Introduction
Colorectal cancer (CRC) is the third most common
type of cancer, which accounts for ~10% of all cases of cancer with
1,400,000 new cases and a cancer-associated mortality rate of
694,000 in 2012 (1). The
incidence of CRC is higher in developed countries, compared with
that in developing countries (1).
A number of risk factors of the disease have been defined to date,
including the consumption of red and processed meat, obesity,
smoking and lack of physical activity (2). Its symptoms and signs vary and
dependent predominantly on the location and metastasis of the
tumor. Localized cancer at an early stage can be cured with
surgical resection, however, advanced stages of cancer, accompanied
with presence of metastasis, is less likely to be cured by surgery
and has a poor prognosis with a significantly lower 5-year survival
rate (3).
CRC is a multistep-process resulting from
accumulative mutations of tumor suppressor genes and oncogenes
(4,5). Increasing studies have demonstrated
that heterogeneous mutations of the Wnt pathway, which potentiate
the pathway activity can contribute to the initiation and
development of CRC (6,7). In addition, the important roles of
adenomatous polyposis coli (APC) mutation and relevant β-catenin
signaling pathway have implicated in CRC (8). A common consensus has been reached
that APC mutation can lead to activation of the Wnt pathway by
regulating the accumulation of β-catenin in cells. In addition to
the Wnt pathway, a previous study suggested that APC mutations
affect CRC development via regulation of the energetic metabolite
pathways (9). Furthermore, a
previous in vitro study revealed that tumor suppressor gene,
mothers against decapentaplegic homolog 4 (SMAD4), if lost,
triggers the tumor suppressive bone morphogenetic protein (BMP)
signaling pathway to exert a metastasis-promoting effect on CRC
(10). By contrast, the tumor
suppressor gene, p53 can induce the expression of micro
(mi)RNA-34a, which affects the interleukin (IL)-6R/signal
transducer and activator of transcription 3/miR-34a feedback loop,
and reduces the rate of CRC progression (11). Despite these considerable
insights, the molecular mechanism of CRC remain to be fully
elucidated.
The interest in examining biomarkers of diseases
using bioinformatics approaches has increased. Scavenger receptor
class A, member 5 (SCARA5) is found to be downregulated and is
involved in CRC (12). Unlike the
above-mentioned studies, the present study investigated the
differentially expressed genes (DEGs) between CRC and control
samples, and then aimed to identify the CRC genes from the DEGs
obtained prior to the functional annotation of the identified CRC
genes through Gene Ontology (GO) and Kyoto Encyclopedia of Genes
and Genomes (KEGG) pathway enrichment analyses. The study aimed to
identify potential molecular targets for CRC. The findings of this
study may provide useful information regarding the pathogenesis of
CRC and lay the foundation for developing novel effective
therapeutic strategies for the management of CRC.
Materials and methods
Data preprocessing
As the present study did not involve any humans or
animals, there was no requirement for ethical approval. For
microarray data preprocessing and DEG identification, the gene
expression dataset (GSE7621) was downloaded from the Gene
Expression Omnibus (GEO) database (http://www.ncbi.nlm.nih.gov/geo/), consisting of 17
CRC tissues samples and 17 matched adjacent noncancerous tissues
samples from patients with CRC.
The probe-level data in the CEL files were converted
into expression measures using Affy package in R software
(http://www.r-project.org/), followed by
data normalization using the robust multiarray average 1 algorithm
(13). The data, prior to and
following normalization were presented as respective box plots.
Subsequently, the probe number was converted into corresponding
gene names, according to the HG-U133_Plus_2 Affymetrix Human Genome
U133 Plus 2.0 Array platform (Affymetrix, Inc., Santa Clara, CA,
USA). Expression values of multiple probes targeting one gene were
averaged, and this average expression value was selected for the
gene.
Following data preprocessing, Significance Analysis
of Microarrays (SAM) was performed to screen for the DEGs between
the CRC samples and noncancerous samples using samr package in R
software (http://cran.r-project.org/web/packages/samr/index.html)
(14). Subsequently, multiple
assessment correction was performed to adjust the P-values using
the Benjamini-Hochberg (BH) method (15). Genes with a false discovery rate
(FDR) <0.05 and fold-change >2 were selected.
Identification of CRC genes
The Database for Annotation, Visualization and
Integrated Discovery (DAVID; http://david.abcc.ncifcrf.gov/) provides a
high-throughput and desirable data-mining environment and combines
functional genomic annotations with intuitive graphical
representations, facilitating the transition between genomic data
and the biological function meaning (16). Using DAVID,
GENETIC_ASSOIATION_DB_DISEASE analysis was performed to identify
the CRC genes from the identified DEGs. The CRC genes were defined
as DEGs that were significantly associated with CRC (P <0.05)
(17).
GO and KEGG pathway enrichment
analysis
GO (http://www.geneontology.org/) functions as a database
to provide vocabularies and classifications associated with the
molecular and cellular structures and functions for biological
annotations of genes (18). GO
terms consist of three categories: Biological process (BP),
cellular component (CC) and molecular function (MF). The KEGG
(http://www.genome.jp/kegg/ or http://www.kegg.jp/) database contains rich
information pertaining to known metabolic pathways and regulatory
pathways, and facilitates the mapping of genes to KEGG pathways for
systemic analysis of gene functions (19).
To provide an insight into the precise biological
function and signaling pathways involved with the CRC genes
identified in the present study, GO and KEGG pathway enrichment
analysis were performed for the upregulated and downregulated DEGs,
respectively. GO terms with P<0.05 and gene count ≥5, and KEGG
pathways with P<0.05 were screened out.
Construction of a PPI network and
identification of hub CRC genes
The Search Tool for the Retrieval of Interacting
Genes (STRING; http://string-db.org/) is a useful
tool, which is capable of providing a comprehensive view of all the
known and predicted interactions and associations among proteins
(20). In order to elucidate the
interactions between the CRC genes in the present study, STRING
online software was used to construct a PPI network using the CRC
genes, and the network was visualized using Cytoscape open-source
software (http://www.cytoscape.org/) (21). In the PPI network, the proteins in
the network served as 'nodes' and the link connecting two nodes
represent a pairwise protein interaction. The degree of a node
corresponds to the number of interactions that the protein is in
possession of. The nodes with the highest degree of connection were
considered the 'hub' genes in the PPI network.
Predicting coding sequence (CDS) and
protein domains of hub genes
GENSCAN (http://genes.mit.edu/GENSCAN.html) is a complex
computer program, which applies a probabilistic model of human
genome structure to predict complete gene structures in human
genomic sequences. The probabilistic model is comprised of detailed
information of exons, introns and intergenic regions of human
genome (22–24). In the present study, to obtain a
better understanding of the structure of the identified hub CRC
genes, GENSCAN was used to predict the CDS of the hub genes. The
corresponding amino acid (aa) sequence was then obtained.
