Introduction
Patients with cancer have an increased risk of
venous thromboembolism (VTE) that varies substantially depending on
the cancer type (1). Clear cell
carcinoma (CCC) of the ovary constitutes 25% of epithelial ovarian
cancer (EOC), exhibiting a higher incidence of VTE than other
cancers (1). D-dimer, a soluble
fibrin-degradation product, is a valuable marker for diagnosing VTE
(2). The D-dimer test is
frequently positive for VTE and inflammatory diseases, cancer,
elderly age, surgery, trauma, pregnancy, and postpartum (3). However, the D-dimer test showed high
sensitivity and negative predictive value (NPV) but low specificity
for detecting VTE events (4).
Imaging techniques, including venous ultrasound, computed
tomography angiography, or both, are needed for diagnosing or
excluding VTE (5). Thus, novel and
effective strategies are needed to improve the specificity of VTE
diagnosis and increase the clinical usefulness of the D-dimer
test.
Tissue factor (TF) initiates the extrinsic
coagulation pathway by binding to factor VIIa (FVIIa) and factor Xa
(FXa) (the extrinsic coagulation initiation complex: TF-FVIIa-FXa)
and plays a vital role in thrombosis development (6). Tissue factor pathway inhibitor (TFPI)
is composed primarily of three Kunitz-type proteinase inhibitor
domains (K1, K2, and K3) that downregulate TF-induced coagulation
cascade (7). Furthermore, TFPI is
classified into TFPI1 and TFPI2 [also known as placental protein 5
(PP5)] (7). Several studies have
demonstrated that TFPI1 downregulation is a valuable clinical
predictor of deep vein thrombosis (DVT) (8-13).
Recently, TFPI2 has been verified as a novel serum marker for
diagnosing EOC, especially CCC (14-16).
However, no study has focused on TFPI2 as a marker for clinically
diagnosing VTE in various cancers, including EOC. Therefore, this
study assesses whether the TFPI2 test helps diagnose VTE in
patients with EOC and positive D-dimer results.
Materials and methods
Patient population
From January 2008 to December 2015, 223 patients
with EOC were treated at the Department of Gynecology, Nara Medical
University Hospital. The patients were invited to give biobanking
consent for future research. Informed consent for secondary use of
biospecimens was obtained in August 2021 by an opt-out approach in
all included patients. This study was approved by the Institutional
Review Board and the Research and Ethical Committee of Nara Medical
University Graduate School of Medicine Kashihara, Japan (no. 3061).
Furthermore, plasma D-dimer levels were measured before surgery or
chemotherapy. However, serum TFPI2 levels were measured using
frozen serum samples from a biobank. Inclusion criteria were: i)
patients undergoing surgery involving lesion removal for
histological evaluation; ii) patients with histologically proven
primary EOC; and iii) patients who could measure serum TFPI2 level
using frozen pretreatment samples. The exclusion criteria were: i)
age below 20 years; ii) a history of other malignancies or ovarian
cancers other than EOC; iii) patients with no residual sample; iv)
patients with negative D-dimer results (D-dimer <1.0 ug/ml); v)
previous VTE; vi) currently suspected or symptomatic DVT or
pulmonary thromboembolism (PTE); vii) lack of preoperative VTE
screening (contraindications to iodinated contrast medium or
emergency surgical intervention); viii) taking any drug affecting
blood coagulation or fibrinolytic parameters (e.g., oral
contraceptives, anticoagulant, antiplatelet drug, and fibrinolytic
drugs); ix) severe concomitant comorbidities; and x) incomplete,
inadequate, or missing data. In addition, the following
clinicopathological, hematological, and imaging data were obtained
from the patients' medical records: age, body mass index (BMI;
kg/m2), serum TFPI2 level (pg/ml), plasma D-dimer level
(µg/ml), serum CA125 level (U/ml), Federation of Gynecology and
Obstetrics (FIGO) (International Federation of Gynecologists and
Obstetricians) stage, histology, presence or absence of residual
tumor, estimated ascites volume, history of VTE, anticoagulant use,
and the presence or absence of comorbidities. Finally, stage
classification was based on the FIGO 2014 guidelines.
