Introduction
MicroRNAs (miRNAs) are a family of ~22 nucleotide
length small noncoding RNAs that regulate gene expression by
translational repression, acting on the 3′-untranslated region
(UTR) of target genes (1–3). A number of studies have demonstrated
that miRNAs function as either tumor suppressors by downregulating
oncogenic targets, or tumor promoters through negatively regulating
tumor-suppressive target mRNAs (4,5). In
either way, miRNAs may be used as biomarkers for the early
diagnosis of cancer and other diseases (6). In addition, miRNAs and the respective
target genes may be utilized in anticancer therapy in addition to
pharmaceutical applications (7).
Among miRNA families with important functions, the miR-125 family
has been reported to be implicated in a variety of carcinomas as
either tumor suppressor or promoter (8). miR-125b has been revealed to exert
tumor-suppressor functions in several cancer types, including
ovarian (9), bladder (10) and breast cancer (11) as well as hepatocellular carcinoma
(12), melanoma (13) and osteosarcoma (14). However, in contrast to its
tumor-suppressive properties, miR-125b may also act as an oncogene
in several cancers; for example, overexpression of miR-125b has
been investigated in several cancer types, including pancreatic
cancer (15) and prostate cancer
(16).
In 1956, Otto Warburg observed that the rate of
glycolysis was abnormally high in cancer cells, yet a smaller
fraction of this glucose was broken down by oxidative
phosphorylation (17). This
‘Warburg effect’ indicates that cancer cells prefer glycolytic
breakdown of glucose for energy, rather than mitochondrial
oxidative phosphorylation, suggesting that targeting cancer cell
metabolism may be an effective approach to selectively treating
malignant cancer cells (18).
Furthermore, dysregulated metabolism of cancer cells has been
associated with drug resistance (19). Chemoresistant cells usually exhibit
an upregulated glucose metabolism.
Lactate dehydrogenase A (LDHA), an enzyme involved
in anaerobic glycolysis, has been reported to contribute to
paclitaxel resistance in breast cancer (20), and glycolysis suppression through
hexokinase II (HK II) inhibition was found to enhance the cytotoxic
effects of daunorubicin and doxorubicin in vitro and in
vivo (21). In addition, the
pyruvate dehydrogenase kinase inhibitor dichloroacetic acid (DCA)
potentiated the anticancer effects of 5-fluorouracil (5-FU) through
initiating the mitochondrial apoptosis pathway (22), suggesting that targeting the
glucose metabolism of cancer cells contributes to the sensitization
of cancer cells to therapeutic agents.
Hepatocellular carcinoma (HCC) is an aggressive and
highly vascular tumor (23).
Currently, the treatment options for HCC depend on the stage and
grade of the tumor. 5-FU-based chemotherapy is widely used in the
treatment of multiple cancer types, and systemic therapy with 5-FU
alone or in combination with other chemotherapeutic agents has been
utilized in the treatment of HCC (24,25).
However, despite impressive initial clinical responses, the
majority of patients eventually develop resistance to 5-FU. The
mechanisms of 5-FU resistance in HCC remain under investigation;
5-FU-resistant colon cancer cells have been reported to exhibit
upregulated glucose metabolism, and the combination of DCA and 5-FU
treatment exerts a synergistic inhibitory effect (22), indicating a close correlation
between dysregulated glucose metabolism and 5-FU resistance.
In the present study, the tumor suppressor
activities of miR-125b were investigated in human HCC cells.
Materials and methods
Cell lines and cell culture
The SMMC-7221, Huh7, MHCC-97L, HepG2, HepG3 and
BEL-7402 human HCC cell lines, along with THLE-2 and THLE-3 normal
human liver cell lines were obtained from the American Type Culture
Collection (Manassas, VA, USA). Human HCC cells were grown in
Williams’ medium E (#W1878; Sigma-Aldrich, Hong-Kong, China)
supplemented with 10% fetal bovine serum (#10099-141; Gibco-BRL,
Carlsbad, CA, USA) and antibiotics. The THLE-2 and THLE-3 cells
were cultured in LHC-8 medium (#12678-017; Gibco-BRL) supplemented
with 70 ng/ml phosphoethanolamine (#P0503; Sigma-Aldrich), 5 ng/ml
epidermal growth factor (#E5036; Sigma-Aldrich), 10% fetal bovine
serum and penicillin-streptomycin (10,000 U/ml, 10 ml/tube stored
at −20°C; #15140-122; Gibco-BRL). All cells were cultured in a
humidified atmosphere of 5% CO2 in air at 37°C.
5-FU-resistant clone 1, clone 2 and pool cells were developed from
HepG3 cells by treatment with gradually increasing concentrations
of 5-FU (#F6627; Sigma-Aldrich) in cell culture medium. The
resistant cells were re-selected every month through doxorubicin
treatment.
Antibodies and reagents
Anti-HK II (rabbit monoclonal, #2867) and
anti-β-actin (rabbit monoclonal, #4967) antibodies were purchased
from Cell Signaling Technology, Inc. (Danvers, MA, USA). Anti-5-FU
antibodies were provided by Sigma-Aldrich.
Glucose uptake assay
Cells were seeded in 12-well plates at
1×105 to 3×105 cells per well. The culture
media was collected at 48 h and stored at −20°C. Glucose uptake was
measured using an Amplex Red Glucose/Glucose Oxidase assay kit
(Molecular Probes, Camarillo, CA, USA). Absorbance was measured at
563 nm using a SpectraMax M5 plate reader (Molecular Devices,
Sunnyvale, CA, USA) and the results were normalized to the quantity
of total glucose compared with that of the control cells.
Lactate production assay
Lactate production in the medium was detected by
using a Lactate assay kit (BioVision, Mountain View, CA, USA). The
results were normalized to the quantity of total lactate compared
with the control cells.
Pre-miRNA or anti-miRNA transfection
miRNA precursors (pre-miRNAs) and miRNA antisense
RNAs (anti-miRNAs) were purchased from Applied Biosystems China
(Beijing, China). Pre-miR-negative (#AM17110; Ambion, Austin, TX,
USA) served as negative controls. Lipofectamine 2000 (Invitrogen
Life Technologies, Carlsbad, CA, USA) was used for the pre-miRNAs
or anti-miRNA transfections. At 48 h after transfection, the
expression levels of miR-125b were detected by quantitative
polymerase chain reaction and the expression levels of HK II, a
target of miR-125b, were analyzed by western blot analysis.
