Introduction
Bone loss may be caused by the imbalance of bone
remodeling. Pathological changes in bone tissue not only depend on
the number of osteoclasts and osteoblasts but also are closely
associated with the apoptosis-mediated lifecycle of these cells
(1). A number of studies have
reported that apoptosis participates in the development and
progression of periodontitis (2,3).
Periodontal pathogens mainly include Gram-negative anaerobic
bacteria. Lipopolysaccharides (LPSs) are a major component of the
cell wall of Gram-negative bacteria and are considered to be a
pivotal factor for inducing the destruction of alveolar bone tissue
and for the development and progression of periodontitis (4,5). LPS
has previously been demonstrated to directly inhibit the
differentiation of periodontal target cells and to increase the
absorption of periodontal tissues (6–8).
Therefore, LPS is of growing concern for its role in alveolar bone
resorption and periodontitis incidence. Previous studies have
revealed that LPS and the metabolites of periodontal pathogens may
affect matrix formation and cellular apoptosis. (9,10).
Regarding this imbalanced process, the biological behavior of
osteoblasts is particularly important. LPS may induce inflammatory
responses in osteoblasts and abnormal osteoblast apoptosis, which
may lead to a disorder in the number of cells coupled between bone
resorption and formation, thereby causing bone loss (10). Currently, there are few effective
treatments for bone destruction caused by bacteria, and a major
objective of bone-loss prevention is to investigate the protection
of osteoblast function and activity in the imbalanced state and to
search for potential drug targets.
Calcitonin gene-related peptide (CGRP) was first
extracted from medullary thyroid carcinoma tissue in 1971 (11). CGRP comprises 37 amino acids and
includes two isomers, α-CGRP and β-CGRP. CGRP was revealed to be
stored as secretory granules in sensory nerve endings (12), and recent studies have demonstrated
that CGRP is widely distributed in the nervous, cardiovascular,
respiratory and digestive systems (13–16).
CGRP exhibits diverse physiological effects in various tissues and
in the immune response; exogenous CGRP may increase the number and
size of osteoblast colonies (17).
In bone tissues, CGRP is not only produced in sensory nerve fibers
and endings, but also generated by osteoblasts, and it functions in
both autocrine and paracrine signaling (18). It has been recently reported that
CGRP may have a potential role in regulating inflammation, as it
lies at the intersection of the nervous and immune systems
(19,20). CGRP was reported to directly affect
CD4+ T helper cells and influence the function of
antigen-presenting cells to regulate adaptive immune responses
(21). Additionally, CGRP was
revealed to suppress the release of inflammatory cytokines, such as
interleukin (IL)-1β, tumor necrosis factor (TNF)-α and carbon
tetrachloride, from monocyte-macrophage cells and dendritic cells
upon stimulation by inactivated bacteria or Toll-like receptor
agonists (22,23). Numerous studies have provided an
understanding that CGRP may be a potential regulator of
inflammation that might inhibit the production of pro-inflammatory
cytokines, as CGRP serves a major regulatory role in the
inflammatory process (24,25). However, the effects of CGRP on
Porphyromonas gingivalis (Pg)LPS-induced osteoblast
apoptosis remained unclear. The present study demonstrated that
CGRP blocks PgLPS-induced cytostatic activity and apoptosis,
whereas TNF-α serves an important opposing role in this
process.
Materials and methods
Cell culture and reagents
Primary osteoblasts were obtained from the calvaria
of BALB/C mice according to the method described previously
(26) and cultured in Dulbecco's
modified Eagle's medium (DMEM; cat. no. 11965; Gibco; Thermo Fisher
Scientific, Inc., Waltham, MA, USA) containing 10% fetal bovine
serum (cat. no. 10100; Gibco; Thermo Fisher Scientific, Inc.), 1.5
g/l sodium bicarbonate (cat. no. 25080; Gibco; Thermo Fisher
Scientific, Inc.), 0.11 g/l sodium pyruvate (cat. no. 11360; Gibco;
Thermo Fisher Scientific, Inc.) and 100 µg/ml
penicillin/streptomycin (cat. no. 15140; Gibco; Thermo Fisher
Scientific, Inc.). Cells were maintained in a humidified atmosphere
containing 5% CO2 at 37°C. Peptide α-CGRP (cat. no.
C0167) and TNF-α (cat. no. T6674) were purchased from Sigma-Aldrich
(Merck KGaA, Darmstadt, Germany). PgLPS (Invitrogen; Thermo Fisher
Scientific, Inc.) was reconstituted in distilled and deionized
water, according to the manufacturer's protocol.
