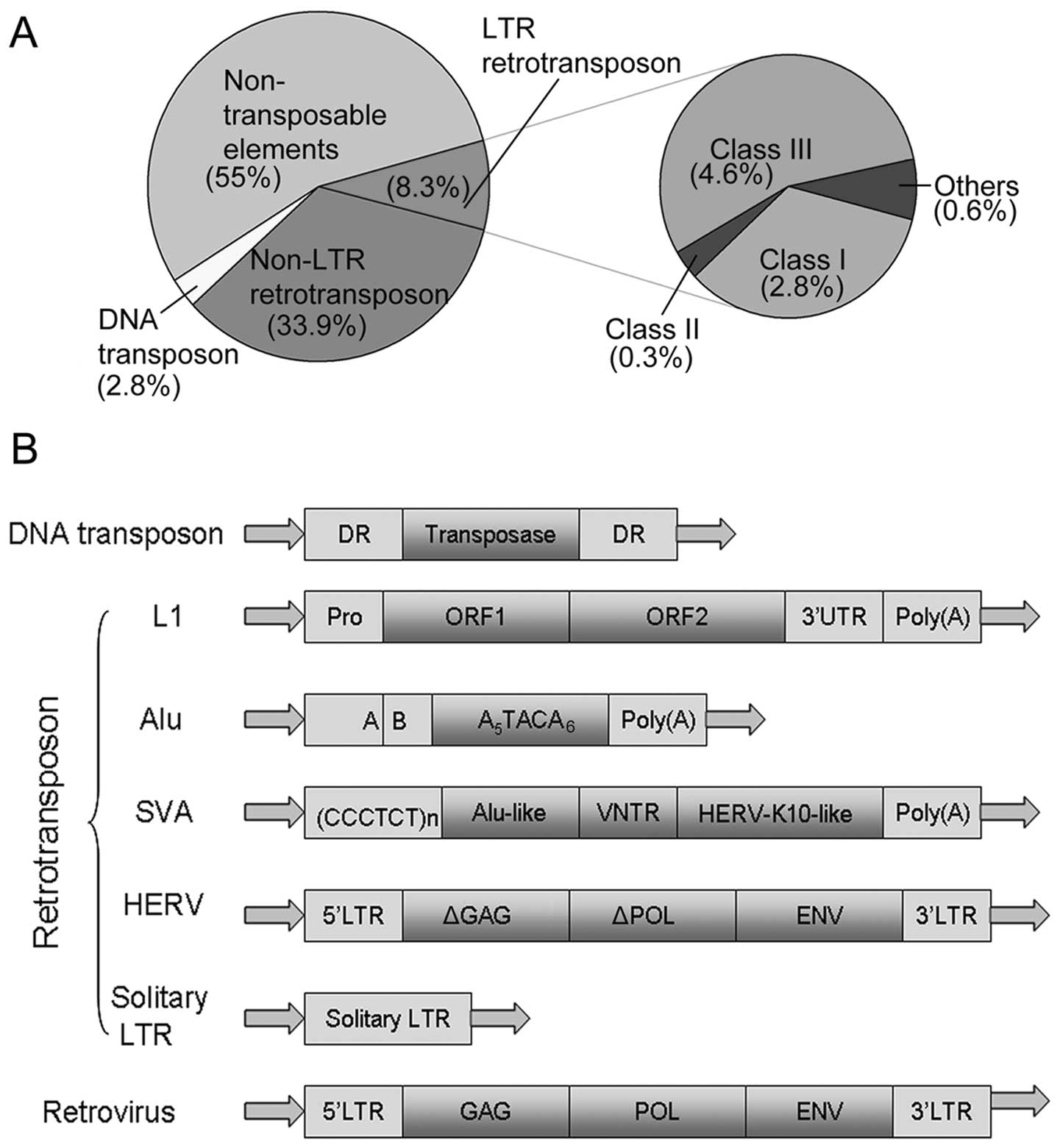
Although the majority of HERV genes are highly
defective with large deletions, stop codons and frameshifts in the
open reading fames, HERV LTR still retain their functions, such as
promoter, enhancer and transcriptional factor-binding site and
potentially regulate their neighboring viral and cellular genes.
Non-LTR retrotransposon has been reported to cause human cancer
through insertional mutagenesis of genes,
retrotransposition-associated mutagenesis, non-allelic homologous
recombination and hypomethylation of retroelement promoters
(8). Previous studies have
reported that LTR elements affected adjacent genes to contribute to
cancer (9–11). However, the molecular mechanism of
LTR in tumorigenesis has yet to be reported. Herein, we briefly
review the characteristics of HERV LTRs, their distribution, as
well as the available evidence for LTR biological significance and
function in physiological and pathological processes. The focus of
this review is on the role of HERV LTR in human tumorigenesis.
