Introduction
Assisted reproductive technology (ART) has
demonstrated the potential to aid infertile couples to conceive
their own offspring since the birth of the first test tube baby in
1978 (1). Since then, novel ART
procedures and technologies have been developed to overcome various
infertility factors, including in vitro fertilization (IVF)
(1), intracytoplasmic sperm
injection (ICSI) (2) and
preimplantation genetic diagnosis (3). One simple but effective procedure was
developed in 1995, and involves a combination of IVF and ICSI using
sibling oocytes (4). With this
procedure, cases of unexpected total fertilization failure can be
avoided. Furthermore, this procedure is beneficial for patients
with unexplained infertility (5),
mild oligoteratoasthenozoospermia or borderline semen parameters
(6,7), severe isolated teratozoospermia
(8) and polycystic ovary syndrome
with normozoospermic semen (9,10).
Embryo selection is a critical issue in procedures
using sibling oocytes. In traditional IVF or ICSI, the embryos used
for embryo transfer (ET) are high-quality embryos; however, in
sibling oocyte procedures, embryo fertilization approaches should
also be considered. At present, the consensus amongst clinicians
and embryologists is that IVF embryos should be selected and
transferred prior to ICSI embryos with the same or marginally
better quality; however, others consider embryo quality to be the
gold standard for embryo selection prior to ET.
Previous studies have mainly focused on clinical
data analysis. Shveiky et al (11) reported that the development
competence of ICSI embryos, including fertilization, pregnancy and
implantation efficiency, was not different from that of IVF
embryos; Plachot et al (12) reported similar results. Becker
et al (13) suggested that
a higher number of top quality embryos were obtained following ICSI
than following IVF in sibling oocyte procedures, but found that the
clinical pregnancy efficiency was similar when IVF (three
pregnancies in 11 patients) and ICSI embryos were transferred
(eight pregnancies in 24 patients). Staessen et al (10) reported that whether the transferred
embryos were from ICSI alone, IVF alone, or a combination of IVF
and ICSI did not affect the pregnancy and implantation rates.
However, Kihaile et al (14) demonstrated that pregnancy and
implantation efficiency is increased when selecting mixed IVF and
ICSI embryos together, and in addition, only fertilization
efficiency by ICIS approach was superior to IVF-ET. In these
clinical studies, different conclusions were suggested based on
different infertility symptoms; therefore, it is difficult to be
certain of the effects of the fertilization approach on the
pregnancy and implantation results.
The other important consideration in selecting
embryos for transfer is single embryo transfer (SET), which has
been utilized in an increasing number of centers. In the USA, the
rate of SET tripled from 4.5% to 14.8% between 2001 and 2009
(15). Thurin et al
(16) performed a double-blind
multicenter trial among 11 clinics in Sweden, which found that the
birth rates were significantly lower following fresh transfer of
one versus two embryos, but the cumulative birth rates were not
statistically different between the treatment groups. Gardner et
al (17) demonstrated that the
ongoing pregnancy rates for SET versus double embryo transfer (DET)
were not significantly different in a study evaluating blastocyst
transfer. Therefore, SET has been demonstrated to be effective in
preventing multiple deliveries without compromising overall
pregnancy rates, although it is key in this strategy to select an
embryo with higher developmental competence, particularly for
sibling oocyte procedures that have two types of embryos from
different fertilization approaches. However, there are no relevant
studies regarding sibling oocyte procedures; certain studies have
focused on candidate genes and comprehensive chromosome screens in
traditional IVF or ICSI procedures (18,19).
In the present study, therefore, the pregnancy and
delivery outcomes of ICSI alone, IVF alone and a mixture of embryos
using mouse SET and multiple embryo transfer (MET) models were
compared, and the effect of transferring embryos from different
fertilization approaches in sibling oocyte procedures on
post-implantation development was investigated.
Materials and methods
All chemicals were purchased from Sigma-Aldrich
(Shanghai, China) unless otherwise indicated.
Ethics statement
The study was approved by the Institutional Review
Board at Tianjin First Central Hospital, Tianjin, China. All
experiments requiring the use of animals were approved by the
Committee on the Ethics of Animal and Medicine of the Peking
University Third Hospital, Beijing, China.
Experimental design
In the first experiment, the general characteristics
of ICSI and IVF embryos were compared, including pre-implantation
development competence, blastocyst cell number and apoptosis
analysis. In the second experiment, the pregnancy and delivery
outcomes were analyzed with two types of SET models. In the first
model, only one blastocyst from IVF or ICSI was transferred into
one female recipient. In the second model, six blastocysts from IVF
or ICSI were transferred into one recipient since mice are
multiparous and normally conceive with ~8–15 fetuses. In the third
experiment, the pregnancy and delivery outcomes were analyzed with
the MET model. The blastocysts were classified into five groups:
IVF, ICSI, and IVF and ICSI combined following a defined ratio of
1:1; 1:2 or 2:1. Twenty-four blastocysts in each group were
transferred into one recipient; a total of six recipients were used
in each group. In the fourth experiment, the cumulative pregnancy
rates in the SET models were calculated and compared with the MET
data.
