Introduction
Pathological scarring is one of the most common
complications that occurs following severe wounds and burns, and
remains an important issue that is difficult to treat by plastic
surgery (1). Pathological scars
affect an individual's appearance and may lead to varying degrees
of functional impairment, which may have psychological and economic
consequences for patients. Currently, the mechanisms underlying the
formation of pathological scars are not well established. Current
therapeutic methods for the treatment of pathological scars include
drug therapy, compression therapy, radiotherapy, laser therapy,
biotherapy, gene therapy and surgical treatment; however, the
therapeutic efficacy of these methods requires improvement
(2).
During the wound-healing process, the proliferation
of fibroblasts and deposition of collagen are conducive to wound
healing. However, the hyperproliferation of fibroblasts and
excessive collagen deposition may lead to hypertrophic scar
formation (3). The transforming
growth factor (TGF)-β1 cytokine is closely associated with scar
formation, and overexpression of TGF-β1 has been demonstrated to
promote scar formation (4). Bone
morphogenetic protein-7 (BMP-7) is a member of the TGF-β
superfamily. Increasing evidence suggests that BMP-7 attenuates
TGF-β-induced fibrogenesis in various tissues (5). Bi et al (6) reported that the TGF-β1/BMP-7 ratio
regulated liver fibrosis in rats. An additional study demonstrated
that maintaining BMP-7 levels and inhibiting TGF-β1 attenuates the
progression of renal fibrosis (7).
Furthermore, it was previously demonstrated that BMP-7 inhibits
TGF-β1-induced fibroblast-like differentiation of rat dermal
papilla cells (8). Based on these
reports, the authors of the current study hypothesized that BMP-7
may serve a role in promoting wound healing and attenuating scar
formation.
In the present study, a mouse model of thermal
injury was established. The effect of exogenous BMP-7 treatment on
scar formation and its potential mechanisms were then investigated,
and the results present a novel theoretical basis for the treatment
of skin scars.
Materials and methods
Chemicals and antibodies
Recombinant human BMP-7 was purchased from Cusabio
Biotech Co., Ltd (Wuhan, China) and dissolved in sterile saline
prior to use. The primary antibodies against the following proteins
employed were as follows: Collagen I (BA0323; Wuhan Boster
Biological Technology, Ltd., Wuhan, China); collagen III (BM1625;
Wuhan Boster Biological Technology, Ltd.); α-smooth muscle actin
(α-SMA; BM0002; Wuhan Boster Biological Technology, Ltd.);
connective tissue growth factor (CTGF; ab6992; Abcam, Cambridge,
UK); TGF-β1 (sc-146; Santa Cruz Biotechnology, Inc., Dallas, TX,
USA); B-cell lymphoma 2 (BCL2; BA0412; Wuhan Boster Biological
Technology, Ltd.); BCL2-associated X (Bax; BA0315; Wuhan Boster
Biological Technology, Ltd.); cleaved caspase-3 (ab2302; Abcam);
Smad1/5/8 (sc-6031R; Santa Cruz Biotechnology, Inc.);
phosphorylated (p)-Smad1/5/8 (sc-12353; Santa Cruz Biotechnology,
Inc.); and β-actin (sc-47778; Santa Cruz Biotechnology, Inc.). The
secondary antibodies were HRP-conjugated Goat anti-Rabbit IgG
(WLA023; Wanleibio, Shenyang, China) and HRP-conjugated Goat
anti-Mouse IgG (WLA024; Wanleibio, Shenyang, China).
Animals and experimental protocol
C57BL/6 mice were purchased from Vital River
Laboratories Co., Ltd. (Beijing, China). The mice (6 mice/cage)
were reared at 22±1°C and 45–55% humidity, fed standard laboratory
chow and had free access to food and water throughout the
experiment. A total of 30 male C57BL/6 mice (18–20 g, 8 weeks) were
randomly divided into the following five groups (n=6/group):
Control, thermal injury, thermal injury + BMP-7 (25 µg), thermal
injury + BMP-7 (50 µg), and thermal injury + BMP-7 (100 µg). The
mouse model of thermal injury was established according to Walker's
method with some modifications (9). Briefly, the mice were anesthetized by
intraperitoneal injection of 10% chloral hydrate (3.5 ml/kg). The
hair on the back was then removed to expose the dorsal skin.
Following disinfection, a 2-cm diameter plastic pipe was placed
directly onto the dorsal skin, with one end was in close contact
with the skin and the other was injected with boiling water. The
boiling water remained in contact with the skin for 10 sec. To
prevent infection, the mice that received thermal injury were
housed individually, and the mice in the thermal injury + BMP-7 (25
µg), thermal injury + BMP-7 (50 µg) and thermal injury + BMP-7 (100
µg) groups were subcutaneously injected once with BMP-7 at the
respective dosages immediately after the treatment with boiling
water. Mice in the control and thermal injury groups were
subcutaneously injected with an equal volume of sterile saline. All
mice were euthanized at 15 days following thermal injury, and
scalded skin tissues were obtained for subsequent experiments. All
animal experiments were performed in strict accordance with
international ethical guidelines (10) and the National Institutes of Health
Guide concerning the Care and Use of Laboratory Animals (11). All animal experiments were approved
by the Institutional Animal Care and Use Committee of Jilin
University (Changchun, China).
Wound healing observation
At 0, 3, 7, 10 and 14 days following thermal injury,
the healing of scalded skin in mice from all experimental groups
was observed and photographed. The wound area was measured using
Photoshop CS5 software (Adobe Systems, Inc., Beijing, China).
Histological analysis
The scar tissues and normal skin tissues were
collected at 15 days following thermal injury, and were fixed in 4%
paraformaldehyde for 24 h at room temperature. Tissues were washed
in an ethanol series before they were embedded in paraffin and
sectioned at 5-µm thickness. Subsequently, routine hematoxylin and
eosin (H&E) and Masson's trichrome staining were performed
according to standard procedures (12). Morphological alterations in scar
tissues were determined by H&E staining, and scar tissue
fibrosis was evaluated by Masson's trichrome staining.