Screening and detection of VTE
Primary VTE screening was performed by evaluating
pathognomonic clinical signs or symptoms and plasma D-dimer levels.
Clinical VTE features were painful leg swelling and tenderness,
tachycardia, dyspnea, shortness of breath, chest pain, acute
cardiovascular dysfunction, and loss of consciousness. Patients
with clinical signs of VTE immediately underwent multidetector-row
computed tomography scanning (SOMATOM Definition Flash, SOMATOM
Definition As, and SOMATOM Emotion; Siemens Healthcare Diagnostics
K.K.) with intravenously injected low-osmolar iodinated contrast
mediums (17). All asymptomatic
patients were initially evaluated by the D-dimer test on admission.
Patients with positive D-dimer results (>1.0 µg/ml) further
underwent bilateral whole-leg compression ultrasonography and
contrast-enhanced CT chest, abdomen, and lower extremity scan to
detect VTE (17). Furthermore, VTE
was detected using a LOGIQ 9 ultrasound scanner (GE Healthcare)
equipped with a 4-7 MHz transducer (17). Furthermore, contrast-enhanced CT
scans were performed from the chest to the pelvis in patients with
negative D-dimer results as a preoperative examination.
Board-certified radiologists diagnosed VTE based on the image
findings.
Collection and preservation of blood
samples
Patients' peripheral venous blood samples were
collected ~4 weeks before surgery or chemotherapy. First, 7 ml
venous blood samples were collected in vacuum tubes containing and
not containing sodium citrate (3.8%) from the cubital veins of
subjects. Then, these tubes were centrifuged at 3,000 x g for at
least 10 min at 4˚C. Finally, the separated sample was divided into
1,000 µl aliquots and stored at -80˚C until further use.
Detection of plasma D-dimer and serum
TFPI2
First, D-dimer concentrations were measured at the
Department of Clinical Laboratory in Nara Medical University
Hospital. Then, plasma D-dimer levels were measured using DD
immunoenzymometric assay kit (Nanopia) using an automated
immunoassay analyzer. Furthermore, VTE screening was performed
based on the D-dimer levels, with a 1.0 µg/ml cutoff value for
plasma D-dimer. Next, TFPI2 concentrations were measured at the
Tosoh diagnostics product divisions (Tosoh Corporation, Kanagawa,
Japan). Additionally, using two anti-TFPI2 monoclonal antibodies,
the TFPI2 concentration in serum samples was measured using E-test
Tosoh II (AIA-PACK TFPI2) with an automated immunoassay
analyzerAIA-2000 (Tosoh Corporation) (9). These assays demonstrated high
reliability for intra-assay (2.2%-2.7%) and inter-assay (2.4%-3.1%)
variability. Finally, measurements were performed by clinical
laboratory technologists who were blinded to the study.
Statistical analysis
Statistical analysis was conducted using SPSS
(Statistical Package for the Social Sciences, v.25, SPSS, Inc.).
Clinicopathological characteristics were compared using the
chi-squared test or Mann-Whitney U test. The
receiver-operating characteristic (ROC) curve was constructed, and
areas under the curve were calculated to illustrate the diagnostic
power of TFPI2. The maximum Youden index determined the best cutoff
value for TFPI2. Furthermore, Spearman's correlation coefficient
was used to analyze the correlation between D-dimer and TFPI2
levels. Finally, P<0.05 was considered to indicate a
statistically significant difference.
Results
Characteristics of the study
population and clinical features
A total of 223 subjects were admitted to our
hospital during the research period. All patients had consented to
biobanking, but 79 patients had no residual serum samples. However,
63 subjects were excluded from the study, including those with
suspicious or symptomatic VTE (n=2), those with negative D-dimer
results (<1.0 µg/ml, n=41), those whose D-dimer level was not
measured (n=12), and those with incomplete data (n=8). Finally, 81
patients with EOC who met the inclusion and exclusion criteria
participated in this study. VTE was objectively detected in 25
patients (30.9%) and absent in 56 patients (69.1%), and participant
selection is shown in Fig. 1.
Baseline characteristics of patients with EOC and with and without
VTE are presented in Table I.