Plasmid DNA transfection
Transfection was performed using Lipofectamine 2000
transfection reagent (Invitrogen Life Technologies) according to
the manufacturer’s instructions. Overexpression vector containing
wild-type HK II (Myc-DDK-tagged; RC209482) was purchased from
Origene Technologies, Inc. (Rockville, MD, USA). At 48 h following
transfection, the cells were collected for further analysis, and an
aliquot was used to prepare whole-cell lysates.
Generation of the HepG3 5-FU-resistant
cell line
HepG3 cells were treated with gradually increasing
concentrations of 5-FU under regular cell culture conditions for
the selection of resistant cells. Following successive treatments
for up to two months, several resistant cell clones were developed
from the parental cell line. 5-FU-resistant clone 1, clone 2 and
pooled clones were used for subsequent experiments. The resistant
cells were selected by 5-FU treatments each month.
Reverse transcription (RT)-PCR
RNA was isolated from cultured cells using an RNeasy
mini-kit (Qiagen, Hilden, Germany) with an on-column DNAse
digestion step according to the manufacturer’s instructions.
Briefly, the lysates of the cells were passed through a Qiashredder
(Qiagen) and the eluted lysates were mixed 1:1 with 70% ethanol.
The lysates were applied to a mini-column and subsequent to washing
and DNAse I digestion, the RNAs were eluted in 30–50 μl RNAse-free
water. The quantity and quality of total RNA samples was checked by
agarose-gel-electrophoresis and a Bioanalyzer RNA 6000 Nano assay
(Agilent, Waldbronn, Germany). For miRNA expression analysis,
RT-PCR was performed using a TaqMan microRNA reverse transcription
kit (Applied Biosystems, China) and TaqMan microRNA assays kit
(Applied Biosystems) following the manufacturer’s instructions. The
precursor miR-125b RT primer used was: GTCGTATCCAGTGCAGGGTCCGAG
GTATTCGCACTGGATACGACAGCACG. RNU6B (RT primer,
TCGTATCCAGTGCAGGGTCCGAGGTATTCG CACTGGATACGACAAAATATGGAAC;
Invitrogen Life Technologies) served as an internal control. All
reactions were performed in triplicate. End-point PCR was conducted
to analyze the expression levels of miR-125b using a mirVana RT-PCR
miRNA detection kit and mirVana RT-PCR Primer sets (#AM1558;
Applied Biosystems China) according to the manufacturer’s
instructions. Human U6 (Invitrogen Life Technologies)served as an
internal control. All reactions were performed in triplicate. The
relative quantities of mRNA were calculated using the comparative
CT method.
Luciferase reporter assay
The pMIR-reporter luciferase vector containing
either the wild-type HK II 3′-UTR or the binding site mutant HK II
3′-UTR and the empty vector were constructed according to methods
previously described (29). For
the luciferase assay, cells were seeded at a density of
2×105 per well in 24-well plates and co-transfected with
pMIR-REPORT luciferase reporters (#AM5795; Ambion) with the 3′-UTRs
of wild-type HK II or binding site mutant HK II, and pre-miR-125b
or pre-miR-negative using Lipofectamine 2000 reagent. After 48 h,
the cells were harvested and lysed with passive lysis buffer
(Promega Corporation, Madison, WI, USA). Luciferase activity was
measured using a dual luciferase reporter assay (Promega
Corporation). The pRL-TK vector (Promega Corporation) served as an
internal control. The results were expressed as relative luciferase
activity (firefly luciferase/renilla luciferase).
Cell viability assay
The cancer cells were treated with 5-FU at the
indicated concentrations for 48 h. The cells were seeded in a
48-well plate, at a density of 1×104 cells/well in 0.2
ml Williams’ medium E or LHC-8 medium containing 10% fetal bovine
serum. Following overnight incubation under the same cultivating
conditions, each well was refreshed with serum-free medium (SFM)
for another day. The cells were then treated with SFM containing
various concentrations of 5-FU. The drug-containing SFM was
refreshed after two days and incubated under the same conditions.
Cell viability was accessed by MTT (Sigma Diagnostics, Inc., St.
Louis, MO, USA) staining for 1 h followed by the addition of DMSO
and by measuring the absorbance at 590 nm with a SpectraMax Plus384
plate reader (Molecular Devices). The relative viability was
obtained from the absorbance at 590 nm of drug-treated cells/the
absorbance at 590 nm of untreated cells. The same experiment was
repeated three times.
Western blot analysis
The cells were harvested and lysed in a buffer
containing 50 mM Tris-HCl, pH 7.5, 150 mM NaCl, 2 mM EDTA, 1%
Triton, 1 mM phenylmethanesulfonyl fluoride and protease inhibitor
cocktail (Sigma Diagnostics, Inc.) for 20 min on ice. The lysates
were cleared by centrifugation at 10,000 × g at 4°C for 10 min. The
supernatants were collected and protein concentrations were
determined by the Bradford assay (Bio-Rad, Hercules, CA, USA). The
proteins were then separated by SDS-PAGE and transferred to a
nitrocellulose membrane (Bio-Rad). Subsequent to blocking with 5%
non-fat dry milk in phosphate-buffered saline (PBS) for 1 h, the
membranes were incubated overnight at 4–8°C with the primary
antibodies in PBS with 5% non-fat dry milk. The membranes were
extensively washed with PBS and incubated with horseradish
peroxidase-conjugated secondary anti-mouse antibody or anti-rabbit
antibody (1:2,000; Bio-Rad). Following additional washes with PBS,
the antigen-antibody complexes were visualized with an enhanced
chemiluminescence kit (Pierce Biotechnology, Inc., Rockford, IL,
USA). The LAS-3000 Imaging system (Fuji, Tokyo, Japan) was used for
the membrane exposure.
Statistical analysis
Statistical evaluation for data analysis was
determined by unpaired Student’s t-test and GraphPad Prism
software, version 5.0 (GraphPad Software, Inc., La Jolla, CA, USA)
was used. All data are shown as the means. P<0.05 was considered
to indicate a statistically significant difference.
