Introduction
Myofibromatosis is the most common fibrous tumor of
infancy or childhood, often misdiagnosed due to its variable
clinical and imaging characteristics; the differential diagnosis
includes metastatic neuroblastoma, rhabdomyosarcoma, histiocytosis
X or hemangiopericytoma (1–3). Examination of a pathological specimen
is mandatory for ruling out malignancies that may require different
management (4).
The clinical behavior of myofibromatosis may vary
between a benign solitary or multicentric form, with involvement of
muscles, bones and subcutaneous tissues, and a diffuse multicentric
form with visceral involvement and a worse prognosis (1,4).
Conservative management and observation are preferred for the
solitary or multicentric forms without visceral involvement, as
they usually undergo spontaneous regression; occasionally, surgery
is suggested to reduce eventual mass effect symptoms. Aggressive
treatment with radiotherapy, chemotherapy and radical surgery is
used for disseminated forms with visceral involvement (1,4,5).
Clinical/radiological diagnosis and follow-up are
crucial for determining disease localization and prognosis
(6).
We herein describe a case of multicentric
musculoskeletal myofibromatosis, investigated with whole-body
magnetic resonance imaging (MRI) to assess different lesions
without X-ray exposure, in order to follow disease evolution and/or
regression, including early exclusion of visceral involvement,
which is mandatory for prognosis and treatment.
Case report