Representative images were observed and photographed under a light
microscope at ×200 magnification.
Western blotting
The protein expression levels in scar tissues were
determined by western blot analysis. Briefly, total protein in
tissues from different groups was extracted using a lysis buffer
(Beyotime Institute of Biotechnology, Haimen, China). Following
centrifugation at 10,000 × g and 4°C for 10 min, the concentration
of protein was determined using a BCA kit (Beyotime Institute of
Biotechnology). Equal quantities of protein (40 µg) were
subsequently separated by 8, 10 or 13% SDS-PAGE and transferred to
polyvinylidene difluoride membranes (EMD Millipore, Billerica, MA,
USA). The membranes were then blocked with 5% skimmed milk for 1 h
at room temperature, before they were incubated with anti-collagen
I (1:400), anti-collagen III (1:400), anti-α-SMA (1:400), anti-CTGF
(1:1,000), anti-TGF-β1 (1:200), anti-BCL2 (1:400), anti-Bax
(1:400), anti-cleaved caspase-3 (1:1,000), anti-Smad1/5/8 (1:200),
anti-p-Smad1/5/8 (1:200) and anti-β-actin (1:1,000) primary
antibodies at 4°C overnight. Membranes were subsequently incubated
with goat anti-rabbit/mouse secondary antibodies (1:5,000) for 45
min at 37°C. The bands were visualized using an enhanced
chemiluminescence detection reagent (Beyotime Institute of
Biotechnology). The bands were photographed and the optical
densities were quantified using Gel-Pro Analyzer software version
4.5 (Media Cybernetics, Bethesda, USA).
TUNEL staining
The level of apoptosis in scar tissues was assessed
by TUNEL staining using an In Situ Cell Death Detection kit
(Roche; Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) according to
the manufacturer's instructions. Briefly, scar tissues were washed
in three times in phosphate-buffered saline and fixed in 4%
paraformaldehyde for 12 h at room temperature. Tissues were
subsequently permeabilized by incubating with 0.1% Triton X-100 for
8 min at room temperature. Slides were then incubated with 50 µl
TUNEL reaction mixture (enzyme solution:label solution=1:9) for 60
min at 37°C in the dark. Following washing with PBS for 5 min three
times, the slides were stained with 50 µl 3′3 diaminobenzidine
(DA1010; Beijing Solarbio Science & Technology Co., Ltd.,
Beijing, China) at room temperature and immediately placed in water
just as it became dark. Then the slides were washed for 5 min three
times, observed and imaged at ×400 magnification under a light
microscope, and the number of apoptotic cells was determined.
Statistical analysis
GraphPad Prism software (version, 5.0; GraphPad
Software, Inc., La Jolla, CA, USA) was used for statistical
analyses. All data are presented as the mean + standard deviation.
One-way analysis of variance followed by Bonferroni's post hoc test
was performed to compare the results among groups. P<0.05 was
considered to indicate a statistically significant difference.
Results
BMP-7 inhibits scar formation in a
mouse model of thermal injury
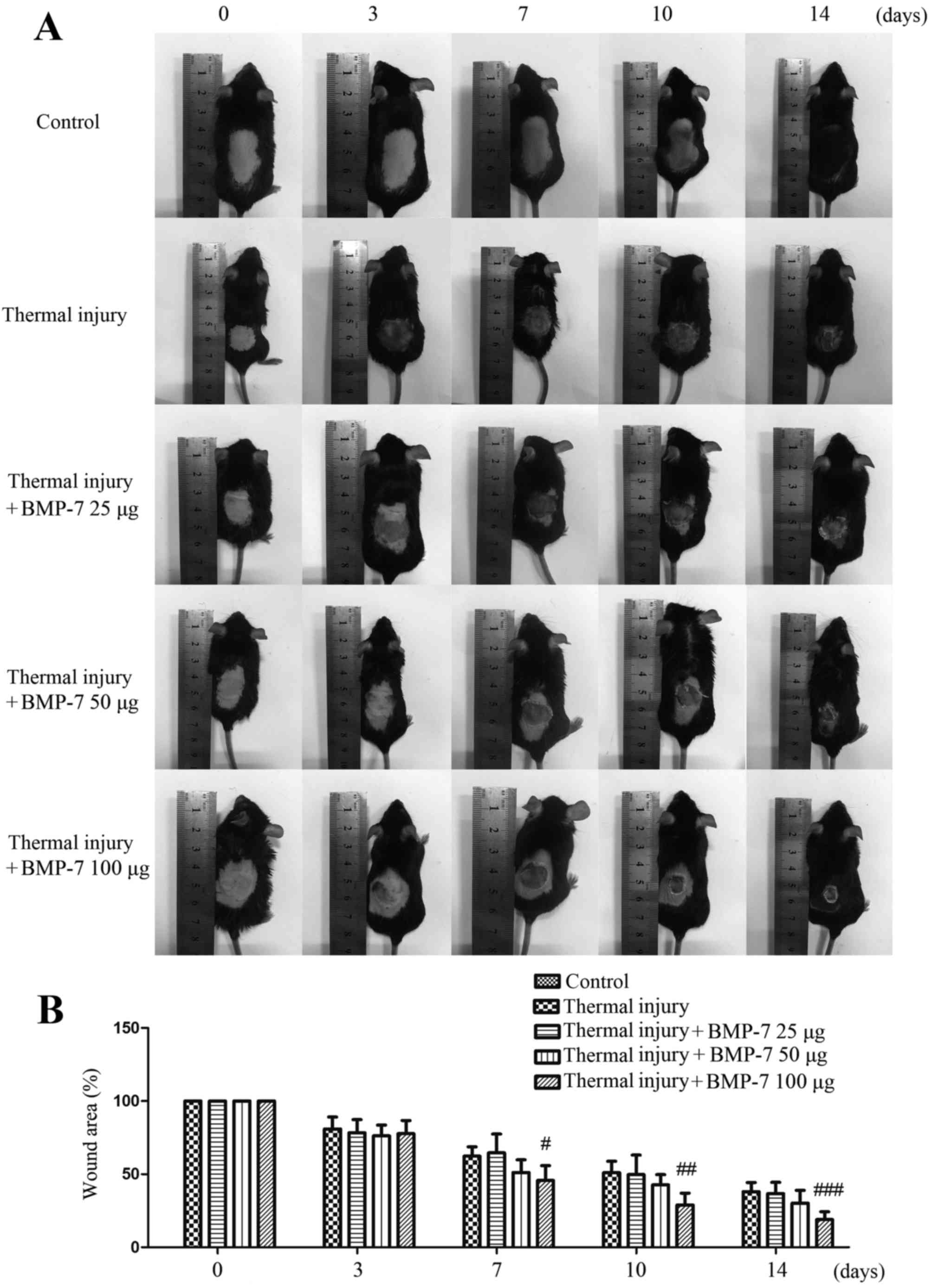
The mouse model of thermal injury was first
established in order to evaluate the effect of BMP-7 on scar
formation. As demonstrated in Fig.
