Introduction
Multiple osteochondroma (MO), previously known as
hereditary multiple exostoses, was first described by Boyer in
1814(1). It occurs as benign
multiple cartilaginous bone tumors in early childhood and ossifies
when skeletal growth is complete. MO occurs at a rate of 1 in
50,000 in Western populations, exhibiting a male-to-female ratio of
1.5(2). It is also inherited in an
autosomal-dominant manner, with a penetrance of approximately 96
and 100% in females and males, respectively (3). Osteochondromas primarily involve the
metaphyses and diaphyses of long bones, including the ribs
(4), and are associated with
reduced skeletal growth, bone deformities, limited joint range of
motion, short stature, premature osteoarthritis, and peripheral
nerve compression (5). Surgeries
for osteochondromas are based on the symptoms, such as pain,
swelling, or reduced range of motion (6). Heterozygous defects in the
exostosin-1 (EXT1) and exostosin-2 (EXT2) genes have
been suggested to induce MO (7,8), and
the lifetime risk of malignant transformation in MO is estimated at
4% (9). Another study suggested
that patients with EXT1 mutations statistically have a 1.5
times higher risk of malignant transformation than those with
unstratified gene or EXT2 mutations (10).
Osteoid osteomas account for approximately 3 and 11%
of all bone tumors and benign ones, respectively (11), with a male-to-female ratio of
approximately 2-3:1. They can occur in any bone of the body.
Osteoid osteomas frequently occur in males aged between 10 and 30
years and affect long bones, such as the femur and tibia (11,12).
They generally occur in the long bones of the lower extremities,
with the femoral neck as the most frequent site (13). Osteoid osteoma has a sclerotic bony
lesion with a diameter of 2 cm known as the nidus and is considered
to induce pain (14). The lesion
exhibited a high level of prostaglandin E2 that caused severe pain;
it was usually treated with nonsteroidal anti-inflammatory drugs
(NSAIDs) (15), which are
typically effective nonsurgical treatments for pain in osteoid
osteomas (15). However, many
patients cannot continue taking NSAIDs in the long term; therefore,
