Introduction
Melanocytes are melanin-producing cells that are
located in the basal layer of the skin epidermis. Melanogenesis is
the physiological process that mediates photoprotection of the skin
from ultraviolet (UV) radiation-induced injury by producing melanin
in the skin and hair (1). However,
overexposure to UV radiation may result in hyperpigmentation
disorders, including melasma, freckles, lentigo, skin wrinkles and
skin cancers, all of which are of medical and cosmetic interest
(2–4).
Melanogenesis is regulated by melanogenic enzymes,
including tyrosinase (TYR), tyrosinase-related protein 1 (TRP-1),
and tyrosinase related protein 2 (TRP-2) (5). TYR is the predominant enzyme that
catalyzes the rate-limiting phase of melanogenesis, and the
inhibition of this enzyme is a typical method used to improve
pigmentation (6). TYR is crucial
for the regulation of melanin synthesis through the hydroxylation
of tyrosine to 3,4-dihydroxyphenylalanine (DOPA) and the oxidation
of DOPA to dopaquinone (7).
Members of the tyrosinase (TYR) gene family (which includes
TYR, TRP-1 and TRP-2) that are responsible for
melanin synthesis are transcriptionally regulated by
microphthalmia-associated transcription factor (MITF), which
regulates differentiation, proliferation and pigmentation of
melanocytes. In addition, mutations in MITF can lead to the
abnormal synthesis of melanin, which results in hyperpigmentation
of the skin and hair (8). For this
reason, compounds that inhibit the expression of MITF may be useful
as depigmentation agents (9).
It is well known that various signaling pathways are
involved in regulating melanogenesis. Phosphorylation of the
mitogen-activated protein kinase (MAPK) signaling pathways lead to
a reduction of melanin synthesis in mouse melanocytes. Several
studies suggested that activation/phosphorylation of the
extracellular signal-regulated kinase (ERK) and c-Jun N-terminal
kinase (JNK) signaling pathways results in the inhibition of
melanogenesis (10,11). This effect has been attributed to
the phosphorylation of MITF at serine 73, which results in MITF
ubiquitination and degradation (12). Thus, the activation of ERK and JNK
signaling inhibits melanogenesis by reducing MITF expression and
TYR activity.
A number of melanin synthesis inhibitors, including
kojic acid and arbutin, have been the focus of previous studies and
are currently being used as functional bio-cutaneous compounds for
whitening of pigmentary disorders such as melasma, dark circles and
post-inflammatory hyperpigmentation (13). Compounds with possible applications
in the cosmetic industry have been the target of ongoing research
and attention. However, the use of melanogenesis inhibitors is
heavily regulated owing to their clinical side effects, and only a
few of them (such as arbutin and kojic acid) are currently used
commercially (14). Therefore, it
would be beneficial to identify additional effective compounds that
may be able to modulate melanogenesis for use in the medical and
cosmetic industry.
The Korean red sea cucumber Stichopus
japonicus is a marine echinoderm of the Holothuroidea family;
it is consumed in East Asian countries, such as China, Indonesia,
Korea and Japan. For centuries, it has been used as a remedy for
inflammation and to treat illnesses or infections from viral and
bacterial pathogens (15). S.
japonicus has also been used as an effective remedy for
treating various internal and external wounds, including athlete's
foot. Several studies have demonstrated that S. japonicus
extracts exhibit a number of biological properties, such as
antioxidant, antifungal, and immunomodulatory activity (16–18).
These extracts have been revealed to contain lectins, peptides,
minerals, vitamins and various other molecules, including
chondroitin sulfates, glycosaminoglycan and polysaccharides. These
compounds may be responsible for the powerful wound-repairing
abilities, and anti-inflammatory, antifungal, and antiviral
properties (19). Based on these
data, S. japonicus extracts may be an excellent candidate
for inclusion in cosmeceutical and skin medical compounds for
treating skin pigmentation disorders including melasma and
post-inflammatory hyperpigmentation (PIH).
However, depigmentation via inhibition of TYR and
melanin synthesis, and the associated molecular mechanism of the
mixture of S. japonicus extracts (MSCE), have not been
examined as a candidate for a skin topical agent to improve
hyperpigmentation. To investigate the mechanistic action of MSCE in
melanin synthesis, the effects of MSCE on melanin synthesis and TYR
expression was investigated. The results suggested that MSCE
treatment inhibited melanogenesis through the ERK signaling pathway
in mouse melanocyte cells.
Materials and methods
Sample preparation and extraction
procedure
Over 1,000 live specimens of S. japonicus
(average body weight 180 g) were obtained from fishermen on the
Korean islands of Sochong and Daechong. Fresh S. japonicus
specimens were divided into the visceral organs and the body, and
were rinsed with clean water. Subsequently, the body was dried
using traditional salting and drying procedures, which involved
drying for 1 day in the sea breeze, and the visceral organs were
frozen at −80 to- 90°C. The dried S. japonicus bodies, which
were prepared at Catholic Kwandong University, International St.
Mary's Hospital (Incheon, Korea), were ground using a blender and
150 g of the resulting powder was dissolved in 2 liters 65% ethanol
for 2 h at room temperature with continual mixing. The solution was
filtered through cotton and underwent reflux extraction using a
COSMOS-660 Universal Vacuum Extractor (Kyungseo Machine Co.,
Incheon, Korea). The extract was concentrated using an EYELA N-12
Vacuum Evaporator connected to an EYELA CA-1112 Low-Temperature
Water Circulator (Tokyo Rikakikai Co., Ltd., Tokyo, Japan) prior to
freeze-drying with a TFD-100 Freeze Dryer (ilShinBioBase Co., Ltd.,
Gyeonggi-do, Korea) and powdered.
Materials
Mushroom TYR, α-melanocyte stimulating hormone
(α-MSH), l-DOPA, phorbol 12-myristate 13-acetate (PMA),
phenylthiourea (PTU), kojic acid and the MAPK/ERK kinase inhibitor
PD98059 were purchased from Sigma-Aldrich (Merck KGaA, Darmstadt,
Germany). The Cell Counting Kit-8 (CCK-8) was purchased from
Dojindo Molecular Technologies, Inc. (Kumamoto, Japan). Dulbecco's
modified Eagle's medium (DMEM), phosphate-buffered saline (PBS),
Roswell Park Memorial Institute (RPMI)-1640 medium, and
penicillin/streptomycin (P/S) were purchased from WelGene, Inc.
