Introduction
Ovarian cancer is one of the most life-threatening
types of epithelial cancer and patients often experience rapid
relapse following chemotherapy. Abnormal thrombosis and accompanied
thrombocytosis can occur in ovarian cancer patients and it has been
suggested that these two factors are involved in cancer metastasis.
Platelets promote metastasis by protecting tumor cells from
destruction by immune cells (1).
They also enhance tumor cell adhesion through a
selectin-GPIIb-dependent mechanism. Deficiencies in the expression
of P-selectin by platelets may prevent cancer cell rolling and
their adhesion to endothelial cells under flow conditions, thereby
reducing the number of metastatic lesions (2). A previous study suggested that
platelets mediate the epithelial-mesenchymal transition (EMT) of
cancer cells (3), as
platelet-derived transforming growth factor-β (TGFβ) and direct
platelet-tumor cell contact activated the TGFβ/Smad and NF-κB
pathways. This induced the transition of tumor cells into an
invasive mesenchymal-like phenotype and enhanced tumor metastasis
in vivo.
Although the role of platelets in cancer migration
is well understood, the details of how platelets are involved in
cancer proliferation remain to be elucidated. There is evidence
indicating that platelets cause cell cycle arrest in cancer cells,
however, thrombocytosis occurs in 10–50% of patients with solid
malignancies (4). This has been
labeled as ‘reactive’ or ‘secondary’, implying that the tumor, even
small tumors, can stimulate platelet production (5). In addition, thromboipoetin (TPO),
which drives thrombopoiesis, is often used to regenerate the
platelet counts in patients following chemotherapy, a treatment
that has been associated with tumor homing (6). Therefore, further clarification of
the association between platelets and tumor proliferation may
assist in elucidating the mechanisms underlying carcinogenesis and
is of clinical importance (7).
In the present study, the correlation between
thrombogenic markers and malignancies of ovarian tumor was
investigated. The expression of coagulant factors from patients
with benign, borderline and malignant phenotypes were determined
and human tumor tissue samples were stained using antibodies
against platelet glycoprotein IIb (CD41), a marker for platelets,
and other biomarkers associated with angiogenesis, including
platelet endothelial cell adhesion molecule 1 (CD31), vascular
endothelial growth factor (VEGF), lisyl oxidase (LOX), focal
adhesion kinase (FAK) and breast cancer anti-estrogen resistance 1
(BCAR1). The roles of platelets in ovarian cancer growth were also
determined in a transplanted mouse model. Tumor growth patterns,
animal survival rate, platelet-depletion and infusion in the murine
tumor model were assessed. The results provided evidence that
platelets contribute to the proliferation of ovarian cancer by
mediating angiogenesis, which may be important for the development
of intervention strategies to treat ovarian cancer.
Materials and methods
Patients and specimens
A total of 140 patients with histologically
confirmed ovarian tumors, including serous cystadenoma (SC),
borderline serous cystadenoma (BSC), serous cystadenocarcinoma
(SC), mucinous cystadenoma (MC), borderline mucinous cystadenoma
(BMC), mucinous cystadenocarcinoma (MCC) and clear cell carcinoma
(CCC), were recruited by the Obstetrics and Gynecology Hospital of
Fudan University between 2008 and 2010. Each histotype contained 20
tissue specimens. The patients all underwent primary surgery at the
the Obstetrics and Gynecology Hospital of Fudan University for
pelvic masses. Surgery was advised if the diameter of the pelvic
mass exceeded 5 cm, and patients with physioloigal ovarian cysts
were excluded. International Federation of Gynecological
Oncologists (FIGO) staging was assigned to the tumors following
surgery if possible.
The patient charts were reviewed to obtain
clinicopathological data regarding age, preoperative blood
coagulation function (activated partial thromboplastin time,
prothrombin time, international normalized ratio, fasting blood
glucose and D-dimer), pre- and post-operative platelet level,
preoperative serum levels of CA125, CA199, carcinoembryonic antigen
and α-fetoprotein, diagnosis, histology, FIGO staging (if any),
residual disease following tumor cytoreductive surgery (if any) and
overall survival rate. Tissue blocks, which were harvested at the
time of surgery and subsequently formalin fixed, paraffin embedded
and archived in the pathology department of the hospital in the
dark at room temperature, were retrieved for further
immunohistochemical (IHC) staining.
Additional fresh tissue specimens from 12 patients
(six with malignant epithelial ovarian tumors and six with benign
tumors) were collected following patient consent for
immunofluorescent staining.
The present study was reviewed and approved by the
Ethics Committee of the Obstetrics and Gynecology Hospital of Fudan
University, Shanghai, China.
IHC
The tissue samples, obtained from all 140 patients
with benign, borderline and malignant ovarian tumors, were
retrieved and serial 4-mm sections were obtained from each block.
The first slide was stained for hematoxylin and eosin to confirm
the pathological diagnosis and the subsequent slides were stained
for CD31, CD 41, FAK, LOX, BCAR1 and VEGF, respectively. Routine
deparaffinization and rehydration procedures were performed
following online protocols (http://www.bioworldantibodies.com/info/IHC.pdf).
For antigen retrieval, the slides were heated at
98°C in either an EDTA-Tris buffer (pH 9.0) or citric acid for 30
min and cooled naturally to room temperature. EDTA-Tris buffer was
used for the FAK and LOX staining, while citric acid was used for
the CD31, CD41, BCAR1 and VEGF staining. Endogenous peroxidase was
blocked using 3% H2O2 in methanol for 10 min
prior to washing with phosphate-buffered saline (PBS). The sections
were then incubated with the following primary antibodies: rabbit
anti-CD31 polyclonal antibody (1:50; ab28364), rabbit anti-CD41
polyclonal antibody (1:200; ab63983), rabbit anti-FAK polyclonal
antibody (1:200; ab4803), rabbit anti-LOX polyclonal antibody
(1:200; ab31238), rabbit anti-BCAR1 polyclonal antibody (1:150;
ab31831) and rabbit anti-VEGF polyclonal antibody (1:200; ab46154)
(Abcam, Cambridge, UK) overnight at 4°C. The secondary antibody
used in this study was Sunpoly-H II (Shanghai Sunbio Co., Shanghai,
China). The slides were rinsed and incubated with horseradish
peroxidase-labeled secondary anti-rabbit antibody detection reagent
(Shanghai BioTech Company, Ltd., Shanghai, China) at room
temperature for 30 min. The bound antibody complexes were stained
for 3–5 min, or until appropriate for microscopic examination with
diaminobenzidine and then counterstained with hematoxylin (30 sec)
and mounted. Negative controls were generated by incubating slides
with PBS instead of the primary antibody.
