Introduction
Stroke, a cerebrovascular disease, is defined as a
sudden neurological deficiency in the brain, which is caused by
either hemorrhage or a lack of blood supply in the brain, termed
ischemia. A total of 80% of strokes are caused by arterial blockage
and consequent ischemia and 20% are caused by hemorrhage. At
present, stroke represents one of the three most dangerous diseases
threatening human health (1). It
has been reported that 70–80% of stroke survivors have a level of
disability, particularly post-apoplectic limb spasm, which has
become an increasingly serious social problem (2). Post-apoplectic limb spasm is one of
the key difficulties, which is faced during rehabilitation therapy
following stroke, due to its high rate of motor impairment,
difficulty of treatment and importance for limb function (3). The identification of effective
therapeutic approaches to treat post-apoplectic limb spasm is
important for stroke rehabilitation.
It is well established that excitotoxicity is the
major pathophysiological mechanism underlying ischemic
stroke-induced brain injury (4).
Furthermore, excitatory amino acid (EAA)-induced excitotoxicity has
been proposed to be involved in post-apoplectic limb spasm.
Ischemia-reperfusion (I/R)-induced injury has been reported to be
associated with numerous biochemical events, a number of which may
result from the release of EAAs and the activation of their
receptors (5). When EAAs reach
toxic levels, they contribute to increased excitability, which
leads to injury (6). The
associated ion channels open and cause excessive Ca2+
influx, which stimulates a cascade of enzymatic processes, leading
to irreversible injury (7). EAAs,
including glutamic acid (Glu) and aspartic acid (Asp) are important
neurotransmitters in the central nervous system (CNS) and have a
major role in synaptic transmission. It is well established that
EAAs are involved in the transmission of sensory information and
motion instruction (8).
A large number of pharmacological agents have been
used in attempt to improve motor impairment. These agents work
through a variety of neurotransmitter- and receptor-associated
mechanisms (9). A number of these
agents have been used in clinical trials with varying results;
however, many of these agents exhibit considerable side effects.
Traditional Chinese medicine has advantages compared with these
other pharmacological agents, including lower side effects, thus
more regular doses may be taken. Traditional Chinese medicines,
including Buyang Huanwu Decotion (10) and Xiao-Xu-Ming decotion (11), have been used for stroke and
muscular therapy to treat post-apoplectic limb spasm with the
advantages of easy administration and few adverse reactions.
Gua Lou Gui Zhi decotion (GLGZD) is a traditional
Chinese medicine that consists of the following extracts: Radix
Trichosanthis, Radix Paeoniae Alba, Ramulus Cinnamomi, Rhizoma
Zingiberis Recens, Radix Glycyrrhizae and Fructus Ziziphi Jujubae.
Clinical trials have found that GLGZD improves limb muscle tension,
benefiting locomotive function and quality of life in patients with
stroke (12–14). However, the effect of GLGZD on the
release of EAAs in the brain during I/R and the underlying
mechanisms have yet to be elucidated.
Middle cerebral artery occlusion (MCAO) followed by
reperfusion is a model of focal ischemia in rats, which resembles
human ischemic stroke (15). The
present study aimed to investigate the levels of EAAs and their
receptors within the brain and whether GLGZD had a protective
effect against I/R through modulating the release of EAAs and the
expression of EEA receptors. Infarct volumes and neurological
deficits were measured and analyzed in five groups.
Materials and methods
Chemicals, reagents and animals
Specific pathogen-free male Sprague-Dawley rats
(weight, 180–220 g) were obtained from the Laboratory Animal Center
of Fujian University of Traditional Chinese Medicine (Fuzhou,
China). The principles of laboratory animal care were followed and
the study was approved by the Ethics Committee of Fujian University
of Traditional Chinese Medicine.
Dried drugs were purchased from Tongchun Drugstore
(Fuzhou, China) and were identified by Professor Yang (College of
Pharmacy, Fujian University of Traditional Chinese Medicine).
Voucher specimens were deposited at the College of Pharmacy of
Fujian University of Traditional Chinese Medicine. Peoniflorin,
liquiritigenin, liquiritin, cinnamaldehyde, cinnamic acid and
glycyrrhizin were purchased from the National Institute for the
Control of Pharmaceutical and Biological Products (Beijing, China).
Methanol and acetonitrile were of high performance liquid
chromatography (HPLC)-grade and purchased from Merck KGaA
(Darmstadt, Germany). Phosphoric acid, hydrochloric acid,
petroleum, chloroform, acetoacetate and N-butanol were used as
analytical reagents and were purchased from Aladdin Reagents Co.,
Ltd. (Shanghai, China). Other reagents were all of analytical
grade. The deionized water used throughout the experiments was
generated using a Millipore water purification system (Billerica,
MA, USA). A CX31 microscope was purchased from Olympus Corporation
(Tokyo, Japan).