(Daegu, Korea). Fetal bovine serum (FBS) was purchased from Gibco
(Thermo Fisher Scientific, Inc., Waltham, MA, USA). Primary
antibodies against ERK (#9102), phosphorylated (p)-ERK (#9101), JNK
(#9252), p-JNK (#9251), p38 (#9212) and p-p38 MAPK (#9211) were
purchased from Cell Signaling Technology, Inc. (Danvers, MA, USA).
Antibodies against TYR (SC-7833), TRP-1 (SC-10443), TRP-2
(SC-74439) and β-actin (SC-47778) were purchased from Santa Cruz
Biotechnology, Inc. (Dallas, TX, USA). The antibody against MITF
(MA5-14146) was purchased from NeoMarkers, Inc. (Portsmouth, NH,
USA). Secondary antibodies specific for horseradish peroxidase
(HRP) goat anti-rabbit immunoglobulin (Ig)G (cat. no. PI-1000), HRP
horse anti-mouse IgG (cat. no. PI-2000), and HRP horse anti-goat
IgG (cat. no. PI-9500) were purchased from Vector Laboratories,
Inc. (Burlingame, CA, USA).
Cell culture
B16F10 mouse melanoma cells were purchased from the
American Type Culture Collection (Manassas, VA, USA). Cells were
cultured in DMEM containing 10% FBS and 1% P/S in 5% CO2
at 37°C. Melan-A immortalized mouse melanocytes were obtained from
Professor Dorothy C. Bennett (St. George's, University of London,
London, UK) (20) and were
cultured in RPMI-1640 medium containing 10% FBS, 1% P/S, and 200 nM
PMA in 10% CO2 at 37°C.
Cell viability assay
The mouse cells were seeded (1×104
cells/well) in 96-well plates and incubated at 37°C. Following a 24
h incubation, the cells were washed with DPBS (WelGene, Inc.,
Daegu, Korea) and replaced with DMEM (B16F10 cells) or RPMI1640
(Melan-A cells), containing MSCE diluted to various concentrations
at 0–100%. Following 24 h at 37°C, the cells were washed with DPBS,
and DMEM (B16F10 cells) or RPMI1640 (Melan-A cells) containing 10%
CCK-8 solution was added. The cells were then incubated at room
temperature and the absorbance was measured at 450 nm using a
microplate reader (SpectraMax 340; Molecular Devices, LLC,
Sunnyvale, CA, USA).
Mushroom TYR activity assay
A mushroom TYR activity assay was assessed by
measuring DOPA oxidase activity using a slightly modified
experimental protocol from a previous study (21). Briefly, 95 µl of 0.1 M sodium
phosphate buffer (pH 6.5) containing the indicated concentrations
of kojic acid (50 µM) and MSCE, and 20 µl of mushroom TYR (1,000
U/ml) were combined in each well, and 20 µl of 5 mM l-DOPA was then
added. The absorbance was measured at 475 nm every 10 min for 1 h
using a microplate reader (SpectraMax 340; Molecular Devices, LLC,
Sunnyvale, CA, USA).
Melanin assay
B16F10 cells characteristically release melanin,
which was measured with slight modifications to a previously
described protocol (22). Briefly,
cells were seeded (1×105 cells/well) in 6-well plates.
Following 24 h at 37°C, the plates were washed with DPBS and the
media was replaced with phenol red-free DMEM containing α-MSH (1
µM), or PD98059 (10 µM) for 30 min at 37°C followed by α-MSH and
MSCE treatment as aforementioned. The cells were subsequently
incubated for 3 days. Absorbance was measured in the cell culture
supernatant at 405 nm using a microplate reader (SpectraMax 340;
Molecular Devices, LLC, Sunnyvale, CA, USA). PTU is an inhibitor of
melanin synthesis, and this compound was employed as a positive
control; BF16F10 cells were treated with PTU (50 µM) for 3 days at
37°C in place of MSCE.
Melan-A cells were seeded (5×105
cells/well) in 24-well plates. Following 24 h incubation, the
plates were washed with DPBS, and then RPMI1640 containing the
indicated concentrations of MSCE or PD98059 (pretreated for 30 min
at 37°C, prior to MSCE treatment) was added. The plates were
subsequently incubated at 37°C for 3 days. Cells were lysed in 110
µl of 1 N NaOH at 55°C for 15 min, and the absorbance was measured
at 470 nm using a microplate reader (SpectraMax 340; Molecular
Devices, LLC, Sunnyvale, CA, USA). All results were normalized to
the total protein concentration of the cell pellet using a
Bicinchoninic Acid Assay kit (Pierce; Thermo Fisher Scientific,
Inc.).
Western blot analysis
Cells were cultured in a 100 mm dish and treated as
aforementioned for 0–720 min. Cells were lysed using a cell lysis
buffer [62.5 mM Tris-HCl (pH 6.8), 2% SDS, 5% β-mercaptoethanol, 2
mM phenylmethylsulfonyl fluoride, Complete Protease Inhibitors
(Roche Diagnostics GmbH, Mannheim, Germany), 1 mM
Na3VO4, 50 mM NaF and 10 mM EDTA] and
centrifuged at 13,000 × g for 20 min at 4°C. Equal amounts (20 µg)
of protein were separated by SDS-PAGE (8–12%) and transferred onto
a polyvinylidene fluoride membrane (EMD Millipore, Billerica, MA,
USA). Following blocking with 5% skim milk for 2 h at room
temperature, the membranes were probed with primary antibodies
(1:1,000) for 24 h at 4°C and then incubated with horseradish
peroxidase-conjugated secondary antibodies (1:1,000) for 3 h at
room temperature. Following washing in TBS- 0.1% Tween-20 (TBS-T)
buffer, hybridized antibodies were detected using an Enhanced
Chemiluminescence detection kit (Amersham; GE Healthcare Life
Sciences, Chalfont, UK) and images were captured using an LAS-1000
Lumino-image Analyzer (Fujifilm, Tokyo, Japan).
