Introduction
Osteosarcoma (OS) is one of the most common types of
primary malignant bone tumor in childhood and adolescence (1). Despite the introduction of effective
chemotherapy and improvements in surgical technology, which has
increased the survival rate to approximately 65–75%, pulmonary
metastasis occurs in 40–50% of OS patients and remains the leading
cause of mortality (2–3). Therefore, it is important to uncover
the molecular mechanisms involved in OS progression, particularly
pulmonary metastasis.
Mechanistically, OS development, as with other types
of cancer, involves multiple genetic alterations, including
oncogene activation and tumor-suppressor gene dysfunction. Over the
previous several decades, substantial knowledge has been gained
regarding the molecular alterations associated with OS
carcinogenesis and several key signaling pathways, including fatty
acid synthase (FASN)/human epidermal growth factor receptor
2/phosphatidylinositol 3-kinase (PI3K)/Akt (5), extracellular signal-regulated
kinases-p38-JNK (6), nuclear
factor-κB (7) and the
mitogen-activated protein kinase pathway (8) have been implicated in OS metastasis.
Dysregulation of the genes associated with these pathways has been
demonstrated to promote OS cell growth and metastasis.
PI3Ks are a family of related intracellular signal
transducer enzymes, which have the capability to phosphorylate the
hydroxyl group at position 3 of the inositol ring of
phosphatidylinositol. PI3Ks are important in tumor cell
differentiation, the cell cycle, apoptosis, growth and metastasis
via the activation of Akt. Various studies have demonstrated that
decreasing the activation of the PI3K/Akt signaling pathway
inhibits cell growth and metastasis in various types of tumor
(9,10). Several small molecule inhibitors of
the PI3K/Akt signaling pathway have been previously developed as
promising antitumor treatments (11–14).
Previous studies have demonstrated that inhibition of PI3K/Akt
significantly suppresses OS cell growth, migration and invasion
(5,15,16).
Therefore, we hypothesized that small molecule inhibitors of the
PI3K/Akt signaling pathway may be novel alternative agents for the
treatment of OS.
The present study investigated the inhibition of the
malignant phenotype of U2-OS and MG-63 cells by
2-(4-morpholinyl)-8-phenyl-chromone (LY294002), a commonly used
pharmacological inhibitor that selectively inhibits the PI3K-Akt
pathway, via downregulation of the PI3K/Akt/FASN signaling
pathway.
Materials and methods
Cell lines and cell culture
The human OS cell lines, U2-OS and MG-63, were
obtained from the American Type Culture Collection (Manassas, VA,
USA) and cultured in Dulbecco’s modified Eagle’s medium (DMEM;
HyClone, Waltham, MA, USA) supplemented with 10% fetal bovine serum
(FBS; Sigma Aldrich, St. Louis, MO, USA) in a humidified atmosphere
of 5% CO2 at 37°C.
Cell growth assay
MG-63 and U2-OS cell lines were cultured in 96-well
tissue culture plates at a density of 5,000 cells/well in minimum
essential medium (Solarbio, Beijing, China) containing 10% FBS and
2 mM l-glutamine. Following attachment overnight, the medium was
replaced and cells were incubated with increasing concentrations
(5, 10, 20, 40, 80 and 160 μM) of LY294002 (Sigma Aldrich).
Following treatment for 48 h, MTT assays were performed at a
wavelength of 490 nm in triplicate. The inhibition ratio was
calculated and the concentration-viability curves were fitted by
the OriginPro 7.5 program (OriginLab, Northampton, MA, USA). The
half maximal inhibitory concentration (IC50) values of
LY294002 were determined. All experiments were performed in
triplicate.
Fluorescence-activated cell sorting
(FACS)
MG-63 and U2-OS cells in the exponential growth
phase were treated with varying concentrations of LY294002 for 24
h. Cells were then fixed with 70% ethanol and stained with
propidium iodide. FACS analysis was performed to determine the
percentage of apoptotic cells and cell cycle distribution by using
the EPICS XL flow cytometer (Beckman Coulter, Miami, FL, USA) and
System II software V4.16 (Coulter Electronics, Miami, FL, USA). All
experiments were performed in triplicate.