The immunoreactivity staining was characterized
quantitatively using a semi-quantitative scoring system, as
described previously (8). The
number and intensity of positive cells was quantified using
Image-Pro Plus 6.0 software (Media Cybernetics, Inc., Rockville MD,
USA) in a blinded-manner. A series of five random images on several
sections were captured to obtain a mean value.
The microvascular density (MVD) of the containing
CD31-stained slides was assessed using light microscopy (Eclipse
Ni-U; Nikon Corp., Tokyo, Japan) in the areas having the highest
number of capillaries and small venules (neovascular hot spots).
Subsequently, the microvessels were counted on 10 selected (x200)
fields of the ‘hot plot’ by the same investigator in a
blinded-manner. The MVD was defined as the mean vessel count
obtained in these fields, as reported by Ma et al (9).
Immunofluorescent staining
The freshly harvested tissues were
formaldehyde-fixed prior to freezing and were sectioned using a
cryostat (Leika CM1850; Leica Microsystems GmbH, Nussloch, Germany)
to 4 μm. The tissue sections were then incubated with rabbit
anti-CD41 polyclonal antibody (1:100; ab63983), mouse anti-CD31
monoclonal antibody (1:100; ab24590) and/or mouse anti-VEGF
monoclonal antibody (1:200; ab1316), which were all purchased from
Abcam, overnight at 4°C. The tissue sections were then incubated
with a secondary goat anti-rabbit fluorescein isothiocyanate
(111-095-003; Jackson ImmunoResearch Laboratories, Inc., West
Grove, PA, USA) and goat anti-mouse Rhodamine (115-295-003; Jackson
ImmunoResearch Laboratories, Inc.) at 37°C for 1 h. The nuclei were
counterstained with diamidinophenylindole (Sigma-Aldrich, St.
Louis, MO, USA) and fluorescent images were captured using
microscopy (BX51; Olympus, Tokyo, Japan) and analyzed using
Image-Pro Plus software for colocalization.
Tumor growth and survival rates in the
mouse model
To assess the effects of different treatments on the
tumor growth and mouse survival rates, SKOV3 human ovarian cancer
xenografts in athymic nude mice were used. The animals were
purchased from Biomodel (Shanghai Research Center for Model
Organisms), and were kept under standard conditions of 25°C, 40–60%
humidity and a 12-h light/dark cycle, and access water and food
ad libitum. Tumor loci were generated by injecting
4×106 tumor cells subcutaneously into 6–8-week-old
female mice (weight, 18–20 g) on day 0.
For the thrombin and hirudin treatment groups, prior
to tumor transplantation, the ovarian tumor cells were pretreated
with thrombin, an activator of platelets (1 U/106 cells;
Sigma-Aldrich) or hirudin, a specific inhibitor of thrombin (0.1
μg/106 cells) donated by Professor Houyan Song, Fudan
University (Shanghai, China) for 10 mins. The other groups were
treated 7 days after tumor transplantation with either: Platelet
depletion, involving administration of rat anti-mouse GPIb
immunoglobulin (Ig)G antibody (2 μg/g, Emfret Analytics,
Eibelstadt, Germany) via the tail veins once every 5 days; Isotype
control, involving administration of rat IgG (2 μg/g, Emfret
Analytics) via the tail veins once every 5 days; platelet infusion,
in which the platelets first separated from the platelet-rich
plasma of the blood by centrifugation, washed by gel filtration and
resuspended in buffer containing 137 mm NaCl, 4 mm KCl, 0.5 mm
MgCl2, 0.5 mm Na2HPO4, 11.1 mm
dextrose, 0.1% bovine serum albumin and 10 mm
N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid
(pH 7.4), as previously described (10). Briefly, in the platelet infusion
group, platelets were first separated from the platelet rich plasma
(PRP) of blood by centrifugation at 280 × g for 8 min, the plasma
and buffy coat were gently transferred to a fresh tube and the
centrifugation was repeated at 280 × g for 4 min. Platelets were
isolated by filtering the resulting PRP through a Sepharose 2B
(Sigma-Aldrich) column equilibrated with Pipes buffer (25 mM Pipes,
137 mM NaCl, 4 mM KCl, 0.1% dextrose; pH 7.0) and then resuspended
in buffer containing 137 mM NaCl, 4 mM KCl, 0.5 mM
MgCl2, 0.5mM Na2HPO4, 11.1 mM
dextrose, 0.1% bovine serum albumin, 10mM
N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid
(pH 7.4). The platelets (6×108) from two donor mice in
buffer were then injected into a single recipient xenograft mouse
via the tail vein and platelet infusion was repeated every 5 days
until experimental termination or TPO, involving administration of
recombinant human (rh)TPO (50 U/mouse/day; 3Sbio, Inc., Shenyang,
China) for 7 days.
The tumor volume and the number of mice remaining
alive following tumor cell xenograft (lethalities were due to
dyscrasia of the tumor growth) were measured on days 7, 12, 17, 22,
27, 32 and 37. The volumes of the in situ tumors were
defined as V=πab2/6 (a, major diameter; b, minor
diameter) (11) and measured using
calipers.
Effect of platelets on early developing
and fast growing periods in vivo
Based on previous in vitro studies
demonstrating the effects of platelets on the promotion of primary
ovarian tumor growth (12), the
present study evaluated the role of platelets in tumor growth
during different growing periods of carcinogenesis (early
developing, vs. fast growing periods).
In the early platelet depletion group
(E-PD/E-non-immune), the mice received either rat anti-mouse GPIb
or control rat IgG antibodies via the tail vein 1 day prior to
subcutaneous inoculation of the tumors (−1 day). The antibody
regimes were repeated every 5 days until experimental termination.
In the late platelet depletion group (L-PD/L-non-immune), the mice
received either rat anti-mouse GPIb or control rat IgG via the tail
vein 11 days post-tumor inoculation, which was repeated every 5
days until experiment termination.
In the early TPO treatment group (E-TPO), from -4
day, the mice were injected subcutaneously with rhTPO for 4 days.
The mice then received injection once every 4 days (rhTPO injection
on days 1 and 5). For the late-TPO treatment group (L-TPO),
starting from day 11, the mice received with TPO for 4 days
(between days 11 and 14). The purpose of starting the TPO
injections at -4 days for the E-TPO group was to enable the
platelet counts to rise 2-fold above that of the control at day 0.
This treatment enabled the platelet count to be comparable to that
of platelet infusion.
