brain tissue homogenates and cellular lysates
were separated using 12% SDS-polyacrylamide gel electrophoresis
(SDS-PAGE) and electronically transferred to nitrocellulose
membranes. After blocking with 5% non-fat milk powder in TBST
(phosphate-buffered saline, pH 7.6, containing 0.05% Tween-20), the
membranes were incubated with 1:1,000 diluted anti-MARK4 pAb,
1:5,000 diluted PrP specific mAb 3F4, 1:1,000 diluted anti-PrP mAb
1E4, 1:1,000-diluted anti-neuron specific enolase mAb (NSE; Abcam),
1:1,000 diluted anti-human β-actin mAb, 1:1,000 diluted anti-tau
mAb tau13 (Santa Cruz Biotechnology, Inc.), or 1:1,000 diluted
anti-p-tau at Ser262 pAb TAU [pS262] (Biosource, USA) at 4°C
overnight. After washing thrice with TBST, the membranes were
incubated with 1:8,000 diluted HRP-conjugated anti-rabbit or
anti-mouse IgG (Boehringer, Germany) in TBST at room temperature
for 1 h, followed by detection of signals with an enhanced
chemiluminescence detection kit (Amersham Pharmacia Biotech,
USA).
Peptides
Peptides PrP106-126 (KTNMKHMAGAAAAG AVVGGLG) and
scrambled (scr) peptide PrP106-126 (AVHT GLGAMAALNMVVGGAAGL) were
synthesized and purified by Invitrogen (USA). Peptides were freshly
dissolved in dimethyl sulfoxide (DMSO) to a concentration of 50 μM
before each experiment.
Cell culture
The adherent human neuroblastoma cell line SK-N-SH
and rat pheochromocytoma cell line PC12 were cultured in Dulbecco’s
modified Eagle’s medium (DMEM) supplemented with 10% fetal calf
serum (Gibco, USA). Cells were maintained at 37°C in a humidified
5% CO2 atmosphere.
Cell viability assays
SK-N-SH and PC12 cells at logarithmic growth stage
were trypsinized and seeded on a 96-well plate at a concentration
of 104 cells/well before peptide treatments. Twelve
hours after incubation with different concentrations of peptides,
cell viability was determined using a commercially available Cell
Counting kit (CCK-8; Dojindo, Japan). Briefly, 10 μl of CCK-8
reagent were added to each well and incubated at 37°C for 1 h or
until the media turned yellow. Absorbance was measured at 450 nm
with a spectrophotometer. Each experiment was performed in
triplicate and repeated at least three times.
Quantitative real-time PCR (qRT-PCR)
Total-RNA from cells was extracted using the RNeasy
mini kit (Qiagen, USA) reagent according to the manufacturer’s
instructions. Reverse transcription was performed with the Reverse
Transcription System (Promega, USA). Briefly, 1 μl of each RT
product was employed into subsequent PCR reactions. The primers for
MARK4 were designed based on the sequences of human MARK4 in
GenBank (NM_001199867.1) including MARK4(Hu), sense,
5′-GGCTATGAGGGTGAGGAGT TGAA-3′ and antisense, 5′-GCGGTGGTAGGTGGAAG
AGG-3′; β-actin, sense, 5′-CT ACAATGAGCTGCGTGTGGC-3′ and antisense,
5′-CAGG TCCAGACGCAGGATGGC-3′. qRT-PCR was performed on a 7900 Fast
Real-Time PCR System (Applied Biosystems, USA) using the following
conditions: 94°C for 15 sec, 56°C for 40 sec, and 72°C for 30 sec
for 40 cycles. The expression level of mRNA was determined relative
to that of the β-actin control. All real-time PCR reactions were
performed in triplicate.
Statistical analyses
Statistical analyses were performed using the SPSS
17.0 statistical package. Quantitative analysis of immunoblot
images was carried out using the ImageJ software. All data are
presented as the mean ± SD. One-way ANOVA was used to assess the
differences of OD450 values of CCK-8 between treatment groups with
mock group. p-values <0.05 were statistically significant.
Results
Decreases in MARK4 levels in the brain
tissues of the scrapie-infected rodents at the terminal stages of
the disease
To assess the potential changes in MARK4 levels in
the brain tissues with TSEs, 4 hamsters infected with agent 263K
(Ha-263K) and 4 mice infected with agent 139A (Mo-139A) were used
in this study. The clinical features and the presence of
PrPSc in the brain tissues of the scrapie-infected
animals have been previously reported (8,22).