1, the scalded wound surface of mice in each experimental group
was examined. Among the three different doses of BMP-7 tested, 100
µg significantly decreased wound size and inhibited scar formation
at 7, 10 and 14 days following thermal injury when compared with
the untreated thermal injury group.
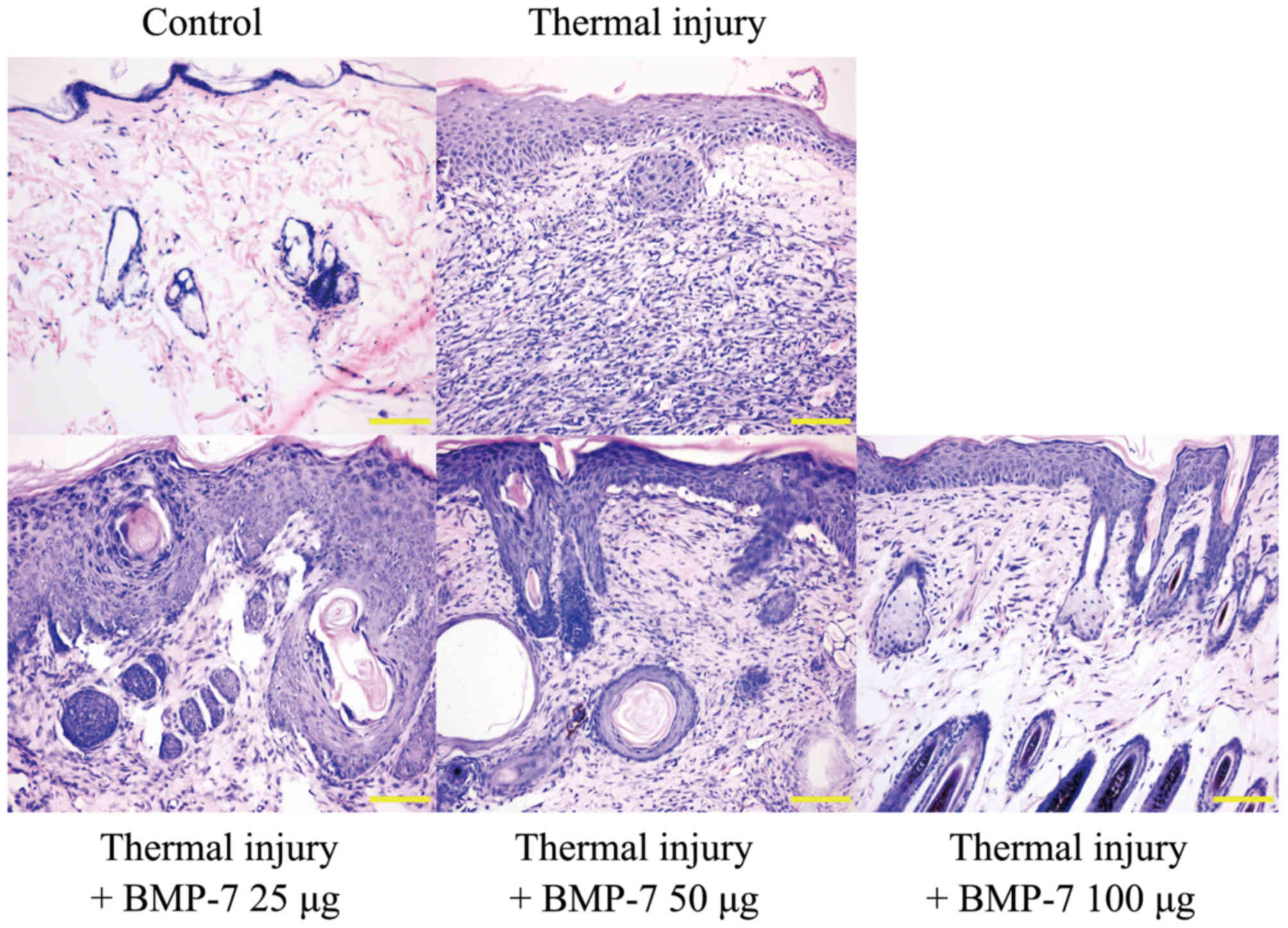
H&E staining was used to examine
histopathological morphologic alterations in the scalded tissues.
The results demonstrated that the epidermis and dermis in the
scalded skin tissue were thicker when compared with the control
group (Fig. 2). In addition, there
was obvious inflammatory cell infiltration, and the skin suffered
structural damage to a certain degree. However, treatment with
BMP-7 attenuated these histopathological morphologic alterations
when compared with tissues from the untreated thermal injury group
(Fig. 2). These results indicate
that BMP-7 demonstrates a potential inhibitory effect on scar
formation.