Each of the E-TPO, E-PD, E-non-immune, L-TPO, L-PD
and L-non-immune groups were comprised of 8 mice. For all treatment
groups, the mice were sacrificed by cervial dislocation on day 21.
The volumes of the in situ tumors were defined as
V=πab2/6, measured using calipers.
Statistical analysis
For descriptive statistics, box plots were used to
graphically depict groups of immunoreactivity data and clinical
parameters. The bottom and top of the box represent the lower and
upper quartile, respectively, the band close to the middle of the
box represents the median and open circles indicate outliers
(values >1.5 × interquartile range). Pearson’s or Spearman’s
rank correlation coefficient were used to evaluate the correlations
between the variables when the two variables were continuous or
when at least one variable was ordinal. The association between
various clinical and pathological parameters was compared using
χ2 tests. The present study compared continuous
variables using Student’s t-test or analysis of variance (ANOVA)
and used non-parametric tests (Mann-Whitney), when appropriate, to
compare differences. P<0.05 was considered to indicate a
statistically significant difference. All statistical analyses were
performed using SPSS 16.0 software (SPSS, Inc., Chicago, IL,
USA).
Results
Patients with a poor prognosis are prone
to thrombophilia
To compare the differential expression levels of
coagulation factors and molecular markers among a variety of human
pathological conditions, clinical specimens from 140 patients with
SC, BSC, SCC, MC, BMC, MCC and CCC were used for analysis of
coagulation indices and IHC markers (each histotype had 20
specimens, respectively). No differences in ages were apparent
among the clinical categories (mean=46.1; standard deviation=17.8).
The malignant histotypes (SCC, MC and CCC) had a higher D-dimer
level, a marker for fibrin generation (Fig. 1A) and a shorter thrombin clotting
time compared with the benign samples, as analyzed by ANOVA
(P<0.05; Fig. 1B). Prior to
surgery, the carcinoma patients had a significantly higher platelet
count compared with either the borderline cystadenoma or
cystadenoma (281±103×109, 262±108×109,
209±41×109, respectively; P=0.03). By contrast,
following tumor removal surgery, the platelet counts were equal in
the three groups. Notably, the preoperative platelet counts were
positively correlated with the clinical stages in the malignant
histotypes (r=0.494; P=0.004).
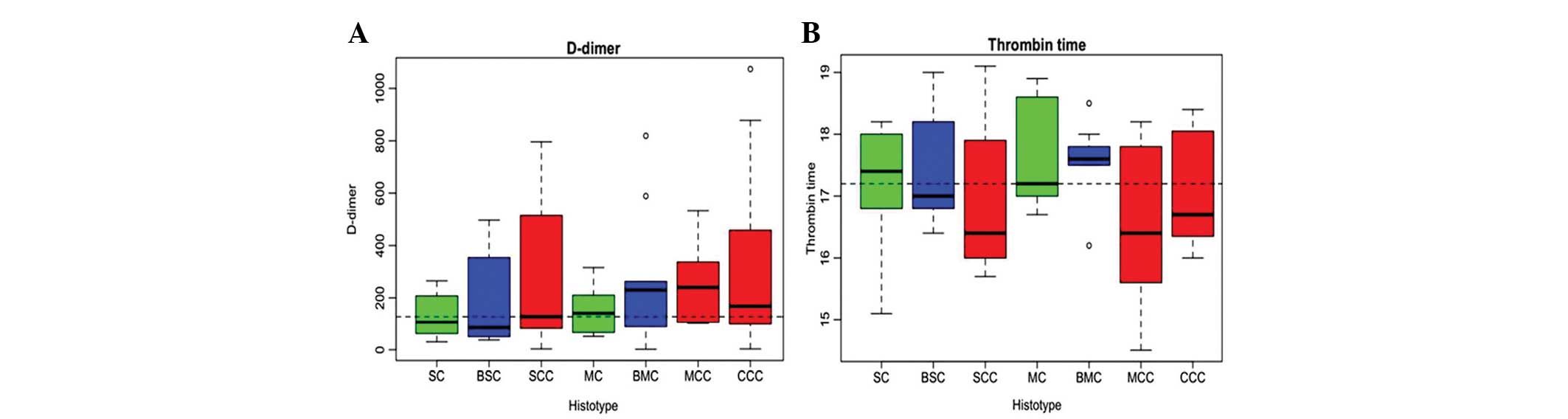 | Figure 1(A) Malignant histotypes (SCC, MC and
CCC) had higher levels of D-dimer (marker for fibrin generation)
and (B) shorter thrombin clotting time scompared with the benign
samples. The coagulation indices of clinical specimens from 140
patients with SC, BSC, SCC, MC, BMC, MCC, CCC were analyzed. No
differences in ages were observed among clinical categories
(mean=46.1, standard deviation=17.8). Bars represent the maximum
and minimum values, open circle indicate outliers. SC, serous
cystadenoma; BSC, borderline serous cystadenoma; SCC, serous
cystadenocarcinoma; MC, mucinous cystadenoma; BMC, borderline
mucinous cystadenoma; MCC, mucinous cystadenocarcinoma; CCC, clear
cell carcinoma. |
To analyze the phenotypes of the cancer specimens,
angiogenic, tumorigenic and thrombogenic markers, including CD41,
CD31 (MVD), BCAR1, VEGF, LOX and FAK, were specifically selected.
CD41, a platelet marker and receptor (GPIIb/IIa), mediates platelet
adhesion to endothelial cells and augments endothelial angiogenesis
(13). Tumor cell adhesion to
endothelium-bound platelets is mediated by GPIIb-IIIa in flow
(14). BCAR1, also referred to as
p130 Crk-associated substrate, is located at the sites of adhesion
and is associated with tumor migration, colony formation and
anchorage-independent growth (15–18).
FAK is responsible for the uninhibited proliferation of cancer
cells, their protection from apoptosis, invasion, migration,
adhesion and spreading, as well as tumor angiogenesis (19). VEGF also induces angiogenesis and
tumor growth (20). There were
significant differences in the expression levels of CD41, BCAR1,
LOX, FAK, VEGF and CD31 (MVD) in the different histoypes
(P<0.05; Fig. 2A–F). Regrouping
of the seven histotypes into three groups: cystadenoma, borderline
cystadenoma and carcinoma, according to their outcomes, revealed
that patients with carcinoma expressed significantly more CD41,
BCAR1, LOX, FAK, VEGF and MVD compared with the borderline or
cystadenoma groups (Fig. 3A–F). In
addition, the expression levels of CD41, CD31 (MVD), BCAR1, VEGF,
LOX and FAK were positively correlated with the clinical stages in
the carcinoma (SCC, MC and CCC) samples (r=0.528, 0.613, 0.575,
0.575, 0.43, 0.571 and 0.589, respectively). The expression levels
were significantly higher in patients diagnosed with late stage
cancer compared to those with early stage cancer (P<0.05).