Invasion assay
The invasiveness of the OS cells was measured using
BD BioCoat™ BD Matrigel™ Invasion Chambers (BD Bioscience, Franklin
Lakes, NJ, USA) according to the manufacturer’s instructions. The
medium in the lower chamber contained 15% FBS as a source of
chemoattractant. The cells were suspended in serum-free medium
containing LY294002 (20 and 40 μM) and added to the upper chambers
simultaneously (2×103 cells in 0.1 ml). The cells that
passed through the Matrigel-coated membrane were stained with
Diff-Quik (Sysmex, Kobe, Japan) and images were captured. Cell
invasion was quantified by direct microscopic visualization and
cell counting. Invading cells were counted from five randomly
selected fields under an inverted microscope. The cells not treated
with LY294002 were used as a control. Three independent experiments
were performed.
Migration assay
Cell migration was assessed by determining the
ability of the cells to move into a cellular space in a
two-dimensional wound healing assay in vitro. In brief, the
cells were cultured in six-well tissue culture plastic dishes at a
density of 5×106 cells/well and then subsequently
treated with LY294002 (20 and 40 μM ) for 24 h. The cells were
denuded by dragging a rubber policeman (Fisher Scientific, Hampton,
NH, USA) through the center of the plate well. The culture plates
were rinsed with phosphate-buffered saline (PBS) and fresh
quiescent medium alone or with 10% bovine serum albumin was added.
The cells were subsequently incubated at 37°C for 24 h. Images of
the cells were captured at 0 and 24 h and the migrated distance was
measured. The migration rate was counted from five randomly
selected fields under an inverted microscope (Olympus, Tokyo,
Japan). The cells not treated with LY294002 were used as a control.
Three independent experiments were performed.
Western blot analysis
U2-OS and MG-63 cells in the exponential growth
phase were treated with LY294002 (20, 40, 80 and 160 μM) for 24 h.
The cells were subsequently washed with cold PBS. Total cellular
protein was extracted using radioimmunoprecipitation assay lysis
buffer containing 60 μg/ml phenylmethanesulfonyl fluoride and the
protein concentration was determined using a Bradford protein
assay. Equal quantities of protein were electrophoresed by 10%
SDS-PAGE (Solarbio) and transferred onto a pure nitrocellulose
blotting membrane (0.22 μm pores; Solarbio). The membranes were
blocked with 5% Difco skimmed milk for 1 h at room temperature and
then incubated with primary antibodies (monoclonal rabbit anti-Akt,
anti-p-Akt and anti-FASN IgG; 1:2,000; Santa Cruz Biotechnology,
Inc., Santa Cruz, CA, USA) overnight at 4°C. The membranes were
washed prior to incubation with the appropriate
peroxidase-conjugated secondary antibodies (monoclonal mouse
anti-rabbit; 1:5,000; Santa Cruz Biotechnology, Inc.). The immune
complexes were detected with a Pro-light HRP kit (Tiangen Biotech
Co., Ltd., Beijing, China). All experiments were performed in
triplicate.
Reverse transcription quantitative
polymerase chain reaction (RT-qPCR)
Semi-quantitative PCR was used to detect FASN mRNA
levels. Total RNA was extracted from cells using TRIzol reagent
(Invitrogen Life Technologies, Carlsbad, CA, USA). The total RNA
concentration was determined by spectrophotometry at 260 nm and the
purity was determined by calculating the 260/280 ratio with a
BioPhotometer (Eppendorf, Hamburg, Germany). The Two-Step kit
(Promega Corporation, Madison, WI, USA) was used to obtain cDNA
according to the manufacturer’s instructions, which was
subsequently used as the template for amplification. The following
primers were used to amplify target sequences: FASN, forward
5′-AACTCCATGTTTGGTGTTTG-3′ and reverse 5′-CACATGCGGTTTAATTGTG-3′,
171 bp (Sangon Biotech Shanghai Co., Ltd., Shanghai, China); GAPDH,
forward 5′-CAGGGCTGCTTTTAACTCTGGT-3′ and reverse
5′-GATTTTGGAGGGATCTCGCT-3′, 199 bp (Sangon Biotech Shanghai Co.,
Ltd.). Following amplification, DNA electrophoresis was performed
on standard 1% agarose gels and DNA was labeled and visualized
using ethidium bromide. Images were captured using the Canon
Digital IXUS 900Ti (Canon Inc., Tokyo, Japan). All experiments were
repeated six times.