The protein domain of the hub genes was predicted
using the Pfam database (http://pfam.sanger.ac.uk/), which is a widely used
database containing information of protein domains and families,
and assists in determining the structure of proteins (25). Finally, the structure of the
obtained protein domain was visualized using Cn3D software version
4.0 (http://www.ncbi.nlm.nih.gov/Structure/CN3D/cn3d.shtml;).
The Cn3D ('see in 3D') is a molecular graphics program, which
facilitates viewing of the 3D structure of a protein sequence. An
interactive view of sequences and sequences alignments is also
available with Cn3D (26). The
structures of the proteins encoded by the hub genes were analyzed
using Conserved Domain Database (CDD). It is a protein annotation
database that provides rich information concerning protein
structure and function (http://www.ncbi.nlm.nih.gov/Structure/cdd/cdd.shtml).
Results
Data preprocessing
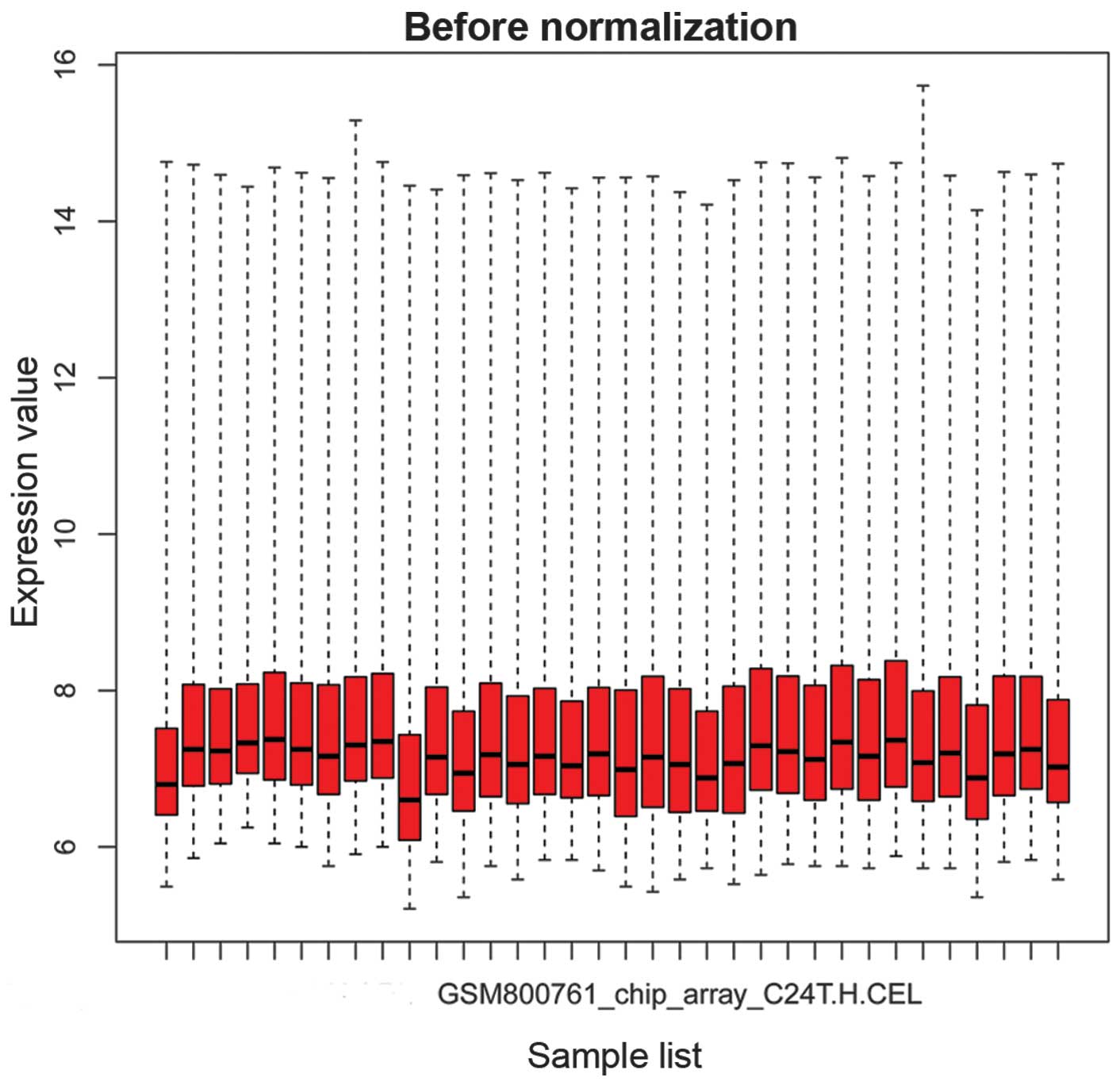
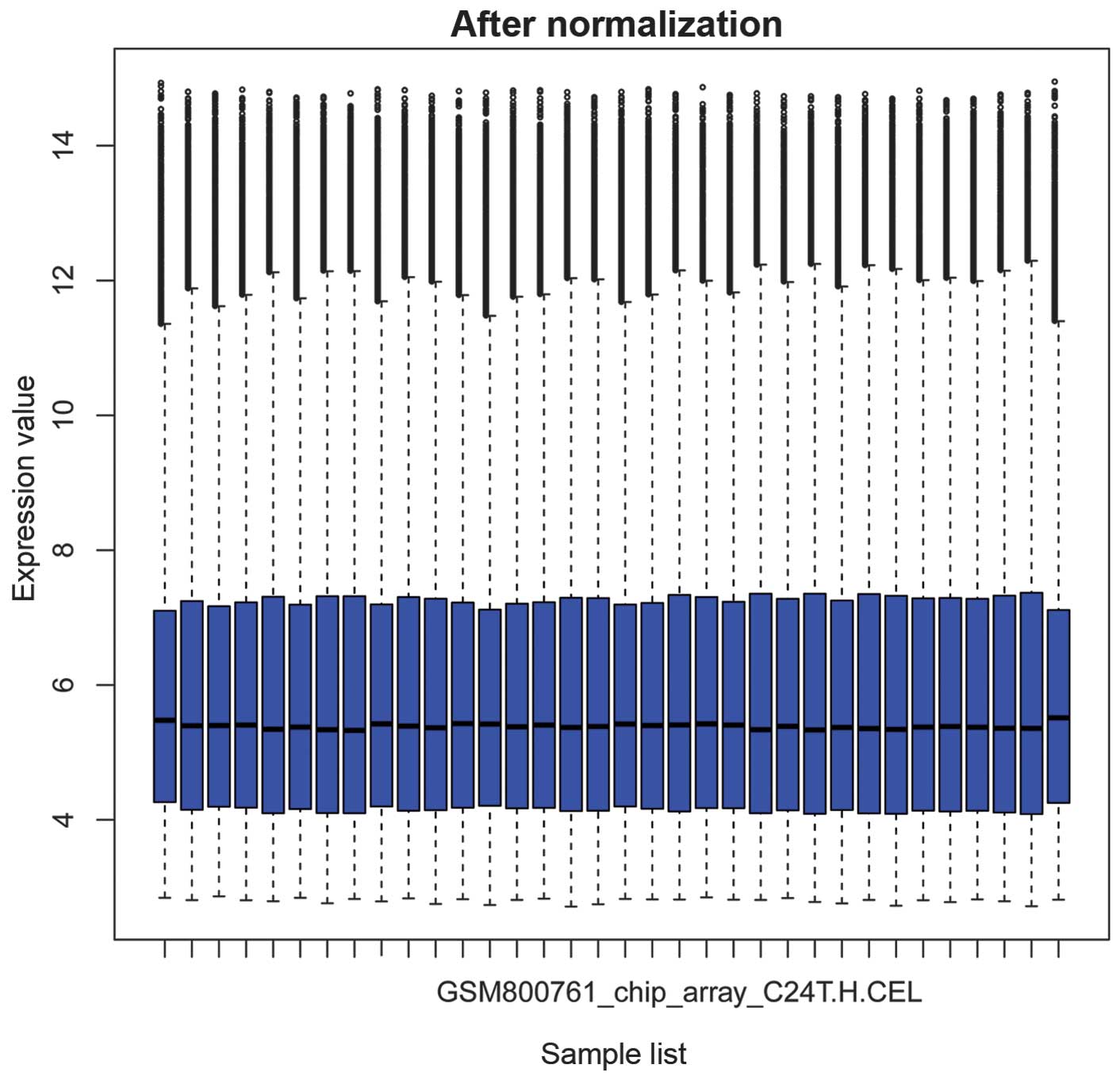
The box plots in Figs.