BMP-7 suppresses collagen deposition
and fibrotic protein expression in scar tissues
Collagen deposition in the scar tissues of mice from
each experimental group was then determined. Masson's trichrome
staining was performed to evaluate collagen fiber area density and
arrangement in scar tissues. As demonstrated in Fig. 3A, the collagen fibers were stained
blue. BMP-7 treatment was associated with a reduced collagen area
in scar tissues when compared with the untreated thermal injury
group. The arrangement of collagen fibers in the scar tissues of
mice from all BMP-7-treated groups was more loose and regular when
compared with mice in the untreated thermal injury group.
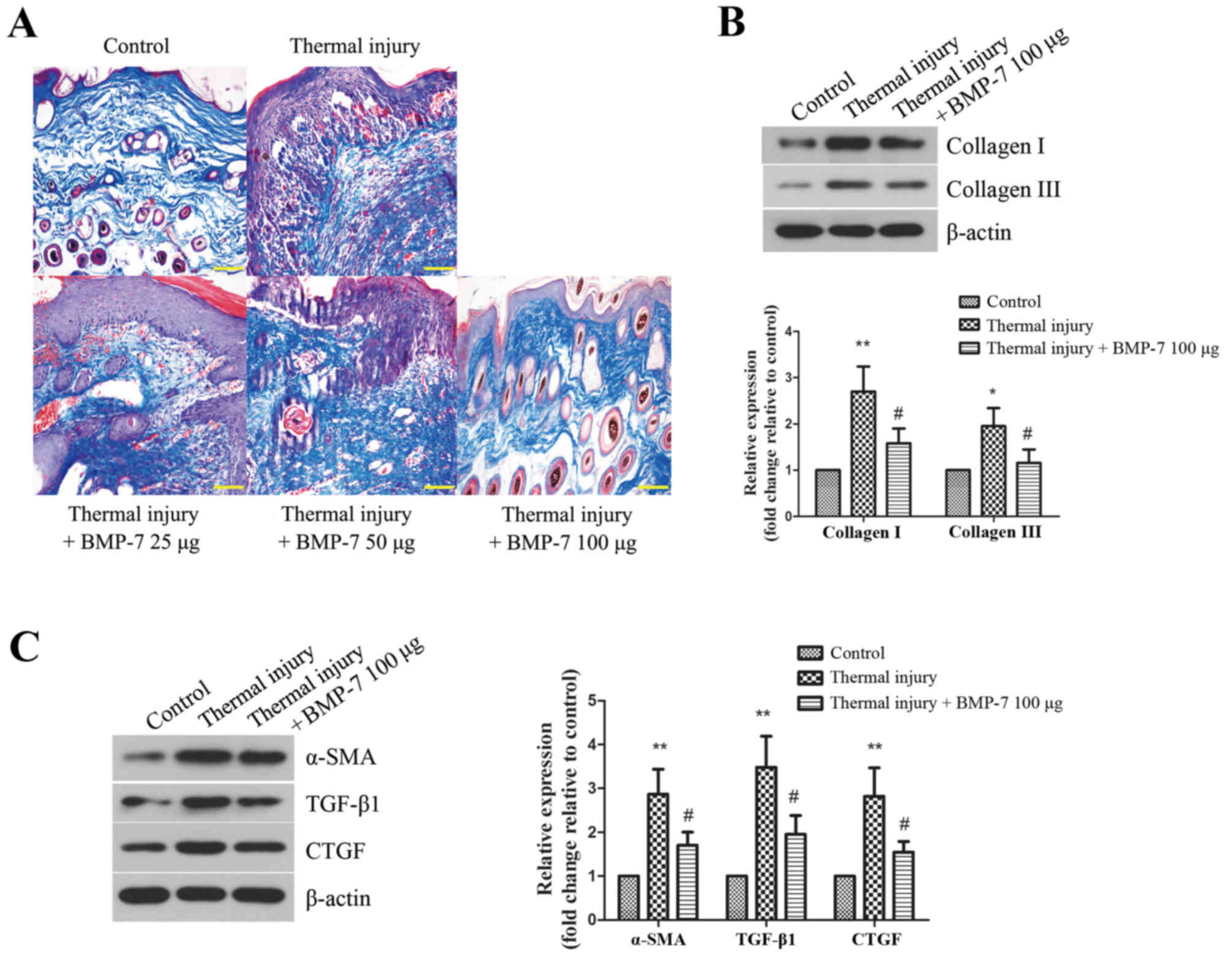 | Figure 3.BMP-7 suppresses collagen deposition
and fibrotic protein expression in scar tissues. (A) Masson's
trichrome staining was performed to examine collagen fibers in skin
tissues of mice from the different groups (magnification, ×200;
scale bars, 100 µm). (B) The protein expression levels of collagen
I and collagen III were determined by western blot analysis, and
protein levels were quantified by densitometric analysis. β-actin
was used as a loading control. (C) Protein expression levels of
fibrosis-associated proteins, α-SMA, TGF-β1 and CTGF, as determined
by western blot analysis. Protein levels were quantified by
densitometric analysis. The results are presented as the mean +
standard deviation (n=6), and are representative of three
independent experiments. *P<0.05 and **P<0.01 vs. control
group; #P<0.05 vs. untreated thermal injury group.
BMP-7, bone morphogenetic protein-7; α-SMA, α-smooth muscle actin;
TGF, transforming growth factor; CTGF, connective tissue growth
factor. |
Based on the above results, BMP-7 with the dose of
100 µg had the most inhibitory effect on scarring. Therefore, 100
µg BMP-7 was chosen for the following experiments. The protein
expression levels of collagen I and collagen III were detected by
western blot analysis. As demonstrated in Fig. 3B, the levels of collagen I and III
were increased significantly in the thermal injury group when
compared with the control group. By contrast, treatment with 100 µg
BMP-7 significantly reduced collagen I and III protein expression
levels when compared with the thermal injury group.
α-SMA, TGF-β1 and CTGF are proteins that are
involved in promoting fibrosis, which leads to scar formation
(13). In the present study, the
expression levels of these proteins in mice from all experimental
groups were determined by western blot analysis. As demonstrated in
Fig. 3C, thermal injury induced a
significant increase in the protein expression levels of α-SMA,
TGF-β1 and CTGF when compared with the control group. By contrast,
treatment with 100 µg BMP-7 significantly reduced the protein
expression levels of α-SMA, TGF-β1 and CTGF when compared with the
mice in the untreated thermal injury group.