Results
LY294002 inhibits OS cell growth
The inhibitory effect of LY294002 on the
growth of U2-OS and MG-63 cell lines was examined using an MTT
assay. The results revealed that MG-63 and U2-OS cell lines were
sensitive to LY294002 and that LY294002 inhibits U2-OS and MG-63
cell proliferation in a time- and dose-dependent manner (Fig. 1). IC50 values at 24 h
were 62.98 and 55.2 μM, respectively.
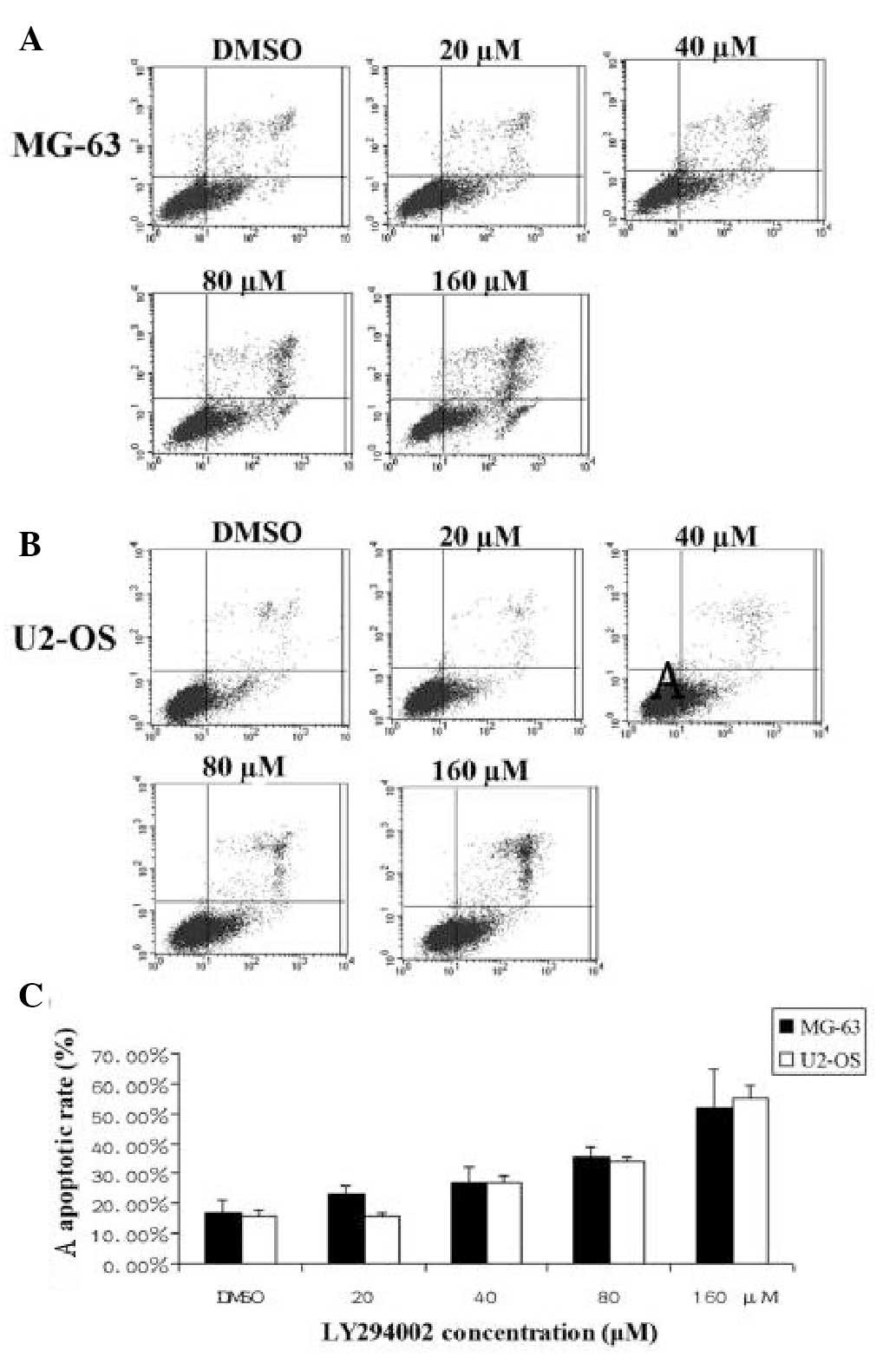
LY294002 induces OS cell apoptosis
FACS analysis was used to examine the mechanism of
LY294002 inhibition of OS cell apoptosis. Gradient concentrations
(20, 40, 80 and 160 μM) of LY294002 were added to U2-OS and MG-63
cell cultures in the exponential growth phase. The treated and
untreated cell samples were obtained and fixed for FACS analysis 24
h later. FACS analysis revealed that LY294002-induced apoptosis of
U2-OS and MG-63 cells increased with increasing concentration of
the inhibitor (Fig. 2). The data
indicated that LY294002 induced OS apoptosis in a dose-dependent
manner.
LY294002 inhibits OS cell invasion
Transwell invasion assays were used to examine the
effect of LY294002 on U2-OS and MG-63 cell invasion. The results
demonstrated that the proportion of invading cells of these cell
samples treated with LY294002 was significantly lower than that of
the untreated cells and the inhibitory effect of LY294002 on cell