Additionally, the median age was 60 years (range, 37-83 years), and
CCC accounted for 22.2% of all EOC. During diagnosis, 66.7% of
patients had advanced EOC. However, compared with the non-VTE
group, the VTE group had higher D-dimer and TFPI2 levels (P=0.004
and P=0.001, respectively). In contrast, there were no differences
between both groups in age, BMI, CA125 level, ascites volume,
residual tumor, histology, and FIGO stage.
 | Table IClinicopathological characteristics of
patients with epithelial ovarian cancer, with and without VTE. |
Table I
Clinicopathological characteristics of
patients with epithelial ovarian cancer, with and without VTE.
| Factor | VTE (n=25) | non-VTE (n=56) | P-value |
|---|
| Age, year, median
(range) | 58 (40-81) | 61 (37-83) | 0.546a |
| BMI,
kg/m2, median (range) | 22.6 (17.0-36.0) | 22.0 (15.2-34.3) | 0.101a |
| TFPI2, pg/ml, median
(range) | 472.2
(199.4-1,288.4) | 279.1
(116.1-5,824.2) | 0.001a |
| D-dimer, µg/ml,
median (range) | 5.1 (1.2-35.2) | 2.6 (1.1-15.5) | 0.004a |
| CA125, U/ml, median
(range) | 1,021.6
(17.5-8,386.0) | 503.3
(8.2-43,170.9) | 0.236a |
| Massive
ascitesc, n | | | |
|
No | 13 | 31 | 0.779b |
|
Yes | 12 | 25 | |
| Residual tumor,
n | | | |
|
No | 8 | 21 | 0.663b |
|
Yes | 17 | 35 | |
| Histology, n | | | |
|
CCC | 6 | 12 | 0.797b |
|
Non-CCC | 19 | 44 | |
| FIGO
staged, n | | | |
|
I/II | 9 | 18 | 0.734b |
|
III/IV | 16 | 38 | |
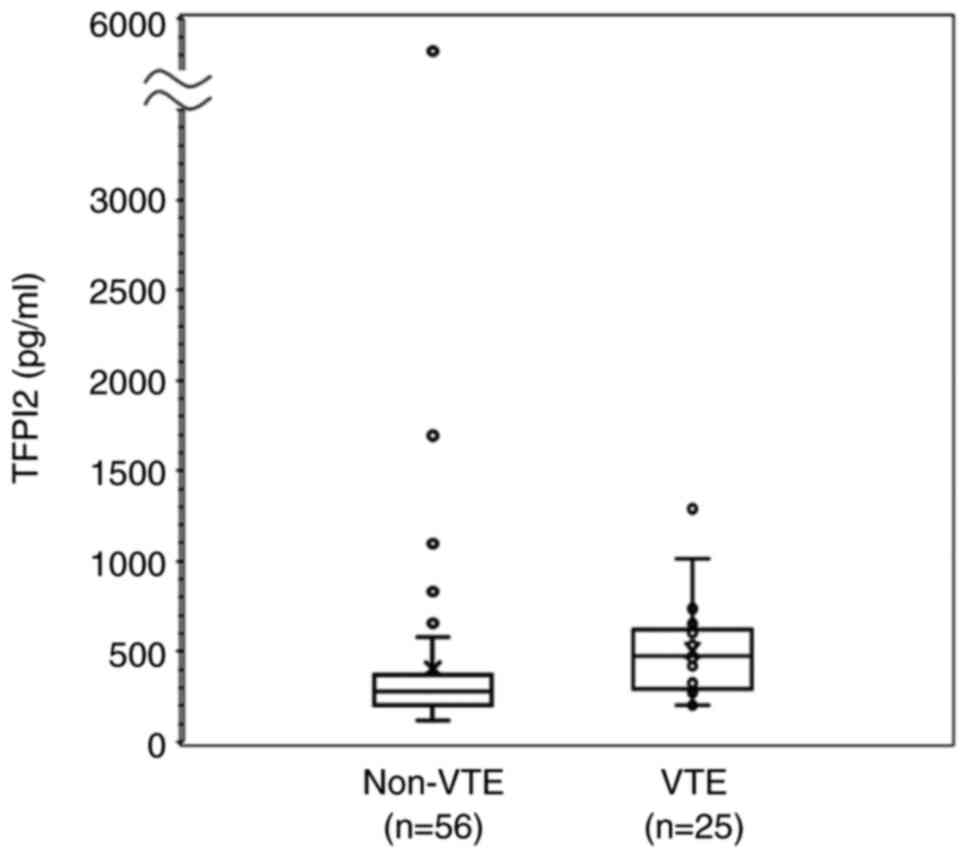
Serum TFPI2 level in patients with VTE
and non-VTE
We examined serum TFPI2 distribution in patients
with non-VTE (n=56) and VTE (n=25) by Box and Whisker plot
analysis. Also, serum TFPI2 levels in patients with VTE are
significantly higher than those with non-VTE (Mann-Whitney U
test, P=0.001; Fig. 2).