HERV LTR elements are retained in the vicinity of
genes in order to regulate their expression during primate
evolution. LTR elements and nearby genes appear to have forged a
mutually beneficial relationship. LTR elements can benefit from
genes by surviving from one generation to the next, while genes can
benefit from the characteristics of LTR. For example, genes could
also obtain their enhancer (12),
promoter (sometimes bidirectional) (12–16), polyadenylation [poly(A)] sites
(17,18) from LTR. Alteration of the
expression of adjacent genes usually results from LTR. For example,
an explanation of LTR promoter activity involves the expression of
amylase in the human parotid glands, where integration of HERV-E in
reverse orientation upstream of the pancreatic amylase gene
promotes its expression and release into saliva (19). In addition, bidirectional promoter
activity is observed in HERV-K LTR for the neighboring reporter
gene (20). As an alternative
promoter LTR is capable of enhancing the transcription from a
native promoter of apolipoprotein CI and the endothelin B receptor
genes (13). HERV-K LTR revealed
its tissue-specific enhancer activity (21). LTR may lead to polyadenylation of
spliced chromosomal transcripts in human genes (HHLA2 and HHLA3) as
it contains a poly(A) sequence (17). LTR can provide alternative and
aberrant sites for transcript splicing and encode an additional
carboxy-terminal sequence in the human leptin receptor (22). Similarly, LTR acts as one of at
least two alternative promoters for the human
β1,3-galactosyltransferase 5 gene and is the dominant promoter in
the colon (15). These functions
of LTR are essential for LTR to regulate adjacent gene
expression.
The most abundant transcriptional activation of HERV
LTR may be observed in the placenta and embryonic tissues. Early
studies of LTR transcript in placenta emphasized the expression of
primary placenta of LTR of different HERV families [ERV3 (23); HERV-F (24); HERV-W (25,26)]. For example, 5′LTR regulates the
expression of its downstream viral env gene in placenta trophoblast
(26), while the ERV-9 regulatory
region acts as a promoter to form a novel transcript of p63 in
order to replenish cutaneous epithelial stem cells and maintain the
fidelity of the female germ line (27). Further examples include the
placenta-specific and tissue-specific expression of adjacent genes
driven by LTR (28). These
findings suggest that HERV LTR expression in human placenta is
frequently active in human physical biology.
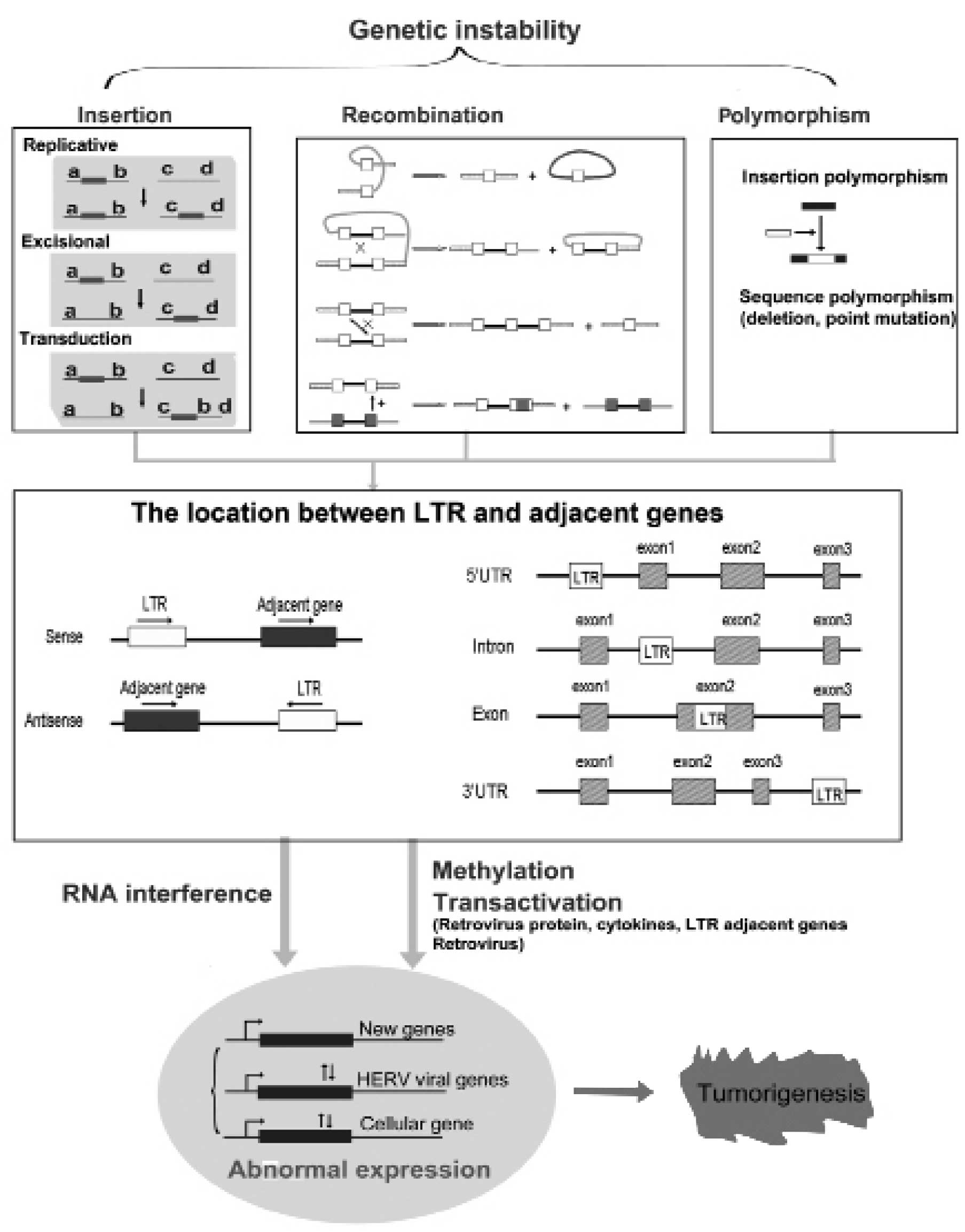
The role of HERV LTR in tumorigenesis as well as the
possible mechanisms of how HERV LTR contributes to the development
of human cancer was investigated. An outline of potential
mechanisms of HERV LTR in human tumorigenesis is provided in
Fig. 2. The four aspects of
genetic instability, methylation, transactivation and RNA
interference by antisense transcripts are discussed below.
