The concentration range of biological macromolecules
such as ribonucleoproteins, polysaccharides, nucleic acids,
proteins and others inside cells is 80-400 mg/ml (1). In response to such high
concentrations, organisms have developed various conserved
mechanisms to prevent the chaotic aggregation of proteins by
allowing these proteins to form different higher-order complexes
with multiple biological functions as a response to different types
of environmental stress (2).
There are typically two types of higher-order
assemblies: i) Stable and rigid protein-protein interactions that
generate ordered, solid-like macromolecular complexes; and ii)
complexes consisting of weaker and more dynamic molecules. In
biology, the term 'aggregation' is commonly used to describe
assemblies formed under pathological conditions, where the
molecules in the aggregate are irreversibly disrupted and often
considered as pathogenic factors. Aggregation represents a
prominent characteristic of irreversible biological processes. By
contrast, the term 'condensation' refers to reversible and dynamic
molecules which can be redissolved to perform their respective
functions, and their assembly is tightly monitored within the
intracellular environment (3).
However, these two types of higher-order protein assemblers are not
completely independent. Disruptions in protein homeostasis under
pressure or under pathological conditions can result in an
imbalance of biomolecular condensation, ultimately leading to the
uncontrolled collapse of these structures, which in turn triggers
the irreversible aggregation and misfolding of protein
constituents, and often leads to the transformation of aged or
solidified condensates into aggregates (4,5).
Hypoxia is a prevalent environmental stressor
encountered by aerobic organisms and a common property of
pathological disorders such as bacterial infections, inflammation,
impairment, cardiovascular disease (CVD) and cancer (6,7).
Eukaryotes have developed a rapid and well-conserved hypoxia
response mechanism. More specifically, hypoxia induces the
production of cellular reactive oxygen species (ROS) and
acidification of the cellular environment due to decreased oxygen
supply (8). Several studies have
examined the stress responses of mitochondria and endoplasmic
reticulum (ER) under hypoxic conditions, and they showed that the
protein folding process is impaired and protein homeostasis is
disrupted (9,10). Kaufman et al (9) recently revealed that
hypoxia-induced insolubility of specific proteins in nematodes; it
was revealed that oxygen depletion and adenosine triphosphate (ATP)
could disturb the intracellular equilibrium, leading to
uncontrolled aggregation. However, eukaryotic cells have evolved
conserved molecular chaperones and protein autophagy networks to
maintain balance (6). There is
also increasing evidence that uncontrolled protein homeostasis and
condensate aging are involved in hypoxia-related diseases,
providing a probable cause for the relationship between hypoxic
stress and related diseases (11-13).
A hypoxic environment may induce an imbalance of
protein homeostasis and aggregation. This imbalance can also
activate the assembly of biomolecular condensates, which play
crucial roles as organelles without membrane and are regulated by
multiple mechanisms related to environmental stress (3). Stress granules (SGs) (14), glycolytic bodies (G-bodies)
(15) and processing bodies
(P-bodies) (16) contribute to
cell survival under stress conditions and induce metabolic
reprogramming in hypoxic environments.
In the present review, the aim was to summarize
hypoxia-induced aggregate behaviors and discuss their functions and
regulatory mechanisms, hoping that the information provided in the
review could help us to gain better insights into the mechanisms
underlying neuromedicine, altitude medicine and the tumor
microenvironment.
Hypoxia is a common stressor for aerobic cells that
can lead to cell acidification, oxidative stress, cell cycle arrest
and death (17). Using
transmission electron microscopy, recent studies have revealed the
presence of abundant electron-dense deposits, which represent
aggregates of unfolded and misfolded proteins in neurons exposed to
ischemic-hypoxic brain injury (18,19). During hypoxic stress, the
obstruction of protein folding serves as the primary cause of
protein aggregation, prompting eukaryotes to develop unfolded
protein responses as a regulatory mechanism (20,21). In the current study, a
comprehensive review of the mechanisms involved in hypoxia-induced
aggregation of unfolded and misfolded proteins, and the cellular
strategies relying to this phenomenon is presented.
The number of large multidomain proteins notably
increases from prokaryotes to eukaryotes. These proteins exhibit
diverse conformations, and as their protein configurations become
more complex, the possibility of misfolding increases (22). Hydrophobic amino acid residues,
unstructured regions in folding intermediates and misfolded
proteins are often exposed to solvents, leading to aggregation
(23). Aggregates are primarily
driven by liquid-liquid phase separation (LLPS) or hydrophobic
forces, depending on the concentration (24). While most aggregates are
amorphous, the aggregation of certain proteins leads to the
formation of amyloid fibers characterized by β strands normal to
the long fibril axis (cross-β structure) (25). Before fiber formation, amyloid
often exists in an oligomeric state, and both types of aggregates
play crucial roles in diseases (26). For instance, cerebral blood flow
decreased in patients with early Alzheimer's disease (AD) (27). Increased binding of oligomeric
β-amyloid protein (Aβ) to ROS leads to vasoconstriction around
brain cells, contributing to decreased cerebral blood flow, which
may initiate a cascade reaction involving amyloid Aβ itself or the
fibrous Aβ, which is important for driving cognitive decline
(27,28). Thus, it is necessary to
understand the mechanisms underlying hypoxia-induced protein
aggregation for elucidating the pathogenesis of neurodegenerative
disease and developing intervention strategies.
Molecular chaperones play an important role in
maintaining protein homeostasis, and assist other proteins in
acquiring functionally active conformations without affecting their
final structure. Different types of molecular chaperones receive
newly synthesized protein chains from ribosomes to ensure effective
folding and minimize aggregate formation by guiding them through
appropriate folding pathways (26). As proteins are structurally
dynamic, proteostasis occurs via a network of chaperones and
protein degradation mechanisms that continuously monitor the
proteome (29,30). Chaperones help prevent chain
compaction and misfolding, and facilitate the removal of protein
aggregates through lysosomal-autophagy degradation (31). Before degradation, the
depolymerization of aggregates is cooperatively carried out by heat
shock proteins (Hsps) such as Hsp70, Hsp110 and Hsp40 (32,33). The clearance pathways involving
proteasomes and lysosomes are intricately linked to the Hsp70 and
Hsp90 chaperone systems through specialized ubiquitin ligases such
as the co-chaperone C-terminus of the Hsc70-interacting protein and
the BAG domain (34,35).
However, under hypoxia conditions, the regulatory
network of protein homeostasis is disrupted, and numerous molecular
chaperones are affected by hypoxic stress. Nguyen et al
(36) observed notable global
reductions of ATP-dependent Hsp70 and Hsp90 (83 and 78%,
respectively) after 24 h of hypoxia treatment. Conversely, the
protein expression of the ATP-independent Hsp27 and Hsp40 in the
brain, heart and muscle remained constant throughout the 24-h
hypoxia treatment. However, with prolonged hypoxia, the expression
of the Hsp27 and Hsp40 genes in these tissues was also reduced,
suggesting that the protein expression of these chaperones may also
eventually decrease under hypoxia. These results suggest that
energy conservation is prioritized over cytoprotective protein
chaperoning in naked mole-rat tissues during acute hypoxia.
Although ATP-independent partners do not require ATP to regulate
their functional cycle passive histone aggregation (37), aggregate bursts under low oxygen
stress also suggest that these ATP-independent partners cannot
remedy the homeostatic imbalance caused by the energy gap. In fact,
the effects of hypoxic stress on protein chaperones are not
machine-made, for example, C2C12 cells induce Hsp70 gene expression
through a similar mechanism to heat stress during acute hypoxia
(38). However, macrophages
exposed to 5% oxygen for 24 h notably reduced Hsp70 expression and
recovered after reoxygenation (39). Proteomics indicated that Hsp72
downregulation in the cerebral cortex of rats after 5 days of
hypoxia reached its lowest level (40). In addition, the Hsp90 chaperone
family TRAP1 has been found to be frequently induced in tumors and
regulate energy metabolism after HIF-1 stabilization (41), and hypoxia can also reduce the
transcription of cyclin B1 in liver cancer cells through Hsp90
(42). These contradictory
results may be due to differences in the function and distribution
of molecular chaperons, and the crosstalk between hypoxia stress
and chaperons may need further exploration.
Disulfide bonds are commonly found in protein
domains located in the cytoplasmic membrane and enhance protein
stability. The cleavage of disulfide bonds triggers the function of
some secreted soluble proteins and cell-surface receptors (43). Oxidative protein folding refers
to the restorative process through which proteins containing
disulfide bonds transit from fully reduced and unfolded states to
their original bioactive forms (44-46). Koritzinsky et al (47) used 35S labeling and suggested
that the production of disulfide bonds was limited by hypoxic
surroundings and that protein folding recovered upon oxygen
restoration (48,49). This evidence suggested that
oxygen depletion may seriously impede disulfide bonding leading to
protein misfolding.
In brief, oxygen deprivation disrupts protein
folding through multiple mechanisms, including inhibiting disulfide
bond formation, inactivation of molecular chaperones and elevation
of ROS levels (50,51). Prolonged accumulation of
misfolded proteins may eventually result in the formation of
pathological protein aggregates (Fig. 1). This shift contributes to the
development of neurodegenerative diseases, such as AD, Huntington's
disease (HD) and Parkinson's disease (PD) (3,52,53). Moreover, hypoxic-ischemic
encephalopathy (HIE) occurs when the brain is exposed to oxygen
deprivation and ischemia. Newborns often experience HIE due to
birth asphyxia, causing an unfavorable prognosis owing to cerebral
dysfunction, neuronal cell death and neurological deficits.
Notably, marked molecular and subcellular changes observed in the
brain cells of patients with HIE include protein misfolding,
aggregation and organelle damage (54). The disruption of protein
homeostasis is also closely related to cardiac hypertrophy,
cardiomyopathy and heart failure caused by cardiovascular hypoxia
(55). Soluble protein oligomers
have been observed in the myocardial cells of patients with
idiopathic dilated cardiomyopathy, non-ischemic cardiomyopathy, or
hypertrophic heart disease (56). Similarly, aggregation of abnormal
and ubiquitinated proteins has been detected in the heart of
individuals with dilated cardiomyopathy or ischemic heart disease
(57). Pattison et al
(58) previously demonstrated
that the expression of ectopic gene that containing 83 glutamine
repeats in cardiomyocytes promoted the cohesive accumulation and
aggregation of pre-glutamine amyloid oligomers, increasing protein
deposition, cardiac muscle cell death and heart failure.
Cellular proteostasis is tightly controlled by a
network of molecular chaperones. In addition to counteracting
abnormal folding and aggregation by directly binding to misfolded
proteins (59), chaperones also
assist the ubiquitin-proteasome system (UPS) (60) and the autophagy-lysosome system
in degrading aggregators for proteostasis (61).
The lysosomal-mediated autophagy degradation pathway
is a major hunter for clearing protein aggregates, especially in
neurodegenerative diseases (62). Most neurodegenerative diseases
involve pathological abnormal protein aggregates, developing
neurofibrillary tangles. For example, Aβ and C-terminal fragments
of the amyloid precursor protein in AD, mutant α-synuclein in PD,
polyglutamine-expanded huntingtin in HD, and mutant superoxide
dismutase 1 and TAR DNA-binding protein 43 (TDP-43) in ALS
(63-65). These protein aggregates mainly
target the autophagy lysosomal degradation pathway, and chaperone
proteins play a key role in this process. Specific aggrephagy
receptors have been reported in yeast S. cerevisiae (Atg19)
and C. elegans (SEPA-1 and EPG-7) (66-68). Recently, Ma et al
(69) reported the function of
the TRiC subunit chaperonin-containing TCP-1 subunit 2 (CCT2) in
aggrephagy in mammals and yeast. CCT2 promotes autophagosome
incorporation and clearance of protein aggregates with little
liquidity by interacting with ATG8s and aggregation-prone proteins
independent of cargo ubiquitination. The dual function of CCT2, as
a chaperone and an aggrephagy receptor, enables double-layer
maintenance of proteostasis.
Cellular stress and aging can lead to a decrease in
protein homeostasis. In addition to the inhibition of protein
chaperone activity by hypoxia metabolism, notably,
hypoxia-reoxygenation treatment dysregulates key molecules that
maintain autophagy-lysosomal flux in primary human trophoblasts,
notably reduced autophagosomes and autolysosomes (70). The expression of ubiquitin
26S-proteasome E3 ligase, autophagolysosomal degradation related
mRNA transcripts and proteins, and integrated stress response
markers were also decreased after 12 days of hypoxic feeding
(71).
The UPS system is strongly associated with
regulating biomolecular condensation (60). More specifically, ubiquitin and
other post-translational modifications act as agents of phase
separation, and are essential for the formation of condensates and
ubiquitin-proteasome system activity (5). It is noteworthy that previous
studies demonstrated that polyubiquitin chains can function as
multivalent molecules that can drive either the assembly or the
disassembly of condensates via interactions with various
ubiquitin-binding proteins (72,73).
Cellular responses to hypoxia primarily aim to
enhance cell survival and restore oxygen equilibrium. In the
context of uncontrolled protein folding, the accumulation of
unfolded or misfolded proteins within the ER or mitochondrial space
leads to activation of UPR (50,53). Through its distinct signalling
network, the UPR pathway restores protein homeostasis, alleviates
the burden of protein aggregation and maintains cell viability
(74-78). The heavy-chain-binding protein
(BIP), a member of the Hsp70 family, is a crucial chaperone that
triggers UPR activation. BIP enters the ER by binding to
hydrophobic amino acids to prevent incorrect folding and
polymerization of the polypeptide chains. This is followed by ATP
binding and subsequent release of the bound polypeptides through
ATP hydrolysis (79).