they usually require surgery. Recently, Fittall et al
reported that recurrent rearrangements of FOS or FOSB
were found in osteoblastomas and osteoid osteomas (16). However, to our knowledge, no
association has been reported between EXT1/EXT2 and
FOS/FOSB.
Although MO and osteoid osteoma are rare, their
coexistence has never been reported. Therefore, we describe a case
of coexisting MO and osteoid osteoma that was treated with marginal
excision and curettage at our institution.
Case report
A 16-year-old male patient with a 3-month history of
left calf pain and a bony mass on the left knee from the previous
hospital was referred to the Department of Orthopedic Surgery,
University of the Ryukyus in December 2019. The patient had a
family history of MO, for which the mother and maternal grandmother
underwent surgery. The patient was 158 cm tall, weighed 46 kg, and
had no relevant medical history. Although the patient did not
report night pain, physical examination showed multiple bony
prominences around both knee joints and tenderness on the medial
side of the left lower leg on a 60 mm pain visual analog scale
(VAS). Laboratory data showed that inflammatory reactions and serum
alkaline phosphatase levels were not elevated. Radiography revealed
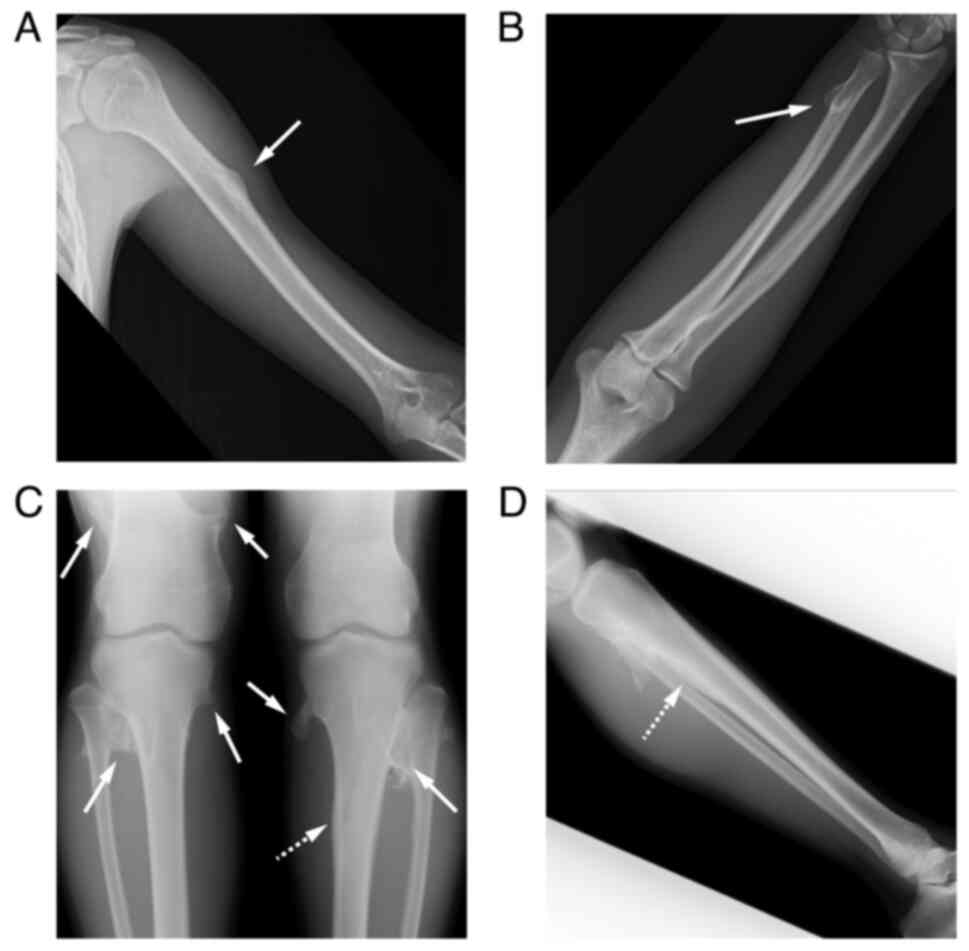
MOs on the left proximal humerus, right distal ulna, right distal
femur, proximal tibiae, and fibulas (Fig. 1A-D). Additionally, the epiphyseal
line was almost closed. Computed tomography (CT) revealed a
fungiform osteochondroma on the medial side of the proximal tibia
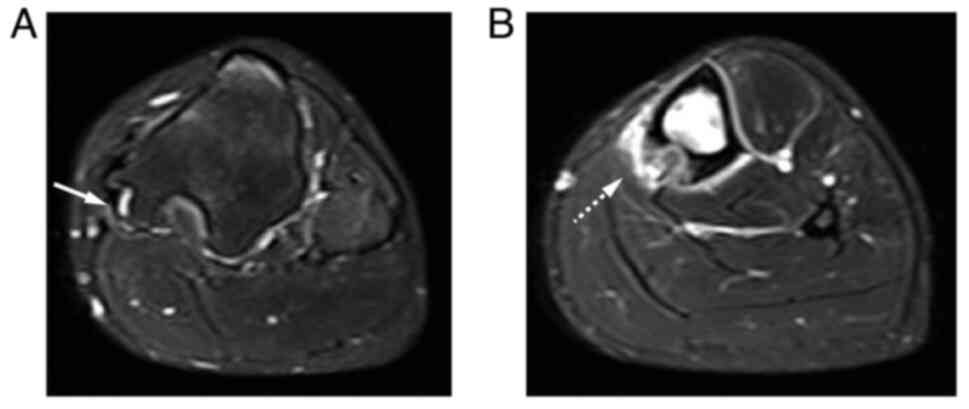
and the nidus of the medial shaft of the tibia (Fig. 2A and B). Magnetic resonance imaging revealed a
high-intensity layer of the cartilaginous cap on the fungiform
osteochondroma of the proximal tibia on short-tau inversion
recovery (STIR) images (Fig. 3A).
The nidus surrounding the bone edema was also confirmed in the left
tibial shaft on the STIR image (Fig.
3B). A 99mTc bone scintigraphy showed strong
accumulation in the medial tibial shaft. Based on the family
history and imaging findings, the patient was diagnosed with MO and
solitary osteoid osteoma of the left tibial shaft.