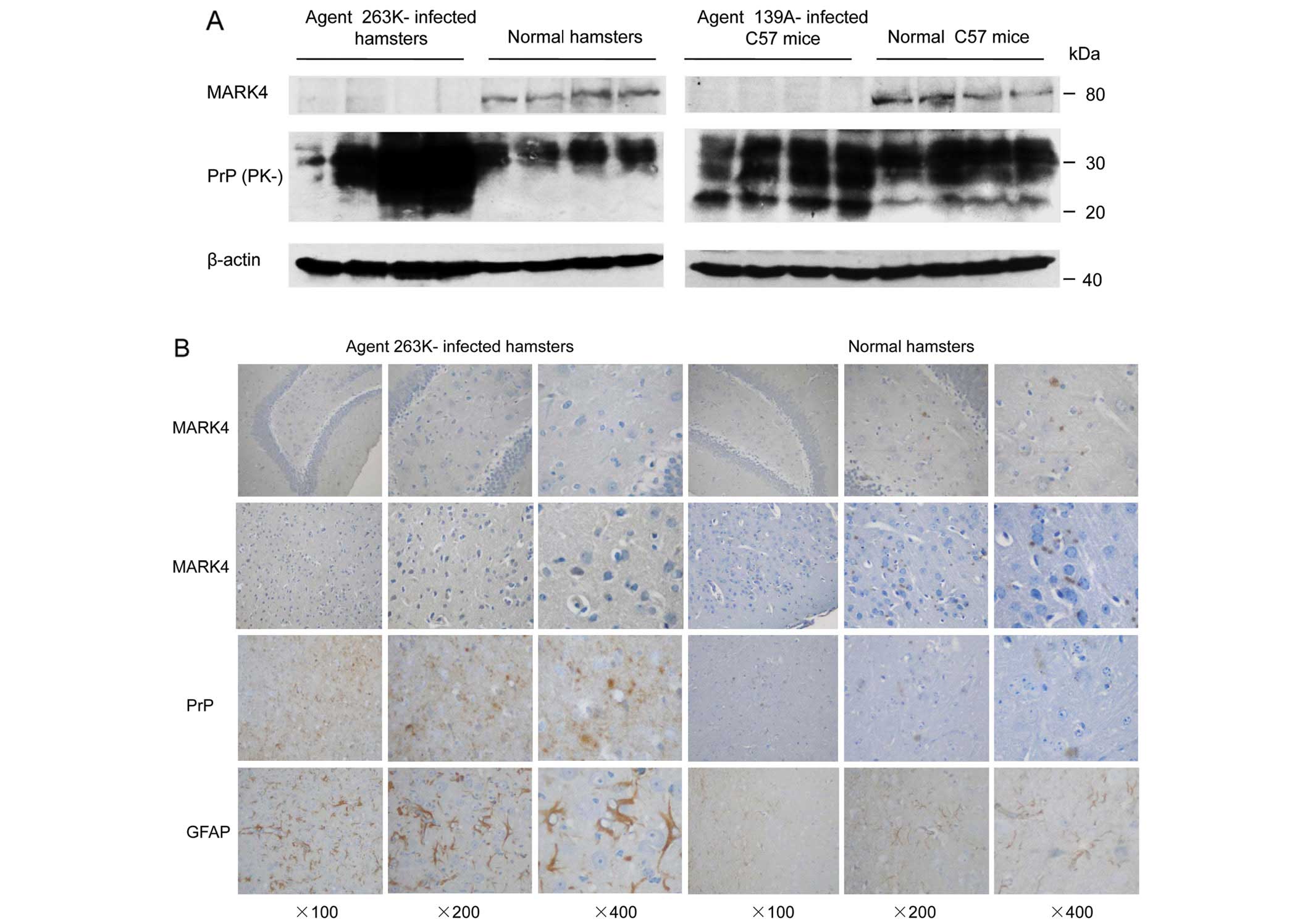
The amounts of MARK4 and total PrP were evaluated with western blot
analyses using individual antibodies. In accompaniment with large
amounts of total PrP signals, almost no MARK4 signal was observed
in the brain homogenates of two different kinds of scrapie-infected
animals, whereas a clear MARK4 specific signal was observed in
brain homogenates prepared from normal hamsters and mice at the
same age (Fig. 1A).
To further investigate the effects of decreased
MARK4 in scrapie-infected animals, the presence of MARK4 in the
brain tissues of Ha-263K-infected animals were analyzed with IHC.
Additionally, PrPSc deposits and astrogliosis were
monitored. As expected, large quantities of PrPSc
deposits were observed in the hippocampus and cortex of Ha-263K
animals, which accompanied different sizes of vacuolation, but were
not observed in the normal controls (Fig. 1B). More GFAP positively stained
long and fibrous-like cells were detected in brain tissues of
Ha-263K, while only small, filament-like structures appeared in
wild-type animals (Fig. 1B). More
round and granular MARK4 positively stained particles were
monitored in the brain tissues of normal hamsters, but were almost
unobservable in that of Ha-263K (Fig.
1B). The data strongly indicate that the levels of MARK4 in the
brain tissues of scrapie experimental rodents are severely
repressed at the terminal stages of diseases.
Decreases in MARK4 protein levels in the
brain tissues of a G114V gCJD patient, but not in a FFI
patient
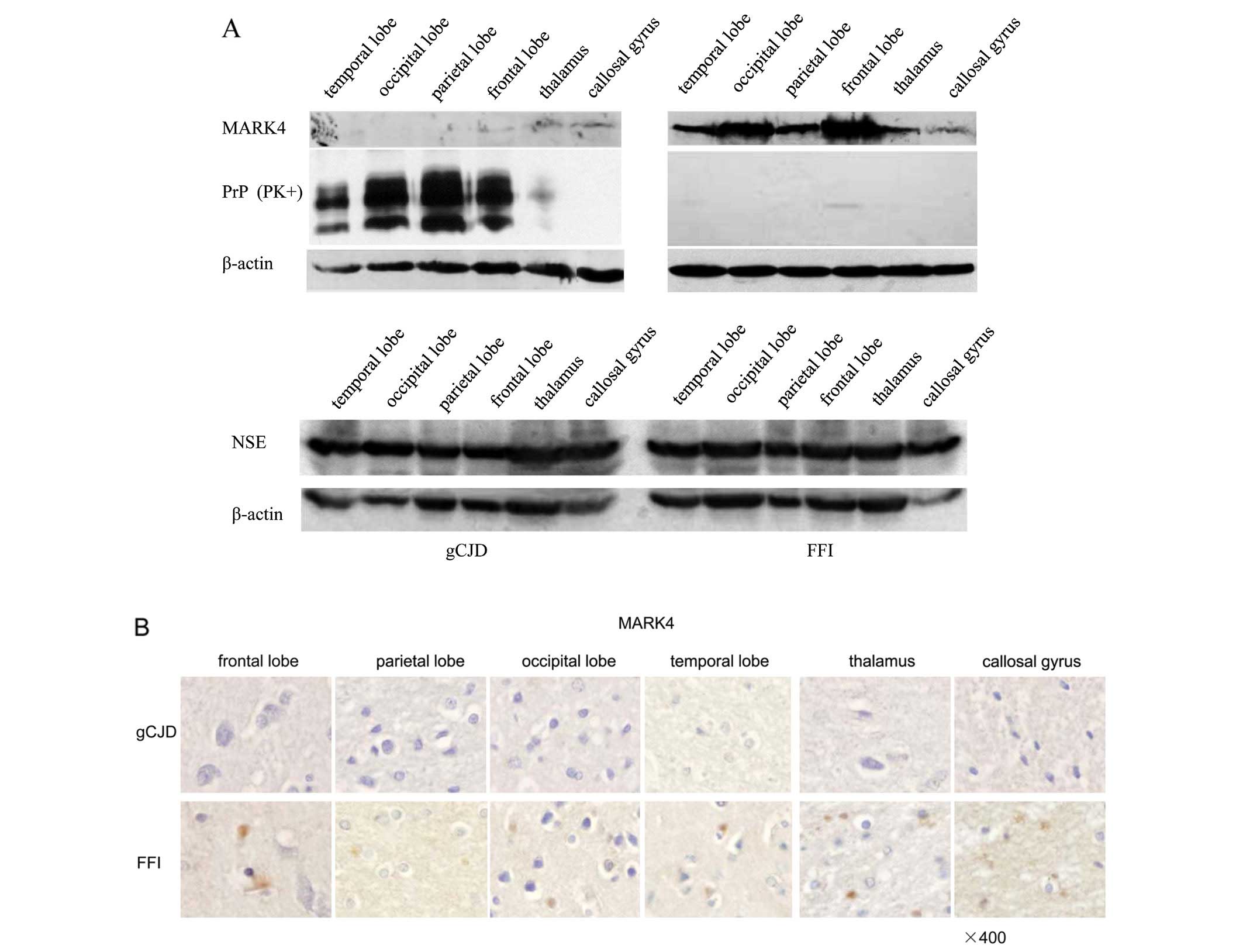
To address the state of MARK4 in human prion
diseases, the protein expression levels of MARK4 in six different
brain regions, including the frontal lobe, parietal lobe, occipital
lobe, temporal lobe, thalamus and callosal gyrus, from a G114V gCJD
patient and a D178N FFI patient were assessed using western
blotting. In accordance with the observations in scrapie-infected
animals, in the six preparations from a G114V gCJD patient
(Fig. 2A, left panel), there were
almost no MARK4 signal observed in the four cortex regions. These
regions were deposited with a mass of PrPSc, although
weak, noticeable MARK4 bands in thalamus and callosal gyrus were
deposited with a few PrPSc. Surprisingly, the
preparations from a D178N FFI patient had nearly undetectable