Environmental stress leads to misfolded proteins accumulating,
causing the release of BIPs (80). The released BIPs undergo
phosphorylation and polymerization, triggering the activation of
protein kinase R (PKR)-like ER kinases (PERKs) and
inositol-requiring enzyme-1 (IRE1) (81). Additionally, activating
transcription factor (ATF) 6 is switched to the Golgi apparatus and
convered to soluble and active cytoplasmic ATF6 (82-84). These PERK, IRE1 and ATF6 sensors
constitute three distinct signalling pathways within the UPR
(80,85). Hypoxia induces BIP expression in
both cancer and endothelial cells (86-88). Hypoxia can activate the PERK
signalling pathway in various models (89-91), and the phosphorylation of
eukaryotic initiation factor 2 (eIF2) mediated by PERK was observed
within minutes of hypoxic exposure, with a reduced response rate as
the oxygen concentration increased (92). To alleviate ER stress, UPR
signalling inhibits protein aggregation by reducing protein
synthesis flux and activating the transcriptional program of
molecular chaperones.
The hypoxia-mediated UPR has been well demonstrated
in the tumor microenvironment, and exposure of solid tumors to
intermittent hypoxia may lead to high ROS levels and UPR activation
(93-95). For example, increased ATF4
expression has been shown in numerous hypoxic and nutrient-deprived
tumors (96) and can mediate
autophagy under hypoxia (97).
Immunohistochemical staining demonstrated increased expression of
ATF4 in hypoxic, perinecrotic regions distal to the tumour
vasculature, consistent with a nutrient-deprived mechanism of
translational activation. In addition, the distribution of p-eIF2a
and p-GCN2 signal demonstrated considerable association in serial
sections, consistently, spontaneous mouse tumours also contain
greater levels of p-eIF2a and ATF4 than corresponding normal tissue
(98). PERK and ATF4 protect
glioblastoma cells exposed to cyclic hypoxia or radiotherapy from
oxidative damage (99,100). In human cervical cancer, PERK
activation leads to the accumulation of oncogenic
lysosomal-associated membrane protein 3, thus increasing the
aggressiveness of these cells (99).
Mitochondria are the primary consumers of oxygen
within cells. Early mitochondrial dysfunction is implicated in
numerous hypoxic diseases such as cancer and neurodegenerative
diseases (17,101,102). The efficiency of mitochondrial
oxidative phosphorylation is markedly reduced under hypoxic
conditions due to mitochondrial perinuclear localization and
fragmentation mediated by CHCHD4 (103-105). Mitochondria contain their
inherent genetic information and rely on stress response systems to
translate and fold encoded proteins, and refold nuclear-encoded
proteins (106). Maintenance of
protein homeostasis in this organelle involves unique molecules
such as Hsp60 and the peptidase lon peptidase 1 (106,107). Under hypoxic conditions,
mitochondria can also experience unfolded or misfolded proteins
aggregating. For example, using C. elegans, Kaufman et
al (9) identified 65
preferentially insoluble mitochondrial proteins and 110 generally
insoluble mitochondrial proteins during hypoxia, and reported that
the abundance of hypoxia-induced mitochondrial protein aggregates
(HIMPA) increased notably with the severity of hypoxia.
Additionally, Yan et al (108) reported that disruption of
mitochondrial proteostasis and mitochondrial protein aggregation
are early processes involved in hypoxia in C. elegans. Like
in the ER, mitochondria also activate their own UPR, which is known
as the mitochondrial UPR (UPRmt). The UPRmt is classically
considered as a transcriptional response that increases the
expression of mitochondrial chaperones to protein misfolding and
aggregation in mitochondria (109-111). In C. elegans, the UPRmt
was found to be regulated by sensitizing transcription factor
associated with stress 1 (ATFS-1), which is a transcription factor
within mitochondrial and nuclear localization sequences, and dual
subcellular localization. ATFS-1 is transported into the
mitochondrial matrix and then degraded by LON proteases under
steady-state conditions. The transport of ATFS-1 is downregulated
in mitochondrial dysfunction, and ATFS-1 is subsequently
transported to the nucleus to stimulate transcriptional responses
(111,112). Additional regulatory mechanisms
may exist in mammalian cells, with ATF5 acting as a functional
ortholog of ATFS-1 (113). In
addition, ATF4 and the C/EBP homologous protein activating are
important in the activation of UPRmt (114,115). Activation of UPRmt to
mitochondrial stress in cancer could maintain mitochondrial
integrity and tumor growth (116). A recent study by Sutandy et
al (117) showed that UPRmt
signaling is prompted by the release of two individual signals in
the cytosolmitochondrial ROS (mtROS) and mitochondrial protein
precursors in the cytosol, leading to the release of HSF1 by Hsp70,
which results in nuclear translocation and transcription of UPRmt
genes (117).
The expression of these transcription factors is
mediated by eIF2α kinase phosphorylation (118). Recently, Guo et al
(119) delineated the
relationship between mitochondrial stress and the relay of ATF4.
Heme-regulated initiation factor 2 α kinase (HRI) is a necessary
eIF2 kinase for this relay. A genome-wide CRISPRi screen identified
two upstream signaling factors for HRI: The OMA1 zinc
metallopeptidase (OMA1), as a mitochondrial stress-activated
protease, and the DAP3 binding cell death enhancer 1 (DELE1)
associating with the inner mitochondrial membrane. Mitochondrial
stress results in DELE1 cleavage by OMA1 and its accumulation in
the cytosol, which interacts with HRI and increases eIF2 kinase
activity. These results indicated that the UPRmt and UPR signaling
pathways can been interlinked via eIF2α (Fig. 2) (109-111).
HIMPA consistently alleviates hypoxia-induced cell
death, and UPRmt activation had the same effect. However, UPRmt is
not necessarily protective against hypoxia-induced cell death
(108). It is the
overactivation of UPRmt that can induce cell death as in the case
of UPR (118), and the
relationship of HIMPA with UPRmt and its crosstalk with UPR needs
to be explored further.
Previous studies have suggested that the cytosol is
not uniform in which proteins diffuse freely, but rather formed
biomolecular condensates with phase separation (52,120). Previous studies have shown that
cytoplasmic proteins or RNAs are organized into distinct
biomolecular condensates (52,121,122). These condensates, also known as
organelles without membrane, employ the cytoskeleton for targeted
transport. These proteins serve as the center for biochemical
reactions, act as signaling hubs and execute a wide range of
physiological functions when required (123). LLPS is a principal method for
condensing of biological macromolecules. This gives rise to a
resemblance of 'order' within the seemingly 'chaotic' cells and a
new framework for organization of macromolecules (121,124).
Inside the cell, LLPS formation first requires that
the macromolecule (protein, DNA, or RNA) in the solution reaches a
certain concentration threshold, knowing that an excessive
threshold can induce phase separation under suitable pH and
temperature conditions (121,125). Biological macromolecules exist
in two forms: A diluted state in solution and a concentrated state
in 'droplets' (126), and the
two forms are dynamically interchangeable as the relevant
conditions shift (3,127,128). Cells can regulate the
concentration at which specific proteins form droplets by altering
post-translational modifications (129), and then assemble into
biomolecular condensates by recruiting relevant macromolecular
components. A protein or RNA that acts as the phase separation
scaffold or starter in the assembly process is called the 'scaffold
molecule', and the assembled material is called the 'client
molecule' (130). The currently
recognized 'scaffold-client' molecular model of the assembly of
biomolecular condensates is described below (121,131). In addition, condensates are
also controlled by the protein quality control machinery, which
includes molecular chaperones and protein degradation systems
(132). With enriching in
specific proteins and other components, condensates can execute
various biological functions in different cellular compartments.
These effects can be attributed to condensation including the
promotion (133) or inhibition
of biochemical reactions (134), reduction of protein
concentrations (135),
detection of fluctuating in the environment (136) and mechanical forces (137).
In hypoxic environments or hypoxia-related disease
models, certain biomolecular condensates are equipped with cellular
regulatory functions and are used to regulate the metabolism of
cells or maintain their survival (Table I). This section summarizes the
activation mechanisms and physiological functions associated with
these hypoxia-induced condensates.
SGs are assemblies of non-translating messenger
ribonucleoprotein granules, various non-membrane-bound cellular
compartments that contain high concentrations of proteins and RNA
(138), and are close to UPR
(139). The formation of SGs is
facilitated by interactions between mRNAs and mRNA-binding
proteins, translation initiation factors, the 40S ribosomal subunit
(a myriad of RNA-binding proteins) and translationally stalled
mRNAs (139,140). Once the cells return to a
normal and non-stressful environment, SGs disperse and protein
translation is reinstated (141). Eukaryotic cells use SGs to
redirect limited resources from protein synthesis to survival and
stress resistance.
The core of the SG central node of this network
incorporates the G3BP SG assembly factor 1 (G3BP1), which serves as
a molecular switch instigating RNA-dependent LLPS in response to
elevated concentrations of free RNA in cells. G3BP1 is also capable
of modulating LLPS propensity via three different inherently
disordered regions. The core SG network can be simultaneously
reinforced or weakened by altering G3BP1-binding factors (142). The assembly activation cues of
SGs coalesce with UPR signals to create networks that maintain
protein homeostasis (139,143). The conventional assembly
process of SGs is mediated by eIF2 phosphorylation. The eIF2 kinase
family includes PERK, PKR, general control non-depressible 2
(GCN2), and HRI (144,145). In a hypoxic environment, eIF2
phosphorylation is induced by PERK and activated through UPR
signaling or the OMA1-DELE1-HRI pathway, which is initiated by
UPRmt (119). The
phosphorylation state of eIF2 is regulated by its interaction with
eIF2β, and this interaction inhibits the conversion of GDP to GTP
by eIF2β, resulting in a decrease in the concentration of ternary
complex eIF2-GTP-tRNA Met (146,147). Consequently, the RNA-binding
protein TIA1 and T-cell-restricted intracellular antigen-associated
protein (TIAR) stimulate the formation of the noncanonical 48S
preinitiation complex (148).
This complex, unable to recruit the 60S ribosomal subunit
translation, can be used for SG assembly (148-150).
In addition to being mediated by UPR activation,
hypoxia causes the assembly of SGs through several other pathways.
Hypoxia is frequently associated with nutrient scarcity, and
mammalian cells can sense alterations in amino acid levels through
the GCN2 and mTORC1 pathways (151). Amino acid deprivation inhibits
mTORC1-mediated protein translation but stimulates angiogenesis via
the GCN2-ATF4 amino acid starvation response pathway, that is
independent of HIF-1 (152,153). GCN2 also promotes eIF2
stimulation and collaborates with PERK to shield hypoxic cells from
apoptosis (154). Furthermore,
hypoxia generally triggers type I interferon (IFN) pathway
inhibition and reduces IFN secretion, which could lead to
uncontrolled double-stranded RNA (dsRNA) expression (155). As a stressor, dsRNA can incite
the phosphorylation of eIF2α via PKR. This phosphorylation results
in the formation of SGs, which serve as an antiviral core (156). To summarize, hypoxia instigates
the activation of SG assemblies (Fig. 3).
Hypoxia-induced SG assembly effectively improves
cell viability, which has been well demonstrated in the hypoxic
microenvironment of cancer. Apoptosis-related molecules that
accumulate within SGs assembled by cancer cells manifested
antiapoptotic effects (157),
and the development of hypoxia-induced SGs causes drug resistance
in cancer (158). By
pharmaceutically impeding hypoxia-induced SG formation in HeLa
cells, Timalsina et al (159) managed to decrease drug
resistance in hypoxic microenvironments. A study by Attwood et
al (160) showed that
hypoxia increased the number of late apoptotic/necrotic
glioblastoma cells during the raloxifene-induced delay in SG
dissolution. Liu et al (161) provided that hypoxic conditions
could result in FUS-circTBC1D14-associated SG formation in the
cytoplasm after PRMT1 modification, thus contributing to the
maintenance of cellular homeostasis and promoting tumor progression
in triple-negative breast cancer.
In rodent models, SGs were found to protect
hepatocytes against hypoxia-induced damage by reducing apoptosis.
With the increased expression of the SG marker proteins G3BP1 and
TIA-1, the degree of liver injury, HIF-1α and apoptosis induced by
acute liver failure decreases (162). In addition, Hu et al
(163) found that impaired SGs
are important in the pathogenesis of spinal muscular atrophy.
It is noteworthy that nematodes and rat
cardiomyocytes produced characteristic SGs in mitochondria
stimulated by sublethal hypoxia. Mitochondrial SGs are involved in
early mitochondrial pathology and are closely associated with UPRmt
(14).
Metabolic flux is an important intracellular change
that occurs during hypoxic stress. When cellular oxidative
phosphorylation is impaired by hypoxia, glycolysis becomes the
primary source of energy (172,173). Although the glycolytic pathway
has a shorter energy supply pathway, the total amount of ATP
produced is lower than that produced during oxidative
phosphorylation (174). To meet
the ATP required for survival and speed up the flow of glycolysis,
cells integrate the enzymes required for glycolysis and other
scaffold proteins through LLPS to form a special biomolecular
condensate, called a glycolytic body (G-body) (15,175).
Under hypoxic conditions, glycolytic enzymes are
compartmentalized into cytoplasmic structures (176), and analogous condensates form
were also found in C. elegans neurons (177). Therefore, Jin et al
(15) demonstrated that under
hypoxic conditions, cells assemble non-membrane organelles that
include glycolytic enzymes, called G-bodies. They also found that
glucose consumption increased, and that the level of glycolytic
intermediates decreased in cells with G bodies. It is noteworthy
that the formation of G-bodies increases the glycolytic output in
hypoxia (15) via glycolytic
enzymes such as phosphofructokinase, pyruvate kinase, acetyl-CoA
carboxylase and yeast pyruvate kinase Cdc19 (178-181). These enzymes can catalyze the
rate-limiting step in glycolysis and be utilized to increase the
glycolysis rate under hypoxic conditions. While the mechanism of
G-body activation has not been elucidated. Gregory et al
(182) detected hundreds of
RNA-binding proteins in G-bodies using genomic and proteomic
methods. The failure of nonspecific endonucleases to maintain the
structural integrity of G-bodies suggests that the assembly of
G-bodies replying to hypoxia is likely mediated by an RNA-dependent
phase separation mechanism (182). The enzymes involved in the
formation of G-body aggregates follow a specific order
post-nucleation, and the entry of each metabolic enzyme into the
G-body is tightly regulated (183). The multiple glycolysis enzymes
within phase separation may function to enhance the activity and
increase the reaction rate in energy production, thereby forming
'metabolons' during hypoxic stress (184).