invasion was increased with increasing concentration of the
inhibitor (Fig. 3A and B). The
data indicated that LY294002 inhibits OS cell invasion in
vitro.
LY294002 decreases OS cell migration
The effect of LY294002 on the migration of U2-OS and
MG-63 cells was evaluated using wound healing assays. The results
revealed that the migration rate of cells treated with LY294002 was
significantly lower when compared with that of the untreated cells
and the migration rate decreased with increasing concentration of
LY294002 (Fig. 3C and D). The
results suggested that LY294002 inhibits OS cell migration.
LY294002 downregulates the activation of
the PI3K/Akt/FASN pathway
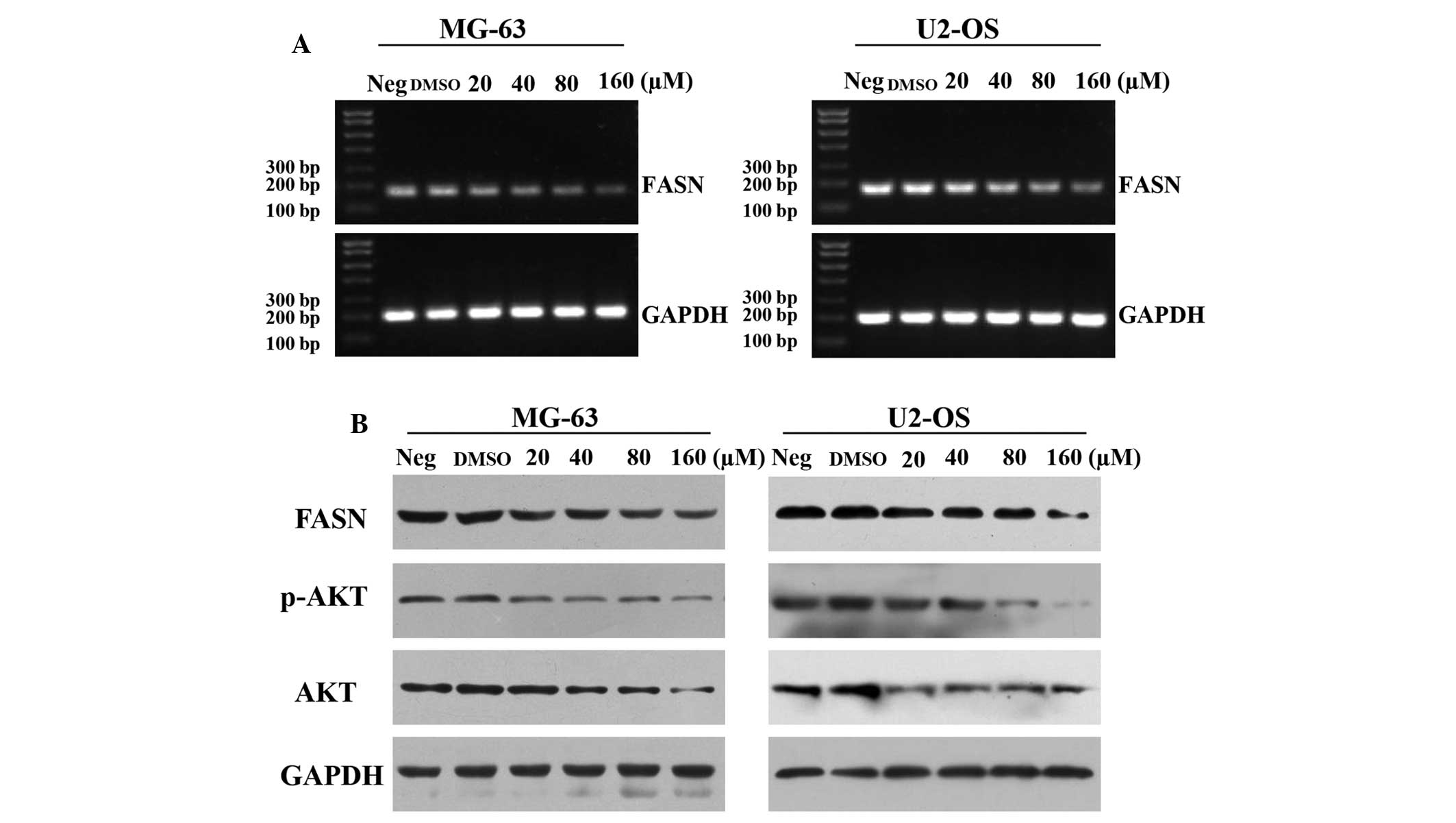
To investigate the potential molecular mechanisms,
RT-qPCR was performed to detect the expression of FASN mRNA in
U2-OS and MG-63 cell lines to evaluate the inhibitory effect of
LY294002 on FASN expression. The results revealed that the mRNA
expression of FASN was significantly downregulated with increasing
concentrations of LY294002 (Fig.
4A). Furthermore, the expression levels of p-Akt, Akt and FASN
protein in U2-OS and MG-63 cell lines were measured using western
blot analysis. The results revealed that p-Akt and FASN protein
expression levels in cells treated with LY294002 were significantly
lower compared with untreated cells (Fig. 4B). The present findings indicated