In the IVF experiments, the oocytes were retrieved
from C57/BL6 female mice and the sperm were collected from DBA/2
strain male mice; the offspring were thus designated B6D2F1 with a
black coat color. In the ICSI experiments, the oocytes were also
retrieved from C57/BL6 female mice, but the sperm were collected
from C3H/HeJ strain male mice; the offspring were designated B6C3F1
with an agouti coat color.
Mice
All mice were purchased from the Vital River Company
(Beijing, China). The mice used in this study were 8–10 weeks of
age, and housed and bred in the Animal Center of the Medical
College of Peking University (Beijing, China), according to
national legislation for animal care. All mice were maintained
individually under controlled temperature and lighting conditions,
and provided food and water ad libitum.
C57/BL6 strain female mice (black coat color) were
used for oocyte retrieval. The DBA/2 strain male mice (dilute brown
coat color) and C3H/HeJ strain male mice (agouti coat color) were
used as sperm donors. ICR strain female mice (white coat color)
were used as ET recipients.
Oocyte collection
Oocytes were collected from the fallopian tubes of
adult C57/BL6 strain female mice that were superovulated by
injection of 10 IU pregnant mare serum gonadotropins (Hua Fu
Biotechnology Company, Tianjin, China) and 10 IU human chorionic
gonadotropin (hCG; Hua Fu Biotechnology Company), administered 48 h
apart. Following the retrieval of the oocytes from the oviducts
14–16 h after administration of hCG, the cumulus-oocyte complexes
(COCs) were separated into two groups. The COCs from the left
oviduct ampulla were digested with 100 U/ml hyaluronidase, and the
naked oocytes were cultured in M16 medium at 37°C and 5%
CO2 until ICSI micromanipulation. The COCs from the
right oviduct ampulla were used for IVF experiments. The COCs were
placed directly into CZB-culture medium at 37°C and 5%
CO2 without any treatment, and cultured for ~1 h prior
to IVF manipulation.
IVF
Conventional IVF was performed in human tubal fluid
(HTF) medium [Vitrolife AB (publ), Göteborg, Sweden], as described
in a previous study (20). The
sperm were collected from the cauda epididymis of adult DBA/2
strain male mice and capacitated at 37°C under 5% CO2
and 95% humidity for 1–2 h. The COCs were then moved and
co-cultured with motile sperm (final concentration,
1–2×106/ml). After 6 h, the COCs were removed from the
HTF medium and transferred to the M16 medium (Sigma-Aldrich, St.
Louis, MO, USA). Pronuclear formation was considered to represent
successful fertilization.
ICSI
ICSI was performed with a 10-mm internal diameter
Piezo-driven micropipette (Prime Tech, Ltd., Ibaraki, Japan), as
previously described (20). The
sperm were collected from the cauda epididymis of adult C3H/HeJ
strain male mice, and resuspended in Krebs–Ringer bicarbonate
solution at a density of 5–106/ml prior to incubation at
37°C with 5% CO2 in air for 45 min prior to ICSI. Groups
of oocytes were moved from M16 medium to M2 medium (Sigma-Aldrich,
St. Louis, MO, USA), and the spermatozoan heads were injected into
the oocyte cytoplasm, assisted with a Piezo apparatus (Prime Tech
Corp., Tokyo, Japan). The injected oocytes were cultured at 37°C
under 5% CO2 and 95% humidity, and pronuclear formation
was considered to represent successful fertilization after 6 h.
Blastocyst differential staining
The blastocyst cell number was determined, as
previously described (20). The
blastocysts were transferred to the medium with 0.5% pronase on day
four, and the zona pellucida was removed. The blastocysts were then
exposed to rabbit anti-mouse whole serum (Sigma-Aldrich, St. Louis,
MO, USA) for ~1 h, and rinsed three times in buffer medium for 5
min. The blastocysts were treated with guinea pig complement
(Sigma-Aldrich, St. Louis, MO, USA) and the treated blastocysts
were then stained with propidium iodide (10 mg/ml) and Hoechst
33342 (1 mg/ml). Twenty embryos in each group were used for
counting.
Apoptosis analysis
Apoptosis was analyzed using a terminal
deoxynucleotidyl-transferase-mediated dUTP nick end labeling
(TUNEL) apoptosis detection kit (Millipore, Billerica, MA, USA).