Preparation of GLGZD
According to ‘Jinkui Yaolue’ (16), GLGZD consists of Radix
Trichosanthis, Radix Paeoniae Alba, Ramulus Cinnamomi, Rhizoma
Zingiberis Recens, Radix Glycyrrhizae and Fructus Ziziphi Jujubae.
In order to obtain the GLGZD water extract, volatile oils were
extracted from Ramulus Cinnamomi and Rhizoma Zingiberis Recens.
Other medical materials were decocted through boiling in distilled
water twice for 1 h. The solution was then dried under vacuum to
obtain a final concentration of 1.08 g/ml. GLGZD was then stored
for further analysis.
HPLC analysis of GLGZD
The GLGZD extract was subjected to HPLC analysis.
Sample solutions were injected into the HPLC system (Shimadzu
Corporation, Kyoto, Japan) for analysis in triplicate. The HPLC
system was equipped with a LC-20A pump system, SPD-M20A photodiode
array detector and Diamonsil® C18
reversed-phase column (I.D. 4.6×250 mm, 5 μm). Separation was
achieved using a linear gradient program for mobile phase A
(acetonitrile) and mobile phase B (water containing 0.1% phosphoric
acid). The elution was initiated with a gradient of 95% B for 45
min, followed by 68% for 15 min and 52% for 5 min. Flow rate and
injection volume were 1.0 ml/min and 10 μl, respectively.
Focal cerebral I/R model
An intraluminal suture method was used for the
induction of focal cerebral ischemia. Rats were anesthetized using
10% chloral hydrate solution (0.3 ml/100 g body weight;
intraperitoneal injection). MCAO was induced using an intraluminal
suture method as described previously, but with certain
modifications (15). In brief, the
left common carotid artery (CCA) and the external carotid artery
(ECA) were exposed. A 3-0 surgical monofilament nylon suture was
then inserted from the ECA into the internal carotid artery (ICA)
and was used to occlude the origin of the left MCA until light
resistance was felt (18–20 mm from the CCA bifurcation). After 2 h
of MCAO, the nylon suture was withdrawn, followed by 2 h of
reperfusion.
Experimental grouping and treatment
Sixty rats were divided into the following five
experimental groups and underwent the following treatments:
Sham-operated group (n=12), the rats were subjected to surgical
procedure, but MCAO was not induced, except for exposure of the
right ICA and the right ECA; MCAO model group (n=12), rats received
normal saline and underwent MCAO surgery; positive control group
(n=12), rats received piracetam (6 g/kg body weight) and underwent
MCAO surgery; GLGZD low-dose group (n=12), rats received GLGZD (3.6
g/kg body weight) and underwent MCAO surgery; and GLGZD high-dose
group (n=12), rats received GLGZD (14.2 g/kg body weight) and
underwent MCAO surgery. In the GLGZD treatment groups, GLGZD was
administered once a day for seven days.
Scoring neurological defects
Rats were scored based on a five-point scale
(17). The scale ratings were as
follows: 0, no neurological symptoms; 1, unable to completely
extend the front jaw on the other side; 2, rotating while crawling
and falling to the contralateral side; 3, unable to walk without
assistance; and 4, unconsciousness. Rats with a score of 1–3 were
considered successful models and were included in the study. A
sample of the ipsilateral cortex was taken at the indicated time
and used for sample preparation.
Behavioral tests were performed on the rats after 60
min of ischemia followed by seven days of exercise. Each rat was
scored based on a five-point scale as described previously
(18).
Screen tests
In order to investigate motor function recovery in
the rats subjected to ischemia, motor performance was measured
prior to surgery and on days one and eight following surgery. In
order to measure the muscle strength of the forelimbs, a net screen
was used (19). The trial
commenced subsequent to placing the rat on the horizontal screen on
the ground. The screen was turned over 90° within 2 sec by raising
one side gradually. The screen was maintained in this position for
5 sec.
The duration of time for which rats held on to the
net screen was recorded in seconds. The scoring criterion was as
follows: 5, holding on to the screen and climbing upward; 4,
holding on to the screen with forelimbs and not falling down; 3,
holding on to the screen temporally, but slipping a certain
distance; 2, falling down to the ground within the test period; 1,
falling down to the ground as soon as the screen was at 90°.