Results
miR-125b acts as a tumor suppressor in
human HCC cells
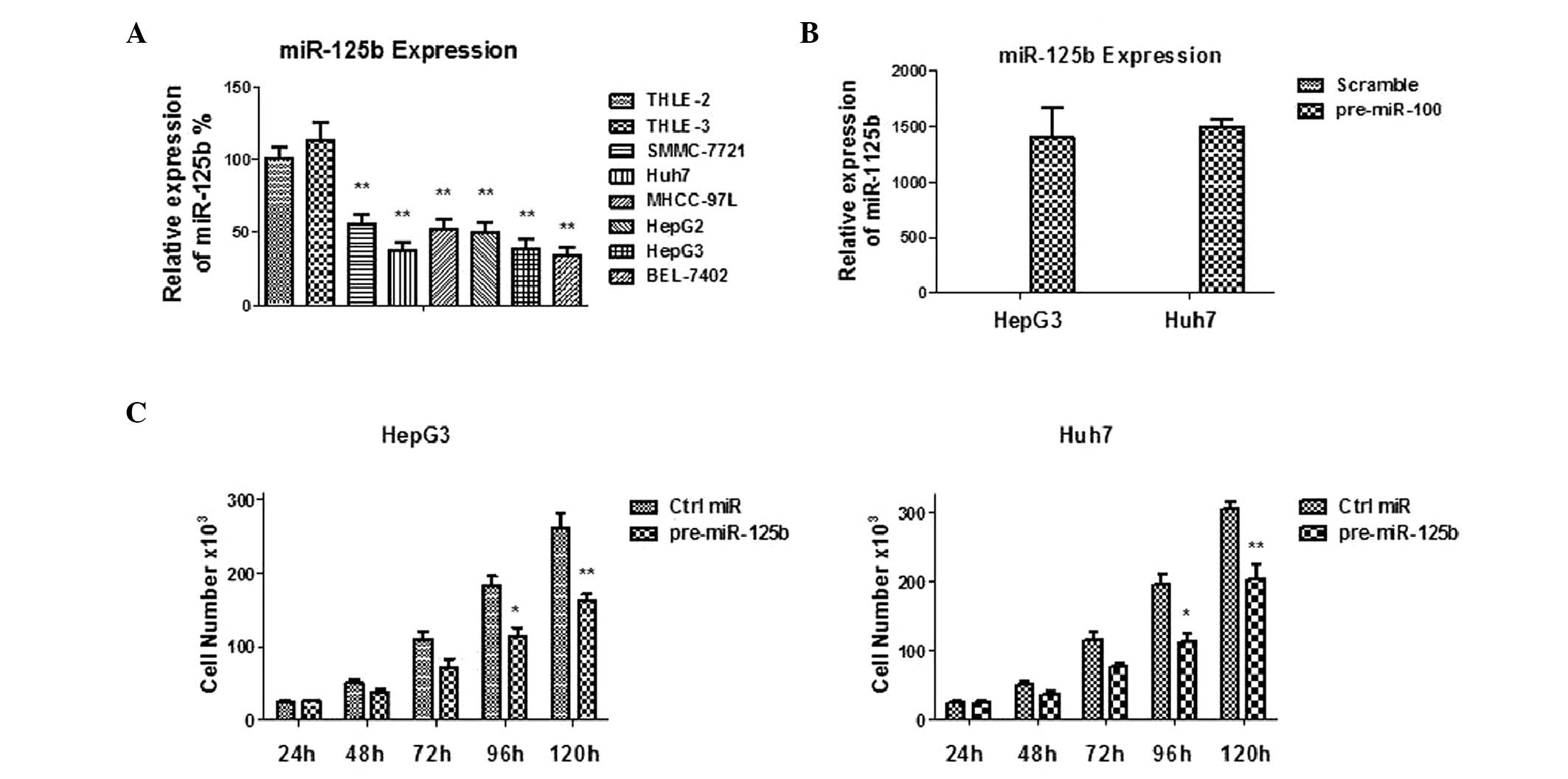
miR-125b has been reported to act as a tumor
suppressor in multiple tumors. In the present study, the expression
levels of miR-125b in multiple types of human HCC cells were
compared with those in normal human hepatocyte cells. A significant
reduction in the expression levels of miR-125b was observed in the
HCC cell lines compared with the normal hepatocytes (Fig. 1A), suggesting that miR-125b may be
a tumor suppressor in human liver cancer cells. The observation
that miR-125b was downregulated in liver cancer cells prompted
investigation into the detailed functions of miR-125b through
transient transfection of miR-125b into two liver cancer cell
lines, HepG3 and Huh7 (Fig. 1B).
The cell growth rates were significantly reduced in
miR-125b-overexpressing liver cancer cells (Fig. 1C), suggesting that miR-125b acts as
a tumor suppressor in human HCC cells.
miR-125b is downregulated in
5-FU-resistant HCC cells
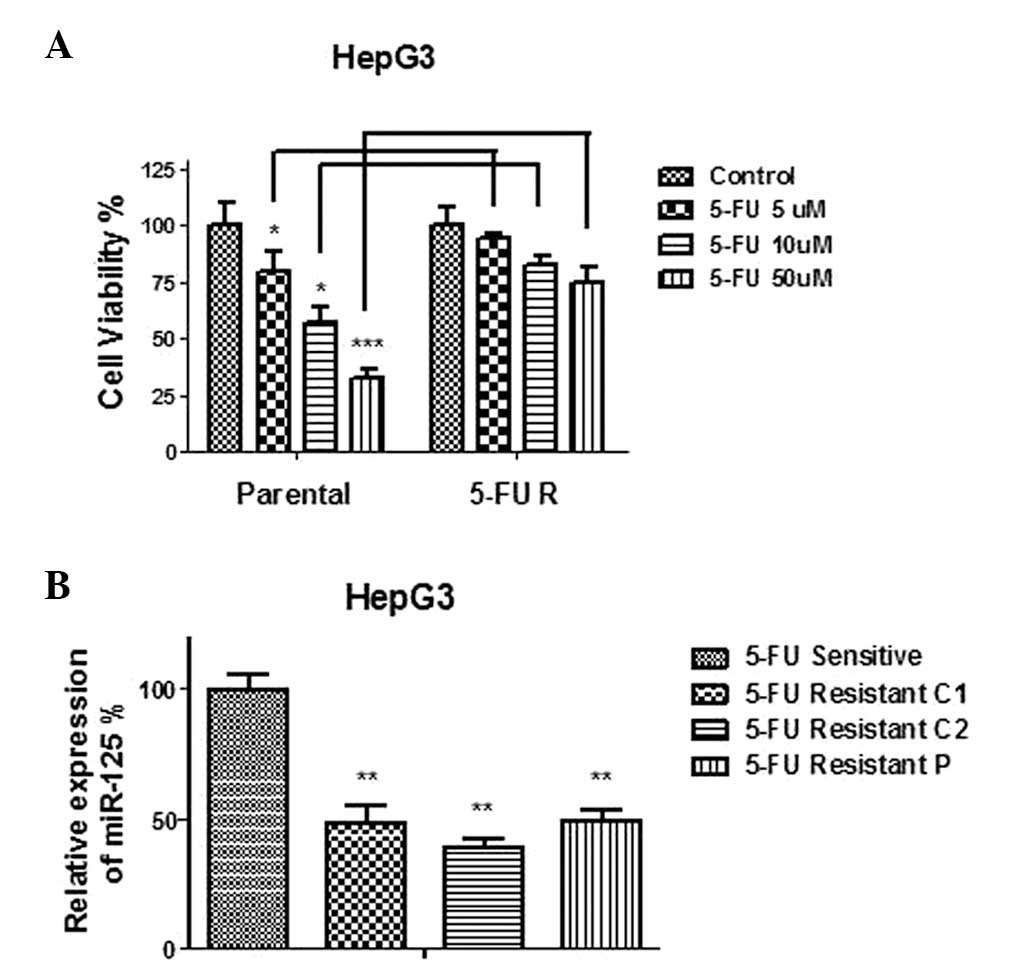
As discussed above, chemotherapy resistance is a
major obstacle that limits the effectiveness of clinical management
of liver cancer. The dysregulated expression of tumor suppressor
genes negatively correlates with chemoresistance of cancer cells.