Notably, cells that are unable to form G-bodies
undergo abnormal division and yield nonviable daughter cells during
hypoxia, and the formation of G-bodies represents a conserved
adaptive response that maintains the energy requirements of the
cells (15).
Fatty acids consist a major fuel in various cells.
The depletion of oxygen substrate severely inhibits the fatty acid
β oxidative energy pathway of the cell, and the accumulated excess
fatty acids are transformed into triglycerides for storage
(185,186). The ER participates in
synthesizing these triglycerides, which are subsequently stored in
biomolecular condensate called LDs (187). LDs are dynamic lipid
compartments that can effectively manage fluctuating cellular
lipids. Following oxygen restoration and activation of fatty acid
oxidation, LDs are broken down by neutral lipase, and the liberated
fatty acids serve as substrates for mitochondrial oxidation,
leading to energy production (188). LDs contain core lipid
components, and are surrounded by an amphipathic lipid layer
(189). Almost all organisms
synthesize LDs, whose formation is initiated by the synthesis of
neutral lipids (NLs) (190).
Overnutrition or various stressors prompts cells to produce NLs in
the ER bilayer (191,192), where the synthesized NLs mix
with phospholipids on the membrane and diffuse in the ER bilayer
(193). When the NL
concentration exceeds the nucleation threshold, LLPS drives LD
formation to prevent NL accumulation in the ER membrane (194).
LD formation and degradation are controlled by
numerous enzymes and LD-associated proteins. Hypoxia-inducible
LD-associated protein (HILPDA) is a paramount LD-associated protein
induced by HIF-1 and fatty acid expression. It localizes in the LDs
of several cell types, and is situated near the ER and LDs within
cells. HILPDA directly inhibits the activity of adipose
triglyceride lipase via physical interaction and encourages LD
accumulation by stimulating triglyceride synthesis (202,203). These findings suggest that
under hypoxic stress, not only proteins and RNA, but also lipids
can be orchestrated to assemble into specific molecular biopolymers
for survival.
Hypoxic stress can induce the formation of protein
condensates, which play a role in promoting basic biochemical
processes. For instance, prolyl hydroxylases are involved in
regulating molecular responses to oxygen availability. These
proteins hydroxylate HIF-α, enabling its ubiquitination and
degradation (204,205). Increased expression of HIF can
lead to the generation of ROS, which modulates HIF-α stabilization
in conjunction with prolyl hydroxylase domain proteins (PHD)
(206,207). The PDH family has a function in
regulating HIF through the condensation of PDH3, a protein
expressed in response to oxygen deprivation that contributes to
neural cell death. PDH3 forms subcellular condensates in the
presence of oxygen, but its condensation is notably decreased under
hypoxia (208,209). The formation of PDH3
condensates relies on microtubules and involves the integration of
components from the 26S proteasome, chaperones and ubiquitin. The
PHD2 condensates exhibit liquid characteristics similar to other
condensates (210). When PHD3
is actively expressed under normoxia, it leads to the condensation
of proteasome components, triggering apoptosis in HeLa cells.
Apoptosis occurs in cells prone to PHD3 condensation and is
observed before apoptosis (210).
In conclusion, cells initiate the assembly of
biomolecular condensates to sustain cell survival and regulate
metabolism in response to hypoxic conditions. A concise overview of
the crucial components and biological functions of hypoxic-related
biomolecular condensates is presented in Table I, which can provide valuable
information for future research on hypoxic-related diseases.
Exposure of cells to hypoxia leads to the
impairment of cytochrome C oxidase activity, resulting in the
generation of ROS and the inhibition of ATP synthesis (17,219,220).
ATP-driven protein chaperones and molecular motors
play crucial roles in activating molecular condensation and
regulating solubilization. ATP-dependent depolymerases are
responsible for dissolving aggregates and reordering them for
refolding or degradation (221,222). In yeast, the ATP-generating
enzyme Cdc19 is incorporated into SGs to form reversible amyloid
structures under stressful conditions (223,224). Rapid re-solubilization of these
amyloids is essential for ATP generation and subsequent breakdown
of SGs (180). Increasing
energy metabolism enhances Cdc19 re-solubilization in yeast, while
the recruitment and aggregation of the ATP-dependent chaperones
Hsp104 and Ssa2 can enhance the efficiency of solubilization
(225).
The formation of misfolded protein aggregates is
regulated by molecular chaperones. Small Hsp sequesters such as
yeast Hsp26 can promote misfolded protein aggregating, facilitating
subsequent refolding (226). In
yeast, the Hsp70 protein cooperates with Hsp104 disaggregate to
solubilize aggregated proteins with ATP (227). Energy-dependent processes or
molecular machinery also participate in regulating the extent of
fiber formation within condensates. These processes could restrict
the formation of structures when dynamic condensates are required,
and facilitate their formation and growth when static condensates
are necessary. This explains the reason numerous higher-order
assemblies contain molecular chaperones, ATP-dependent
depolymerases and molecular motors (131,228). A previous study in newborn rats
subjected to unilateral carotid ligation and then exposed to
hypoxia for 80 min showed varying levels of hsp72 mRNA expression
in the area of ATP reduction induced during hypoxia recovery
(229). In renal epithelial
cells, Hsp72 expression is increased in response to ATP depletion,
especially after thermal preconditioning (230). Other studies have shown that
hypoxia/reoxygenation or ATP depletion can reduce Hsp60 levels,
induce Bax transfer to mitochondria and cause apoptosis (231). Although it is unclear whether
ATP produced from glycolysis under hypoxia is inadequate to support
molecular chaperones, these results also suggest a strong link
between hypoxia-induced ATP depletion and changes in protein
chaperones.
With the role of ATP in driving enzymatic activity,
more direct evidence arises from the hydrophilic tripolyphosphate
and a relatively hydrophobic adenosine ring, which provide ATP with
amphiphilic properties (232,233). Patel et al (234) demonstrated that ATP could
prevent the liquid-liquid phase separation of FUS, and even
dissolve previous droplets within the liquid phase compartment.
This effect was also observed for TATA-Box Binding Protein
Associated Factor 15, heterogeneous nuclear Ribonucleoprotein A
(hnRNPA) 3 and phosphogluconolactonase 3 in the liquid phase
compartment. Increasing the ATP concentration to 2 mM in the
chamber achieved a similar solubilization effect by inhibiting
protein aggregate formation and maintaining protein solubility
(234). These findings provide
a new direction for understanding disorders associated with
aberrant amyloid aggregation or a hypoxic environment.
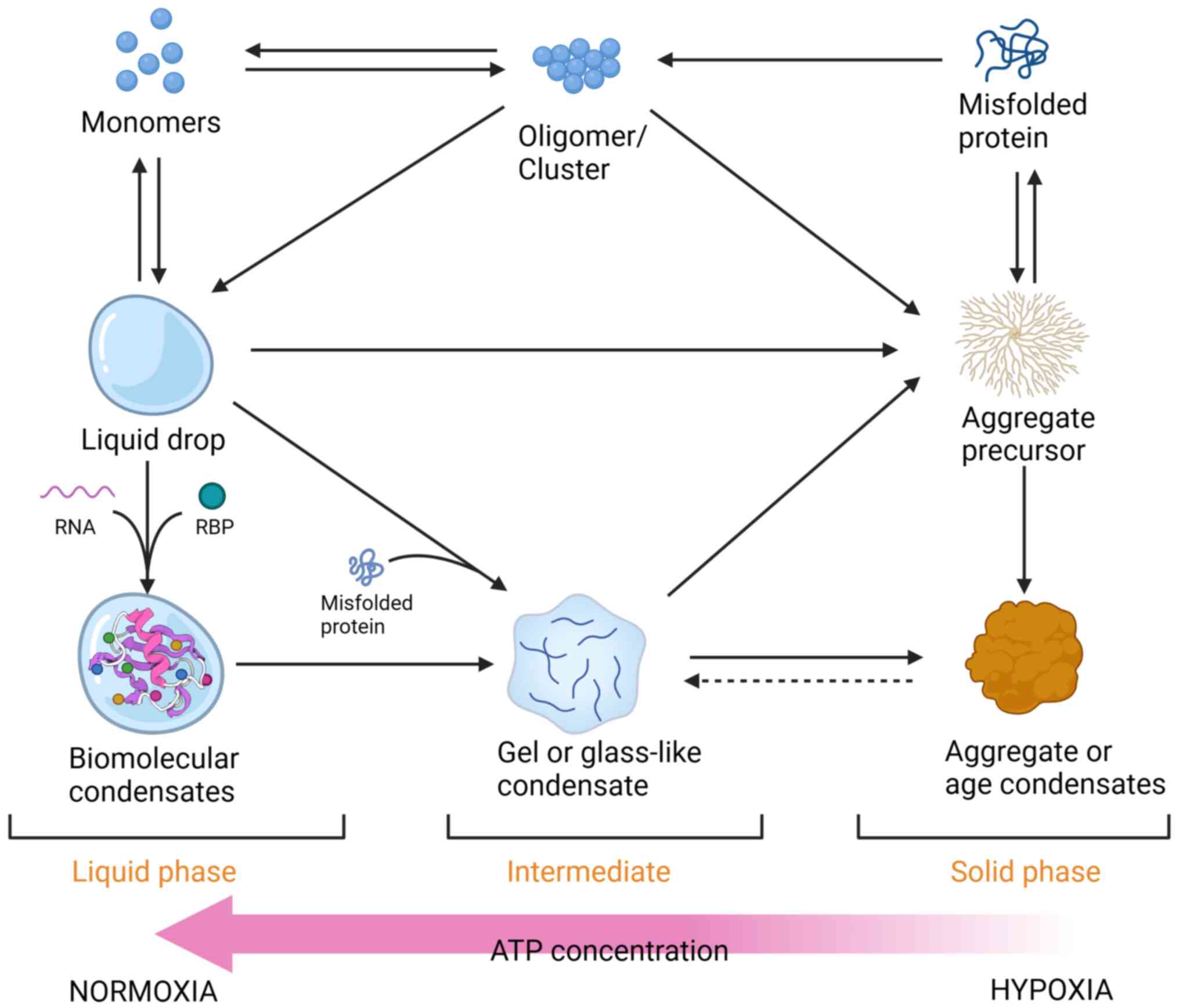
Previous studies have often focused on either the
assembly of aggregates or the formation of healthy molecular
condensates (13-15,53-55). However, they have rarely
considered them together, resulting in conceptual separation
between these macromolecular structures. Protein aggregates and
condensates are closely related because they both involve
higher-order assemblies with stoichiometric ratios (3).
Under different conditions, protein aggregates or
condensates can originate from intermediate clusters as aggregation
or droplet precursors. Another possible mechanism of protein
aggregation involves the initial formation of a droplet as an
intermediate aggregate, which undergoes a transition into a solid
state. Recent studies focusing on proteins such as FUS, hnRNPA1,
TDP43 and Tau associated with neurodegenerative diseases including
ALS, AD and PD have shown that liquid-phase condensation precedes
protein aggregation and amyloid formation (235-237). However, multiple studies have
suggested that cross-β (or amyloid) interactions are involved in
the formation of protein aggregates, and amyloid fibril formation
is frequently found in phase-separated proteins in vitro
(238,239). This means that the condensates
can be mutually converted to some extent.
The assembled molecular condensate itself can also
be transformed into a more solid-like state, a process known as
aging or hardening (136,240,241). The aging pathway of
agglomerates involves the gradual transition of glass-like
condensates from a fluid state to a more solid-like state. These
glass-like condensates undergo continuous changes in their
properties but do not fully solidify (242). Their behavior is influenced by
multiple factors, such as temperature and density, which affect
their propensity for undergoing transitions (243). Over time, glass-like
condensates show reduced elasticity and shrinkage, indicating an
increase in molecular contacts and aggregation (244,245). Another method of transformation
is gelation, with weak or strong interaction forces, and coacervate
components result in the formation of a physical gel such as the
gel formed by the extracellular matrix protein elastin (246,247). High concentrations of proteins,
lack of physiological chaperones and low water content are factors
contributing to condensate aging (248). Conversely, it has been revealed
that cells can prevent condensate aging by altering the condensate
composition (249), thereby
minimizing the potential for conformational changes in the protein
aggregation pathway. This regulatory process is often associated
with energy-consuming processes (249). However, the regulatory
mechanism that prevents aggregate aging is impaired in a hypoxic
environment with a notable decrease in ATP levels (37,90,221,228). Additionally, the cosolvent
effect of ATP is weakened under these conditions, resulting in an
increased propensity of proteins to aggregate. Several factors
collectively contribute to the aging of condensates (Fig. 4).
In conclusion, under physiological conditions,
dynamic equilibrium is maintained between the liquid and solid
phases within cells through the vigilant regulation of an intricate
network of molecular chaperones and regulatory mechanisms. However,
in various disease associated with hypoxia or hypoxic stimulation,
the function of molecular chaperones is disrupted, leading to the
accumulation of misfolded proteins and subsequent formation of
numerous aggregates, thereby compromising protein homeostasis. It
is hypothesized that this phenomenon is closely linked to
hypoxia-induced ATP depletion.