2,3,5-triphenyltetrazolium chloride (TTC)
staining and measurement of brain ischemic infarct volume
The six rats in each group were sacrificed by
chloral hydrate and decapitated in order to remove the brain to
measure the infarct volume following I/R. The brain was placed at
20°C for ~10 min, then cut into six coronal slices continuously
from front to back using a blade. The brain tissues were immersed
into 2% TTC solution (T8877; Sigma-Aldrich, St. Louis, MO, USA) in
phosphate-buffered saline (PBS; pH 7.4) and stained at 37°C for 1 h
and turned over several times. Subsequent to staining, the viable
cerebral tissue was stained red while the infarcted cerebral tissue
remained pale. Images were captured using a high-resolution digital
camera (IXUS130; Canon, Tokyo, Japan) and analyzed using Image-Pro
Plus (Media Cybernetics, Inc., Rockville, MD, USA). Infarct volume
was quantified using the Motic Med 6.0 Digital Medical Image
Analysis system (Motic Instruments Inc., Richmond, Canada), The
infarct size was calculated as a percentage of the viable cerebral
tissue of the whole brain (20).
Analysis of Glu, Asp and glycine (Gly)
levels in the cerebrospinal fluid
Cerebrospinal fluid was collected and the Glu, Asp
and Gly levels were analyzed using a Hitachi automatic amino acid
analyzer (21).
Cerebral histopathology
Brain samples were collected from six rats in each
group for cerebral histopathological analysis. The brain samples
were paraffin-embedded, sliced and stained with hematoxylin and
eosin. Histopathological changes were observed using a light
microscope.
Immunohistochemistry
Paraffin-embedded brain tissue samples (0.5×0.5×0.1
cm) were used for immunohistochemical analysis of
N-methyl-D-aspartic acid receptor (NMDAR) and glutamate receptor
(GluR) 1/2/3/4 expression. Briefly, the paraffin sections were
dewaxed, repaired using citric acid, incubated with 3%
H2O2, washed using PBS and blocked with
normal goat serum. Sections were then incubated with primary
antibodies (polyclonal rabbit Anti-NMDA-NR and Anti-GluR1/2/3/4;
Beijing Biosynthesis Co., Ltd., Beijing, China) at room temperature
for 2 h, washed using PBS and incubated with biotinylated secondary
antibodies. Sections were then washed, incubated with horseradish
peroxidase-labeled streptavidin and stained with
3,3′-diaminobenzidine. PBS was used to replace the primary antibody
as a negative control. Five high-power fields (magnification, ×400)
were randomly selected in each slide.
Statistical analysis
Data are presented as the mean ± standard deviation.
Analysis of variance was performed to determine significant
differences between the groups. SPSS 16.0 statistical software
(SPSS, Inc., Chicago, IL, USA) was used for statistical analyses.
P<0.05 was considered to indicate a statistically significant
difference.
Results
HPLC of GLGZD
Various mobile phase systems were used to resolve
the active constituents of GLGZD. The proposed method of analysis
gave simultaneous quantification of six compounds. The individual
constituents were identified by comparing their peaks, UV spectra
and retention times against their corresponding reference standards
(Fig. 1). The percentage content
of each compound was estimated using a calibration curve. The
compounds present in the GLGZD extract were peoniflorin (74.2
mg/g), liquiritigenin (0.92 mg/g), liquiritin (0.09 mg/g), cinnamic
acid (0.20 mg/g), cinnamaldehyde (2.13 mg/g) and glycyrrhizic acid
(1.14 mg/g).
Effect of GLGZD on infarct volume
As shown in Fig. 2,
the infarct volume in the MCAO model group was found to be
significantly higher than that in the sham-operated group.
Furthermore, seven days following ischemia, infarct volumes were
observed to be decreased in the GLGZD groups compared with the MCAO
model group. The infarct volumes in the MCAO model group vs. the
GLGZD groups were as follows: 0.77±0.13 vs. 0.29±0.12% for the
GLGZD low-dose group and 0.77±0.13 vs. 0.18±0.12% (P<0.05) for
the GLGZD high-dose group. Infarct volume was more greatly reduced
in the GLGZD high-dose group than the GLGZD low-dose group. The
infarct volumes in the MCAO model group vs. the piracetum group was
0.77±0.13 vs. 0.56±0.07% (P<0.05), showing that the results were
significantly different between the two groups.
 | Figure 2Effect of GLGZD on cerebral infarction
in focal cerebral ischemia-reperfusion injury rats. (A) Following
the treatment conditions, cerebral tissues from rats from each
group were coronally sectioned into 2-mm thick sections and stained
using 2,3,5-triphenyl tetrazolium chloride. Images were captured
using a high-resolution digital camera. (B) Infarct volume was
quantified using the Motic Med 6.0 Digital Medical Image Analysis
system, which was represented as a percentage of the total brain
volume. Data are presented as the mean ± standard error from five
individual rats in each group. *P<0.05, vs.
sham-operated group and #P<0.05, vs. MCAO model
group. GLGZD, Gua Lou Gui Zhi decotion; MCAO, middle cerebral
artery occlusion; H, high-dose; L, low-dose. |
Effect of GLGZD on neurological behavior
and motor performance
The effect of GLGZD on neurological and motor
function were assessed by measuring neurological and motor
performance. The induction of MCAO for 60 min followed by
reperfusion for 2 h caused marked changes in rat behavior. The
neurological behavior of the rats that were subjected to MCAO was
measured using the scoring method described by Longa et al
(15). Motor performance was
measured using a screen test scoring method in order to investigate
the motor function recovery of the rats subjected to MCAO.
