Introduction
Cervical carcinoma is the second most common type of
malignant cancer and the fourth leading cause of cancer-associated
mortality in females worldwide (1). Currently, surgery, chemoradiotherapy,
HPV vaccines and associated biological therapy are the main
modalities for the treatment of cervical cancer; however, they all
have limitations. The HPV vaccines are only effective for HPV types
16 and 18, however, there are other high-risk subtypes that are
also able to cause cervical cancer (2). Patients with cervical carcinoma
undergoing surgery and chemoradiotherapy have a survival advantage
(3), however, data suggested that
the recurrence incidence of cervical cancer diagnosed in females
accounts for ~35%, and 90% were found within three years after the
initial management (4). It was
demonstrated that certain cervical cancer cells were not eradicated
by current therapeutics.
The cancer stem cell (CSC) theory provided novel
insights into the recurrent formation of tumors following surgery
or chemoradiotherapy in cancer patients. CSCs possess certain
properties, including a capacity for self-renewal, chemoresistance,
the ability to differentiate into mature, specialized cancer cell
types as well as a high tumorigenic potential that may correlate
with the initiation, progression and recurrence of cancer (5). CSCs have been reported in multiple
types of solid tumor and in cultured cancer cell lines, including
brain (6), breast (7), colon (8) and prostate (9), as well as cervical cancer cell lines
(10). Almost all the cancer
stem-like cells have been isolated and cultured in serum-free
medium supplemented with adequate mitogens, including basic
fibroblast growth factor and epidermal growth factor, and incubated
for 2–6 weeks, which is costly, time-consuming and ineffective
(11–13). Recently, a novel method for
overcoming these drawbacks and limitations, termed the nonadhesive
culture system, was used to successfully isolate and enrich CSCs
from established human oral squamous cell carcinoma (OSCC) cell
lines (14). To establish a
reliable in vitro model of CSCs of cervical cancer for basic
and preclinical studies, the present study was designed to enrich
and identify stem-like cells from human cervical cancer cells
(HeLa), and to further characterize their CSC properties.
Materials and methods
Cell line and culture
The human cervical cancer cell line, HeLa was
obtained from the Shanghai Cell Biology Institute of the Chinese
Academy of Sciences (Shanghai, China). The parental adherent
monolayer HeLa cells were maintained in Dulbecco’s modified Eagle’s
medium (DMEM) supplemented with 10% fetal bovine serum (FBS),
penicillin (100 U/ml) and streptomycin (100 μg/ml) in a humidified
atmosphere of 50 μg/ml CO2 at 37°C.
Tumor sphere culture
The tumor spheres of HeLa cells were cultured using
the nonadhesive culture system described by Chen et al
(14) with minor modifications.
Briefly, the parental adherent monolayer HeLa cells were collected
and plated in 100-mm dishes coated with agarose at a density of
5×104 cells, and the culture medium was altered every
other day until tumor spheres were formed.
Colony formation assay
The colony forming ability of the parental adherent
monolayer and tumor sphere HeLa cells were assayed by replating
them in 6-well plates (200 cells/well). Following 12 days of
incubation, the cells were stained with 0.5% crystal violet in
absolute ethanol, and colonies with >50 cells were counted under
a dissection microscope [Olympus (China) Co., Ltd., Beijing,
China]. Three independent experiments were performed.
Tumor sphere formation and self-renewal
assay
The tumor spheres were collected by gentle
centrifugation, disaggregated with Accutase (Sigma-Aldrich, St.
Louis, MO, USA) to generate single cells and passaged every 5–7
days when the spheres reached a diameter of 100 μm. To evaluate
tumor sphere forming efficiency, single tumor sphere cells derived
from the parental or tumor spheres were plated into 96 wells at
varying densities; the lowest density was one cell per well.
Following 12 days of culture, the sphere number of each well was
counted. Sphere forming efficiency was calculated as the sphere
number divided by the initial single cell number plated and
expressed as a percentage (15).
In addition, the wells with only one cell were monitored. The
spheres derived from single cells were marked and images of the
spheres were captured every day.
Toluidine blue staining
To evaluate the light cell (LC) and dark cell (DC)
populations in the parental adherent monolayer and tumor sphere
HeLa cells, the two cell suspensions were stained with toluidine
blue staining buffer containing 10 mM HEPES buffer (pH 7.4), 2 mM
EDTA, 0.5% bovine serum albumin (BSA) and 0.4% toluidine blue
(Sigma-Aldrich) for 5 min at room temperature (RT) (7). Images of the cells were captured with
a photocamera-equipped light microscope [Olympus (China) Co.,
Ltd.]. An average of six fields/sample was analyzed and three
independent experiments were performed.
Chemotherapy sensitivity and resistance
assays
The chemoresistance of the parental adherent
monolayer and tumor sphere HeLa cells was assessed using a modified
MTT assay (16). Briefly,
2×103 cells per well were seeded in 96-well plates in
100 μl culture medium (three wells per group). Following 24 h, the
cells were treated with various concentrations of cisplatin and
epirubicin, respectively, for 72 h. Subsequently, 10 μl MTT
solution was added to each well and the plate was incubated in the
dark for an additional 4 h at 37°C. The cells were then lysed in a
buffer containing 10% sodium dodecyl sulfate in 0.01 M HCl. The
absorbance at 570 nm was measured using a microplate reader
(Bio-Rad, Richmond, CA, USA), using wells without cells as blanks.
The effects of cisplatin and epirubicin on the viabilities of
adherent monolayer and tumor sphere HeLa cells were expressed as
the %cytoviability using the following formula: %cytoviability =
A570 of treated cells/A570 of control cells ×
100% (17). Three independent
experiments were performed.
Invasion assay
The invasion assay was performed using 24-transwell
chambers (Costar, Bodenheim, Germany). Briefly, the parental
adherent monolayer and tumor sphere HeLa cells were resuspended in
serum-free DMEM at a concentration of 4×105 cells/ml.