Immunocytochemistry
Cells were seeded (1×105 cells/well) onto
glass coverslips pre-coated with poly-l-lysine (Sigma-Aldrich;
Merck KGaA) and incubated for 24 h at 4°C. Slides were fixed with
4% paraformaldehyde for 15 min at room temperature. Following
washing with PBS, cells were treated with 0.01% Triton X-100
(Sigma-Aldrich; Merck KGaA) and then blocked with 2% bovine serum
albumin (Merck KGaA) in TBS-T for 1 h at room temperature. The
cells were incubated with an anti-TYR antibody (1:100) at 4°C
overnight. Following washing with TBS-T + 0.1% Tween-20 buffer,
cells were incubated for 3 h at 4°C with goat anti-rabbit
IgG-fluorescein isothiocyanate secondary antibodies (1:1,000;
SC-2012, Santa Cruz Biotechnology, Inc.) and mounted with
fluorescent mounting medium containing DAPI (Golden Bridge
International, Inc., Bothell, WA, USA). Cell morphology was
observed using a FluoView FV10i fluorescence microscope and a DP70
Digital Microscope Camera (both from Olympus Corporation, Tokyo,
Japan). Images were processed using the proprietary DP70 Controller
software (Ver.03.03; Olympus Corporation).
Statistical analysis
Statistical analyses were performed using SPSS 18.0
(SPSS, Inc., Chicago, IL, USA). There were three replicates for
each of the data measurements. Results are presented as the mean ±
standard deviation. Data were analyzed by one-way analysis of
variance, followed by Tukey's multiple comparison tests. P<0.05
was considered to indicate a statistically significant
difference.
Results
Effects of MSCE on mouse melanocyte
cell viability
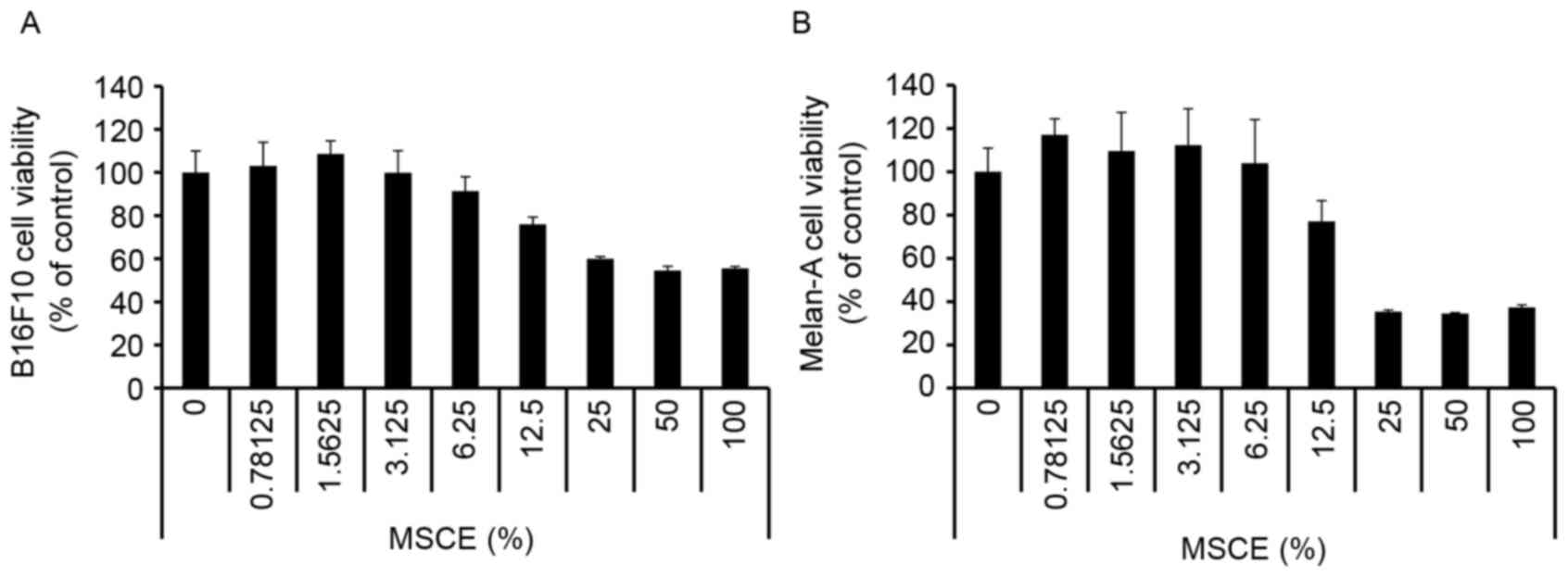
First, a CCK-8 assay was performed to evaluate the
cytotoxicity of MSCE on B16F10 mouse melanoma cells and Melan-A
normal mouse melanocytes. Cells were treated with MSCE at a
concentration range of 0–100%. As demonstrated in Fig. 1, there was no detectable
cytotoxicity at concentrations ranging from 0.78–6.25%. Therefore,
the MSCE concentration range of 1.25–5% was used for the subsequent
experiments.
Effects of MSCE on melanogenesis in
B16F10 and Melan-A cells
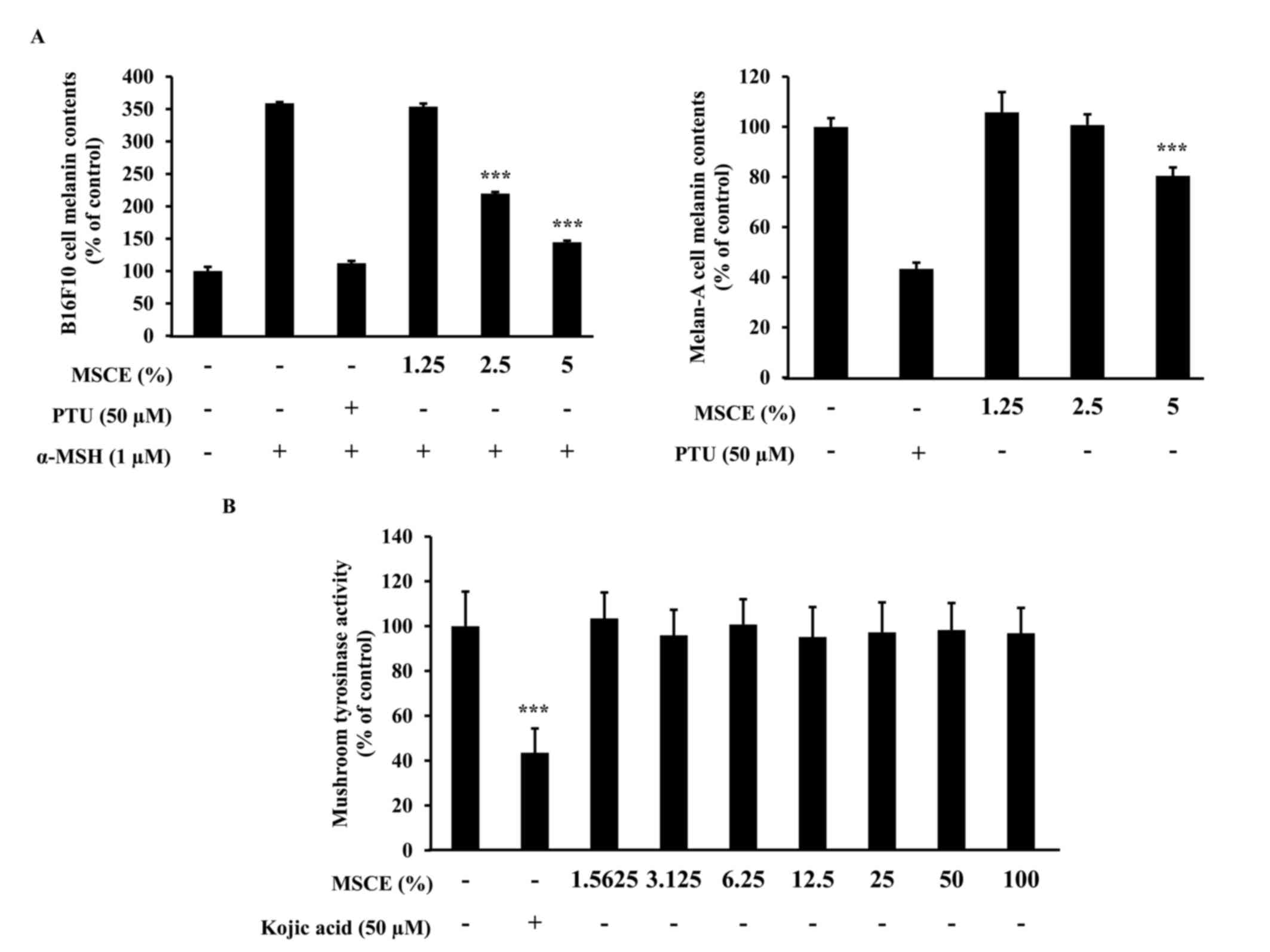
The effects of MSCE treatment on melanogenesis in
mouse cells were examined. To determine whether MSCE inhibits
melanin production in mouse cells, melanin content was measured
following 3 days treatment with MSCE at concentrations of 1.25–5%.
