Introduction
Age-related macular degeneration (AMD) is a major
cause of visual impairment worldwide. As the world population ages,
the number of patients with AMD is expected to increase to 288
million by 2040, which will impose a heavy economic and social
burden on modern society (1).
Age, high blood pressure, atherosclerosis, diabetic retinopathy,
smoking, alcohol abuse and genetics are factors that increase AMD
risk. However, the oxidative stress, inflammation and choroidal
vascular dysfunction induced by these high-risk factors are
considered key pathological events in AMD pathogenesis (2). Retinal pigment epithelium (RPE) and
Müller cells are susceptible to oxidative damage due to their high
oxygen consumption during retinal metabolism (3-5).
Oxidative stress is the main cause of RPE cell senescence,
RPE-Bruch's membrane-choroid complex dysfunction, and ultimately
drusen formation (6).
Neovascularization, a typical sign of exudative AMD, may also be
due to RPE-Bruch's membrane-choroid complex dysfunction and drusen
formation, such that dry AMD can eventually progress to wet AMD
(7). Most patients with early
AMD have mild symptoms that progress relatively slowly; by the time
severe visual impairment has occurred, most patients have
progressed to having wet AMD. Uniform treatment protocols have been
developed for wet AMD, and a wide range of treatment options is
available with favorable results (8). Unfortunately, there is no unified
treatment protocol for early AMD, highlighting the growing demand
for personalized treatment of early AMD (9). Therefore, it is particularly
important to identify new personalized drugs or protocols for early
AMD intervention. Existing evidence indicates that early AMD
treatment should focus on rescuing retinal cells from oxidative
stress and inflammatory damage, especially RPE and Müller cells
(10,11). Several groups, including the
authors', have been focusing on the underlying mechanisms of
reactive oxygen species (ROS)-induced retinal cell damage and
developing drugs or therapeutic targets to inhibit or even reverse
this process (12,13) to achieve precise and personalized
treatment.
Research on personalized medicine in the field of
ophthalmology has progressed recently. Chemical compounds
originating from traditional Chinese herbal medicines are drawing
increasing attention in personalized medicine because of their
ability to influence certain pathways without systemic toxic
effects (14). Additionally,
compounds from traditional Chinese medicine can provide a broader
range of options for individualized treatment. Tripterygium
wilfordii is a traditional Chinese medicine widely used to
treat various inflammatory and autoimmune diseases such as
rheumatoid arthritis, Behçet's disease and multiple sclerosis
(15). Triptolide (Tl) and
triptonide (Tn) are active derivatives of Tripterygium
wilfordii that have attracted marked attention in recent years
(16). Tl has demonstrated
anti-inflammatory effects in the treatment of immune diseases by
activating the Kelch-like ECh-associated protein 1/nuclear factor
erythrocyte 2-related factor 2/antioxidant response element
(Keap1/Nrf2/ARE) signaling pathway. However, toxicity and side
effects have limited its clinical application (15). Previous studies did not find
toxic effects in mice treated with 20-fold the effective dose of
Tn, and subsequent studies confirmed Tn as a potential agent with
powerful anti-inflammatory and antioxidant effects (17,18). Although understanding of the role
of Tn in certain diseases has increased recently, its potential
activity against retinal oxidative damage remains unexplored.
The Keap1/Nrf2/ARE pathway is one of the most
critical defense mechanisms against oxidative stresses (19). Oxidative damage caused by various
factors can cause conformational changes in the Keap1-Cul3-E3
ubiquitin ligase and interfere with Nrf2 ubiquitination.
Subsequently, Nrf2 translocates to the nucleus by heterodimerizing
with the sMAF protein and binding to the ARE/electrophilic reaction
element, inducing a series of cell protective genes, such as heme
oxygenase-1 (HO1), NAD(P)H: quinone oxidoreductase 1 (NQO1), and
gamma-glutamyl-cysteine ligase catalytic subunits (20). Thus, the activation of Nrf2 may
lead to a remarkable antioxidant response and protect the cells
from oxidative stress damage (21).
Using in vitro and in vivo
experiments, the present study intended to verify whether a low Tn
concentration could effectively protect retinal cells from
oxidative damage, inhibit retinal inflammation, stabilize retinal
structure, and promote functional recovery; it was also aimed to
confirm Tn as a potential highly efficient Nrf2 signaling pathway
activator, thus providing strong evidence for AMD treatment, and to
some extent, address the clinical need for personalized
treatments.
Materials and methods
Chemicals and reagents
Tn and N-Methyl-D-aspartic acid (NMDA) were obtained
from Sigma-Aldrich; Merck KGaA. All antibodies used in the present
study were acquired from Cell Signaling Technology, Inc., Asbcam
and Thermo Fisher Scientific, Inc., as detailed in Table SI. All primers used in the
present study were provided by Sangon Biotech Co., Ltd., and their
sequences are listed in Table
SII.
Cell cultures
Human umbilical vein endothelial cells (HUVECs,
AC337632), human retinal capillary endothelial cells (HRCECs,
AC340334) and Müller cells (MIO-M1, YS1695C) were obtained from
Nanjing Saiyan Biotechnology Co., Ltd. and stably cultured in
Dulbecco's Modified Eagle's Medium (DMEM) (Gibco; Thermo Fisher
Scientific, Inc.) supplemented with 10% fetal bovine serum (Gibco;
Thermo Fisher Scientific, Inc.) in a humidified incubator
containing 5% CO2 at 37°C. ARPE-19 (cat. no. CL-0026)
cells were purchased from ProCell Life Science & Technology
Co., Ltd. and maintained in Ham's F12 nutrient medium (Gibco;
Thermo Fisher Scientific, Inc.) supplemented with 10% fetal bovine
serum in the same environment.
Model establishment and mice
grouping
In total, 50 C57BL/6 male mice (6-8 weeks old;
weight, 20-25 g) provided by Nanjing Junke Bioengineering Co., Ltd.
were used for the in vivo studies. All animals received
ad libitum access to food and water in a pathogen-free
environment with a constant temperature (22±2°C) and (50±10%)
humidity, and maintained in a 12/12-h light/dark cycle during the
experiment. The animal experimental procedures used in the present
study were approved (approval no. 2303048) by the Ethics Committee
of Nanjing Medical University (Nanjing, China).
In the establishment of the animal model, the
methods and criteria in the previous authoritative literature were
strictly followed (22-25). The animals were randomly assigned
to one of five groups: i) control; ii) light-induced retinal
neurodegeneration model: Mice were acclimatized to a 12-h dark
environment before the experiments, and they were exposed to 8000
lX of white light for 12 h/day for 7 days after mydriasis; iii) Tn
+ light damage: 2 μl of Tn (1 mg/ml) was intravitreally
injected 24 h before exposure to light; iv) NMDA: 20 nmol of NMDA
was intravitreally injected 2 days before the experiments; and v)
Tn + NMDA: 2 μl of Tn (1 mg/ml) was intravitreally injected
24 h before the NMDA treatment. All mice were euthanized by
cervical dislocation after anesthesia with 90 mg/kg ketamine and
7.5 mg/kg xylazine injected intraperitoneally, and the retinal
tissue was subsequently removed.
Cell viability and cytotoxicity
assays
Cell viability tests were performed using MTT and
CCK-8 assays (Biosharp Life Sciences). Cells were seeded in 96-well
plates, with 100 μl of cell suspension containing 5,000
cells injected into each well, and five parallel replicates were
prepared. Cells were incubated with 25, 50, 100, 250, or 500 nM Tn
for 24 h at 37°C. For the CCK-8 assay, post-incubation, a precise
volume of 10 μl of CCK-8 reagent was dispensed into each
designated well, followed by a subsequent incubation period of 1 h
at 37°C. This was succeeded by spectrophotometric measurement of
the absorbance at a wavelength of 450 nm to assess cell viability.
For the MTT assay, upon completion of the initial incubation
period, 50 μl of MTT solution were administered to each
well. The plates were then incubated at 37°C for another 4 h to
reduce the MTT to formazan. Subsequently, the supernatant was
aspirated without disturbing the cell monolayer, and 150 μl
of DMSO was added to each well to solubilize the crystals. The
plates were then shaken for 10 min on a shaker. The absorbance was
spectrophotometrically measured at 490 nm to assess
cytotoxicity.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
The Müller and ARPE-19 cells were treated with
different Tn concentration levels 24 h before RT-qPCR assays. RNA
was extracted from cells and tissues using Trizol™ reagent (Thermo
Fisher Scientific, Inc.), and followed the manufacturer's protocol
for reverse transcription using PrimeScript RT Reagent kit (cat.
no. RR037A; Takara Bio USA, Inc.) at 37°C for 15 min and 85°C for 5
sec. RT-qPCR was performed using the SYBR Green qPCR SuperMix kit
(Invitrogen; Thermo Fisher Scientific, Inc.) with a PikoReal
Real-Time PCR System (Thermo Fisher Scientific, Inc.). The
thermocycling conditions were as follows: Initial denaturation at
95°C for 2 min, followed by 35 cycles at 94°C for 45 sec, 56°C for
30 sec, and 72°C for 45 sec. Glyceraldehyde-3-phosphate
dehydrogenase was used as a control. All data were analyzed using
the 2−ΔΔCq method (26).
ROS estimation
ROS levels were measured using a ROS Assay kit (cat.
no. S0033S; Beyotime Institute of Biotechnology) according to the
manufacturer's protocol. Cells were treated with 50 nM Tn at 37°C
for 6 h or 8 h at a density of 5×105 cells/ml, and then
with H2O2, followed by incubation of cells
with DCFH-DA (10 μM) for 30 min at 37°C. Finally, flow
cytometry was used to detect fluorescence intensity.
Dihydroethidium (DHE) staining was used to determine the level of
super oxidation in ARPE-19 cells using a DHE staining kit (cat. no.
ab145360; Abcam), according to the manufacturer's protocols.
Flow cytometry and apoptosis
detection
ARPE-19 and Müller cells were incubated with Tn at
10, 25, 50, and 100 nM for 6 h at 37°C at a density of
5×105 cells/ml before stimulation with
H2O2 (300 μM/ml). The cell suspension
was counter-stained with fluorescein isothiocyanate-labeled Annexin
V and propidium iodide (PI) for 10 min at 37°C in the dark.
Apoptosis was measured using a CytoFLEX flow cytometer (Beckman
Coulter, Inc.) and analyzed using CytExpert 2.2 (Beckman Coulter,
Inc.). Early apoptosis was detected using the JC-1 staining kit
(cat. no. C2006; Beyotime Institute of Biotechnology) according to
the manufacturer's protocol.
Western blotting
Western blot assay protocols were applied as
previously described (12).
