Introduction
Inflammatory bowel disease (IBD), often referred to
as Crohn's disease or ulcerative colitis (UC), is characterized by
gastrointestinal tract inflammation-related disorders (1,2).
While Crohn's disease can affect any part of gastrointestinal
tract, ulcerative colitis only affects the colon and rectum
(3). Although genetic factors or
microbiota dysregulation were reported to be the primary factors
accounting for the occurrence of IBD in current conceptions
(4,5), the detailed pathogenesis of IBD still
remains unknown.
Growing evidence has indicated that disruption of
intestinal microorganism homeostasis will arouse the immune
responses against intracellular bacteria and fungi (6). Even though effector T cells such as T
helper (h) 17 cells, interleukin (IL)-22 producing T cells as well
as the cytokines they produced [IL-17A, IL-22, interferon (IFN)-γ
fight against the pathogens, they may also cause unwanted tissue
destruction, immunopathology and autoimmunity (7–9).
Therefore, therapies should focus on restraining T-cell responses
in IBD.
Regulatory T cells, by constitutively expressing
IL-2 receptor CD25, serve an essential role in maintaining
homeostasis thought constraining the reactivity of effector T cells
as well as blocking pro-inflammatory cytokine production to inhibit
the progression of IBD (10,11).
Similar to conventional Th cells, T regulatory (reg) cells can be
divided into different subsets such as natural Treg cells, induced
Tregs and CD8+ Treg cells characterized by different
patterns of cytokine and chemokine receptors. Currently, the role
of different Treg subsets in modulating inflammatory bowel disease
has still not been addressed carefully and systematically.
Therefore, in the present study, the authors sought
to determine the percentage of different subsets in
well-established dextran sodium sulfate (DSS)-induced colitis
mimicking chronic intestinal inflammation in mice at different time
points. Efforts were also made to evaluate the change of different
Treg subsets at different stage of patients diagnosed with
ulcerative colitis compared with enrolled healthy controls.
Materials and methods
Mice
A total of 30 5-week-old male mice (C57BL/6; weight,
20–22 g) were purchased from Liaoning Changsheng Biotechnology Co.,
Ltd. (Benxi, China) and kept under specific pathogen-free
conditions with free access to food and water and regular 12-h
light/dark cycle in the Experimental Animal Center of China Medical
University (Shenyang, China). All the animal experiments were
conducted according to the Ethnics of China Medical University (no.
KY201511).
Human subjects and ethics
Patients diagnosed of ulcerative colitis and treated
at The First Affiliated Hospital of Jinzhou Medical School
(Jinzhou, China) from August 2014 to August 2015 were recruited
into the study. Age- and sex-matched healthy volunteers were also
enrolled as healthy controls. Patients details are shown in
Table I. Informed consent was
obtained from all the subjects and ethics were approved by The
First Affiliated Hospital of Jinzhou Medical School (Jinzhou,
China; no. KY201511).
 | Table I.Basic characteristics of ulcerative
colitis patients. |
Table I.
Basic characteristics of ulcerative
colitis patients.
|
| Ulcerative colitis
(n=23) |
|---|
| No. of patients
(M/F) | 11/12 |
| Age (year) | 38.6±3.4 |
| C-reactive protein
(mg/l) | 12.1±3.8 |
| No. in acute
stage | 16 |
| No. in remittent
stage | 7 |
Induction of colitis
3% DSS (molecular weight, 4,000Da; Sigma-Aldrich;
Merck KGaA, Darmstadt, Germany) were added in drinking water for 7
days to induce acute colitis in mice. Regular drinking water was
changed from day 8. Mice were euthanized at days 0, 4, 7, 10, 20
and 30, respectively. Mesenteric lymph nodes (MLN), peripheral
blood and the colon were collected at the same time.
Peripheral blood mononuclear cell
(PBMC) isolation
Fresh sodium heparinized blood samples were diluted
by PBS, carefully layered on Ficoll density gradient and
centrifuged at 1,000 × g for 20 min in room temperature (25°C).
PBMCs were isolated from the interface and transferred into new
tubes, washed with PBS for two times.