1 and 2 show the microarray
data priot to and following normalization, respectively. In
Fig. 1, the horizontal black
lines representing the median gene expression value for each sample
were varied. By contrast, the median gene expression vales appear
on a straight line in Fig. 2,
suggesting that the data following normalization was suitable for
further analysis.
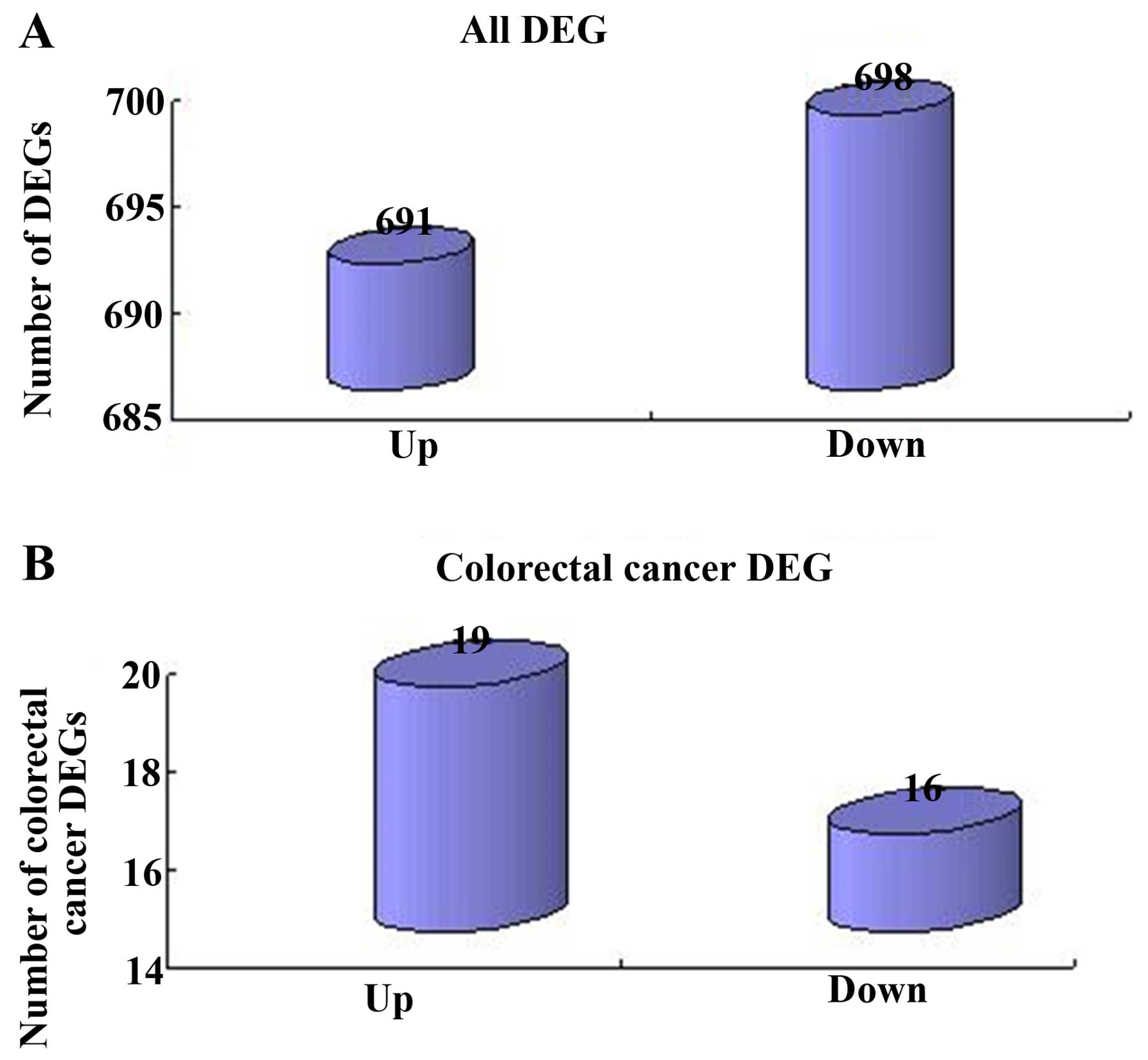
Identification of DEGs and CRC genes
Following data normalization, a total of 1,389 DEGs
were screened between the CRC and noncancerous samples, from which
691 genes were upregulated and 698 genes were downregulated
(Fig. 3A). Consequently, from the
obtained DEGs, 35 CRC genes were identified, including 19
upregulated and 16 downregulated genes (Fig. 3B).