The patient underwent surgery to relieve the pain,
and marginal excision of the osteochondroma of the left proximal
tibia was performed. Cone-beam CT-guided curettage was also
performed during the same surgery for osteoid osteoma of the tibial
shaft. Notably, the pain in the left calf resolved, and the pain
VAS score was 0 mm immediately after surgery. Pathological
examinations revealed an osteoid formation surrounded by
osteoblasts in the osteoid osteoma lesion and a hyaline cartilage
cap in the osteochondroma lesion (Fig.
4A and B). However, no bony
prominence or pain recurrence occurred at the final follow-up 7
months postoperatively, and the patient could skateboard.
Discussion
We report, for the first time, to our knowledge, a
case of coexisting osteochondroma and osteoid osteoma, which were
located in the proximal tibia and tibial shaft. Although genetic
examinations, such as EXT1/EXT2 and
FOS/FOSB (7,8,16),
were not performed in this case, pathological examination confirmed
the proximal tibia and tibial shaft lesions as osteochondroma and
osteoid osteoma, respectively. Therefore, further investigation on
the genetic status of this case may be warranted to help understand
the genetic/pathologic mechanisms of coexisting MO and osteoid
osteoma.
Multiple osteoid osteomas are very rare. To our
knowledge, a total of 37 cases of multiple osteoid osteomas have
been reported in the literature (17-20).
Aynaci et al reported 24 cases of multiple osteoid osteomas
in the same bone, which included one case they reported (19). Six cases of multiple osteoid
osteomas in adjacent bones have been reported (19,21),
starting with Larsen et al (21), whereas sven cases in two widely
separated bones have been documented (17,20),
beginning with de Ga et al (17). Although osteochondroma and osteoid
osteoma coexisted in the tibia in this case, there might be an
etiological linkage between multiple osteoid osteomas and the
coexistence of osteochondroma and osteoid osteoma.
Furthermore, marginal excision of the osteochondroma
and CT-guided curettage of the osteoid osteoma on the proximal
tibia and tibial shaft, respectively, were performed in this case,
and the patient recovered from tibial pain immediately after
surgery, even though during short-term follow-up. Although no
recurrence of osteochondroma or osteoid osteoma was observed during
short-term follow-up, the patient should undergo long-term
follow-up to observe whether other bones with osteochondromas may
develop osteoid osteoma in the future.
In conclusion, we described the first case of
coexisting MO and osteoid osteoma, and marginal excision for
osteochondroma and curettage for osteoid osteoma effectively
improved the symptoms.
Acknowledgements
Not applicable.
Funding
Funding: This work was supported in part by the Japan Society
for the Promotion of Science (JSPS) KAKENHI Grant-in-Aid for
Scientific Research (C) (grant no. 21K09207) and a Grant-in-Aid for
Early-Career Scientists (grant no. 23K15718). Any options,
findings, conclusions or recommendations expressed in this material
are those of the authors and do not necessarily reflect the views
of the authors' organization or JSPS.
Availability of data and materials
All data generated and analyzed during this study
are included in this published article.
Authors' contributions
RK, HO, YA, KM, YT and KN conceived the study. RK,
HO and YT contributed to the data acquisition. RK, HO, YA, KM, YT
and KN wrote and edited the manuscript. HO and YT performed surgery
and postoperative management, respectively. YT and KN revised the
manuscript for intellectual content. RK and YT confirm the
authenticity of all the raw data. All authors have read and
approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Written informed consent for the publication and use
of images was obtained from the patient and their guardians.
Competing interests
YT is on the editorial board of the Cancer Diagnosis
and Prognosis. KN is on the editorial board of the Journal of
Orthopaedic Research and is a board member of the International
Society for the Study of Lumbar Spine.
References
|
1
|
Hennekam RC: Hereditary multiple
exostoses. J Med Genet. 28:262–266. 1991.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Bukowska-Olech E, Trzebiatowska W, Czech
W, Drzymala O, Frak P, Klarowski F, Klusek P, Szwajkowska A and
Jamsheer A: Hereditary multiple exostoses-a review of the molecular
background, diagnostics, and potential therapeutic strategies.