that LY294002 downregulates the activation of the PI3K/Akt/FASN
pathway.
Discussion
A significant number of studies on OS were published
during the 1950s and 1960s, revealing poor 5-year survival rates of
<15% (17–19). Following the discovery of effective
chemotherapy, the 5-year survival rates for patients treated with
intensive multi-drug chemotherapy and aggressive local control have
been reported to be 55–80% (20–22).
However, chemotherapy fails to eliminate all OS cells due to
intrinsic or acquired drug resistance, which is the most common
cause of tumor recurrence and resultant poor clinical outcomes
(23). Therefore, there is an
urgent requirement to develop new drugs with improved
chemotherapeutic effects for the management of OS.
LY294002, a selective inhibitor of PI3K, is the
first artificially synthesized small molecular inhibitor of
PI3Kα/β/δ, which acts on the adenosine triphosphate-binding site of
the enzyme and is also more stable than wortmannin in solution.
LY294002 inhibits cell proliferation and induces apoptosis by
downregulating the activation of AKT/PKB. Semba et al
demonstrated that LY294002 inhibits cell growth and induces
apoptosis by decreasing the phosphorylation of Akt (Ser473) in
colon cancer cell lines (24).
Previous studies revealed that LY294002 inhibits cell growth and
induces apoptosis in various types of cancer (25,26).