Lamina proprialymphocytes (LPL)
preparation
Colon tissues were removed of Peyer's patches,
washed with PBS, cut into 1–2 mm small pieces and shaken in RPMI
1640 (Sigma-Aldrich; Merck KGaA) supplemented with fetal bovine
serum (Sigma-Aldrich; Merck KGaA) and 5 mM EDTA (Sigma-Aldrich;
Merck KGaA) at 100 × g and 25°C for 20 min. The supernatants were
discarded to remove epithelial cells and intraepithelial
lymphocytes. The remained fragments were then incubated with 5 ml 1
mg/ml collagenase type IV (Sigma-Aldrich; Merck KGaA), shaken at
100 × g and 25°C for between 45 min to 1 h. The procedure was
repeated twice. Supernatant of digested solution were collected,
carefully layered on 45/75% discontinuous Percoll gradients (GE
Healthcare Life Sciences, Chalfont, UK) and centrifuged at 800 × g
for 20 min at room temperature. LPLs were harvested, washed with
PBS and suspended in RPMI 1640 medium.
Flow cytometry
Human fluorescein isothiocyanate-conjugated
anti-CD25, phycoerythrin (PE)-conjugated transforming growth factor
(TGF)-β, peridinin chlorophyll protein complex-conjugated anti-CD4,
Alexa Fluor 647 conjugated Foxp3, PE-Cy7-conjugated IL-10,
BV421-conjugated anti-CD127 and APC/Cy7-conjugated anti-CXCR5 were
purchased from BD Pharmingen (San Diego, CA, USA). Isolated
mononuclear cells were stained with various antibody cocktails at
4°C for 30 min, then washed with PBS. For intracellular staining,
cells were washed, fixed, permeabilized and stained with
intracellular antibody following manufacturer's instructions (BD
Biosciences, Franklin Lakes, NJ, USA). Stained cells were detected
using FACSCalibur flow cytometry (BD Biosciences). The results were
analyzed using FlowJo software (version 9; FlowJo, LLC, Ashland,
OR, USA).
ELISA
Serum cytokine levels were analyzed using the
following commercially available ELISA kits according to the
manufacturer's protocol: MMP-2, Total MMP-2 Quantikine ELISA kit
(cat. no. MMP200; Quantikine, R&D Systems, Inc., Minneapolis,
MD, USA); thymic stromal lymphopoietin, Legend Max™ Human TSLP
ELISA kit with Pre-coated Plates (cat. no. 434208; BioLegend, Inc.,
San Diego, CA, USA); MMP-9, Legend Max™ Human MMP-9 ELISA kit with
Pre-coated Plates (cat. no. 440707; BioLegend, Inc.); IL-17A,
Legend Max™ Human IL-17A ELISA kit with Pre-coated Plates (cat. no.
433918; BioLegend, Inc.); IL-25, Human IL-17E/IL-25 AccuSignal
ELISA kit (cat. no. KOA0468; Rockland Immunochemicals, Inc.,
Limerick, PA, USA); and eotaxin, Human Eotaxin ELISA kit (cat. no.
KOA0159; Rockland Immunochemicals, Inc.).
Statistical analysis
Comparisons between two groups were performed using
Student's t-test and comparisons among multiple groups were
performed using one-way analysis of variance followed by Tukey's
post-hoc test. Data are expressed as the mean ± standard error of
the mean. P<0.05 was considered to indicate as statistically
significant difference.