Notably, the serum CA-125 levels were positively correlated with
the expression levels of CD41 and MVD (data not shown).
 | Figure 2Differences in the expression levels
of CD41, BCAR1, LOX, FAK, VEGF and CD31 (MVD) in different
histoypes. (A) CD41; (B) BCAR1; (C) LOX; (D) FAK; (E VEGF and (F)
MVD staining among the different histotype groups. Clinical
specimens from 140 patients with SC, BSC, SCC, MC, BMC, MCC and CCC
were subject to analysis of immunohistochemical markers. No
differences in ages were observed among the clinical categories
(mean=46.1, standard deviation=17.8). Bars represent the minimum
and maximum values, open circles indicate outliers. SC, serous
cystadenoma; BSC, borderline serous cystadenoma; SCC, serous
cystadenocarcinoma; MC, mucinous cystadenoma; BMC, borderline
mucinous cystadenoma; MCC, mucinous cystadenocarcinoma; CCC, clear
cell carcinoma; LOX, lysyl oxidase; FAK, focal adhesion kinase;
VEGF, vascular endothelial growth factor; MVD, microvascular
density. |
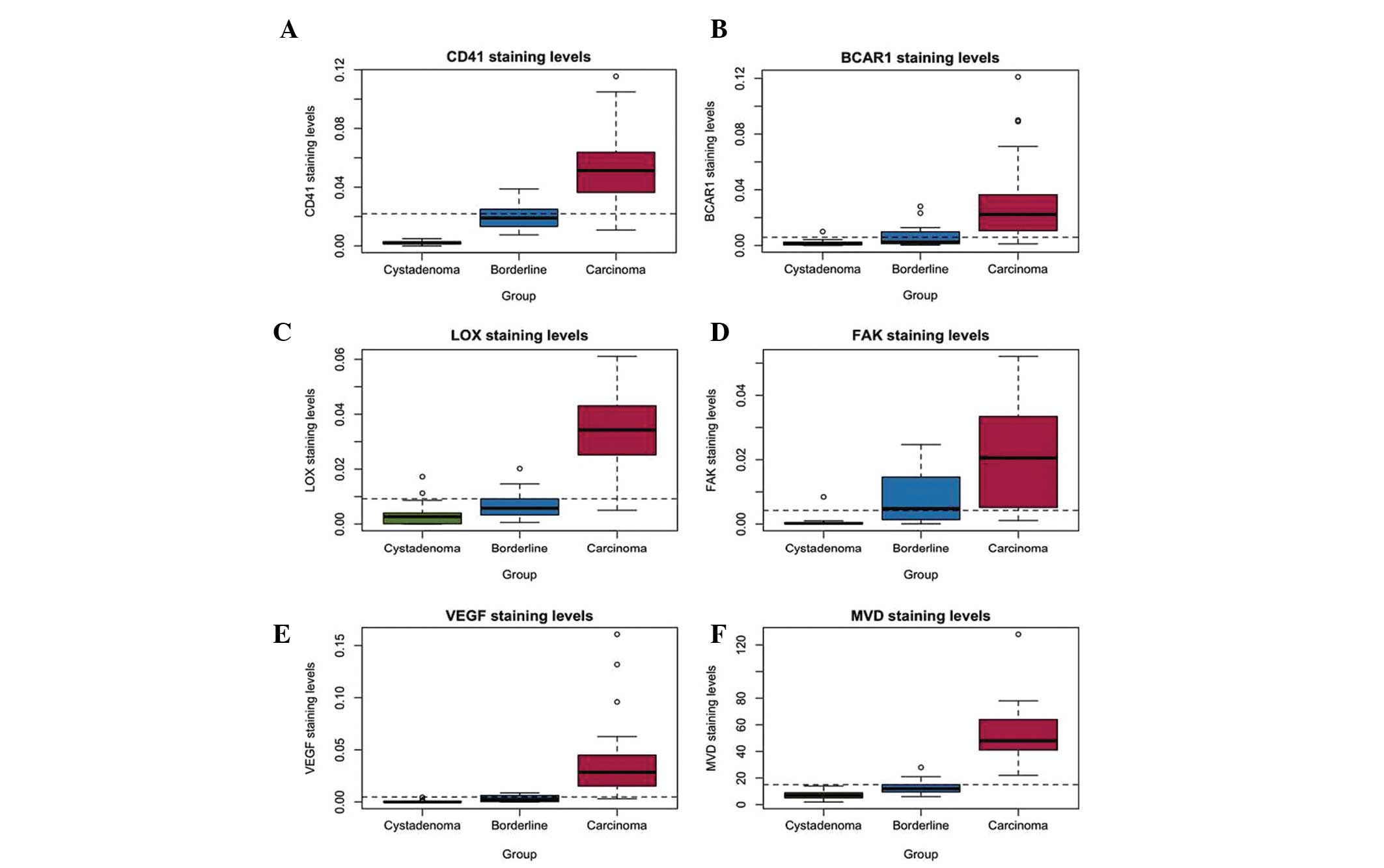 | Figure 3Expression levels of CD41, BCAR1,
LOX, FAK, VEGF and MVD in patients with carcinoma, borderline
cystadenoma or cystadenoma group. (A) CD41, (B) BCAR1, (C) LOX, (D)
FAK, (E) VEGF and (F) MVD staining among the different histotype
groups. Histotypes were regrouped into cystadenoma, borderline and
carcinoma. Bars represent the minimum and maximum values, open
circles indicate outliers. CD41, platelet glycoprotein IIb; BCAR1,
breast cancer anti-estrogen resistance 1; LOX, lysyl oxidase; FAK,
focal adhesion kinase; VEGF, vascular endothelial growth factor;
MVD, microvascular density. |
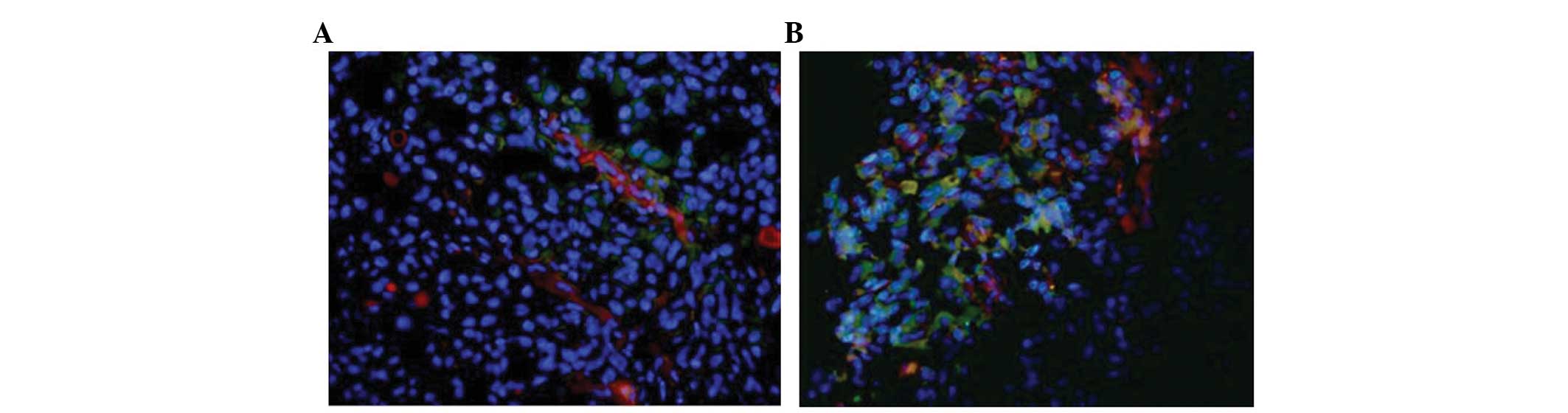
In the carcinoma samples, the expression of CD41
overlapped with that of CD31 and VEGF (Fig. 4A and B), suggesting that the
platelets may contribute to the generation of carcinoma by inducing
angiogenesis. Furthermore, platelets formed aggregates surrounding
the tumor.