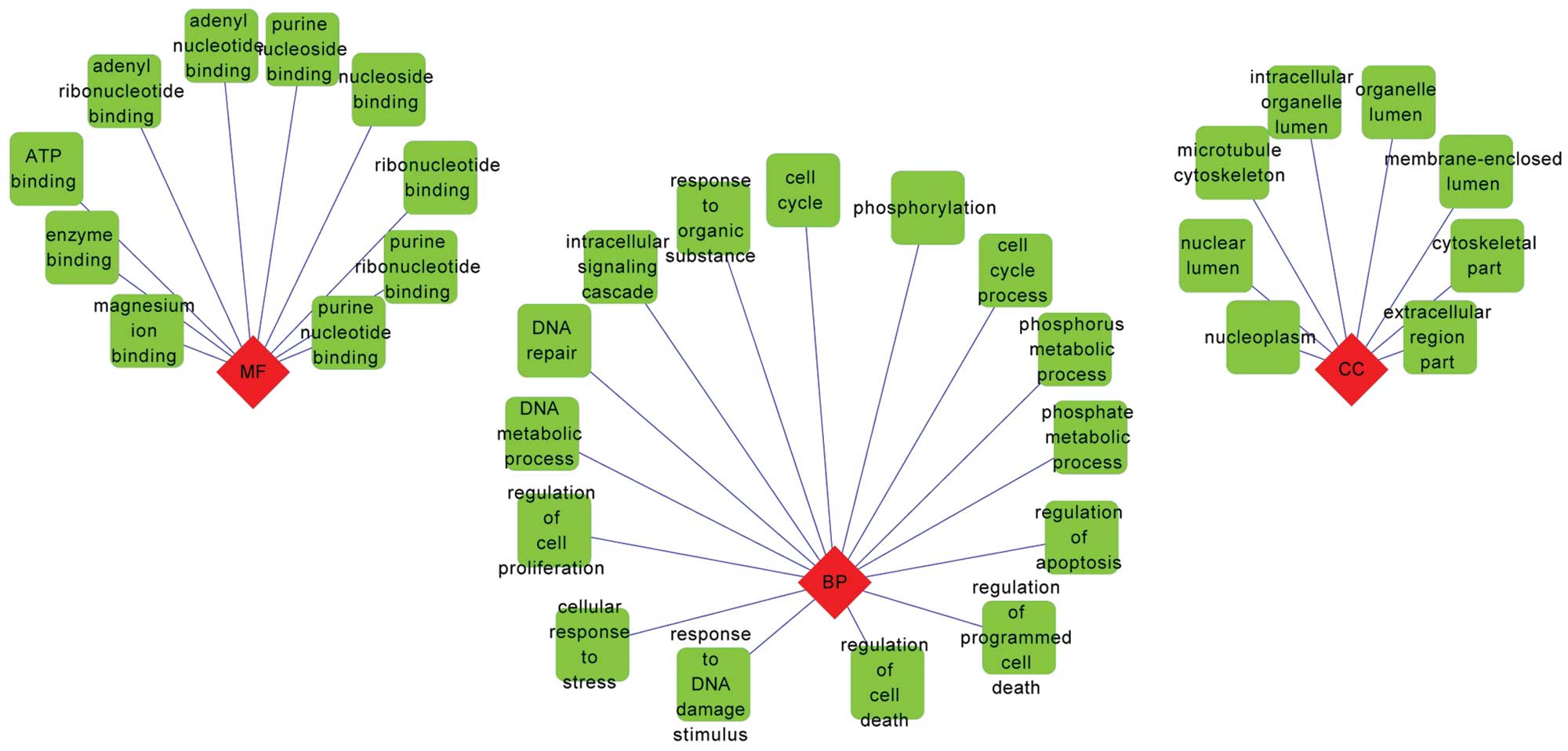
GO and KEGG pathway enrichment
analysis
The result of GO enrichment analysis revealed that
the upregulated CRC genes were enriched in a number of BP terms
associated with cell proliferation and DNA metabolism (Fig. 4). By contrast, the downregulated
genes were predominately associated with response to organic
substance and regulation of cell proliferation (Fig. 5).
As shown in Table
I, the base excision repair, cell cycle, bladder cancer and p53
signaling pathways were critical for the upregulated CRC genes. The
downregulated NAT1 and NAT2 genes were enriched in the caffeine
metabolism pathway.
 | Table IKEGG pathways enriched with
colorectal cancer genes. |
Table I
KEGG pathways enriched with
colorectal cancer genes.
| KEGG pathway | Gene | Change in
expression |
|---|
| Caffeine
metabolism | NAT1, NAT2 | Down |
| Base excision
repair | PCNA, POLB,
APEX1 | Up |
| Cell cycle | CCND1, PCNA, CHEK1,
CHEK2 | Up |
| Bladder cancer | CCND1, IL8,
MMP1 | Up |
| p53 signaling
pathway | CCND1, CHEK1,
CHEK2 | Up |
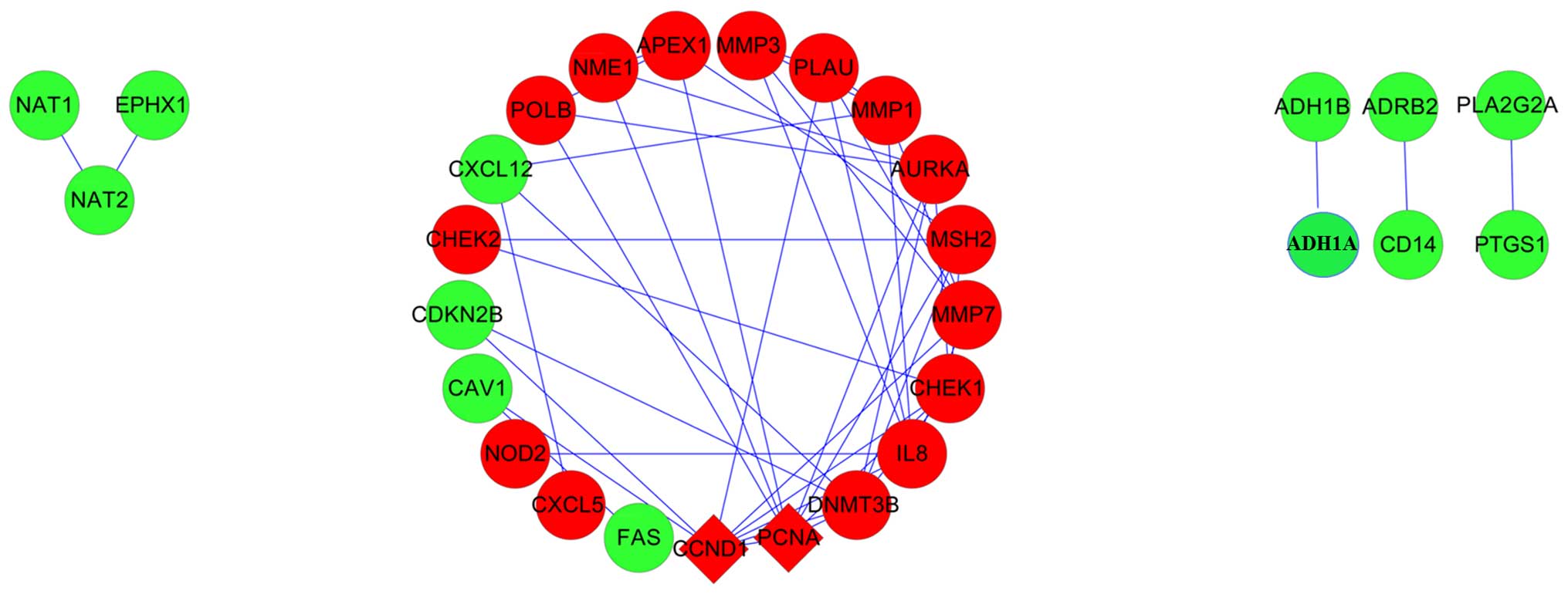
Identification of hub genes from the PPI
network
A PPI network was constructed using the obtained CRC
genes (Fig. 6). The two
upregulated genes, CCND1 and (PCNA), were determined as the hub
genes, as they were identified to exhibit the highest degrees of
connection.