Therapeutic strategies aimed at preventing aberrant
protein aggregation and aging of biomolecular condensates have
shown promising results in managing ailments, particularly
neurodegenerative diseases (250). Currently, the US FDA has
endorsed a broad array of drugs capable of diminishing the
production of Aβ aggregates, which have been shown to be effective
at prolonging patient survival (251). The treatment mechanisms of
these drugs fall into the following three categories: i) Create
drug-protein chaperones that mimic the activity of natural
chaperones, or the synthesis of small molecules that assist in
stabilizing the folded protein conformation, thereby preventing
protein aggregation, and examples of such drugs include aducanumab
(252) and ALZT-OP1 (253); ii) indirect disruption of the
signaling pathway that governs aggregation, and several inhibitors,
including CNP520 (254) and
JNJ-54861911 (255), have been
created to target β-site amyloid precursor protein cleaving enzyme
signaling in an AD model; and iv) burgeoning approaches include
regulating hypoxia signals, addressing the hypoxic state, or
mitigating the chronic impact of hypoxia. Numerous small molecules
are being explored for their ability to alleviate the toxic effects
of protein aggregates induced by hypoxic stress. One of these
molecules, melatonin, effectively prevents chemical injury and
impedes the synthesis and formation of Aβ (256). The administration of vitamin
B6/B12/folate and choline notably improved in hypoxia-induced
memory impairment by effectively curtailing tau
hyperphosphorylation at several sites associated with AD (257).
Within the context of cancer models, a study
revealed that LLPS which alters some of the target proteins could
be used as a direction for cancer treatment. Our previous study
presented evidence that baicalin can serve as a potential therapy
for non-small cell lung cancer by altering the solid state of
cyclic GMP-AMP synthase (CGAS) in hypoxic microenvironments and
thereby improving mobility (259). Additionally, hypoxia has been
verified to inhibit the activation of the CGAS-stimulator of the
IFN gene signaling pathway (260). P53 is known as a tumor
suppressor protein. Once p53 is mutated, it will result in phase
separation phase transition (261), so it provides a promising
strategy to investigate new therapeutic targets focusing on p53
aggregates (262).
However, the limitations, cost and side effects of
current aggregate targeted therapy remain an issue in clinical
practice. It is widely acknowledged that both neurodegenerative
diseases and cancer are multifactorial conditions with numerous
hypotheses. Consequently, therapies targeting a single potential
factor are deemed unsatisfactory (263). For instance, aducanumab, an
aggregate-targeting drug for AD, exhibited adverse symptoms in ~25%
of patients with amyloid-related imaging abnormalities during a
comprehensive safety evaluation of a Phase 3 study involving 3,285
participants (264). The
mandatory exclusion criterion for aducanumab treatment is the
presence of abnormal amyloid proteins in the brain. However,
available data indicate that 20-40% of patients with early-stage AD
do not exhibit abnormal amyloid deposition, rendering aducanumab
ineffective for these individuals (265). Furthermore, there are
substantial risks associated with ALZT-OP1 due to previous clinical
failures and an incomplete understanding of the pathophysiological
role of Aβ in AD (253).
Therefore, in the case of hypoxic-related pathology
or hypoxic stress, it is crucial to acquire a comprehensive
understanding of the intricate interplay between hypoxic stress and
macromolecular aggregate and condense behaviors. Consequently, an
effective dual-pronged treatment strategy should be implemented:
Prevention of hypoxic injury and precise intervention targeting
aggregation and its behavior. This approach holds promising
therapeutic prospects for clinical intervention.
Hypoxic environments are stress conditions that can
lead to ATP depletion, cell acidification, disulfide bond
inhibition, ER mitochondrial stress and other reactions. The
accumulation of misfolded proteins induced by hypoxia promotes the
development of pathological aggregates, resulting in neuronal
damage. Disruption of protein homeostasis and accumulation of Aβ
are directly involved in this process. Various hypoxia-related
diseases, including AD, ALS, HIE, heart failure and cancer, are
characterized by disturbances in protein homeostasis.
Simultaneously, hypoxic pressure triggers the assembly of specific
biomolecular condensates in cells. These condensates, with their
distinct folding patterns, core types and recruited molecules, are
responsible for specific activities related to cell viability,
metabolic processes and protein homeostasis. Our understanding of
these aggregates may provide deeper insights into the interplay
between biochemical processes during hypoxic stress and
macromolecular phase separation. Interconversion between
aggregation and condensation occurs through intermediate states
under specific conditions. Misfolded proteins caused by hypoxia
tend to aggregate, accelerating the aging process of certain phase
separation droplets.
Efforts have been made to develop small molecules
that specifically target hypoxic stress and protein aggregation
mechanisms. They have already been employed in clinical
interventions for the treatment of hypoxic injuries and
neurodegenerative disorders. A comprehensive understanding of
aggregates and condensates provides insight into the biochemical
processes of hypoxic stress based on LLPS, which enhances the
understanding of the mechanisms underlying protein disturbances and
hypoxia-related diseases. In summary, the present study may also
open up new possibilities for the advancement of therapeutic
strategies and drug development.
However, studies of aggregates and LLPS condensates
still face limitations in clinical treatment and in vivo
investigations due to the lack of suitable testing methodologies.
The biological relevance of the aggregates was validated without
affecting LLPS-related parameters such as protein structure and
cellular physiology including pH, ionic strength and others.
Not applicable.
WX, LF and CL conceived and designed the review. CL
drafted the manuscript. BH revised and polished the manuscript. HY
and KW collated the literature. Data authentication is not
applicable. All authors read and approved the final version of the
manuscript.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
Not applicable.
The present review was sponsored by the National Natural Science
Foundation of China (grant no. 32371185), the Shanghai Science and
Technology Plan Project (grant no. 23010504200), the Shanghai
Talent Development Fund (grant no. 2020125), the Key Lab of
Exercise and Health Sciences of the Ministry of Education (Shanghai
University of Sports; grant no. 2022KF001) and the Shanghai Key Lab
of Human Performance (Shanghai University of Sports; grant no.
11DZ2261100).
|
1
|
Kuznetsova IM, Turoverov KK and Uversky
VN: What macromolecular crowding can do to a protein. Int J Mol
Sci. 15:23090–23140. 2014.
|
|
2
|
Gaudelet T, Malod-Dognin N and Pržulj N:
Higher-order molecular organization as a source of biological
function. Bioinformatics. 34:i944–i953. 2018.
|
|
3
|
Alberti S and Hyman AA: Biomolecular
condensates at the nexus of cellular stress, protein aggregation
disease and ageing. Nat Rev Mol Cell Biol. 22:196–213. 2021.
|
|
4
|
Savastano A, Flores D, Kadavath H, Biernat
J, Mandelkow E and Zweckstetter M: Disease-associated tau
phosphorylation hinders tubulin assembly within tau condensates.
Angew Chem Int Ed Engl. 60:726–730. 2021.
|
|
5
|
Amzallag E and Hornstein E: Crosstalk
between biomolecular condensates and proteostasis. Cells.
11:24152022.
|
|
6
|
Burtscher J, Mallet RT, Burtscher M and
Millet GP: Hypoxia and brain aging: Neurodegeneration or
neuroprotection? Ageing Res Rev. 68:1013432021.
|
|
7
|
Eltzschig HK and Carmeliet P: Hypoxia and
inflammation. N Engl J Med. 364:656–665. 2011.
|
|
8
|
Schito L and Rey S: Cell-autonomous
metabolic reprogramming in hypoxia. Trends Cell Biol. 28:128–142.
2018.
|
|
9
|
Kaufman DM, Wu X, Scott BA, Itani OA, Van
Gilst MR, Bruce JE and Crowder CM: Ageing and hypoxia cause protein
aggregation in mitochondria. Cell Death Differ. 24:1730–1738.
2017.
|
|
10
|
Dasmeh P and Wagner A: Yeast Proteins may
reversibly aggregate like amphiphilic molecules. J Mol Biol.
434:1673522022.
|
|
11
|
Wilson DM III, Cookson MR, Van Den Bosch
L, Zetterberg H, Holtzman DM and Dewachter I: Hallmarks of
neurodegenerative diseases. Cell. 186:693–714. 2023.
|
|
12
|
Kohler V and Andréasson C: Reversible
protein assemblies in the proteostasis network in health and
disease. Front Mol Biosci. 10:11555212023.
|
|
13
|
Spannl S, Tereshchenko M, Mastromarco GJ,
Ihn SJ and Lee HO: Biomolecular condensates in neurodegeneration
and cancer. Traffic. 20:890–911. 2019.
|
|
14
|
Sun CL, Van Gilst M and Crowder CM:
Hypoxia-induced mitochondrial stress granules. Cell Death Dis.
14:4482023.
|
|
15
|
Jin M, Fuller GG, Han T, Yao Y, Alessi AF,
Freeberg MA, Roach NP, Moresco JJ, Karnovsky A, Baba M, et al:
Glycolytic enzymes coalesce in G bodies under hypoxic stress. Cell
Rep. 20:895–908. 2017.
|
|
16
|
Saito K, Kondo E and Matsushita M:
MicroRNA 130 family regulates the hypoxia response signal through
the P-body protein DDX6. Nucleic Acids Res. 39:6086–6099. 2011.
|
|
17
|
Lee P, Chandel NS and Simon MC: Cellular
adaptation to hypoxia through hypoxia inducible factors and beyond.
Nat Rev Mol Cell Biol. 21:268–283. 2020.
|
|
18
|
Liu C, Gao Y, Barrett J and Hu B:
Autophagy and protein aggregation after brain ischemia. J
Neurochem. 115:68–78. 2010.
|
|
19
|
Hu BR, Martone ME, Jones YZ and Liu CL:
Protein aggregation after transient cerebral ischemia. J Neurosci.
20:3191–3199. 2000.
|
|
20
|
Wouters BG and Koritzinsky M: Hypoxia
signalling through mTOR and the unfolded protein response in
cancer. Nat Rev Cancer. 8:851–864. 2008.
|
|
21
|
Koumenis C and Wouters BG: 'Translating'
tumor hypoxia: Unfolded protein response (UPR)-dependent and
UPR-independent pathways. Mol Cancer Res. 4:423–436. 2006.
|
|
22
|
Gidalevitz T, Prahlad V and Morimoto RI:
The stress of protein misfolding: From single cells to
multicellular organisms. Cold Spring Harb Perspect Biol.
3:a0097042011.
|
|
23
|
Rahman A, Saikia B, Gogoi CR and Baruah A:
Advances in the understanding of protein misfolding and aggregation
through molecular dynamics simulation. Prog Biophys Mol Biol.
175:31–48. 2022.
|
|
24
|
Chiti F and Dobson CM: Protein misfolding,
functional amyloid, and human disease. Annu Rev Biochem.
75:333–366. 2006.
|
|
25
|
Riek R: The three-dimensional structures
of amyloids. Cold Spring Harb Perspect Biol. 9:a0235722017.
|
|
26
|
Balchin D, Hayer-Hartl M and Hartl FU: In
vivo aspects of protein folding and quality control. Science.
353:aac43542016.
|
|
27
|
Korte N, Nortley R and Attwell D: Cerebral
blood flow decrease as an early pathological mechanism in
Alzheimer's disease. Acta Neuropathol. 140:793–810. 2020.
|
|
28
|
Nortley R, Korte N, Izquierdo P,
Hirunpattarasilp C, Mishra A, Jaunmuktane Z, Kyrargyri V, Pfeiffer
T, Khennouf L, Madry C, et al: Amyloid β oligomers constrict human
capillaries in Alzheimer's disease via signaling to pericytes.
Science. 365:eaav95182019.
|
|
29
|
Park SH, Kukushkin Y, Gupta R, Chen T,
Konagai A, Hipp MS, Hayer-Hartl M and Hartl FU: PolyQ proteins
interfere with nuclear degradation of cytosolic proteins by
sequestering the Sis1p chaperone. Cell. 154:134–145. 2013.
|
|
30
|
Heck JW, Cheung SK and Hampton RY:
Cytoplasmic protein quality control degradation mediated by
parallel actions of the E3 ubiquitin ligases Ubr1 and San1. Proc
Natl Acad Sci USA. 107:1106–1111. 2010.
|
|
31
|
Ciechanover A and Kwon YT: Degradation of
misfolded proteins in neurodegenerative diseases: Therapeutic
targets and strategies. Exp Mol Med. 47:e1472015.
|
|
32
|
Rampelt H, Kirstein-Miles J, Nillegoda NB,
Chi K, Scholz SR, Morimoto RI and Bukau B: Metazoan Hsp70 machines
use Hsp110 to power protein disaggregation. EMBO J. 31:4221–4235.
2012.
|
|
33
|
Nillegoda NB, Kirstein J, Szlachcic A,
Berynskyy M, Stank A, Stengel F, Arnsburg K, Gao X, Scior A,
Aebersold R, et al: Crucial HSP70 co-chaperone complex unlocks
metazoan protein disaggregation. Nature. 524:247–251. 2015.
|
|
34
|
Gamerdinger M, Hajieva P, Kaya AM, Wolfrum
U, Hartl FU and Behl C: Protein quality control during aging
involves recruitment of the macroautophagy pathway by BAG3. EMBO J.
28:889–901. 2009.
|
|
35
|
Quintana-Gallardo L, Martín-Benito J,
Marcilla M, Espadas G, Sabidó E and Valpuesta JM: The cochaperone
CHIP marks Hsp70- and Hsp90-bound substrates for degradation
through a very flexible mechanism. Sci Rep. 9:51022019.
|
|
36
|
Nguyen VC, Deck CA and Pamenter ME: Naked
mole-rats reduce the expression of ATP-dependent but not
ATP-independent heat shock proteins in acute hypoxia. J Exp Biol.
222:jeb2112432019.
|
|
37
|
Mitra R, Wu K, Lee C and Bardwell JCA:
ATP-independent chaperones. Annu Rev Biophys. 51:409–429. 2022.
|
|
38
|
Benjamin IJ, Kröger B and Williams RS:
Activation of the heat shock transcription factor by hypoxia in
mammalian cells. Proc Natl Acad Sci USA. 87:6263–6267. 1990.
|
|
39
|
Degrossoli A, Colhone MC, Arrais-Silva WW
and Giorgio S: Hypoxia modulates expression of the 70-kD heat shock
protein and reduces Leishmania infection in macrophages. J Biomed
Sci. 11:847–854. 2004.
|
|
40
|
Hernández R, Blanco S, Peragón J, Pedrosa
JÁ and Peinado MÁ: Hypobaric hypoxia and reoxygenation induce
proteomic profile changes in the rat brain cortex. Neuromolecular
Med. 15:82–94. 2013.
|
|
41
|
Laquatra C, Sanchez-Martin C, Dinarello A,
Cannino G, Minervini G, Moroni E, Schiavone M, Tosatto S, Argenton
F, Colombo G, et al: HIF1α-dependent induction of the mitochondrial
chaperone TRAP1 regulates bioenergetic adaptations to hypoxia. Cell
Death Dis. 12:4342021.
|
|
42
|
Zhang J, Li H, Huang Z, He Y, Zhou X,
Huang T, Dai P, Duan D, Ma X, Yin Q, et al: Hypoxia attenuates
Hsp90 inhibitor 17-DMAG-induced cyclin B1 accumulation in
hepatocellular carcinoma cells. Cell Stress Chaperones. 21:339–348.