TFPI2 diagnostic value
ROC curve analysis was performed to assess the TFPI2
level between VTE and non-VTE groups in patients with EOC and
positive D-dimer results (Fig. 3).
The area under the ROC curve was 0.729 [95% confidence interval
(CI) 0.614-0.844]. Using the Youden index, the optimal cutoff value
for the TFPI2 level was set at 398.9 pg/ml. With this cutoff value,
the sensitivity, specificity, PPV, and NPV of the TFPI2 test were
64.0, 80.4, 59.3, and 83.3%, respectively, in diagnosing VTE.
Correlations between D-dimer and TFPI2
levels
The correlation between D-dimer and TFPI2 levels is
presented in Fig. 4. The Spearman
correlation coefficient between the levels of D-dimer and TFPI2 was
0.063 (P=0.574). Finally, there was no significant correlation
between D-dimer and TFPI2 levels.
Discussion
TFPI2 is a novel serodiagnostic marker for EOC
(14-16).
TFPI2 levels were significantly increased in patients with CCC than
those without CCC, suggesting that TFPI2 may help diagnose CCC
(14-16).
The TFPI2 test has been covered by the Japanese national health
insurance system since April 2021. Furthermore, we showed for the
first time that TFPI2 might effectively diagnose asymptomatic VTE
in patients with EOC and positive D-dimer assay results.
Additionally, in patients with EOC and positive D-dimer results,
the sensitivity and specificity of the TFPI2 test were 64.0 and
80.4%, respectively, in diagnosing VTE, with a cutoff value of
398.9 pg/ml. Finally, 80.4% of patients with TFPI2 levels <398.9
pg/ml were VTE negative.
First, EOC, especially CCC, increases the risk of
VTE (1,18). DVT was detected in 22.7% of
patients with EOC, which was higher than those (<10%) reported
in other malignancies (19).
Additionally, women with CCC were associated with a 2.5 times
higher risk for VTE than women with other histological types of EOC
(18). More than 80% of women with
DVT were asymptomatic, and accurately predicting VTE before
treatment is challenging (18).
Therefore, a reliable, non-invasive diagnostic and screening tool
for accurately evaluating VTE is needed. D-dimer testing is the
most widely used for VTE evaluation, but its low specificity
requires definitive confirmation by imaging techniques (20). Venous ultrasonography and
contrast-enhanced CT scans are expensive and time-consuming, so
novel biomarkers that enhance the diagnostic performance combined
with D-dimer are required. Furthermore, TFPI2 may be a
serodiagnostic marker that fits this purpose. It is unclear whether
two-step screening with D-dimer and TFPI2 is clinically more
beneficial than D-dimer and ultrasonography, but it may reduce the
burden on the ultrasound specialist. However, we cannot compare the
diagnostic performance of TFPI2 with that of D-dimer because the
former was not measured in all patients. In addition, the
sensitivity and specificity of TFPI2 are targeted for patients with
a D-dimer >1 µg/ml.