The blastocysts were fixed in 4% paraformaldehyde in
phosphate-buffered saline (PBS) for 20 min at 4°C, then subjected
to permeabilization for 20 min at room temperature with PBS
containing 0.2% Triton X-100. Briefly, the fixed and permeabilized
blastocysts were labeled with the TUNEL reaction mixture for 60 min
at 37°C. The blastocyst nuclei were also stained with DAPI. The
results were then analyzed using a Nikon fluorescence microscope
(A1R; Nikon, Tokyo, Japan). Twenty embryos in each group were
analyzed.
Embryo transfer
The blastocysts were transferred to the uterus of
2.5 day post coitum (dpc) pseudopregnant ICR mice. On day
17.5 dpc, the pregnant mice delivered naturally.
Statistical analysis
The results were evaluated using a χ2
test or t-test with SPSS 13.0 software (SPSS, Inc., Chicago, IL,
USA) and P<0.05 was considered to indicate a statistically
significant difference.
Results
General characteristics of IVF and ICSI
embryos
The pre-implantation development competence was
analyzed and compared between IVF and ICSI embryos. The
fertilization and cleavage efficiencies in the two groups were not
identified to be significantly different at the PN and 2-cell stage
(P>0.05), but the development efficiency of the IVF embryos was
significantly higher than the ICSI embryos at the 8-cell and
blastocyst stages (P<0.05; Fig.
1A). To assess the embryo quality in the two groups, the
blastocyst and apoptotic cell numbers were analyzed by differential
staining and TUNEL detection methods, respectively. The results
revealed that the inner cell mass (ICM) cell number and the ratio
of ICM were higher in the IVF group (P<0.05), although there was
no difference in the trophoderm cell number between the IVF and
ICSI groups (P>0.05). A higher cell apoptosis ratio was observed
in ICSI blastocysts (P<0.05; Fig.
1B).
Pregnancy and delivery outcomes of ICSI
and IVF embryos in SET models
In the SET model I, only one blastocyst from IVF or
ICSI was transferred into the recipients. The results revealed that
67.5% of the recipients receiving a single IVF blastocyst became
pregnant, which is comparable to single ICSI blastocysts (60%;
P>0.05). On day 17.5, a healthy neonate was born by natural
delivery from each pregnant recipient. In ICSI groups, 24 neonates
were born from a total 40 blastocysts, and in IVF group, 27
neonates were born from a total 40 blastocysts (P>0.05). In the