2016.
|
|
43
|
Hogg PJ: Disulfide bonds as switches for
protein function. Trends Biochem Sci. 28:210–214. 2003.
|
|
44
|
Braakman I and Hebert DN: Protein folding
in the endoplasmic reticulum. Cold Spring Harb Perspect Biol.
5:a0132012013.
|
|
45
|
Meyer AJ, Riemer J and Rouhier N:
Oxidative protein folding: State-of-the-art and current avenues of
research in plants. New Phytol. 221:1230–1246. 2019.
|
|
46
|
Narayan M: Revisiting the formation of a
native disulfide bond: Consequences for protein regeneration and
beyond. Molecules. 25:53372020.
|
|
47
|
Koritzinsky M, Levitin F, van den Beucken
T, Rumantir RA, Harding NJ, Chu KC, Boutros PC, Braakman I and
Wouters BG: Two phases of disulfide bond formation have differing
requirements for oxygen. J Cell Biol. 203:615–627. 2013.
|
|
48
|
Bulleid NJ: Disulfide bond formation in
the mammalian endoplasmic reticulum. Cold Spring Harb Perspect
Biol. 4:a0132192012.
|
|
49
|
Braakman I and Bulleid NJ: Protein folding
and modification in the mammalian endoplasmic reticulum. Annu Rev
Biochem. 80:71–99. 2011.
|
|
50
|
Saaranen MJ and Ruddock LW: Applications
of catalyzed cytoplasmic disulfide bond formation. Biochem Soc
Trans. 47:1223–1231. 2019.
|
|
51
|
Csordás G, Weaver D and Hajnóczky G:
Endoplasmic reticulum-mitochondrial contactology: Structure and
signaling functions. Trends Cell Biol. 28:523–540. 2018.
|
|
52
|
Shin Y and Brangwynne CP: Liquid phase
condensation in cell physiology and disease. Science.
357:eaaf43822017.
|
|
53
|
Wang M and Kaufman RJ: Protein misfolding
in the endoplasmic reticulum as a conduit to human disease. Nature.
529:326–335. 2016.
|
|
54
|
Hua C, Ju WN, Jin H, Sun X and Zhao G:
Molecular chaperones and hypoxic-ischemic encephalopathy. Neural
Regen Res. 12:153–160. 2017.
|
|
55
|
Gouveia M, Xia K, Colón W, Vieira SI and
Ribeiro F: Protein aggregation, cardiovascular diseases, and
exercise training: Where do we stand? Ageing Res Rev. 40:1–10.
2017.
|
|
56
|
Okada K, Minamino T, Tsukamoto Y, Liao Y,
Tsukamoto O, Takashima S, Hirata A, Fujita M, Nagamachi Y, Nakatani
T, et al: Prolonged endoplasmic reticulum stress in hypertrophic
and failing heart after aortic constriction: Possible contribution
of endoplasmic reticulum stress to cardiac myocyte apoptosis.
Circulation. 110:705–712. 2004.
|
|
57
|
Tannous P, Zhu H, Nemchenko A, Berry JM,
Johnstone JL, Shelton JM, Miller FJ Jr, Rothermel BA and Hill JA:
Intracellular protein aggregation is a proximal trigger of
cardiomyocyte autophagy. Circulation. 117:3070–3078. 2008.
|
|
58
|
Pattison JS, Sanbe A, Maloyan A, Osinska
H, Klevitsky R and Robbins J: Cardiomyocyte expression of a
polyglutamine preamyloid oligomer causes heart failure.
Circulation. 117:2743–2751. 2008.
|
|
59
|
Kim YE, Hipp MS, Bracher A, Hayer-Hartl M
and Hartl FU: Molecular chaperone functions in protein folding and
proteostasis. Annu Rev Biochem. 82:323–355. 2013.
|
|
60
|
Liang P, Zhang J and Wang B: Emerging
roles of ubiquitination in biomolecular condensates. Cells.
12:23292023.
|
|
61
|
Kaushik S and Cuervo AM: The coming of age
of chaperone-mediated autophagy. Nat Rev Mol Cell Biol. 19:365–381.
2018.
|
|
62
|
Park H, Kang JH and Lee S: Autophagy in
neurodegenerative diseases: A hunter for aggregates. Int J Mol Sci.
21:33692020.
|
|
63
|
Deng Z, Purtell K, Lachance V, Wold MS,
Chen S and Yue Z: Autophagy receptors and neurodegenerative
diseases. Trends Cell Biol. 27:491–504. 2017.
|
|
64
|
Menzies FM, Fleming A, Caricasole A, Bento
CF, Andrews SP, Ashkenazi A, Füllgrabe J, Jackson A, Jimenez
Sanchez M, Karabiyik C, et al: Autophagy and neurodegeneration:
Pathogenic mechanisms and therapeutic opportunities. Neuron.
93:1015–1034. 2017.
|
|
65
|
Frake RA, Ricketts T, Menzies FM and
Rubinsztein DC: Autophagy and neurodegeneration. J Clin Invest.
125:65–74. 2015.
|
|
66
|
Lin L, Yang P, Huang X and Zhang H, Lu Q
and Zhang H: The scaffold protein EPG-7 links cargo-receptor
complexes with the autophagic assembly machinery. J Cell Biol.
201:113–129. 2013.
|
|
67
|
Scott SV, Guan J, Hutchins MU, Kim J and
Klionsky DJ: Cvt19 is a receptor for the cytoplasm-to-vacuole
targeting pathway. Mol Cell. 7:1131–1141. 2001.
|
|
68
|
Zhang Y, Yan L, Zhou Z, Yang P, Tian E,
Zhang K, Zhao Y, Li Z, Song B, Han J, et al: SEPA-1 mediates the
specific recognition and degradation of P granule components by
autophagy in C. elegans. Cell. 136:308–321. 2009.
|
|
69
|
Ma X, Lu C, Chen Y, Li S, Ma N, Tao X, Li
Y, Wang J, Zhou M, Yan YB, et al: CCT2 is an aggrephagy receptor
for clearance of solid protein aggregates. Cell. 185:1325–1345.e22.
2022.
|
|
70
|
Cheng S, Huang Z, Jash S, Wu K, Saito S,
Nakashima A and Sharma S: Hypoxia-reoxygenation impairs
autophagy-lysosomal machinery in primary human trophoblasts
mimicking placental pathology of early-onset preeclampsia. Int J
Mol Sci. 23:56442022.
|
|
71
|
de Theije CC, Schols AMWJ, Lamers WH,
Neumann D, Köhler SE and Langen RCJ: Hypoxia impairs adaptation of
skeletal muscle protein turnover- and AMPK signaling during
fasting-induced muscle atrophy. PLoS One. 13:e02036302018.
|
|
72
|
Dao TP and Castañeda CA:
Ubiquitin-modulated phase separation of shuttle proteins: Does
condensate formation promote protein degradation? Bioessays.
42:e20000362020.
|
|
73
|
Cabe M, Rademacher DJ, Karlsson AB,
Cherukuri S and Bakowska JC: PB1 and UBA domains of p62 are
essential for aggresome-like induced structure formation. Biochem
Biophys Res Commun. 503:2306–2311. 2018.
|
|
74
|
Walter P and Ron D: The unfolded protein
response: From stress pathway to homeostatic regulation. Science.
334:1081–1086. 2011.
|
|
75
|
Kim R, Emi M, Tanabe K and Murakami S:
Role of the unfolded protein response in cell death. Apoptosis.
11:5–13. 2006.
|
|
76
|
Karagöz GE, Acosta-Alvear D and Walter P:
The unfolded protein response: detecting and responding to
fluctuations in the protein-folding capacity of the endoplasmic
reticulum. Cold Spring Harb Perspect Biol. 11:a0338862019.
|
|
77
|
Hetz C and Papa FR: The unfolded protein
response and cell fate control. Mol Cell. 69:169–181. 2018.
|
|
78
|
You K, Wang L, Chou CH, Liu K, Nakata T,
Jaiswal A, Yao J, Lefkovith A, Omar A, Perrigoue JG, et al: QRICH1
dictates the outcome of ER stress through transcriptional control
of proteostasis. Science. 371:eabb68962021.
|
|
79
|
Kopp MC, Larburu N, Durairaj V, Adams CJ
and Ali MMU: UPR proteins IRE1 and PERK switch BiP from chaperone
to ER stress sensor. Nat Struct Mol Biol. 26:1053–1062. 2019.
|
|
80
|
Hetz C: The unfolded protein response:
Controlling cell fate decisions under ER stress and beyond. Nat Rev
Mol Cell Biol. 13:89–102. 2012.
|
|
81
|
Bertolotti A, Zhang Y, Hendershot LM,
Harding HP and Ron D: Dynamic interaction of BiP and ER stress
transducers in the unfolded-protein response. Nat Cell Biol.
2:326–332. 2000.
|
|
82
|
Ye J, Rawson RB, Komuro R, Chen X, Davé
UP, Prywes R, Brown MS and Goldstein JL: ER stress induces cleavage
of membrane-bound ATF6 by the same proteases that process SREBPs.
Mol Cell. 6:1355–1364. 2000.
|
|
83
|
Haze K, Yoshida H, Yanagi H, Yura T and
Mori K: Mammalian transcription factor ATF6 is synthesized as a
transmembrane protein and activated by proteolysis in response to
endoplasmic reticulum stress. Mol Biol Cell. 10:3787–3799.
1999.
|
|
84
|
Schröder M and Kaufman RJ: The mammalian
unfolded protein response. Annu Rev Biochem. 74:739–789. 2005.
|
|
85
|
Münch C: The different axes of the
mammalian mitochondrial unfolded protein response. BMC Biol.
16:812018.
|
|
86
|
Binet F and Sapieha P: ER stress and
angiogenesis. Cell Metab. 22:560–575. 2015.
|
|
87
|
Sun LL, Chen CM, Zhang J, Wang J, Yang CZ
and Lin LZ: Glucose-regulated protein 78 signaling regulates
hypoxia-induced epithelial-mesenchymal transition in A549 cells.
Front Oncol. 9:1372019.
|
|
88
|
Raiter A, Weiss C, Bechor Z, Ben-Dor I,
Battler A, Kaplan B and Hardy B: Activation of GRP78 on endothelial
cell membranes by an ADAM15-derived peptide induces angiogenesis. J
Vasc Res. 47:399–411. 2010.
|
|
89
|
Wang Y, Alam GN, Ning Y, Visioli F, Dong
Z, Nör JE and Polverini PJ: The unfolded protein response induces
the angiogenic switch in human tumor cells through the PERK/ATF4
pathway. Cancer Res. 72:5396–5406. 2012.
|
|
90
|
Scheuner D, Song B, McEwen E, Liu C,
Laybutt R, Gillespie P, Saunders T, Bonner-Weir S and Kaufman RJ:
Translational control is required for the unfolded protein response
and in vivo glucose homeostasis. Mol Cell. 7:1165–1176. 2001.
|
|
91
|
Liu L, Cash TP, Jones RG, Keith B,
Thompson CB and Simon MC: Hypoxia-induced energy stress regulates
mRNA translation and cell growth. Mol Cell. 21:521–531. 2006.
|
|
92
|
Koumenis C, Naczki C, Koritzinsky M,
Rastani S, Diehl A, Sonenberg N, Koromilas A and Wouters BG:
Regulation of protein synthesis by hypoxia via activation of the
endoplasmic reticulum kinase PERK and phosphorylation of the
translation initiation factor eIF2alpha. Mol Cell Biol.
22:7405–7416. 2002.
|
|
93
|
Dewhirst MW, Cao Y and Moeller B: Cycling
hypoxia and free radicals regulate angiogenesis and radiotherapy
response. Nat Rev Cancer. 8:425–437. 2008.
|
|
94
|
Almendros I, Martínez-García MÁ,
Campos-Rodríguez F, Riveiro-Falkenbach E, Rodríguez-Peralto JL,
Nagore E, Martorell-Calatayud A, Hernández Blasco L, Bañuls Roca J,
Chiner Vives E, et al: Intermittent hypoxia is associated with high
hypoxia inducible factor-1α but not high vascular endothelial
growth factor cell expression in tumors of cutaneous melanoma
patients. Front Neurol. 9:2722018.
|
|
95
|
Yoon DW, So D, Min S, Kim J, Lee M,
Khalmuratova R, Cho CH, Park JW and Shin HW: Accelerated tumor
growth under intermittent hypoxia is associated with
hypoxia-inducible factor-1-dependent adaptive responses to hypoxia.
Oncotarget. 8:61592–61603. 2017.
|
|
96
|
Singleton DC and Harris AL: Targeting the
ATF4 pathway in cancer therapy. Expert Opin Ther Targets.
16:1189–1202. 2012.
|
|
97
|
Rouschop KM, van den Beucken T, Dubois L,
Niessen H, Bussink J, Savelkouls K, Keulers T, Mujcic H, Landuyt W,
Voncken JW, et al: The unfolded protein response protects human
tumor cells during hypoxia through regulation of the autophagy
genes MAP1LC3B and ATG5. J Clin Invest. 120:127–141. 2010.
|
|
98
|
Ye J, Kumanova M, Hart LS, Sloane K, Zhang
H, De Panis DN, Bobrovnikova-Marjon E, Diehl JA, Ron D and Koumenis
C: The GCN2-ATF4 pathway is critical for tumour cell survival and
proliferation in response to nutrient deprivation. EMBO J.