Following treatment, cells were collected and lysed using RIPA
lysis buffer (Beyotime Institute of Biotechnology) to extract
proteins. Protein concentration was determined using the BCA assay
to ensure consistent protein loading across samples. Protein
samples were mixed with SDS-PAGE loading buffer and denatured by
boiling. During the electrophoresis phase, equal quantities of
proteins (20 μg/lane) were separated using a 10% SDS-PAGE
gel. Subsequently, the proteins on the gel were transferred onto a
polyvinylidene fluoride membranes. After the transfer, the membrane
was blocked with 5% non-fat milk for 2 h at room temperature on a
shaking platform. It was then incubated with primary antibodies
overnight at 4°C with gentle agitation. On the following day, the
primary antibodies were removed, and the membrane was incubated
with secondary antibodies for 1 h at room temperature. According to
the instructions of the ECL kit (Biosharp Life Sciences), the
luminescent substrates A and B were mixed in equal volumes under
light-protected conditions to prepare the luminescent working
solution. The solution was then evenly applied to the membrane and
allowed to incubate for at least 1 min. Finally, the membrane was
scanned using the Tannon 5200 (Tanon Science and Technology Co.,
Ltd.) equipment to observe the protein bands, and ImageJ software
(https://imagej.net; National Institutes of Health) was
used for quantitative densitometric analysis of the protein
bands.
Immunofluorescence staining of retinal
tissues
Retinal tissues from the different groups were fixed
with 4% paraformaldehyde at 4°C overnight and dehydrated in 30%
sucrose for 24 h. The retinal tissues were then embedded in the
compound at the optimal cutting temperature and stored at −80°C
after being cut into 10-μm sections at −25°C. The sections
were incubated for 45 min at 37°C in 5% bovine serum albumin
(neoFroxx GmbH) in phosphate buffered saline containing 0.5% Triton
X-100 and then incubated with anti-NeuN primary antibodies
overnight at 4°C and then washed with phosphate-buffered saline
containing 0.05% Tween, and incubated with the Alexa Fluor™ 647
secondary antibody (1:1,000; Thermo Fisher Scientific, Inc.) for 4
h at room temperature and shielded from light. Finally, the
sections were observed and images were captured under a
fluorescence microscope (Olympus Corporation).
Hematoxylin and eosin (H&E) staining
of retinal tissues
H&E staining kit (cat. no. C0105S; Beyotime
Institute of Biotechnology) was used to examine the retinal layers
in light-damaged and NMDA-treated mice. The eyes were fixed in 4%
paraformaldehyde at 4°C overnight. They were then embedded in
paraffin and cut into 5 μm-thick slices, which were securely
adhered to glass slides. After dewaxing and hydration, the sections
were stained in hematoxylin solution for 10 min at room
temperature, followed by eosin staining for 1 min. The stained
sections were dehydrated, cleared, and mounted with neutral balsam
(Biosharp Life Sciences). The sections were observed and
photographed under a fluorescence microscope.
Terminal deoxynucleotidyl
transferase-mediated dUTP nick-end labeling (TUNEL) assay
The TUNEL Apoptosis Assay kit (cat. no. C1091;
Beyotime Institute of Biotechnology) was used to detect retinal
cell apoptosis in light-damaged and NMDA-treated mice. Eyes were
preserved in 4% paraformaldehyde and fixed overnight at 4°C.
Following this, the specimens were embedded in paraffin, sectioned
into 5 μm-thick slices and firmly adhered to the glass
slides. Post-dewaxing and hydration, the sections underwent
protease K (Beyotime Institute of Biotechnology) treatment at 37°C
for 30 min. Subsequently, they were incubated with the TUNEL
reaction mixture at 37°C for 1 h in a light-protected environment.
To visualize the nuclei, cells were stained with 5 μg/ml of
4',6-diamidino-2-phenylindole (DAPI) (Beyotime Institute of
Biotechnology) for 10 min at room temperature, maintaining light
protection throughout. Sections were observed under a fluorescence
microscope with a 1.1-mm field of view, and images were captured
from three randomly selected fields of view for each section.
Apoptotic retinal cells were clearly labeled with green
fluorescence, while the nuclei showed blue fluorescence due to DAPI
staining.
Electroretinography (ERG)
The experimental mice were acclimated to the dark
for at least 12 h before changes in retinal electrical activity
were evaluated. The pupils of the mice were dilated using a 1%
tropicamide solution (Alcon). ERGs were recorded in both eyes by
placing a wire electrode (Nihon Kohden) contacting the cornea with
the reference electrode in the forehead and ground electrode in the
tail. Data were analyzed for a- and b-waves in each group.
Cell functional assays
The 5-ethynyl-2'-deoxyuridine (EdU) staining,
Transwell migration, invasion and tube formation assays were
performed to observe the proliferative ability of endothelial
cells. HUVECs were pretreated with Tn at 250 and 500 nM, followed
by stimulation with 10 ng/ml vascular endothelial growth factor
(VEGF) in 24-well plates for 24 h. Endothelial cells were incubated
with EdU (Beyotime Institute of Biotechnology) at 37°C for 2 h and
images were captured under a fluorescence microscope after fixation
and rinsing. Transwell migration and invasion assays were performed
to evaluate their influence on endothelial cell migration and
invasion. For the migration assay, no Matrigel (Corning, Inc.) was
added to the upper chamber of the Transwell inserts (Corning,
Inc.). For the invasion assay, the Matrigel was diluted with DMEM
medium at a ratio of 1:8, and 100 μl of the diluted Matrigel
was evenly spread onto the upper chamber of the Transwell inserts,
which was then incubated at 37°C for 3 h. Subsequently, treated
cells were suspended in serum-free DMEM medium and adjusted to a
density of 1×105 cells, and then seeded onto the upper
chamber of Transwell inserts of 8.0-μm pore size treated or
untreated with Matrigel. The lower chamber was filled with 600
μl of DMEM medium containing 10% serum to support cell
migration. After incubation, the migratory cells were fixed with
methanol for 10 min at room temperature, stained with 0.5% crystal
violet solution for 30 min, and counted under a light microscope.
Tube formation assays were designed to evaluate the angiogenic
ability of endothelial cells. After the same treatment as
aforementioned, the cells were added to Matrigel for 6 h. The
capillary network was observed using a bright-field microscope. All
results were quantified using the ImageJ software (https://imagej.net).
Laser-induced choroidal
neovascularization (CNV)
A total of 3 days before laser irradiation, Tn and
normal saline injections were performed in the vitreous cavity of
mice. CNV was induced using a 532-nm laser, as previously described
(27). The mice were
anesthetized with ketamine (90 mg/kg) and xylazine (7.5 mg/kg) by
intraperitoneal injection, followed by pupil dilation. Laser
photocoagulation was then performed using a laser photocoagulator
(75 μm spot size, 100 msec duration, and 100 mW) to rupture
the Bruch's membrane. A 10-mm coverslip was used to view the
retinal vessels. Choroids were harvested for immunohistochemical
staining 7 days after photocoagulation. Mouse eyes of different
groups were fixed in 4% paraformaldehyde at 4°C for 1 h. The
choroids were isolated by microsurgery and flat laid on slides. The
flat mounts were incubated in 5% bovine serum albumin in phosphate
buffered saline containing 1% Triton X-100 at 37°C for 45 min.
Subsequently, flat mounts were incubated with isolin B4 (iB4; cat.
no. I21411; 1:50; Thermo Fisher Scientific, Inc.) overnight at 4°C,
protected from light. The next day, the flat mounts were washed
with phosphate-buffered saline containing 0.05% Tween and viewed
under the fluorescence microscope and images were captured.
Statistical analyses
All data for the present study was obtained from at
least three independent experiments and expressed as the mean ±
standard error of the mean. In conducting statistical difference
analysis, the unpaired Student's t-test was used to evaluate the
disparity between two data groups. For comparisons involving
multiple data groups, ANOVA was first used to examine the overall
variability among the groups, followed by Tukey's multiple
comparisons test to further identify the groups that exhibited
significant differences. Data analyses were conducted using
GraphPad Prism 8.0.0 for Windows (GraphPad Software; Dotmatics).
P<0.05 was considered to indicate a statistically significant
difference.
Results
Tn: Novel and efficient activator of Nrf2
signaling
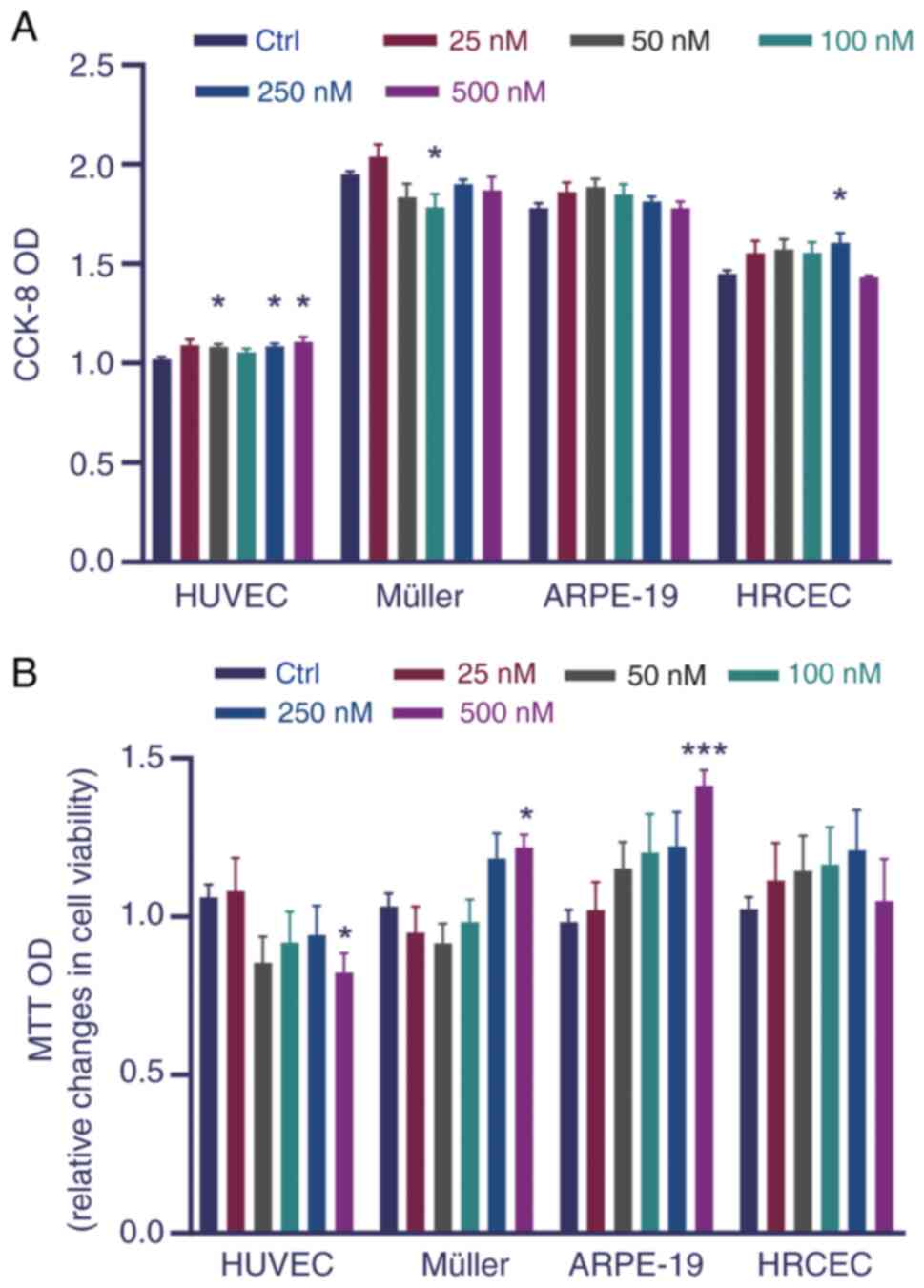
The potential effects of Tn on the viability of
HUVECs, HRCECs and Müller and ARPE-19 cells were first analyzed to
evaluate its influence on cytotoxicity. No pronounced reduction in
cell viability or significant cytotoxicity were observed in
vitro (Fig. 1A and B).