SET model II, six blastocysts from IVF or ICSI were transferred
into the recipients. The pregnancy efficiency did not differ
significantly between the IVF and ICSI groups (83.3 vs. 75.0%;
P>0.05), nor did the delivery rate (P>0.05) (Table I).
 | Table IThe pregnancy and delivery outcomes
of IVF and ICSI blastocyst using models SET I and II. |
Table I
The pregnancy and delivery outcomes
of IVF and ICSI blastocyst using models SET I and II.
| SET model | Group | No. of
recipients | No. of transferred
embryos per recipient | Total no. of
transferred embryos | No. of pregnant
mice (%) | No. of neonates
(%) |
|---|
| I | ICSI | 40 | 1 | 40 | 24 (60.0) | 24 (60.0) |
| IVF | 40 | 1 | 40 | 27 (67.5) | 27 (67.5) |
| II | ICSI | 24 | 6 | 144 | 18 (75.0) | 72 (50.0) |
| IVF | 24 | 6 | 144 | 20 (83.3) | 92 (63.9) |
Pregnancy and delivery outcomes of ICSI
and IVF embryos in MET models
In an effort to replicate human double or triple ET,
24 blastocysts were transferred into each recipient. Five MET
groups were designed as follows: A) 24 Blastocysts from ICSI; B) 16
blastocysts from ICSI and eight blastocysts from IVF; C) 12
blastocysts from ICSI and 12 blastocysts from IVF; D) Eight
blastocysts from ICSI and 16 blastocysts from IVF; and E) 24
blastocysts from IVF. All recipients became pregnant following ET
in all five groups and the delivery efficiency between groups did
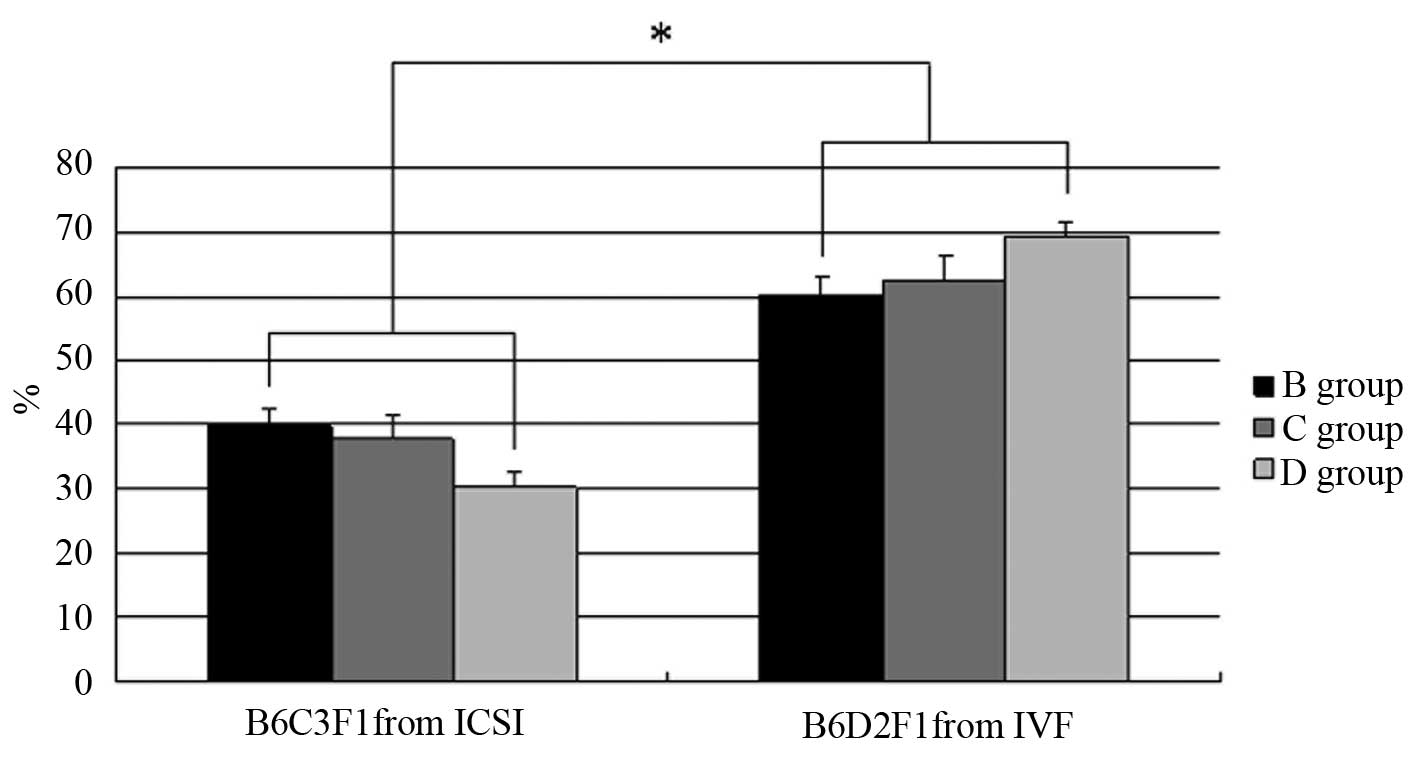
not differ. Based on an analysis of neonates from IVF or ICSI
blastocysts, mice with a black coat color were B6D2F1 from IVF
blastocysts and mice with an agouti coat color were B6C3F1 from
ICSI blastocysts (Table II). With
the exceptions of group A, in which ICSI blastocysts alone were
transferred, and group E, in which IVF blastocysts alone were
transferred, the birth rates of B6D2F1 and B6C3F1 mice were
compared. The results indicated that the delivery rate of ICSI
blastocysts was <40% (39.82, 37.61 and 30.32% in B, C and D
groups, respectively). By contrast, the IVF neonatal ratio of all
the neonates was >60% (60.18, 62.39 and 69.68% in B, C and D
groups, respectively), which was significantly higher than that of
the ICSI counterparts (P<0.01; Fig.
2).
 | Table IIThe pregnancy and delivery outcomes
of IVF and ICSI blastocyst using MET model. |
Table II
The pregnancy and delivery outcomes
of IVF and ICSI blastocyst using MET model.