Cell viability assay
Cell viability was evaluated using a Cell Counting
kit-8 (CCK-8; cat. no. C0038; Beyotime Institute of Biotechnology,
Haimen, China), according to the manufacturer's protocol.
Osteoblasts (1.0×104 cells/well) were seeded in 96-well
plates and cultured at 4°C overnight. Cells were treated with PgLPS
at different concentrations (0, 25, 50, 100, 500 and 1,000 ng/ml)
in the aforementioned culture medium for different lengths of time
(0, 6, 12, 24, 48 or 72 h). Cells cultured in medium with 0 ng/ml
PgLPS or for 0 h incubation were used as the controls. Following
treatment, 10 µl of CCK-8 was added to each well and incubated for
2 h, and the absorbance of each well was measured with an iMark 680
Microplate Absorbance Reader (Bio-Rad Laboratories, Inc., Hercules,
CA, USA) at 450 nm. All experiments were performed independently
and repeated three times. The cell viabilities were normalized to
the control group using SPSS software for Windows (version 18.0;
SPSS, Inc., Chicago, IL, USA).
ELISA
Osteoblasts (1×104 cells/well) were
pretreated with 100 nM CGRP at 37°C for 30 min, followed by
treatment with 500 ng/ml PgLPS at 37°C for 48 h. Cells cultured in
medium with 0 ng/ml PgLPS or for 0 h incubation were used as the
controls ELISA kits for TNF-α (cat. no. MTA00B), IL-1β (cat. no.
MLB00C), IL-6 (cat. no. M6000B), monocyte chemotactic protein 1
(MCP-1; cat. no. MJE00) and MCP-2 (cat. no. DY790) were all
purchased from R&D Systems China Co., Ltd. (Shanghai, China)
and used to determine the concentrations in cell culture
supernatants according to the manufacturer's protocols.
The cells were centrifuged at 71.04 × g for 5 min at
25°C, and then 100 µl supernatant was collected and assessed for
concentration of the above proteins. The optical density of each
well was measured with a microplate reader at 450 nm and normalized
to the control group. The level of absorbance for each tested
sample was measured using the Microplate Reader 550 (Bio-Rad
Laboratories, Inc.). The data were analyzed by using SPSS software
for Windows (version 18.0; SPSS, Inc., Chicago, IL, USA).
Apoptosis assay
Osteoblasts (1×104 cells/well) were
pretreated with 100 nM CGRP at 37°C for 30 min, followed by
treatment with 500 ng/ml PgLPS at 37°C for 48 h. Cells
(1×104 cells/well) were treated with PgLPS at different
concentrations (0, 25, 50, 100, 500 and 1,000 ng/ml) in the
aforementioned culture medium for different lengths of time (0, 6,
12, 24, 48 or 72 h). Cells cultured in medium with 0 ng/ml PgLPS or
for 0 h incubation were used as the controls.
Apoptosis was assessed by flow cytometry using an
Annexin V-fluorescein isothiocyanate (FITC)/propidium iodide (PI)
staining kit (cat. no. 556547; BD Biosciences, Franklin Lakes, NJ,
USA), according to the manufacturer's protocol. Briefly, cells were
collected and washed twice with cold PBS, resuspended in staining
buffer containing Annexin V-FITC (0.025 µg/ml) and PI (1 µg/ml) and
incubated for 15 min at 25°C in the dark. Cells were washed twice
with PBS and apoptotic cells were analyzed by FACScan flow
cytometer and CELLQuest software (version 4.0.2; BD Biosciences,
Franklin Lakes, NJ, USA).
Western blot analysis
Osteoblast cultures (1×104 cells/well)
were stimulated with 500 ng/ml PgLPS at 37°C for various lengths of
time (0, 6, 12, 24, and 48 h). CGRP protein expression was
assessed. Osteoblasts (1×104 cells/well) were pretreated
with 100 nM CGRP at 37°C for 30 min, followed by treating with 500
ng/ml PgLPS at 37°C for 48 h. Cleaved (c)-Caspase-8, (c)-Caspase-3
and TNF-α protein expression was assessed.