A 3-month-old male infant, asymptomatic, born to
term after an uneventful pregnancy, without a noteworthy family
history, was referred to the University Hospital Paolo Giaccone in
April, 2014, due to multiple, firm, painless swellings on the trunk
and abdomen, covered by flesh-coloured skin.
The laboratory tests were inconclusive and
misleading, including a mild increase in neuronspecific enolase to
27 ng/ml (normal, <15 ng/ml).
An ultrasound (US) examination revealed multiple
heterogeneously hypoechogenic lesions, with scarce inner and
peripheral vascularization, located in the left breastplate, right
deep intercostal muscles and the rectus abdominis muscles
bilaterally. Grade 2 left pyelectasis was also detected on
abdominal US (image not shown).
Due to the suspicion of neuroblastoma based on US
examination and laboratory tests, a nodule located in the right
rectus muscle was selected for removal, due to its easy
accessibility and low deep subcutaneous invasion, and was
subsequently analyzed by histology. The tumor was characterized by
tangles of spindleshaped elements, it was smooth muscle actin
(SMA)+, desmin− and S100−, rich in
cellular components in the periphery and exhibiting central
hyalinization. No mitotic activity or cell atypia were identified,
and the diagnosis of infantile myofibromatosis was established.
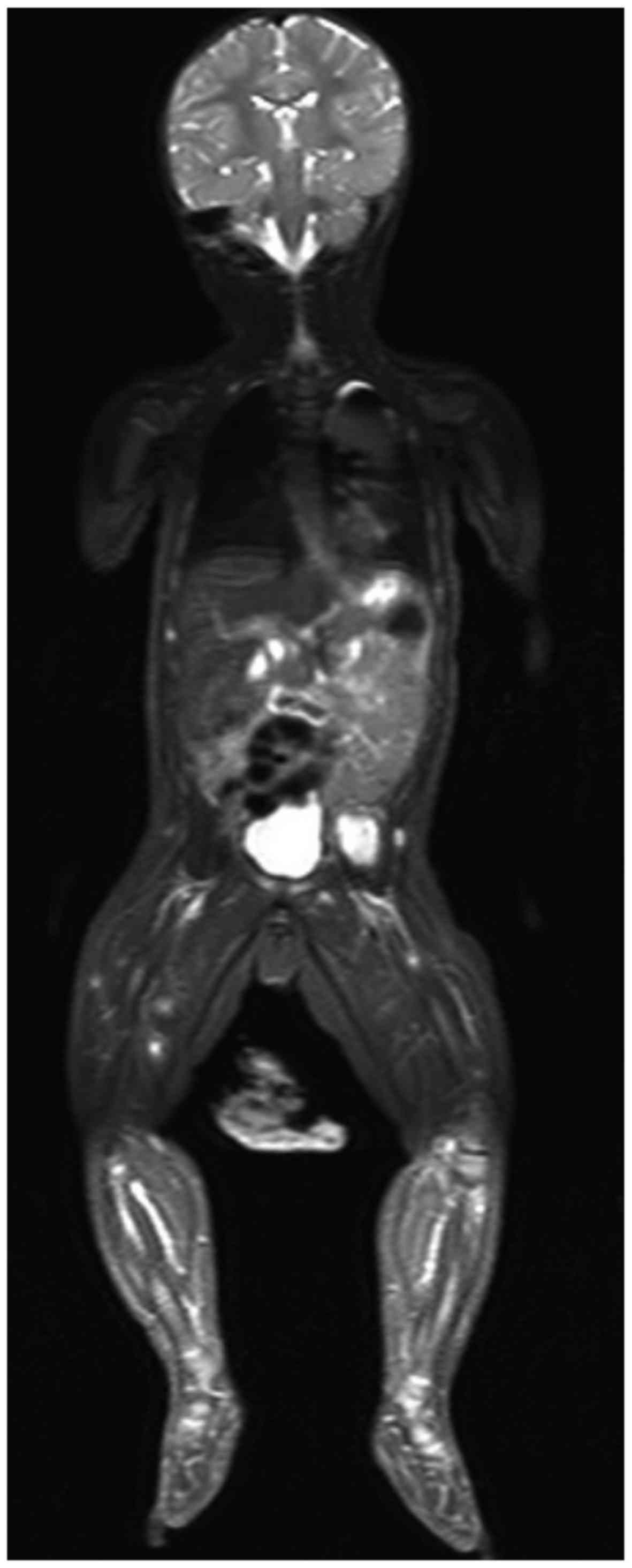
Whole-body MRI was performed for panoramic
localization of the nodules using short TI inversion recovery