As shown in Fig. 3A and
B, the rats in the MCAO model group that were subjected to I/R
injury, exhibited severe neurological deficit (score: 2) and motor
disorder, and showed circling towards the contralateral side with a
reduced mobility compared with the rats in the sham-operated group
(score: 0). Rats in the GLGZD groups showed significant
improvements in behavior between days 1 and 7, particularly those
in the high-dose group. Rats in the piracetum group showed
improvements in behavior between days 5 and 7, while no changes in
neurological function or motor performance were observed in the
rats in the sham-operated group.
Effect of GLGZD on levels of the EAAs
Glu, Asp and Gly in the cerebrospinal fluid
The excessive release of EAAs in the brain following
I/R injury is closely associated with post-apoplectic limb spasm.
As shown in Table I, MCAO induced
significant increases in the levels of the excitatory
neurotansmitters Glu, Asp and Gly. The levels of Glu, Asp and Gly
in the low- and high-dose GLGZD groups were lower than those in the
MCAO model group, but higher than those in the sham-operated group.
Similarly, compared with the MCAO model group, the levels of Glu,
Asp and Gly in the piracetum group decreased and were significantly
different (P<0.05). These findings show that GLGZD had a
modulatory effect on EAA levels.
 | Table ILevels of the excitatory amino acids
Glu, Asp and Gly in the cerebrospinal fluid. |
Table I
Levels of the excitatory amino acids
Glu, Asp and Gly in the cerebrospinal fluid.
| Group | Asp | Glu | Gly |
|---|
| Sham-operation | 3.00±0.12. | 4.27±0.72. | 1.02±0.04. |
| MCAO model | 4.84±0.23a | 6.34±0.66a | 1.65±0.02a |
| Piracetam | 3.25±0.15b | 4.27±0.68b | 0.85±0.06b |
| GLGZD-L | 3.36±0.26. | 4.64±0.81b | 0.96±0.04. |
| GLGZD-H | 3.34±0.11b | 4.43±0.75b | 0.72±0.03b |
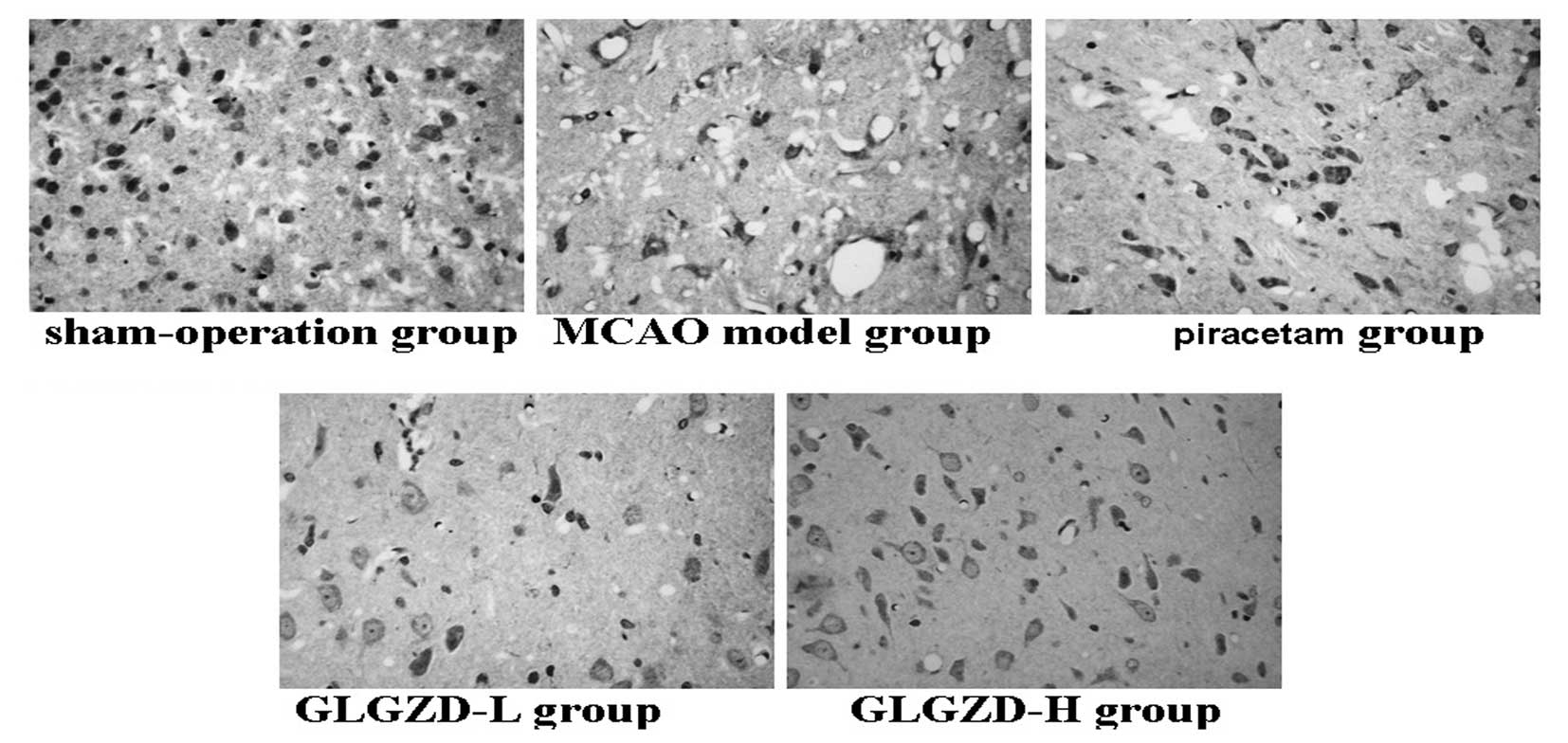
Cerebral histopathology
As shown in Fig. 4,
the cortical neural cells in the sham-operated group were observed
to be arranged orderly and to exhibit normal cell morphology, with
clearly visible structures and integrity. The cell membranes and