In cells treated with α-MSH, melanin levels in the media increased
compared with untreated cells, due to an induction of melanogenesis
(Fig. 2A). However, cells
co-treated MSCE exhibited a dose-dependent reduction in melanin
pigmentation in B16F10 and Melan-A cells, compared with cells
treated with α-MSH alone (Fig.
2A). PTU, a well-known TYR inhibitor (4), was used as a positive control; PTU
treatment inhibited melanin pigmentation compared with cells
treated with α-MSH alone (Fig.
2A).
Melanogenesis is regulated by an enzymatic cascade
that is under the control of TYR (23). For this reason, various compounds
that have been developed for skin-whitening purposes directly
inhibit TYR (24). The effect of
MSCE on TYR activity was examined using a mushroom TYR activity
assay. Treatment with 50 µM kojic acid, a direct inhibitor of TYR
that was used in this assay as a positive control, resulted in
significant inhibition of TYR activity (Fig. 2B). However, treatment with various
concentrations of MSCE did not inhibit mushroom TYR activity
(Fig. 2B). These results
demonstrated that the inhibitory effect of MSCE on melanogenesis
was not mediated by the direct inhibition of TYR.
Effects of MSCE on melanogenesis
signaling pathways in B16F10 and Melan-A cells
To investigate whether the inhibitory effect of MSCE
is related to the melanogenesis signaling pathways, mouse cells
were treated with α-MSH and/or MSCE, and the protein expression
levels of TYR, TRP-1, TRP-2 and MITF was examined by western
blotting. As demonstrated in Fig.
3A, the protein expression levels of TYR, TRP-1 and TRP-2 were
reduced following co-treatment with MSCE for 24, 48 or 72 h,
compared with the α-MSH-treated group in B16F10 cells. In addition,
MITF protein expression levels were reduced in a time-dependent
manner compared with the α-MSH-treated group in B16F10 cells
(Fig. 3A). These results indicated
that MSCE treatment inhibited melanin production through the
reduction of MITF and TYR expression levels. The inhibitory effect
of MSCE on TYR expression in the mouse cell lines was confirmed by
immunocytochemistry. The results indicated that TYR levels were
reduced following MSCE treatment for 72 h, compared with the
α-MSH-treated group in B16F10 cells and with the untreated group in
Melan-A cells (Fig. 3B).