| Group | No. of IVF
embryos | No. of ICSI
embryos | No. of
recipients | No. of transferred
embryos per recipient | Total no. of
transferred embryos | No. of pregnant
mice (%) | No. of neonates
(%) |
|---|
| A | 0 | 24 | 6 | 24 | 144 | 6 (100.0) | 70 (48.6) |
| B | 8 | 16 | 6 | 24 | 144 | 6 (100.0) | 59 (41.0) |
| C | 12 | 12 | 6 | 24 | 144 | 6 (100.0) | 74 (51.4) |
| D | 16 | 8 | 6 | 24 | 144 | 6 (100.0) | 63 (43.8) |
| E | 24 | 0 | 6 | 24 | 144 | 6 (100.0) | 71 (49.3) |
Cumulative pregnancy efficiency in SET
and MET models by mathematical equation
With the establishment of SET and MET models, the
cumulative pregnancy efficiency (CPE) was further analyzed using a
mathematical equation. If A was designated as pregnancy efficiency
in the SET model, the pregnancy probability is A when the mouse is
ET for the first time, A × (1-A) for the second time and A ×
(1-A)2 for the third time. When the mouse is ET for N
times, the probability is A × (1-A)n-1. Finally, the CPE
is the sum of the probability for each ET, and the equation is as
follows: ∑n=A+A × (1-A)+A × (1-A)2+A ×
(1-A)n-1=1-(1-A)n
The equation suggests that the CPE is closely
associated with the single pregnancy efficiency and with the ET
times (and also indicates the number of available ET embryos). When
the two values were higher, the cumulative pregnancy efficiency was
100%.
To validate the accuracy of the equation, in the
present study, the previous data were calculated using this
equation. In the ICSI group, the pregnancy rates in MET and SET I
and II were 100, 75 and 60%, respectively. Thus, if 24 embryos were
transferred four times, and six embryos were transferred each time,
the CPE was 99.6%. If the embryos were transferred 24 times (using
only one embryo each time), the CPE was also 99.9%. Similar results
were obtained when calculating the data in the IVF groups.
Discussion
One of the key concerns in the sibling oocyte
procedure is the embryo selection for ET, as it remains unknown
whether the embryo quality or the embryo fertilization method is
more important. Moreover, with the wide application of SET in
numerous ART centers, this issue is increasingly important in
sibling oocyte and other ART procedures.
Using the SET and MET models, the pregnancy and
delivery efficiencies on embryo selection in the sibling oocyte
procedure were calculated in the present study. The results
demonstrate that the pre-implantation development competence and
embryo quality were marginally affected by ICSI micromanipulation;
however, the ICSI or IVF blastocyst development competence to term
was similar in SET models I and II. Furthermore, the IVF, ICSI, 16
ICSI + 8 IVF, 12 ICSI + 12 IVF and 8 ICSI + 16 IVF groups
demonstrated similar pregnancy and delivery efficiencies in the MET
model, but in the ICSI/IVF combination groups, IVF blastocysts
exhibited greater implantation and delivery competence. Finally,
the mathematical equation indicated that the CPE was not any lower
in the SET models, and that it was closely associated with the
single pregnant efficiency and ET times.
It is well-known that ICSI has been mainly applied
to male factor infertility since the first baby was born using this
technology in 1992 (2). However,
certain studies have indicated potentially harmful effects of ICSI
on embryo development and the resulting fetus in human and mouse
models (21,22). In the present study, development
competence in mouse ICSI embryos was decreased from the beginning
of the 8-cell stage. This result was in agreement with previous
studies using mice (22), and
similar results have been found in humans (23). Cell number and apoptosis are
regarded to be better methods to identify blastocyst quality
compared with cell morphology (24). Cell apoptosis is regarded as a
cellular activity in which the embryos eliminate abnormal cells and
prepare for implantation; however, certain studies have indicated
that the apoptosis cell number in blastocysts is affected by
culture conditions (25–27). In the present study, the ICSI and
IVF embryos were cultured in the same culture medium, suggesting
that the abnormal cell number was increased by ICSI technology.
To evaluate the post-implantation development
competence of ICSI and IVF embryos in sibling oocyte procedures,
and to simulate the clinical setting, the SET model I was
established, which involves only one embryo, using IVF or ICSI in
recipients. However, mice are mammals with multiple gestations,
which is different from humans, a generally monotocous mammal.
Thus, the SET model II was established, which involves six embryos,
with IVF or ICSI transfer to recipients. Normally the litter size
of ICR mice is 8–12, thus transferring six embryos is reasonable
for the SET model. In a mouse study, the ET number is often >12
to ensure pregnancy, but the transferred embryos are usually at the
2-cell stage (22,28). In the current study, the embryos
were selected at the blastocyst stage as the development competence
of ICSI embryo beyond two cells was decreased compared with the IVF
groups. To eliminate potential differences from pre-implantation,
the blastocysts were selected in the current study. In the human
clinical setting, different clinical outcomes were reported in
different centers. Bhattacharya et al (29) summarized and analyzed the data from
four UK centers and indicated decreased pregnancy and delivery
outcomes when transferring ICSI embryos compared with IVF embryos.
However, La Sala et al (30) reported inconsistent results and
suggested that ICSI embryos have similar outcomes with IVF embryos
following ET in a retrospective analysis. Hourvitz et al
(31) also obtained similar
results to La Sala et al (30) by analyzing a database from July
2000 and October 2004. The incompatible results may be attributed
to multiple factors, including differences between patients,
centers, regions and nationalities.