Cells were treated and collected by centrifugation
at 71.04 × g for 1 min at 4°C. Following washing with PBS, cells
were lysed in radioimmunoprecipitation assay buffer [50 mM Tris,
(pH 7.4) 150 mM NaCl; 1% sodium deoxycholate, 1 mM sodium
orthovanadate; 1% Triton X-100; 0.1% SDS; 10 µg/ml aprotinin; 10
µg/ml leupeptin and 1 mM phenylmethylsulfonyl fluoride] on ice for
30 min. Supernatants were collected by centrifugation at 18,759 × g
for 5 min at 4°C. Protein concentrations were measured using an
Enhanced BCA Protein Assay Reagent (cat. no. P0010; Beyotime
Institute of Biotechnology). A total of 30 µg cellular protein was
boiled for 10 min and separated by 12% SDS-PAGE. Proteins were
transferred to a polyvinylidene difluoride membrane (cat. no.
162-0177; Bio-Rad Laboratories, Inc.) at 50 V for 3 h at 4°C.
Following blocking with 5% non-fat dried milk for 2 h at room
temperature, membranes were incubated with primary antibodies as
follows: CRGP (1:1,000; cat. no. 14959; CST Biological Reagents
Company Ltd., Shanghai, China), cleaved (c)-Caspase-8 (1:500; cat.
no. 9505; CST Biological Reagents Company Ltd.), c-Caspase-3
(1:500; cat. no. 9664; CST Biological Reagents Company Ltd.), TNF-α
(1:1,000; cat. no. ab6671; Abcam, Shanghai, China) and actin
(1:1,000; cat. no. sc-8432; Santa Cruz Biotechnology, Inc., Dallas,
TX, USA) overnight at 4°C. Membranes were washed three times in 1X
TBS +0.1% Tween-20 (TBST), followed by incubation with horseradish
peroxidase (HRP)-conjugated goat anti-rabbit (1:2,000; cat. no.
474-1506; Kirkegaard & Perry Laboratories, Inc.; SeraCare Life
Sciences, Milford, MA, USA) or HRP-conjugated goat anti-mouse
(1:2,000; cat. no. 474-1806; Kirkegaard & Perry Laboratories,
Inc.; SeraCare Life Sciences) secondary antibodies for 2 h at room
temperature. Following washing by 1X TBST, protein bands were
visualized using the Enhanced Chemiluminescence kit (cat. no.
170-5061; Bio-Rad Laboratories, Inc.), according to the
manufacturer's protocol. Densitometric analysis of the blots was
performed using Quantity One Software (version 4-2; Bio-Rad
Laboratories, Inc.) and normalized to actin expression levels.
Statistical analysis
All data are expressed as the mean ± standard
deviation from at least three independent experiments. Statistical
analysis was performed using one-way analysis of variance, followed
by a Newman-Keuls Student's t-test for multiple comparisons. The
analysis was conducted using SPSS software for Windows (version
18.0; SPSS, Inc.). P<0.05 was considered to indicate a
statistically significant difference.
Results
PgLPS induces the cell viability
inhibition and apoptosis
To assess the effects of PgLPS on osteoblast
viability, osteoblasts were first cultured with different
concentrations of PgLPS for 48 h and Cell viability was assessed by
CCK-8 and apoptosis was assessed by flow cytometry assay as stated
above.
Osteoblast viability was significantly reduced by
PgLPS treatment at concentrations of 50, 100, 500 and 1,000 ng/ml
(P<0.05; Fig. 1A); no
significant difference was identified between cells treated with
500 or 1,000 ng/ml PgLPS. Osteoblasts were subsequently stimulated
with 500 ng/ml PgLPS for 0-72 h, followed by measurement of
viability. The results indicated that as the stimulation time
increased, the viability of the osteoblasts significantly decreased
within 48 h (P<0.01; Fig. 1B),
no significant difference was identified between 48 and 72 h
treatment. Analysis of apoptotic rates revealed that osteoblast
apoptosis increased with increasing concentrations of PgLPS and
with increasing treatment time (Fig.
1C and D, respectively).