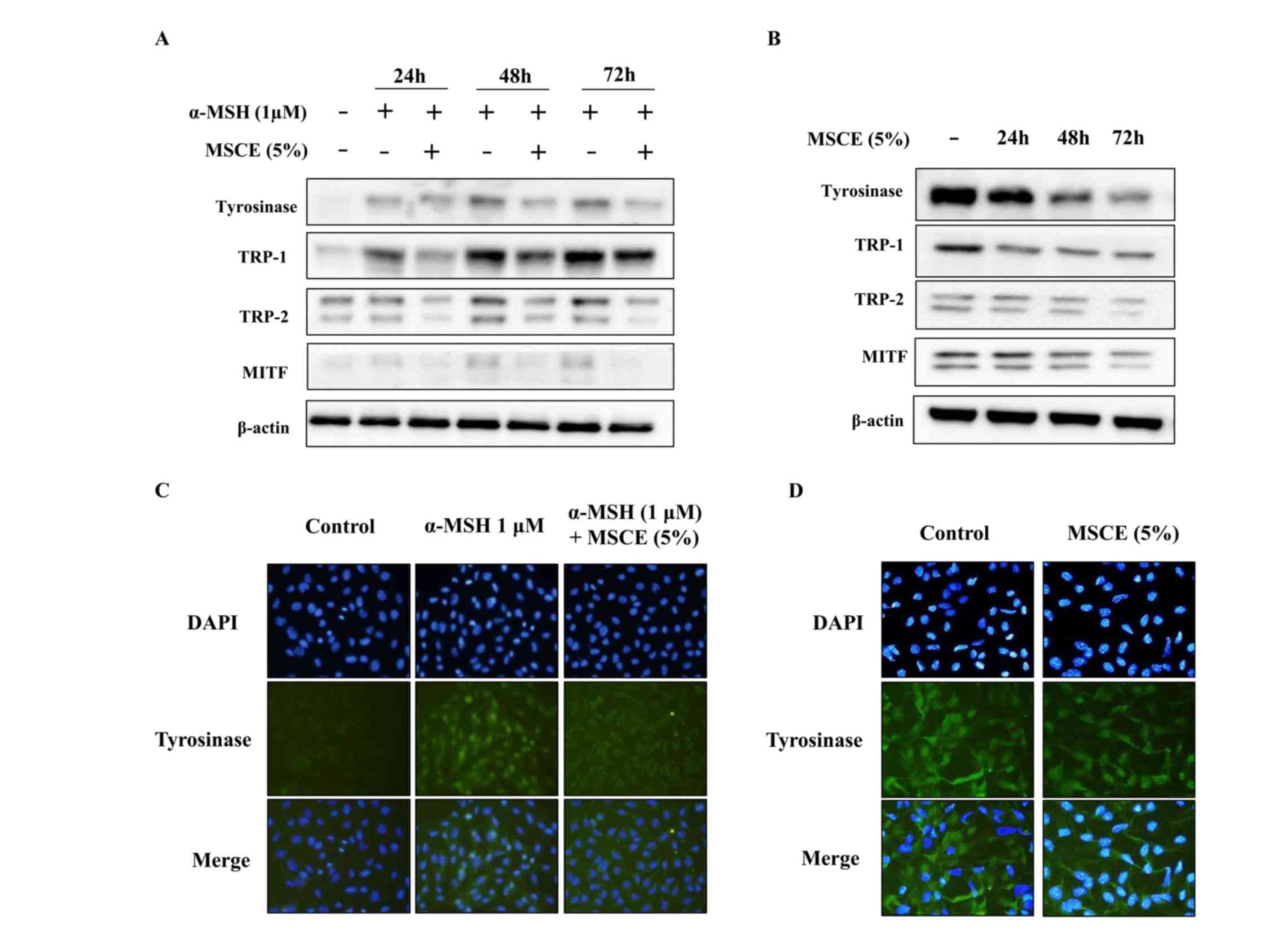 | Figure 3.Effects of MSCE on
melanogenesis-related proteins. B16F10 mouse melanoma cells (left
panels) and Melan-A mouse normal melanocytes (right panels) were
treated with MSCE (5%) for 24, 48 or 72 h. B16F10 cells were
additionally induced towards melanogenesis with 1 µM α-MSH. (A)
Cell lysates were analyzed by western blotting for TYR, TRP-1,
TRP-2 and MITF protein expression. β-actin was used as loading
control. (B) Expression of TYR was also examined by
immunocytochemical staining. Representative images of (C) B16F10
cells and (D) Melan-A melanocytes from experiments performed in
triplicate depict TYR expression (green), nuclear DAPI staining
(blue) and merged signals (magnification, ×200). α-MSH,
α-melanocyte stimulating hormone; MITF, microphthalmia-associated
transcription factor; MSCE, mixed Stichopus japonicus
extract; TRP, tyrosinase related protein; TYR, tyrosinase. |
Effects of MSCE on signal transduction
pathways in mouse cells
In an attempt to identify the underlying mechanisms
involved in the MSCE-mediated cell depigmentation, the effects of
MSCE treatment (5%) on the activation of ERK, JNK and p38 MAPKs
were examined by western blot analysis. In B16F10 cells treated
with MSCE for up to 720 min, ERK and JNK phosphorylation was
greatly increased at the 30 and 60 min time points, respectively
(Fig. 4A). By contrast, p38
phosphorylation increased in expression slightly (Fig. 4A). In Melan-A cells, ERK
phosphorylation was notably increased at 10 min following MSCE
treatment, whereas p38 phosphorylation was slightly increased at
120 min. However, no change to JNK levels (Fig. 4B). To determine the possible
involvement of p38 and JNK signaling on the MSCE-mediated
inhibitory effect, cells were treated with the JNK and p38
inhibitors (SP600125 and SB203580, respectively) and melanin
production was analyzed following MSCE treatment. The results
revealed no effect of the inhibitors on melanin production
following MSCE treatment (data not shown).
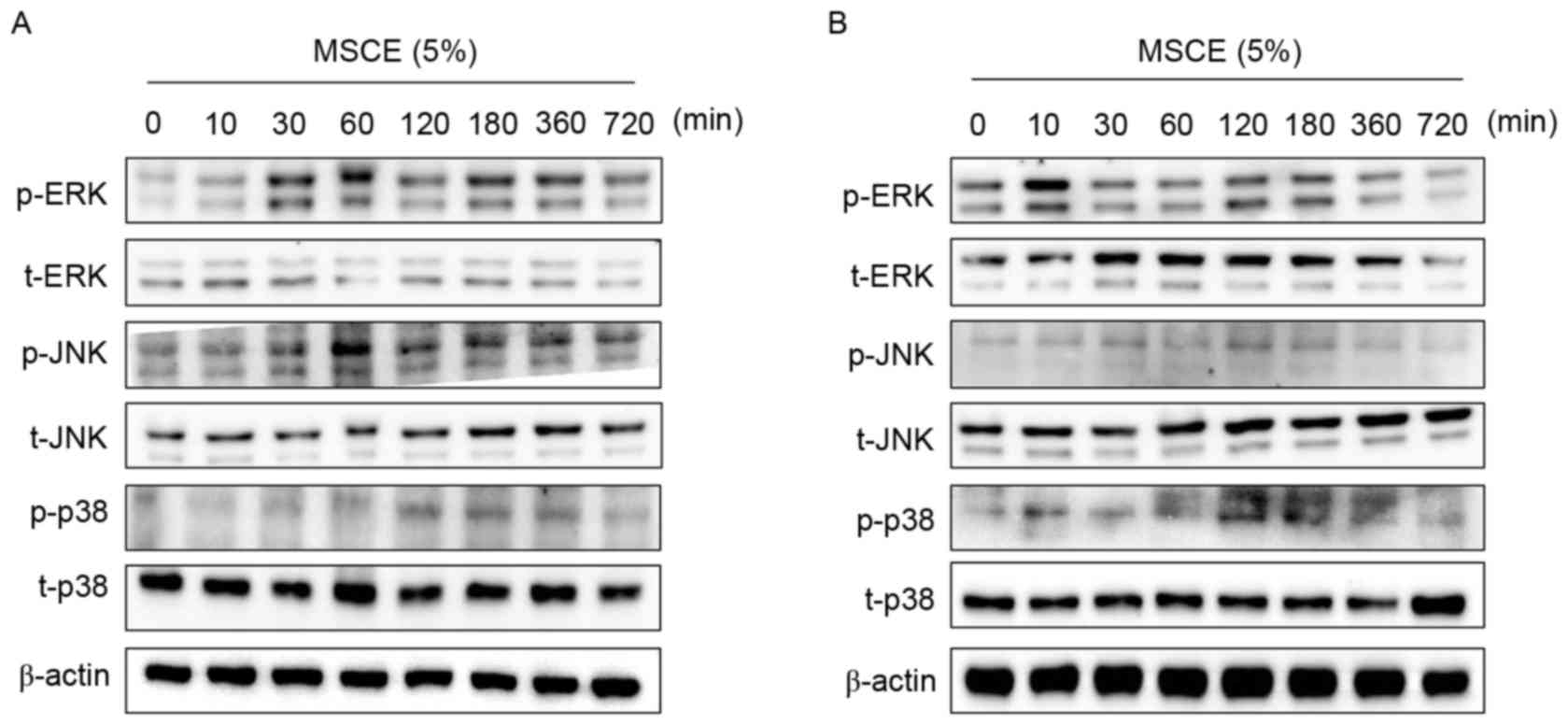 | Figure 4.Effects of MSCE on signal transduction
pathways in mouse melanocyte cells. Following 24 h of serum
starvation, (A) B16F10 mouse melanoma cells and (B) Melan-A mouse
normal melanocytes were treated with 5% MSCE for the indicated time
periods. Cell lysates were harvested for western blot analysis
using primary antibodies against p-ERK, t-ERK, p-JNK, t-JNK, p-p38
and t-p38; β-actin was used as the loading control. The results are
representative of triplicate experiments. α-MSH, α-melanocyte