Second, TFPI, including TFPI1 and TFPI2, mainly
inhibits blood coagulation induced by TF. TF, a potent initiator of
the extrinsic coagulation cascade, can be released into the
systemic circulation from viable tumor cells, especially CCC,
activating the coagulation system and increasing the risk of DVT
(21). TFPI1 levels were
significantly downregulated in patients with DVT compared with
non-DVT patients (8-13),
suggesting that low TFPI1 levels increase DVT risk. Surprisingly,
TFPI2 levels were increased in patients with VTE compared with
TFPI1 levels. Elevated TFPI2 should suppress TF activity,
inhibiting thrombus formation. Rather, elevated TFPI2 is a
compensatory mechanism that may reflect beneficial adaptations to
maintain blood coagulation homeostasis. In contrast, TFPI2, unlike
TFPI1, acts as a potent plasmin inhibitor. Plasmin inactivates
coagulation factors, FV, FVIII, FIX, and FX (22). Plasmin is also involved in
fibrinolysis and contributes to fibrin degradation and clot removal
(23). TFPI2 may inhibit the
removal of organized clots by plasmin. Elucidation of the mechanism
of TFPI2 production and thrombosis formation in EOC is awaited.
Finally, there are several limitations to this
study. First, this is a single-center study conducted in the
Department of Gynecology at University Hospital. Second, This study
is limited to the diagnosis of asymptomatic DVT in patients with
positive D-dimer results, hampering the broader applicability in
clinical practice. Third, there is currently no evidence that TFPI2
has higher specificity than D-dimer. Further study is needed to
assess whether TFPI2 provides the clinical utility with the current
D-dimer test to improve VTE diagnosis.
Conclusively, TFPI2 may improve the diagnostic
performance of asymptomatic VTE in patients with EOC and positive
D-dimer results. However, a prospective study using a larger cohort
of patients with EOC is needed to determine if the combination test
of the two markers helps screen for detecting VTE.
Acknowledgements
Not applicable.
Funding
Funding: This study was supported by the Japan Society for the
Promotion of Science (JSPS) (grant no. 20K09647).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
HK made substantial contributions to conception and
design. RM, SY and RK were responsible for the acquisition of data.
NO and SM contributed to measuring TFPI2 levels. YY was responsible
for the analysis and interpretation of data. RM and YY confirm the
authenticity of all the raw data. The first draft of the manuscript
was written by RM under the guidance of HK. All authors have read
and approved the final manuscript.
Ethics approval and consent to
participate
The present study was approved by the medical ethics
committee of the Nara Medical University (approval no. 3061;
Kashihara, Japan). Written informed consent was obtained from each
patient. All procedures followed were in accordance with the
ethical standards of the responsible committee on human
experimentation (institutional and national) and with the Helsinki
Declaration of 1964 and its later amendments.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Matsuura Y, Robertson G, Marsden DE, Kim
SN, Gebski V and Hacker NF: Thromboembolic complications in
patients with clear cell carcinoma of the ovary. Gynecol Oncol.
104:406–410. 2007.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Weitz JI, Fredenburgh JC and Eikelboom JW:
A test in context: D-dimer. J Am Coll Cardiol. 70:2411–2420.
2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Hou H, Ge Z, Ying P, Dai J, Shi D, Xu Z,
Chen D and Jiang Q: Biomarkers of deep venous thrombosis. J Thromb
Thrombolysis. 34:335–346. 2012.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Wells PS, Anderson DR, Rodger M, Forgie M,
Kearon C, Dreyer J, Kovacs G, Mitchell M, Lewandowski B and Kovacs
MJ: Evaluation of D-dimer in the diagnosis of suspected deep-vein
thrombosis. N Engl J Med. 349:1227–1235. 2003.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Andrews EJ Jr and Fleischer AC: Sonography
for deep venous thrombosis: Current and future applications.
Ultrasound Q. 21:213–225. 2005.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Kocatürk B and Versteeg HH: Tissue
factor-integrin interactions in cancer and thrombosis: Every Jack
has his Jill. J Thromb Haemost. 11 (Suppl 1):S285–S293.
2013.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Peraramelli S, Thomassen S, Heinzmann A,
Rosing J, Hackeng TM, Hartmann R, Scheiflinger F and Dockal M:
Inhibition of tissue factor:factor VIIa-catalyzed factor IX and
factor X activation by TFPI and TFPI constructs. J Thromb Haemost.
12:1826–1837. 2014.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Fei X, Wang H, Yuan W, Wo M and Jiang L:
Tissue factor pathway inhibitor-1 is a valuable marker for the
prediction of deep venous thrombosis and tumor metastasis in
patients with lung cancer. Biomed Res Int.