To investigate the function of miR-125b in liver cancer cell
chemosensitivity, a 5-FU-resistant cancer cell line was generated
using HepG3 cells by gradually treating the cells with elevated
concentrations of 5-FU. Two HepG3 5-FU-resistant colonies were
selected and the remainder were combined as a 5-FU-resistant pool.
Fig. 2A shows that the resistant
cells were significantly less sensitive to regular 5-FU treatments
compared with the parental cells. The parental cells exhibited a
IC50 at ~10 μM, while the viability of 5-FU-resistant
cells at 10 μM was significantly higher (P<0.05; Fig. 2A). The expression levels of
miR-125b were measured in the 5-FU-resistant clone1, clone2 and
pool cells. The results revealed that miR-125b was significantly
downregulated in all 5-FU-resistant clones (Fig. 2B), suggesting that miR-125b
functions as a tumor suppressor in liver cancer cells.
5-FU-resistant liver cancer cells exhibit
increased glucose metabolism
In accordance with the Warburg effect, evidence
supports the hypothesis that dysregulated cellular metabolism is
associated with drug resistance in cancer therapy. To analyze the
putative mechanisms for miR-125b-mediated sensitization to
chemotherapy in HCC cells, the glucose metabolism in 5-FU-resistant
and -sensitive cells was measured in the present study. Notably,
glucose metabolism, which is normally upregulated in chemoresistant
cancer cells, was markedly increased in 5-FU-resistant cells.
Lactate production (P<0.05; Fig.
3A) and glucose uptake (P<0.05; Fig. 3B) were significantly increased in
5-FU-resistant cells, indicating that dysregulated metabolism may
contribute to chemoresistance.
HK II is a direct target of miR-125b
To further elucidate the mechanism for
miR-125b-mediated sensitization to 5-FU in liver cancer cells,
potential targets of miR-125b were investigated by searching miRNA
databases. The three public miRNA databases (TargetScan, Pictar and
MicroRNA) all predicted HK II to be a target of miR-125b; the
3′-UTR of HK II contains a highly conserved binding site for
miR-125b (Fig. 4A). HKs catalyze
the first committed step in glucose metabolism and are important in
tumor initiation and maintenance. Thus far, to the best of our
knowledge, no publication has reported HK II to be a direct target
of miR-125b in HCC cells. To determine whether miR-125b affects HK
II in chondrosarcoma cells, pre-miR-125b and anti-miR-125b were
transfected into HepG3 cells. The overexpression of miR-125b
markedly downregulated HK II protein expression levels in
comparison with a control group, but inhibition of miR-125b
restored HK II expression levels in HepG3 cells. (Fig. 4B). To identify whether miR-125b
directly targets the 3′-UTR of HK II mRNA, luciferase reporter
analysis was performed by co-transfecting the cells with a vector
containing pMIR reporter-luciferase fused with the original
sequence or a predicted binding site mutant of the 3′-UTR of HK II
mRNA, and pre-miR-100 or control microRNA. Overexpression of
miR-125b significantly reduced the luciferase activity of the
reporter with wild-type HK II 3′-UTR by ~60% in HepG3 cells
(P<0.01; Fig. 4C). No
inhibitory effects of miR-125b on the activity of the reporter with
binding site mutant HK II 3′-UTR were detected (Fig. 4C). In conclusion, these results
demonstrated that HK II is a direct target of miR-125b in HCC
cells.