However, the mechanism of the anti-OS effect of LY294002 remains to
be elucidated. In the present study, the results demonstrated that
LY294002 induces MG-63 and U2-OS cell apoptosis and inhibits growth
in a dose-dependent manner. Furthermore, the inhibitory effect of
LY294002 on MG-63 and U2-OS cell migration and invasion was also
revealed. The present findings suggest that LY294002 inhibits the
malignant phenotype of OS cells in vitro and LY294002 may be
an alternative treatment option for the management of OS.
The role of the PI3K/Akt signaling pathway in OS
invasion and migration has been confirmed. Substantiated studies
demonstrate that LY294002 inhibits malignant phenotypes of cells in
various types of tumor via modulating the activity of the PI3K/Akt
signaling pathway (25,27). The molecular mechanisms associated
with LY294002 inhibition of the malignant phenotype of OS cells
were also analyzed. The current results demonstrated that the Akt
and p-Akt protein in MG-63 and U2-OS cells was inhibited by
LY294002 in a dose-dependent manner. It indicated that LY294002 may
modulate the activation of the PI3K/Akt signaling pathway in OS
cells.
FASN is an enzyme crucial for endogenous lipogenesis
in mammals as it is responsible for catalyzing the synthesis of
long-chain fatty acids. FASN is increased in a variety of human
tumors and has been demonstrated to be associated with tumor cell
growth, apoptosis and metastasis (28–31).
It has been previously demonstrated that inhibition of FASN through
use of pharmacological inhibitors or RNA interference significantly
inhibits OS cell growth, migration and invasion in vitro and
in vivo (32,33). Previous studies have demonstrated
that the activation of the PI3K/Akt pathway modulates FASN
expression in tumor cells (32,33).
It is therefore hypothesized that the inhibition of the PI3K/Akt
signaling pathway by LY294002 may modulate the expression of FASN,
resulting in inhibition of the malignant phenotype in OS cells. To
confirm these hypotheses, the inhibitory effect of LY294002 on the
expression of FASN mRNA and protein was investigated by RT-qPCR and
western blot analysis. The results revealed that the expression of
FASN was inhibited by LY294002 in a dose-dependent manner.
Taken together, the present findings indicated
LY294002 suppressed the malignant phenotype of OS cells by
downregulating the PI3K/Akt/FASN signaling pathway in vitro.
However, the tumor microenvironment is important in the malignant
phenotype of tumor cells. Further experiments are required to
elucidate whether LY294002 may be a new, alternative drug in
vivo and whether targeting the PI3K/Akt/FASN pathway may be a
potential treatment strategy for treating OS.
Acknowledgements
The present study was supported by grants from the
National Natural Science Foundation of China (grant no. 81260400),
the Natural Science Foundation of Jiangxi Province (grant no.
20114BAB205093) and the Jiangxi Provincial Department of Public
Health (grant no. 20143052).
References
|
1
|
Mirabello L, Troisi RJ and Savage SA:
Osteosarcoma incidence and survival rates from 1973 to 2004: data
from the Surveillance, Epidemiology, and End Results Program.
Cancer. 115:1531–1543. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bacci G, Briccoli A, Rocca M, et al:
Neoadjuvant chemotherapy for osteosarcoma of the extremities with
metastases at presentation: recent experience at the Rizzoli
Institute in 57 patients treated with cisplatin, doxorubicin, and a
high dose of methotrexate and ifosfamide. Ann Oncol. 14:1126–1134.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ta HT, Dass CR, Choong PF and Dunstan DE:
Osteosarcoma treatment: state of the art. Cancer metastasis Rev.
28:247–263. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kager L, Zoubek A, Potschger U, et al:
Primary metastatic osteosarcoma: presentation and outcome of
patients treated on neoadjuvant Cooperative Osteosarcoma Study
Group protocols. J Clin Oncol. 21:2011–2018. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wang TF, Wang H, Peng AF, et al:
Inhibition of fatty acid synthase suppresses U-2 OS cell invasion
and migration via downregulating the activity of HER2/PI3K/AKT
signaling pathway in vitro. Biochem Biophys Res Commun.