Hence, Tn 10-100 nM was used in subsequent experiments.
Nrf2/Keap1/ARE signaling is widely acknowledged as
one of the most critical antioxidant signaling pathways. The Nrf2
cascade protects against oxidative injury in vitro (19). To investigate whether Tn
activates the Nrf2/Keap1 signaling pathway, the mRNA expression
levels of Nrf2, Keap1 and Nrf2 downstream genes, including HO1 and
NQO1, were measured in Müller (Fig.
2A-D) and ARPE-19 cells (Fig.
2E-H). The RT-qPCR results showed that following Tn
administration, the mRNA levels of Nrf2, HO1 and NQO1 increased
significantly in Müller and ARPE-19 cells. By contrast, the mRNA
level of Keap1 was significantly reduced in Tn-treated cells.
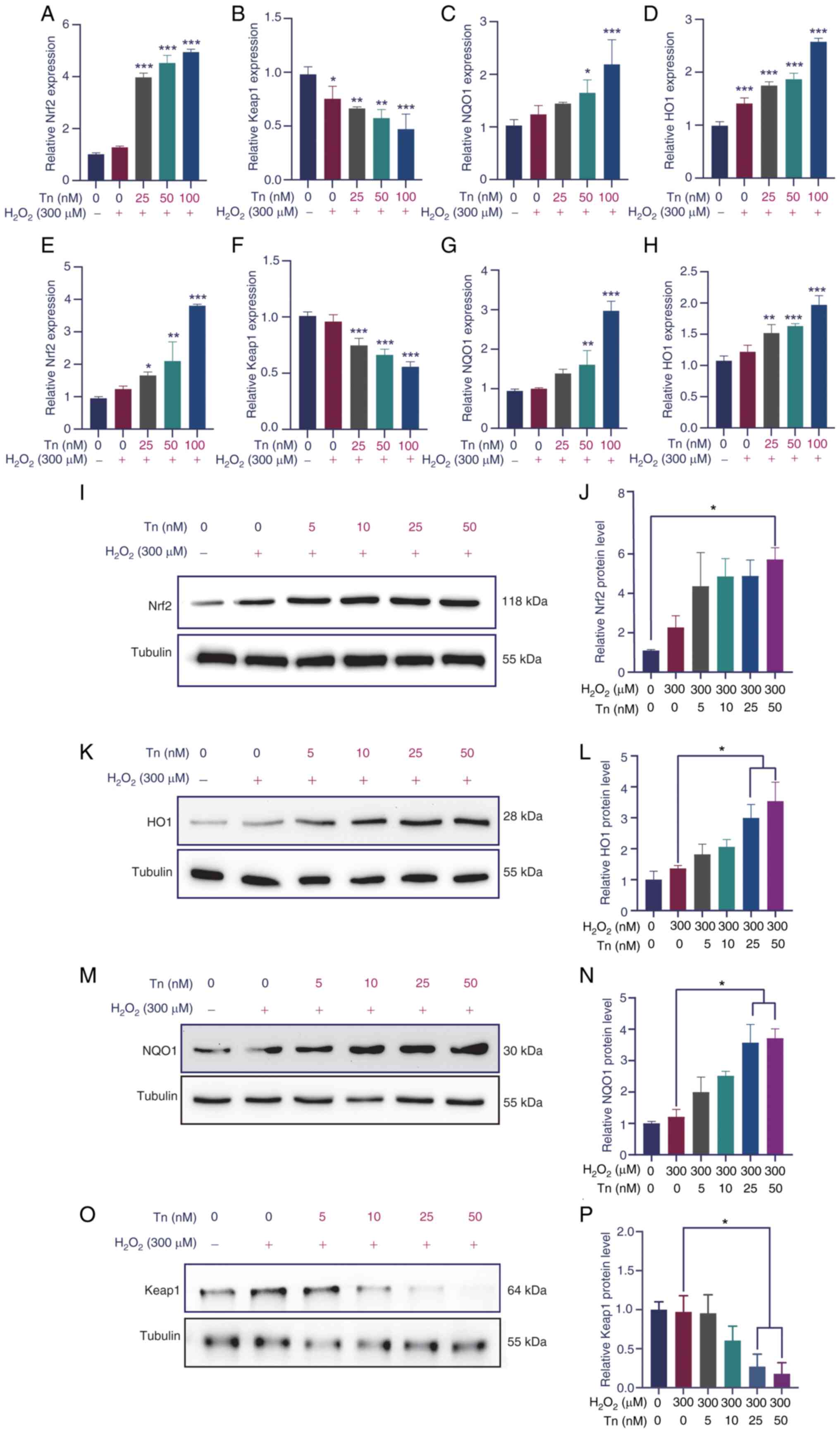 | Figure 2Tn administration activates the
expression of Nrf2 downstream target genes in Müller and ARPE-19
cells under oxidative-stress conditions. (A-D) In the
H2O2-induced Müller cells oxidative stress
model, Tn significantly increased the RNA levels of Nrf2 and Nrf2
downstream antioxidant genes, while suppressing Keap1 levels,
compared with the non-treated group. (E-H) ARPE-19 cells exhibited
the same trend. (I-P) In the H2O2-induced
ARPE-19 cells oxidative stress models, Tn administration
significantly elevated the protein expression levels Nrf2, HO1 and
NQO1, and similarly inhibited Keap1 protein expression. Data are
expressed as the mean ± standard error of the mean.
*P<0.05, **P<0.01 and
***P<0.005 (n=5). Tn, triptonide; Nrf2, nuclear
factor erythrocyte 2-related factor 2; Keap1, Kelch-like
ECh-associated protein 1; HO1, heme oxygenase-1; NQO1, NAD(P)H:
quinone oxidoreductase 1. |
Western blotting was used to evaluate the protein
expression of the Nrf2/Keap1 cascade in ARPE-19 cells. As expected,
in Tn-treated ARPE-19 cells, the protein expression levels of Nrf2
and its downstream effectors, HO1 and NQO1, exhibited a significant
concentration-dependent increase compared with the control group
(Fig. 2I-N). Concurrently, the
protein expression level of Keap1 also demonstrated a corresponding
concentration-dependent decrease (Fig. 2O and P). These results indicated
that Tn may have been an efficient activator of the Nrf2/Keap1
signaling cascade.
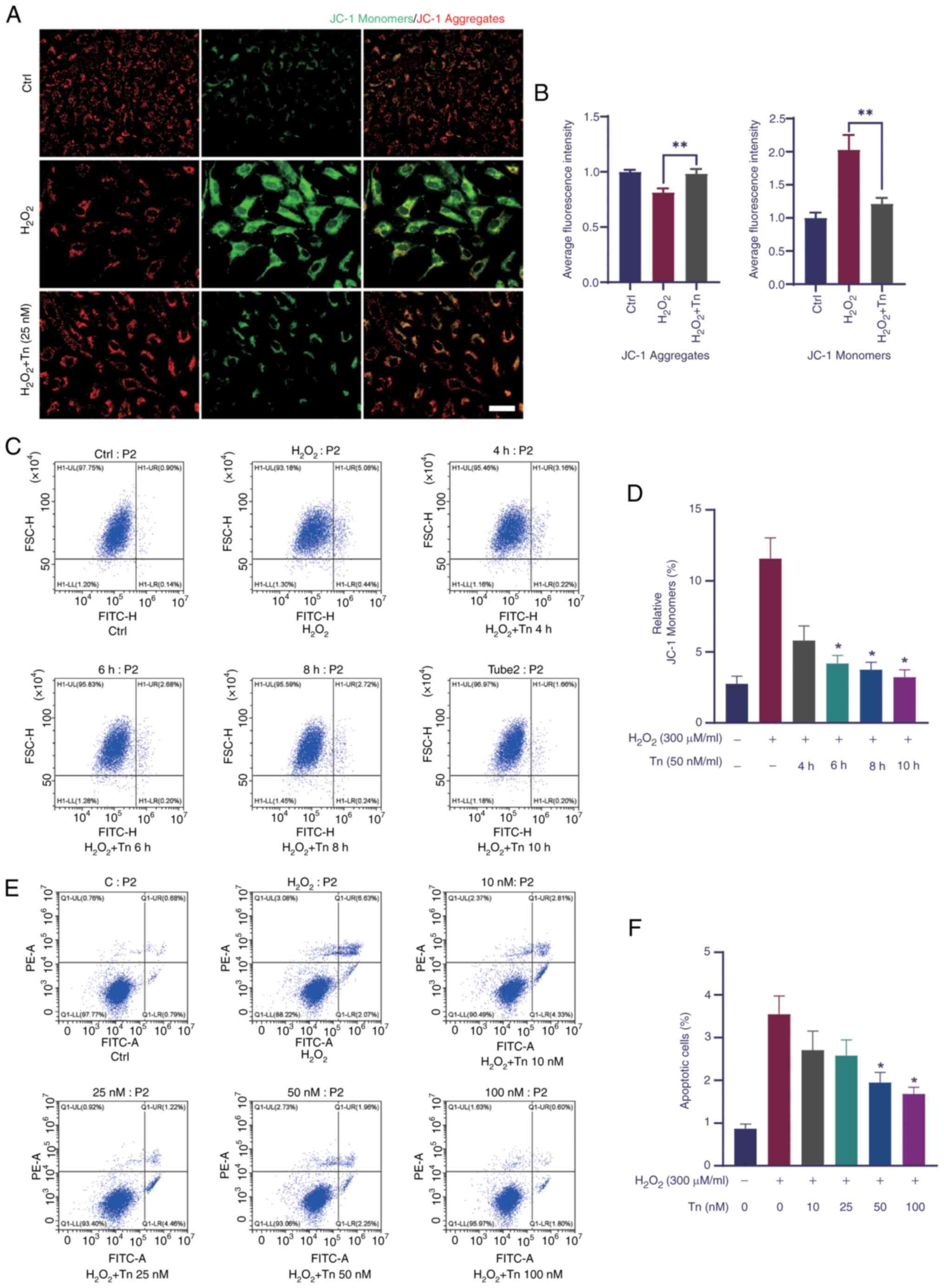
Tn significantly alleviates apoptosis
caused by oxidative injury in vitro
Oxidative stress is an important pathogenic factor
in the development of retinal injury and diseases, especially AMD
(28). To confirm the protective
effects of Tn against H2O2-induced oxidative
stress, the mitochondrial potential of ARPE-19 cells was examined
using the JC-1 assay. The change in fluorescence from red to green
indicates a marked reduction in mitochondrial potential (29). The fluorescence analysis using
confocal microscopy and flow cytometry revealed that Tn effectively
ameliorated the reduction in mitochondrial membrane potential
(Fig. 3A-D), indicating that the
Tn cytoprotective effect was correlated with oxidative stress and
apoptosis. Furthermore, double labeling of Müller cells was
performed with PI and Annexin V to verify the Tn cytoprotective
effect. Flow cytometric assays showed that the percentage of PI-and
Annexin V-positive cells in the Tn-pretreated group was
significantly lower than that in the
H2O2-stimulated group (Fig. 3E and F). These results suggested
that Tn substantially reduced the mitochondrial membrane potential
and thus protected retinal cells from
H2O2-induced apoptosis in ARPE-19 and Müller
cells.