Results
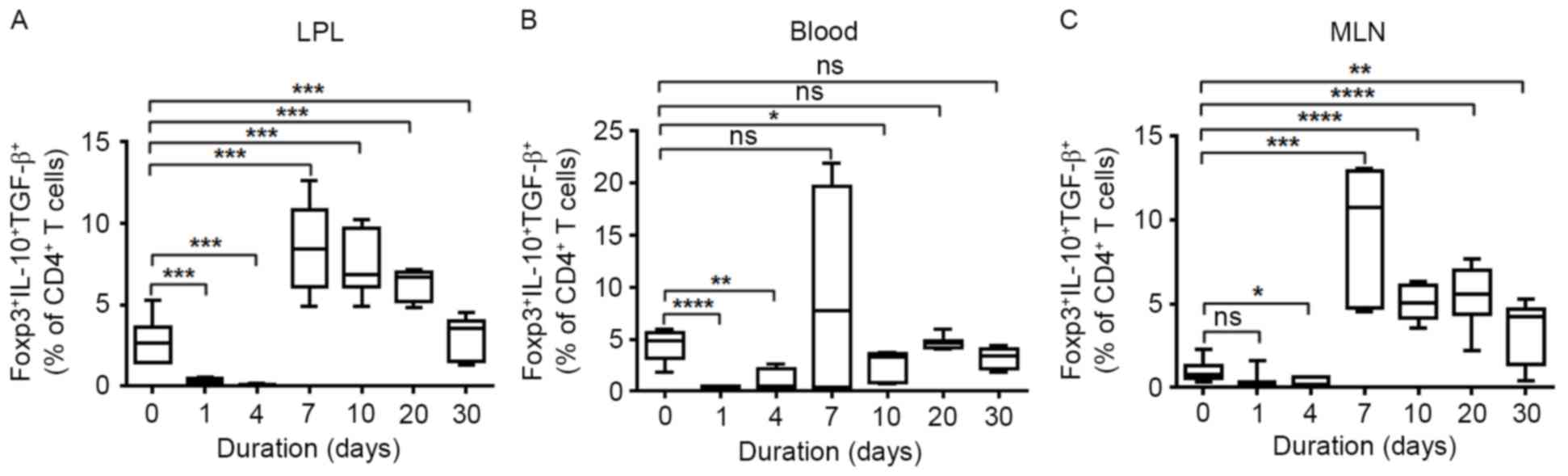
Changes of natural regulatory T-cell
subsets in DSS-induced colitis
The best-characterized regulatory T cell population
is the natural regulatory T cell population. To determine whether
the natural regulatory T cell subset serve a role in alleviating
intestinal bowel disease-associated mucosal injury, the authors
measured the percentage of Foxp3+ IL-10+
TGF-β+ natural Tregs at different time points in DSS
drinking water-treated mice. The proportion of nTregs in LPL,
peripheral blood and MLNs was significantly decreased in the acute
phage at days 1 and 4 following DSS water treatment. At 7 days
following oral DSS administration, nTregs in LPLs and MLNs were
significantly elevated compared with the baseline (Fig. 1A and C). Although slightly elevated
nTregs were observed in PBMCs at 7 days, it decreased to a normal
level at the chronic stage of DSS-induced IBD (Fig. 1B).
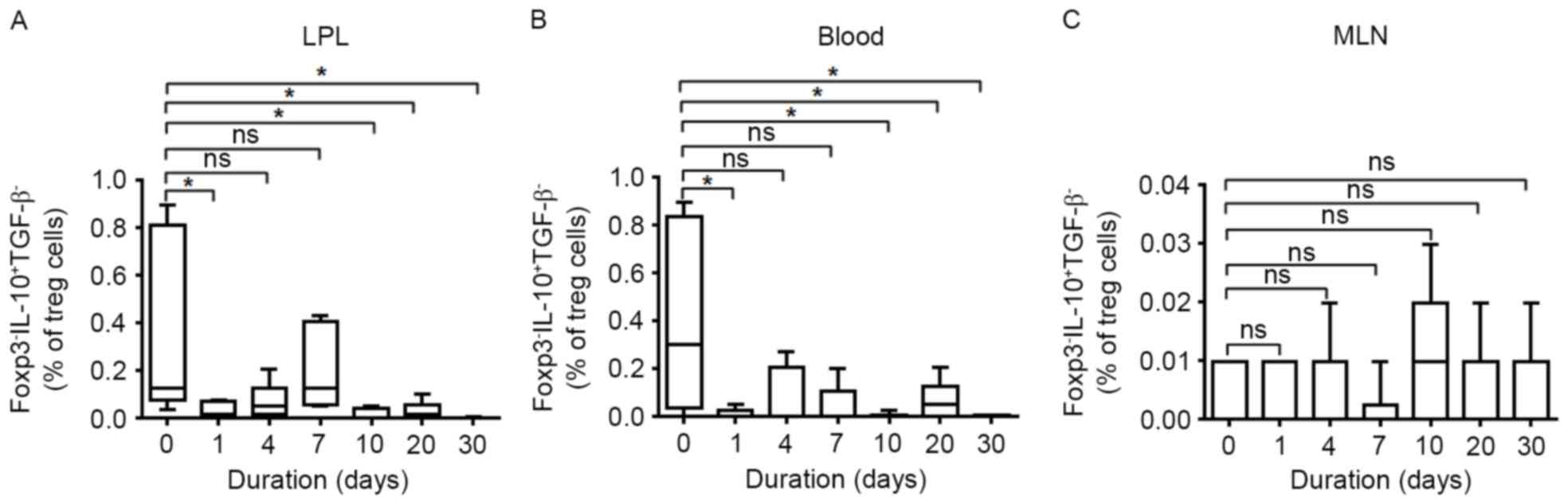
The level of induced regulatory T-cell
subsets in DSS-induced colitis
The frequency of Foxp3− IL-10+
TGF-β− induced Tregs (Tr1) in LPLs, MLNs and PBMCs was
also measured. As presented in Fig.