(STIR) and diffusion-weighted whole-body imaging with background
body signal suppression (DWIBS) sequences in the coronal or axial
planes and volumetric 3D reformatting, to provide a detailed
assessment of the lesions and an accurate estimation of the disease
status.
The MRI revealed a significant number of nodules
(~70 lesions), with almost exclusive muscle localization, in the
head, trunk, abdomen, and upper and lower limbs.
STIR revealed multiple widespread lesions,
characterized by a hyperintense core, surrounded by a rim of low
signal (Fig. 1). The largest lesion
was detected in the left iliopsoas muscle (34×26×20 mm), whereas no
compression or infiltration of adjacent tissues was observed, other
than mild ureteral compression.
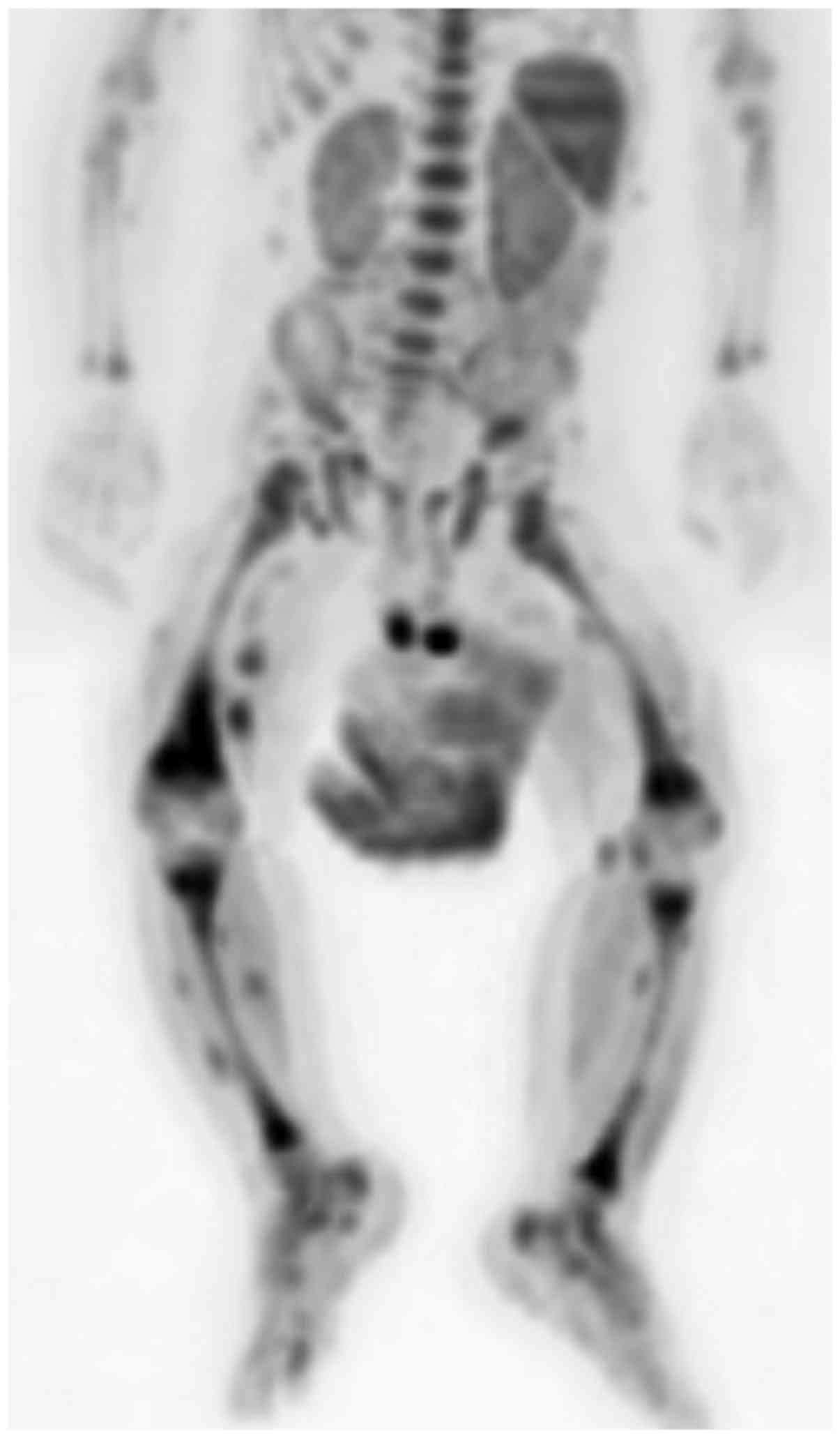
DWIBS displayed multiple widespread loci of high
intensity (Fig. 2), which on the
apparent diffusion coefficient map exhibited an intermediate inner
signal and a lower signal on their rim.
A blurred mass in the IX right rib was also
identified; thus, to assess bone invasion and lung involvement, an
unenhanced lowdose computed tomography (CT) scan was performed,
which revealed a round cortical enlargement, but without a
sclerotic rim, in the IX rib; the lungs were of otherwise normal
appearance.
On the basis of the histological and radiological
characteristics, the diagnosis of multicentric musculoskeletal
myofibromatosis without visceral involvement was confirmed.
Clinical observation was considered the best approach in this case.