Genetic instability is one of the key features
associated with cancer causation and progression (40,41). Instability exists at two distinct
levels. In a small subset of tumors, instability is observed at the
nucleotide level and results in base substitutions, deletions or
insertions of a few nucleotides, such as polymorphisms. In the
majority of other cancers, instability is observed at the
chromosome level, resulting in losses and gains of whole
chromosomes or large portions, for example insertion mutation and
recombination. As a mobile element HERV LTR possesses the feature
of genetic instability. Therefore, HERV LTR affects human genome
instability involvement in tumorigenesis via three principal
mechanisms: insertion, recombination and polymorphism (Fig. 2).
Retroviruses mediate malignant transformation via
insertional mutagenesis or expression of viral genes. Similarly,
HERV is also involved in human tumorigenesis via the mechanism of
insertion (42). HERV LTR
amplifies in the human genome by generating new integration events.
The amplification of HERV integration in the human genome consists
of three processes: replication, excision and transduction
(Fig. 2). Insertions are
therefore considered beneficial, negative or neutral. HERV LTR
elements insert into genomic regions and contain crucial biological
functions, resulting in insertional mutagenesis during evolution.
Abnormal expression of novel gene and adjacent gene are also caused
by the insertion of LTR. Among the examples of human cancers caused
by LTR-mediated insertion mutagenesis, the expression of PTN gene
contributes to the highly aggressive growth of human
choriocarcinoma (42).
LTR-associated insertional mutagenesis can contribute to cancer via
germ-line and somatic mutations, either of which can directly lead
to the onset of malignant transformation.
Solitary LTR is formed by homologous recombination
between two full-length HERV families. A variety of different
recombination products are shown in Fig. 2 (43), including recombination between the
two LTRs of a single provirus, homologous recombination between two
HERV on the same chromosome, recombination between the 3′ and
5′LTRs of one HERV and gene conversion between the non-homologous
genes. Homologous recombination between two proviruses results in
substantial deletion and rearrangements of cellular DNA. Gene
conversion leads to non-homologous gene exchange with no proviral
loss. This occurs predominantly through contributions to
recombination, which is frequently detected in cancer (44). LTR33 and LTR7 are formed by
recombination and can regulate DNAJC15 gene expression in cancer
cells (38). Therefore, LTR
recombination may be crucial for LTR to be involved in cancer
progression.
The existence of polymorphism provides one
explanation of how a ubiquitous gene causes disease in only a
proportion of individuals. The polymorphism is subdivided into two
broad categories: sequence and insertional polymorphisms. Only a
few sequence polymorphisms have been described, partly due to the
difficulty in identifying them against the background of closely
similar proviruses. Using specific PCR primers spanning 5′LTRs of
K115 and K113, Jha et al (45) reported the presence of three
single nucleotide polymorphism sites in the K113 5′LTR and four in
the K115 5′LTR that together constituted four haplotypes for K113.
At present, there is little convincing evidence that any sequence
polymorphism of a HERV provides susceptibility to human disease.
For insertion polymorphisms, although most HERV families became
integrated into the human genome millions of years ago, a new class
of insertionally polymorphic HERV-K family members has recently
been described (46,47). HERV-K113 and HER-K115 insertion
has been reported to be involved in various types of cancer
(48–51). New insertion polymorphism of HERVs
could serve as novel genetic risk factors and thus provide new
insight for research into HERV LTR and cancer.