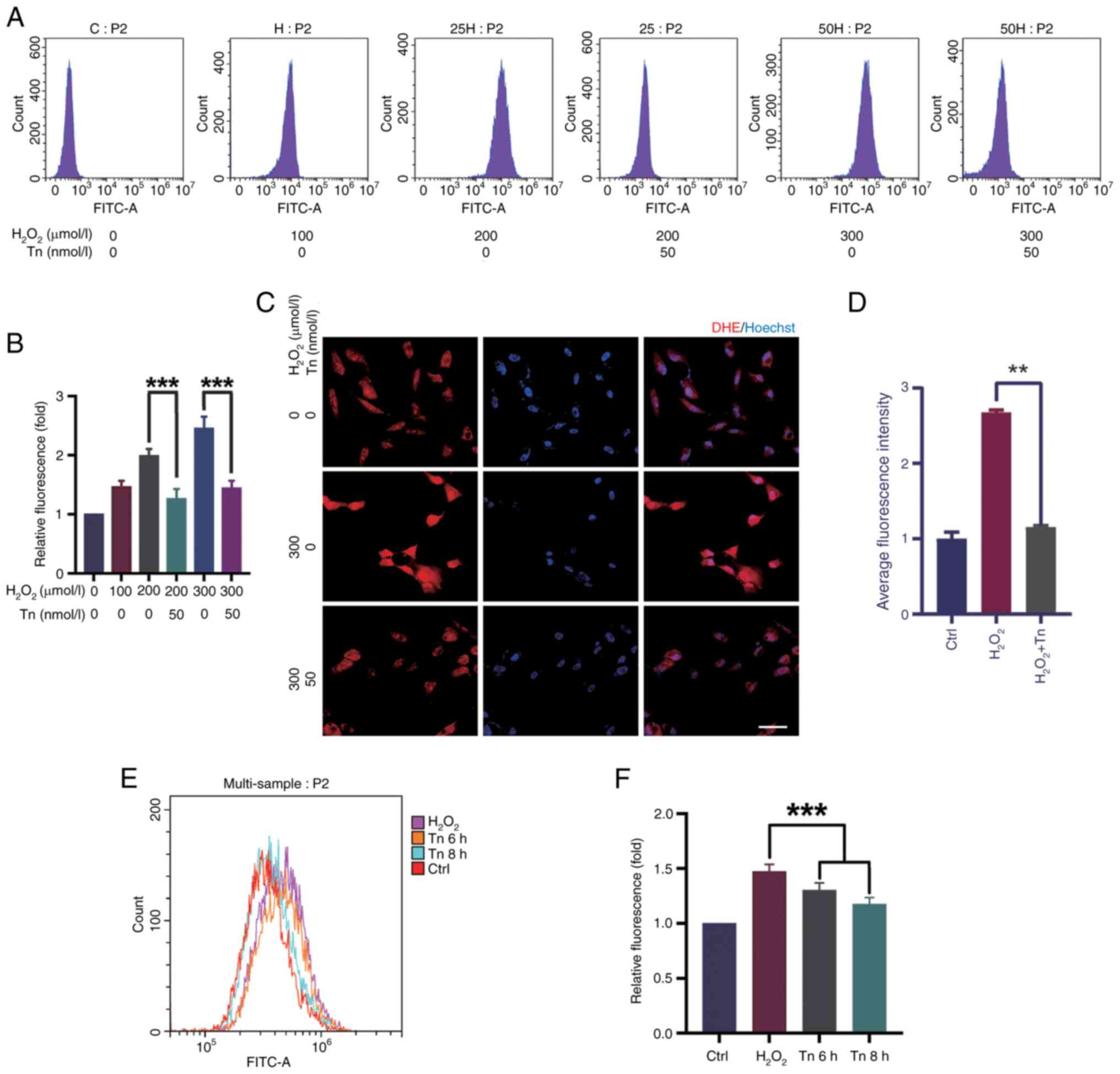
Tn decreases
H2O2-induced ROS production in vitro
It was investigated whether the decrease in
apoptosis in ARPE-19 and Müller cells was due to a decrease in ROS
production induced by Tn. To examine the effect of Tn on
antioxidants, the ROS expression was evaluated in Müller cells and
ARPE-19 cells. Flow cytometry revealed that exposure to
H2O2 at a concentration of 300 μM for
18 h resulted in high ROS levels. Simultaneously, Tn significantly
suppressed ROS levels in Müller and ARPE-19 cells under stressed
conditions. Tn pretreatment decreased ROS optical density compared
with the H2O2-induced group, indicating that
Tn enhanced the antioxidant capacity of Müller and ARPE-19 cells
(Fig. 4A, B, E and F). DHE
staining, a sensitive probe for detecting ROS generation in cells,
showed similar results. After H2O2
stimulation, ARPE-19 cells had a substantial increase in DHE
staining intensity, which was significantly decreased in the Tn
pretreatment group (Fig. 4C and
D). These results illustrated that Tn pretreatment
significantly alleviated ROS production induced by
H2O2 in ARPE-19 and Müller cells.
Tn significantly reduces overall retinal
inflammatory response and oxidative stress
In vivo experiments were performed to verify
the protective effects of Tn injection into the mouse retina
against oxidative stress injury. Firstly, the concentration of Tn
was determined by referring to existing literature (30) and through calculations based on
the molecular weight of Tn (358.39 Da) and anticipated in
vivo bioavailability. At this concentration, Tn can generate
significant biological effects without inducing notable toxic
effects on the retina (Fig.
S1). As previously described, the mouse retinal photodamage and
NMDA injury models are relatively effective to evaluate retinal
oxidative stress injury (22,31).
First, RT-qPCR analysis of the genes involved in the
Nrf2 pathway was performed. The results showed that in the light-
and NMDA-induced retinal models, the mRNA levels of Nrf2
pathway-related genes increased, which was more elevated in the Tn
pretreatment group, although the mRNA changes in the light-induced
model were not significant (Fig.
5A-C). To further investigate the effect of Tn on oxidative
protection, the relative mRNA levels of inflammatory cytokines and
genes related to oxidative stress were measured. The results of
RT-qPCR demonstrated that the mRNA expression of inflammatory
factors, such as interleukin (IL)-6 and IL-1β, significantly
increased in the NMDA-treated group; in the Tn pretreatment these
mRNA expression levels decreased (Fig. 5D and E). Western blotting further
validated the aforementioned results (Fig. 5F). Genes related to the oxidative
stress response, including glutathione synthetase (GSS),
glutathione S-transferase (GST), superoxide dismutase 2 (SOD2), and
glutamine peroxidase 1 (GPX1), almost doubled in the light-induced
group and tripled in the NMDA-induced group, which plunged to a
level equivalent to that of the control group after Tn intravitreal
injection (Fig. 5G and H). The
RT-qPCR results indicated that Tn promoted anti-inflammatory and
anti-oxidative activities and protected against light- and
NMDA-induced damage in vivo.
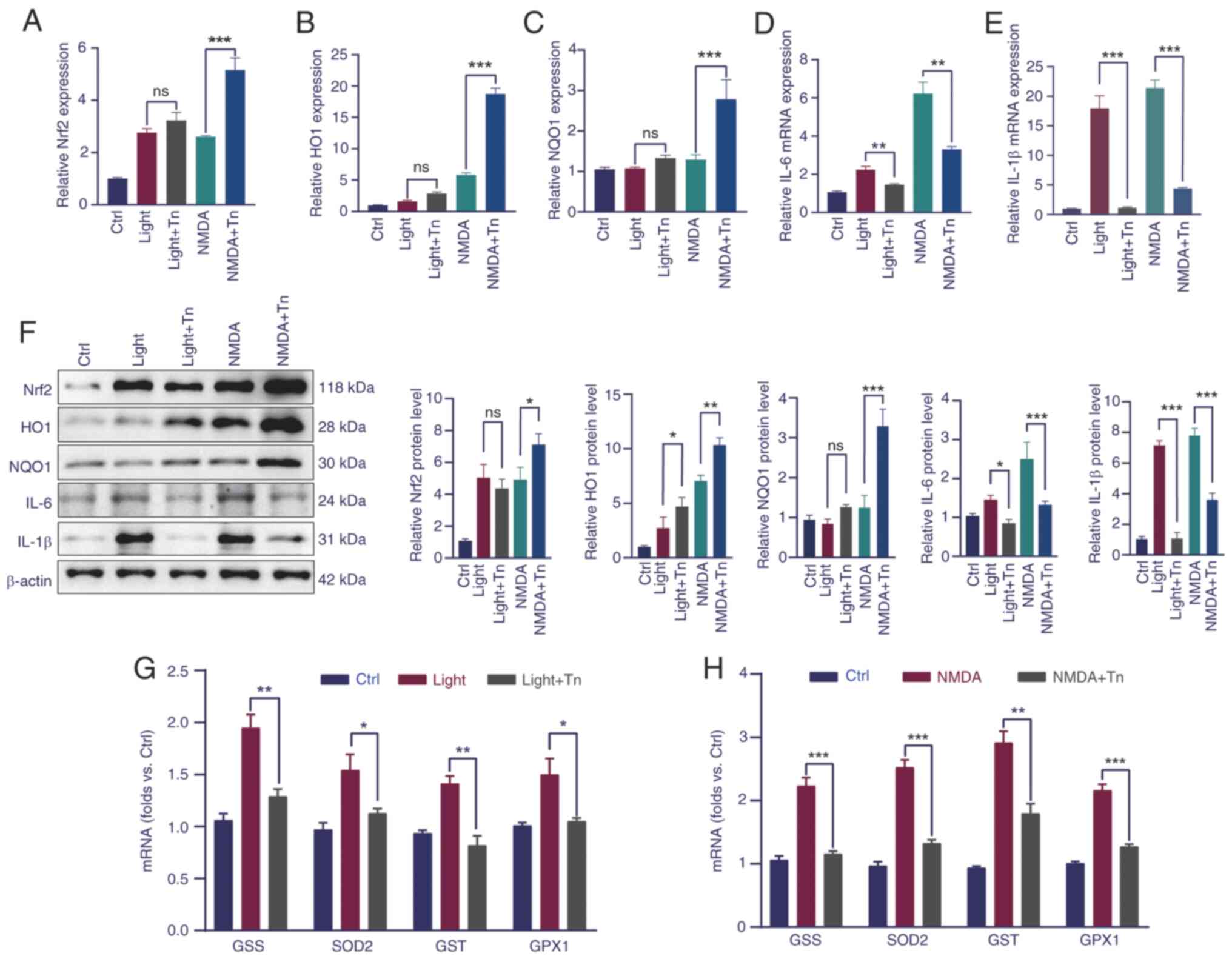 | Figure 5Tn reduces the expression of
inflammatory cytokines and increases oxidative stress-related genes
in light-induced and NMDA-induced oxidative stress models. (A-C) In