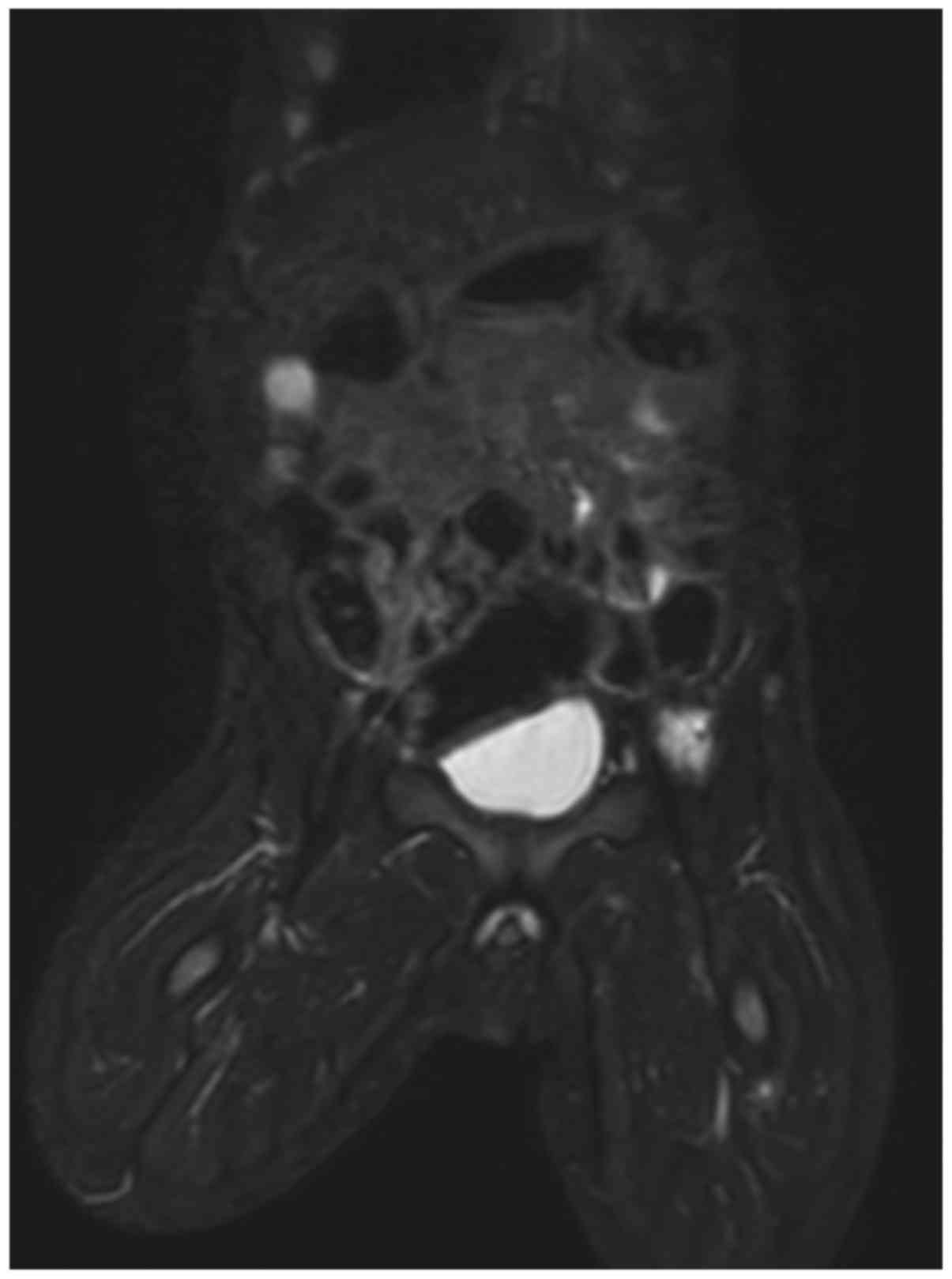
The patient was discharged after 1 week and follow-up with clinical
examination and whole-body MRI re-evaluation was performed at 1
year, demonstrating a reduction in the size and number of the
lesions (Fig. 3). A noticeable
regression of disease was achieved and further clinical follow-up
was scheduled. The last physical examination was performed 2 years
after the first referral, reporting no palpable nodules or other
symptoms. Therefore, no further hospital follow-up was planned. The
patient's parents gave their consent to the publication of the case
details and associated images.
Discussion
Myofibromatosis is the most common fibrous tumor of
childhood, with 88% of the cases occurring prior to the age of 2
years. The lesions may be present at birth, or may develop during
early infancy; solitary tumors mainly affect males, whilst the
multicentric form is more common in females (1,7). The
precise etiology remains unknown, but genetic factors, autosomal
dominant as well as recessive, appear to be implicated (5).