440:229–234. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chen HJ, Lin CM, Lee CY, et al: Kaempferol
suppresses cell metastasis via inhibition of the ERK-p38-JNK and
AP-1 signaling pathways in U-2 OS human osteosarcoma cells. Oncol
Rep. 30:925–932. 2013.PubMed/NCBI
|
|
7
|
Zhao Z, Wu MS, Zou C, et al:
Downregulation of MCT1 inhibits tumor growth, metastasis and
enhances chemotherapeutic efficacy in osteosarcoma through
regulation of the NF-kappaB pathway. Cancer Lett. 342:150–158.
2013. View Article : Google Scholar
|
|
8
|
Zhang S, Guo W, Ren TT, Lu XC, Tang GQ and
Zhao FL: Arsenic trioxide inhibits Ewing’s sarcoma cell
invasiveness by targeting p38 (MAPK) and c-Jun N-terminal kinase.
Anticancer Drugs. 23:108–118. 2012. View Article : Google Scholar
|
|
9
|
Liu R, Li X, Gao W, et al: Monoclonal
antibody against cell surface GRP78 as a novel agent in suppressing
PI3K/AKT signaling, tumor growth and metastasis. Clin Cancer Res.
19:6802–6811. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang YH, Dong YY, Wang WM, et al: Vascular
endothelial cells facilitated HCC invasion and metastasis through
the Akt and NF-kappaB pathways induced by paracrine cytokines. J
Exp Clin Cancer Res. 32:512013. View Article : Google Scholar
|
|
11
|
Zito CR, Jilaveanu LB, Anagnostou V, et
al: Multi-level targeting of the phosphatidylinositol-3-kinase
pathway in non-small cell lung cancer cells. PLoS One.
7:e313312012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Shukla S, Bhaskaran N, Babcook MA, Fu P,
Maclennan GT and Gupta S: Apigenin inhibits prostate cancer
progression in TRAMP mice via targeting PI3K/Akt/FoxO pathway.
Carcinogenesis. 35:452–460. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Li J, Cao B, Zhou S, et al:
Cyproheptadine-induced myeloma cell apoptosis is associated with
inhibition of the PI3K/AKT signaling. Eur J Haematol. 91:514–521.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Qin H, Du X, Zhang Y and Wang R:
Platycodin D, a triterpenoid saponin from Platycodon grandiflorum,
induces G2/M arrest and apoptosis in human hepatoma HepG2 cells by
modulating the PI3K/Akt pathway. Tumour Biol. 35:1267–1274. 2014.
View Article : Google Scholar
|
|
15
|
Liu ZL, Mao JH, Peng AF, et al: Inhibition
of fatty acid synthase suppresses osteosarcoma cell invasion and
migration via downregulation of the PI3K/Akt signaling pathway in
vitro. Mol Med Rep. 7:608–612. 2013.
|
|
16
|
Long XH, Zhang GM, Peng AF, Luo QF, Zhang
L, Wen HC, et al: Lapatinib alters the malignant phenotype of
osteosarcoma cells via downregulation of the activity of the
HER2-PI3K/AKT-FASN axis in vitro. Oncol Rep. Epub 2013/11/01.
2013.PubMed/NCBI
|
|
17
|
McKenna RJ, Schwinn CP and Higinbotham NL:
Osteogenic sarcoma in children. CA Cancer J Clin. 16:26–28. 1966.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Fraser GM and Aterman K: Osteogenic
Sarcoma in Young Children: A Case Report with Review of Literature.
Can Med Assoc J. 87:74–78. 1962.PubMed/NCBI
|
|
19
|
Pearson D and Steward JK: Malignant
disease in juveniles. Proc R Soc Med. 62:685–688. 1969.PubMed/NCBI
|
|
20
|
Iwamoto Y, Tanaka K, Isu K, et al:
Multiinstitutional phase II study of neoadjuvant chemotherapy for
osteosarcoma (NECO study) in Japan: NECO-93J and NECO-95J. J Orthop
Sci. 14:397–404. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Meyers PA, Schwartz CL, Krailo M, et al:
Osteosarcoma: a randomized, prospective trial of the addition of
ifosfamide and/or muramyl tripeptide to cisplatin, doxorubicin, and
high-dose methotrexate. J Clin Oncol. 23:2004–2011. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Jawad MU, Cheung MC, Clarke J, Koniaris LG
and Scully SP: Osteosarcoma: improvement in survival limited to
high-grade patients only. J Cancer Res Clin Oncol. 137:597–607.