Analysis of CDS and protein domain of the
hub genes
The CDSs and aa sequences of CCND1 and PCNA was
obtained, following which the protein domains were retrieved for
CCND1 and PCNA (Table II).
According to the Conserved Domains database, the N-terminal and
C-terminal domains of PCNA were topologically identical:
Proliferating cell nuclear antigen, in which three PCNA molecules
formed a closed ring zoning DNA duplex.
 | Table IIProtein domains of CCND1 and
PCNA. |
Table II
Protein domains of CCND1 and
PCNA.
| Coding-region | Family | Description | E-value |
|---|
| PCNA | PCNA_N | Proliferating cell
nuclear antigen, N-terminal domain | 2.8e-63 |
| PCNA_C | Proliferating cell
nuclear antigen, C-terminal domain | 1.5e-67 |
| CCND1 | Cyclin_N | Cyclin, N-terminal
domain | 1.8e-30 |
| Cyclin_C | Cyclin, C-terminal
domain | 5.6e-22 |
However, the N-terminal and C-terminal domains of
CCND1 were different. Cyclin_N: Cyclins modulated cyclin dependent
kinases (CDKs) and human cyclin-O was a type of uracil-DNA
glycosylase associated with other cyclins. Cyclins contain 2
domains of similar all-alpha fold, of which this family corresponds
with the N-terminal domain. Cyclin_C: Cyclins regulated CDKs and
human CCNO was a uracil-DNA glycosylase, linked to other cyclins.
Cyclins contain 2 domains of similar all-alpha fold, of which this
family corresponds with the C-terminal domain.
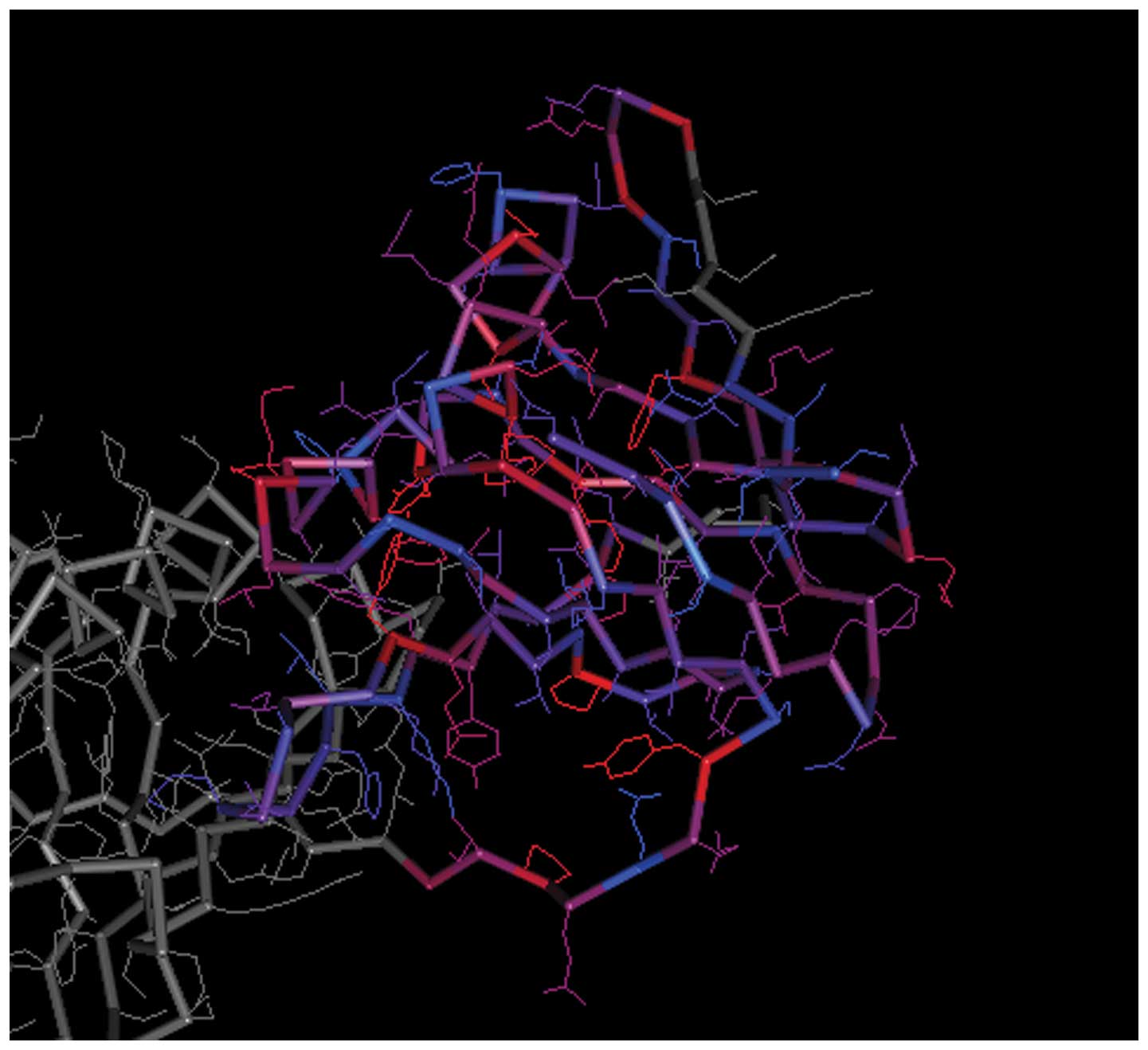
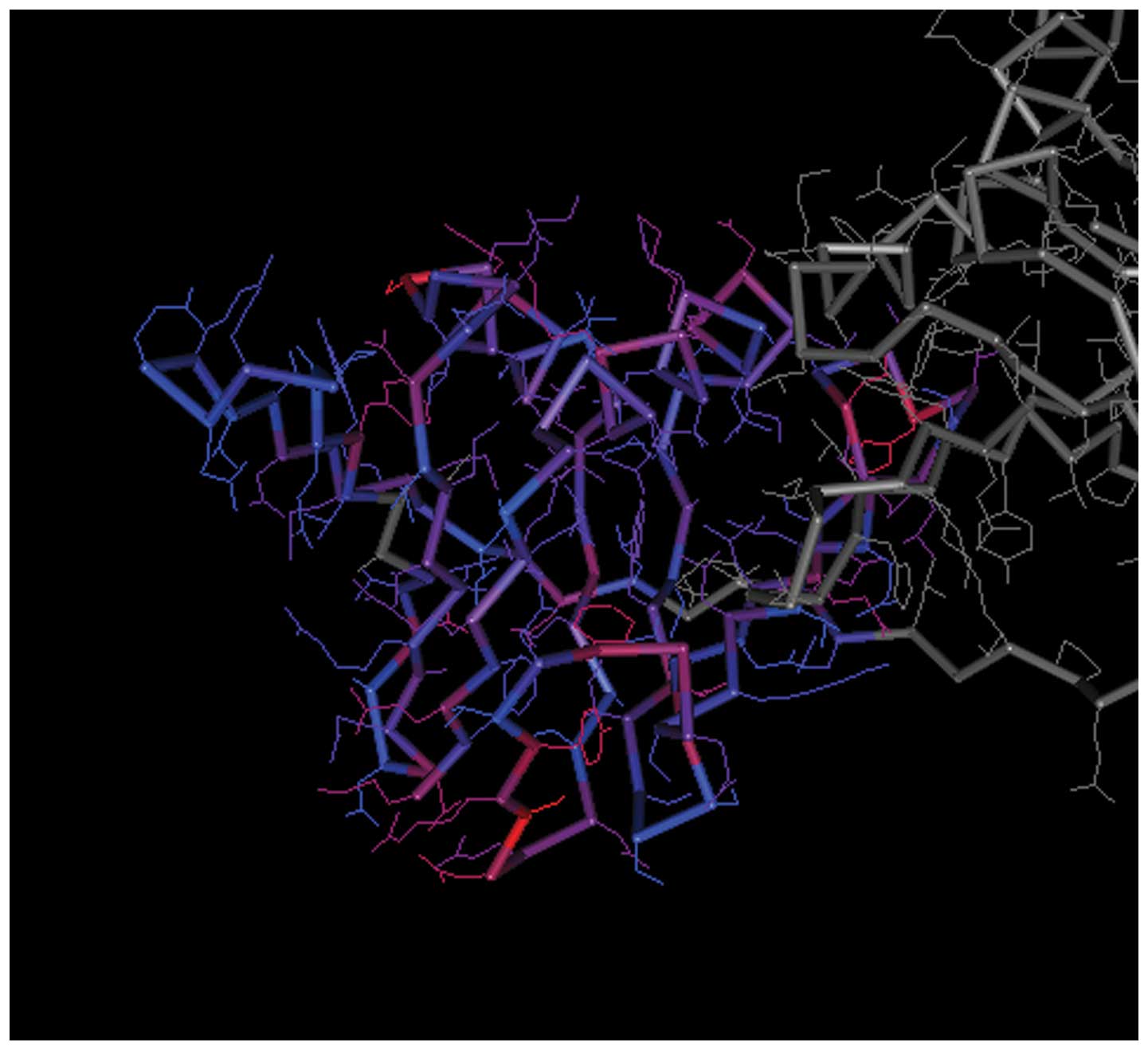
The spatial structures of the C-terminal and
N-terminal domains of PCNA are shown in Figs. 7 and 8, respectively. For PCNA, the structure
of the N-terminal domain was similar to that of the C-terminal
domain. The C-terminal and N-terminal domains of CCND1 are shown in
Figs. 9 and 10, respectively, in which the structure
of the N-terminal domain of CCND1 differed from that of the
C-terminal domain.
Discussion
Although the mortality rates of CRC have reduced
substantially, resulting from improved diagnosis and treatment of
the disease, it remains a primary health concern, which is life
threatening (27). The aim of the
present study was to determine the key genes involved in CRC and
their underlying mechanism. A total of 1,389 DEGs were screened out
between the CRC and noncancerous samples. From the 1,389 identified
DEGs, 35 CRC genes were further screened, which included 19
upregulated and 16 downregulated genes. GO enrichment analysis
revealed that the downregulated CRC genes were predominately
associated with response to organic substance and regulation of
cell proliferation, whereas the upregulated CRC genes were
predominantly involved in a number of biological functions
pertaining to cell death and DNA repair. KEGG pathway analysis
revealed that the downregulated CRC genes were enriched in the
caffeine metabolism pathway, whereas the upregulated CRC genes were
involved in base excision repair, cell cycle, bladder cancer and
p53 signaling pathways. In the PPI network, CCND1 and PCNA were
determined as hub genes, which had a number of interactions with
other CRC genes. Subsequent structural analysis of the two hub
genes revealed that the C-terminal and N-terminal domains were
similar in PCNA, but different in CCND1.