nuclei were normal, as was the tissue interspace, which exhibited
no edema or inflammatory cell infiltration. The cortical region of
the ischemic side of the brain in the rats in the MCAO model group
exhibited visible disorder and high levels of cell necrosis, with
cells showing nucleolar shrinkage and breakdown, as well as
vacuolar degeneration. Furthermore, the mesenchymal cells
demonstrated high levels of edema and inflammatory cell
infiltration compared with those in the MCAO model group. The
cortical neural cells in the rats in the GLGZD groups showed
decreased pathological changes compared with those in the MCAO
model group. Neuronal degeneration and necrosis, as well as
disorderly cell arrangement, tissue edema, nuclear dissolution and
nucleolar shrinkage were observed in the cells in the GLGZD
treatment groups; however, the quantity and extent was decreased
compared with that in the MCAO model group. Similarly, compared
with the MCAO model group, the cortical neural cells in the rats in
the piracetum groups exhibited reduced pathological changes
Immunohistochemistry
Semi-quantitative scoring was performed according to
the ratio of positively stained cells and the staining intensity
(22,23). Immunohistochemical scoring revealed
a significant increase in NMDAR and GluR1/3/4, and a significant
decrease in GluR2 in the hippocampus of the MCAO model rats
compared with the sham rats (Fig.
5; Table II). After seven
days of high-dose GLGZD administration, the scores were further
reduced to 4.4±0.6 for NMDAR (P<0.05), 4.4±0.7 for GluR1
(P<0.05), 3.6±0.3 for GluR3 (P<0.01) and 3.6±0.7 for GluR4,
while they were upregulated to 3.6±0.3 for GluR2 (P<0.01). For
the piracetum groups, the scores were reduced to 4.0±0.1 for NMDAR
(P<0.05), 4.2±0.6 for GluR1 (P<0.05), 3.6±0.4 for GluR3
(P<0.01) and 2.8±0.5 for GluR4, while they were upregulated to
4.0±0.0 for GluR2 (P<0.01). Alteration in the expression of
NMDAR and GluR1/2/3/4 was neutralized with GLGZD treatment.
 | Table IIImmunohistochemical scores of NMDAR
and GluR1/2/3/4 expression in brain tissue. |
Table II
Immunohistochemical scores of NMDAR
and GluR1/2/3/4 expression in brain tissue.