2011. View Article : Google Scholar
|
|
23
|
Ferrari S, Smeland S, Mercuri M, et al:
Neoadjuvant chemotherapy with high-dose Ifosfamide, high-dose
methotrexate, cisplatin, and doxorubicin for patients with
localized osteosarcoma of the extremity: a joint study by the
Italian and Scandinavian Sarcoma Groups. J Clin Oncol.
23:8845–8852. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Semba S, Itoh N, Ito M, Harada M and
Yamakawa M: The in vitro and in vivo effects of
2-(4-morpholinyl)-8-phenyl-chromone (LY294002), a specific
inhibitor of phosphatidylinositol 3′-kinase, in human colon cancer
cells. Clin Cancer Res. 8:1957–1963. 2002.PubMed/NCBI
|
|
25
|
Chen Z, Yang L, Liu Y, et al: LY294002 and
Rapamycin promote coxsackievirus-induced cytopathic effect and
apoptosis via inhibition of PI3K/Akt/mTOR signaling pathway. Mol
Cell Biochem. 385:169–177. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Hu L, Zaloudek C, Mills GB, Gray J and
Jaffe RB: In vivo and in vitro ovarian carcinoma growth inhibition
by a phosphatidylinositol 3-kinase inhibitor (LY294002). Clin
Cancer Res. 6:880–886. 2000.PubMed/NCBI
|
|
27
|
Wempe SL, Gamarra-Luques CD and Telleria
CM: Synergistic lethality of mifepristone and LY294002 in ovarian
cancer cells. Cancer Growth Metastasis. 6:1–13. 2013.PubMed/NCBI
|
|
28
|
Liu ZL, Wang G, Peng AF, Luo QF, Zhou Y
and Huang SH: Fatty acid synthase expression in osteosarcoma and
its correlation with pulmonary metastasis. Oncol Lett. 4:878–882.
2012.PubMed/NCBI
|
|
29
|
Porta R, Blancafort A, Casoliva G, et al:
Fatty acid synthase expression is strongly related to menopause in
early-stage breast cancer patients. Menopause. 21:188–191. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Tao BB, He H, Shi XH, et al: Up-regulation
of USP2a and FASN in gliomas correlates strongly with glioma grade.
J Clin Neurosci. 20:717–720. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Rahman MT, Nakayama K, Ishikawa M, et al:
Fatty acid synthase is a potential therapeutic target in estrogen
receptor-/progesterone receptor-positive endometrioid endometrial
cancer. Oncology. 84:166–173. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Liu ZL, Wang G, Shu Y, Zou PA, Zhou Y and
Yin QS: Enhanced antitumor activity of epirubicin combined with
cerulenin in osteosarcoma. Mol Med Rep. 5:326–330. 2012.
|
|
33
|
Liu ZL, Zhou Y, Luo QF, et al: Inhibition
of fatty acid synthase supresses osteosarcoma cell invasion and
migration. Indian J Pathol Microbiol. 55:163–169. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Yeh CW, Chen WJ, Chiang CT, Lin-Shiau SY
and Lin JK: Suppression of fatty acid synthase in MCF-7 breast
cancer cells by tea and tea polyphenols: a possible mechanism for
their hypolipidemic effects. Pharmacogenomics J. 3:267–276.
2003.PubMed/NCBI
|
|
35
|
Van de Sande T, De Schrijver E, Heyns W,
Verhoeven G and Swinnen JV: Role of the phosphatidylinositol
3′-kinase/PTEN/Akt kinase pathway in the overexpression of fatty
acid synthase in LNCaP prostate cancer cells. Cancer Res.
62:642–646. 2002.PubMed/NCBI
|