Front Genet. 12(759129)2021.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Schmale GA, Conrad EU III and Raskind WH:
The natural history of hereditary multiple exostoses. J Bone Joint
Surg Am. 76:986–992. 1994.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Stieber JR and Dormans JP: Manifestations
of hereditary multiple exostoses. J Am Acad Orthop Surg.
13:110–120. 2005.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Clement ND and Porter DE: Can deformity of
the knee and longitudinal growth of the leg be predicted in
patients with hereditary multiple exostoses? A cross-sectional
study. Knee. 21:299–303. 2014.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Bottner F, Rodl R, Kordish I, Winklemann
W, Gosheger G and Lindner N: Surgical treatment of symptomatic
osteochondroma. A three- to eight-year follow-up study. J Bone
Joint Surg Br. 85:1161–1165. 2003.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Ahn J, Ludecke HJ, Lindow S, Horton WA,
Lee B, Wagner MJ, Horsthemke B and Wells DE: Cloning of the
putative tumour suppressor gene for hereditary multiple exostoses
(EXT1). Nat Genet. 11:137–143. 1995.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Stickens D, Clines G, Burbee D, Ramos P,
Thomas S, Hogue D, Hecht JT, Lovett M and Evans GA: The EXT2
multiple exostoses gene defines a family of putative tumour
suppressor genes. Nat Genet. 14:25–32. 1996.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Jones KB, Pacifici M and Hilton MJ:
Multiple hereditary exostoses (MHE): Elucidating the pathogenesis
of a rare skeletal disorder through interdisciplinary research.
Connect Tissue Res. 55:80–88. 2014.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Fei L, Ngoh C and Porter DE:
Chondrosarcoma transformation in hereditary multiple exostoses: A
systematic review and clinical and cost-effectiveness of a proposed
screening model. J Bone Oncol. 13:114–122. 2018.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Lee EH, Shafi M and Hui JH: Osteoid
osteoma: A current review. J Pediatr Orthop. 26:695–700.
2006.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Zeng H, He H, Tong X, Wang Z, Luo R and
Liu Q: Osteoid osteoma of the proximal femur: Pitfalls in diagnosis
and performance of open surgical resection. Front Surg.
9(922317)2022.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Zhang Y and Rosenberg AE: Bone-forming
tumors. Surg Pathol Clin. 10:513–535. 2017.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Hasegawa T, Hirose T, Sakamoto R, Seki K,
Ikata T and Hizawa K: Mechanism of pain in osteoid osteomas: An
immunohistochemical study. Histopathology. 22:487–491.
1993.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Greco F, Tamburrelli F and Ciabattoni G:
Prostaglandins in osteoid osteoma. Int Orthop. 15:35–37.
1991.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Fittall MW, Mifsud W, Pillay N, Ye H,
Strobl AC, Verfaillie A, Demeulemeester J, Zhang L, Berisha F,
Tarabichi M, et al: Recurrent rearrangements of FOS and FOSB define
osteoblastoma. Nat Commun. 9(2150)2018.PubMed/NCBI View Article : Google Scholar
|
|
17
|
de Ga K, Bateni C, Darrow M, McGahan J,
Randall RL and Chen D: Polyostotic osteoid osteoma: A case report.
Radiol Case Rep. 15:411–415. 2020.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Schajowicz F and Lemos C: Osteoid osteoma
and osteoblastoma. Closely related entities of osteoblastic
derivation. Acta Orthop Scand. 41:272–291. 1970.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Aynaci O, Turgutoglu O, Kerimoglu S, Aydin
H and Cobanoglu U: Osteoid osteoma with a multicentric nidus: A
case report and review of the literature. Arch Orthop Trauma Surg.
127:863–866. 2007.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Beck SE, Schwab JH, Rosenthal DI,
Rosenberg AE and Grottkau BE: Metachronous osteoid osteoma of the
tibia and the T7 vertebral body: A case report. J Bone Joint Surg
Am. 93(e73)2011.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Larsen LJ, Mall JC and Ichtertz DF:
Metachronous osteoid-osteomas: Report of a case. J Bone Joint Surg
Am. 73:612–614. 1991.PubMed/NCBI
|