| Group | NMDAR | GluR1 | GluR2 | GluR3 | GluR4 |
|---|
| Sham-operation | 1.8±0.4. | 2.0±0.6. | 5.6±0.4. | 4.8±0.5 | 3.2±0.5 |
| MCAO model | 5.6±0.2a | 5.2±0.7a | 1.6±0.6a | 5.6±0.4 | 4.0±0.9 |
| Piracetam | 4.0±0.1b | 4.2±0.6b | 4.0±0.0c | 3.6±0.4c | 2.8±0.5 |
| GLGZD-L | 4.8±0.5. | 4.6±0.1. | 3.2±0.4c | 4.4±0.7 | 4.4±0.7 |
| GLGZD-H | 4.4±0.6b | 4.4±0.7b | 3.6±0.3c | 3.6±0.7 | 3.6±0.7 |
Discussion
Stroke often causes motor impairment, which may be
defined as a velocity-dependent increase in resistance to passive
stretch or movement, resulting in abnormal signaling between the
CNS and the muscles, leading to muscle excitability (24). In the CNS, EAAs and their receptors
have been shown to have a crucial role in motor impairment
following cerebral I/R injury. Glu is the predominant excitatory
neurotransmitter in the brain, and the subsequent activation of its
receptors induces an increase in intracellular calcium and the
generation of free radicals, leading to cell death, necrosis or
apoptosis (25). Asp is present in
high levels in the CNS within the hypothalamus and cerebellum, and
is an excitatory neurotransmitter of intermediate neurons, which
causes neuron depolarization and generates excitatory postsynaptic
potential with slow and persistent function (26). It is well established that Gly has
an important role in inhibitory synaptic transmission in the spinal
cord and the brain stem. Furthermore, it has been reported that Gly
is an important facilitator of glumate function in the cerebrum,
through an allosteric site on the NMDA receptor (27). Therefore, Gly may have excitatory
and inhibitory roles in the CNS.
Ionotropic glutamate receptors consist of three
subclasses: NMDA, AMPA and kainate receptors, termed based on their
characteristic agonists. AMPA and kainate receptors are often known
as non-NMDA receptors. NMDA and non-NMDA glutamate receptors have a
role in mediating motor impairment (28). NMDAR toxicity is dependent on
extracellular Ca2+ and occurs as a consequence of high
Ca2+ influx directly through the receptor-gated ion
channel in the receptor (29).
However, the majority of AMPA receptor (AMPAR) channels have poor
Ca2+ permeability (30). AMPA-type GluRs are heteromeric
channels comprised of a combination of GluR1-4 subunits (31), with changes in the expression of
GluR1-4 being associated with development, aging, epilepsy and
alterations in synaptic efficacy. The presence of the GluR2 subunit
determines the permeability of the AMPAR to Ca2+ and
other divalent cations. The majority of neurons exhibit high levels
of GluR2 expression, which renders these cells impermeable to
calcium influx thorugh AMPARs (32). Thus, AMPAR Ca2+
permeability is considered to be determined by the relative
abundance of GluR2 subunits in the heteromeric AMPA receptors. It
has been reported that ischemic conditions may alter the subunit
composition of AMPA receptors towards a more
Ca2+-permeable combination, which may promote neuronal
degeneration (33).
GLGZD has been used for the treatment of
post-apoplectic limb spasm. Yang et al (14) showed that GLGZD had a significant
therapeutic effect on post-apoplectic limb spasm. The present study
aimed to investigate the effect of GLGZD on focal I/R
insult-induced motor impairment and behavioral disturbance, by
inducing cerebral I/R injury in rats. GLGZD was found to improve
neurological and motor performance. Furthermore, the present study
identified a large infarct volume mainly in the cortex and striatum
(or in the transient focal cerebral ischaemic brains) of the
ipsilateral hemisphere of the brain in the ischemic rats.
Pretreatment with GLGZD at high- and low-doses was observed to
reduce the infarct volume compared with the sham-operated group.
This finding suggests that oral administration of GLGZD extracts
significantly inhibits reperfusion-induced neuronal cell death. In
the present study, behavioral tests were used to assess the success
of MCAO. Screen tests were used to assess behavioral dyskinesia
caused by cerebral infarction. The findings suggest that GLGZD
improves stroke-induced neurobehavioral abnormalities in rats.
In the present study, the quantity of Glu and Asp
were found to increase markedly in the brain tissue of the rats in
the MCAO model group, which indicates that the cytotoxicity of EAAs
is involved in the establishment of ischemic brain injury and
delayed nerve damage. The level of Gly in the brain tissue was
observed to increase significantly in the MCAO model group, which
may have a role in cerebral ischemia injury and a synergistic toxic
effect with Glu. GLGZD treatment was found to inhibit the release
of Glu and Asp during I/R.
Immunohistochemistry of the NMDAR revealed that the
mice in the low- and high-dose GLGZD groups exhibited decreased
positive NMDA expression, which may reduce excessive activation of
the NMDA receptor, as well as reduce nerve cell cytotoxicity,
necrosis and apoptosis. Immunohistochemistry of AMPAR subunits
revealed that the the mice in the low- and high-dose GLGZD groups
exhibited increased GluR2 expression, but decreased GluR1, 3 and 4
expression, which may prevent Ca2+ influx and have a
protective effect against nerve injury. Thus, the neuroprotective
effects of GLGZD may be partially due to its capacity to attenuate
excitotoxic Glu and Asp accumulation. The effect of Gly and its
receptors on cerebral I/R injury requires further
investigation.