PrPres signals, while MARK4 specific bands were repeatedly observed
in all tested regions (Fig. 2A,
right panel). NSE-specific blots revealed similar signal
intensities between the brain tissues of the two cases (Fig. 2A). The slides of the six brain
regions from a G114V gCJD and a FFI patient were also screened with
MARK4-specific IHC. In line with the results from our western blot
analysis, clear, round and granular MARK4 positive-stained
particles were observed in the brain regions of FFI, but not in the
brain tissues of G114V gCJD (Fig.
2B). This result indicates that decreased MARK4 in brain
tissues may be a common feature in TSEs, which is likely linked to
the deposits of PrPSc.
Decreases of MARK4 are time-relative
events correlated with the deposits of PrPSc
To ascertain possible dynamic alterations of MARK4
in the brain tissues of scrapie experimental hamsters during the
incubation periods, brain samples infected with the agent Ha-263K
at 20, 40, 60 and 80 days post-inoculation (dpi) were collected.
PrP specific western blot analysis identified PK-resistant PrP
signals (PrPres) in 40 dpi preparations, but not in control and 20
dpi (Fig. 3A). The signal
intensities of PrPres became more intense with prolonged
incubation, showing a time dependency for this signal. In contrast,
MARK4 was clearly detectable in the brain tissues of normal
hamsters, which significantly weakened in samples infected with
Ha-263K at 20 dpi, and continually decreased with time, eventually
disappearing in the Ha-263K sample at 80 dpi (Fig. 3A). Quantitative analyses of the
gray values of PrPSc and MARK4 of each sample,
normalized to the individual values of β-actin, revealed two
opposite fluctuating curves, the increasing curve of
PrPSc and the declining curve of MARK4, along with the
incubation period (Fig. 3B).
These results illustrate that the levels of MARK4 in the brains of
the scrapie-infected animals decline with prolonged incubation,
which correlates with the increase of PrPSc.
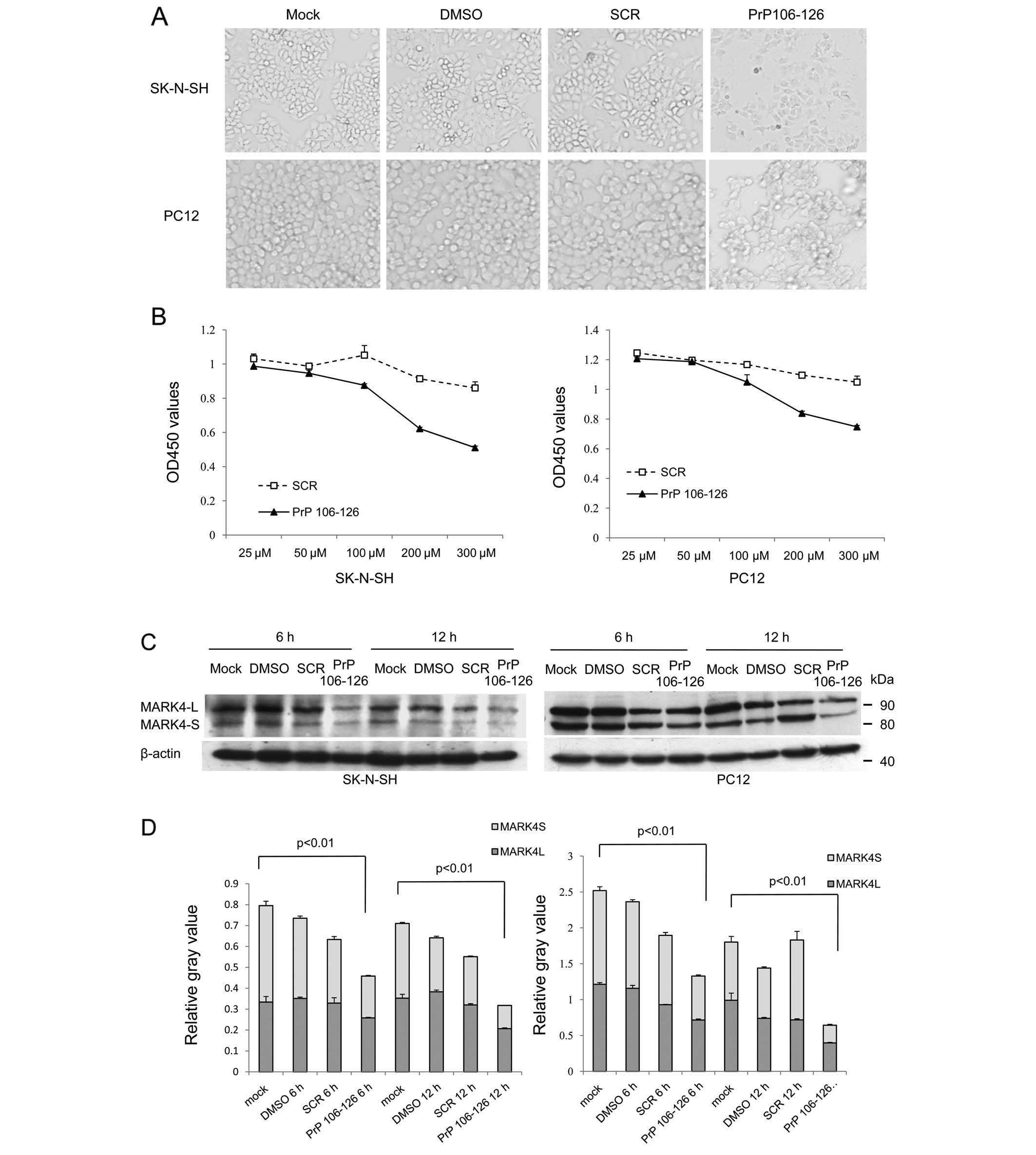
Exposure to peptide PrP106-126 reduces
the levels of MARK4 in cultured cells
Peptide PrP106-126 shows cytotoxicity in several
cultured cells in vitro, which may partially mimic the
features of PrPSc (23–25). To assess the possible changes in
cellular MARK4 following cytotoxicity induced by exposure to