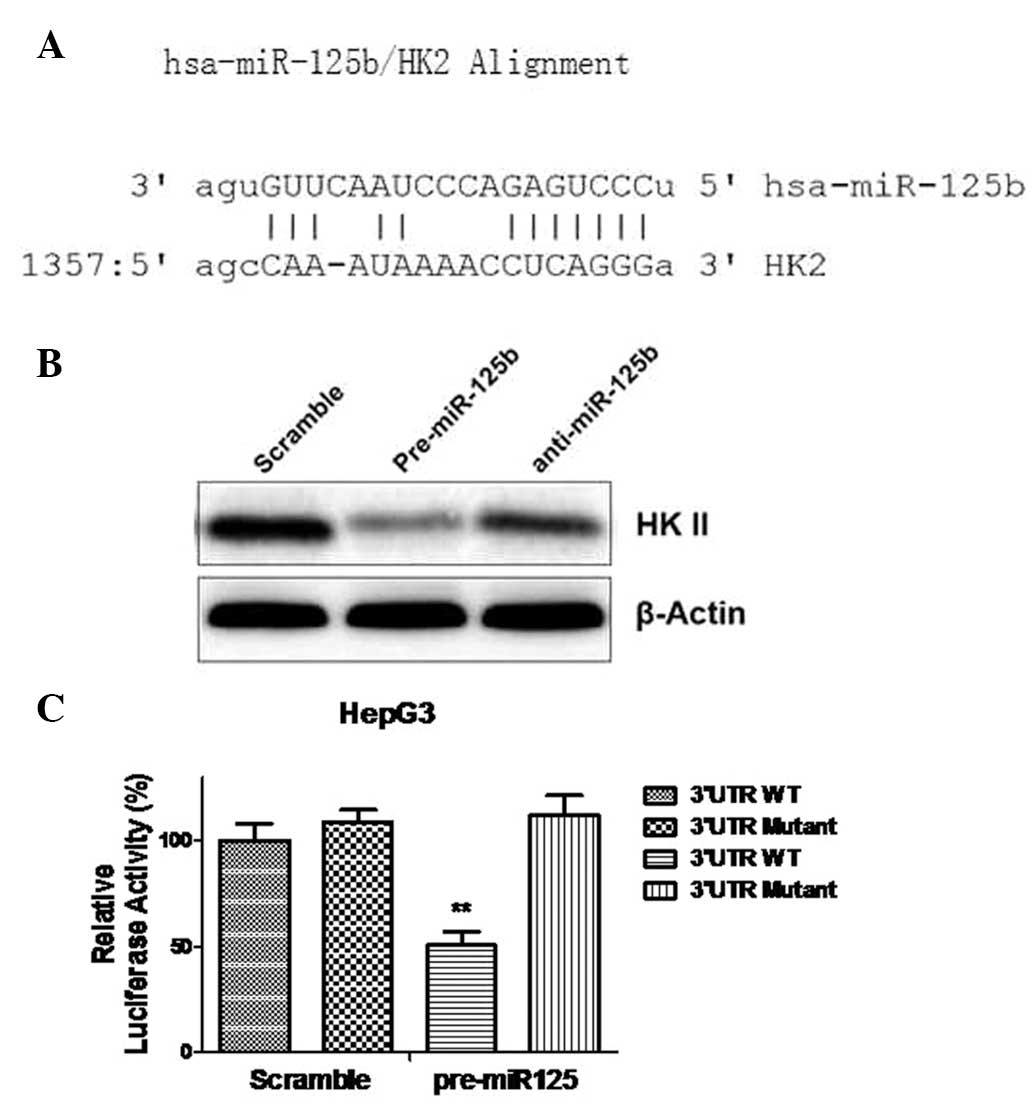 | Figure 4HK II is a direct target of miR-125b
in hepatocellular carcinoma cells. (A) Target prediction with
Targetscan.org: The 3′UTR of HK II contains putative binding sites
for miR-125b. (B) HepG3 cells were transfected with 200 nM
pre-miR-negative (Scramble), pre-miR-125b or anti-miR-125b for 48
h. Cell lysates were prepared for Western blot analysis. β-actin
served as a loading control. (C) HepG3 cells were co-transfected
with luciferase reporter plasmids with WT HK II 3′-UTR or mutant HK
II 3′-UTR, and pre-miR-125b or pre-miR-negative (Scramble) using
Lipofectamine 2000 reagent. At 48 h post-transfection, the cells
were harvested and lysed with passive lysis buffer. Luciferase
activities were measured by dual luciferase reporter assay. The
pRL-TK vector served as an internal control. The results were
expressed as relative luciferase activity (firefly
luciferase/renilla luciferase). Columns, mean of three independent
experiments; bars, standard error of the mean.
**P<0.01 vs. control. HK, hexokinase; hsa, Homo
sapiens; UTR, untranslated region; WT, wild-type; miR,
microRNA. |
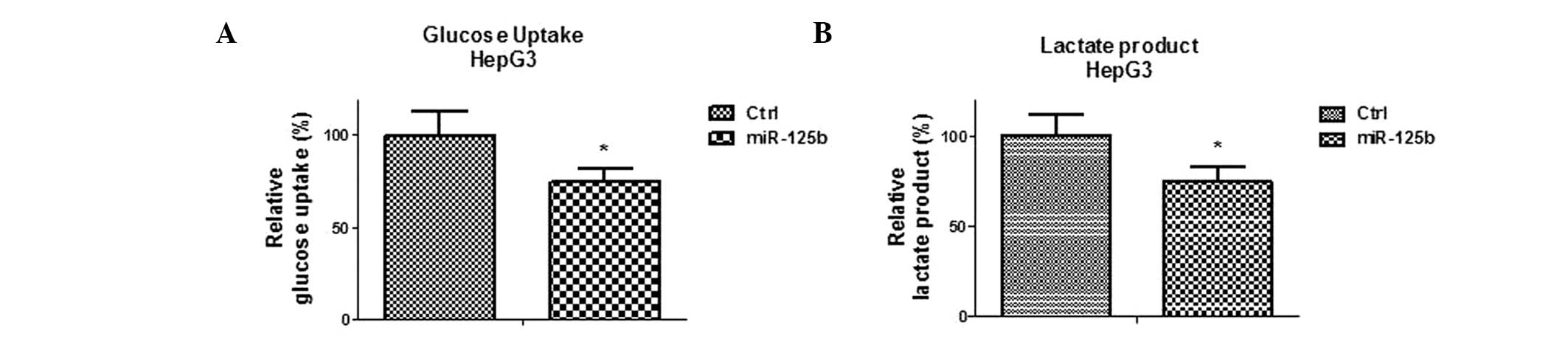
Overexpression of miR-125b in liver
cancer cells inhibits glucose metabolism
The above results revealed that HK II is a direct
target of miR-125b. HK II has been established as essential in
catalyzing the first committed step in glucose metabolism. To
identify whether miR-125b regulates glucose metabolism in liver
cancer cells, glucose uptake and lactate production in
miR-125b-overexpressing HepG3 cells were compared with those in
cells transfected with control microRNA. The data revealed
significant inhibitions of glucose uptake and lactate production in
the miR-125b-overexpressing cells (P<0.05; Fig. 5A and B), indicating that miR-125b
may sensitize liver cancer cells to 5-FU through downregulation of
glucose metabolism. In conclusion, the results in Figs. 3 and 5 revealed a correlation between
miR-125b-mediated suppression of glucose metabolism and 5-FU
sensitivity.