In conclusion, the present study established a HPLC
method to qualitatively and quantitatively analyze the effect of
GLGZD on EEAs and their receptors in the brains of MCAO model rats.
The findings provides important insight into GLGZD as a
neuroprotective agent.
Acknowledgements
This research was done in state key laboratory of
Chinese pharmacies of Fujian Provincial Department of Science and
Technology, Collaborative Innovation Center for Rehabilitation
Technology and TCM Rehabilitation Research Center of SATCM. It was
funded by the Important Subject of Fujian Province Science and
technology hall of China (2012Y0041) and the important subject of
Fujian province Education Hall of China (JA12176).
Abbreviations:
|
GLGZD
|
Gua Lou Gui Zhi decotion
|
|
MCAO
|
middle cerebral artery occlusion
|
|
CSF
|
cerebrospinal fluid
|
|
NMDA
|
N-methyl-D-aspartic acid
|
|
AMPA
|
α-amino-3-hydroxy-5-methyl-4-isoxazole-propionic acid
|
|
TTC
|
2,3,5-triphenyl tetrazolium
chloride
|
|
EAAs
|
excitatory amino acids
|
|
CNS
|
central nervous system
|
References
|
1
|
Zeng MF, Pan LM, Zhu HX, Zhang QC and Guo
LW: Comparative pharmacokinetics of baicalin in plasma after oral
administration of Huang-Lian-Jie-Du-Tang or pure baicalin in MCAO
and sham-operated rats. Fitoterapia. 81:490–496. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Riddoch MJ, Glyn WH and Bateman A: Stroke
issues in recovery and rehabilitation. Physiotherapy. 81:689–694.
1995.
|
|
3
|
Lamontagne A, Stephenson JL and Fung J:
Physiological evaluation of gait disturbances post stroke. Clin
Neurophysiol. 118:717–729. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wang HW, Liou KT, Wang YH, et al:
Deciphering the neuroprotective mechanisms of Bu-yang Huan-wu
decotion by an integrative neurofunctional and genomic approach in
ischemic stroke mice. J Ethnopharmacol. 138:22–23. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Li C, Yan Z, Yang J, Chen H, Li H, Jiang Y
and Zhang Z: Neuroprotective effects of resveratrol on ischemic
injury mediated by modulating the release of neurotransmitter and
neuromodulator in rats. Neurochem Int. 56:495–500. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Herz RC, Gaillard PJ, Wildt DJ and
Versteeg DH: Differences in striatal extracellular amino acid
concentrations between Wistar and Fischer 344 rats after middle
cerebral artery occlusion. Brain Res. 715:163–171. 1996. View Article : Google Scholar
|
|
7
|
Phillis JW and O’Regan MH:
Characterization of modes of release of amino acids in the
ischemic/reperfused rat cerebral cortex. Neurochem Int. 43:461–467.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Doble A: The role of excitotoxicity in
neurodenenerative disease: implications for therapy. Pharmacol
Ther. 81:163–221. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Van HA, Sarre S, Smolders I and Michotte
Y: Determination of amino acids associated with cerebral ischaemia
in rat brain microdialysates using narrowbore liquid chromatography
and fluorescence detection. J Neurosci Methods. 144:63–71. 2005.
View Article : Google Scholar
|
|
10
|
Chern CM, Liao JF, Wang YH and Shen YC:
Melatonin ameliorates neural function by promoting endogenous
neurogenesis through the MT2 melatonin receptor in ischemic-stroke
mice. Free Radic Biol Med. 52:1634–1647. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhu XH, Li SJ, Hu HH, Sun LR, Das M and
Gao TM: Neuroprotective effects of Xiao-Xu-Ming decotion against
ischemic neuronal injury in vivo and in vitro. J Ethnopharmacol.
37:38–46. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sun X: Research on formula treating
paralysis and spasticity from ‘treatise on febrile and
miscellaneous diseases’. Zhongguo Zhong Yi Ji Chu Yi Xue Za Zhi.