2, DSS treatment significantly decreased the percentage of Tr1
cells at days 1 and 4. During the recovery time, the frequency of
Tr1 cells continued declining in both LPLs and peripheral blood.
Few changes were observed in MLNs at various time points by
DSS-treatment.
Analysis of CD127−induced
regulatory T-cell frequency in DSS-induced colitis
Following this, the authors determined the change of
CD127−inducible (i) Treg population in DSS-induced
colitis by flow cytometry. As indicated in Fig. 3, a significantly lower percentage
of CD127− Treg was observed in LPLs, MLNs and peripheral
blood at days 1 and 4 following DSS treatment. However, the
frequency of CD127− Treg cells was only slightly
restored but remained at a low level following 7 days in
DSS-treated mice.
The proportion of CD8+
Tregs in DSS-treated mice
As a small proportion of CD8+ Tregs were
also able to inhibit hyper-responsive effective T cells in
autoimmune diseases, efforts were made to determine the role of
CD8+ Tregs in DSS-induced murine IBD. The proliferation
of CD8+ Tregs was only enhanced in LPL at 7 days
following DSS treatment, but reduced to a normal level at days 10,
20 and 30 and no significant differences in MLNs and PBMCs was
discovered at any stage compared with the baseline (Fig. 4).
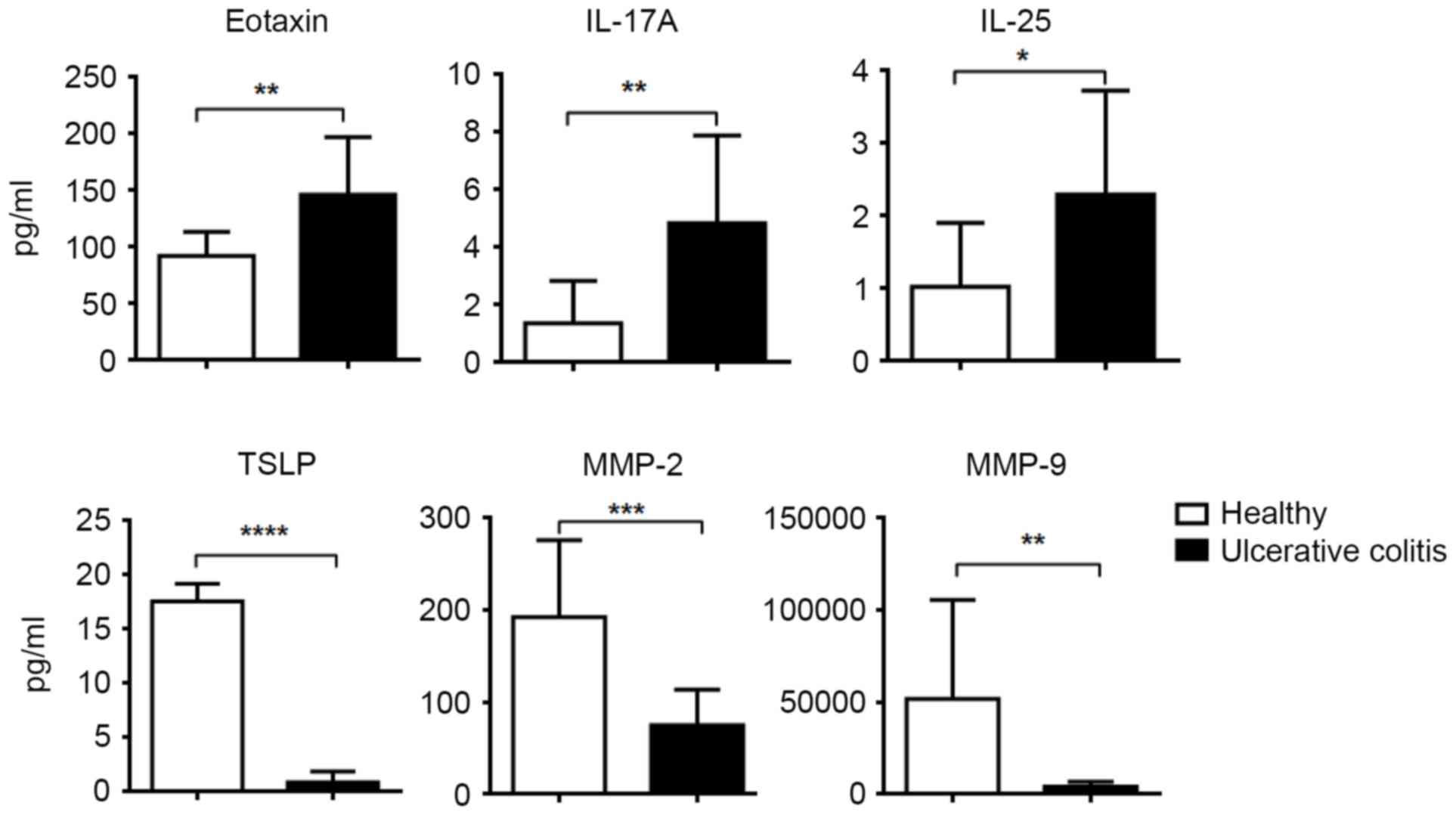
The frequency of Treg subsets in