Myofibromatosis clinically presents as single mass
or diffuse multiple nodules that may develop anywhere, from the
skin to subcutaneous tissues or viscera, characterized by
spindle-shaped cells, SMA and vimentin positivity, related to
myofibroblast proliferation; the lesions are often associated with
richly vascular central areas, hemangiopericytomalike, frequently
displaced by sheets of hyalinization or necrosis and calcification
due to extensive apoptosis (3–5).
According to several authors, hemangiopericytoma and infantile
myofibromatosis are a continuous spectrum of a single myofibroblast
disease (8). Therefore, the
appearance on radiological investigation may vary on the basis of
the abovementioned criteria (6).
Prognosis and treatment depend on the disease type,
namely the solitary or multicentric form with subcutaneous,
muscular and osseous involvement, or the multicentric form with
visceral spread. Solitary or multicentric disease without visceral
involvement is usually associated with a very good prognosis with
spontaneous regression, whereas the multicentric form with visceral
spread requires aggressive surgery and/or radiotherapy and
chemotherapy; however, the prognosis is often poor, particularly
when the lungs are involved (1,4,5). A solitary visceral form has rarely been
reported, and its prognosis and treatment depend on the involved
organ and associated symptoms (7).
For the abovementioned reasons, the determination of
disease extent and progression with imaging are mandatory (6). US is the firststep examination, due to
its availability, lack of ionizing radiation, and no need for
sedation; however, the findings are often nonspecific, and US
cannot fully investigate deep lesions, bone spread, visceral and
lung involvement (3).
The lesions of myofibromatosis appear on CT scan as
masses that exhibit soft tissue attenuation, or mildly higher,
although lower attenuation is also possible due to necrosis, and
sparse hyperdense loci of calcification may be present as well
(3,5).
However, CT scan should be avoided when possible,
due to ionizing radiation exposure and its relatively low contrast
resolution for soft tissues. CT should be limited to determining
eventual lung or bone involvement; the latter is usually seen as
lytic lesions, with or without cortical sclerosis, which is more
commonly observed when the lesions start to heal. Pulmonary
involvement is usually correlated with a worse prognosis (3–5).
MRI is considered a gold standard imaging technique
for soft tissue evaluation, due to its ability to provide a
radiationfree detailed assessment of the disease, even without
contrast media administration; it also enables early detection of
visceral involvement in myofibromatosis (6). MRI may also provide high-accuracy
musculoskeletal images and distinguish between various tissues,
such as muscle, fibrous tissue and fat.
In radiological literature, myofibromatosis nodules
usually display different MRI signals, depending on the main
pattern of the lesions, or on their ‘age’, according to several
authors (5,8): Younger nodules, characterized by an
immature, highly vascular central component (hemangiopericytoma
like) surrounded by bundles of myofibroblasts, are iso/hypointense
on T1W images before and after contrast compound administration,
and exhibit heterogeneous gadolinium (Gd) enhancement and high
signal on T2W sequences (5,6). In older lesions, the immature core is
replaced by mature myofibroblasts, or more often involutes in
sheets of hyalinization or necrosis and calcification. T1W
sequences usually reveal intermediate or lower intensity compared
with the surrounding muscle, and noticeable peripheral
heterogeneous Gd enhancement (target appearance), whereas in T2W
images they display a hyperintense core due to central necrosis.
Inner low signal foci in all sequences are detected in case of
inner lesion calcifications (3,5,6).
MRI plays a pivotal role in cases of widespread soft
tissue nodules characterized by myofibroblast proliferation in
pediatric patients, particularly when performed as whole-body MRI,
in order to evaluate multiple diffuse lesions (9).
Until recently, whole-body evaluation of
multicentric disease was only performed by nuclear medicine,
involving exposure to high doses of radiation. MRI, due to the new
dedicated coils, self-sliding table, new reconstruction software
and use of fast sequences, may substitute PET in certain cases of
cancer staging and in other systemic diseases (9).
Whole-body MRI enables a complete imaging survey
within a short time and allows panoramic detailed imaging of all
lesions sized >6 mm, it is able to investigate spread to
contiguous organs and may also be used in the follow-up to
determine lesion reduction and shrinkage, as in the present case.
Fast image acquisition is also useful in pediatric patients to
reduce sedation time during MRI acquisition (9,10).