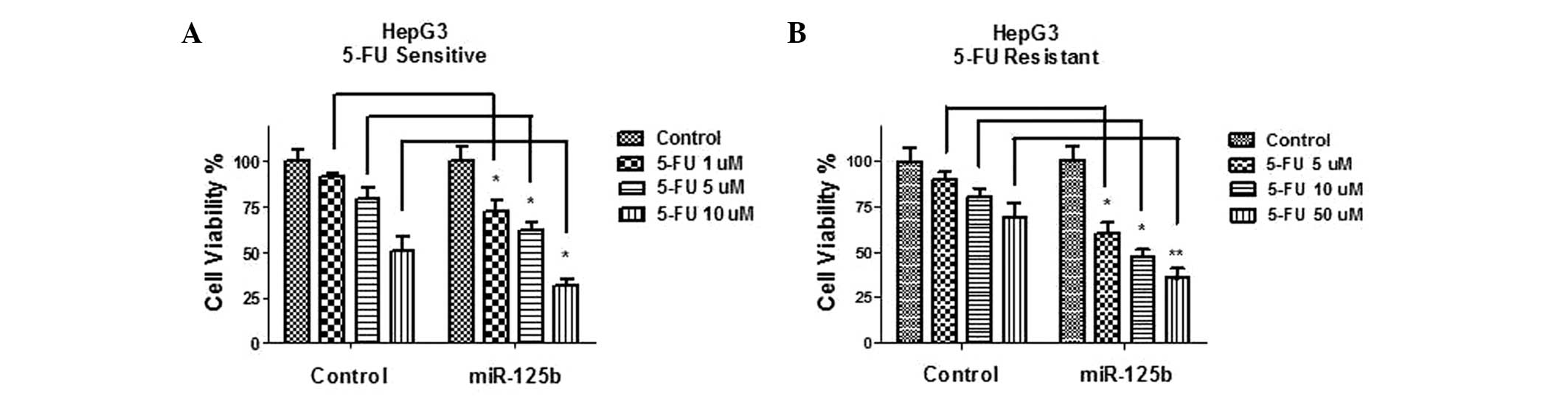
Overexpression of miR-125b resensitizes
5-FU-resistant HCC cells through the inhibition of glucose
metabolism
To determine the mechanisms accounting for
miR-125b-mediated 5-FU sensitivity, miR-125b and control microRNA
were transfected into HepG3 5-FU-sensitive and -resistant cells,
and the cells were then treated with the indicated concentrations
of 5-FU for 48 h. The effect of miR-125b overexpression on cell
viability was examined following 5-FU administration. Treatment
with 5-FU resulted in a marginal inhibition of cell viability in
the control 5-FU-sensitive and -resistant HepG3 cells (Fig. 6A and B). However, when miR-125b was
overexpressed in 5-FU-sensitive and -resistant liver cancer cells,
the susceptibility of the cells to 5-FU was significantly increased
(Fig. 6A and B), suggesting that
miR-125 is involved in the development of 5-FU resistance. To
examine whether sensitization to 5-FU by miR-125b occurred through
inhibition of the glucose metabolism, an overexpression vector
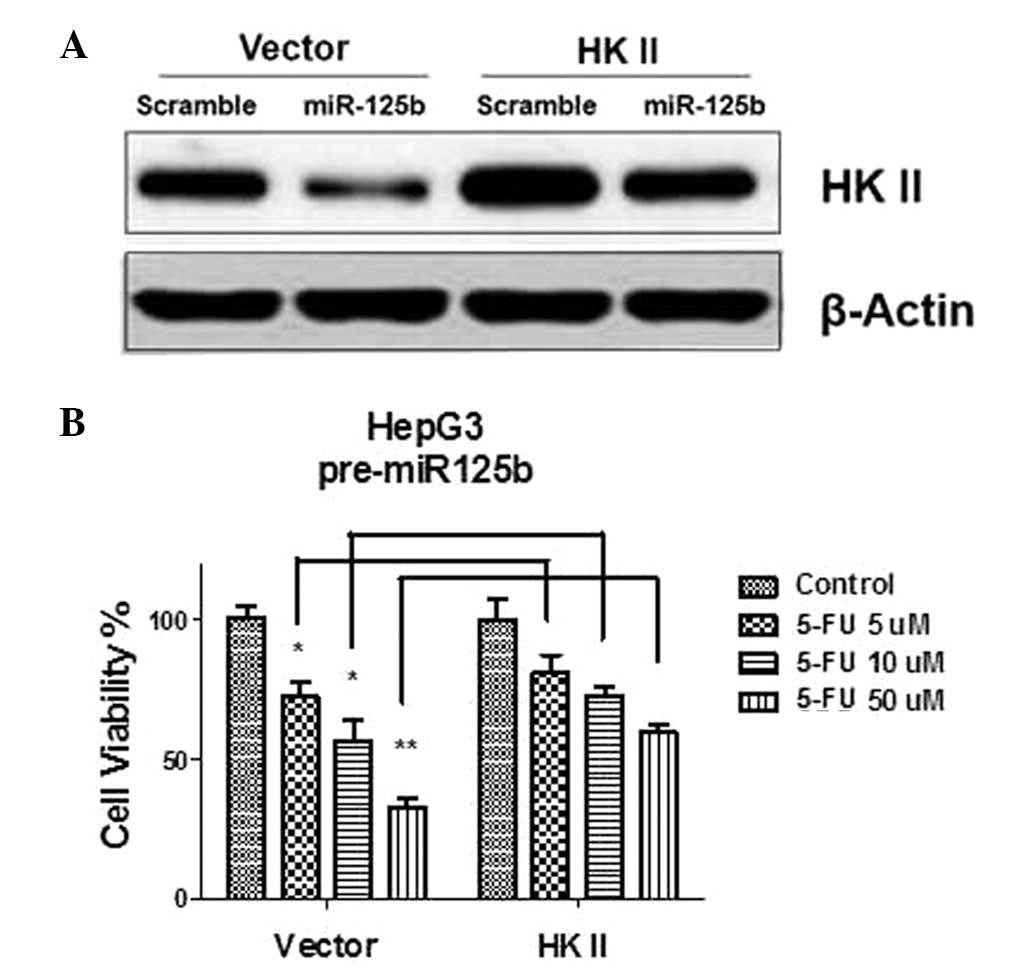
containing wild-type HK II was transfected into miR-125
pre-transfected HepG3 cells. Exogenous overexpression of HK II in
miR-125 pre-transfected cells restored the expression levels of HK
II to non-transfection levels (Fig.
7A). The sensitivity of the transfected HepG3 cells to 5-FU was
then measured (Fig. 7B). The
results demonstrated that exogenous upregulation of the glucose
metabolism by overexpression of HK II rendered miR-125b
overexpressing HepG3 cells significantly more resistant to 5-FU in
comparison to cells not transfected with HK II. In conclusion, the
results suggested that the overexpression of miR-125b sensitized
HCC cells to 5-FU through inhibition of HK II.