The upper chamber was loaded with 100 μl cell suspension and the
lower chamber was loaded with 500 μl DMEM with 15% FBS. Following
culture for 48 h, the cells in the upper chamber were removed using
a cotton swab and the lower chamber filter was fixed with 4%
paraformaldehyde and stained with crystal violet. The number of
cells that migrated to the undersurface of the membrane was counted
and six randomly selected fields were analyzed. Three independent
experiments were performed.
Western blotting analysis of Oct4 and
Sox2 protein expression
The proteins of the parental adherent and tumor
sphere cells were prepared with RIPA lysis buffer (Beyotime
Institute of Biotechnology, Shanghai, China), separated by 12%
SDS-PAGE, transferred onto PVDF membranes using a semi-dry blotting
apparatus (Bio-Rad, Hercules, CA, USA), and blocked in 5% non-fat
milk. The membranes were subsequently incubated with the
corresponding primary antibodies, as indicated: a rabbit
anti-β-actin (Beijing Biosynthesis Biotechnology Co., Ltd.,
Beijing, China) diluted 1:300, rabbit anti-Oct4 and rabbit
anti-Sox2 (BioLegend, San Diego, CA) diluted 1:1,000. Antibody
recognition was detected with peroxidase-conjugated goat
anti-rabbit IgG (H+L) secondary antibody (Zhongshan Goldenbridge
Biotechnology Co., Ltd, Beijing, China) used at 1:3,000 dilutions.
Antibody-bound proteins were detected with a BeyoECL Plus kit
(Beyotime Institute of Biotechnology) and western blotting analysis
system (Universal Hood II, Bio-Rad, USA ).
Detection of aldehyde dehydrogenase 1
(ALDH1) and SOX2 by immunofluorescence staining
The parental adherent monolayer and tumor sphere
HeLa cells were fixed with 4% paraformaldehyde for 10 min,
permeabilized with 0.01% Triton X-100 and inhibited with 5% BSA in
phosphate-buffered saline (PBS). The cells were then incubated with
rabbit anti-ALDHA1-fluorescein isothiocyanate (FITC) polyclonal
antibody (Beijing Biosynthesis Biotechnology Co., Ltd., Beijing,
China) and rabbit anti-SOX2-FITC (Epitomics, Inc., Burlingame, CA,
USA) in 1% BSA and PBS with Tween-20 in a humidified chamber for 1
h at RT, respectively. DAPI (Sigma-Aldrich) was used for nuclear
counterstaining. Images were captured using a Leica DMI400B
inverted fluorescence microscope linked to a DFC340FX camera (Leica
Microsystems GmbH, Wetzlar, Germany). Three independent experiments
were performed.
Adipogenic differentiation assay
For the adipogenic differentiation assay, tumor
sphere HeLa cells were seeded in 6-well plates, cultured with DMEM
containing 5% FBS, supplemented with 10 μM insulin, 1 μM
dexamethasone, 200 μM indomethacin and 3-isobutyl-1-methylxanthine
(Sigma-Aldrich). The culture medium was altered twice a week and
the appearance of lipid droplets was monitored every day. Following
incubation for 15 days, the medium was aspirated and the cells were
washed with PBS and fixed with 4% paraformaldehyde in PBS for 30
min. Then, the cells were incubated with Oil Red O dye
(Sigma-Aldrich) at RT for 30 min. The dye was carefully removed,
washed with PBS and counterstained with hematoxylin. Images were
captured using a Leica DMI400B inverted fluorescence microscope
linked to a DFC340FX camera (Leica Microsystems GmbH).
Statistical analysis
The data are expressed as the mean ± standard
deviation. All data were analyzed using the software SAS V9.1 (SAS
Institute Inc., Cary, NC, USA). Student’s t-test was used to
analyze the statistical difference. P<0.05 was considered to
indicate a statistically significant difference.
Results
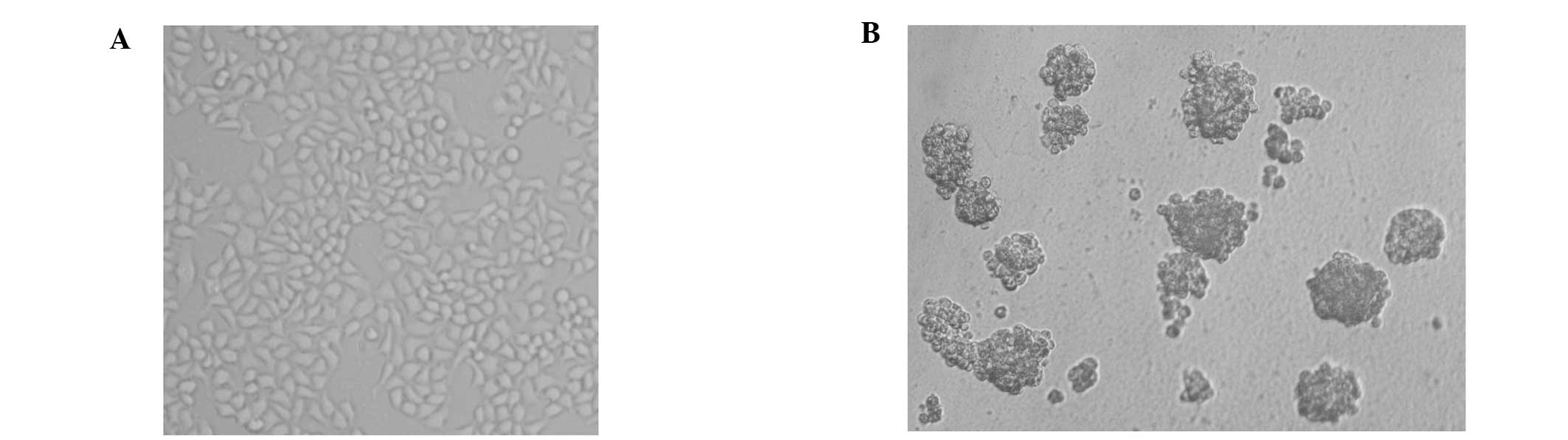
Morphological characteristics
The parental HeLa cells cultured with DMEM
supplemented with 10% FBS grew as an adherent monolayer (Fig. 1A), while HeLa cells cultured under
the nonadhesive culture system formed typical tumor spheres
(Fig. 1B).