It has been well-established that the primary
function of N-acetyltransferase is in catalyzing the acetylation of
arylamines and aromatic amines, as well as the detoxication of
environmental toxins (28). NAT1
and NAT2 are two members of NAT family, of which NAT1 is
distributed in almost all tissues, whereas NAT2 is detected
predominantly in the liver. Although divergence exists in the
distribution of NAT1 and NAT2, which locate on chromosome 8, the
two are involved in metabolizing drugs and other xenobiotics, and
are involved in folate catabolism and caffeine metabolism (29). In addition, the overexpression of
NAT1 can result increase the survival rates of patients with cancer
and has been recommended as a viable target for developing therapy
against cancer (30). The
association between NAT2 and CRC is a source of controversy. A
meta-analysis, combining evidence from 40 studies, revealed that
NAT2 phenotypes may not be linked to the progression of CRC
(31). Another study demonstrated
that people with fast NAT2 acetylator phenotypes are at increased
risk of CRC (32). The present
study found that NAT1 and NAT2 were critical downregulated genes
for CRC and were predominantly enriched in the caffeine metabolism
pathway. The conflicting results between these studies may be
partly attributed to the specificity of population and
inter-individual variations. A previous large retrospective study
provided novel insight into the prevention of CRC, demonstrating
that coffee intake appears to reduce the susceptibility to colon
cancer, particularly in proximal cancer (33). These findings suggest that
downregulated NAT1 and NAT2 may facilitate the initiation and
development of CRC, through effects on the caffeine metabolism
pathway.
Cyclin D1 is the protein encoded by CCND1 gene,
belonging to the cyclin family that regulates CDKs. Cyclin D1 is
involved in modulating cell cycle (34,35) and suggests the possibility that
cyclin D1 may affect the development of CRC through modulation of
the cancer cell cycle. Accumulating evidence has revealed that
cyclin D1 polymorphisms may be associated with the prognosis of
patients with advanced CRC, administrated with cetuximab, and
confer susceptibility to CRC (36,37). However, a prospective,
population-based study suggested a novel viewpoint that the
expression of cyclin D1 is a preferred biomarker for predicting the
prognosis of male patients with CRC, but not female patients
(38). Furthermore, a previous
bioinformatics study suggested that microRNA may exert an antitumor
effect on colon cancer via targeting cyclin D1 (39). These studies demonstrated the
importance of cyclin D1 in CRC. In agreement with the studies, the
present study identified CCND1 as a hub gene in the PPI network,
which exhibited numerous interactions with other genes, adding
further support to suggestion that cyclin D1 is pivotal in CRC. The
present study also demonstrated, through detailed analysis of the
protein structure and spatial structure of the hub genes, that the
N-terminal and C-terminal domains of CCND1 were topologically
distinct. The protein domains of CCND1 may serve as targets for the
development of novel therapies.