peptide PrP106-126, neuroblastoma cell line SK-N-SH and rat
pheochromocytoma cell line PC12 were exposed to different
concentrations of PrP106-126. Both cell lines showed distinct
morphological changes and low cell viability when exposed to
PrP106-126, but did not show obvious changes when exposed to DMSO
or the same amount of scramble PrP106-126 peptide (Fig. 4A and B). After treatment with PrP
peptides for 6 and 12 h, the levels of MARK4 were evaluated using
western blot analyses. Two MARK4-specific bands in both cell
lysates migrate near the positions of 85 and 80 kDa (Fig. 4C), representing large and small
fragments of MARK4 (MARK4-L and MARK4-S), respectively. The protein
level of MARK4-L and MARK4-S both decreased after PrP106-126
treatment compared with scramble peptide or DMSO treatment.
Quantitative analyses of the relative gray numerical values
normalized to that of β-actin revealed that the levels of MARK4-L
and MARK4-S in the cells treated with PrP106-126 were significantly
lower than the mock-treated cells, whereas the cells exposed to
DMSO and scrambled PrP peptide were slightly decreased without
significant difference (Fig. 4D).
The reductions of MARK4-S after treatment of PrP106-126 were even
more remarkable. Prolonging the treatment times of PrP106-126
resulted in significant reductions of MARK4 in both cell lines.
To address the expression profile of MARK4 in the
cells after challenging with PrP106-126, a qRT-PCR specific for
MARK4 was performed. RT-PCR assays using extracted RNA from the
cells exposed to PrP106-126 for 6 and 12 h as well as the cells
receiving DMSO or scramble PrP peptide for 12 h revealed a 325-bp
amplified fragment after using RNA isolated from cells which were
further verified to be MARK4-specific sequences (data not shown).
Real-time PCR for MARK4 demonstrated that the amounts of MARK4
transcripts in the cells exposed to PrP106-126 were profoundly
downregulated when compared with that of the controls, showing a
statistically significant difference both in the SK-N-SH and PC12
cell lines (Fig. 5 and Table I). Meanwhile, treatment with
PrP106-126 for 12 h decreased the MARK4 expression in cells more
than the 6 h treatment. These data suggest that PrP106-126
downregulates the expression of the endogenous MARK4 when it is
cytotoxic to cultured cells.
 | Table I.Comparison of the amplification data