peripheral blood of patients with ulcerative colitis
A total of 23 patients diagnosed of ulcerative
colitis were recruited and serum inflammatory cytokines were
measured. Higher levels of eotaxin, IL-17A, IL-25 but lower levels
of thymic stromal lymphopoietin, matrix metalloproteinase (MMP)-2,
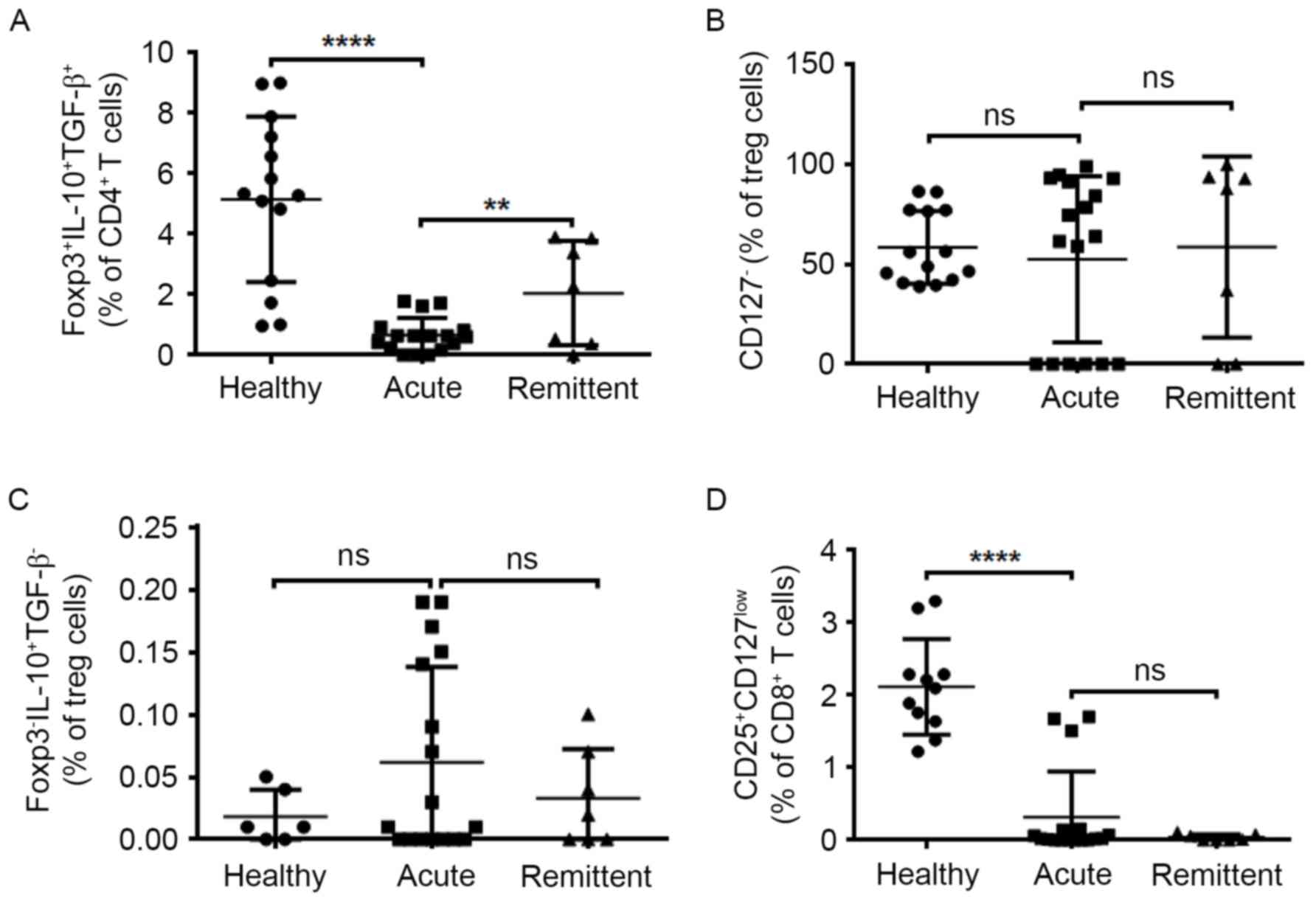
MMP-9 were identified compared with healthy controls (Fig. 5). The frequency of four Treg
subsets in peripheral blood was also measured by flow cytometry. In
good accordance, decreased percentage of nTregs were reported in
peripheral blood of patients with UC in the acute stage compared
with healthy controls and during remittent stage, a significantly
elevated percentage of nTregs was observed (Fig. 6A). The level of CD8+
Tregs decreased significantly in the acute stage and this was not
recovered in remittent stage (Fig.
6D). Both CD127− Tregs and Tr1 did not present any
increased tendency either in acute stage and remittent stage
(Fig. 6B and C). Collectively,
these data in humans and mice suggested that Foxp3+
IL-10+ TGF-β+ natural Tregs may exhibit
suppressive activity of against activated immune cells and protect
from mucosal injury during chronic stage in DSS-induced intestinal
bowel disease.
Discussion
Regulatory T cells serve an important role in
maintaining intestinal homeostasis and preventing unwanted tissue