Genetic instability of LTR leads to the its wide
distribution in the human genome, while the activation of LTR can
be controlled by DNA methylation. For example, Gimenez et al
(52) have recently reported that
hypomethylation of the promoter domain of the HERV U3 element
appears to be a prerequisite for the increased expression in tumor
tissues compared to normal tissues. Colon cancer cells are treated
with DNA methylation and histone deacetylase inhibitors, and RT-PCR
results show that the expression patterns of HERV-H are
significantly altered in several colon cancer cells. The finding
suggests that the hypomethylation context affects the expression of
HERV-H elements in colon cancer cells (53). Increased HERV-K expression in
melanomas may be due to increased promoter activity and
demethylation of the 5′LTR (54).
Thus, overexpression of the HERV sequence in cell lines is
correlated with the demethylation of LTR.
HERV and LTR are usually oriented opposite to the
transcription direction of corresponding host genes (55–57). Antisense transcripts affect the
sense partner gene function by modifying the transcriptional and
post-transcriptional regulation processes. Findings of a previous
study (55) demonstrated that
from 10 HERV-K LTRs which were localized in introns of unique human
genes, nine exhibited opposite orientation to the transcription
direction of the corresponding human genes. A hypothesis was
propounded that LTR affects the gene expression by initiation of
the antisense RNA synthesis (55). Subsequently, several reports
supported the hypothesis (57–61). Antisense transcript of HERV-Ec1
affected the expression of cytosolic phospholipase A2 in urothelial
carcinoma (57). A novel exon
cassette is derived from the antisense transcript of the HERV-K
element (58). Intronic RNAs
arising from U3 of ERV-9 LTR are expressed as both sense and
antisense transcripts, with the antisense transcript being
expressed at higher levels compared to the sense expression in
malignant cells (59). LTR from
exogenous retrovirus HTLV can also generate antisense manuscripts
such as the HTLV-1 basic leucine zipper factor (HBZ) (60). It has been reported that HBZ is
consistently expressed and remains intact in all ATL cases and
HTLV-1-infected individuals, where it promotes cell proliferation
(61). Antisense transcripts are
able to form double-stranded RNA and may recruit RNAi machinery.
The abovementioned studies suggest that the abnormal expression of
antisense transcripts of LTR retrotransposon may be a causative
factor for tumorigenesis.
HERV LTR could also be reactivated by environmental
factors including cytokines (62), radiation (63,64) and proteins of exogenous
retroviruses (65,66). Radiation induced the epigenetic
regulation of HERV-R 5′LTR and upregulated HERV-R env expression
(63). HSV-1 activated the LTR
activity of HERV-W to enhance their potential oligodendrotoxic and
immunopathogenic effects (67).
HSV-1 infection also induced the LTR-directed transcription of the
HERV-K. HSV-1 immediate-early ICP0 protein was able to upregulate
the activity of HERV-K LTR (66).
Relative levels of transcripts encoding HERV-W elements and
cellular genes are transactivated by viral infection in different
cell lines by regulating the transcriptional activity (68). Several HERV LTRs could be
activated by HTLV Tax and were involved in diseases (65). High-level expression of HERV-K has
been demonstrated to be activated by the MITF-M gene in melanomas,
breast cancers and teratocarcinomas (69). Of note, these factors can also
target ERVs, for example, a HERV-K (HML-2) consensus element can be
inhibited by APOBEC3F (70). Such
LTR reactivation can enhance its impact on the surrounding genes in
the pathological process of human cancer.
In summary, cumulative evidences indicate that LTR
may be involved in the process of tumorigenesis at various levels.
The potential role of LTR in human cancer appears much more complex
(Fig. 2). LTR can be involved in
tumorigenesis in four distinct ways, i.e., genetic instability,
hypomethylation, transactivation and RNA interference. Insertional
mutagenesis, recombination and polymorphism of LTR have been found
to contribute to its genetic instability. Genetic instability can
lead to diverse distribution and high copy numbers of LTR in the
human genome. LTR can exhibit a sense or antisense orientation of
their neighboring genes. By contrast, LTR can also be located in
the 5′UTR, intron, exon and 3′UTR region of their neighboring
genes. Genome-wide hypomethylation, transactivation of LTR and RNA
interference by antisense orientation of LTR can regulate the
expression of human cellular, viral and novel genes. Abnormal
expression of cancer-associated genes may also contribute to
tumorigenesis. Thus, identification of the distribution, structure
and functional characteristics of LTR is extremely important to
elucidate the mechanism involved in LTR regulation of
cancer-associated gene abnormal expression in the process of
tumorigenesis and development. Given the strong evidence for the
abnormal expression of LTR adjacent genes in human tumors, it can
be hypothesized that similar unknown genes or other
tumor-associated genes may also affect various types of human
cancer. Although a causative role of HERV LTR in human tumors has
not been reported, HERV LTR is a potential contributory factor in
various types of human cancer. Further investigations should
include, for example, the novel insertion of HERV LTR in the human
genome, RNA interference by antisense transcript of LTR,
distribution of HERV LTR in different human chromosomes and the
identification of LTR-related tumor genes.
|
1
|
Bannert N and Kurth R: Retroelements and
the human genome: new perspectives on an old relation. Proc Natl
Acad Sci USA. 101(Suppl 2): S14572–S14579. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
de Parseval N and Heidmann T: Human
endogenous retroviruses: from infectious elements to human genes.