Discussion
The ‘Warburg effect’ describes the unique metabolic
phenomenon where tumor cells preferentially utilize the markedly
less efficient glycolytic process for energy production, even in
the presence of oxygen (17,18).
A high rate of glycolysis in cancer has been observed, occurring
through upregulation of the key enzymes HK II, glucose transporter
1, pyruvate kinase M2 isoform (PKM2), pyruvate dehydrogenase kinase
1 and LDHA (19). The ability to
reduce the metabolic rate in cancer cells has been investigated as
one mechanism of cancer suppressors. Evidence supports the
hypothesis that dysregulated cellular metabolism is associated with
drug resistance in cancer therapy (19). Researchers have attempted to use
the unique bioenergetic properties of cancer cells to enhance the
therapeutic efficacy of cytotoxic chemotherapy. In the glycolytic
pathway, LDHA has been shown to contribute to
paclitaxel/trastuzumab resistance in breast cancer (20), and inhibition of HK II was
demonstrated to restore drug sensitivity in doxorubicin-resistant
colon cancer cells (21).
Therefore, targeting cancer cell metabolism with glycolysis
inhibitors administered in combination with conventional
chemotherapeutic agents may provide a synergistic inhibitory effect
in the treatment of 5-FU-resistant liver cancer cells.
miRNAs have been identified as exhibiting oncogenic
and tumor suppressor-like functions in the processes of cellular
proliferation, differentiation and apoptosis. microRNAs, including
miR-221 (26), miR-222 (27) and miR-106b (28), have previously been reported to
function as oncogenes. Conversely, the majority of downregulated
microRNAs, including miR-16 (29),
miR-100 (30), miR-29a/b (31) and miR-146a/b (32), have been reported to act as tumor
suppressors. Increasing evidence has demonstrated that miR-125b is
crucial in diverse cellular processes and numerous carcinomas.
However, little is known regarding the role of miRNAs in
chemoresistance to drugs such as 5-FU. In the present study, the
differential expression profiles of miR-125b in normal human liver
cells and human HCC cells were examined. miR-125b was found to be
downregulated in liver cancer cells and overexpression of miR-125b
significantly suppressed liver cancer cell growth, supporting the
conclusion that miR-125b acts as a tumor suppressor in human HCC
cells.
Thus far, several targets of miR-125b in cancers
have been identified. The ErbB2 and ErbB3 oncogenes were reported
to be targets of miR-125b in breast cancer cells (11). Other studies have shown that the
tumor suppressor p53 is a direct target of miR-125b, and
suppression of p53 gene expression by overexpression of miR-125b
inhibits p53-dependent apoptosis in human neuroblastoma cells and
human lung fibroblast cells (33).
Furthermore, miR-125b has been reported to reduce the expression
levels of the MUC1 oncoprotein, whose silencing promotes DNA
damage-induced apoptosis in breast cancer cells (34).
HK II catalyzes the first irreversible step of the
glycolytic pathway, where glucose is phosphorylated to
glucose-6-phosphate with consumption of adenosine triphosphate. In
addition to miR-125b, cancer glycolysis has also been reported to
be regulated by miR-143, via the targeting of HK II (35). Furthermore, PKM2 is a target of the
tumor-suppressive microRNA-326 and regulates the survival of glioma
cells (36), demonstrating that
targeting metabolic enzymes by microRNA results in downregulation
of cancer cell metabolic processes. 5-FU is a widely used
chemotherapeutic agent for the treatment of several types of
cancer. 5-FU resistance may result in subsequent recurrence and
metastasis of cancer, ultimately causing cancer-associated
mortality. Although extensive investigations have been conducted
with regard to 5-FU resistance in cancer cells, the specific
mechanisms involved remain poorly understood. In the present study,
HK II was identified as a direct target of miR-125b in HCC cells.
Inhibition of HK II by overexpression of miR-125b resulted in
suppression of the glucose metabolism pathway, which sensitized
liver cancer cells to 5-FU. Restoration of HK II by exogenous
transfection of HK II rendered liver cancer cells resistant to
5-FU, indicating that overexpression of miR-125b sensitizes
5-FU-resistant HCC cells through the inhibition of HK II.
In conclusion, the role of miR-125b in acquired 5-FU
resistance was investigated in multiple human HCC cell lines.
Compared with 5-FU-sensitive cells, 5-FU-resistant cells exhibited
reduced expression levels of miR-125b, and transfection of
pre-miR-125b into liver cancer cells resulted in an increased
sensitivity of 5-FU-resistant cells to 5-FU. Since drug resistance
is a phenotype of malignant cancer cells, the finding that miR-125b
expression levels are negatively correlated with 5-FU resistance in
HCC cells is consistent with the reported functions of miR-125b. In
addition, 5-FU-resistant cells exhibited higher glucose metabolic
activity than 5-FU-sensitive cells, and miR-125 was identified to
downregulate glucose metabolism by directly targeting HK II. These
results identified miR-125b as a tumor suppressor-like microRNA,
which has great potential as a diagnostic and prognostic
biomarker.
Acknowledgements
The authors would like to thank Dr Chao Yu at the
Department of Hepatobiliary Surgery, Affiliated Hospital of Guiyang
Medical College for editorial assistance. This study received
support from the Chinese National Natural Science Foundation (grant
no. 81160311) and the Chinese Postdoctoral Foundation (grant no.
2013-52-134523).
References
|
1
|
Ameres SL and Zamore PD: Diversifying
microRNA sequence and function. Nat Rev Mol Cell Biol. 14:475–488.
2013. View
Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bushati N and Cohen SM: microRNA
functions. Annu Rev Cell Dev Biol. 23:175–205. 2007. View Article : Google Scholar
|
|
3
|
Di Leva G, Garofalo M and Croce CM:
MicroRNAs in Cancer. Annu Rev Pathol. 9:287–314. 2014.
|
|
4
|
Shenouda SK and Alahari SK: MicroRNA
function in cancer: oncogene or a tumor suppressor? Cancer
Metastasis Rev. 28:369–378. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhang B, Pan X, Cobb GP and Anderson TA:
microRNAs as oncogenes and tumor suppressors. Dev Biol. 302:1–12.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wang D, Qiu C, Zhang H, Wang J, Cui Q and
Yin Y: Human microRNA oncogenes and tumor suppressors show
significantly different biological patterns: from functions to
targets. PLoS One. 5:2010.
|
|
7
|
Hong L, Yang Z, Ma J and Fan D: Function
of miRNA in controlling drug resistance of human cancers. Curr Drug
Targets. 14:1118–1127. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sun YM, Lin KY and Chen YQ: Diverse
functions of miR-125 family in different cell contexts. J Hematol
Oncol. 6:62013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Guan Y, Yao H, Zheng Z, Qiu G and Sun K:
MiR-125b targets BCL3 and suppresses ovarian cancer proliferation.