BMP-7 induces fibroblast apoptosis in
scar tissues
The level of apoptosis in scar tissues was
determined using a TUNEL assay. As demonstrated in Fig. 4A, the level of TUNEL-positive
staining was lower in the scar tissues of mice in the thermal
injury group compared with those of the control group. By contrast,
treatment with 100 µg BMP-7 markedly enhanced the level of
apoptosis in scar tissues when compared with the thermal injury
group (Fig. 4A). In order to
validate these observations, the expression of apoptosis-associated
proteins was then determined by western blot analysis. As
demonstrated in Fig. 4B, treatment
with 100 µg BMP-7 significantly decreased the protein expression
levels of BCL2, and significantly increased the levels of Bax and
cleaved caspase-3 when compared with the thermal injury group.
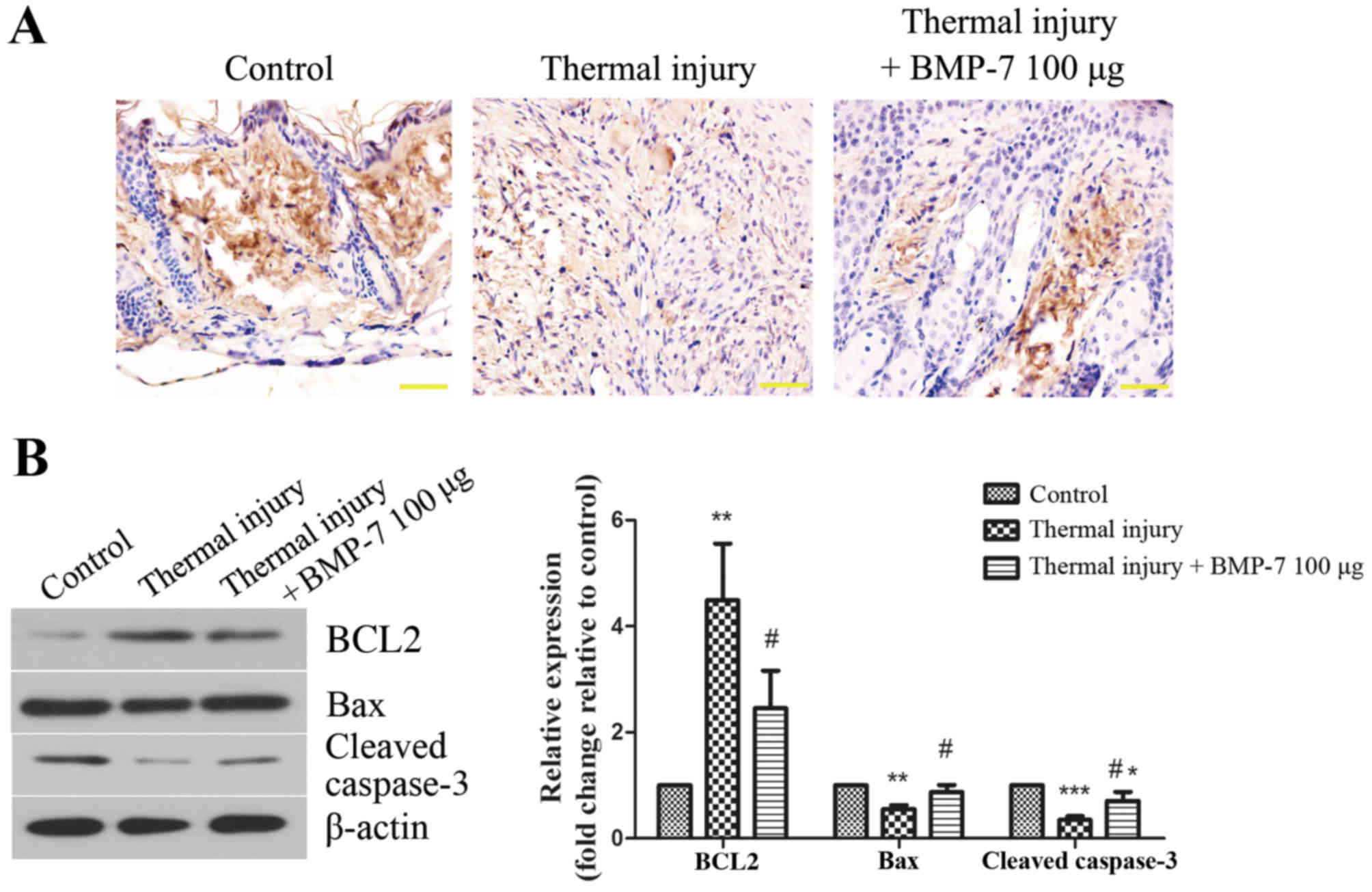 | Figure 4.BMP-7 induces fibroblast apoptosis in
scar tissues. (A) Effect of BMP-7 on apoptosis in scar tissues was
evaluated using a TUNEL assay (magnification, ×400; scale bars, 50
µm). (B) The protein expression levels of apoptosis-associated
proteins, BCL2, Bax and cleaved caspase-3 were assessed by western
blot analysis, and protein levels were quantified by densitometric
analysis. β-actin was used as a loading control. The results are
presented as the mean + standard deviation (n=6), and are
representative of three independent experiments. *P<0.05,
**P<0.01 and ***P<0.001 vs. control group;
#P<0.05 vs. untreated thermal injury group. BMP-7,
bone morphogenetic protein-7; BCL2, B-cell lymphoma 2; Bax,
BCL2-associated X. |
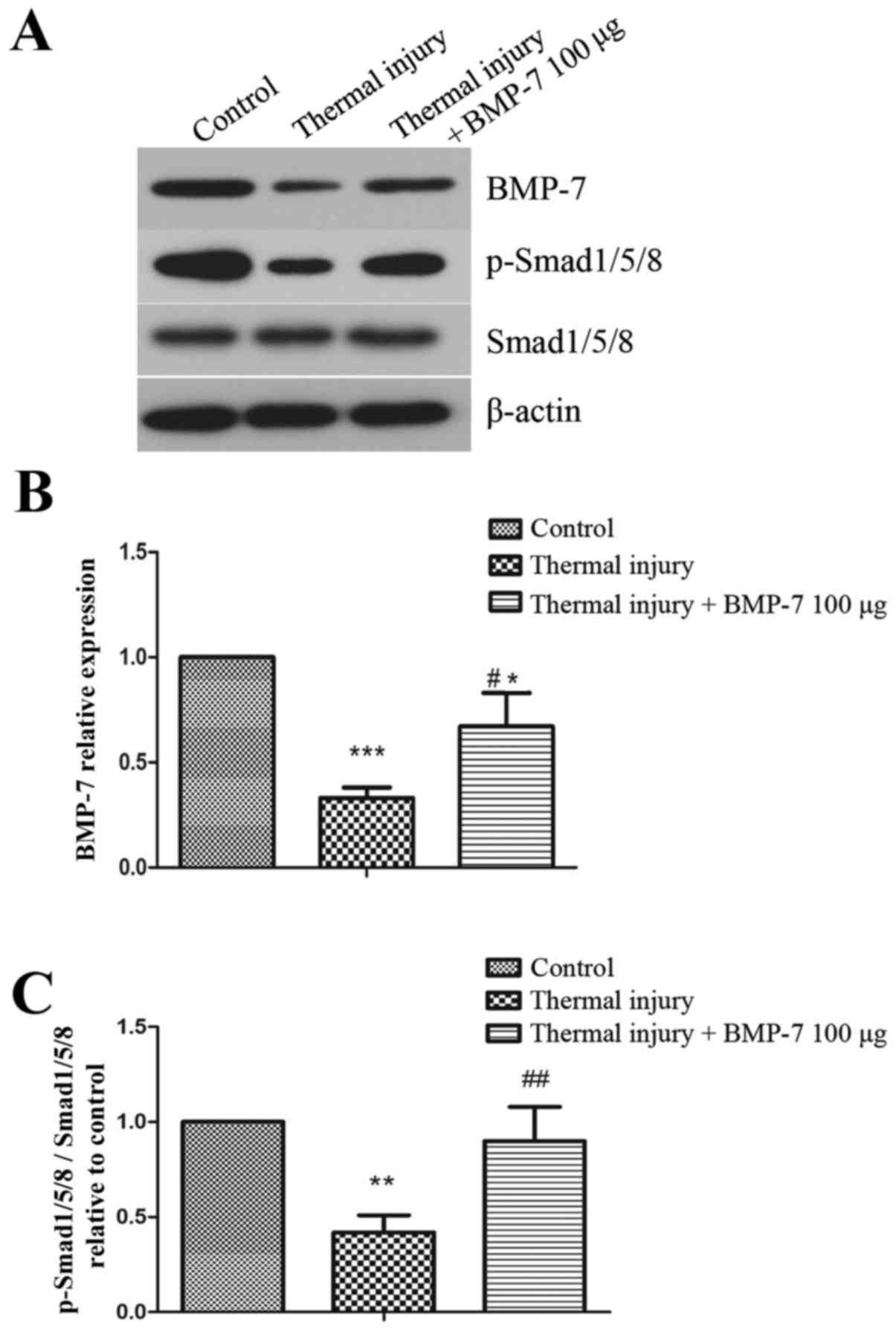
BMP-7 activates the BMP-7/Smad1/5/8
signaling pathway
In order to investigate the molecular mechanisms
underlying BMP-7-mediated suppression of scar formation, the
present study examined the BMP-7/Smad1/5/8 signaling pathway. As
demonstrated in Fig. 5, the
protein expression levels of BMP-7 and p-Smad1/5/8 were
significantly decreased in scar tissues induced by thermal injury
when compared with the control group. However, injection of
exogenous 100 µg BMP-7 significantly enhanced the level of