the NMDA-induced models stimulating oxidative stress, Nrf2 and
antioxidant response element-dependent genes HO1 and NQO1
increased, which further increased in Tn pretreatment groups. (D
and E) Reverse transcription-quantitative PCR assays of retinal
inflammatory cytokines were reduced after Tn pretreatment in the
light and NMDA-induced oxidative stress models. (F) In the light
and NMDA-induced oxidative stress models, Tn significantly
increased the levels of Nrf2, HO1 and NQO1 protein expression, and
similarly inhibited the protein expression of retinal inflammatory
cytokines. (G and H) Similarly, HO1 and NQO1 anti-oxidative genes
were reduced in Tn pretreatment group. Data are expressed as the
mean ± standard error of the mean. *P<0.05,
**P<0.01 and ***P<0.005. (n=5). Tn,
triptonide; NMDA, N-Methyl-D-aspartic acid; Nrf2, nuclear factor
erythrocyte 2-related factor 2; HO1, heme oxygenase-1; NQO1,
NAD(P)H: quinone oxidoreductase 1; Ctrl, control; GPX1, glutamine
peroxidase 1; GSS, glutathione synthetase; GST, glutathione
S-transferase; SOD2, superoxide dismutase 2. |
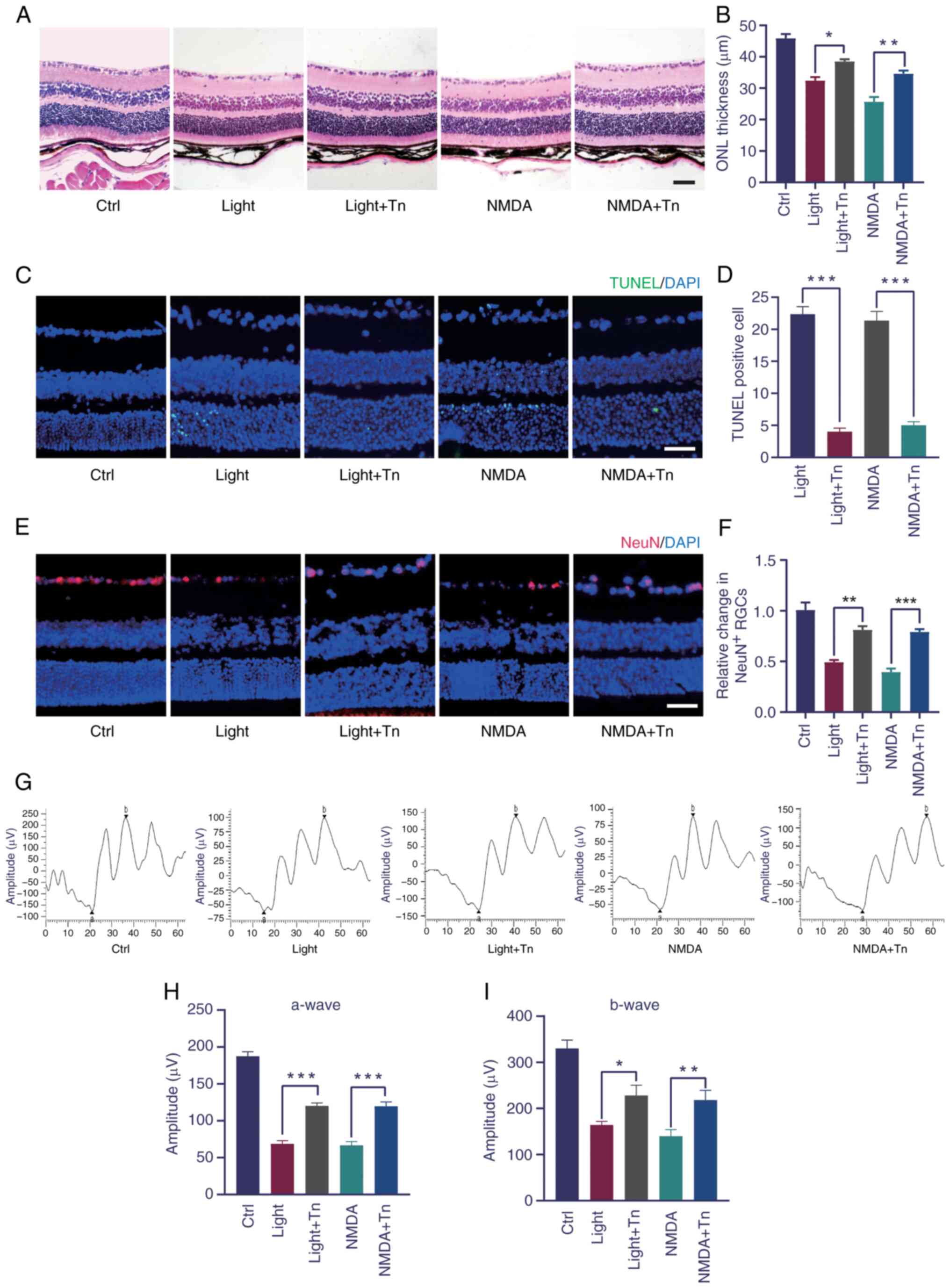
Tn ameliorates retina function in
vivo
Using the light- and NMDA-induced oxidative stress,
it was tested whether intravitreally injected Tn could ameliorate
the retinal structure and function. H&E staining revealed that
light- and NMDA-induced retinal injury significantly decreased the
outer nuclear layer thickness, which was alleviated by Tn
pretreatment (Fig. 6A and B).
The TUNEL assay demonstrated that light exposure and NMDA treatment
significantly increased the number of apoptotic retinal cells. Tn
pretreatment significantly decreased the number of apoptotic
retinal cells (Fig. 6C and D).
Immunohistochemistry was performed to determine whether Tn affected
the number of retinal ganglion cells. The number of NeuN-positive
cells decreased in the light-damaged and NMDA-treated groups and
increased in the Tn-pretreated group (Fig. 6E and F). These results identified
that Tn pretreatment protected against light- and NMDA-induced
retinal degeneration in vivo. To determine the protective
effect of Tn on retinal function, ERGs were performed in the mice.
A decrease was found in the a- and b-wave amplitudes in the retinas
of the light-damaged and NMDA-induced groups. However, this
dysfunction was partially reversed in the treatment group, as
demonstrated by the ERG a- and b-wave amplitudes (Fig. 6G-I).
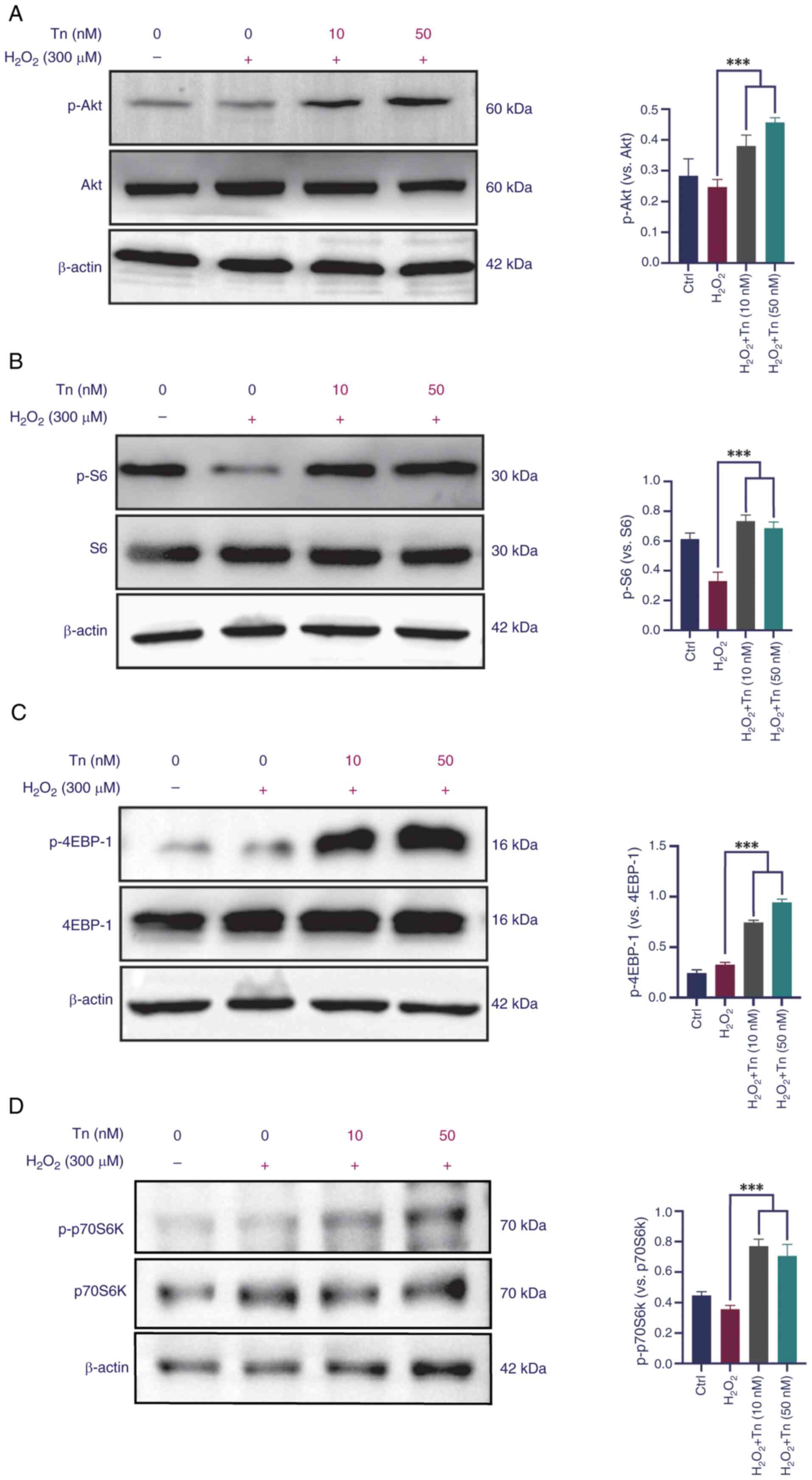
Tn effects on the expression of the
PI3K/Akt/mTOR signaling pathway
PI3K/Akt/mTOR signaling is vital for Nrf2
phosphorylation and nuclear translocation, as confirmed in a
previous study by the authors (12). Western blotting was performed to
investigate whether Tn pretreatment affected activation of the Akt
signaling pathway. The present results demonstrated that Tn
phosphorylated Akt and its downstream target, S6, in ARPE-19 cells
(Fig. 7A and B). Furthermore,
mTOR downstream proteins 4EBP-1 and p70S6K1 were significantly
phosphorylated in the Tn-pretreated groups (Fig. 7C and D). Increased expression of
the signaling proteins revealed that Tn pretreatment greatly
enhanced Akt pathway activation in ARPE-19 cells when treated with
H2O2. Hence, it was deduced that the
Tn-mediated effects on Nrf2 activation likely depend on the
PI3K/Akt/mTOR signaling pathway. Thus, it was reported that Tn
protects ARPE-19 cells from oxidative stress by activating the
PI3K/Akt/mTOR signaling axis.