29:2082–2096. 2010.
|
|
99
|
Mujcic H, Nagelkerke A, Rouschop KM, Chung
S, Chaudary N, Span PN, Clarke B, Milosevic M, Sykes J, Hill RP, et
al: Hypoxic activation of the PERK/eIF2α arm of the unfolded
protein response promotes metastasis through induction of LAMP3.
Clin Cancer Res. 19:6126–6137. 2013.
|
|
100
|
Mudassar F, Shen H, O'Neill G and Hau E:
Targeting tumor hypoxia and mitochondrial metabolism with
anti-parasitic drugs to improve radiation response in high-grade
gliomas. J Exp Clin Cancer Res. 39:2082020.
|
|
101
|
Wheaton WW and Chandel NS: Hypoxia. 2.
Hypoxia regulates cellular metabolism. Am J Physiol Cell Physiol.
300:C385–C393. 2011.
|
|
102
|
Garcia-Bermudez J, Baudrier L, La K, Zhu
XG, Fidelin J, Sviderskiy VO, Papagiannakopoulos T, Molina H,
Snuderl M, Lewis CA, et al: Aspartate is a limiting metabolite for
cancer cell proliferation under hypoxia and in tumours. Nat Cell
Biol. 20:775–781. 2018.
|
|
103
|
Thomas LW, Staples O, Turmaine M and
Ashcroft M: CHCHD4 regulates intracellular oxygenation and
perinuclear distribution of mitochondria. Front Oncol.
7:712017.
|
|
104
|
Al-Mehdi AB, Pastukh VM, Swiger BM, Reed
DJ, Patel MR, Bardwell GC, Pastukh VV, Alexeyev MF and Gillespie
MN: Perinuclear mitochondrial clustering creates an oxidant-rich
nuclear domain required for hypoxia-induced transcription. Sci
Signal. 5:ra472012.
|
|
105
|
Kim H, Scimia MC, Wilkinson D, Trelles RD,
Wood MR, Bowtell D, Dillin A, Mercola M and Ronai ZA: Fine-tuning
of Drp1/Fis1 availability by AKAP121/Siah2 regulates mitochondrial
adaptation to hypoxia. Mol Cell. 44:532–544. 2011.
|
|
106
|
Melber A and Haynes CM: UPRmt
regulation and output: A stress response mediated by
mitochondrial-nuclear communication. Cell Res. 28:281–295.
2018.
|
|
107
|
Peter B, Waddington CL, Oláhová M,
Sommerville EW, Hopton S, Pyle A, Champion M, Ohlson M, Siibak T,
Chrzanow ska-Lightowlers ZMA, et al: Defective mitochondrial
protease LonP1 can cause classical mitochondrial disease. Hum Mol
Genet. 27:1743–1753. 2018.
|
|
108
|
Yan J, Sun CL, Shin S, Van Gilst M and
Crowder CM: Effect of the mitochondrial unfolded protein response
on hypoxic death and mitochondrial protein aggregation. Cell Death
Dis. 12:7112021.
|
|
109
|
Yoneda T, Benedetti C, Urano F, Clark SG,
Harding HP and Ron D: Compartment-specific perturbation of protein
handling activates genes encoding mitochondrial chaperones. J Cell
Sci. 117:4055–4066. 2004.
|
|
110
|
Durieux J, Wolff S and Dillin A: The
cell-non-autonomous nature of electron transport chain-mediated
longevity. Cell. 144:79–91. 2011.
|
|
111
|
Nargund AM, Pellegrino MW, Fiorese CJ,
Baker BM and Haynes CM: Mitochondrial import efficiency of ATFS-1
regulates mitochondrial UPR activation. Science. 337:587–590.
2012.
|
|
112
|
Nargund AM, Fiorese CJ, Pellegrino MW,
Deng P and Haynes CM: Mitochondrial and nuclear accumulation of the
transcription factor ATFS-1 promotes OXPHOS recovery during the
UPR(mt). Mol Cell. 58:123–133. 2015.
|
|
113
|
Fiorese CJ, Schulz AM, Lin YF, Rosin N,
Pellegrino MW and Haynes CM: The transcription factor ATF5 mediates
a mammalian mitochondrial UPR. Curr Biol. 26:2037–2043. 2016.
|
|
114
|
Quirós PM, Prado MA, Zamboni N, D'Amico D,
Williams RW, Finley D, Gygi SP and Auwerx J: Multi-omics analysis
identifies ATF4 as a key regulator of the mitochondrial stress
response in mammals. J Cell Biol. 216:2027–2045. 2017.
|
|
115
|
Michel S, Canonne M, Arnould T and Renard
P: Inhibition of mitochondrial genome expression triggers the
activation of CHOP-10 by a cell signaling dependent on the
integrated stress response but not the mitochondrial unfolded
protein response. Mitochondrion. 21:58–68. 2015.
|
|
116
|
Inigo JR and Chandra D: The mitochondrial
unfolded protein response (UPRmt): Shielding against
toxicity to mitochondria in cancer. J Hematol Oncol. 15:982022.
|
|
117
|
Sutandy FXR, Gößner I, Tascher G and Münch
C: A cytosolic surveillance mechanism activates the mitochondrial
UPR. Nature. 618:849–854. 2023.
|
|
118
|
Anderson NS and Haynes CM: Folding the
mitochondrial UPR into the integrated stress response. Trends Cell
Biol. 30:428–439. 2020.
|
|
119
|
Guo X, Aviles G, Liu Y, Tian R, Unger BA,
Lin YT, Wiita AP, Xu K, Correia MA and Kampmann M: Mitochondrial
stress is relayed to the cytosol by an OMA1-DELE1-HRI pathway.
Nature. 579:427–432. 2020.
|
|
120
|
Alberti S, Gladfelter A and Mittag T:
Considerations and challenges in studying liquid-liquid phase
separation and biomolecular condensates. Cell. 176:419–434.
2019.
|
|
121
|
Banani SF, Lee HO, Hyman AA and Rosen MK:
Biomolecular condensates: Organizers of cellular biochemistry. Nat
Rev Mol Cell Biol. 18:285–298. 2017.
|
|
122
|
Zhang H, Ji X, Li P, Liu C, Lou J, Wang Z,
Wen W, Xiao Y, Zhang M and Zhu X: Liquid-liquid phase separation in
biology: Mechanisms, physiological functions and human diseases.
Sci China Life Sci. 63:953–985. 2020.
|
|
123
|
Hirose T, Ninomiya K, Nakagawa S and
Yamazaki T: A guide to membraneless organelles and their various
roles in gene regulation. Nat Rev Mol Cell Biol. 24:288–304.
2023.
|
|
124
|
Brangwynne CP, Eckmann CR, Courson DS,
Rybarska A, Hoege C, Gharakhani J, Jülicher F and Hyman AA:
Germline P granules are liquid droplets that localize by controlled
dissolution/condensation. Science. 324:1729–1732. 2009.
|
|
125
|
Kim J, Lee H, Lee HG and Seo PJ: Get
closer and make hotspots: Liquid-liquid phase separation in plants.
EMBO Rep. 22:e516562021.
|
|
126
|
Alberti S, Saha S, Woodruff JB, Franzmann
TM, Wang J and Hyman AA: A user's guide for phase separation assays
with purified proteins. J Mol Biol. 430:4806–4820. 2018.
|
|
127
|
Shrinivas K and Brenner MP: Phase
separation in fluids with many interacting components. Proc Natl
Acad Sci USA. 118:e21085511182021.
|
|
128
|
Galves M, Rathi R, Prag G and Ashkenazi A:
Ubiquitin signaling and degradation of aggregate-prone proteins.
Trends Biochem Sci. 44:872–884. 2019.
|
|
129
|
Snead WT and Gladfelter AS: The control
centers of biomolecular phase separation: How membrane surfaces,
PTMs, and active processes regulate condensation. Mol Cell.
76:295–305. 2019.
|
|
130
|
Sanchez-Burgos I, Espinosa JR, Joseph JA
and Collepardo-Guevara R: Valency and binding affinity variations
can regulate the multilayered organization of protein condensates
with many components. Biomolecules. 11:2782021.
|
|
131
|
Jain S, Wheeler JR, Walters RW, Agrawal A,
Barsic A and Parker R: ATPase-modulated stress granules contain a
diverse proteome and substructure. Cell. 164:487–498. 2016.
|
|
132
|
Hipp MS, Kasturi P and Hartl FU: The
proteostasis network and its decline in ageing. Nat Rev Mol Cell
Biol. 20:421–435. 2019.
|
|
133
|
Case LB, Zhang X, Ditlev JA and Rosen MK:
Stoichiometry controls activity of phase-separated clusters of
actin signaling proteins. Science. 363:1093–1097. 2019.
|
|
134
|
Franzmann TM, Jahnel M, Pozniakovsky A,
Mahamid J, Holehouse AS, Nüske E, Richter D, Baumeister W, Grill
SW, Pappu RV, et al: Phase separation of a yeast prion protein
promotes cellular fitness. Science. 359:eaao56542018.
|
|
135
|
Klosin A, Oltsch F, Harmon T, Honigmann A,
Jülicher F, Hyman AA and Zechner C: Phase separation provides a
mechanism to reduce noise in cells. Science. 367:464–468. 2020.
|
|
136
|
Riback JA, Katanski CD, Kear-Scott JL,
Pilipenko EV, Rojek AE, Sosnick TR and Drummond DA:
Stress-triggered phase separation is an adaptive, evolutionarily
tuned response. Cell. 168:1028–1040.e19. 2017.
|
|
137
|
Shin Y, Chang YC, Lee DSW, Berry J,
Sanders DW, Ronceray P, Wingreen NS, Haataja M and Brangwynne CP:
Liquid nuclear condensates mechanically sense and restructure the
genome. Cell. 175:1481–1491.e13. 2018.
|
|
138
|
Spector DL: SnapShot: Cellular bodies.
Cell. 127:10712006.
|
|
139
|
Protter DSW and Parker R: Principles and
properties of stress granules. Trends Cell Biol. 26:668–679.
2016.
|
|
140
|
Damgaard CK and Lykke-Andersen J:
Translational coregulation of 5'TOP mRNAs by TIA-1 and TIAR. Genes
Dev. 25:2057–2068. 2011.
|
|
141
|
Gwon Y, Maxwell BA, Kolaitis RM, Zhang P,
Kim HJ and Taylor JP: Ubiquitination of G3BP1 mediates stress
granule disassembly in a context-specific manner. Science.
372:eabf65482021.
|
|
142
|
Yang P, Mathieu C, Kolaitis RM, Zhang P,
Messing J, Yurtsever U, Yang Z, Wu J, Li Y, Pan Q, et al: G3BP1 is
a tunable switch that triggers phase separation to assemble stress
granules. Cell. 181:325–345.e28. 2020.
|
|
143
|
Bartoszewska S and Collawn JF: Unfolded
protein response (UPR) integrated signaling networks determine cell
fate during hypoxia. Cell Mol Biol Lett. 25:182020.
|
|
144
|
Donnelly N, Gorman AM, Gupta S and Samali
A: The eIF2α kinases: Their structures and functions. Cell Mol Life
Sci. 70:3493–3511. 2013.
|
|
145
|
Wek RC, Jiang HY and Anthony TG: Coping
with stress: eIF2 kinases and translational control. Biochem Soc
Trans. 34:7–11. 2006.
|
|
146
|
Beilsten-Edmands V, Gordiyenko Y, Kung JC,
Mohammed S, Schmidt C and Robinson CV: eIF2 interactions with
initiator tRNA and eIF2B are regulated by post-translational
modifications and conformational dynamics. Cell Discov.
1:150202015.
|
|
147
|
Kedersha N and Anderson P: Stress
granules: Sites of mRNA triage that regulate mRNA stability and
translatability. Biochem Soc Trans. 30:963–969. 2002.
|
|
148
|
Kedersha N, Chen S, Gilks N, Li W, Miller
IJ, Stahl J and Anderson P: Evidence that ternary complex
(eIF2-GTP-tRNA(i) (Met))-deficient preinitiation complexes are core
constituents of mammalian stress granules. Mol Biol Cell.
13:195–210. 2002.
|
|
149
|
Anderson P and Kedersha N: Stressful
initiations. J Cell Sci. 115:3227–3234. 2002.
|
|
150
|
Anderson P and Kedersha N: Stress
granules: The tao of RNA triage. Trends Biochem Sci. 33:141–150.
2008.
|
|
151
|
Darnell AM, Subramaniam AR and O'Shea EK:
Translational control through differential ribosome pausing during
amino acid limitation in mammalian cells. Mol Cell. 71:229–243.e11.
2018.
|
|
152
|
Eleftheriadis T, Pissas G, Antoniadi G,
Liakopoulos V, Tsogka K, Sounidaki M and Stefanidis I: Differential
effects of the two amino acid sensing systems, the GCN2 kinase and
the mTOR complex 1, on primary human alloreactive CD4+
T-cells. Int J Mol Med. 37:1412–1420. 2016.
|
|
153
|
Longchamp A, Mirabella T, Arduini A,
MacArthur MR, Das A, Treviño-Villarreal JH, Hine C, Ben-Sahra I,
Knudsen NH, Brace LE, et al: Amino acid restriction triggers
angiogenesis via GCN2/ATF4 regulation of VEGF and H2S
production. Cell. 173:117–129.e14. 2018.
|
|
154
|
Liu Y, László C, Liu Y, Liu W, Chen X,
Evans SC and Wu S: Regulation of G(1) arrest and apoptosis in
hypoxia by PERK and GCN2-mediated eIF2alpha phosphorylation.
Neoplasia. 12:61–68. 2010.
|
|
155
|
Miar A, Arnaiz E, Bridges E, Beedie S,
Cribbs AP, Downes DJ, Beagrie RA, Rehwinkel J and Harris AL:
Hypoxia induces transcriptional and translational downregulation of
the type I IFN pathway in multiple cancer cell types. Cancer Res.