2017(8983763)2017.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Sidelmann JJ, Bladbjerg EM, Gram J,
Münster AM and Jespersen J: Tissue factor pathway inhibitor relates
to fibrin degradation in patients with acute deep venous
thrombosis. Blood Coagul Fibrinolysis. 19:405–409. 2008.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Dahm A, Rosendaal FR, Andersen TO and
Sandset PM: Tissue factor pathway inhibitor anticoagulant activity:
Risk for venous thrombosis and effect of hormonal state. Br J
Haematol. 132:333–338. 2006.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Dahm A, Van Hylckama Vlieg A, Bendz B,
Rosendaal F, Bertina RM and Sandset PM: Low levels of tissue factor
pathway inhibitor (TFPI) increase the risk of venous thrombosis.
Blood. 101:4387–4392. 2003.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Amini-Nekoo A, Futers TS, Moia M, Mannucci
PM, Grant PJ and Ariëns RA: Analysis of the tissue factor pathway
inhibitor gene and antigen levels in relation to venous thrombosis.
Br J Haematol. 113:537–543. 2001.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Fedi S, Gori AM, Falciani M, Cellai AP,
Aglietti P, Baldini A, Vena LM, Prisco D, Abbate R and Gensini GF:
Procedure-dependence and tissue factor-independence of
hypercoagulability during orthopaedic surgery. Thromb Haemost.
81:874–878. 1999.PubMed/NCBI
|
|
14
|
Arakawa N, Miyagi E, Nomura A, Morita E,
Ino Y, Ohtake N, Miyagi Y, Hirahara F and Hirano H: Secretome-based
identification of TFPI2, a novel serum biomarker for detection of
ovarian clear cell adenocarcinoma. J Proteome Res. 12:4340–4350.
2013.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Arakawa N, Kobayashi H, Yonemoto N,
Masuishi Y, Ino Y, Shigetomi H, Furukawa N, Ohtake N, Miyagi Y,
Hirahara F, et al: Clinical significance of tissue factor pathway
inhibitor 2, a serum biomarker candidate for ovarian clear cell
carcinoma. PLoS One. 11(e0165609)2016.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Miyagi E, Arakawa N, Sakamaki K, Yokota
NR, Yamanaka T, Yamada Y, Yamaguchi S, Nagao S, Hirashima Y,
Kasamatsu Y, et al: Validation of tissue factor pathway inhibitor 2
as a specific biomarker for preoperative prediction of clear cell
carcinoma of the ovary. Int J Clin Oncol. 26:1336–1344.
2021.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Kawaguchi R, Furukawa N and Kobayashi H:
Cut-off value of D-dimer for prediction of deep venous thrombosis
before treatment in ovarian cancer. J Gynecol Oncol. 23:98–102.
2012.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Duska LR, Garrett L, Henretta M, Ferriss
JS, Lee L and Horowitz N: When ‘never-events’ occur despite
adherence to clinical guidelines: The case of venous
thromboembolism in clear cell cancer of the ovary compared with
other epithelial histologic subtypes. Gynecol Oncol. 116:374–377.
2010.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Ebina Y, Uchiyama M, Imafuku H, Suzuki K,
Miyahara Y and Yamada H: Risk factors for deep venous thrombosis in
women with ovarian cancer. Medicine (Baltimore).
97(e11009)2018.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Bates SM, Jaeschke R, Stevens SM, Goodacre
S, Wells PS, Stevenson MD, Kearon C, Schunemann HJ, Crowther M,
Pauker SG, et al: Diagnosis of DVT: Antithrombotic therapy and
prevention of thrombosis, 9th ed: American college of chest
physicians evidence-based clinical practice guidelines. Chest. 141
(Suppl 2):e351S–e418S. 2012.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Geddings JE and Mackman N: Tumor-derived
tissue factor-positive microparticles and venous thrombosis in
cancer patients. Blood. 122:1873–1880. 2013.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Hoover-Plow J: Does plasmin have
anticoagulant activity? Vasc Health Risk Manag. 6:199–205.
2010.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Medcalf RL: Fibrinolysis: from blood to
the brain. J Thromb Haemost. 15:2089–2098. 2017.PubMed/NCBI View Article : Google Scholar
|