Tn regulates angiogenic function in
endothelial cells
VEGF is involved in angiogenesis, which is a key
pathological change in exudative AMD (32). HUVECs were pretreated with Tn
(250 or 500 nM) and stimulated with VEGF (10 ng/ml). The
VEGF-treated group had increased viability and endothelial cell
proliferation compared with the control group. However, EdU
staining demonstrated that pretreatment with Tn reduced endothelial
cell proliferation and rescued the effects of VEGF (Fig. S2A). Further investigation using
Transwell migration and invasion assays showed that VEGF-mediated
migration and infiltration were interrupted after Tn administration
(Fig. S2B and C). Matrigel tube
formation assay results indicated that Tn decreased the ability of
HUVECs to form tubes (Fig.
S2D). In addition, in vivo experiments using
laser-induced CNV models revealed that the neovascular area in the
Tn-pretreated group was significantly smaller than that in the
saline-treated control group (Fig.
S3). Therefore, it was concluded that Tn reduced the angiogenic
function of endothelial cells.
Discussion
It has been shown that retinal tissue is highly
sensitive to oxidative stress due to high oxygen consumption during
phototransduction (33).
Therefore, oxidative stress is a key factor in accelerating the
pathological progression of ocular diseases. The AMD
pathophysiological course involves various changes, including RPE
injury, Bruch's membrane lesions, neurodegenerative changes, and
abnormal elevation of proinflammatory and proangiogenic cytokines
(34,35). RPE cell senescence was previously
considered to initiate AMD; although, senescence and degeneration
of RPE cells involve pathological processes such as oxidative
stress damage and apoptosis, which are considered important causes
of AMD (6). However, Müller
cells are the first cell type to show metabolic abnormalities
during retinal degeneration due to their high aerobic metabolism
(36-38). The peaks of glutamine and
glutathione are more than double the normal levels in the Müller
cells of patients with early dry and wet AMD (38). Müller cells activated under
hypoxic or high-glucose conditions secrete angiogenic factors such
as VEGF and basic fibroblast growth factor, which promote
endothelial cell proliferation (39,40). Therefore, future studies on the
pathological changes of RPE and Müller cells in AMD are expected to
reveal therapeutic targets for maintaining or restoring healthy
retinal function.
Previously, numerous edible plants (cauliflowers,
melons, blueberries and legumes) and traditional Chinese herbs have
been extensively studied for their powerful antioxidant capacity
and low toxicity, and are expected to be antioxidant therapeutics
(41,42). Tn is a key bioactive small
molecule extracted from Tripterygium wilfordii, a plant
utilized in traditional Chinese medicine, which has a molecular
structure similar to that of Tl, but is significantly less toxic
than Tl (43,44). Tn can play a neuroprotective role
by regulating MAPKs and NF-κB pathways, inhibiting microglia
activation, inflammatory factor release, oxidative stress and
calcium overload, antagonizing excitatory toxicity, and promoting
the synthesis of neurotrophic factors (16,17). The results of the present study
revealed that Tn was an efficient Nrf2 activator with the potential
for use in the treatment of diseases such as AMD. An oxidative
stress model of Müller and ARPE-19 cells induced by
H2O2 was constructed, and the results showed
that Tn pretreatment at different concentrations had no obvious
cytotoxicity in retinal cells. Mitochondria are the main source of
ROS in AMD and mitochondrial DNA is a sensitive target of oxidative
stress. Therefore, early oxidative damage is often accompanied by
the destruction of mitochondrial transmembrane potential (3,45,46). It was found that nanomolar levels
of Tn effectively ameliorated the reduction in mitochondrial
membrane potential; flow cytometry and JC-1 staining also confirmed
the anti-oxidative stress and anti-apoptotic effects of Tn. To
further investigate the antioxidant effect of Tn, ROS production in
ARPE-19 and Müller cells was detected by flow cytometry and DHE
staining. As previously described, light-induced retinal oxidative
stress and NMDA-induced neuro-excitotoxicity models have been used
to study oxidative stress conditions in AMD and other ocular
diseases because of their proposed ability to induce oxidative
stress, mitochondrial dysfunction and retinal inflammation
(22-25). Tn pretreatment significantly
decreased the expression of oxidative stress-related genes and
inflammatory factors in the retinal tissues of the two models; and
the results obtained by both western blotting and RT-qPCR methods
were highly consistent with each other. In addition, H&E
staining and immunofluorescence results indicated that Tn improved
retinal function and sustained the integrity of retinal morphology,
which was further verified by ERG.
The transcription factor Nrf2 is a key signaling
molecule that regulates cell survival and maintains redox
homeostasis. It mediates the expression of antioxidant genes and
phase II detoxification enzymes by activating ARE signals. It is
considered as a potential intervention target for numerous
oxidative stress-related ocular diseases (47,48). Oxidative stress stimulates Nrf2
dissociation from Keap1 and subsequent nuclear translocation. Gene
transcription related to antioxidant protection, such as HO1, NQO1
and SOD (49), is activated by
Nrf2 when it interacts with downstream AREs. Some natural plant
components (such as quercetin, ginsenosides, astaxanthin and
curcumin) have been shown to inhibit oxidative damage in ARPE-19
cells by activating Nrf2 signaling (50-53). Previous studies by the authors
have found that salvianolic acid A, ginsenosides Rg1, Rg3, 3h-1,
2-dimercaptiol-3-thione, and other Chinese herbal extracts or small
molecular compounds are effective Nrf2 activators, inducing
phosphorylation of Nrf2 dependent on Akt-mTORC1, with potential
therapeutic value for oxidative stress-related retinal degenerative
diseases (12,13,54). Based on these results, the mRNA
expression levels of Nrf2, Keap1 and the transcription levels of
the downstream target genes HO1 and NQO1 were determined. The
RT-qPCR results showed that the mRNA levels of Nrf2, HO1 and NQO1
significantly increased after Tn administration in ARPE-19 and
Müller cells. This indicates that Tn can indeed activate the Nrf2
signaling pathway and promote the expression of these antioxidant
genes. Notably, the mRNA level of Keap1 decreased slightly after Tn
treatment. This may be due to the direct modification of Keap1 by
Tn, leading to decreased stability or transcriptional inhibition.
However, this decrease was not significant, suggesting that Tn
mainly exerts its therapeutic effects by promoting the activation
of Nrf2, rather than by directly inhibiting Keap1 expression.
Considering that the core pathological process in the early stage
of AMD mainly affects RPE cells and photoreceptor cells (while
Müller cells may play a relatively indirect role, primarily
providing nutritional support and neuroprotective functions as
glial cells within the retina and are not the direct targets of
damage), the protein expression levels of Nrf2, Keap1 and their
downstream targets HO1 and NQO1 were further measured in ARPE-19
cells using western blotting. The results indicated that Tn not
only promotes the transcription of these genes except Keap1, but
also effectively facilitates their protein translation and
accumulation.
Although Tn was confirmed to be an effective
activator of Nrf2 in the present study, promoting the expression of
downstream antioxidant proteins, the underlying signal transduction
mechanism has not been fully elucidated. Akt-mTORC1 is a key signal
for cell proliferation and survival (55). Existing evidence suggests that
the phosphorylation of Nrf2 is dependent on the activation of
Akt-mTORC1 and that the PI3K/Akt/mTORC1 pathway mediates activation
of the Keap1/Nrf2/ARE antioxidant pathway (56,57). Specifically, the activation of
PI3K triggers the phosphorylation of phosphatidylinositol (PI) to
generate 3-phosphoinositide (PIP3). PIP3, as a second messenger,
recruits and activates Akt, leading to its phosphorylation.
Phosphorylated Akt not only further activates downstream mTORC1 but
also stabilizes Nrf2 by phosphorylating and inhibiting GSK3-β, a
kinase that negatively regulates Nrf2. By inhibiting GSK3-β, Akt
protects Nrf2 from degradation, promoting its nuclear translocation
and transcriptional activity (58,59). Therefore, it was attempted to
observe whether Tn activated the PI3K/Akt/mTORC1 pathway and its
downstream target genes. It was found that Tn pretreatment
significantly increases the phosphorylation levels of Akt, S6, as
well as the mTORC1 downstream target proteins 4EBP-1 and p70S6K
after H2O2 exposure, indicating that the Tn
effect in rescuing the antioxidant and anti-inflammatory pathways
of Nrf2/HO1 may have arisen from reactivation of the
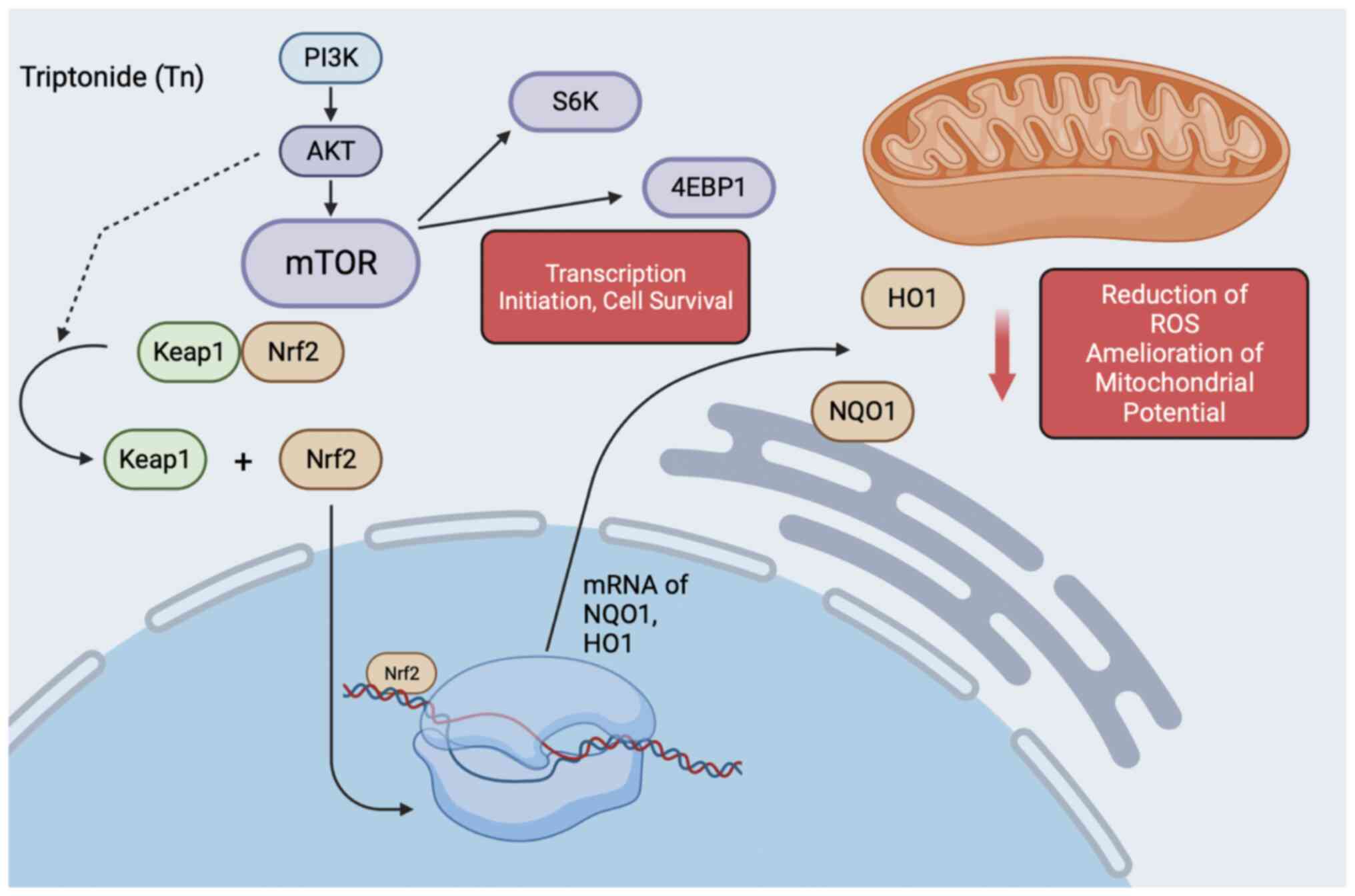
PI3K/Akt/mTORC1 axis (Fig.