The PCNA protein, essential for DNA synthesis and
DNA repair, resides in the nucleus and serves as a cofactor of DNA
polymerase δ. Of particular concern is that PCNA is can cooperate
with diverse partners in a coordinated manner, and is involved in
several pathways, including DNA repair, DNA synthesis and cell
cycle regulation (40). The
expression of PCNA at the invasive tumor margin has been suggested
to be positively correlated with the invasion and metastasis
potential of CRC (41). In
addition, it is also significantly associated with the liver
metastasis of CRC (42). These
evidence lead to a general acceptance that PCNA is implicated in
CRC. Consistently, the present study provided evidence that PCNA
was another hub gene of the PPI network, interacting with several
other genes. Unlike CCND1, PCNA possessed almost identical
N-terminal and C-terminal domains, allowing for the development of
therapies that target the specific structure of PCNA.
In the present study, the upregulated PCNA and CCND1
were enriched in cell cycle pathway, consistent with the results of
the GO enrichment analysis, suggesting that the upregulated genes
were significantly linked to a variety of biological processes
concerning cell and DNA metabolism, including cell cycle, cell
cycle process, DNA repair and regulation of cell proliferation. In
addition to the cell cycle pathway, CCND1 was also closely
associated with bladder cancer and p53 signaling pathways. p53, a
crucial tumor suppressor gene, acts as gatekeeper for cell growth
and division, and the expression of p53 is associated with clinical
outcomes and pathological characteristics of patients with CRC
(43). Based on the findings of
the present study, it was hypothesized that upregulated CCND1 may
enhance the growth and proliferation of CRC cells through
regulation of the cell cycle and p53 signaling pathways, and that
upregulated PCNA promotes the progression of CRC via effects on the
cell cycle pathway.
The present study had limitations due to the small
sample size, therefore, further investigations with large sample
sizes are warranted. Further in vivo and in vitro
investigations are also required to validate the findings of the
present study.
In conclusion, the present study found that PCNA,
CCND1, NAT1 and NAT2 may be pivotal genes for CRC. The upregulation
of PCNA and CCND1 may accentuate the development of CRC through
regulation of the cell cycle and p53 signaling pathways, and
downregulation of NAT1 and NAT2 may potentiate progression of the
disease by the targeting caffeine metabolism pathway. In addition,
the C-terminal and N-terminal domains were similar in PCNA, but
different in CCND1. These findings suggested the potential use of
these genes as molecular biomarkers in the early diagnosis and
monitoring of CRC. They also offer potential in developing a range
of potent therapies against CRC, which target the unique protein
domains of PCNA and CCND1.
References
|
1
|
Stewart BW and Wild C: World Cancer Report
2014. World Health Organization; 2014
|
|
2
|
Stegeman I, de Wijkerslooth TR, Stoop EM,
van Leerdam ME, Dekker E, van Ballegooijen M, Kuipers EJ, Fockens
P, Kraaijenhagen RA and Bossuyt PM: Colorectal cancer risk factors
in the detection of advanced adenoma and colorectal cancer. Cancer
Epidemiol. 37:278–283. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Nelson H, Petrelli N, Carlin A, Couture J,
Fleshman J, Guillem J, Miedema B, Ota D and Sargent D; National
Cancer Institute Expert Panel: Guidelines 2000 for colon and rectal
cancer surgery. J Natl Cancer Inst. 93:583–596. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Markowitz SD and Bertagnolli MM: Molecular
origins of cancer: Molecular basis of colorectal cancer. N Engl J
Med. 361:2449–2460. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Goyette MC, Cho K, Fasching CL, Levy DB,
Kinzler KW, Paraskeva C, Vogelstein B and Stanbridge EJ:
Progression of colorectal cancer is associated with multiple tumor
suppressor gene defects but inhibition of tumorigenicity is
accomplished by correction of any single defect via chromosome
transfer. Mol Cell Biol. 12:1387–1395. 1992.PubMed/NCBI
|
|
6
|
Vermeulen L, De Sousa E, Melo F, van der
Heijden M, Cameron K, de Jong JH, Borovski T, Tuynman JB, Todaro M,
Merz C, Rodermond H, et al: Wnt activity defines colon cancer stem
cells and is regulated by the microenvironment. Nat Cell Biol.
12:468–476. 2010. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Segditsas S and Tomlinson I: Colorectal
cancer and genetic alterations in the Wnt pathway. Oncogene.
25:7531–7537. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Fodde R, Smits R and Clevers H: APC,
signal transduction and genetic instability in colorectal cancer.
Nat Rev Cancer. 1:55–67. 2001. View
Article : Google Scholar
|
|
9
|
Yoshie T, Nishiumi S, Izumi Y, Sakai A,
Inoue J, Azuma T and Yoshida M: Regulation of the metabolite
profile by an APC gene mutation in colorectal cancer. Cancer Sci.
103:1010–1021. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Voorneveld PW, Kodach LL, Jacobs RJ, Liv
N, Zonnevylle AC, Hoogenboom JP, Biemond I, Verspaget HW, Hommes
DW, de Rooij K, et al: Loss of SMAD4 alters BMP signaling to
promote colorectal cancer cell metastasis via activation of Rho and
ROCK. Gastroenterology. 147:196–208. e132014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Rokavec M, Öner MG, Li H, Jackstadt R,
Jiang L, Lodygin D, Kaller M, Horst D, Ziegler PK, Schwitalla S, et
al: IL-6R/STAT3/miR-34a feedback loop promotes EMT-mediated
colorectal cancer invasion and metastasis. J Clin Invest.
124:1853–67. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Khamas A, Ishikawa T, Shimokawa K, Mogushi
K, Iida S, Ishiguro M, Mizushima H, Tanaka H, Uetake H and Sugihara
K: Screening for epigenetically masked genes in colorectal cancer
Using 5-Aza-2′-deoxycytidine, microarray and gene expression
profile. Cancer Genomics Proteomics. 9:67–75. 2012.PubMed/NCBI
|
|
13
|
Gautier L, Cope L, Bolstad BM and Irizarry
RA: affy - analysis of Affymetrix GeneChip data at the probe level.
Bioinformatics. 20:307–315. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Tibshirani R, Hastie T and Narasimhan B:
samr: SAM: Significance Analysis of Microarrays. R package version
1.26. 2011, http://cran.r-project.org/web/packages/samr/index.html;
Date accessed June 29, 2011.
|
|
15
|
Thissen D, Steinberg L and Kuang D: Quick
and easy implementation of the Benjamini-Hochberg procedure for
controlling the false positive rate in multiple comparisons. J Educ
Behav Stat. 27:77–83. 2002. View Article : Google Scholar
|
|
16
|
Dennis G Jr, Sherman BT, Hosack DA, Yang
J, Gao W, Lane HC and Lempicki RA: DAVID: Database for annotation,
visualization, and integrated discovery. Genome Biol. 4(3)2003.
|
|
17
|
Huang W, Sherman BT and Lempicki RA:
Systematic and integrative analysis of large gene lists using DAVID
bioinformatics resources. Nat Protoc. 4:44–57. 2009. View Article : Google Scholar
|
|
18
|
Harris MA, Clark J, Ireland A, Lomax J,
Ashburner M, Foulger R, Eilbeck K, Lewis S, Marshall B, Mungall C,
et al: Gene Ontology Consortium: The Gene Ontology (GO) database
and informatics resource. Nucleic Acids Res. 32:D258–D261. 2004.