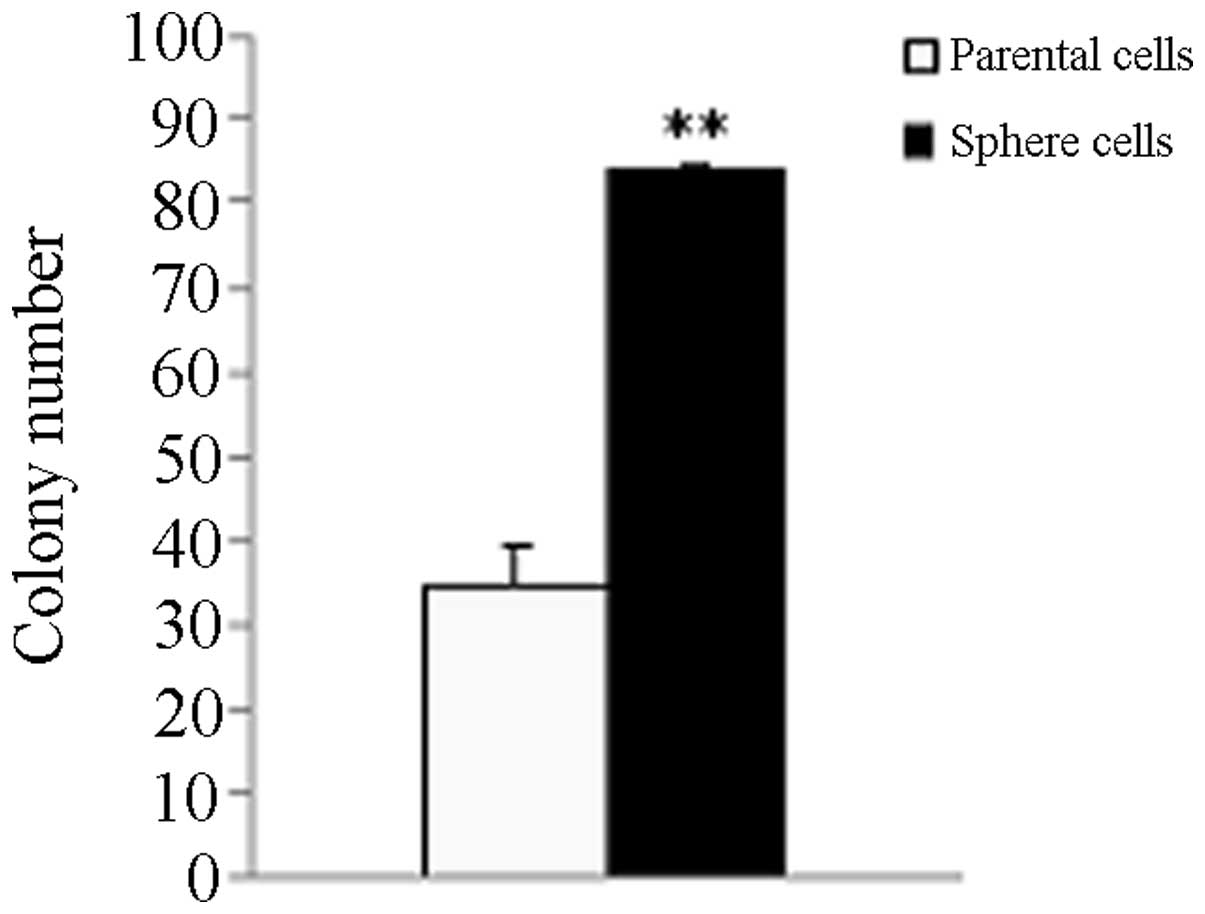
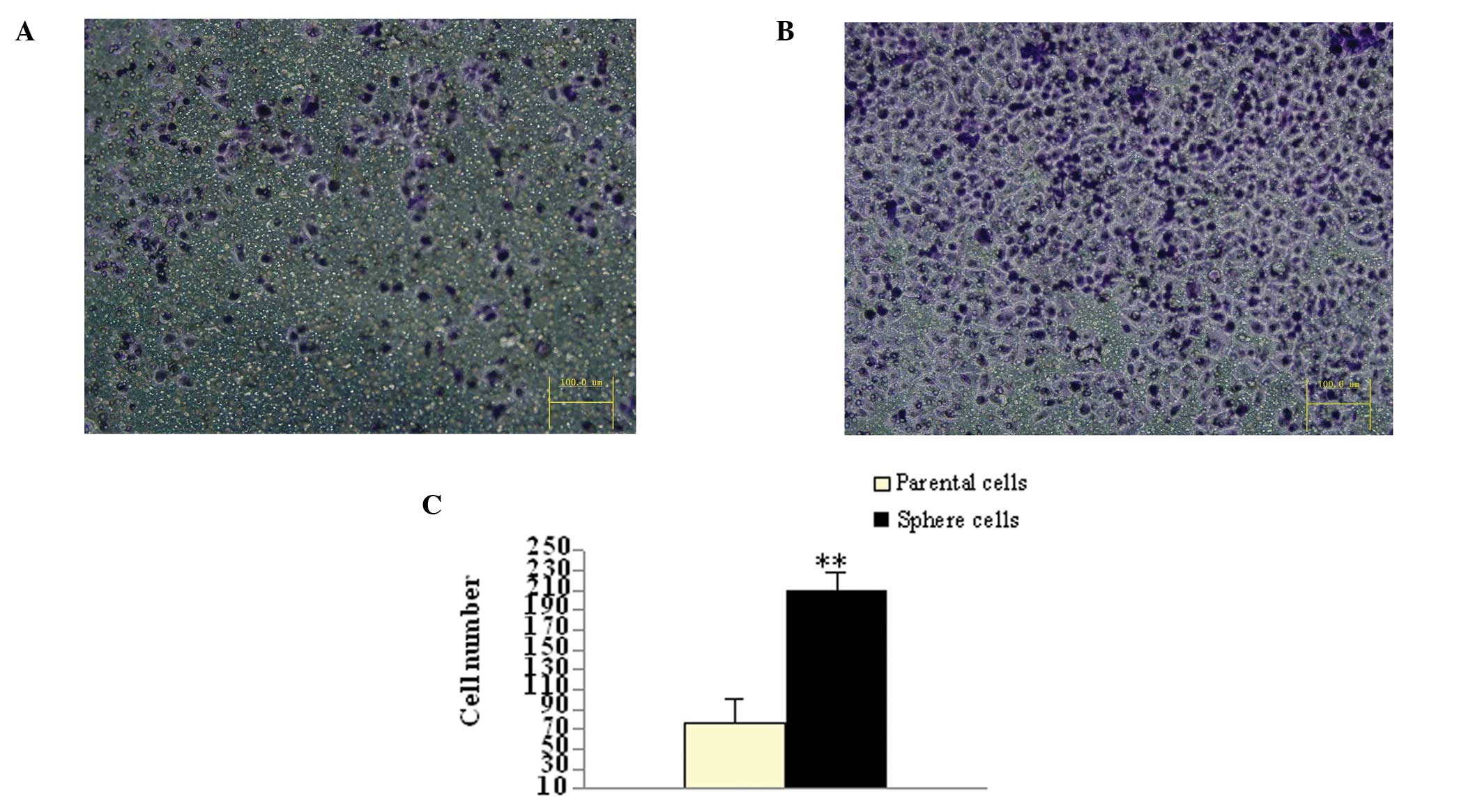
Tumor sphere cells exhibit a higher
colony forming efficiency compared with parental adherent monolayer
cells
The parental HeLa cells generated 34.67±4.51
colonies and the tumor sphere cells generated 83.67±8.50. The tumor
sphere cells exhibited a higher colony forming efficiency compared
with the parent adherent monolayer cells (P<0.01; Fig. 2).
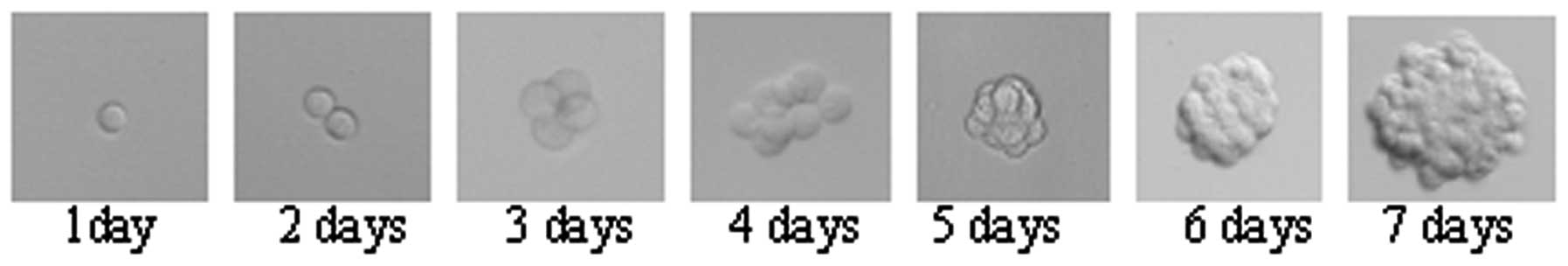
Tumor sphere cells exhibit a high
self-renewal potential
The sphere formation assay has been universally used
to evaluate the property of progenitors or stem cells. The parental
HeLa cells grew as an adherent monolayer in DMEM containing 10% FBS
(Fig. 1A). When plated in an
agarose coated nonadhesive culture system, they grew as floating,
three-dimensional tumor spheres and reached 100 μm in diameter
following 7 days (Fig. 1B). Tumor
spheres were passaged and plated into 96 wells at varying
densities; the lowest density was one cell per well. Following 12
days of culture, the sphere formation efficiency of tumor sphere
HeLa cells was 40.79±1.8% (data not shown). Tumor sphere formation
from single cells was observed (Fig.
3).