Effects of platelets and thrombosis on
cancer and survival rates in a xenograft mouse model
During several stages of tumor development, tumor
cells acquire different capacities to sustain proliferation, evade
growth suppressors and activate invasion (21). The interactions between tumors and
platelets in blood vessels are critical for tumor attachment and
transendothelial migration. To examine the role of platelets and
coagulation in tumor development in situ, nude mice were
subcutaneously transplanted with SKOV3 ovarian tumor cells prior to
further treatments, including platelet depletion, platelet
infusion, TPO injection and thrombin and hirudin pretreatment. The
depletion of platelets with the injection of anti-GPIb IgG resulted
in a decrease in tumor volume compared with the injection of
control non-immune antibody alone (Fig. 5). The inhibition of tumour growth
by platelet depletion remained after 5 days (Fig. 5B). In addition, the effect of
platelet depletion was increasingly potent with tumor progression
(Fig. 5B–E). At day 27, platelet
depletion reduced tumor volume by 29% compared with the control
samples (Fig. 5E). However, the
mice receiving platelet infusion exhibited a 1.14-fold increase in
their median tumor size at day 12. The tumor-promoting effect of
platelets was sustained until day 27. Notably, at day 32 the median
tumor size of the platelet infusion group was comparable to that of
the control (Fig. 5F), which was
possibly due to the mice succumbing to mortality.
 | Figure 5Effects of platelets in growth of
tumor volume. Nude mice were subcutaneously inoculated with SKOV3
tumor cells and grouped according to treatment type: Non-immune
group (injection of platelet non-immune antibody with 2 μg/g
control rat IgG every 5 days); depletion group (injecting 2 μg/g
platelet depletion rat anti-mouse GPIb IgG antibody every 5 days);
infusion group (infusing platelets from two donor mice into a
single recipient every 5 days); TPO group (subcutaneous injection
of 50 U/mouse/day TPO for 7 days); Thrombin group (thrombin
treatment prior to subcutaneous transplantation); hirudin group
(hirudin treatment prior to subcutaneous transplantation) and
control group (tumor subcutaneous transplantation without any
treatment). The tumor volumes (mm3) were recorded on
days (A) 7, (B) 12, (C) 17, (D) 22, (E) 27 and (F) 32. Bars
represent the minimum and maximum values, open circles indicate
outliers. Non-Im, non-immune group; Infn, infusion group; Dplt,
depletion group; Hrdn, hirudin group; Thmb, thrombin group; TPO,
thrombopoietin. |
The injection of TPO, which is a factor regulating
platelet production, resulted in the most profound induction of
tumor growth day 12 and between days 22 and 27 (Fig. 5B–E). When the transplanted tumor
was pre-incubated with hirudin, an antithrombin reagent, the tumor
proliferation was reduced (Fig.
5A–E). Furthermore, the tumor-suppression effect of hirudin
peaked at day 32. However, the tumor-supressing effect of hirudin
treatment was less significant than that of platelet depletion.
Pretreatment of the tumor cells with thrombin, an enzyme that
converts fibrinogen to fibrin, increased the tumor size compared
with the non-immune antibody control (Fig. 5A–D). The difference was observed
from the beginning of tumor measurement until day 27.
The effect of different treatments on the survival
rate of mice bearing a human ovarian tumor are shown in Fig. 6. The median survival rate for the
non-immune antibody treated group was 33.5 days. Exogenous infusion
of platelets resulted in regional transplant tumor growth and
reduced the median survival rate to 30 days (Fig. 6). The median survival rate for mice
receiving platelet-depleting antibody, leading to experimental
thrombocytopenia, was 39 days. In addition, 9 of the 10 mice
receiving the platelet-depleting antibody survived >39 days.
Pretreatment of the tumor cells with hirudin
increased the mouse survival rates. By contrast, pretreatment with
thrombin decreased the mouse survival rates. Notably, TPO had a
dual effect on survival rate. Initially, it caused a rapid decrease
in survival rate, while at the later time point, it prolonged the
rates. TPO has been suggested to have additional functions to
promoting platelet generation, which may affect survival rates in a
non-specific manner (22).