Cytogenet Genome Res. 110:318–332. 2005.PubMed/NCBI
|
|
3
|
Leib-Mosch C, Haltmeier M, Werner T, et
al: Genomic distribution and transcription of solitary HERV-K LTRs.
Genomics. 18:261–269. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tonjes RR, Lower R, Boller K, et al:
HERV-K: the biologically most active human endogenous retrovirus
family. J Acquir Immune Defic Syndr Hum Retrovirol. 13(Suppl 1):
S261–S267. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Glazkova DV, Nadezhdin EV, Vinogradova TV,
et al: Nucleotide sequences of long terminal repeats of the human
endogenous retrovirus (LTR HERV-K) on the short arm of chromosome
7: identification, analysis and evaluation of transcriptional
activity. Genetika. 39:702–708. 2003.(In Russian).
|
|
6
|
Kurdyukov SG, Lebedev YB, Artamonova II,
et al: Full-sized HERV-K (HML-2) human endogenous retroviral LTR
sequences on human chromosome 21: map locations and evolutionary
history. Gene. 273:51–61. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Smit AF: Interspersed repeats and other
mementos of transposable elements in mammalian genomes. Curr Opin
Genet Dev. 9:657–663. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Belancio VP, Roy-Engel AM and Deininger
PL: All y’all need to know ‘bout retroelements in cancer. Semin
Cancer Biol. 20:200–210. 2010.
|
|
9
|
Lamprecht B, Walter K, Kreher S, et al:
Derepression of an endogenous long terminal repeat activates the
CSF1R proto-oncogene in human lymphoma. Nat Med. 16:571–579. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Papadimitriou E, Mikelis C, Lampropoulou
E, et al: Roles of pleiotrophin in tumor growth and angiogenesis.
Eur Cytokine Netw. 20:180–190. 2009.PubMed/NCBI
|
|
11
|
Sin HS, Huh JW, Kim DS, et al:
Transcriptional control of the HERV-H LTR element of the GSDML gene
in human tissues and cancer cells. Arch Virol. 151:1985–1994. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Feuchter A and Mager D: Functional
heterogeneity of a large family of human LTR-like promoters and
enhancers. Nucleic Acids Res. 18:1261–1270. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Medstrand P, Landry JR and Mager DL: Long
terminal repeats are used as alternative promoters for the
endothelin B receptor and apolipoprotein C-I genes in humans. J
Biol Chem. 276:1896–1903. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Dunn CA and Mager DL: Transcription of the
human and rodent SPAM1/PH-20 genes initiates within an ancient
endogenous retrovirus. BMC Genomics. 6:472005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Dunn CA, Medstrand P and Mager DL: An
endogenous retroviral long terminal repeat is the dominant promoter
for human beta1,3-galactosyltransferase 5 in the colon. Proc Natl
Acad Sci USA. 100:12841–12846. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Dunn CA, van de Lagemaat LN, Baillie GJ
and Mager DL: Endogenous retrovirus long terminal repeats as
ready-to-use mobile promoters: the case of primate beta3GAL-T5.
Gene. 364:2–12. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mager DL, Hunter DG, Schertzer M and
Freeman JD: Endogenous retroviruses provide the primary
polyadenylation signal for two new human genes (HHLA2 and HHLA3).
Genomics. 59:255–263. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Baust C, Seifarth W, Germaier H, Hehlmann
R and Leib-Mosch C: HERV-K-T47D-Related long terminal repeats
mediate polyadenylation of cellular transcripts. Genomics.
66:98–103. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Samuelson LC, Wiebauer K, Snow CM and
Meisler MH: Retroviral and pseudogene insertion sites reveal the
lineage of human salivary and pancreatic amylase genes from a
single gene during primate evolution. Mol Cell Biol. 10:2513–2520.
1990.PubMed/NCBI
|
|
20
|
Domansky AN, Kopantzev EP, Snezhkov EV,
Lebedev YB, Leib-Mosch C and Sverdlov ED: Solitary HERV-K LTRs
possess bi-directional promoter activity and contain a negative
regulatory element in the U5 region. FEBS Lett. 472:191–195. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ruda VM, Akopov SB, Trubetskoy DO, et al:
Tissue specificity of enhancer and promoter activities of a
HERV-K(HML-2) LTR. Virus Res. 104:11–16. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kapitonov VV and Jurka J: The long
terminal repeat of an endogenous retrovirus induces alternative
splicing and encodes an additional carboxy-terminal sequence in the
human leptin receptor. J Mol Evol. 48:248–251. 1999. View Article : Google Scholar
|
|
23
|
Kato N, Pfeifer-Ohlsson S, Kato M, Larsson
E, Rydnert J, Ohlsson R and Cohen M: Tissue-specific expression of
human provirus ERV3 mRNA in human placenta: two of the three ERV3
mRNAs contain human cellular sequences. J Virol. 61:2182–2191.