View Article : Google Scholar
|
|
19
|
Kanehisa M, Goto S, Sato Y, Furumichi M
and Tanabe M: KEGG for integration and interpretation of
large-scale molecular data sets. Nucleic Acids Res. 40:D109–D114.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Franceschini A, Szklarczyk D, Frankild S,
Kuhn M, Simonovic M, Roth A, Lin J, Minguez P, Bork P, von Mering
C, et al: STRING v9.1: Protein-protein interaction networks, with
increased coverage and integration. Nucleic Acids Res.
41:D808–D815. 2013. View Article : Google Scholar :
|
|
21
|
Saito R, Smoot ME, Ono K, Ruscheinski J,
Wang P-L, Lotia S, Pico AR, Bader GD and Ideker T: A travel guide
to Cytoscape plugins. Nat Methods. 9:1069–1076. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Burge C and Karlin S: Prediction of
complete gene structures in human genomic DNA. J Mol Biol.
268:78–94. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Marasco E, Ross A, Dawson J, Moroose T and
Ambrose T: Detecting STR peaks in degraded DNA samples. Proceedings
of the 4th International Conference of Bioinformatics and
Computational Biology; pp. 1–8. LasVegas, USA. 2012
|
|
24
|
McEvoy CR, Seshadri R and Firgaira FA:
Large DNA fragment sizing using native acrylamide gels on an
automated DNA sequencer and GENESCAN software. Biotechniques.
25:464–470. 1998.PubMed/NCBI
|
|
25
|
Punta M, Coggill PC, Eberhardt RY, Mistry
J, Tate J, Boursnell C, Pang N, Forslund K, Ceric G, Clements J, et
al: The Pfam protein families database. Nucleic Acids Res.
40:D290–D301. 2012. View Article : Google Scholar :
|
|
26
|
Wang Y, Geer LY, Chappey C, Kans JA and
Bryant SH: Cn3D: sequence and structure views for Entrez. Trends
Biochem Sci. 25:300–302. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Jemal A, Ward E and Thun M: Declining
death rates reflect progress against cancer. PLoS One. 5. pp.
e95842010, View Article : Google Scholar
|
|
28
|
Evans DA: N-acetyltransferase. Pharmacol
Ther. 42:157–234. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Minchin RF, Hanna PE, Dupret J-M, Wagner
CR, Rodrigues-Lima F and Butcher NJ: Arylamine N-acetyltransferase
I. Int J Biochem Cell Biol. 39:1999–2005. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Butcher NJ and Minchin RF: Arylamine
N-acetyltransferase 1: A novel drug target in cancer development.
Pharmacol Rev. 64:147–165. 2012. View Article : Google Scholar
|
|
31
|
Zhang L, Zhou J, Wang J, Liang G, Li J,
Zhu Y and Su Y: Absence of association between N-acetyltransferase
2 acetylator status and colorectal cancer susceptibility: Based on
evidence from 40 studies. PLoS One. 7:e324252012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Lilla C, Verla-Tebit E, Risch A, Jäger B,
Hoffmeister M, Brenner H and Chang-Claude J: Effect of NAT1 and
NAT2 genetic polymorphisms on colorectal cancer risk associated
with exposure to tobacco smoke and meat consumption. Cancer
Epidemiol Biomarkers Prev. 15:99–107. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Sinha R, Cross AJ, Daniel CR, Graubard BI,
Wu JW, Hollenbeck AR, Gunter MJ, Park Y and Freedman ND:
Caffeinated and decaffeinated coffee and tea intakes and risk of
colorectal cancer in a large prospective study. Am J Clin Nutr.
96:374–381. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Baldin V, Lukas J, Marcote MJ, Pagano M
and Draetta G: Cyclin D1 is a nuclear protein required for cell
cycle progression in G1. Genes Dev. 7:812–821. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Jirawatnotai S, Hu Y, Michowski W, Elias
JE, Becks L, Bienvenu F, Zagozdzon A, Goswami T, Wang YE, Clark AB,
et al: A function for cyclin D1 in DNA repair uncovered by protein
interactome analyses in human cancers. Nature. 474:230–234. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhang W, Gordon M, Press OA, Rhodes K,
Vallböhmer D, Yang DY, Park D, Fazzone W, Schultheis A, Sherrod AE,
et al: Cyclin D1 and epidermal growth factor polymorphisms
associated with survival in patients with advanced colorectal
cancer treated with Cetuximab. Pharmacogenet Genomics. 16:475–483.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Yang Y, Wang F, Shi C, Zou Y, Qin H and Ma
Y: Cyclin D1 G870A polymorphism contributes to colorectal cancer
susceptibility: Evidence from a systematic review of 22
case-control studies. PLoS One. 7:e368132012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Wangefjord S, Manjer J, Gaber A, Nodin B,
Eberhard J and Jirström K: Cyclin D1 expression in colorectal
cancer is a favorable prognostic factor in men but not in women in
a prospective, population-based cohort study. Biol Sex Differ.
2(10)2011. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Nie J, Liu L, Zheng W, Chen L, Wu X, Xu Y,
Du X and Han W: microRNA-365, downregulated in colon cancer,
inhibits cell cycle progression and promotes apoptosis of colon
cancer cells by probably targeting Cyclin D1 and Bcl-2.
Carcinogenesis. 33:220–225. 2012. View Article : Google Scholar
|
|
40
|
Maga G and Hübscher U: Proliferating cell
nuclear antigen (PCNA): A dancer with many partners. J Cell Sci.
116:3051–3060. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Teixeira CR, Tanaka S, Haruma K, Yoshihara
M, Sumii K and Kajiyama G: Proliferating cell nuclear antigen
expression at the invasive tumor margin predicts malignant
potential of colorectal carcinomas. Cancer. 73:575–579. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Sumiyoshi Y, Yamashita Y, Maekawa T, Sakai
N, Shirakusa T and Kikuchi M: Expression of CD44, vascular
endothelial growth factor, and proliferating cell nuclear antigen
in severe venous invasional colorectal cancer and its ionship to
liver metastasis. Surg Today. 30:323–327. 2000. View Article : Google Scholar
|
|
43
|
Yu SJ, Yu JK, Ge WT, Hu HG, Yuan Y and
Zheng S: SPARCL1, Shp2, MSH2, E-cadherin, p53, ADCY-2 and MAPK are
prognosis-related in colorectal cancer. World J Gastroenterol.
17:2028–2036. 2011. View Article : Google Scholar : PubMed/NCBI
|