Effects of platelets at different ovarian
cancer developmental stages
To assess the roles of platelets at different cancer
developmental stages (early developing and fast growing periods),
two different methods of platelet depletion and TPO injection were
developed, as mentioned previously. E-TPO treatment led to a
significantly larger tumor volume (3778.1±737.7 mm3) and
stripped tumor weight (2.48 g) compared with L-TPO treatment
(3,075.9±405.9 mm3 and 1.91 g) as shown in Fig. 7A and B (P<0.05). By contrast,
E-PD treatment resulted in smaller tumor volume (1,599.0±435.9
mm3) and stripped tumor weight (0.9 g) compared with
those treated with L-PD (1,660.8±294.1 mm3 and 1.06 g;
Fig. 7A and B), although the
difference was not statistically significant (P>0.05). For the
control groups, tumor volume (2,327.9±410.2 mm3) and
stripped tumor weight (1.69 g) following E-non-immune treatment
were not significantly different compared with those of the
L-non-immune treatment group (2,301.5±423.7 mm3 and 1.42
g; Fig. 7A and B).
 | Figure 7Effects of E-PD and L-PD (rat
antimouse GPIb IgG antibody, 2 μg/g): E-non-immune and L-non-immune
(control rat IgG, 2 μg/g) and E-TPO and L-TPO on (A) tumor volumes
(πab2/6, mm3) and (B) net tumor weight (g). n=8 in each
group. Bars represent the minimum and maximum values, open circles
indicate outliers. Nonimmune, non-immune antibody injection; PD,
platelet-depletion; TPO, thrombopoetin, E-, early; L-, late; Ig,
immunoglobulin. |
Fluorescent staining revealed that TPO treatment
resulted in more significant overlapping of CD41 and CD31 compared
with non-immune antibody treatment (Fig. 8). Mice in the E-TPO group had a
higher levels of CD41 and CD31 colocalization compared with those
in the L-TPO group.
 | Figure 8Effects of E-PD and L-PD and E-TPO
and L-TPO treatment on the colocalization of CD41 and CD31 in tumor
tissues. Images were captured under ×400 magnification using a
fluorescent microscope. Blue, diamidinophenylindole; green, CD41
and red, CD31. Nonimmune, non-immune antibody injection; PD,
platelet-depletion; TPO, thrombopoietin; E-, early; L-, late; CD41,
platelet glycoprotein IIb CD31, platelet endothelial cell adhesion
molecule 1. |
Discussion
Thrombocytosis is a notable characteristic
associated with cancer progression. However, despite the fact that
the metastasis-promoting effect of platelets has been investigated,
the role of platelets in primary tumor proliferation remains to be
full elucidated. A number of studies have suggested that platelets
facilitate hematogenous metastasis (1,2,14,23–25).
In blood flow, tumor cells roll and firmly adhere to
endothelium-bound platelets. This dynamic adhesion process is
dependent on P-selectin and glycoprotein IIb (2,14,25).
Deficiency in these two proteins reduces the number of metastatic
lesions (2). The present study
demonstrated that tumour malignancy was positively correlated with
the enhanced expression of CD41, CD31 (MVD), BCAR1, FAK, LOX and
VEGF using IHC. By examining human ovarian cancer cells
transplanted into nude mice, the present study demonstrated that
platelets were relevant for tumor development and mouse survival
rates. Early treatment with TPO resulted in a larger tumor loci
compared with later treatment, suggesting that efficient neoplasia
requires angiogenesis and platelets.
The clinical samples derived from malignant
carcinoma had higher expression levels of thrombogenic and
angiogenic markers, including D-dimer, based on IHC analysis.
D-dimer is a degraded product of fibrin generation (26,27)
and its presence, which reflects the activation of the coagulation
system and fibrinolysis, this has been associated with clinical
tumor growth, invasion and metastasis (27). The present study revealed that
malignant cancer patients had a shorter thrombin time, suggesting
that the production of thrombin increased. In the tumor
microenvironment, thrombin can bind to its receptor PAR-1 on the
endothelium and tumor cells, inducing angiogenesis and cancer
proliferation (28). Certain types
of cancer, including colon carcinoma and melanoma, express tissue
factors on their surface to augment local thrombogenesis. However,
whether increased coagulation is the cause or the consequence of
malignancy of cancer requires further investigation.
BCAR1 is one of the Crk-associated substrate protein
family members and a variety of mutations in this protein confers
susceptibility to ovarian cancer (16). Reduction in the expression levels
of BCAR1 resulted in reduced tumor growth following docetaxel
chemotherapy. BCAR1 forms a phosphorylation-dependent signaling
complex with FAK and Src kinase, promoting the adhesion-mediated
breast cancer survival rate (29).
FAK is an important adhesion regulator, which has been suggested to
promote cancer metastasis, angiogenesis and tumor progression
(30). Elevated levels of FAK in
serous ovarian carcinoma are associated with decreased patient
survival rates (31). Studies have
also suggested that VEGF-induced vascular endothelium permeability
is mediated by FAK (32). The
present study revealed that malignant cancer patients had increased
levels of FAK, BCAR1 and VEGF. These findings implied that
increased ‘cross-talk’ of platelets with either soluble factors or
P-selectin may enhance tumor survival rate.
The expression levels of the LOX family proteins,
which are secreted by tumors and have been investigated
extensively, increased in various types of cancer including head
and neck squamous cell carcinoma (33), breast (33,34),
colorectal (35,36) and prostate (37) cancer. Although numerous studies
have supported the roles of the LOX family members in tumor
suppression and in promoting metastasis, a number failed to address
the underlying mechanism. The potential contributions of LOX, BCAR1
and FAK in the malignant progression of ovarian cancer requires
further investigation.
The observation that the expression pattern of VEGF
overlapped with that of CD41, suggested that platelet aggregation
may promote angiogenesis and tumor proliferation. The coexpression
of CD41/CD31 and CD41/VEGF in tissue samples implied that platelets
promoted local ovarian cancer growth through a mechanism of
pro-angiogenesis. The effect of platelets on angiogenesis may be
dependent on the secretion of endothelial growth factors, including
platelet-derived growth factor (38). Further investigation using a
platelet-tumor co-culture model are required to delineate the
soluble factor expression levels.
Thrombocytosis was observed in patients with
carcinomas. The cause of malignancy-associated thrombocytosis has
been assumed to be attributable to interleukin-6 (IL-6) which
potently promotes megakaryocyte maturation and enhances platelet
production, although the cellular source of IL-6 may differ between
patients (39). Using a murine
tumor model, the present study demonstrated that platelet depletion
or pretreatment with hirudin considerably reduced the tumor volume
and increased the median mouse survival rate. These results
suggested that platelets were important for tumor growth. In
addition, the mice with early platelet-depletion occurred at an
early stage, had a smaller tumor size compared with mice with late
stage platelet-depletion. Furthermore, mice receiving early TPO
injection had a larger tumor size compared with those receiving
late TPO injection. However, whether the produced platelets
affected tumor growth and angiogenesis remains to be elucidated.