1987.PubMed/NCBI
|
|
24
|
Kjellman C, Sjogren HO, Salford LG and
Widegren B: HERV-F (XA34) is a full-length human endogenous
retrovirus expressed in placental and fetal tissues. Gene.
239:99–107. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Mallet F, Bouton O, Prudhomme S, et al:
The endogenous retroviral locus ERVWE1 is a bona fide gene involved
in hominoid placental physiology. Proc Natl Acad Sci USA.
101:1731–1736. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Prudhomme S, Oriol G and Mallet F: A
retroviral promoter and a cellular enhancer define a bipartite
element which controls env ERVWE1 placental expression. J Virol.
78:12157–12168. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Liu M and Eiden MV: Role of human
endogenous retroviral long terminal repeats (LTRs) in maintaining
the integrity of the human germ line. Viruses. 3:901–905. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Landry JR and Mager DL: Functional
analysis of the endogenous retroviral promoter of the human
endothelin B receptor gene. J Virol. 77:7459–7466. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Seidl C, Donner H, Petershofen E, Usadel
KH, Seifried E, Kaltwasser JP and Badenhoop K: An endogenous
retroviral long terminal repeat at the HLA-DQB1 gene locus confers
susceptibility to rheumatoid arthritis. Hum Immunol. 60:63–68.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Pascual M, Martin J, Nieto A, Giphart MJ,
van der Slik AR, de Vries RR and Zanelli E: Distribution of
HERV-LTR elements in the 5′-flanking region of HLA-DQB1 and
association with autoimmunity. Immunogenetics. 53:114–118.
2001.PubMed/NCBI
|
|
31
|
Hegyi H: GABBR1 has a HERV-W LTR in its
regulatory region - a possible implication for schizophrenia. Biol
Direct. 8:52013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kamp C, Hirschmann P, Voss H, Huellen K
and Vogt PH: Two long homologous retroviral sequence blocks in
proximal Yq11 cause AZFa microdeletions as a result of
intrachromosomal recombination events. Hum Mol Genet. 9:2563–2572.
2000. View Article : Google Scholar
|
|
33
|
Goering W, Ribarska T and Schulz WA:
Selective changes of retroelement expression in human prostate
cancer. Carcinogenesis. 32:1484–1492. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Armbruester V, Sauter M, Krautkraemer E,
et al: A novel gene from the human endogenous retrovirus K
expressed in transformed cells. Clin Cancer Res. 8:1800–1807.
2002.PubMed/NCBI
|
|
35
|
Chen T, Meng Z, Gan Y, et al: The viral
oncogene Np9 acts as a critical molecular switch for co-activating
beta-catenin, ERK, Akt and Notch1 and promoting the growth of human
leukemia stem/progenitor cells. Leukemia. Jan 11–2013.(Epub ahead
of print). View Article : Google Scholar
|
|
36
|
Boese A, Sauter M, Galli U, et al: Human
endogenous retrovirus protein cORF supports cell transformation and
associates with the promyelocytic leukemia zinc finger protein.
Oncogene. 19:4328–4336. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Singh S, Kaye S, Francis N, Peston D, Gore
M, McClure M and Bunker C: Human endogenous retrovirus K (HERV-K)
rec mRNA is expressed in primary melanoma but not in benign naevi
or normal skin. Pigment Cell Melanoma Res. 26:426–428. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Sin HS, Huh JW, Kim DS, et al: Endogenous
retrovirus-related sequences provide an alternative transcript of
MCJ genes in human tissues and cancer cells. Genes Genet Syst.
81:333–339. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Romanish MT, Lock WM, van de Lagemaat LN,
Dunn CA and Mager DL: Repeated recruitment of LTR retrotransposons
as promoters by the anti-apoptotic locus NAIP during mammalian
evolution. PLoS Genet. 3:e102007. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Lengauer C, Kinzler KW and Vogelstein B:
Genetic instabilities in human cancers. Nature. 396:643–649. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Lin J, Gan CM, Zhang X, et al: A
multidimensional analysis of genes mutated in breast and colorectal
cancers. Genome Res. 17:1304–1318. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Schulte AM, Lai S, Kurtz A, Czubayko F,
Riegel AT and Wellstein A: Human trophoblast and choriocarcinoma
expression of the growth factor pleiotrophin attributable to
germ-line insertion of an endogenous retrovirus. Proc Natl Acad Sci
USA. 93:14759–14764. 1996. View Article : Google Scholar
|
|
43
|
Stoye JP: Endogenous retroviruses: still
active after all these years? Curr Biol. 11:R914–R916.