destructions during the progression of IBD by inhibiting the
reactivity of effector immune cells (12,13).
In the present study, the authors utilized a well-established
DSS-induced murine colitis model and measured the percentage of
different Treg subsets at different time points. It was observed
that Foxp3+ IL-10+ TGF-β+ natural
Tregs may exhibit suppressive activity against activated immune
cells and protect from mucosal injury during chronic stage in
DSS-induced intestinal bowel disease.
nTreg cells, identified by the expression of
CD4+ CD25+ FOXP3+ by flow
cytometry, were the major component of Treg cells and they control
the immune cell expansion either by cell-cell contact or through
IL-10 or TGF-β secretion (14,15).
In an earlier study, by quantifying and characterizing nTreg
populations in the LPL and peripheral blood of patients with and
without IBD, Lord et al (16) observed a significantly enhanced
expression of inhibitory receptors CD39, CTLA-4 and PD-1 in
inflamed mucosa compared with controls, suggesting that nTregs are
effectively activated in IBD patients. In the current study, the
percentage of Foxp3+ IL-10+ TGF-β+
natural Tregs was measured at different time points in murine
DSS-induced colitis and different stage of UC patients. It was
suggested that, in the acute stage, the proportion of nTregs was
decreased, but this recovered and expanded in the remittent stage.