To simulate DET or TET, MET models were established
in which 24 blastocysts were transferred to the recipients.
Furthermore, to simulate the mixture of ICSI and IVF transferred
embryos in the clinical setting, three mixture MET models were
established following the possible mixture ratio between IVF and
ICSI (1:2, 1:1 and 2:1) in the two groups. The pregnancy rate in
each group reached 100% and the delivery rate between groups was
also similar. In a human study, Pandian et al (32) also reported that the likelihood of
a live birth in a fresh IVF cycle in SET was significantly
decreased; however, certain studies have revealed that there are no
significant differences between SET and MET (33,34).
Notably, the IVF embryos in the mixture groups exhibited greater
implantation and delivery ability than the ICSI counterpart,
independent of the number of IVF embryos (10, 15 or 20 in the three
mixture groups), but the ICSI and IVF embryos exhibited similar
implantation ability when only one type of embryo was transferred.
In the human clinical setting, Shveiky et al (11) reported that the pregnancy and
implantation rates in ICSI and IVF embryos did not differ for
sibling oocytes. Moreover, Staessen et al (10) concluded that implantation and
delivery outcomes were not different when using ICSI, IVF or a
mixture of embryos for ET in sibling oocyte procedures. These
results were consistent with the findings of the SET and MET models
in the present study.
In humans it is almost impossible to distinguish
neonates developed from IVF or ICSI embryos, but this issue is
easier to resolve in the mouse models using different coat colors
for mouse neonates from IVF or ICSI embryos. The present study
indicated that IVF embryos exhibited more robust competitiveness in
the process of implantation compared with ICSI counterparts in the
mixture groups, which could reflect embryo quality. Montag et
al (35) reported that
successful hatching in vitro is dependent on a sufficiently
high number of embryonic cells, which enables the blastocyst
expansion and zona shedding that is required prior to implantation.
Desai et al (36) suggested
that the cell number is one of the key parameters to predict
clinical outcome in ART. These studies indicate that embryos with a
greater cell number have more opportunities to implant into the
uterus. In the present study, IVF embryos had a greater cell number
in ICM and lesser apoptosis cell number compared with ICSI embryos,
which may result in higher competitiveness when combined for ET. In
biological evolution, acceptance of superior embryos by the uterus
is a possible protective mechanism in the process of
reproduction.
In the present study, the pregnancy efficiency was
lower in the SET models compared with the MET model; however, the
two models exhibited similar CPEs. SET has the advantage of
decreasing the multiple pregnancies in ART, and certain clinical
studies have shown similar CPE results to those of the current
study (37,38). Despite these results, how to select
a ‘healthy’ embryo remains unknown, particularly for sibling oocyte
procedures, although a number of studies have focused on screening
particular molecular markers in cumulus cells (39,40).
Based on the established ET models, a mathematical equation for
mouse ET was developed. Predicting pregnancy efficacy in ART has
been performed using Kaplan-Meier curves, but rarely by equation
(41–44). Lannon et al (45) previously developed an equation in
humans using multiple factors to predict pregnancy potential. In
the current study, certain extra factors, including
freezing/thawing, individual differences, disease factors and the
effects of repeated ET on physiology and psychology, were removed
from consideration as mouse models were used. The core factors, SET
pregnancy rate and ET times (ET times were equal to the available
ET number if the SET was applied), were revealed to affect the CPE
using the equation developed in the present study. Thus, increasing
uterine receptivity by improving ET technology and adjusting female
pre-conception physiology (e.g., endometrium blood flow and
thickness, and hormone levels), as well as increasing embryo
development competence and quality by modifying the culture system,
is required prior to improving the CPE.
In conclusion, the present study has provided a
criterion for selecting embryos in ART, particularly for sibling
oocyte procedures using SET and MET models. Embryo quality was the
primary factor while selecting embryos. IVF embryos are more
competitive than ICSI embryos in the process of implantation in MET
mixture groups, but the fertilization methods did not affect the
overall pregnancy and delivery rates. Finally, a mathematical
equation demonstrated that the SET pregnancy rate and ET times were
key factors with regard to CPE. The present study provides methods
to select embryos prior to ET and may aid in promoting the SET
application in sibling oocyte procedures.