2001.PubMed/NCBI
|
|
44
|
Romanish MT, Cohen CJ and Mager DL:
Potential mechanisms of endogenous retroviral-mediated genomic
instability in human cancer. Semin Cancer Biol. 20:246–253. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Jha AR, Pillai SK, York VA, et al:
Cross-sectional dating of novel haplotypes of HERV-K 113 and HERV-K
115 indicate these proviruses originated in Africa before Homo
sapiens. Mol Biol Evol. 26:2617–2626. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Turner G, Barbulescu M, Su M,
Jensen-Seaman MI, Kidd KK and Lenz J: Insertional polymorphisms of
full-length endogenous retroviruses in humans. Curr Biol.
11:1531–1535. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Dangel AW, Mendoza AR, Baker BJ, Daniel
CM, Carroll MC, Wu LC and Yu CY: The dichotomous size variation of
human complement C4 genes is mediated by a novel family of
endogenous retroviruses, which also establishes species-specific
genomic patterns among Old World primates. Immunogenetics.
40:425–436. 1994. View Article : Google Scholar
|
|
48
|
Burmeister T, Ebert AD, Pritze W,
Loddenkemper C, Schwartz S and Thiel E: Insertional polymorphisms
of endogenous HERV-K113 and HERV-K115 retroviruses in breast cancer
patients and age-matched controls. AIDS Res Hum Retroviruses.
20:1223–1229. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Wang-Johanning F, Radvanyi L, Rycaj K, et
al: Human endogenous retrovirus K triggers an antigen-specific
immune response in breast cancer patients. Cancer Res.
68:5869–5877. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Contreras-Galindo R, Kaplan MH, Leissner
P, et al: Human endogenous retrovirus K (HML-2) elements in the
plasma of people with lymphoma and breast cancer. J Virol.
82:9329–9336. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Golan M, Hizi A, Resau JH, Yaal-Hahoshen
N, Reichman H, Keydar I and Tsarfaty I: Human endogenous retrovirus
(HERV-K) reverse transcriptase as a breast cancer prognostic
marker. Neoplasia. 10:521–533. 2008.PubMed/NCBI
|
|
52
|
Gimenez J, Montgiraud C, Pichon JP, et al:
Custom human endogenous retroviruses dedicated microarray
identifies self-induced HERV-W family elements reactivated in
testicular cancer upon methylation control. Nucleic Acids Res.
38:2229–2246. 2010. View Article : Google Scholar
|
|
53
|
Liang Q, Xu Z, Xu R, Wu L and Zheng S:
Expression patterns of non-coding spliced transcripts from human
endogenous retrovirus HERV-H elements in colon cancer. PLoS One.
7:e299502012. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Stengel S, Fiebig U, Kurth R and Denner J:
Regulation of human endogenous retrovirus-K expression in melanomas
by CpG methylation. Genes Chromosomes Cancer. 49:401–411. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Buzdin AA, Lebedev IuB and Sverdlov ED:
Human genome-specific HERV-K intron LTR genes have a random
orientation relative to the direction of transcription, and,
possibly, participated in antisense gene expression regulation.
Bioorg Khim. 29:103–106. 2003.(In Russian).
|
|
56
|
Li F, Nellaker C, Yolken RH and Karlsson
H: A systematic evaluation of expression of HERV-W elements;
influence of genomic context, viral structure and orientation. BMC
Genomics. 12:222011. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Gosenca D, Gabriel U, Steidler A, et al:
HERV-E-mediated modulation of PLA2G4A transcription in urothelial
carcinoma. PLoS One. 7:e493412012. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Kim DS and Hahn Y: Human-specific
antisense transcripts induced by the insertion of transposable
element. Int J Mol Med. 26:151–157. 2010.PubMed/NCBI
|
|
59
|
Xu L, Elkahloun AG, Candotti F, et al: A
novel function of RNAs arising from the long terminal repeat of
human endogenous retrovirus 9 in cell cycle arrest. J Virol.
87:25–36. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Gaudray G, Gachon F, Basbous J,
Biard-Piechaczyk M, Devaux C and Mesnard JM: The complementary
strand of the human T-cell leukemia virus type 1 RNA genome encodes
a bZIP transcription factor that down-regulates viral
transcription. J Virol. 76:12813–12822. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Arnold J, Zimmerman B, Li M, Lairmore MD
and Green PL: Human T-cell leukemia virus type-1 antisense-encoded
gene, Hbz, promotes T-lymphocyte proliferation. Blood.
112:3788–3797. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Katsumata K, Ikeda H, Sato M, et al:
Cytokine regulation of env gene expression of human endogenous
retrovirus-R in human vascular endothelial cells. Clin Immunol.