8:644–645. 2010.(In Chinese).
|
|
13
|
Zhang L and Ai H: Effects of Gua Lou Gui
Zhi decotion on c-fos and c-jun in epileptic rats. Sichuan Hua xi
Zhong Yi Yao Yan Jiu Suo. 23:21–22. 2005.(In Chinese).
|
|
14
|
Yang C, Chen L and Tao J: New usage of a
classical formula-Gua Lou Gui Zhi decotion. Liaoning Zhong Yi Za
Zhi. 8:166–167. 2012.(In Chinese).
|
|
15
|
Longa EZ, Weinstein PR, Carlson S and
Cummins R: Reversible middle cerebral artery occlusion without
craniectomy in rats. Stroke. 20:84–91. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang Z: Jinkui Yaolue. Lin Y, Yang P, Hou
X and Yang Y: Synopsis of Golden Chamber. Macmillan Press; Beijing:
pp. 203–204. 2008, (In Chinese).
|
|
17
|
Guo J, Liu L, Ma C, Xu B, Duan X and Wang
B: Effect of restraint stress on depression-like behaviors in rats
after transient focal cerebral ischemic injury. Neural Regen Res.
2:390–394. 2007. View Article : Google Scholar
|
|
18
|
Yin J, Tu C, Zhao J, Ou D, Chen G, Liu Y
and Xiao X: Exogenous hydrogen sulfide protects against global
cerebral ischemia/reperfusion injury viaitsanti-oxidative,
anti-inflammatory and anti-apoptotic effects in rats. Brain Res.
1491:188–196. 2013. View Article : Google Scholar
|
|
19
|
Benedek A, Moricz K, Juranyi Z, Gigler G,
Levay G, Harsing LG Jr, Matyus P, Szénási G and Albert M: Use of
TTC staining for the evaluation of tissue injury in the early
phases of reperfusion after focal cerebral ischemia in rats. Brain
Res. 1116:159–165. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gao WJ, Qian T and Cong B: Effect of
Yishen Jiangzhuo Decotion on the EAA content of hippocampus in
cerebral ischemia reperfusion rats. Chin J Intergrative Med
Cardio/Cerebrovasc Dis. 1:319–321. 2003.
|
|
21
|
Guo C, Tong L, Xi MM, Yang HF, Dong H and
Wen AD: Neuroprotective effect of calycosin on cerebral ischemia
and reperfusion injury in rats. J Ethnopharmacol. 144:768–774.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Soslow RA, Dannenberg AJ, Rush D, Woerner
BM, Khan KN, Masferrer J and Koki AT: Cox-2 is expressed in human
pulmonary, colonic, and mammary tumors. Cancer. 89:2637–2645. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhang Y, Xu W, Li H, Zhang X, Xia Y, Chu K
and Chen L: Therapeutic effects of total alkaloids of
Tripterygium wilfordii Hook f. on collagen-induced arthritis
in rats. J Ethnopharmacol. 45:699–705. 2013.
|
|
24
|
Pisano F, Miscio G, Del Conte C, Pianca D,
Candeloro E and Colombo R: Quantitative measures of spasticity in
post-stroke patients. Clin Neurophysiol. 111:1015–1022. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sucher NJ: Insights from molecular
investigations of traditional Chinese herbal stroke medicines:
implications for neuroprotective epilepsy therapy. Epilepsy Behav.
8:350–362. 2006. View Article : Google Scholar
|
|
26
|
D’Aniello A: D-Aspartic acid: an
endogenous amino acid with an important neuroendocrine role. Brain
Res Rev. 53:215–234. 2007.PubMed/NCBI
|
|
27
|
Yang ZZ, Li J, Li SX, Feng W and Wang H:
Effect of ginkgolide B on striatal extracellular amino acids in
middle cerebral artery occluded rats. J Ethnopharmacol.
136:117–122. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kocsis P, Tarnawa I, Szombathelyi Z and
Farkas S: Participation of AMPA-and NMDA-type excitatory amino acid
receptors in the spinal reflex transmission, in rat. Brain Res
Bull. 60:81–91. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Tu W, Xu X, Peng L, et al: DAPK1
Interaction with NMDA receptor NR2B subunits mediates brain damage
in stroke. Cell. 140:222–234. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Xiong ZG, Zhu XM, Chu XP, et al:
Neuroprotection in ischemia: blocking calcium-permeable
acid-sensing ion channels. Cell. 118:687–698. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kamphuis W, Klooster J and Dijk F:
Expression of AMPA-type glutamate receptor subunit (GluR2) in
ON-bipolar neurons in the rat retina. J Comp Neurol. 455:172–186.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Dijk F and Kamphuis W: Ischemia-induced
alterations of AMPA-type glutamate receptor subunit expression
patterns in the rat retina - an immunocytochemical study. Brain
Res. 997:207–221. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Cebers G, Cebere A and Liljequist S:
Metabolic inhibition potentiates AMPA-induced Ca2+
fluxes and neurotoxicity in rat cerebellar granule cells. Brain
Res. 779:194–204. 1998. View Article : Google Scholar : PubMed/NCBI
|