8).
CNV is the core pathological change in wet AMD
(60,61). Interestingly, it was found that
Tn significantly inhibited pathological neovascularization, both
in vitro and in vivo. Specifically, Tn attenuated
VEGF-induced vascular endothelial cell proliferation and migration,
and a significantly decreased level of pathological angiogenesis
was observed in laser-induced CNV (animal models of exudative AMD)
(62).
The present study indicated that Tn can induce the
activation of Nrf2 signaling by activating the PI3K/Akt signaling
pathway, thereby improving the structure and function of the
retina. However, in addition to this core mechanism, there may be
other signaling mechanisms that interact with the effects of Tn.
There is a close interaction between the PI3K/Akt and NF-κB
signaling pathways. Akt, as a key molecule downstream of PI3K, can
inhibit the nuclear translocation and transcriptional activity of
NF-κB by phosphorylating the IκB kinase (IKK) complex, thereby
achieving negative regulation (63). Therefore, Tn may indirectly
inhibit the excessive activation of NF-κB by activating the
PI3K/Akt signaling pathway. Furthermore, autophagy, a highly
conserved cellular self-degradation process, is crucial for
clearing damaged organelles and protein aggregates and maintaining
intracellular homeostasis. The activation of autophagy is closely
related to the regulation of the PI3K/Akt signaling pathway. Akt,
as a key molecule in this pathway, can regulate autophagic activity
by influencing the expression of autophagy-related genes and the
initiation of the autophagic process (64). Tn may promote the clearance of
harmful substances within retinal cells by activating the
autophagic signaling pathway.
Tn, as a potent Nrf2 activator, exhibits significant
antioxidant effects on the retina. In AMD, retinal cells are
chronically exposed to high oxidative stress, leading to cellular
dysfunction and death. Therefore, by activating the Nrf2 signaling
pathway, Tn holds promise as an effective antioxidant protective
agent for patients with AMD. Furthermore, Tn is capable of
modulating inflammatory signaling pathways, such as NF-κB, and
suppressing the expression of inflammation-related genes, thereby
attenuating retinal inflammation. This mechanism contributes to the
protection of retinal cells from inflammatory damage and the
slowing of AMD progression. Notably, Tn also demonstrates
remarkable anti-angiogenic properties. It can inhibit the
proliferation and migration of vascular endothelial cells, thus
suppressing the formation of pathological neovascularization. This
mechanism provides a potential therapeutic strategy for patients
with wet AMD.
However, AMD is a complex retinal disease; combining
Tn with other drugs or therapies may lead to more comprehensive
therapeutic outcomes. In the treatment of AMD, high doses of
antioxidant vitamins and minerals, such as vitamin C, vitamin E,
beta-carotene and zinc, have been extensively studied and applied
(65). These substances
effectively scavenge free radicals and reduce retinal oxidative
stress. When combined with Tn, they can provide additional
antioxidant protection by synergistically activating the Nrf2
pathway, thereby delaying the progression of AMD. Furthermore, the
combination of Tn with anti-VEGF drugs, such as ranibizumab and
aflibercept, may also demonstrate promising results. This
combination not only synergistically controls the condition of AMD,
reduces the growth and leakage of abnormal blood vessels, but also
provides additional antioxidant protection. In some severe cases of
AMD, glucocorticoids are used to control retinal inflammation
(66). However, long-term or
high-dose steroid use may increase the risk of side effects such as
cataracts and glaucoma. Therefore, combining low-dose steroids with
Tn may be a strategy to balance efficacy and safety. Finally, cell
therapy, particularly the transplantation of stem cells or RPE
cells, offers promise in restoring the structure and function of
the retina (67). When combined
with Tn, this therapeutic approach can be further optimized as Tn
promotes the survival and integration of transplanted cells through
its antioxidant and anti-inflammatory actions, thereby enhancing
the overall treatment outcome.
The present study also has some limitations.
Firstly, further studies are needed to investigate the specific
mechanism of Tn on the Nrf2 signaling pathway by in vivo and
silencing experiments. Secondly, the reactivation of the
PI3K/AKT/mTOR signaling pathway by Tn could be further verified
using specific inhibitors or activators. Additionally, to improve
evaluation of the protective effects of Tn, it can be compared with
other known retinal protective strategies such as antioxidants and
anti-inflammatory agents to improve identification of the
superiority or uniqueness of Tn. Finally, the current study did not
address the evaluation of the long-term efficacy of Tn in retinal
diseases. In the future, rigorous long-term follow-up trials will
be conducted by the authors to systematically assess both the
sustained efficacy and safety profile of Tn in the treatment of
retinal diseases.
In conclusion, the results of the present study
suggest that nanomolar concentrations of Tn protected retinal cells
against oxidative damage and inflammation. Activation of Tn-induced
Nrf2 signaling may be realized by activating PI3K/Akt/mTOR
signaling, thus enhancing the protective effect on cells. The in
vivo studies demonstrated that intravitreal injection of Tn
protected mice from light- and NMDA-induced retinal damage and
dysfunction. Consequently, it is reasonable to hypothesize that Tn
is a highly potent Nrf2 activator that could become a new
therapeutic agent for retinal oxidative stress injury and
pathological neovascular ocular diseases such as AMD.
Supplementary Data
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
KL designed and supervised the study. JinL, JiaL and
YC performed the experiments, analyzed data, followed-up
experimental supplement and wrote the manuscript. JY, YS and LL
participated in parts of the experiments. JiaL, YC and KL confirm
the authenticity of all the raw data. All authors read and approved
the final manuscript.
Ethics approval and consent to
participate
The animal experimental procedures in the present
study were approved (approval no. 2303048) by the Ethics Committee
of Nanjing Medical University (Nanjing, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgements
Not applicable.
Funding
The present study was supported by the National Natural Science
Foundation of China (grant nos. 82171080 and 82101156).
References
|
1
|
Thomas CJ, Mirza RG and Gill MK:
Age-related macular degeneration. Med Clin North Am. 105:473–491.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hanus J, Anderson C and Wang S: RPE
necroptosis in response to oxidative stress and in AMD. Ageing Res
Rev. 24:286–298. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Liang FQ and Godley BF: Oxidative
stress-induced mitochondrial DNA damage in human retinal pigment
epithelial cells: A possible mechanism for RPE aging and
age-related macular degeneration. Exp Eye Res. 76:397–403. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tu W, Wang H, Li S, Liu Q and Sha H: The
anti-inflammatory and anti-oxidant mechanisms of the keap1/Nrf2/ARE
signaling pathway in chronic diseases. Aging Dis. 10:637–651. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ulyanova T, Szél A, Kutty RK, Wiggert B,
Caffé AR, Chader GJ and van Veen T: Oxidative stress induces heme
oxygenase-1 immunoreactivity in Müller cells of mouse retina in
organ culture. Invest Ophthalmol Vis Sci. 42:1370–1374.
2001.PubMed/NCBI
|
|
6
|
Flores R, Carneiro Â, Vieira M, Tenreiro S
and Seabra MC: Age-related macular degeneration: Pathophysiology,
management, and future perspectives. Ophthalmologica. 244:495–511.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhang X and Sivaprasad S: Drusen and
pachydrusen: The definition, pathogenesis, and clinical
significance. Eye (Lond). 35:121–133. 2021. View Article : Google Scholar
|
|
8
|
Hernández-Zimbrón LF, Zamora-Alvarado R,
Ochoa-De la Paz L, Velez-Montoya R, Zenteno E, Gulias-Cañizo R,
Quiroz-Mercado H and Gonzalez-Salinas R: Age-related macular
degeneration: New paradigms for treatment and management of AMD.
Oxid Med Cell Longev. 2018:83746472018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
García-Layana A, Cabrera-López F,
García-Arumí J, Arias-Barquet L and Ruiz-Moreno JM: Early and
intermediate age-related macular degeneration: Update and clinical
review. Clin Interv Aging. 12:1579–1587. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cabral de Guimaraes TA, Daich Varela M,
Georgiou M and Michaelides M: Treatments for dry age-related
macular degeneration: Therapeutic avenues, clinical trials and
future directions. Br J Ophthalmol. 106:297–304. 2022. View Article : Google Scholar
|
|
11
|
Damico FM, Gasparin F, Scolari MR, Pedral
LS and Takahashi BS: New approaches and potential treatments for
dry age-related macular degeneration. Arq Bras Oftalmol. 75:71–76.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Li KR, Yang SQ, Gong YQ, Yang H, Li XM,
Zhao YX, Yao J, Jiang Q and Cao C: 3H-1,2-dithiole-3-thione
protects retinal pigment epithelium cells against Ultra-violet
radiation via activation of Akt-mTORC1-dependent Nrf2-HO-1
signaling. Sci Rep. 6:255252016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Li KR, Zhang ZQ, Yao J, Zhao YX, Duan J,
Cao C and Jiang Q: Ginsenoside Rg-1 protects retinal pigment
epithelium (RPE) cells from cobalt chloride (CoCl2) and hypoxia
assaults. PLoS One. 8:e841712013. View Article : Google Scholar
|
|
14
|
Ong FS, Kuo JZ, Wu WC, Cheng CY, Blackwell
WLB, Taylor BL, Grody WW, Rotter JI, Lai CC and Wong TY:
Personalized medicine in ophthalmology: From pharmacogenetic
biomarkers to therapeutic and dosage optimization. J Pers Med.
3:40–69. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Liu R, Li X, Huang N, Fan M and Sun R:
Toxicity of traditional Chinese medicine herbal and mineral
products. Adv Pharmacol. 87:301–346. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Tang B, Zhu J, Zhang B, Wu F, Wang Y, Weng
Q, Fang S, Zheng L, Yang Y, Qiu R, et al: Therapeutic potential of
triptolide as an anti-inflammatory agent in dextran sulfate
sodium-induced murine experimental colitis. Front Immunol.
11:5920842020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Liu Q: Triptolide and its expanding
multiple pharmacological functions. Int Immunopharmacol.
11:377–383. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zheng YL, Lin JF, Lin CC and Xu Y:
Anti-inflammatory effect of triptolide. Zhongguo Yao Li Xue Bao.
15:540–543. 1994.In Chinese. PubMed/NCBI
|
|
19
|
Buendia I, Michalska P, Navarro E, Gameiro
I, Egea J and León R: Nrf2-ARE pathway: An emerging target against
oxidative stress and neuroinflammation in neurodegenerative
diseases. Pharmacol Ther. 157:84–104. 2016. View Article : Google Scholar
|
|
20
|
Baird L and Yamamoto M: The molecular
mechanisms regulating the KEAP1-NRF2 pathway. Mol Cell Biol.