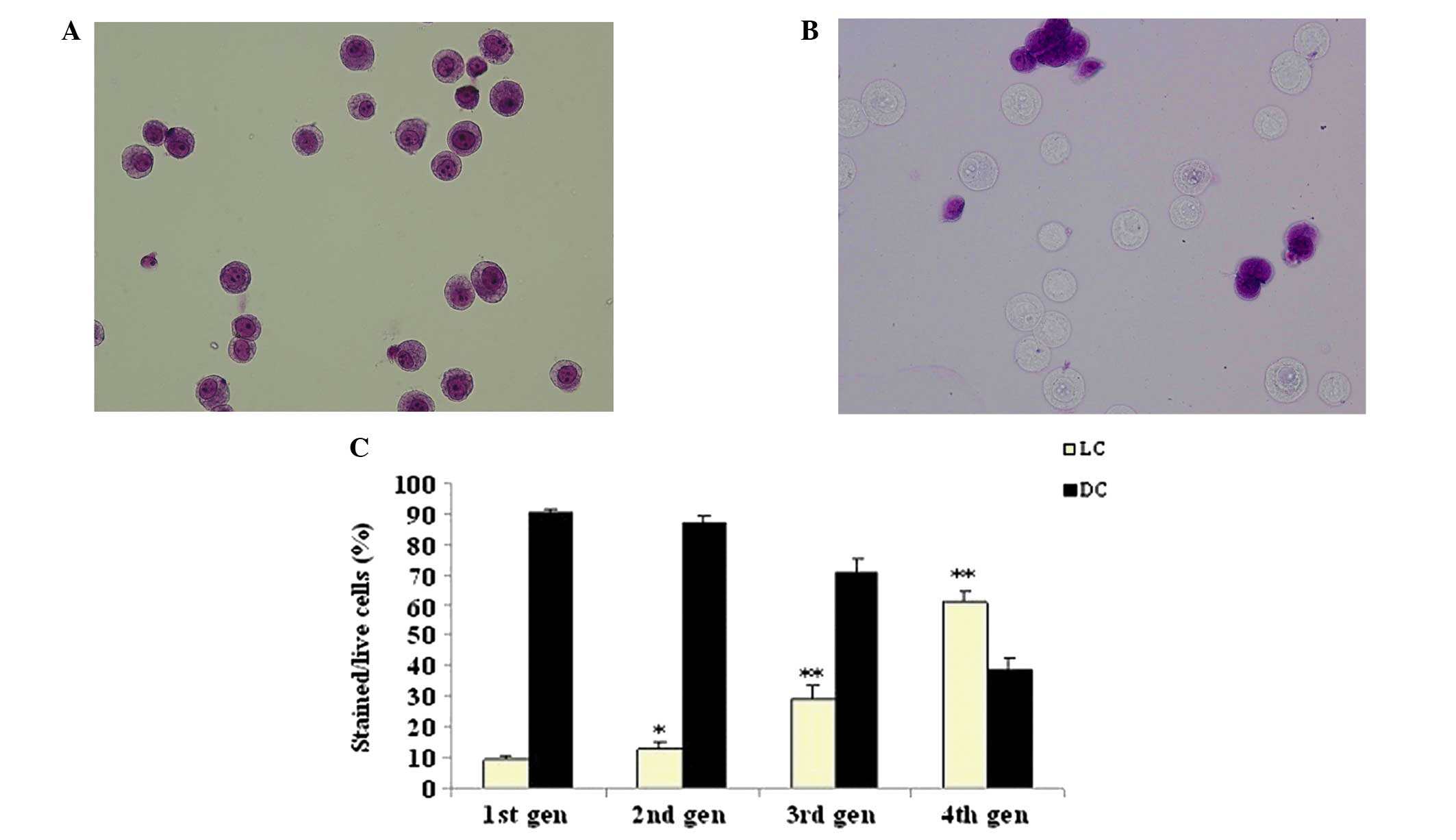
Toluidine blue pale LC and DC populations
in the parental adherent monolayer and tumor sphere HeLa cells
The parental adherent monolayer and tumor sphere
HeLa cells contained distinct LC and DC populations (Fig. 4A and B). The number of LCs in tumor
sphere HeLa cells (60.94%) was higher than those in the parental
adherent monolayer HeLa cells (2.2%; Fig. 4B; P<0.01). LC populations in the
tumor sphere increased gradually between 9.48+0.9 and 60.94+3.2%
(Fig. 4C) following four
passages.
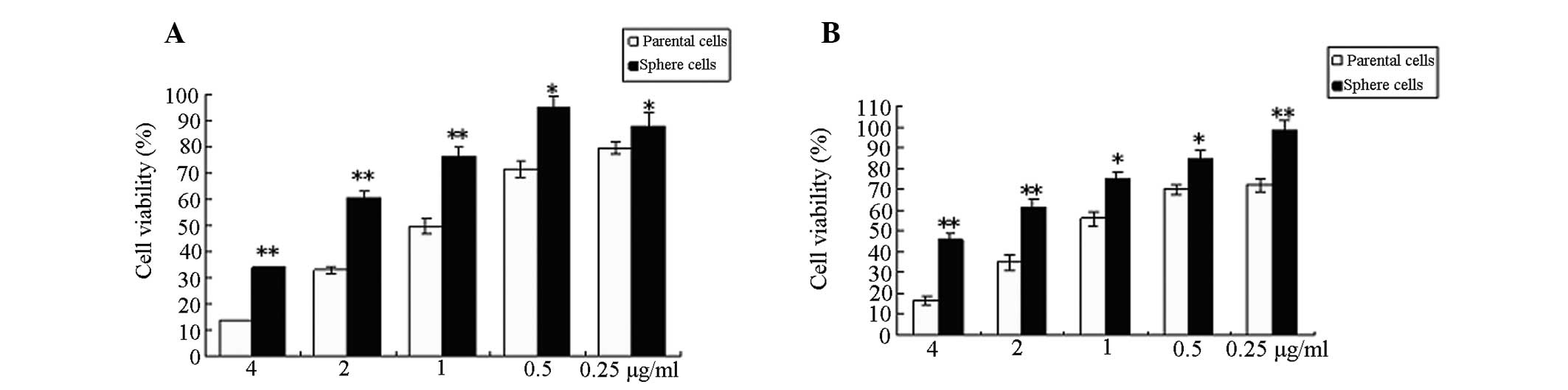
Tumor sphere HeLa cells are resistant to
chemotherapy compared with parent adherent monolayer HeLa
cells
To assess the chemoresistance of the parental
adherent monolayer and tumor sphere HeLa cells, the two cell
populations were treated with cisplatin and epirubicin,
respectively for 48 h. The viability of tumor sphere HeLa cells was
higher than adherent monolayer HeLa cells at the same concentration
of the drug (P<0.05 or P<0.01; Fig. 5A and B).
Tumor sphere cells exhibit a high
invasive capacity
The invasive capacity of the parental adherent
monolayer and tumor sphere HeLa cells were determined using a
transwell invasion assay. Following incubation for 48 h, the number
of cells that penetrated the transwell membrane (208±18/field;
magnification, ×200) of tumor spheres was higher than the parental
adherent monolayer HeLa cells (75±24/field; magnification, ×200;
P<0.01; Fig. 6).