Consistent with the findings of Lord et al (16), the data also suggested that
dysfunction of nTregs may lead to the development of IBD in the
acute stage, however, enhanced nTreg proliferation during the
chronic stage may contribute to the suppression of immunopathology
in IBD.
iTreg cells that develop from conventional
CD4+ T cells in the periphery can be classified into
CD127- (FOXP3+) and FOXP3−. Both iTreg
subsets suppress inflammation through IL-10 production (17,18).
Adoptive transfer of iTreg cells can modulate innate and adaptive
immune responses, control inflammation and promote epithelial cell
homeostasis (19). Furthermore,
IL-10 produced by iTreg was sufficient to suppress immune response
when is nTreg present but IL-10 is deficient (20). The authors' examinations did not
identify any differences in the percentage of Foxp3−
IL-10+ TGF-β− and CD127− iTregs in
the acute or remittent stage of human IBD patients compared with
healthy individuals. Only a significantly decreased percentage of
Foxp3− IL-10+ TGF-β− and
CD127− iTreg was observed in LPL of DSS-induced colitis
at 4 and 7 days. Interestingly, the frequency of Foxp3−
IL-10+ TGF-β− iTreg cells became decreased
and CD127− iTreg only slightly increased in the chronic
stage. The authors suggest that the decreased percentage of
Foxp3− IL-10+ TGF-β− and CD127-
iTreg may lead to the autoimmune responses and tissue damage in the
acute phage of IBD, however, it could not be ruled out that iTreg
takes part in promoting intestinal homeostasis during the recovery
stage. Further study is required to confirm the role of iTreg cells
in IBD.
Since the pioneering work of Rifa'i et al
(21) demonstrated CD8+
CD122+ T subsets that suppress conventional T-cell
responses. In addition, CD8+ Tregs are emerging as an
important subset of T suppressors (22). CD8+ regulatory T-cell
populations are also believed to serve an important role in the
control of mucosal immunity and Ménager-Marcq et al
(23) indicated that naturally
occurring CD8+ CD28− regulatory T lymphocytes
can prevent experimental IBD in mice. Analysis the frequency of
CD8+ Treg cells demonstrated different changes in mice
and human. While CD8+ Treg cells decrease significantly
in patients with acute UC and remained in low levels at the
remittent stage, enhanced proliferation of LPL-derived
CD8+ Treg was observed at 7 days in DSS-induced murine
colitis. The authors speculated that CD8+ Treg served a
regulatory role in the late phase of murine IBD and loss of
CD8+ Treg activity may contribute to persistent chronic
inflammation in human IBD individuals.
In conclusion, the present data highlighted the
essential role of Foxp3+ IL-10+
TGF-β+ natural Tregs in controlling immune-mediated
pathology and consequent tissue damage of IBD in both human and
mice. Further studies are still needed to draw a definite
conclusion on whether natural Treg therapies were more effective in
the cure of human IBD compared to other Treg subsets.
References
|
1
|
Dixon LJ, Kabi A, Nickerson KP and
McDonald C: Combinatorial effects of diet and genetics on
inflammatory bowel disease pathogenesis. Inflamm Bowel Dis.
21:912–922. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Giusti S, Tani U and Neri E: Inflammatory
bowel diseasesCT Colonography Atlas. Springer; pp. 75–83. 2013,
View Article : Google Scholar
|
|
3
|
Baumgart DC and Sandborn WJ: Inflammatory
bowel disease: Clinical aspects and established and evolving
therapies. Lancet. 369:1641–1657. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Jostins L, Ripke S, Weersma RK, Duerr RH,
McGovern DP, Hui KY, Lee JC, Schumm LP, Sharma Y, Anderson CA, et
al: Host-microbe interactions have shaped the genetic architecture
of inflammatory bowel disease. Nature. 491:119–124. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Knights D, Lassen KG and Xavier RJ:
Advances in inflammatory bowel disease pathogenesis: Linking host
genetics and the microbiome. Gut. 62:1505–1510. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Round JL and Mazmanian SK: The gut
microbiota shapes intestinal immune responses during health and
disease. Nat Rev Immunol. 9:313–323. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Li L, Shi QG, Lin F, Liang YG, Sun LJ, Mu
JS, Wang YG, Su HB, Xu B, Ji CC, et al: Cytokine IL-6 is required
in Citrobacter rodentium infection-induced intestinal Th17
responses and promotes IL-22 expression in inflammatory bowel
disease. Mol Med Rep. 9:831–836. 2014.PubMed/NCBI
|
|
8
|
Troncone E, Marafini I, Pallone F and
Monteleone G: Th17 cytokines in inflammatory bowel diseases:
Discerning the good from the bad. Int Rev Immunol. 32:526–533.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Jiang WY, Zhang XF and Zhang HJ: Elevated
levels of Th17 cells and Th17-related cytokines in patients with
inflammatory bowel disease. J Gastroenterol Hepatol. 28:368.