p-Smad1/5/8 when compared with the untreated thermal injury
group.
Discussion
To the best of the author's knowledge, the present
study is the first to report that BMP-7 treatment suppresses scar
formation in a mouse model of thermal injury. Pathological scarring
is a common fibrotic disease that occurs during wound healing
(14). Currently, there are
numerous treatments with clearly proven efficiency or doubtful
benefits for this condition. However, the optimal treatment has yet
to be identified. Therefore, investigating novel therapeutic
methods and understanding the underlying mechanisms is important
for improving treatment strategies for patients with pathological
scarring. BMP-7 has been reported to inhibit TGF-β1-induced
epithelial-mesenchymal transition, and has therefore been
identified as an anti-fibrosis cytokine with potential applications
in the treatment of fibrotic diseases (15). Until recently, the effect of BMP-7
on scar formation was unknown. In the present study, recombinant
human BMP-7 was injected into the scalded skin of mice, and the
effect on scar formation and the underlying mechanisms were
investigated. The results demonstrated that exogenous BMP-7
promoted wound healing and inhibited scar formation. This provides
a novel insight into the molecular role of BMP-7 in skin wound
healing, and indicates that BMP-7 may present a potential
therapeutic target for the treatment of pathological scarring.
It is generally accepted that hypertrophic scars are
primarily characterized by disturbances in collagen metabolism.
Collagen, as the major component of extracellular matrix, is the
structural and functional protein of skin. However, upon serious
trauma, collagen deposits in hypertrophic scar fibroblasts and the
expression of collagen I and III are increased (16). The results of the present study
demonstrated that BMP-7 treatment reduced excessive collagen
deposition in the scar tissues induced by thermal injury, and the
thermal injury-induced increases in the expression of collagen I
and III were significantly inhibited by BMP-7 treatment. The
cytoskeletal protein, α-SMA, is a typical marker of a contractile
phenotype in myofibroblasts (17).