Protein expression of Oct4 and SOX2
increases in tumor sphere HeLa cells compared with the parental
adherent monolayer
Western blotting demonstrated that there was little
expression of Oct4 and SOX2 in the parental adherent monolayer HeLa
cells, while their expression was evident in tumor sphere HeLa
cells cultured under a nonadhesive culture system (Fig. 7).
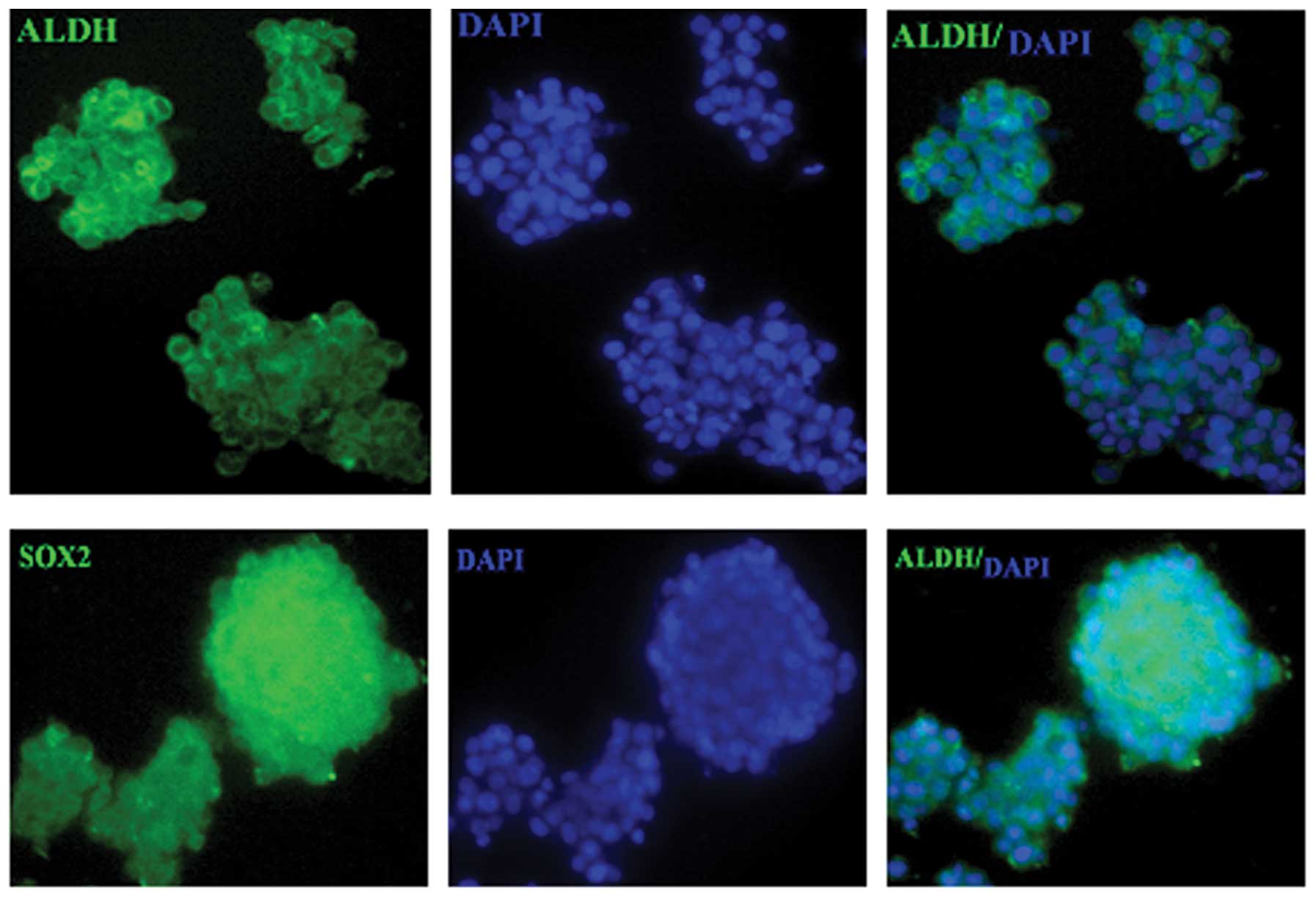
Tumor sphere HeLa cells express high
levels of the putative stem cell markers, ALDH1 and SOX2
To detect the expression of putative CSC markers in
tumor sphere HeLa cells, the parental adherent monolayer and tumor
sphere HeLa cells were examined for ALDH1 and SOX2 protein
expression. The parental adherent monolayer HeLa cells scarcely
expressed ALDH1 and SOX2, while stable ALDH1 and SOX2 expression
was detected in tumor sphere HeLa cells (Fig. 8).