of MARK4-specific mRNAs in various cell preparations by real-time
PCR. |
Table I.
Comparison of the amplification data
of MARK4-specific mRNAs in various cell preparations by real-time
PCR.
| MARK4 Average
Ct | β-actin Average
Ct | Average
ΔCta |
ΔΔCtb |
2−ΔΔCt |
|---|
| PC-12 | | | | | |
| Mock | 23.74 | 13.84 | 9.90 | 0.00 | 1.00±0.00 |
| DMSO | 23.67 | 13.53 | 10.14 | 0.24 | 0.85±0.07 |
| SCR | 23.76 | 13.82 | 9.94 | 0.04 | 0.97±0.11 |
| PrP 6 h | 23.59 | 12.98 | 10.61 | 0.71 | 0.61±0.04 |
| PrP 12 h | 25.01 | 13.36 | 11.65 | 1.75 | 0.30±0.02 |
| SK-N-SH | | | | | |
| Mock | 22.56 | 13.10 | 9.46 | 0.00 | 1.00±0.00 |
| DMSO | 22.76 | 13.11 | 9.65 | 0.04 | 0.97±0.11 |
| SCR | 22.85 | 13.20 | 9.65 | 0.06 | 0.96±0.12 |
| PrP 6 h | 23.64 | 13.62 | 10.02 | 0.60 | 0.66±0.04 |
| PrP 12 h | 24.01 | 13.20 | 10.81 | 1.01 | 0.49±0.05 |
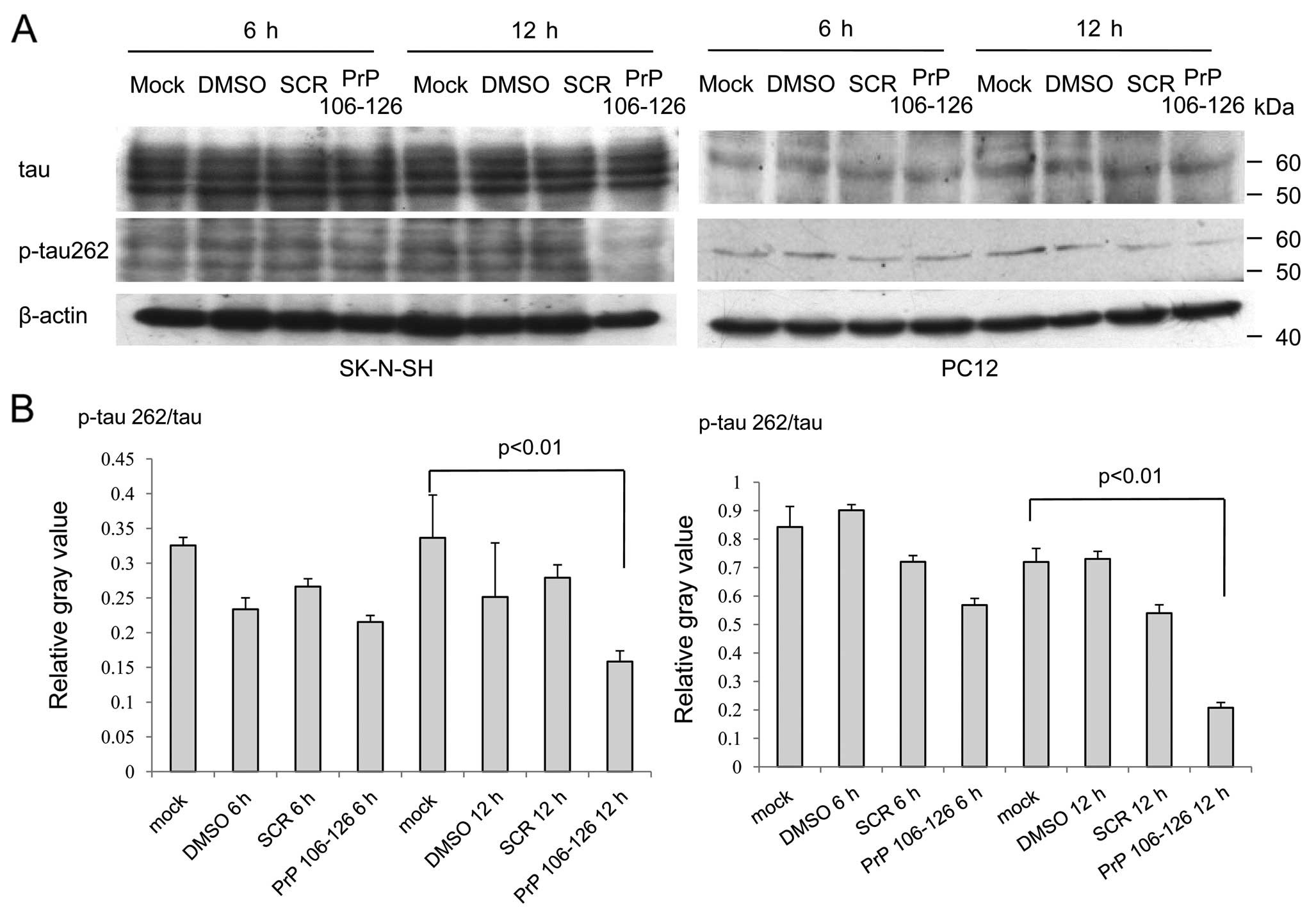
Phosphorylated tau at serine 262 is
downregulated in the PrP106-126-treated cells and in
scrapie-infected hamster brains
Tau is a group of molecular mass proteins of 45–66
kDa with multiple phosphorylation sites. To test whether p-tau262,
which is considered a substrate of MARK4 (26), was influenced as MARK4 decreased,
the lysates of PC12 and SK-N-SH cells treated with PrP106-126 were
tested using western blot analyses with a mAb for total tau (tau13)
and a pAb specific for p-tau at Ser262 [TAU (pS262)]. In SK-N-SH
cells, several bands were detected after staining with mAb tau13
and pAb TAU (pS262). Among them, only the signal of p-tau262 in the
cells after exposure of PrP106-126 for 12 h was markedly weak
(Fig. 6A). In PC12 cells, one
signal band was observed after immunoblotting with mAb tau13 and
pAb TAU (pS262). Similarly, the signal of p-tau262 in the cells
exposed to PrP106-126 for 12 h was significantly weaker compared to
the other cells (Fig. 6A). The
relative gray values of the signals of total tau and p-tau262 from
each reaction were normalized to β-actin. Analyses of the ratios of
the digital data of p-tau262 to that of total tau revealed
significantly lower values (p<0.05) in the preparations treated
with PrP106-126 for 12 h, both in SK-N-SH and PC-12 cells (Fig. 6B). These finding highlight that
treatment of PrP106-126 in cultured cells does not alter the level
of total tau, but induces a reduction of p-tau262.