Int J Cancer. 128:2274–2283. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Huang L, Luo J, Cai Q, et al:
MicroRNA-125b suppresses the development of bladder cancer by
targeting E2F3. Int J Cancer. 128:1758–1769. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Scott GK, Goga A, Bhaumik D, Berger CE,
Sullivan CS and Benz CC: Coordinate suppression of ERBB2 and ERBB3
by enforced expression of micro-RNA miR-125a or miR-125b. J Biol
Chem. 282:1479–1486. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Liang L, Wong CM, Ying Q, et al:
MicroRNA-125b suppressesed human liver cancer cell proliferation
and metastasis by directly targeting oncogene LIN28B2. Hepatology.
52:1731–1740. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kappelmann M, Kuphal S, Meister G,
Vardimon L and Bosserhoff AK: MicroRNA miR-125b controls melanoma
progression by direct regulation of c-Jun protein expression.
Oncogene. 32:2984–2991. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Liu LH, Li H, Li JP, et al: miR-125b
suppresses the proliferation and migration of osteosarcoma cells
through down-regulation of STAT3. Biochem Biophys Res Commun.
416:31–38. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bloomston M, Frankel WL, Petrocca F, et
al: MicroRNA expression patterns to differentiate pancreatic
adenocarcinoma from normal pancreas and chronic pancreatitis. JAMA.
297:1901–1908. 2007. View Article : Google Scholar
|
|
16
|
Shi XB, Xue L, Yang J, et al: An
androgen-regulated miRNA suppresses Bak1 expression and induces
androgen-independent growth of prostate cancer cells. Proc Natl
Acad Sci USA. 104:19983–19988. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Vander Heiden M, Cantley LC and Thompson
CB: Understanding the Warburg effect: the metabolic requirements of
cell proliferation. Science. 324:1029–1033. 2009.PubMed/NCBI
|
|
18
|
Najafov A and Alessi DR: Uncoupling the
Warburg effect from cancer. Proc Natl Acad Sci USA.
107:19135–19136. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhao Y, Butler EB and Tan M: Targeting
cellular metabolism to improve cancer therapeutics. Cell Death Dis.
4:e5322013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhou M, Zhao Y, Ding Y, et al: Warburg
effect in chemosensitivity: targeting lactate dehydrogenase-A
re-sensitizes taxol-resistant cancer cells to taxol. Mol Cancer.
9:332010. View Article : Google Scholar
|
|
21
|
Nakano A, Tsuji D, Miki H, et al:
Glycolysis inhibition inactivates ABC transporters to restore drug
sensitivity in malignant cells. PLoS One. 6:e272222011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Tong J, Xie G, He J, Li J, Pan F and Liang
H: Synergistic antitumor effect of dichloroacetate in combination
with 5-fluorouracil in colorectal cancer. J Biomed Biotechnol.
2011:7405642011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Singh S, Singh PP, Roberts LR and Sanchez
W: Chemopreventive strategies in hepatocellular carcinoma. Nat Rev
Gastroenterol Hepatol. 11:45–54. 2014. View Article : Google Scholar
|
|
24
|
Abdel-Hamid NM and Morsy MA: Novel
biochemical pathways for 5-Fluorouracil in managing experimental
hepatocellular carcinoma in rats. J Membr Biol. 234:29–34. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yoshida H, Onda M, Tajiri T, et al:
Hepatocellular carcinoma responding to chemotherapy with 5-FU.
Hepatogastroenterology. 47:1120–1121. 2000.PubMed/NCBI
|
|
26
|
Lupini L, Bassi C, Ferracin M, et al:
miR-221 affects multiple cancer pathways by modulating the level of
hundreds messenger RNAs. Front Genet. 4:642013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Visone R, Russo L, Pallante P, et al:
MicroRNAs (miR)-221 and miR-222, both overexpressed in human
thyroid papillary carcinomas, regulate p27Kip1 protein levels and
cell cycle. Endocr Relat Cancer. 14:791–798. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ivanovska I, Ball AS, Diaz RL, et al:
MicroRNAs in the miR-106b family regulate p21/CDKN1A and promote
cell cycle progression. Mol Cell Biol. 28:2167–2174. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhou R, Li X, Hu G, Gong AY, Drescher KM
and Chen XM: miR-16 targets transcriptional corepressor SMRT and
modulates NF-kappaB-regulated transactivation of interleukin-8
gene. PLoS One. 7:e307722012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Henson BJ, Bhattacharjee S, O’Dee DM,
Feingold E and Gollin SM: Decreased expression of miR-125b and
miR-100 in oral cancer cells contributes to malignancy. Genes
Chromosomes Cancer. 48:569–582. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Balkhi MY, Iwenofu OH, Bakkar N, et al:
miR-29 acts as a decoy in sarcomas to protect the tumor suppressor
A20 mRNA from degradation by HuR. Sci Signal. 6:ra632013.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Li Y, VandenBoom TG II, Wang Z, et al:
miR-146a suppresses invasion of pancreatic cancer cells. Cancer
Res. 70:1486–1495. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Le MT, Teh C, Shyh-Chang N, et al:
MicroRNA-125b is a novel negative regulator of p53. Genes Dev.
23:862–876. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Rajabi H, Jin C, Ahmad R, McClary C, Joshi
MD and Kufe D: Mucin 1 Oncoprotein Expression Is Suppressed by the
miR-125b Oncomir. Genes Cancer. 1:62–68. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Peschiaroli A, Giacobbe A, Formosa A, et
al: miR-143 regulates hexokinase 2 expression in cancer cells.
Oncogene. 32:797–802. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kefas B, Comeau L, Erdle N, Montgomery E,
Amos S and Purow B: Pyruvate kinase M2 is a target of the
tumor-suppressive microRNA-326 and regulates the survival of glioma
cells. Neuro Oncol. 12:1102–1112. 2010. View Article : Google Scholar : PubMed/NCBI
|