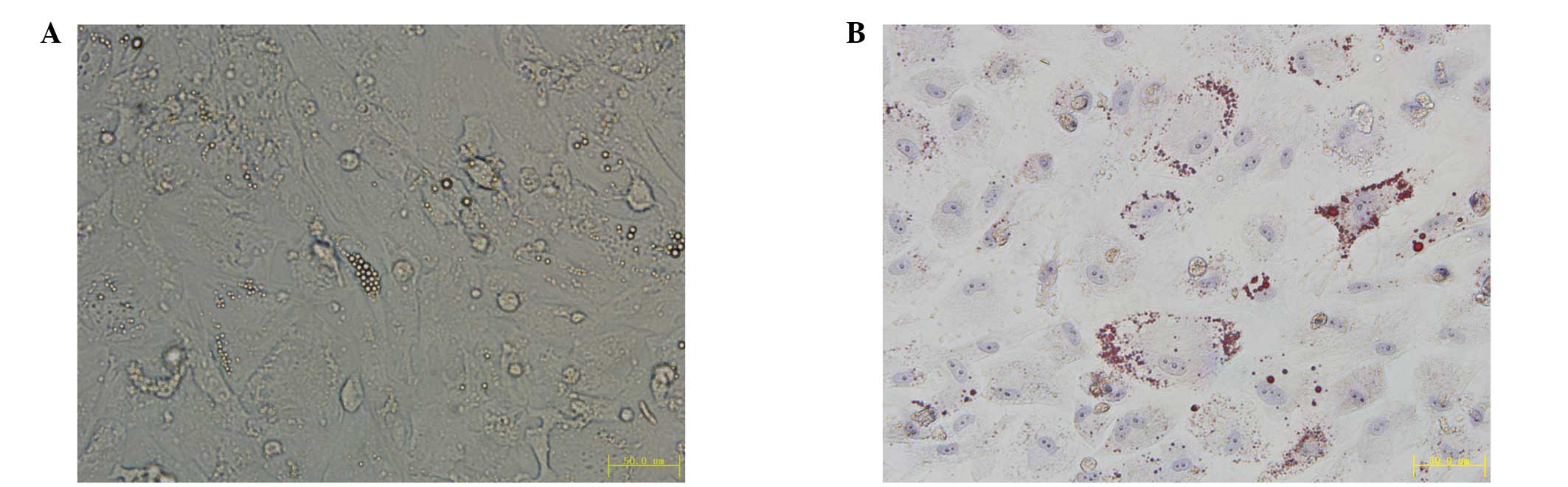
Tumor sphere HeLa cells are induced to
differentiate into adipocytes
To determine the multipotent differentiation
potential of the tumor sphere cells, the tumor sphere HeLa cells
were cultured in adipogenic differentiation media for 15 days and
small lipid droplets were observed in the cytoplasm of cancer
cells. The lipid droplets increased in size with time (Fig. 9A) and were confirmed by Oil Red O
staining (Fig. 9B).
Discussion
Cervical carcinoma, a prevalent disease, is
considered to be the second most common type of malignant cancer
and the fourth leading cause of cancer-associated mortality in
females worldwide (1). Cervical
cancer has a high recurrence rate and high risk of metastasis
following conventional therapy, leading to a high mortality rate
(2–4). Previous studies indicated that a
small population of CSCs appear to be responsible for tumor
initiation and progression and also for the resistance to
conventional treatment (18,19).
Recently, isolation, identification and selective eradication of
CSCs using targeted drugs have become a major focus in basic and
clinical cancer studies (20,21).
Therefore, for investigating and developing agents targeting CSCs
for cancer therapeutics, a reliable model of CSCs is crucial for
basic and preclinical studies. However, there are currently no
universal markers for the isolation and identification of CSCs in
any type of cancer (14). Cell
lines cultured with defined serum-free culture conditions are a
commonly used method for enriching CSCs from mixed populations and
has been particularly important in establishing in vitro
models for CSC expansion (22).
However, it is costly, time-consuming and ineffective (11–13).
Using the nonadhesive culture method to enrich CSCs
from the human OSCC cell line was cost-effective and simple
(14).
In the present study, human cervical cancer
stem-like cells were enriched and expanded using a nonadhesive
culture system and the majority of cells formed typical tumor
spheres (Fig. 1B). The colony and
tumor sphere formation efficiency of cancer stem-like cells from
tumor spheres was higher than the parental adherent monolayer HeLa
cells (Fig. 2; P<0.01). Single
cells derived from tumor spheres were able to generate the second
tumor spheres (Fig. 3), which
reflects the self-renewal potential of cancer stem-like cells. The
cancer stem-like cells from tumor spheres stained pale with
toluidine blue (LCs; Fig. 4) and
were endowed with features of CSCs (7). Stemness-associated genes Oct4 and
SOX2 and putative stem cell markers, ALDH and Oct4 were expressed
in the tumor sphere cells but not in the parental adherent
monolayer HeLa cells.
Positive stemness markers and the ability to form
spheres are considered to be hallmarks of CSCs (7,14,22,23),
which are endowed with chemoresistance (18,19)
and a high invasive capacity (24). In the present study, the cancer
stem-like cells from tumor spheres were more resistant to cisplatin
and epirubicin (Fig. 5) and
exhibited a higher invasive potential (Fig. 6) than the Bcrp1-positive cervical
CSCs (25). These findings
demonstrated that the tumor sphere cells cultured in this
nonadhesive culture system exhibited stemness (18,19,24,25).
The findings in the present study suggested that the cancer
stem-like cells that were enriched and expanded under this
experimental condition may be useful for basic and preclinical
studies of cervical CSCs and or other solid CSCs.
Inducing the differentiation of CSCs, aimed at
attacking the stemness of CSCs and reducing their chemo and
radioresistance, represents a novel modality for cancer
stem-cell-targeting therapy (26).
Adipogenic differentiation induction of cancer cells was previously
reported in breast cancer cells (27–29)
and prostate cancer cells (30);
however, to the best of our knowledge, no studies have investigated
the adipogenic differentiation induction of cervical CSCs. In the
present study, adipogenic differentiation was induced in tumor
sphere HeLa cells. This suggested that the stemness phenotype of
cervical CSCs was able to be reversed and highlights a promising
avenue for the therapeutics of cervical cancer through
differentiation induction of CSCs.
In conclusion, the cervical cancer stem-like cells
were enriched and expanded using a nonadhesive culture system. The
enriched cancer stem-like cells exhibited the CSC phenotype and may
be a useful model for investigating and developing substances
targeting CSCs for the basic and preclinical investigations of
therapeutics of cervical cancer and/or other types of solid cancer.
The stemness phenotype of cervical CSCs was able to be reversed and
the differentiation induction of cervical CSCs may be a novel
modality in the treatment and/or prevention of human cervical
cancer, and thus requires further investigation.
Acknowledgements
The authors would like to thank Dr Peizhi Zhuo, Dr
Ju Li and Dr Zhengdong Niu for their assistance in performing the
experiments for this study.