2013.
|
|
10
|
Lim SM, Jeong JJ, Choi HS, Chang HB and
Kim DH: Mangiferin corrects the imbalance of Th17/Treg cells in
mice with TNBS-induced colitis. Int Immunopharmacol. 34:220–228.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Omenetti S and Pizarro TT: The Treg/Th17
Axis: A dynamic balance regulated by the gut microbiome. Front
Immunol. 6:6392015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fantini MC, Rizzo A, Fina D, Caruso R,
Becker C, Neurath MF, Macdonald TT, Pallone F and Monteleone G:
IL-21 regulates experimental colitis by modulating the balance
between Treg and Th17 cells. Eur J Immunol. 37:3155–3163. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yamada A, Arakaki R, Saito M, Tsunematsu
T, Kudo Y and Ishimaru N: Role of regulatory T cell in the
pathogenesis of inflammatory bowel disease. World J Gastroenterol.
22:2195–2205. 2016.PubMed/NCBI
|
|
14
|
Chen Y, Sun R, Wu X, Cheng M, Wei H and
Tian Z: CD4+CD25+Regulatory T cells inhibit natural killer cell
hepatocytotoxicity of hepatitis B virus transgenic mice via
membrane-bound TGF-β and OX40. J Innate Immun. 8:30–42. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Langenhorst D, Gogishvili T, Geyer B and
Hunig T: Regulation of IL-10 production in ‘natural’ regulatory T
cells. Wien Klin Wochen. 120:170. 2008.
|
|
16
|
Lord JD, Shows DM, Chen J and Thirlby RC:
Human blood and mucosal regulatory T cells express activation
markers and inhibitory receptors in inflammatory bowel disease.
PLoS One. 10:e01364852015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Verhagen J and Wraith DC: Antigen-specific
iTreg cells require both IL-10 and CTLA-4 to control autoimmune
disease. Immunology. 140:140. 2013.
|
|
18
|
Singh A, Seavey C, Horvath K and Mohiuddin
M: Induced T regulatory (iTreg) cells suppress T and B cell immune
responses. Xenotransplantation. 18:271–272. 2011.
|
|
19
|
Haribhai D, Chatila TA and Williams CB:
Immunotherapy with iTreg and nTreg cells in a murine model of
inflammatory bowel disease. Methods Mol Biol. 1422:197–211. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Schmitt EG, Haribhai D, Williams JB,
Aggarwal P, Jia S, Charbonnier LM, Yan K, Lorier R, Turner A,
Ziegelbauer J, et al: IL-10 produced by induced regulatory T cells
(iTregs) controls colitis and pathogenic Ex-iTregs during
immunotherapy. J Immunol. 189:5638–5648. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Rifa'i M, Kawamoto Y, Nakashima I and
Suzuki H: Essential roles of CD8+CD122+ regulatory T cells in the
maintenance of T cell homeostasis. J Exp Med. 200:1123–1134. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Jebbawi F, Fayyad-Kazan H, Merimi M,
Lewalle P, Verougstraete JC, Leo O, Romero P, Burny A, Badran B,
Martiat P and Rouas R: A microRNA profile of human CD8(+)
regulatory T cells and characterization of the effects of microRNAs
on Treg cell-associated genes. J Transl Med. 12:2182014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ménager-Marcq I, Pomié C, Romagnoli P and
van Meerwijk JP: CD8+CD28- regulatory T lymphocytes prevent
experimental inflammatory bowel disease in mice. Gastroenterology.
131:1775–1785. 2006. View Article : Google Scholar : PubMed/NCBI
|