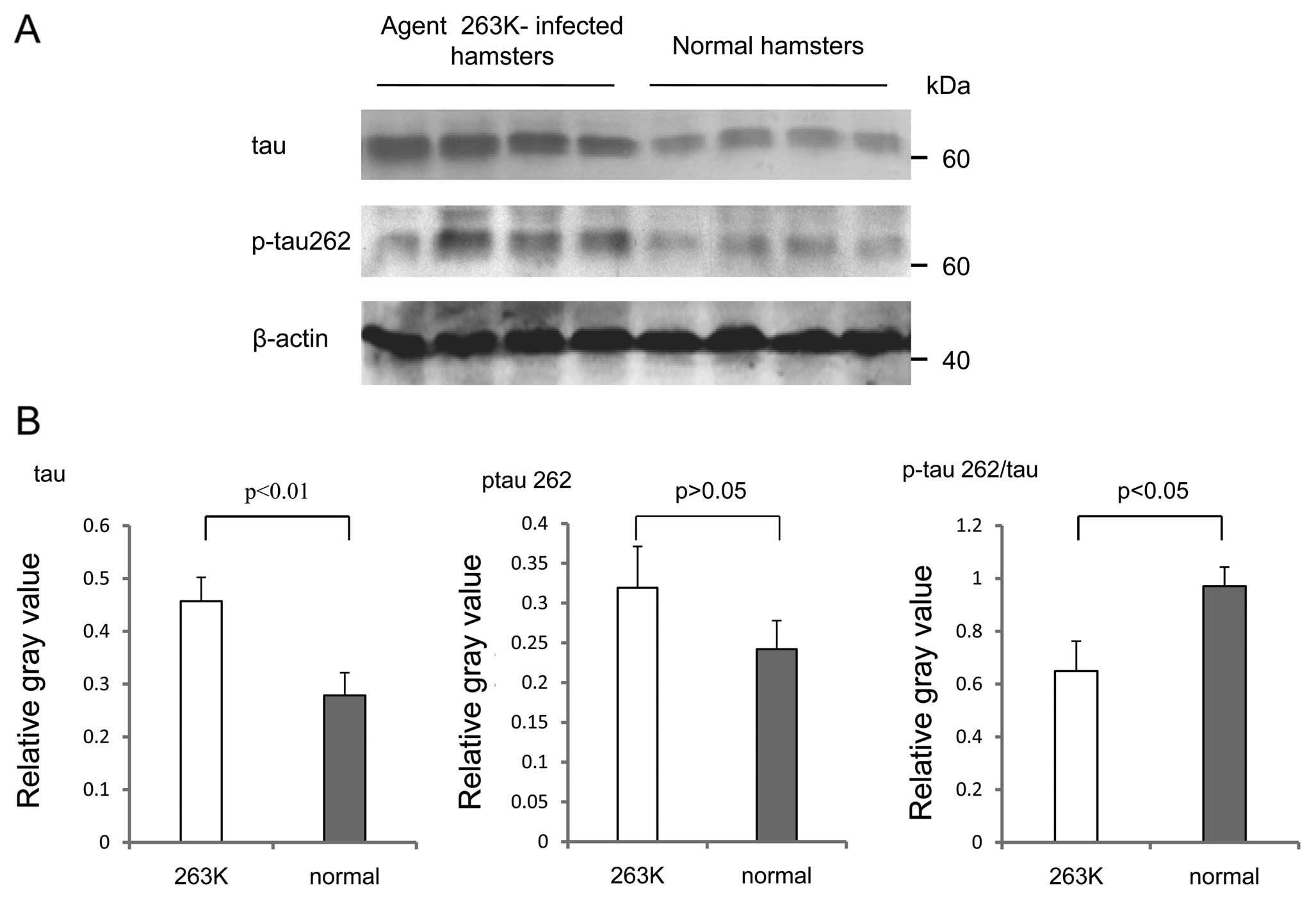
Four brain homogenates from either Ha-263K or normal
hamsters were also analyzed using western blot analyses with the
antibodies against tau and p-tau262. In line with our previous data
(8), the levels of total tau
increased in the brain tissues of Ha-263K, showing a statistically
significant difference in the signal intensity compared with that
of normal controls (p<0.01) (Fig.
7B). The levels of p-tau262 in Ha-263K also increased but were
not statistically significant between the two groups (p>0.05)
(Fig. 7B). Calculations of the
ratios of p-tau262 to total tau identified that the average value
in Ha-263K was clearly lower than that of the normal control
(p<0.05) (Fig. 7B). This
result may suggest that although the amounts of total tau in
scrapie-infected rodents increased at the terminal stage of the
disease, the portion of p-tau262 decreased.
Discussion
A comprehensive sequence analysis of the human
genome (27) revealed that the
human MARK gene family consists of four paralogous genes. MARK4
shares 75% homology with MARK1-3 and is predominantly expressed in
the brain (11).
In differentiated neuroblastoma cells, MARK4 is
localized predominantly at the tips of neurite-like processes and a
tandem affinity-purified MARK4 protein complex contains α-, β- and
γ-tubulin (11). MARK4 is present
in a number of neuronal populations and MARK4-S is upregulated in
human neural progenitor cells (HNPCs) and NTera2 (NT2) cells from
the very early stages of neuronal differentiation, suggesting that
MARK4 is a neuron-specific marker in the CNS (28). Upregulation of MARK4-S during
neuronal differentiation indicates that it may play a specialized
role in neurons.
In our ongoing study on the global gene expression
profiles in the brain tissues of a G114V gCJD patient, we found
that the transcriptional levels of MARK4 are significantly lower
than those of the control (unpublished data). In this study, we
first used both western blot analysis and IHC assays to show that
the MARK4 expression levels are disrupted in the brain tissues of
two different scrapie-infected experimental rodents at the terminal
stages of their diseases. Similarly, MARK4 was hardly detectable in
various brain regions of the G114V gCJD human sample. These
phenomena correlate well with the pathological features of most
human and animal TSEs, representing severe neuron loss.
Our dynamic assays conducted on the brain tissues of
a scrapie 263K experimental hamster, collected over several
incubation periods, showed a clear opposing relationship between
the increase of PrPSc and the decrease of MARK4.
Additionally, the reduction of MARK4 in brain tissues seems to
appear earlier than the presence of detectable PrPSc in
this model. Such phenomena appear to be verified by the
observations in further assays with two different genetic human
prion diseases, in which almost no MARK4 was detectable in the
brain tissues of a G114V gCJD case with PrPSc, but MARK4
could be detected in the brain tissues of a FFI case without
detectable PrPSc. This highlights that the disruption of
MARK4 expression in TSEs closely accompanies the deposits of
PrPSc, which possibly reflects the different situations
of neuron loss among various TSE subtypes.
NSE is a widely used biomarker for neurons. Although
neuronal loss is morphologically observed, due to the lack of
normal human brain tissues, we cannot address the exact status of
neuron damage in CNS of the G114V gCJD and FFI cases at the
terminal stages. However, NSE-specific signals are repeatedly
detected in the brain tissues of G114V gCJD and FFI cases using
western blot analysis and IHC assays (unpublished data). The
intensities of the NSE signals in these two cases differ slightly
in this study, highlighting a possibility that MARK4-positive
neurons are more susceptible and more affected. Astrogliosis is
another hallmark in most human and animal TSEs. Our previous
studies on these two human genetic prion diseases have revealed
abundant gliosis with GFAP-specific IHC assays (20,21). It appears that the astrogliosis in
the brain tissues have little influence on the MARK4
expression.
The influence of PrPSc on the MARK4
expression partially reappears in cultured cells exposed to the
synthetic peptide PrP106-126, which is believed to retain the
ability of PrPSc to aggregate into amyloid-like fibrils
and the tendency to adopt a mostly β-sheet structure (29,30). Along with the cytotoxicity of
PrP106-126, the cellular MARK4 levels drop remarkably. Not
surprisingly, cultured cell lines used in this study possess two
different lengths of MARK4, MARK4-L and MARK4-S, which are
different from the MARK4 profile observed in brain tissues. These
results are in accordance with the distribution of two different
MARK4 splicing isoforms. MARK4-L protein is highly expressed in
cancers, such as hepatocarcinoma, Kato et al (12) and glioma cell lines (14,31), but infrequently in brains. Two
cell lines used in our study are both cancer-derived cells.