stimulating hormone; ERK, extracellular signal-regulated kinase;
JNK, c-Jun N-terminal kinase; MSCE, mixed Stichopus
japonicus extract; p, phosphorylated; p38, mitogen-activated
protein kinase 14; t, total. |
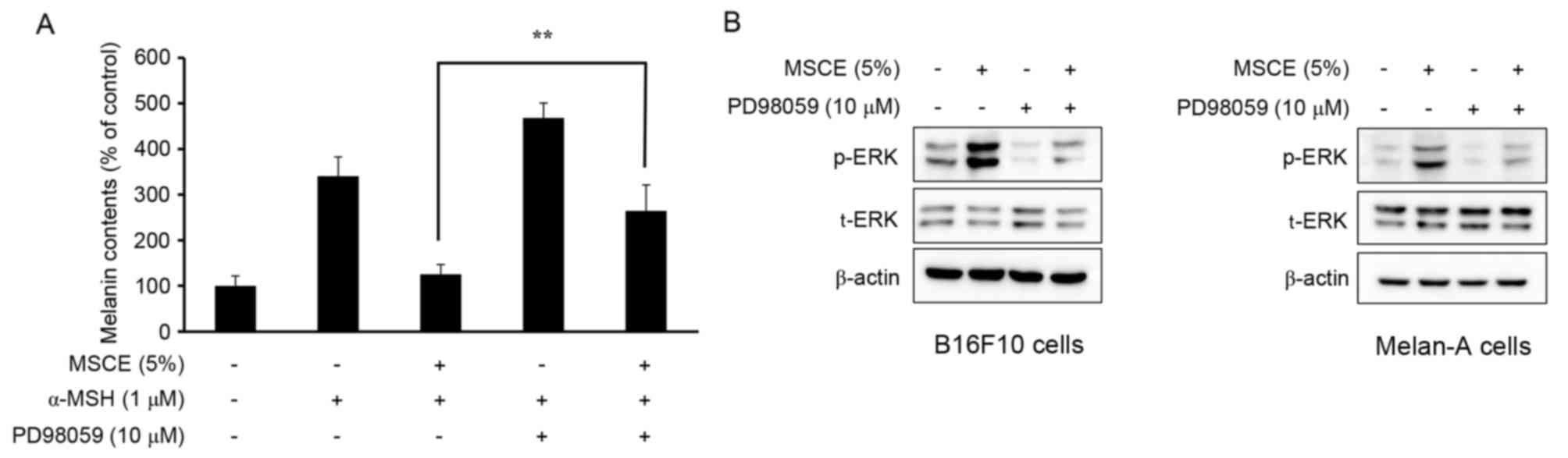
Effects of MSCE on melanogenesis and
ERK phosphorylation
The phosphorylation of ERK has been reported to
inhibit TYR expression, which subsequently reduces cellular melanin
production (25). Therefore, the
present study examined whether the MSCE-induced activation of the
ERK signaling pathway may be important for the MSCE-mediated
inhibition of melanin production in B16F10 and Melan-A cells. For
this purpose, mouse melanocyte cells were treated with MSCE for 30
min in the presence or absence of PD98059, a MEK inhibitor. As
demonstrated in Fig. 5A, the
melanin contents of B16F10 cells co-treated with α-MSH and PD98059
were higher compared with cells treated with α-MSH alone. Melan-A
cells treated with PD98059 did not demonstrate any significant
difference in melanin levels (data not shown). This synergistic
effect of α-MSH and PD98059 on melanin content was reduced by MSCE
treatment (Fig. 5A). The effects
of PD98059 treatment on MSCE-induced ERK phosphorylation was
examined by western blotting, which revealed a reduction of ERK
phosphorylation levels in the MSCE-treated mouse cells following
PD98059 addition (Fig. 5B). These
results indicated that ERK phosphorylation may be involved in the
MSCE-mediated reduction of melanogenesis.
Discussion
Pigmentary disorders, including melasma, lentigo,
post-inflammatory hyperpigmentation and dark circles, are
associated with abnormal regulations of melanin synthesis or
melanosome transfer (26). These
disorders irregularly darken the skin of patients, and may cause
discomfort or nervousness in everyday life. Several depigmentation
compounds have been created and have both medical and clinical
applications in skin. However, many of these compounds produce
toxic and unwanted side effects owing to the inclusion of
potentially harmful ingredients, such as mercury and high levels of
hydroquinone (27). For this
reason, research has been focused on the development of safe and
effective alternative whitening compounds. Various natural products
originating from marine organisms and plants have been investigated
for effects on pigmentation, including TYR, TRP-1 and TRP-2
expression, and are used as traditional whitening compounds
(28). The present study
demonstrated the inhibitory effects of MSCE on melanin synthesis in
B16F10 and Melan-A cells and its underlying mechanism.