The skin exhibits excessive contraction during wound healing as a
result of α-SMA overexpression at the site of injury, which finally
induces scar formation (18,19).
Therefore, α-SMA is considered to be closely associated with scar
contracture, and inhibiting the production of α-SMA is conducive to
preventing scarring during wound repair. According to the results
of the current study, the protein expression levels of α-SMA were
upregulated in thermal injury-induced scar tissues, which was
significantly inhibited by BMP-7 treatment. Skin fibroblasts
synthesize and secrete CTGF, which demonstrates a strong regulatory
effect on the proliferation and differentiation of fibroblasts
(20). As abnormally high
expression of CTGF is known to serve a key role in the development
of scar formation, CTGF has become a focus of anti-scarring
research (21). In addition, CTGF
is a direct downstream effector of TGF-β1, and mediates the
profibrogenic effects of TGF-β1 in various diseases, such as
cicatrix, which is characterized by the scarring of healed wounds
(22–24). The results of the present study
demonstrated that BMP-7 significantly inhibited the expression of
CTGF in thermal injury-induced wound tissues. Taking into account
the results of previous studies, the anti-fibrotic effects of BMP-7
were significant, and may have therefore been the primary mechanism
for inhibiting scar formation.
Apoptosis is a programmed cell death mechanism. A
previous study has demonstrated that the level of apoptosis in
pathological scar-derived fibroblasts is lower when compared with
normal skin (25). An additional
report indicated that the imbalance between fibroblast
proliferation and apoptosis is an important factor in scar
formation (26). Therefore,
inducing apoptosis in fibroblasts may be an effective strategy for
the treatment of pathological scarring. The results of the present
study demonstrated that the level of apoptosis in scar tissues was
increased significantly following treatment with BMP-7 when
compared with untreated controls. The BCL2 protein family regulates
cell apoptosis, and involves two types of functionally opposing
proteins: Anti-apoptotic proteins, such as BCL2, and pro-apoptotic
proteins, such as Bax. The BCL2/Bax ratio is a major factor for the
regulation of apoptosis. Caspase-3 is an important member of the
caspase cascade reaction, and functions as a key downstream
regulatory protein in apoptotic signaling pathways. Activation of
caspase-3 is responsible for the induction of the majority of
apoptotic signaling pathways (27). The results of the current study
demonstrated that BMP-7 treatment significantly reduced the
BCL2/Bax ratio and increased the expression of cleaved caspase-3
when compared with the untreated thermal injury group. In addition,
the results indicated that the level of apoptosis in scar tissues
from mice in the thermal injury group were reduced when compared
with the control group, which may have been due to the
hyperproliferation of fibroblasts in scar tissues. However, the
molecular mechanisms underlying this reduction in apoptosis in scar
tissues requires further investigation.
The Smad signaling pathway serves important roles in
collagen metabolism, and has been confirmed as one of the most
extensively studied signaling pathways in scar formation. Among
different Smad proteins, p-Smad1/5/8 is used as a marker of
BMP/Smad signaling (28). BMP-7
serves a role in preventing fibrosis by activating the BMP/Smad
signaling pathway in various diseases. Yang et al (28) demonstrated that BMP-7 inhibited
silica-induced pulmonary fibrosis in rats by upregulating the
expression of p-Smad1/5/8. In addition, p-Smad1/5/8 was
demonstrated to be involved in BMP-7 signaling pathway-induced
anti-fibrotic effects on fibrous capsule formation on the surface
of orthopedic metallic prosthetic implants (29). Furthermore, increased expression of
p-Smad1/5/8 was observed in mediating the anti-fibrosis effects of
BMP-7 in the cornea (30) and lung
(31). Consistent with the results
of these studies, the present study demonstrated that BMP-7
treatment significantly increased the expression of p-Smad1/5/8 at
thermal injury sites when compared with untreated controls.
In conclusion, the present study provides evidence
that BMP-7 promotes wound healing, inhibits collagen formation and
the progression of fibrosis, and induces fibroblast apoptosis.
Activation of the BMP/Smad signaling pathway may be the underlying
mechanism responsible for the suppressive effects of BMP-7 on scar
formation. Although the effect of BMP-7 on scar formation and its
potential mechanisms require further research, the authors suggest
that BMP-7 may be a potential therapeutic target for the treatment
of patients with pathological scarring.
References
|
1
|
Sun X, Cheng L, Zhu W, Hu C, Jin R, Sun B,
Shi Y, Zhang Y and Cui W: Use of ginsenoside Rg3-loaded electrospun
PLGA fibrous membranes as wound cover induces healing and inhibits
hypertrophic scar formation of the skin. Colloids Surf B
Biointerfaces. 115:61–70. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Tredget EE, Levi B and Donelan MB: Biology
and principles of scar management and burn reconstruction. Surg
Clin North Am. 94:793–815. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zeng G, Zhong F, Li J, Luo S and Zhang P:
Resveratrol-mediated reduction of collagen by inhibiting
proliferation and producing apoptosis in human hypertrophic scar
fibroblasts. Biosci Biotechnol Biochem. 77:2389–2396. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Jagadeesan J and Bayat A: Transforming
growth factor beta (TGFbeta) and keloid disease. Int J Surg.
5:278–285. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Weiskirchen R and Meurer SK: BMP-7
counteracting TGF-beta1 activities in organ fibrosis. Front Biosci
(Landmark Ed). 18:1407–1434. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bi WR, Xu GT, Lv LX and Yang CQ: The ratio
of transforming growth factor-β1/bone morphogenetic protein-7 in
the progression of the epithelial-mesenchymal transition
contributes to rat liver fibrosis. Genet Mol Res. 13:1005–1014.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tasanarong A, Kongkham S, Thitiarchakul S
and Eiam-Ong S: Vitamin E ameliorates renal fibrosis in ureteral
obstruction: Role of maintaining BMP-7 during
epithelial-to-mesenchymal transition. J Med Assoc Thai. 94 Suppl
7:S10–S18. 2011.PubMed/NCBI
|
|
8
|
Bin S, Li HD, Xu YB, Qi SH, Li TZ, Liu XS,
Tang JM and Xie JL: BMP-7 attenuates TGF-β1-induced fibroblast-like
differentiation of rat dermal papilla cells. Wound Repair Regen.