80:5245–5256. 2020.
|
|
156
|
Eiermann N, Haneke K, Sun Z, Stoecklin G
and Ruggieri A: Dance with the Devil: Stress granules and signaling
in antiviral responses. Viruses. 12:9842020.
|
|
157
|
Takahashi M, Higuchi M, Matsuki H, Yoshita
M, Ohsawa T, Oie M and Fujii M: Stress granules inhibit apoptosis
by reducing reactive oxygen species production. Mol Cell Biol.
33:815–829. 2013.
|
|
158
|
Lee AK, Klein J, Fon Tacer K, Lord T,
Oatley MJ, Oatley JM, Porter SN, Pruett-Miller SM, Tikhonova EB,
Karamyshev AL, et al: Translational repression of G3BP in cancer
and germ cells suppresses stress granules and enhances stress
tolerance. Mol Cell. 79:645–659.e9. 2020.
|
|
159
|
Timalsina S, Arimoto-Matsuzaki K, Kitamura
M, Xu X, Wenzhe Q, Ishigami-Yuasa M, Kagechika H and Hata Y:
Chemical compounds that suppress hypoxia-induced stress granule
formation enhance cancer drug sensitivity of human cervical cancer
HeLa cells. J Biochem. 164:381–391. 2018.
|
|
160
|
Attwood KM, Robichaud A, Westhaver LP,
Castle EL, Brandman DM, Balgi AD, Roberge M, Colp P, Croul S, Kim
I, et al: Raloxifene prevents stress granule dissolution, impairs
translational control and promotes cell death during hypoxia in
glioblastoma cells. Cell Death Dis. 11:9892020.
|
|
161
|
Liu Y, Liu Y, He Y, Zhang N, Zhang S, Li
Y, Wang X, Liang Y, Chen X, Zhao W, et al: Hypoxia-induced
FUS-circTBC1D14 stress granules promote autophagy in TNBC. Adv Sci
(Weinh). 10:e22049882023.
|
|
162
|
Li WY, Yang F, Li X, Wang LW and Wang Y:
Stress granules inhibit endoplasmic reticulum stress-mediated
apoptosis during hypoxia-induced injury in acute liver failure.
World J Gastroenterol. 29:1315–1329. 2023.
|
|
163
|
Hu L, Mao S, Lin L, Bai G, Liu B and Mao
J: Stress granules in the spinal muscular atrophy and amyotrophic
lateral sclerosis: The correlation and promising therapy. Neurobiol
Dis. 170:1057492022.
|
|
164
|
Youn JY, Dyakov BJA, Zhang J, Knight JDR,
Vernon RM, Forman-Kay JD and Gingras AC: Properties of stress
granule and P-body proteomes. Mol Cell. 76:286–294. 2019.
|
|
165
|
Kedersha N, Stoecklin G, Ayodele M, Yacono
P, Lykke-Andersen J, Fritzler MJ, Scheuner D, Kaufman RJ, Golan DE
and Anderson P: Stress granules and processing bodies are
dynamically linked sites of mRNP remodeling. J Cell Biol.
169:871–884. 2005.
|
|
166
|
Moon SL, Morisaki T, Khong A, Lyon K,
Parker R and Stasevich TJ: Multicolour single-molecule tracking of
mRNA interactions with RNP granules. Nat Cell Biol. 21:162–168.
2019.
|
|
167
|
Luo Y, Na Z and Slavoff SA: P-bodies:
Composition, properties, and functions. Biochemistry. 57:2424–2431.
2018.
|
|
168
|
Lee JI and Namkoong S: Stress granules
dynamics: Benefits in cancer. BMB Rep. 55:577–586. 2022.
|
|
169
|
Jud MC, Czerwinski MJ, Wood MP, Young RA,
Gallo CM, Bickel JS, Petty EL, Mason JM, Little BA, Padilla PA and
Schisa JA: Large P body-like RNPs form in C. elegans oocytes
in response to arrested ovulation, heat shock, osmotic stress, and
anoxia and are regulated by the major sperm protein pathway. Dev
Biol. 318:38–51. 2008.
|
|
170
|
Bett JS, Ibrahim AF, Garg AK, Kelly V,
Pedrioli P, Rocha S and Hay RT: The P-body component USP52/PAN2 is
a novel regulator of HIF1A mRNA stability. Biochem J. 451:185–194.
2013.
|
|
171
|
Carbonaro M, O'Brate A and Giannakakou P:
Microtubule disruption targets HIF-1alpha mRNA to cytoplasmic
P-bodies for translational repression. J Cell Biol. 192:83–99.
2011.
|
|
172
|
Gutierrez G: Cellular energy metabolism
during hypoxia. Crit Care Med. 19:619–626. 1991.
|
|
173
|
Hollinshead KE and Tennant DA:
Mitochondrial metabolic remodeling in response to genetic and
environmental perturbations. Wiley Interdiscip Rev Syst Biol Med.
8:272–285. 2016.
|
|
174
|
Newsholme EA and Start C: Regulation in
metabolism. John Wiley and Sons; New York and London: pp.
3491973
|
|
175
|
TeSlaa T, Bartman CR, Jankowski CSR, Zhang
Z, Xu X, Xing X, Wang L, Lu W, Hui S and Rabinowitz JD: The source
of glycolytic intermediates in mammalian tissues. Cell Metab.
33:367–378.e5. 2021.
|
|
176
|
Miura N, Shinohara M, Tatsukami Y, Sato Y,
Morisaka H, Kuroda K and Ueda M: Spatial reorganization of
Saccharomyces cerevisiae enolase to alter carbon metabolism under
hypoxia. Eukaryot Cell. 12:1106–1119. 2013.
|
|
177
|
Jang S, Nelson JC, Bend EG,
Rodríguez-Laureano L, Tueros FG, Cartagenova L, Underwood K,
Jorgensen EM and Colón-Ramos DA: Glycolytic enzymes localize to
synapses under energy stress to support synaptic function. Neuron.
90:278–291. 2016.
|
|
178
|
Webb BA, Dosey AM, Wittmann T, Kollman JM
and Barber DL: The glycolytic enzyme phosphofructokinase-1
assembles into filaments. J Cell Biol. 216:2305–2313. 2017.
|
|
179
|
Narayanaswamy R, Levy M, Tsechansky M,
Stovall GM, O'Connell JD, Mirrielees J, Ellington AD and Marcotte
EM: Widespread reorganization of metabolic enzymes into reversible
assemblies upon nutrient starvation. Proc Natl Acad Sci USA.
106:10147–10152. 2009.
|
|
180
|
Saad S, Cereghetti G, Feng Y, Picotti P,
Peter M and Dechant R: Reversible protein aggregation is a
protective mechanism to ensure cell cycle restart after stress. Nat
Cell Biol. 19:1202–1213. 2017.
|
|
181
|
Kohnhorst CL, Kyoung M, Jeon M, Schmitt
DL, Kennedy EL, Ramirez J, Bracey SM, Luu BT, Russell SJ and An S:
Identification of a multienzyme complex for glucose metabolism in
living cells. J Biol Chem. 292:9191–9203. 2017.
|
|
182
|
Fuller GG, Han T, Freeberg MA, Moresco JJ,
Ghanbari Niaki A, Roach NP, Yates JR III, Myong S and Kim JK: RNA
promotes phase separation of glycolysis enzymes into yeast G bodies
in hypoxia. Elife. 9:e484802020.
|
|
183
|
Yoshimura Y, Hirayama R, Miura N, Utsumi
R, Kuroda K, Ueda M and Kataoka M: Small-scale hypoxic cultures for
monitoring the spatial reorganization of glycolytic enzymes in
Saccharomyces cerevisiae. Cell Biol Int. 45:1776–1783. 2021.
|
|
184
|
Fuller GG and Kim JK: Compartmentalization
and metabolic regulation of glycolysis. J Cell Sci.
134:jcs2584692021.
|
|
185
|
Lu H, Gao Z, Zhao Z, Weng J and Ye J:
Transient hypoxia reprograms differentiating adipocytes for
enhanced insulin sensitivity and triglyceride accumulation. Int J
Obes (Lond). 40:121–128. 2016.
|
|
186
|
Gordon GB, Barcza MA and Bush ME: Lipid
accumulation of hypoxic tissue culture cells. Am J Pathol.
88:663–678. 1977.
|
|
187
|
Gross DA and Silver DL: Cytosolic lipid
droplets: from mechanisms of fat storage to disease. Crit Rev
Biochem Mol Biol. 49:304–326. 2014.
|
|
188
|
Lass A, Zimmermann R, Oberer M and Zechner
R: Lipolysis-a highly regulated multi-enzyme complex mediates the
catabolism of cellular fat stores. Prog Lipid Res. 50:14–27.
2011.
|
|
189
|
Farese RV Jr and Walther TC: Lipid
droplets finally get a little R-E-S-P-E-C-T. Cell. 139:855–860.
2009.
|
|
190
|
Thiam AR and Ikonen E: Lipid droplet
nucleation. Trends Cell Biol. 31:108–118. 2021.
|
|
191
|
Walther TC, Chung J and Farese RV Jr:
Lipid droplet biogenesis. Annu Rev Cell Dev Biol. 33:491–510.
2017.
|
|
192
|
Olzmann JA and Carvalho P: Dynamics and
functions of lipid droplets. Nat Rev Mol Cell Biol. 20:137–155.
2019.
|
|
193
|
Santinho A, Salo VT, Chorlay A, Li S, Zhou
X, Omrane M, Ikonen E and Thiam AR: Membrane curvature catalyzes
lipid droplet assembly. Curr Biol. 30:2481–2494.e6. 2020.
|
|
194
|
Zoni V, Khaddaj R, Campomanes P, Thiam AR,
Schneiter R and Vanni S: Lipid droplet biogenesis is driven by
liquid-liquid phase separation. bioRxiv. 7774662020.
|
|
195
|
Walther TC and Farese RV Jr: Lipid
droplets and cellular lipid metabolism. Annu Rev Biochem.
81:687–714. 2012.
|
|
196
|
Ward PS and Thompson CB: Signaling in
control of cell growth and metabolism. Cold Spring Harb Perspect
Biol. 4:a0067832012.
|
|
197
|
Baenke F, Peck B, Miess H and Schulze A:
Hooked on fat: The role of lipid synthesis in cancer metabolism and
tumour development. Dis Model Mech. 6:1353–1363. 2013.
|
|
198
|
Koizume S and Miyagi Y: Lipid droplets: A
key cellular organelle associated with cancer cell survival under
normoxia and hypoxia. Int J Mol Sci. 17:14302016.
|
|
199
|
Qiu B, Ackerman D, Sanchez DJ, Li B,
Ochocki JD, Grazioli A, Bobrovnikova-Marjon E, Diehl JA, Keith B
and Simon MC: HIF2α-dependent lipid storage promotes endoplasmic
reticulum homeostasis in clear-cell renal cell carcinoma. Cancer
Discov. 5:652–667. 2015.
|
|
200
|
Bailey AP, Koster G, Guillermier C, Hirst
EM, MacRae JI, Lechene CP, Postle AD and Gould AP: Antioxidant role
for lipid droplets in a stem cell niche of Drosophila. Cell.
163:340–353. 2015.
|
|
201
|
Rysman E, Brusselmans K, Scheys K,
Timmermans L, Derua R, Munck S, Van Veldhoven PP, Waltregny D,
Daniëls VW, Machiels J, et al: De novo lipogenesis protects cancer
cells from free radicals and chemotherapeutics by promoting
membrane lipid saturation. Cancer Res. 70:8117–8126. 2010.
|
|
202
|
de la Rosa Rodriguez MA and Kersten S:
Regulation of lipid droplet homeostasis by hypoxia inducible lipid
droplet associated HILPDA. Biochim Biophys Acta Mol Cell Biol
Lipids. 1865:1587382020.
|
|
203
|
de la Rosa Rodriguez MA, Deng L, Gemmink
A, van Weeghel M, Aoun ML, Warnecke C, Singh R, Borst JW and
Kersten S: Hypoxia-inducible lipid droplet-associated induces DGAT1
and promotes lipid storage in hepatocytes. Mol Metab.
47:1011682021.
|
|
204
|
Semenza GL: Hypoxia-inducible factors in
physiology and medicine. Cell. 148:399–408. 2012.
|
|
205
|
Watts ER and Walmsley SR: Inflammation and
hypoxia: HIF and PHD isoform selectivity. Trends Mol Med. 25:33–46.
2019.
|
|
206
|
Willson JA, Arienti S, Sadiku P, Reyes L,
Coelho P, Morrison T, Rinaldi G, Dockrell DH, Whyte MKB and
Walmsley SR: Neutrophil HIF-1α stabilization is augmented by
mitochondrial ROS produced via the glycerol 3-phosphate shuttle.
Blood. 139:281–286. 2022.
|
|
207
|
Chandel NS, McClintock DS, Feliciano CE,
Wood TM, Melendez JA, Rodriguez AM and Schumacker PT: Reactive
oxygen species generated at mitochondrial complex III stabilize
hypoxia-inducible factor-1alpha during hypoxia: A mechanism of O2
sensing. J Biol Chem. 275:25130–25138. 2000.
|
|
208
|
Hopfer U, Hopfer H, Jablonski K, Stahl RA
and Wolf G: The novel WD-repeat protein Morg1 acts as a molecular
scaffold for hypoxia-inducible factor prolyl hydroxylase 3 (PHD3).
J Biol Chem. 281:8645–8655. 2006.
|
|
209
|
Wong BW, Kuchnio A, Bruning U and
Carmeliet P: Emerging novel functions of the oxygen-sensing prolyl
hydroxylase domain enzymes. Trends Biochem Sci. 38:3–11. 2013.
|
|
210
|
Rantanen K, Pursiheimo J, Högel H, Himanen
V, Metzen E and Jaakkola PM: Prolyl hydroxylase PHD3 activates
oxygen-dependent protein aggregation. Mol Biol Cell. 19:2231–2240.