Moreover, although PrP106-126 is able to downregulate the levels of
both MARK4 isoforms, it elicits a stronger reduction for the
MARK4-S isoform. As a kinase, MARK4 mediates the phosphorylation on
tau at Ser262. Our data illustrate that PrP106-126 reduces the
cellular MARK4 levels and subsequently reduces the amounts of
p-tau262, but does not change the levels of cellular total tau.
Meanwhile the ratios of p-tau262/total tau in the brain tissues of
scrapie-infected hamsters show downregulation, although the
absolute amounts of p-tau262 and total tau are increased. These
data suggest a positive correlation between MARK4 and p-tau262.
Previously, we discoverd that the tau profile in the
brain tissues of hamsters infected with scrapie strains 263K or
139A show particular changes that are possibly associated with the
alteration of phosphate kinases, in which p-tau at Ser202/Thr205
increased and p-tau at Ser396 and Ser404 decreased, accompanied by
increased CDK5 and reduced GSK3β levels (8). Hyperphosphorylated tau, which can
aggregate into typical paired helical filaments (PHFs), is the main
component of the neurofibrillary tangles found in the brains of
patients with Alzheimer’s disease (32,33). However, the relationship between
toxicity and phosphorylation of tau has not been clearly
illustrated. Microtubule binding activity may play a crucial role
in the regulation of tau toxicity. Abnormality in tau
phosphorylation will induce instability or detachment of the
cytoskeleton.
The reason why the MARK4 expression is decreased in
the terminal stages of prion diseases remains unclear. One
possibility may be due to the mistaken degradation of MARK4. MARK4
belongs to the AMP-activated protein kinase (AMPK) family, which
functions as a metabolic fuel gauge that is activated in response
to environmental stressors needed to restore the cellular energy
balance (34). The AMPK-related
kinases are polyubiquitinated in vivo (35). The polyubiquitination of proteins
is the triggering signal that leads to the degradation of the
protein in the proteasome. It has been reported that MARK4 works as
a partner, showing significant interactions with the human
PrPC interactome in a high-density microarray analysis
(36). One may presume that
during TSE progression, MARK4 interacts with PrPSc and
triggers degradation by the proteasome.
It has been reported that tau inhibits
kinesin-dependent transport of peroxisomes, neurofilaments, and
Golgi-derived vesicles into neurites. In particular, tau inhibits
the transport of an amyloid precursor protein into axons and
dendrites, causing its accumulation in the cell body (37). The expression of tau protein in
CHO cells reveals that both of the microtubule motors, kinesin and
dynein, are inhibited (38).
However, this inhibition can be regulated by MARK. Expression
levels of MARK causes the phosphorylation of MAPs at their KXGS
motifs, thereby detaching MAPs from the microtubules and
facilitating the transport of particles (16). Hence, the repressed level of MARK4
in the brains of prion diseases may block the transportation
through microtubules and accelerate disease progression.
Acknowledgements
This study was supported by the China
Mega-Project for Infectious Disease (2009ZX10004-101,
2008ZX10004-008), the National Basic Research Program of China (973
Program) (2007CB310505), the Chinese National Natural Science
Foundation grant (30800975) and the SKLID Development grant
(2008SKLID102, 2011SKLID204 and 2011SKLID211).
References
|
1.
|
SB PrusinerPrionsProc Natl Acad Sci
USA951336313383199810.1073/pnas.95.23.133639811807
|
|
2.
|
B CaugheyB ChesebroPrion protein and the
transmissible spongiform encephalopathiesTrends Cell
Biol75662199710.1016/S0962-8924(96)10054-417708907
|
|
3.
|
J CastillaP SaaC HetzC SotoIn vitro
generation of infectious scrapie
prionsCell121195206200510.1016/j.cell.2005.02.01115851027
|
|
4.
|
G LegnameIV BaskakovHO NguyenSynthetic
mammalian prionsScience305673676200410.1126/science.1100195
|
|
5.
|
MP McKinleyDC BoltonSB PrusinerA
protease-resistant protein is a structural component of the scrapie
prionCell355762198310.1016/0092-8674(83)90207-66414721
|
|
6.
|
A DesaiTJ MitchisonMicrotubule
polymerization dynamicsAnnu Rev Cell Dev
Biol1383117199710.1146/annurev.cellbio.13.1.83
|
|
7.
|
GG GundersenEvolutionary conservation of
microtubule-capture mechanismsNat Rev Mol Cell
Biol3296304200210.1038/nrm77711994749
|
|
8.
|
GR WangS ShiC GaoChanges of tau profiles
in brains of the hamsters infected with scrapie strains 263 K or
139 A possibly associated with the alteration of phosphate
kinasesBMC Infect Dis1086201010.1186/1471-2334-10-8620356412
|
|
9.
|
CF DongS ShiXF WangThe N-terminus of PrP
is responsible for interacting with tubulin and fCJD related PrP
mutants possess stronger inhibitive effect on microtubule assembly
in vitroArch Biochem
Biophys4708392200810.1016/j.abb.2007.11.00718037369
|
|
10.
|
XL LiGR WangYY JingCytosolic PrP induces
apoptosis of cell by disrupting microtubule assemblyJ Mol
Neurosci43316325201110.1007/s12031-010-9443-920838930
|
|
11.
|
B TrinczekM BrajenovicA EbnethG
DrewesMARK4 is a novel microtubule-associated proteins/microtubule
affinity-regulating kinase that binds to the cellular microtubule
network and to centrosomesJ Biol
Chem27959155923200410.1074/jbc.M304528200
|
|
12.
|
T KatoS SatohH OkabeIsolation of a novel
human gene, MARKL1, homologous to MARK3 and its involvement in
hepatocellular
carcinogenesisNeoplasia349200110.1038/sj.neo.790013211326310
|
|
13.
|
A SchneiderR LaageO von
AhsenIdentification of regulated genes during permanent focal
cerebral ischaemia: characterization of the protein kinase
9b5/MARKL1/MARK4J
Neurochem8811141126200410.1046/j.1471-4159.2003.02228.x
|
|
14.