93:75–80. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Lee JR, Ahn K, Kim YJ, Jung YD and Kim HS:
Radiation-induced human endogenous retrovirus (HERV)-R env gene
expression by epigenetic control. Radiat Res. 178:379–384. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Reiche J, Pauli G and Ellerbrok H:
Differential expression of human endogenous retrovirus K
transcripts in primary human melanocytes and melanoma cell lines
after UV irradiation. Melanoma Res. 20:435–440. 2010.PubMed/NCBI
|
|
65
|
Toufaily C, Landry S, Leib-Mosch C,
Rassart E and Barbeau B: Activation of LTRs from different human
endogenous retrovirus (HERV) families by the HTLV-1 tax protein and
T-cell activators. Viruses. 3:2146–2159. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Kwun HJ, Han HJ, Lee WJ, Kim HS and Jang
KL: Transactivation of the human endogenous retrovirus K long
terminal repeat by herpes simplex virus type 1 immediate early
protein 0. Virus Res. 86:93–100. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Ruprecht K, Obojes K, Wengel V, et al:
Regulation of human endogenous retrovirus W protein expression by
herpes simplex virus type 1: implications for multiple sclerosis. J
Neurovirol. 12:65–71. 2006. View Article : Google Scholar
|
|
68
|
Nellaker C, Yao Y, Jones-Brando L, Mallet
F, Yolken RH and Karlsson H: Transactivation of elements in the
human endogenous retrovirus W family by viral infection.
Retrovirology. 3:442006. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Katoh I, Mirova A, Kurata S, et al:
Activation of the long terminal repeat of human endogenous
retrovirus K by melanoma-specific transcription factor MITF-M.
Neoplasia. 13:1081–1092. 2011.PubMed/NCBI
|
|
70
|
Lee YN and Bieniasz PD: Reconstitution of
an infectious human endogenous retrovirus. PLoS Pathog. 3:e102007.
View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Lee JW and Kim HS: Endogenous retrovirus
HERV-I LTR family in primates: sequences, phylogeny, and evolution.
Arch Virol. 151:1651–1658. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Yi JM and Kim HS: Evolutionary implication
of human endogenous retrovirus HERV-H family. J Hum Genet.
49:215–219. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Mager DL and Henthorn PS: Identification
of a retrovirus-like repetitive element in human DNA. Proc Natl
Acad Sci USA. 81:7510–7514. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Blond JL, Beseme F, Duret L, et al:
Molecular characterization and placental expression of HERV-W, a
new human endogenous retrovirus family. J Virol. 73:1175–1185.
1999.PubMed/NCBI
|
|
75
|
Gifford R and Tristem M: The evolution,
distribution and diversity of endogenous retroviruses. Virus Genes.
26:291–315. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Strazzullo M, Parisi T, Di Cristofano A,
Rocchi M and La Mantia G: Characterization and genomic mapping of
chimeric ERV9 endogenous retroviruses-host gene transcripts. Gene.
206:77–83. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Tonjes RR, Czauderna F and Kurth R:
Genome-wide screening, cloning, chromosomal assignment, and
expression of full-length human endogenous retrovirus type K. J
Virol. 73:9187–9195. 1999.PubMed/NCBI
|
|
78
|
Wang-Johanning F, Frost AR, Jian B, Epp L,
Lu DW and Johanning GL: Quantitation of HERV-K env gene expression
and splicing in human breast cancer. Oncogene. 22:1528–1535. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Herbst H, Sauter M and Mueller-Lantzsch N:
Expression of human endogenous retrovirus K elements in germ cell
and trophoblastic tumors. Am J Pathol. 149:1727–1735.
1996.PubMed/NCBI
|
|
80
|
Huh JW, Kim DS, Kang DW, et al:
Transcriptional regulation of GSDML gene by antisense-oriented
HERV-H LTR element. Arch Virol. 153:1201–1205. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Sun Q, Yang J, Xing G, Sun Q, Zhang L and
He F: Expression of GSDML associates with tumor progression in
uterine cervix cancer. Transl Oncol. 1:73–83. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Backman SA, Ghazarian D, So K, et al:
Early onset of neoplasia in the prostate and skin of mice with
tissue-specific deletion of Pten. Proc Natl Acad Sci USA.
101:1725–1730. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Tsirmoula S, Dimas K, Hatziapostolou M,
Lamprou M, Ravazoula P and Papadimitriou E: Implications of
pleiotrophin in human PC3 prostate cancer cell growth in vivo.
Cancer Sci. 103:1826–1832. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Landry JR, Rouhi A, Medstrand P and Mager
DL: The Opitz syndrome gene Mid1 is transcribed from a human
endogenous retroviral promoter. Mol Biol Evol. 19:1934–1942. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Kowalski PE, Freeman JD and Mager DL:
Intergenic splicing between a HERV-H endogenous retrovirus and two
adjacent human genes. Genomics. 57:371–379. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Gebefügi E, Brunmeir R, Weierich C, Wolff
H, Brack-Werner R and Leib M: Activation of a HERV-H LTR induces
expression of an aberrant calbindin protein in human prostate
carcinoma cells. Retrovirology. 6(Suppl 2): P482009.
|