Levels of IL-6 are frequently increased in tumor patients and
increases thrombopoiesis only in the presence of TPO, which drives
thrombopoiesis. These findings are supported by the demonstration
in vivo that platelets transfused into mice with orthotopic
ovarian tumors enhanced the proliferation index of the tumors. TPO
regulates all stages of platelet production by promoting the
proliferation and maturation of megakaryocyte progenitors (22). In preclinical studies, TPO
treatment resulted in a rapid increase in platelet counts, but not
cytokines (40,41). Truncated or full-length forms of
TPO can stimulate the production of megakaryocytes and platelets in
humans and enhances platelet recovery following chemotherapy and
TPO may offer potential in treating thrombocytopenia in cancer
patients. In the present study, TPO was administered into the tumor
xenograft mice, resulting in ovarian cancer proliferation and
reduced survival rates. Therefore, clinically, administration of
TPO into patients with the intention of recovering platelet counts
following chemotherapy may facilitate latent tumor growth and
promote relapse.
Rupture of atherosclerotic plaques exposes the
subendothelial collage, which then activates platelets. Activated
platelets express receptors, including GPIb-IX, which is the
receptor for von Willebrand factor and induces platelet aggregation
(42). Platelet aggregation also
requires the binding of integrin GPIIB-IIIa receptor to fibrinogen.
Therefore, platelets may function as a bridge, which assists in the
attachment to blood vessel walls. It has also been demonstrated
that platelets could enhance the proliferation rate of human and
murine ovarian cancer cells by a mechanism that requires direct
cell contact (43). These findings
were supported in the present study in vivo, in which
platelet transfusions into mice with orthotopic ovarian tumors
enhanced the tumour proliferation index.
The present study compared the effect on tumor
growth of early and late platelet depletion by TPO treatment. The
purpose of starting E-TPO from -4 day was to raise platelet counts
by 2-fold on day 0. The results indicated that tumour volume when
platelet depletion occurred at an early stage of tumor formation
(−1 day), were not significantly different compared with platelet
depletion at an late stage of tumor formation (11 days). If
platelet counts were increased by TPO, the tumor volume in early
treatment samples were significantly larger compared with these in
late treatment. This implied that platelets did not contribute to
the early stages of tumor development, however, when tumors reached
a certain size, the role of platelets in tumor proliferation was
manifested. As platelets may secrete signaling/adhesive molecules,
including TGF, which promote angiogenesis and initiate tumor EMT,
they possibly contributed to the later stage of cancer
development.
In conclusion, the present study provided in
vivo evidence that platelets enhanced cell proliferation in
ovarian cancer. Additionally, the clinical data suggested that
malignant carcinoma cells were characteristic of platelet,
angiogenesis and migratory markers. These findings suggested that
thrombosis and angiogenic processes may be associated with tumor
progression. The results of the present study assist in
understanding the underlying mechanism of ovarian cancer growth and
provide insight into new therapeutic targets for treating ovarian
cancer.
Acknowledgements
The authors would like to thank Dr Sun-Wei Guo, who
conceived the hypothesis of this study and assisted with the study
design and data analysis.
Abbreviations:
|
CD31
|
platelet endothelial cell adhesion
molecule 1
|
|
CD41
|
platelet glycoprotein IIb
|
|
TPO
|
thrombopoietin
|
|
IHC
|
immunohistochemistry
|
|
FAK
|
focal adhesion kinase
|
|
LOX
|
lysyl oxidase
|
|
BCAR1
|
breast cancer anti-estrogen resistance
1
|
|
VEGF
|
vascular endothelial growth factor
|
|
SC
|
serous cystadenoma
|
|
BSC
|
borderline serous cystadenoma
|
|
SCC
|
serous cystadenocarcinoma
|
|
MC
|
mucinous cystadenoma
|
|
BMC
|
borderline mucinous cystadenoma
|
|
MCC
|
mucinous cystadenocarcinoma
|
|
CCC
|
clear cell carcinoma
|
|
MVD
|
microvascular density
|
|
FIGO
|
international federation of gynecology
and obstetrics
|
|
EMT
|
epithelial-mesenchymal transition
|
References
|
1
|
Felding-Habermann B, Habermann R, Saldivar
E and Ruggeri ZM: Role of beta3 integrins in melanoma cell adhesion
to activated platelets under flow. J Biol Chem. 271:5892–5900.
1996. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Borsig L, Wong R, Feramisco J, et al:
Heparin and cancer revisited: mechanistic connections involving
platelets, P-selectin, carcinoma mucins, and tumor metastasis. Proc
Natl Acad Sci USA. 98:3352–3357. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Labelle M, Begum S and Hynes RO: Direct
signaling between platelets and cancer cells induces an
epithelial-mesenchymal-like transition and promotes metastasis.
Cancer Cell. 20:576–590. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Buergy D, Wenz F, Groden C and Brockmann
MA: Tumor-platelet interaction in solid tumors. Int J Cancer.
130:2747–2760. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rao AK and Rao DA: Platelets signal and
tumors take off. Blood. 120:4667–4668. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Douglas VK, Tallman MS, Cripe LD and
Peterson LC: Thrombopoietin administered during induction
chemotherapy to patients with acute myeloid leukemia induces
transient morphologic changes that may resemble chronic
myeloproliferative disorders. Am J Clin Path. 117:844–850. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Stone RL, Nick AM, McNeish IA, et al:
Paraneoplastic thrombocytosis in ovarian cancer. N Engl J Med.
366:610–618. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang-Tilz Y, Tilz C, Wang B, Tilz GP and
Stefan H: Influence of lamotrigine and topiramate on MDR1
expression in difficult-to-treat temporal lobe epilepsy. Epilepsia.
47:233–239. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ma S, Liu X, Geng JG and Guo SW: Increased
SLIT immunoreactivity as a biomarker for recurrence in endometrial
carcinoma. Am J Obstet Gynecol. 202:68e61–e68. e112010. View Article : Google Scholar
|
|
10
|
Feng W, Madajka M, Kerr BA, et al: A novel
role for platelet secretion in angiogenesis: mediating bone
marrow-derived cell mobilization and homing. Blood. 117:3893–3902.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Manetta A, Satyaswaroop PG, Hamilton T, et
al: Radio imaging of human ovarian carcinoma xenograft in nude
mice. Gynecol Oncol. 28:292–299. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Holmes CE, Levis JE and Ornstein DL:
Activated platelets enhance ovarian cancer cell invasion in a
cellular model of metastasis. Clin Exp Metastasis. 26:653–661.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Trikha M, Zhou Z, Timar J, et al: Multiple
roles for platelet GPIIb/IIIa and alphavbeta3 integrins in tumor
growth, angiogenesis, and metastasis. Cancer Res. 62:2824–2833.