References
|
1
|
Steptoe PC and Edwards RG: Birth after the
reimplantation of a human embryo. Lancet. 2:3661978. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Palermo G, Joris H, Devroey P and Van
Steirteghem AC: Pregnancies after intracytoplasmic injection of
single spermatozoon into an oocyte. Lancet. 340:17–18. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Handyside AH, Kontogianni EH, Hardy K and
Winston RM: Pregnancies from biopsied human preimplantation embryos
sexed by Y-specific DNA amplification. Nature. 344:768–770. 1990.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Calderón G, Belil I, Aran B, et al:
Intracytoplasmic sperm injection versus conventional in-vitro
fertilization: first results. Hum Reprod. 10:2835–2839.
1995.PubMed/NCBI
|
|
5
|
Li Z, Lin H, Xiao W and Wang Y:
Fertilization of IVF/ICSI using sibling oocytes from couples with
subfertile male or unexplained infertility. J Huazhong Univ Sci
Technolog Med Sci. 24:365–368. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Elizur SE, Levron J, Seidman DS, et al:
Conventional in vitro fertilization versus intracytoplasmic
sperm injection for sibling oocytes in couples with mild
oligoteratoasthenozoospermia and couples with normal sperm. Fertil
Steril. 82:241–243. 2004.
|
|
7
|
van der Westerlaken L, Naaktgeboren N,
Verburg H, et al: Conventional in vitro fertilization versus
intracytoplasmic sperm injection in patients with borderline semen:
a randomized study using sibling oocytes. Fertil Steril.
85:395–400. 2006.
|
|
8
|
Fan W, Li SW, Li L, et al: Outcome of
conventional IVF and ICSI on sibling oocytes in the case of
isolated teratozoospermia. J Assist Reprod Genet. 29:905–910. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hwang JL, Seow KM, Lin YH, et al: IVF
versus ICSI in sibling oocytes from patients with polycystic
ovarian syndrome: a randomized controlled trial. Hum Reprod.
20:1261–1265. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Staessen C, Camus M, Clasen K, et al:
Conventional in-vitro fertilization versus intracytoplasmic sperm
injection in sibling oocytes from couples with tubal infertility
and normozoospermic semen. Hum Reprod. 14:2474–2479. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Shveiky D, Simon A, Gino H, et al: Sibling
oocyte submission to IVF and ICSI in unexplained infertility
patients: a potential assay for gamete quality. Reprod Biomed
Online. 12:371–374. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Plachot M, Belaisch-Allart J, Mayenga JM,
et al: Outcome of conventional IVF and ICSI on sibling oocytes in
mild male factor infertility. Hum Reprod. 17:362–369. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Becker B, Bertrand E, Van Hoeck J, et al:
Outcome of conventional IVF and ICSI on sibling oocytes in patients
suffering from teratospermia. Int J Fertil Womens Med. 51:163–169.
2006.PubMed/NCBI
|
|
14
|
Kihaile PE, Misumi J, Hirotsuru K, et al:
Comparison of sibling oocyte outcomes after intracytoplasmic sperm
injection and in vitro fertilization in severe
teratozoospermic patients in the first cycle. Int J Androl.
26:57–62. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Marsh CA, Farr SL, Chang J, et al: Trends
and factors associated with the Day 5 embryo transfer, assisted
reproductive technology surveillance, USA, 2001–2009. Hum Reprod.
27:2325–2331. 2012.PubMed/NCBI
|
|
16
|
Thurin A, Hausken J, Hillensjö T, et al:
Elective single-embryo transfer versus double-embryo transfer in
in vitro fertilization. N Engl J Med. 351:2392–2402. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Gardner DK, Surrey E, Minjarez D, et al:
Single blastocyst transfer: a prospective randomized trial. Fertil
Steril. 81:551–555. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Uyar A, Torrealday S and Seli E: Cumulus
and granulosa cell markers of oocyte and embryo quality. Fertil
Steril. 99:979–997. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Fragouli E and Wells D: Aneuploidy
screening for embryo selection. Semin Reprod Med. 30:289–301. 2012.
View Article : Google Scholar
|
|
20
|
Yu Y, Ding C, Wang E, et al:
Piezo-assisted nuclear transfer affects cloning efficiency and may
cause apoptosis. Reproduction. 133:947–954. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Feng C, Wang LQ, Dong MY and Huang HF:
Assisted reproductive technology may increase clinical mutation
detection in male offspring. Fertil Steril. 90:92–96. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yu Y, Zhao C, Lv Z, et al: Microinjection
manipulation resulted in the increased apoptosis of spermatocytes
in testes from intracytoplasmic sperm injection (ICSI) derived
mice. PLoS One. 6:e221722011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Griffiths TA, Murdoch AP and Herbert M:
Embryonic development in vitro is compromised by the ICSI
procedure. Hum Reprod. 15:1592–1596. 2000.
|
|
24
|
Van Soom A, Vanroose G and de Kruif A:
Blastocyst evaluation by means of differential staining: a
practical approach. Reprod Domest Anim. 36:29–35. 2001.PubMed/NCBI
|
|
25
|
Lane M and Gardner DK: Increase in
postimplantation development of cultured mouse embryos by amino
acids and induction of fetal retardation and exencephaly by
ammonium ions. J Reprod Fertil. 102:305–312. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Cao YJ, Fan XJ, Shen Z, et al: Nitric
oxide affects preimplantation embryonic development in a rotating
wall vessel bioreactor simulating microgravity. Cell Biol Int.