|
A BeghiniI MagnaniG RoversiThe neural
progenitor-restricted isoform of the MARK4 gene in 19q13.2 is
upregulated in human gliomas and overexpressed in a subset of
glioblastoma cell
linesOncogene2225812591200310.1038/sj.onc.120633612735302
|
|
15.
|
M HasegawaM Morishima-KawashimaK TakioM
SuzukiK TitaniY IharaProtein sequence and mass spectrometric
analyses of tau in the Alzheimer’s disease brainJ Biol
Chem26717047170541992
|
|
16.
|
EM MandelkowE ThiesB TrinczekJ BiernatE
MandelkowMARK/PAR1 kinase is a regulator of microtubule-dependent
transport in axonsJ Cell
Biol16799110200410.1083/jcb.20040108515466480
|
|
17.
|
J ZhangL ChenBY ZhangComparison study on
clinical and neuropathological characteristics of hamsters
inoculated with scrapie strain 263K in different challenging
pathwaysBiomed Environ Sci1765782004
|
|
18.
|
YS KimRI CarpSM CallahanHM
WisniewskiIncubation periods and survival times for mice injected
stereotaxically with three scrapie strains in different brain
regionsJ Gen Virol68695702198710.1099/0022-1317-68-3-6953546594
|
|
19.
|
J YeJ HanQ ShiHuman prion disease with a
G114V mutation and epidemiological studies in a Chinese family: a
case seriesJ Med Case
Rep2331200810.1186/1752-1947-2-33118925969
|
|
20.
|
Q ShiBY ZhangC GaoJ HanGR WangC ChenC
TianXP DongThe pathological and pathogenic characteristics in
various brain regions from a Chinese patient with G114V genetic
CJDNeuropathology325159201210.1111/j.1440-1789.2011.01237.x21732990
|
|
21.
|
XH ShiJ HanJ ZhangClinical,
histopathological and genetic studies in a family with fatal
familial insomniaInfect Genet
Evol10292297201010.1016/j.meegid.2010.01.00720096809
|
|
22.
|
JM ChenC GaoQ ShiDifferent expression
patterns of CK2 subunits in the brains of experimental animals and
patients with transmissible spongiform encephalopathiesArch
Virol15310131020200810.1007/s00705-008-0084-z
|
|
23.
|
G ForloniN AngerettiR ChiesaNeurotoxicity
of a prion protein
fragmentNature362543546199310.1038/362543a08464494
|
|
24.
|
T FlorioS ThellungC AmicoPrion protein
fragment 106–126 induces apoptotic cell death and impairment of
L-type voltage-sensitive calcium channel activity in the GH3 cell
lineJ Neurosci Res543413521998
|
|
25.
|
M EttaicheR PichotJP VincentJ ChabryIn
vivo cytotoxicity of the prion protein fragment 106–126J Biol
Chem27536487364902000
|
|
26.
|
G DrewesB TrinczekS
IllenbergerMicrotubule-associated protein/microtubule
affinity-regulating kinase (p110mark). A novel protein kinase that
regulates tau-microtubule interactions and dynamic instability by
phosphorylation at the Alzheimer-specific site serine 262J Biol
Chem270767976881995
|
|
27.
|
G ManningDB WhyteR MartinezT HunterS
SudarsanamThe protein kinase complement of the human
genomeScience29819121934200210.1126/science.107576212471243
|
|
28.
|
RF MoroniS De BiasiP ColapietroL LarizzaA
BeghiniDistinct expression pattern of microtubule-associated
protein/microtubule affinity-regulating kinase 4 in differentiated
neuronsNeuroscience1438394200610.1016/j.neuroscience.2006.07.052
|
|
29.
|
F TagliaviniF PrelliL VergaSynthetic
peptides homologous to prion protein residues 106–147 form
amyloid-like fibrils in vitroProc Natl Acad Sci
USA90967896821993
|
|
30.
|
YH LiuYL HanJ SongHeat shock protein 104
inhibited the fibrillization of prion peptide 106–126 and
disassembled prion peptide 106–126 fibrils in vitroInt J Biochem
Cell Biol43768774201121296677
|
|
31.
|
I MagnaniC NovielliM BelliniG RoversiL
BelloL LarizzaMultiple localization of endogenous MARK4L protein in
human gliomaCell Oncol31357370200919759416
|
|
32.
|
RD TerryThe pathogenesis of Alzheimer
disease: an alternative to the amyloid hypothesisJ Neuropathol Exp
Neurol5510231025199610.1097/00005072-199655100-000018857998
|
|
33.
|
EM MandelkowJ BiernatG DrewesN GustkeB
TrinczekE MandelkowTau domains, phosphorylation, and interactions
with microtubulesNeurobiol
Aging16355363199510.1016/0197-4580(95)00025-A7566345
|
|
34.
|
M ZunguJC SchislerMF EssopC McCuddenC
PattersonMS WillisRegulation of AMPK by the ubiquitin proteasome
systemAm J Pathol178411201110.1016/j.ajpath.2010.11.03021224036
|
|
35.
|
AK Al-HakimA ZagorskaL ChapmanM DeakM
PeggieDR AlessiControl of AMPK-related kinases by USP9X and
atypical Lys(29)/Lys(33)-linked polyubiquitin chainsBiochem
J411249260200810.1042/BJ2008006718254724
|
|
36.
|
J SatohS ObayashiT MisawaK SumiyoshiK
OosumiH TabunokiProtein microarray analysis identifies human
cellular prion protein interactorsNeuropathol Appl
Neurobiol351635200910.1111/j.1365-2990.2008.00947.x18482256
|
|
37.
|
K StamerR VogelE ThiesE MandelkowEM
MandelkowTau blocks traffic of organelles, neurofilaments, and APP
vesicles in neurons and enhances oxidative stressJ Cell
Biol15610511063200210.1083/jcb.20010805711901170
|
|
38.
|
A EbnethR GodemannK StamerS IllenbergerB
TrinczekE MandelkowOverexpression of tau protein inhibits
kinesin-dependent trafficking of vesicles, mitochondria, and
endoplasmic reticulum: implications for Alzheimer’s diseaseJ Cell
Biol14377779419989813097
|