Although there is no standardized technique for
performing whole-body MRI, the main sequences are STIR and DWIBS.
T1W, with or without contrast administration, increases
specificity, but it is not routinely performed as it prolongs
scanning time (10).
The application of STIR sequences in pediatric MRI
is well accepted, since the signal may aid in differentiating
tissue characteristics; in fact, the majority of the pathological
lesions are proton-rich and have extended T1 and T2 relaxation
times, exhibiting high signal on STIR, along with its ability to
provide images within a short time frame and to better suppress fat
signal compared with other T2 fat-saturated sequences (9,10).
DWIBS sequences provide information on lesion
architecture and enable functional 3D imaging of systemic disease
in freebreathing acquisition, depicting highly cellular tumors and
necrosis as high-signal lesions (9,10).
Moreover, DWIBS acquisitions can be postprocessed,
obtaining whole-body images in coronal planes and, inverting the
grey scale, it is possible to create PET-like images (9).
Whole-body MRI is a well-accepted imaging modality
for staging lymphoma; this modality has been extended to the safe
evaluation of other systemic diseases, found to be useful when a
whole-body panoramic assessment of a disease is required, not only
to establish its true extent, but also during follow-up to
determine its evolution (6,9). It must be mentioned that the
sensitivity of whole-body MRI with T1 and STIR sequences is
comparable to that of PET/CT in the detection of metastatic
diseases. DWIBS sequences increase sensitivity by demonstrating
tissue cellular architecture (9).
Whole-body MRI has shown to be able to provide a
detailed panoramic view of systemic disease along with functional
information within a reasonable time frame, without breath holding
and without radiation exposure, in a single short sedation session.
In the present case, whole-body MRI easily assessed all the sites
of myofibromatosis lesions, and provided detailed information
regarding the extent of the disease, spread in contiguous organs
and its evolution during follow-up.
References
|
1
|
Chung EB and Enzinger FM: Infantile
myofibromatosis. Cancer. 48:1807–1818. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Gopal M, Chahal G, Al-Rifai Z, Eradi B,
Ninan G and Nour S: Infantile myofibromatosis. Pediatr Surg Int.
24:287–291. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Koujok K, Ruiz RE and Hernandez RJ:
Myofibromatosis: Imaging characteristics. Pediatr Radiol.
35:374–380. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Larralde M, Hoffner MV, Boggio P, Abad ME,
Luna PC and Correa N: Infantile myofibromatosis: Report of nine
patients. Pediatr Dermatol. 27:29–33. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Murphey MD, Ruble CM, Tyszko SM,
Zbojniewicz AM, Potter BK and Miettinen M: From the archives of the
AFIP musculoskeletal fibromatoses: radiologic-pathologic
correlation. Radiographics. 29:2143–2173. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Counsell SJ, Devile C, Mercuri E, Allsop
JM, Birch R and Muntoni F: Magnetic resonance imaging assessment of
infantile myofibromatosis. Clin Radiol. 57:67–70. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Menéndez-Arzac R, Valdez-Méndez D, Landa
R, Guzmán S, Cárdenas E and Cano AM: Solitary infantile
gastrointestinal myofibroma: Case report. J Pediatr Surg.
40:1361–1363. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Mentzel T, Calonje E, Nascimento AG and
Fletcher CD: Infantile hemangiopericytoma versus infantile
myofibromatosis. Study of a series suggesting a continuous spectrum
of infantile myofibroblastic lesions. Am J Surg Pathol. 18:922–930.
1994. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Chavhan GB, Babyn PS and Whole-Body MR:
Imaging in Children: Principles, Technique, Current
ApplicationsCurrentApplications, and Future Directions.
Radiographics. 31:1758–1774. 2011. View Article : Google Scholar
|
|
10
|
Teixeira SR, Junior J Elias,
Nogueira-Barbosa MH, Guimarães MD, Marchiori E and Santos MK:
Whole-body magnetic resonance imaging in children: State of the
art. Radiol Bras. 48:111–120. 2015. View Article : Google Scholar : PubMed/NCBI
|