References
|
1
|
Jemal A, Bray F, Center MM, et al: Global
Cancer Statistics. CA Cancer J Clin. 61:69–90. 2011. View Article : Google Scholar
|
|
2
|
Lea JS and Lin KY: Cervical Cancer. Obstet
Gynecol Clin North Am. 39:233–253. 2012. View Article : Google Scholar
|
|
3
|
Green JA, Kirwan JM, Tierney JF, et al:
Survival and recurrence after concomitant chemotherapy and
radiotherapy for cancer of the uterine cervix: a systematic review
and meta-analysis. Lancet. 358:781–786. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Pectasides D, Kamposioras K, Papaxoinis G,
et al: Chemotherapy for recurrent cervical cancer. Cancer Treat
Rev. 34:603–613. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ichim CV and Wells RA: First among equals:
the cancer cell hierarchy. Leuk Lymphoma. 47:2017–2027. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Mao XG, Guo G, Wang P, et al: Maintenance
of critical properties of brain tumor stem-like cells after
cryopreservation. Cell Mol Neurobiol. 30:775–786. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Cioce M, Gherardi S, Viglietto G, et al:
Mammosphere-forming cells from breast cancer cell lines as a tool
for the identification of CSC-like- and early progenitor-targeting
drugs. Cell Cycle. 9:2878–2887. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Su YJ, Lai HM, Chang YW, et al: Direct
reprogramming of stem cell properties in colon cancer cells by
CD44. EMBO J. 30:3186–3199. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhang L, Jiao M, Li L, et al: Tumorspheres
derived from prostate cancer cells possess chemoresistant and
cancer stem cell properties. J Cancer Res Clin Oncol. 138:675–686.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
López J, Poitevin A, Mendoza-Martínez V,
et al: Cancer-initiating cells derived from established cervical
cell lines exhibit stem-cell markers and increased radioresistance.
BMC Cancer. 12:482012.PubMed/NCBI
|
|
11
|
Lee J, Kotliarova S, Kotliarov Y, et al:
Tumor stem cells derived from glioblastomas cultured in bFGF and
EGF more closely mirror the phenotype and genotype of primary
tumors than do serum-cultured cell lines. Cancer Cell. 9:391–403.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hueng DY, Sytwu HK, Huang SM, et al:
Isolation and characterization of tumor stem-like cells from human
meningiomas. J Neurooncol. 104:45–53. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhong Y, Guan K, Guo S, et al: Spheres
derived from the human SK-RC-42 renal cell carcinoma cell line are
enriched in cancer stem cells. Cancer Lett. 299:150–160. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chen SF, Chang YC, Nieh S, et al:
Nonadhesive culture system as a model of rapid sphere formation
with cancer stem cell properties. PLoS One. 7:e318642012.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ponti D, Costa A, Zaffaroni N, et al:
Isolation and in vitro propagation of tumorigenic breast cancer
cells with stem/progenitor cell properties. Cancer Res.
65:5506–5511. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wedel S, Hudak L, Seibel JM, et al:
Molecular targeting of prostate cancer cells by a triple drug
combination down-regulates integrin driven adhesion processes,
delays cell cycle progression and interferes with the cdk-cyclin
axis. BMC Cancer. 11:3752011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kim MJ, Kim YJ, Park HJ, et al: Apoptotic
effect of red wine polyphenols on human colon cancer SNU-C4 cells.
Food Chem Toxicol. 44:898–902. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hu Y and Fu L: Targeting cancer stem
cells: a new therapy to cure cancer patients. Am J Cancer Res.
2:340–356. 2012.PubMed/NCBI
|
|
19
|
Gilbert CA and Ross AH: Cancer stem cells:
cell culture, markers, and targets for new therapies. J Cell
Biochem. 108:1031–1038. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kim RK, Kim MJ, Yoon CH, et al: A new
2-pyrone derivative,
5-bromo-3-(3-hydroxyprop-1-ynyl)-2H-pyran-2-one, suppresses
stemness in glioma stem-like cells. Mol Pharmacol. 82:400–407.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Cheng W, Liu T, Wan X, et al:
MicroRNA-199a targets CD44 to suppress the tumorigenicity and
multidrug resistance of ovarian cancer-initiating cells. FEBS J.
279:2047–2059. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Mather JP: In vitro models. Stem Cells.
30:95–99. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
23
|
Tirino V, Desiderio V, Paino F, et al:
Methods for cancer stem cell detection and isolation. Methods Mol
Biol. 879:513–529. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chinn SB, Darr OA, Peters RD, et al: The
role of head and neck squamous cell carcinoma cancer stem cells in
tumorigenesis, metastasis, and treatment failure. Front Endocrinol
(Lausanne). 3:902012.PubMed/NCBI
|
|
25
|
Zhang SL, Wang YS, Zhou T, et al:
Isolation and characterization of cancer stem cells from cervical
cancer HeLa cells. Cytotechnology. 64:477–484. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Pham PV, Phan NL, Nguyen NT, et al:
Differentiation of breast cancer stem cells by knockdown of CD44:
promising differentiation therapy. J Transl Med. 9:2092011.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zheng ZH, Yang Y, Lu XH, et al:
Mycophenolic acid induces adipocyte-like differentiation and
reversal of malignancy of breast cancer cells partly through PPARγ.
Eur J Pharmacol. 658:1–8. 2011.PubMed/NCBI
|
|
28
|
Yan J, Luo D, Luo Y, et al: Induction of
G1 arrest and differentiation in MDA-MB-231 breast cancer cell by
boehmeriasin A, a novel compound from plant. Int J Gynecol Cancer.
16:165–170. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Chakraborty A, Bodipati N, Demonacos MK,
et al: Long term induction by pterostilbene results in autophagy
and cellular differentiation in MCF-7 cells via ROS dependent
pathway. Mol Cell Endocrinol. 355:25–40. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhau HE, He H, Wang CY, et al: Human
prostate cancer harbors the stem cell properties of bone marrow
mesenchymal stem cells. Clin Cancer Res. 17:2159–2169. 2011.
View Article : Google Scholar : PubMed/NCBI
|