31:24–29. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Xie Y, Puscheck EE and Rappolee DA:
Effects of SAPK/JNK inhibitors on preimplantation mouse embryo
development are influenced greatly by the amount of stress induced
by the media. Mol Hum Reprod. 12:217–224. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kimura Y and Yanagimachi R:
Intracytoplasmic sperm injection in the mouse. Biol Reprod.
52:709–720. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Bhattacharya S, Hamilton MP, Shaaban M, et
al: Conventional in-vitro fertilisation versus intracytoplasmic
sperm injection for the treatment of non-male-factor infertility: a
randomised controlled trial. Lancet. 357:2075–2079. 2001.
View Article : Google Scholar
|
|
30
|
La Sala GB, Nucera G, Gallinelli A, et al:
Spontaneous embryonic loss after in vitro fertilization with
and without intracytoplasmic sperm injection. Fertil Steril.
82:1536–1539. 2004.
|
|
31
|
Hourvitz A, Lerner-Geva L, Elizur SE, et
al: Role of embryo quality in predicting early pregnancy loss
following assisted reproductive technology. Reprod Biomed Online.
13:504–509. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Pandian Z, Templeton A, Serour G and
Bhattacharya S: Number of embryos for transfer after IVF and ICSI:
a Cochrane review. Hum Reprod. 20:2681–2687. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Gerris J: Single-embryo transfer versus
multiple-embryo transfer. Reprod Biomed Online. 18(Suppl 2): 63–70.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Veleva Z, Vilska S, Hydén-Granskog C, et
al: Elective single embryo transfer in women aged 36–39 years. Hum
Reprod. 21:2098–2102. 2006.
|
|
35
|
Montag M, Koll B, Holmes P and van der
Ven: Significance of the number of embryonic cells and the state of
the zona pellucida for hatching of mouse blastocysts in
vitro versus in vivo. Biol Reprod. 62:1738–1744. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Desai NN, Goldstein J, Rowland DY and
Goldfarb JM: Morphological evaluation of human embryos and
derivation of an embryo quality scoring system specific for day 3
embryos: a preliminary study. Hum Reprod. 15:2190–2196. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Wang YA, Sullivan EA, Healy DL and Black
DA: Perinatal outcomes after assisted reproductive technology
treatment in Australia and New Zealand: single versus double embryo
transfer. Med J Aust. 190:234–237. 2009.PubMed/NCBI
|
|
38
|
Rodríguez Barredo DB, Tur Padro R, Mancini
F, Parriego García M, Rodríguez García I, Coroleu Lletget B and
Barri Rague PN: Elective single embryo transfer and cumulative
pregnancy rate: five-year experience in a Southern European
Country. Gynecol Endocrinol. 28:425–428. 2012.PubMed/NCBI
|
|
39
|
Gebhardt KM, Feil DK, Dunning KR, et al:
Human cumulus cell gene expression as a biomarker of pregnancy
outcome after single embryo transfer. Fertil Steril. 96:47–52.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
McKenzie LJ, Pangas SA, Carson SA, et al:
Human cumulus granulosa cell gene expression: a predictor of
fertilization and embryo selection in women undergoing IVF. Hum
Reprod. 19:2869–2874. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Garrido N, Bellver J, Remohí J, et al:
Cumulative newborn rates increase with the total number of
transferred embryos according to an analysis of 15,792 ovum
donation cycles. Fertil Steril. 98:341–346. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Garrido N, Bellver J, Remohí J, et al:
Cumulative live-birth rates per total number of embryos needed to
reach newborn in consecutive in vitro fertilization (IVF)
cycles: a new approach to measuring the likelihood of IVF success.
Fertil Steril. 96:40–46. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Luke B, Brown MB, Wantman E, et al:
Cumulative birth rates with linked assisted reproductive technology
cycles. N Engl J Med. 366:2483–2491. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Malizia BA, Hacker MR and Penzias AS:
Cumulative live-birth rates after in vitro fertilization. N
Engl J Med. 360:236–243. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Lannon BM, Choi B, Hacker MR, et al:
Predicting personalized multiple birth risks after in vitro
fertilization-double embryo transfer. Fertil Steril. 98:69–76.
2012. View Article : Google Scholar : PubMed/NCBI
|