Yoon et al (29) previously reported an effect of sea
cucumber on the expression of several melanogenic proteins,
including TYR, TRP-1, TRP-2 and MITF. Furthering this research, the
present study investigated the mechanism of MSCE as a novel
skin-whitening compound. The results further established that MSCE
functions via the inhibition of TYR, TRP-1, TRP-2 and MITF protein
expression. The effects of MSCE were dose-dependent and did not
produce any critical cytotoxicity at doses between 1.25 and 5% that
were used for the melanogenesis assays. TYR has been reported to
catalyze the rate-limiting phase of melanin synthesis, and it is
the first target of PTU, a compound with important depigmentation
effects that was recently employed for medical and cosmetic
purposes (30). MSCE treatment did
not significantly alter TYR activity when tested on mushroom TYR;
however, MSCE treatment did significantly decrease melanin
synthesis in B16F10 and Melan-A cells in a dose-dependent manner.
These results suggested that MSCE did not directly inhibit TYR
activity, indicating that the effects may be a result of indirect
mechanisms. Thus, the MSCE-induced depigmentation may be the result
of inhibitory effects on the signaling pathways controlling TYR
expression.
To understand the results of the present study, the
mechanisms involved in MSCE-mediated melanogenesis inhibition were
investigated. Western blot analysis of melanogenesis-related
proteins was performed, and the results suggested that MSCE
treatment reduced the protein expression levels of TYR, TRP-1,
TRP-2 and MITF in a time-dependent manner. In addition, MSCE
treatment reduced the melanin content compared with untreated
cells. The effects of MSCE on TYR expression, which regulates
melanin synthesis in B16F10 and Melan-A cells, were confirmed using
immunocytochemical analysis. The results indicated that the TYR
protein expression levels in B16F10 cells and Melan-A cells were
reduced following treatment with MSCE for 72 h compared with
untreated cells.
The MAPKs, including ERK, JNK and p38 MAPK, are
important in regulating melanogenesis (31). To explain the mechanisms underlying
the depigmentation effects of MSCE treatment, changes to the
activation/phosphorylation of ERK, JNK and p38 MAPK were examined
in a time-course MSCE treatment experiment using western blot
analysis for 0–720 min. The results demonstrated that the
phosphorylation of ERK was significantly increased and
phosphorylation of JNK was slightly increased following MSCE
treatment compared with untreated cells; however, p38 MAPK
phosphorylation remained unchanged. These findings suggested that
MSCE-induced depigmentation in B16F10 and Melan-A cells may occur
through MAPK-regulated signaling pathways. To identify which MAPKs
may be mediating MSCE-induced melanogenesis inhibition, B16F10 and
Melan-A cells were analyzed in the presence of SB203580, a p38
inhibitor, and PD98059, a specific MEK inhibitor. Although SB203580
had no effect in melanin production (data not shown), PD98059
markedly increased MSCE-suppressed melanin contents in B16F10
cells. In addition, the PD98059-induced suppression of p-ERK
expression was reversed by MSCE treatment in α-MSH-treated B16F10
and Melan-A cells. Notably, it has previously been reported that
ERK is an important regulator of melanogenesis, as ERK
phosphorylation induces MITF activation and its following
degradation, and thus, inhibits melanin synthesis. In addition, the
phosphorylation of ERK inhibits melanin synthesis through the
suppression of TYR expression (32). The effects of MSCE on melanogenesis
and phosphorylation of ERK demonstrated in the present study are in
accordance with the known role of the ERK signaling pathway in
melanin synthesis.
In conclusion, the present study demonstrated that
MSCE treatment led to the phosphorylation of ERK and JNK, which
suppressed the degradation of MITF, TYR, TRP-1, and TRP-2 in B16F10
and Melan-A cells, subsequently reducing melanin synthesis.
Therefore, the results suggested that MSCE may be a useful
depigmenting compound that may be used in the treatment of unwanted
pigmentation conditions and may be a novel future therapeutic in
the medical and cosmetic industries.
Acknowledgements
The present study was supported by research funding
from Catholic Kwandong University International, St. Mary's
Hospital (grant no. CKURF-201406830001).
References
|
1
|
D'Mello SA, Finlay GJ, Baguley BC and
Askarian-Amiri ME: Signaling pathways in melanogenesis. Int J Mol
Sci. 17:pii: E11442016. View Article : Google Scholar
|
|
2
|
Ichihashi M and Ando H: The maximal
cumulative solar UVB dose allowed to maintain healthy and young
skin and prevent premature photoaging. Exp Dermatol. 23 Suppl
1:S43–S46. 2014. View Article : Google Scholar
|
|
3
|
Sklar LR, Almutawa F, Lim HW and Hamzavi
I: Effects of ultraviolet radiation, visible light, and infrared
radiation on erythema and pigmentation: A review. Photochem
Photobiol Sci. 12:54–64. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Gange RW: Comparison of pigment responses
in human skin to UVB and UVA radiation. Prog Clin Biol Res.
256:475–485. 1988.PubMed/NCBI
|
|
5
|
Huang HC, Hsieh WY, Niu YL and Chang TM:
Inhibitory effects of adlay extract on melanin production and
cellular oxygen stress in B16F10 melanoma cells. Int J Mol Sci.