21:275–281. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Walker HL and Mason AD Jr: A standard
animal burn. J Trauma. 8:1049–1051. 1968. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Tian X, Zheng Y, Li Y, Shen Z, Tao L, Dou
X, Qian J and Shen H: Psychological stress induced zinc
accumulation and up-regulation of ZIP14 and metallothionein in rat
liver. BMC Gastroenterol. 14:322014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhang M, Sun G, Shen A, Liu L, Ding J and
Peng J: Patrinia scabiosaefolia inhibits the proliferation of
colorectal cancer in vitro and in vivo via G1/S cell cycle arrest.
Oncol Rep. 33:856–860. 2015.PubMed/NCBI
|
|
12
|
Apgar JM, Juarranz A, Espada J, Villanueva
A, Cañete M and Stockert JC: Fluorescence microscopy of rat embryo
sections stained with haematoxylin-eosin and Masson's trichrome
method. J Microsc. 191:20–27. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Mun JH, Kim YM, Kim BS, Kim JH, Kim MB and
Ko HC: Simvastatin inhibits transforming growth factor-β1-induced
expression of type I collagen, CTGF and α-SMA in keloid
fibroblasts. Wound Repair Regen. 22:125–133. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kopecki Z and Cowin AJ: Flightless I: An
actin-remodelling protein and an important negative regulator of
wound repair. Int J Biochem Cell Biol. 40:1415–1419. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Weiskirchen R, Meurer SK, Gressner OA,
Herrmann J, Borkham-Kamphorst E and Gressner AM: BMP-7 as
antagonist of organ fibrosis. Front Biosci (Landmark Ed).
14:4992–5012. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ghahary A, Shen YJ, Scott PG, Gong Y and
Tredget EE: Enhanced expression of mRNA for transforming growth
factor-beta, type I and type III procollagen in human post-burn
hypertrophic scar tissues. J Lab Clin Med. 122:465–473.
1993.PubMed/NCBI
|
|
17
|
Muchaneta-Kubara EC and Nahas AM:
Myofibroblast phenotypes expression in experimental renal scarring.
Nephrol Dial Transplant. 12:904–915. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Agarwal C, Britton ZT, Alaseirlis DA, Li Y
and Wang JH: Healing and normal fibroblasts exhibit differential
proliferation, collagen production, alpha-SMA expression, and
contraction. Ann Biomed Eng. 34:653–659. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Nedelec B, Ghahary A, Scott PG and Tredget
EE: Control of wound contraction. Basic and clinical features. Hand
Clin. 16:289–302. 2000.PubMed/NCBI
|
|
20
|
Oemar BS and Lüscher TF: Connective tissue
growth factor. Friend or foe? Arterioscler Thromb Vasc Biol.
17:1483–1489. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Grotendorst GR, Okochi H and Hayashi N: A
novel transforming growth factor beta response element controls the
expression of the connective tissue growth factor gene. Cell Growth
Differ. 7:469–480. 1996.PubMed/NCBI
|
|
22
|
Bradham DM, Igarashi A, Potter RL and
Grotendorst GR: Connective tissue growth factor: A cysteine-rich
mitogen secreted by human vascular endothelial cells is related to
the SRC-induced immediate early gene product CEF-10. J Cell Biol.
114:1285–1294. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yatomi M, Hisada T, Ishizuka T, Koga Y,
Ono A, Kamide Y, Seki K, Aoki-Saito H, Tsurumaki H, Sunaga N, et
al: 17(R)-resolvin D1 ameliorates bleomycin-induced pulmonary
fibrosis in mice. Physiol Rep. 3:pii: e126282015. View Article : Google Scholar
|
|
24
|
Song R, Li G and Li S: Aspidin PB, a novel
natural anti-fibrotic compound, inhibited fibrogenesis in
TGF-β1-stimulated keloid fibroblasts via PI-3K/Akt and Smad
signaling pathways. Chem Biol Interact. 238:66–73. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang H, Chen Z, Li XJ, Ma L and Tang YL:
Anti-inflammatory cytokine TSG-6 inhibits hypertrophic scar
formation in a rabbit ear model. Eur J Pharmacol. 751:42–49. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Akasaka Y, Fujita K, Ishikawa Y, Asuwa N,
Inuzuka K, Ishihara M, Ito M, Masuda T, Akishima Y, Zhang L, et al:
Detection of apoptosis in keloids and a comparative study on
apoptosis between keloids, hypertrophic scars, normal healed flat
scars, and dermatofibroma. Wound Repair Regen. 9:501–506. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Liu Q, Si T, Xu X, Liang F, Wang L and Pan
S: Electromagnetic radiation at 900 MHz induces sperm apoptosis
through bcl-2, bax and caspase-3 signaling pathways in rats. Reprod
Health. 12:652015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yang G, Zhu Z, Wang Y, Gao A, Niu P and
Tian L: Bone morphogenetic protein-7 inhibits silica-induced
pulmonary fibrosis in rats. Toxicol Lett. 220:103–108. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Tan HC, Poh CK, Cai Y and Wang W:
Anti-fibrosis effect of BMP-7 peptide functionalization on cobalt
chromium alloy. J Orthop Res. 31:983–990. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Tandon A, Sharma A, Rodier JT, Klibanov
AM, Rieger FG and Mohan RR: BMP7 gene transfer via gold
nanoparticles into stroma inhibits corneal fibrosis in vivo. PLoS
One. 8:e664342013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Murray LA, Hackett TL, Warner SM, Shaheen
F, Argentieri RL, Dudas P, Farrell FX and Knight DA: BMP-7 does not
protect against bleomycin-induced lung or skin fibrosis. PLoS One.
3:e40392008. View Article : Google Scholar : PubMed/NCBI
|