2002.PubMed/NCBI
|
|
14
|
Dardik R, Savion N, Kaufmann Y and Varon
D: Thrombin promotes platelet-mediated melanoma cell adhesion to
endothelial cells under flow conditions: role of platelet
glycoproteins P-selectin and GPIIb-IIIA. Br J Cancer. 77:2069–2075.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Matsui H, Harada I and Sawada Y: Src,
p130Cas, and Mechanotransduction in Cancer Cells. Genes Cancer.
3:394–401. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Nick AM, Stone RL, Armaiz-Pena G, et al:
Silencing of p130cas in ovarian carcinoma: a novel mechanism for
tumor cell death. J Natl Cancer Inst. 103:1596–1612. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sawada Y, Tamada M, Dubin-Thaler BJ, et
al: Force sensing by mechanical extension of the Src family kinase
substrate p130Cas. Cell. 127:1015–1026. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhang P, Guo A, Possemato A, et al:
Identification and functional characterization of p130Cas as a
substrate of protein tyrosine phosphatase nonreceptor 14. Oncogene.
32:2087–2095. 2013. View Article : Google Scholar :
|
|
19
|
Chatzizacharias NA, Kouraklis GP and
Theocharis SE: Clinical significance of FAK expression in human
neoplasia. Histol Histopathol. 23:629–650. 2008.PubMed/NCBI
|
|
20
|
Goel HL and Mercurio AM: VEGF targets the
tumour cell. Nat Rev Cancer. 13:871–882. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hanahan D and Weinberg RA: Hallmarks of
cancer: the next generation. Cell. 144:646–674. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Prow D and Vadhan-Raj S: Thrombopoietin:
biology and potential clinical applications. Oncology (Williston
Park). 12:1597–1604. 1607–1598; discussion 1611–1594. 1998.
|
|
23
|
Dardik R, Kaufmann Y, Savion N, et al:
Platelets mediate tumor cell adhesion to the subendothelium under
flow conditions: involvement of platelet GPIIb-IIIa and tumor cell
alpha(v) integrins. Int J Cancer. 70:201–207. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Konstantopoulos K and Thomas SN: Cancer
cells in transit: the vascular interactions of tumor cells. Annu
Rev Biomed Eng. 11:177–202. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
McCarty OJ, Mousa SA, Bray PF and
Konstantopoulos K: Immobilized platelets support human colon
carcinoma cell tethering, rolling, and firm adhesion under dynamic
flow conditions. Blood. 96:1789–1797. 2000.PubMed/NCBI
|
|
26
|
Ay C, Dunkler D, Pirker R, et al: High
D-dimer levels are associated with poor prognosis in cancer
patients. Haematologica. 97:1158–1164. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Dirix LY, Salgado R, Weytjens R, et al:
Plasma fibrin D-dimer levels correlate with tumour volume,
progression rate and survival in patients with metastatic breast
cancer. Br J Cancer. 86:389–395. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zigler M, Kamiya T, Brantley EC, Villares
GJ and Bar-Eli M: PAR-1 and thrombin: the ties that bind the
microenvironment to melanoma metastasis. Cancer Res. 71:6561–6566.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Cowell LN, Graham JD, Bouton AH, Clarke CL
and O’Neill GM: Tamoxifen treatment promotes phosphorylation of the
adhesion molecules, p130Cas/BCAR1, FAK and Src, via an
adhesion-dependent pathway. Oncogene. 25:7597–7607. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
McLean GW, Carragher NO, Avizienyte E, et
al: The role of focal-adhesion kinase in cancer - a new therapeutic
opportunity. Nat Rev Cancer. 5:505–515. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ward KK, Tancioni I, Lawson C, et al:
Inhibition of focal adhesion kinase (FAK) activity prevents
anchorage-independent ovarian carcinoma cell growth and tumor
progression. Clin Exp Metastasis. 30:579–594. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Chen XL, Nam JO, Jean C, et al:
VEGF-induced vascular permeability is mediated by FAK. Dev Cell.
22:146–157. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Erler JT, Bennewith KL, Nicolau M, et al:
Lysyl oxidase is essential for hypoxia-induced metastasis. Nature.
440:1222–1226. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kirschmann DA, Seftor EA, Fong SF, et al:
A molecular role for lysyl oxidase in breast cancer invasion.
Cancer Res. 62:4478–4483. 2002.PubMed/NCBI
|
|
35
|
Baker AM, Bird D, Lang G, Cox TR and Erler
JT: Lysyl oxidase enzymatic function increases stiffness to drive
colorectal cancer progression through FAK. Oncogene. 32:1863–1868.
2013. View Article : Google Scholar
|
|
36
|
Baker AM, Cox TR, Bird D, et al: The role
of lysyl oxidase in SRC-dependent proliferation and metastasis of
colorectal cancer. J Natl Cancer Inst. 103:407–424. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Lapointe J, Li C, Higgins JP, et al: Gene
expression profiling identifies clinically relevant subtypes of
prostate cancer. Proc Natl Acad Sci USA. 101:811–816. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Battegay EJ, Rupp J, Iruela-Arispe L, Sage
EH and Pech M: PDGF-BB modulates endothelial proliferation and
angiogenesis in vitro via PDGF beta-receptors. J Cell Biol.
125:917–928. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Anglesio MS, George J, Kulbe H, et al:
IL6-STAT3-HIF signaling and therapeutic response to the
angiogenesis inhibitor sunitinib in ovarian clear cell cancer. Clin
Cancer Res. 17:2538–2548. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Harker LA, Marzec UM, Kelly AB, et al:
Prevention of thrombocytopenia and neutropenia in a nonhuman
primate model of marrow suppressive chemotherapy by combining
pegylated recombinant human megakaryocyte growth and development
factor and recombinant human granulocyte colony-stimulating factor.
Blood. 89:155–165. 1997.PubMed/NCBI
|
|
41
|
Grossmann A, Lenox J, Ren HP, et al:
Thrombopoietin accelerates platelet, red blood cell, and neutrophil
recovery in myelosuppressed mice. Exp Hematol. 24:1238–1246.
1996.PubMed/NCBI
|
|
42
|
Bergmeier W, Piffath CL, Goerge T, et al:
The role of platelet adhesion receptor GPIbalpha far exceeds that
of its main ligand, von Willebrand factor, in arterial thrombosis.
Proc Natl Acad Sci USA. 103:16900–16905. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Cho MS, Bottsford-Miller J, Vasquez HG, et
al: Platelets increase the proliferation of ovarian cancer cells.
Blood. 120:4869–4872. 2012. View Article : Google Scholar : PubMed/NCBI
|