15:16665–16679. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ma H, Xu J, DaSilva NA, Wang L, Wei Z, Guo
L, Johnson SL, Lu W, Xu J, Gu Q and Seeram NP: Cosmetic
applications of glucitol-core containing gallotannins from a
proprietary phenolic-enriched red maple (Acer rubrum) leaves
extract: Inhibition of melanogenesis via down-regulation of
tyrosinase and melanogenic gene expression in B16F10 melanoma
cells. Arch Dermatol Res. 309:265–274. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Camacho-Hübner A and Beermann F: Cellular
and molecular features of mammalian pigmentation-tyrosinase and
TRP. Pathol Biol (Paris). 48:577–583. 2000.(In French). PubMed/NCBI
|
|
8
|
Ishii N, Ryu M and Suzuki YA: Lactoferrin
inhibits melanogenesis by down-regulating MITF in melanoma cells
and normal melanocytes. Biochem Cell Biol. 95:119–125. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Tagashira H, Miyamoto A, Kitamura S,
Tsubata M, Yamaguchi K, Takagaki K and Imokawa G: UVB stimulates
the expression of endothelin B receptor in human melanocytes via a
sequential activation of the p38/MSK1/CREB/MITF pathway which can
be interrupted by a french maritime pine bark extract through a
direct inactivation of MSK1. PLoS One. 10:e01286782015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Han JS, Sung JH and Lee SK:
Antimelanogenesis activity of hydrolyzed ginseng extract (GINST)
via Inhibition of JNK Mitogen-activated protein kinase in B16F10
Cells. J Food Sci. 81:H2085–H2092. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Huang HC, Wei CM, Siao JH, Tsai TC, Ko WP,
Chang KJ, Hii CH and Chang TM: Supercritical fluid extract of spent
coffee grounds attenuates melanogenesis through downregulation of
the PKA PI3K/Akt, and MAPK signaling pathways. Evid Based
Complement Alternat Med. 2016:58602962016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fu YT, Lee CW, Ko HH and Yen FL: Extracts
of Artocarpus communis decrease α-melanocyte stimulating
hormone-induced melanogenesis through activation of ERK and JNK
signaling pathways. ScientificWorldJournal. 2014:7243142014.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hseu YC, Cheng KC, Lin YC, Chen CY, Chou
HY, Ma DL, Leung CH, Wen ZH and Wang HM: Synergistic effects of
linderanolide B Combined with Arbutin, PTU or kojic acid on
tyrosinase inhibition. Curr Pharm Biotechnol. 16:1120–1126. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Desmedt B, Courselle P, de Beer JO,
Rogiers V, Grosber M, Deconinck E and De Paepe K: Overview of skin
whitening agents with an insight into the illegal cosmetic market
in Europe. J Eur Acad Dermatol Venereol. 30:943–950. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang X, Wang L, Che J, Li Z, Zhang J, Li
X, Hu W and Xu Y: Improving the quality of Laminaria japonica-based
diet for Apostichopus japonicus through degradation of its algin
content with Bacillus amyloliquefaciens WB1. Appl Microbiol
Biotechnol. 99:5843–5853. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Liu X, Sun Z, Zhang M, Meng X, Xia X, Yuan
W, Xue F and Liu C: Antioxidant and antihyperlipidemic activities
of polysaccharides from sea cucumber Apostichopus japonicus.
Carbohydr Polym. 90:1664–1670. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Gao F, Li F, Tan J, Yan J and Sun H:
Bacterial community composition in the gut content and ambient
sediment of sea cucumber Apostichopus japonicus revealed by 16S
rRNA gene pyrosequencing. PLoS One. 9:e1000922014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Himaya SW, Ryu B, Qian ZJ and Kim SK: Sea
cucumber, Stichopus japonicus ethyl acetate fraction modulates the
lipopolysaccharide induced iNOS and COX-2 via MAPK signaling
pathway in murine macrophages. Environ Toxicol Pharmacol. 30:68–75.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zohdi RM, Zakaria ZA, Yusof N, Mustapha NM
and Abdullah MN: Sea cucumber (Stichopus hermanii) based hydrogel
to treat burn wounds in rats. J Biomed Mater Res B Appl Biomater.
98:30–37. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Bennett DC, Cooper PJ and Hart IR: A line
of non-tumorigenic mouse melanocytes, syngeneic with the B16
melanoma and requiring a tumour promoter for growth. Int J Cancer.
39:414–418. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Shin HJ, Oh CT, Kwon TR, Beak HS, Joo YH,
Kim JH, Lee CS, Lee JH, Kim BJ, Shin SS and Park ES: A novel
adamantyl benzylbenzamide derivative, AP736, inhibits melanogenesis
in B16F10 mouse melanoma cells via glycogen synthase kinase 3β
phosphorylation. Int J Mol Med. 36:1353–1360. 2015.PubMed/NCBI
|
|
22
|
Oh CT, Lee D, Koo K, Lee J, Yoon HS, Choi
YM, Kwon TR and Kim BJ: Superoxide dismutase 1 inhibits
alpha-melanocyte stimulating hormone and ultraviolet B-induced
melanogenesis in murine skin. Ann Dermatol. 26:681–687. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Casanola-Martin GM, Le-Thi-Thu H,
Marrero-Ponce Y, Castillo-Garit JA, Torrens F, Rescigno A, Abad C
and Khan MT: Tyrosinase enzyme: 1. An overview on a pharmacological
target. Curr Top Med Chem. 14:1494–1501. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Smit N, Vicanova J and Pavel S: The hunt
for natural skin whitening agents. Int J Mol Sci. 10:5326–5349.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Lee WJ, Bang S, Chung BY, Jung H, Oh ES
and Chang SE: Inhibitory effects of N,N,N-trimethyl
phytosphingosine-iodide on melanogenesis via ERK
activation-mediated MITF degradation. Biosci Biotechnol Biochem.
80:121–127. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ebanks JP, Wickett RR and Boissy RE:
Mechanisms regulating skin pigmentation: The rise and fall of
complexion coloration. Int J Mol Sci. 10:4066–4087. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Desmedt B, Rogiers V, Courselle P, de Beer
JO, De Paepe K and Deconinck E: Development and validation of a
fast chromatographic method for screening and quantification of
legal and illegal skin whitening agents. J Pharm Biomed Anal.
83:82–88. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kwak JY, Seok JK, Suh HJ, Choi YH, Hong
SS, Kim DS and Boo YC: Antimelanogenic effects of luteolin
7-sulfate isolated from Phyllospadix iwatensis Makino. Br J
Dermatol. 175:501–511. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yoon WJ, Kim MJ, Koh HB, Lee WJ, Lee NH
and Hyun CG: Effect of Korean Red Sea Cucumber (Stichopus
japonicus) on Melanogenic Protein Expression in Murine B16
Melanoma. Int J Pharmacol. 6:37–42. 2010. View Article : Google Scholar
|
|
30
|
Slominski A, Moellmann G and Kuklinska E:
L-tyrosine, L-dopa and tyrosinase as positive regulators of the
subcellular apparatus of melanogenesis in Bomirski Ab amelanotic
melanoma cells. Pigment Cell Res. 2:109–116. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Yanase H, Ando H, Horikawa M, Watanabe M,
Mori T and Matsuda N: Possible involvement of ERK 1/2 in
UVA-induced melanogenesis in cultured normal human epidermal
melanocytes. Pigment Cell Res. 14:103–109. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Alam MB, Seo BJ, Zhao P and Lee SH:
Anti-Melanogenic activities of heracleum moellendorffii via
ERK1/2-Mediated MITF Downregulation. Int J Mol Sci. 17:pii:
e18442016. View Article : Google Scholar
|