2008.
|
|
211
|
Theodoridis PR, Bokros M, Marijan D,
Balukoff NC, Wang D, Kirk CC, Budine TD, Goldsmith HD, Wang M,
Audas TE and Lee S: Local translation in nuclear condensate amyloid
bodies. Proc Natl Acad Sci USA. 118:e20144571182021.
|
|
212
|
Wang M, Tao X, Jacob MD, Bennett CA, Ho
JJD, Gonzalgo ML, Audas TE and Lee S: Stress-induced low complexity
RNA activates physiological amyloidogenesis. Cell Rep.
24:1713–1721.e4. 2018.
|
|
213
|
Standart N and Weil D: P-bodies: Cytosolic
droplets for coordinated mRNA storage. Trends Genet. 34:612–626.
2018.
|
|
214
|
Majerciak V, Zhou T, Kruhlak MJ and Zheng
ZM: RNA helicase DDX6 and scaffold protein GW182 in P-bodies
promote biogenesis of stress granules. Nucleic Acids Res.
51:9337–9355. 2023.
|
|
215
|
Hallacli E, Kayatekin C, Nazeen S, Wang
XH, Sheinkopf Z, Sathyakumar S, Sarkar S, Jiang X, Dong X, Di Maio
R, et al: The Parkinson's disease protein alpha-synuclein is a
modulator of processing bodies and mRNA stability. Cell.
185:2035–2056.e33. 2022.
|
|
216
|
Loll-Krippleber R and Brown GW: P-body
proteins regulate transcriptional rewiring to promote DNA
replication stress resistance. Nat Commun. 8:5582017.
|
|
217
|
Lavalée M, Curdy N, Laurent C, Fournié JJ
and Franchini DM: Cancer cell adaptability: Turning
ribonucleoprotein granules into targets. Trends Cancer. 7:902–915.
2021.
|
|
218
|
Tsai WC and Lloyd RE: Cytoplasmic RNA
granules and viral infection. Annu Rev Virol. 1:147–170. 2014.
|
|
219
|
Bargiela D, Burr SP and Chinnery PF:
Mitochondria and hypoxia: Metabolic crosstalk in cell-fate
decisions. Trends Endocrinol Metab. 29:249–259. 2018.
|
|
220
|
Taylor CT and Moncada S: Nitric oxide,
cytochrome C oxidase, and the cellular response to hypoxia.
Arterioscler Thromb Vasc Biol. 30:643–647. 2010.
|
|
221
|
Sathyanarayanan U, Musa M, Bou Dib P,
Raimundo N, Milosevic I and Krisko A: ATP hydrolysis by yeast
Hsp104 determines protein aggregate dissolution and size in vivo.
Nat Commun. 11:52262020.
|
|
222
|
Torrente MP and Shorter J: The metazoan
protein disaggregase and amyloid depolymerase system: Hsp110,
Hsp70, Hsp40, and small heat shock proteins. Prion. 7:457–463.
2013.
|
|
223
|
Jakobson CM and Jarosz DF: Metabolites
control stress granule disassembly. Nat Cell Biol. 23:1053–1055.
2021.
|
|
224
|
Grignaschi E, Cereghetti G, Grigolato F,
Kopp MRG, Caimi S, Faltova L, Saad S, Peter M and Arosio P: A
hydrophobic low-complexity region regulates aggregation of the
yeast pyruvate kinase Cdc19 into amyloid-like aggregates in vitro.
J Biol Chem. 293:11424–11432. 2018.
|
|
225
|
Cereghetti G, Wilson-Zbinden C, Kissling
VM, Diether M, Arm A, Yoo H, Piazza I, Saad S, Picotti P, Drummond
DA, et al: Reversible amyloids of pyruvate kinase couple cell
metabolism and stress granule disassembly. Nat Cell Biol.
23:1085–1094. 2021.
|
|
226
|
Haslbeck M, Miess A, Stromer T, Walter S
and Buchner J: Disassembling protein aggregates in the yeast
cytosol. The cooperation of Hsp26 with Ssa1 and Hsp104. J Biol
Chem. 280:23861–23868. 2005.
|
|
227
|
Glover JR and Lindquist S: Hsp104, Hsp70,
and Hsp40: A novel chaperone system that rescues previously
aggregated proteins. Cell. 94:73–82. 1998.
|
|
228
|
Cherkasov V, Hofmann S, Druffel-Augustin
S, Mogk A, Tyedmers J, Stoecklin G and Bukau B: Coordination of
translational control and protein homeostasis during severe heat
stress. Curr Biol. 23:2452–2462. 2013.
|
|
229
|
Kobayashi S and Welsh FA: Regional
alterations of ATP and heat-shock protein-72 mRNA following
hypoxia-ischemia in neonatal rat brain. J Cereb Blood Flow Metab.
15:1047–1056. 1995.
|
|
230
|
Oh DJ, Yu SH and Kang ET: Heat shock
protein expression in adenosine triphosphate depleted renal
epithelial cells. Korean J Intern Med. 19:149–154. 2004.
|
|
231
|
Gupta S and Knowlton AA: Cytosolic heat
shock protein 60, hypoxia, and apoptosis. Circulation.
106:2727–2733. 2002.
|
|
232
|
Eastoe J, Hatzopoulos MH and Dowding PJ:
Action of hydrotropes and alkyl-hydrotropes. Soft Matter.
7:5917–5925. 2011.
|
|
233
|
Subbarao CV, Chakravarthy IPK, Sai
Bharadwaj AVSL and Prasad KMM: Functions of hydrotropes in
solutions. Chem Eng Technol. 35:225–237. 2012.
|
|
234
|
Patel A, Malinovska L, Saha S, Wang J,
Alberti S, Krishnan Y and Hyman AA: ATP as a biological hydrotrope.
Science. 356:753–756. 2017.
|
|
235
|
Patel A, Lee HO, Jawerth L, Maharana S,
Jahnel M, Hein MY, Stoynov S, Mahamid J, Saha S, Franzmann TM, et
al: A liquid-to-solid phase transition of the ALS protein FUS
accelerated by disease mutation. Cell. 162:1066–1077. 2015.
|
|
236
|
Wegmann S, Eftekharzadeh B, Tepper K,
Zoltowska KM, Bennett RE, Dujardin S, Laskowski PR, MacKenzie D,
Kamath T, Commins C, et al: Tau protein liquid-liquid phase
separation can initiate tau aggregation. EMBO J. 37:e980492018.
|
|
237
|
Ray S, Singh N, Kumar R, Patel K, Pandey
S, Datta D, Mahato J, Panigrahi R, Navalkar A, Mehra S, et al:
α-Synuclein aggregation nucleates through liquid-liquid phase
separation. Nat Chem. 12:705–716. 2020.
|
|
238
|
Hughes MP, Sawaya MR, Boyer DR,
Goldschmidt L, Rodriguez JA, Cascio D, Chong L, Gonen T and
Eisenberg DS: Atomic structures of low-complexity protein segments
reveal kinked β sheets that assemble networks. Science.
359:698–701. 2018.
|
|
239
|
Luo F, Gui X, Zhou H, Gu J, Li Y, Liu X,
Zhao M, Li D, Li X and Liu C: Atomic structures of FUS LC domain
segments reveal bases for reversible amyloid fibril formation. Nat
Struct Mol Biol. 25:341–346. 2018.
|
|
240
|
Alberti S and Hyman AA: Are aberrant phase
transitions a driver of cellular aging? Bioessays. 38:959–968.
2016.
|
|
241
|
Harmon TS, Holehouse AS, Rosen MK and
Pappu RV: Intrinsically disordered linkers determine the interplay
between phase separation and gelation in multivalent proteins.
Elife. 6:e302942017.
|
|
242
|
Nakauchi Y, Nishinami S and Shiraki K:
Glass-like protein condensate for the long-term storage of
proteins. Int J Biol Macromol. 182:162–167. 2021.
|
|
243
|
Sadati M, Nourhani A, Fredberg JJ and
Taheri Qazvini N: Glass-like dynamics in the cell and in cellular
collectives. Wiley Interdiscip Rev Syst Biol Med. 6:137–149.
2014.
|
|
244
|
Parry BR, Surovtsev IV, Cabeen MT, O'Hern
CS, Dufresne ER and Jacobs-Wagner C: The bacterial cytoplasm has
glass-like properties and is fluidized by metabolic activity. Cell.
156:183–194. 2014.
|
|
245
|
Iadanza MG, Jackson MP, Hewitt EW, Ranson
NA and Radford SE: A new era for understanding amyloid structures
and disease. Nat Rev Mol Cell Biol. 19:755–773. 2018.
|
|
246
|
Choi JM, Holehouse AS and Pappu RV:
Physical principles underlying the complex biology of intracellular
phase transitions. Annu Rev Biophys. 49:107–133. 2020.
|
|
247
|
Roberts S, Dzuricky M and Chilkoti A:
Elastin-like polypeptides as models of intrinsically disordered
proteins. FEBS Lett. 589:2477–2486. 2015.
|
|
248
|
Garaizar A, Espinosa JR, Joseph JA,
Krainer G, Shen Y, Knowles TPJ and Collepardo-Guevara R: Aging can
transform single-component protein condensates into multiphase
architectures. Proc Natl Acad Sci USA. 119:e21198001192022.
|
|
249
|
Falahati H and Wieschaus E: Independent
active and thermodynamic processes govern the nucleolus assembly in
vivo. Proc Natl Acad Sci USA. 114:1335–1340. 2017.
|
|
250
|
Eisele YS, Monteiro C, Fearns C, Encalada
SE, Wiseman RL, Powers ET and Kelly JW: Targeting protein
aggregation for the treatment of degenerative diseases. Nat Rev
Drug Discov. 14:759–780. 2015.
|
|
251
|
Wilson MR and Zoubeidi A: Clusterin as a
therapeutic target. Expert Opin Ther Targets. 21:201–213. 2017.
|
|
252
|
Sevigny J, Chiao P, Bussière T, Weinreb
PH, Williams L, Maier M, Dunstan R, Salloway S, Chen T, Ling Y, et
al: The antibody aducanumab reduces Aβ plaques in Alzheimer's
disease. Nature. 537:50–56. 2016.
|
|
253
|
Lozupone M, Berardino G, Mollica A,
Sardone R, Dibello V, Zupo R, Lampignano L, Castellana F, Bortone
I, Stallone R, et al: ALZT-OP1: An experimental combination regimen
for the treatment of Alzheimer's disease. Expert Opin Investig
Drugs. 31:759–771. 2022.
|
|
254
|
Neumann U, Ufer M, Jacobson LH,
Rouzade-Dominguez ML, Huledal G, Kolly C, Lüönd RM, Machauer R,
Veenstra SJ, Hurth K, et al: The BACE-1 inhibitor CNP520 for
prevention trials in Alzheimer's disease. EMBO Mol Med.
10:e93162018.
|
|
255
|
Timmers M, Streffer JR, Russu A, Tominaga
Y, Shimizu H, Shiraishi A, Tatikola K, Smekens P, Börjesson-Hanson
A, Andreasen N, et al: Pharmacodynamics of atabecestat
(JNJ-54861911), an oral BACE1 inhibitor in patients with early
Alzheimer's disease: Randomized, double-blind, placebo-controlled
study. Alzheimers Res Ther. 10:852018.
|
|
256
|
Wongprayoon P and Govitrapong P: Melatonin
receptor as a drug target for neuroprotection. Curr Mol Pharmacol.
14:150–164. 2021.
|
|
257
|
Yu L, Chen Y, Wang W, Xiao Z and Hong Y:
Multi-vitamin B supplementation reverses hypoxia-induced tau
hyperphosphorylation and improves memory function in adult mice. J
Alzheimers Dis. 54:297–306. 2016.
|
|
258
|
Li S, Hafeez A, Noorulla F, Geng X, Shao
G, Ren C, Lu G, Zhao H, Ding Y and Ji X: Preconditioning in
neuroprotection: From hypoxia to ischemia. Prog Neurobiol.
157:79–91. 2017.
|
|
259
|
Zheng T, Liu H, Hong Y, Cao Y, Xia Q, Qin
C, Li M, Reiter RJ, Bai Y and Fan L: Promotion of liquid-to-solid
phase transition of cGAS by Baicalein suppresses lung
tumorigenesis. Signal Transduct Target Ther. 8:1332023.
|
|
260
|
Zhao F, Liu A, Gong X, Chen H, Wei J, Chen
B, Chen S, Yang R, Fan Y and Mao R: Hypoxia-induced RNASEH2A limits
activation of cGAS-STING signaling in HCC and predicts poor
prognosis. Tumori. 108:63–76. 2022.
|
|
261
|
Baugh EH, Ke H, Levine AJ, Bonneau RA and
Chan CS: Why are there hotspot mutations in the TP53 gene in human
cancers? Cell Death Differ. 25:154–160. 2018.
|
|
262
|
Ferretti GDS, Quarti J, Dos Santos G,
Rangel LP and Silva JL: Anticancer therapeutic strategies targeting
p53 aggregation. Int J Mol Sci. 23:110232022.
|
|
263
|
Wojtunik-Kulesza K, Rudkowska M and
Orzeł-Sajdłowska A: Aducanumab-hope or disappointment for
Alzheimer's disease. Int J Mol Sci. 24:43672023.
|
|
264
|
Salloway S, Chalkias S, Barkhof F, Burkett
P, Barakos J, Purcell D, Suhy J, Forrestal F, Tian Y, Umans K, et
al: Amyloid-related imaging abnormalities in 2 phase 3 studies
evaluating aducanumab in patients with early alzheimer disease.
JAMA Neurol. 79:13–21. 2022.
|
|
265
|
Rabinovici GD, Gatsonis C, Apgar C,
Chaudhary K, Gareen I, Hanna L, Hendrix J, Hillner BE, Olson C,
Lesman-Segev OH, et al: Association of amyloid positron emission
tomography with subsequent change in clinical management among
medicare beneficiaries with mild cognitive impairment or dementia.
JAMA. 321:1286–1294. 2019.
|