40:e00099–20. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hui Q, Karlstetter M, Xu Z, Yang J, Zhou
L, Eilken HM, Terjung C, Cho H, Gong J, Lai MJ, et al: Inhibition
of the Keap1-Nrf2 protein-protein interaction protects retinal
cells and ameliorates retinal ischemia-reperfusion injury. Free
Radic Biol Med. 146:181–188. 2020. View Article : Google Scholar :
|
|
22
|
Han S, Chen J, Hua J, Hu X, Jian S, Zheng
G, Wang J, Li H, Yang J, Hejtmancik JF, et al: MITF protects
against oxidative damage-induced retinal degeneration by regulating
the NRF2 pathway in the retinal pigment epithelium. Redox Biol.
34:1015372020. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Dai S, Wang C, Feng L, Zhang C, Zhang W,
He Y, Zhou X, Xia X, Chen B and Song W: Protective activity of
tert-butylhydroquinone against oxidative stress and apoptosis
induced by glutamate agonizts in R28 cells and mice retina. Biomed
Pharmacother. 152:1131172022. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ozawa Y: Oxidative stress in the
light-exposed retina and its implication in age-related macular
degeneration. Redox Biol. 37:1017792020. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wu T, Handa JT and Gottsch JD:
Light-induced oxidative stress in choroidal endothelial cells in
mice. Invest Ophthalmol Vis Sci. 46:1117–1123. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
27
|
Lambert V, Lecomte J, Hansen S, Blacher S,
Gonzalez ML, Struman I, Sounni NE, Rozet E, de Tullio P, Foidart
JM, et al: Laser-induced choroidal neovascularization model to
study age-related macular degeneration in mice. Nat Protoc.
8:2197–2211. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Beatty S, Koh H, Phil M, Henson D and
Boulton M: The role of oxidative stress in the pathogenesis of
age-related macular degeneration. Surv Ophthalmol. 45:115–134.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Perelman A, Wachtel C, Cohen M, Haupt S,
Shapiro H and Tzur A: JC-1: Alternative excitation wavelengths
facilitate mitochondrial membrane potential cytometry. Cell Death
Dis. 3:e4302012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yang F, Wu L, Guo X, Wang D and Li Y:
Improved retinal ganglion cell survival through retinal microglia
suppression by a chinese herb extract, triptolide, in the DBA/2J
mouse model of glaucoma. Ocul Immunol Inflamm. 21:378–389. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Sakamoto K, Suzuki T, Takahashi K, Koguchi
T, Hirayama T, Mori A, Nakahara T, Nagasawa H and Ishii K:
Iron-chelating agents attenuate NMDA-Induced neuronal injury via
reduction of oxidative stress in the rat retina. Exp Eye Res.
171:30–36. 2018.PubMed/NCBI
|
|
32
|
Ferrara N: VEGF and intraocular
neovascularization: From discovery to therapy. Transl Vis Sci
Technol. 5:102016.PubMed/NCBI
|
|
33
|
Bruninx R, Betz P and Lepièce G:
Functionnal revalidation of patients with age-related macular
degeneration. Rev Med Liege. 75:711–716. 2020.In French. PubMed/NCBI
|
|
34
|
Mitchell P, Liew G, Gopinath B and Wong
TY: Age-related macular degeneration. Lancet. 392:1147–1159.
2018.PubMed/NCBI
|
|
35
|
Telander DG: Inflammation and age-related
macular degeneration (AMD). Semin Ophthalmol. 26:192–197.
2011.PubMed/NCBI
|
|
36
|
Jones BW, Watt CB, Frederick JM, Baehr W,
Chen CK, Levine EM, Milam AH, Lavail MM and Marc RE: Retinal
remodeling triggered by photoreceptor degenerations. J Comp Neurol.
464:1–16. 2003.PubMed/NCBI
|
|
37
|
Marc RE, Jones BW, Watt CB and Strettoi E:
Neural remodeling in retinal degeneration. Prog Retin Eye Res.
22:607–655. 2003.PubMed/NCBI
|
|
38
|
Pfeiffer RL, Marc RE, Kondo M, Terasaki H
and Jones BW: Müller cell metabolic chaos during retinal
degeneration. Exp Eye Res. 150:62–70. 2016.PubMed/NCBI
|
|
39
|
Amin RH, Frank RN, Kennedy A, Eliott D,
Puklin JE and Abrams GW: Vascular endothelial growth factor is
present in glial cells of the retina and optic nerve of human
subjects with nonproliferative diabetic retinopathy. Invest
Ophthalmol Vis Sci. 38:36–47. 1997.PubMed/NCBI
|
|
40
|
Yafai Y, Iandiev I, Lange J, Yang XM,
Wiedemann P, Bringmann A and Eichler W: Basic fibroblast growth
factor contributes to a shift in the angioregulatory activity of
retinal glial (Müller) cells. PLoS One. 8:e687732013.
|
|
41
|
Baby B, Antony P and Vijayan R:
Antioxidant and anticancer properties of berries. Crit Rev Food Sci
Nutr. 58:2491–2507. 2018.
|
|
42
|
Durazzo A, Lucarini M, Novellino E, Daliu
P and Santini A: Fruit-based juices: Focus on antioxidant
properties-Study approach and update. Phytother Res. 33:1754–1769.
2019.PubMed/NCBI
|
|
43
|
Chang Z, Qin W, Zheng H, Schegg K, Han L,
Liu X, Wang Y, Wang Z, McSwiggin H, Peng H, et al: Triptonide is a
reversible non-hormonal male contraceptive agent in mice and
non-human primates. Nat Commun. 12:12532021.PubMed/NCBI
|
|
44
|
Dong F, Yang P, Wang R, Sun W, Zhang Y,
Wang A, Chen M, Chen L, Zhang C and Jiang M: Triptonide acts as a
novel antiprostate cancer agent mainly through inhibition of mTOR
signaling pathway. Prostate. 79:1284–1293. 2019.PubMed/NCBI
|
|
45
|
Estaquier J, Vallette F, Vayssiere JL and
Mignotte B: The mitochondrial pathways of apoptosis. Adv Exp Med
Biol. 942:157–183. 2012.PubMed/NCBI
|
|
46
|
Green DR: The mitochondrial pathway of
apoptosis: Part I: MOMP and beyond. Cold Spring Harb Perspect Biol.
14:a0410382022.PubMed/NCBI
|
|
47
|
Nakagami Y: Nrf2 is an attractive
therapeutic target for retinal diseases. Oxid Med Cell Longev.
2016:74693262016.PubMed/NCBI
|
|
48
|
Nam LB and Keum YS: Binding partners of
NRF2: Functions and regulatory mechanisms. Arch Biochem Biophys.
678:1081842019.PubMed/NCBI
|
|
49
|
Ma Q: Role of nrf2 in oxidative stress and
toxicity. Annu Rev Pharmacol Toxicol. 53:401–426. 2013.PubMed/NCBI
|
|
50
|
Alhasani RH, Biswas L, Tohari AM, Zhou X,
Reilly J, He JF and Shu X: Gypenosides protect retinal pigment
epithelium cells from oxidative stress. Food Chem Toxicol.
112:76–85. 2018.
|
|
51
|
Du W, An Y, He X, Zhang D and He W:
Protection of kaempferol on oxidative stress-induced retinal
pigment epithelial cell damage. Oxid Med Cell Longev.
2018:16107512018. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Li Z, Dong X, Liu H, Chen X, Shi H, Fan Y,
Hou D and Zhang X: Astaxanthin protects ARPE-19 cells from
oxidative stress via upregulation of Nrf2-regulated phase II
enzymes through activation of PI3K/Akt. Mol Vis. 19:1656–1666.
2013.PubMed/NCBI
|
|
53
|
Zhao B, Wang Z, Han J, Wei G, Yi B and Li
Z: Rhizoma Paridis total saponins alleviate H2O2-induced oxidative
stress injury by upregulating the Nrf2 pathway. Mol Med Rep.
21:220–228. 2020.
|
|
54
|
Zhang H, Liu YY, Jiang Q, Li KR, Zhao YX,
Cao C and Yao J: Salvianolic acid A protects RPE cells against
oxidative stress through activation of Nrf2/HO-1 signaling. Free
Radic Biol Med. 69:219–228. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Manning BD and Toker A: AKT/PKB signaling:
Navigating the network. Cell. 169:381–405. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Hu H, Hao L, Tang C, Zhu Y, Jiang Q and
Yao J: Activation of KGFR-Akt-mTOR-Nrf2 signaling protects human
retinal pigment epithelium cells from Ultra-violet. Biochem Biophys
Res Commun. 495:2171–2177. 2018. View Article : Google Scholar
|
|
57
|
Portelli SS, Hambly BD, Jeremy RW and
Robertson EN: Oxidative stress in genetically triggered thoracic
aortic aneurysm: Role in pathogenesis and therapeutic
opportunities. Redox Rep. 26:45–52. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Koundouros N and Poulogiannis G:
Phosphoinositide 3-kinase/Akt signaling and redox metabolism in
cancer. Front Oncol. 8:1602018. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Wang L, Chen Y, Sternberg P and Cai J:
Essential roles of the PI3 kinase/Akt pathway in regulating
Nrf2-dependent antioxidant functions in the RPE. Invest Ophthalmol
Vis Sci. 49:1671–1678. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Deng Y, Qiao L, Du M, Qu C, Wan L, Li J
and Huang L: Age-related macular degeneration: Epidemiology,
genetics, pathophysiology, diagnosis, and targeted therapy. Genes
Dis. 9:62–79. 2021. View Article : Google Scholar
|
|
61
|
Fleckenstein M, Keenan TDL, Guymer RH,
Chakravarthy U, Schmitz-Valckenberg S, Klaver CC, Wong WT and Chew
EY: Age-related macular degeneration. Nat Rev Dis Primers.
7:312021. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Yang TJ, Yao MD, Sun YN, Li XM, Jiang Q
and Yan B: Suppression of choroidal neovascularization by silencing
of long non-coding RNA IPW. Aging (Albany NY). 13:10584–10602.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Ozes ON, Mayo LD, Gustin JA, Pfeffer SR,
Pfeffer LM and Donner DB: NF-kappaB activation by tumour necrosis
factor requires the Akt serine-threonine kinase. Nature. 401:82–85.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Wang RC, Wei Y, An Z, Zou Z, Xiao G,
Bhagat G, White M, Reichelt J and Levine B: Akt-mediated regulation
of autophagy and tumorigenesis through beclin 1 phosphorylation.
Science. 338:956–959. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Agrón E, Mares J, Clemons TE, Swaroop A,
Chew EY and Keenan TDL; AREDS and AREDS2 Research Groups: Dietary
nutrient intake and progression to late age-related macular
degeneration in the age-related eye disease studies 1 and 2.
Ophthalmology. 128:425–442. 2021. View Article : Google Scholar
|
|
66
|
Narayanan R and Kuppermann BD:
Corticosteroids and anti-complement therapy in retinal diseases.
Handb Exp Pharmacol. 242:309–320. 2017. View Article : Google Scholar
|
|
67
|
O'Neill HC, Limnios IJ and Barnett NL:
Advancing a stem cell therapy for age-related macular degeneration.
Curr Stem Cell Res Ther. 15:89–97. 2020. View Article : Google Scholar
|