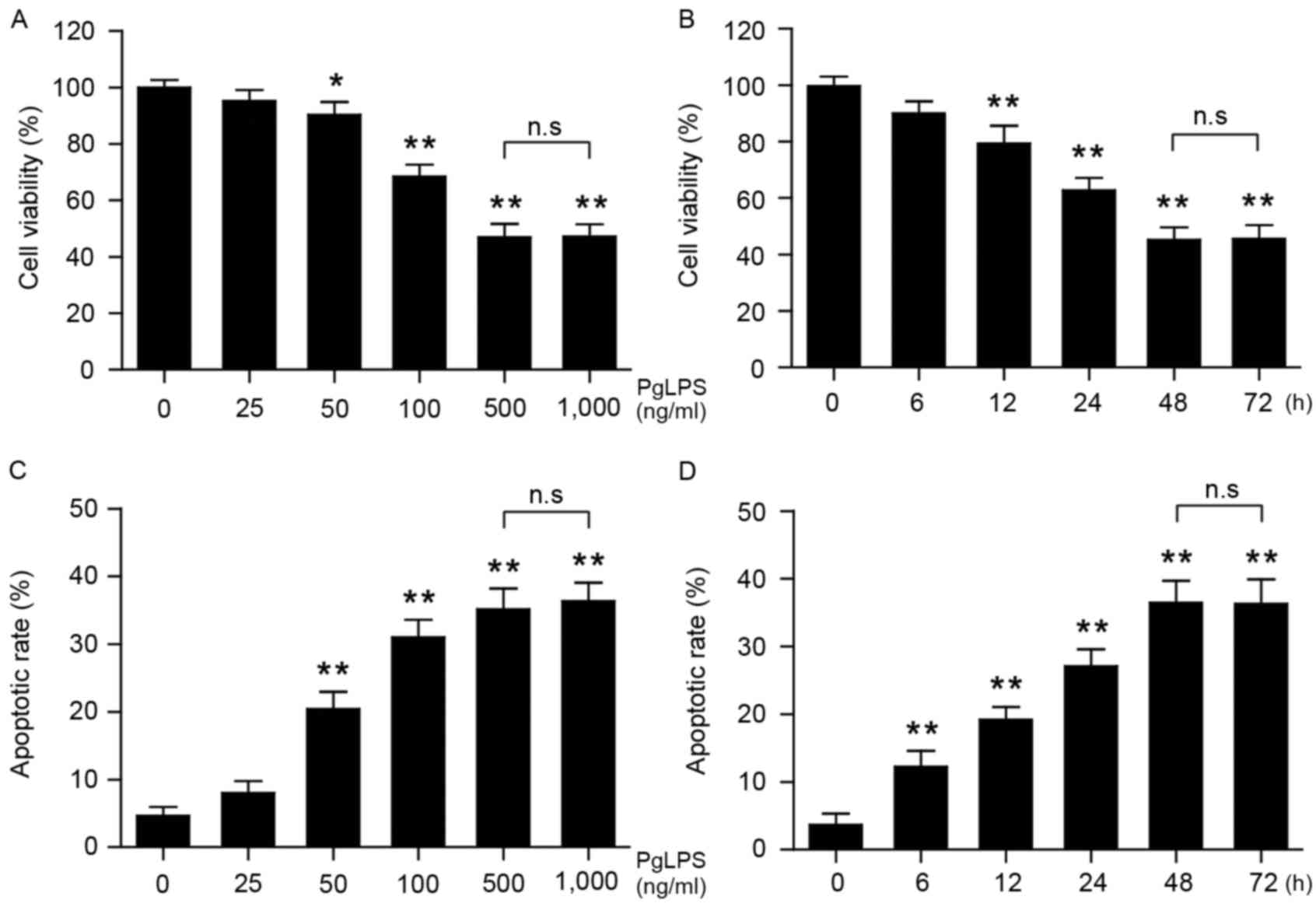 | Figure 1.Effects of PgLPS on osteoblast cell
viability and apoptosis. Osteoblasts were treated with either (A)
0, 25, 50, 100, 500 or 1,000 ng/ml PgLPS for 48 h, or (B) 500 ng/ml
PgLPS for 0, 6, 12, 24, 48 or 72 h. Cell viability was measured by
Cell Counting kit-8 assay, and the data were normalized to the
control group (0 ng/ml PgLPS or 0 h incubation, respectively). (C
and D) Osteoblasts were treated as indicated in A and B, and
apoptosis was evaluated by flow cytometry. Data are present the
mean ± standard deviation from three separate experiments;
*P<0.05 and **P<0.01 vs. the respective control (0 h or 0
ng/ml PgLPS). PgLPS, Porphyromonas gingivalis
lipopolysaccharide. |
CGRP attenuates cell viability
inhibition and apoptosis induced by PgLPS
Osteoblast cultures (1×104 cells/well)
were stimulated with 500 ng/ml PgLPS at 37°C for various lengths of
time (0, 6, 12, 24, and 48 h). Western blot analysis demonstrated
that a transient increase occurred in CGRP protein expression at 6
h following PgLPS stimulation (Fig.
2A); thereafter, CGRP protein levels gradually and
significantly decreased over time, between 12 and 48 h.
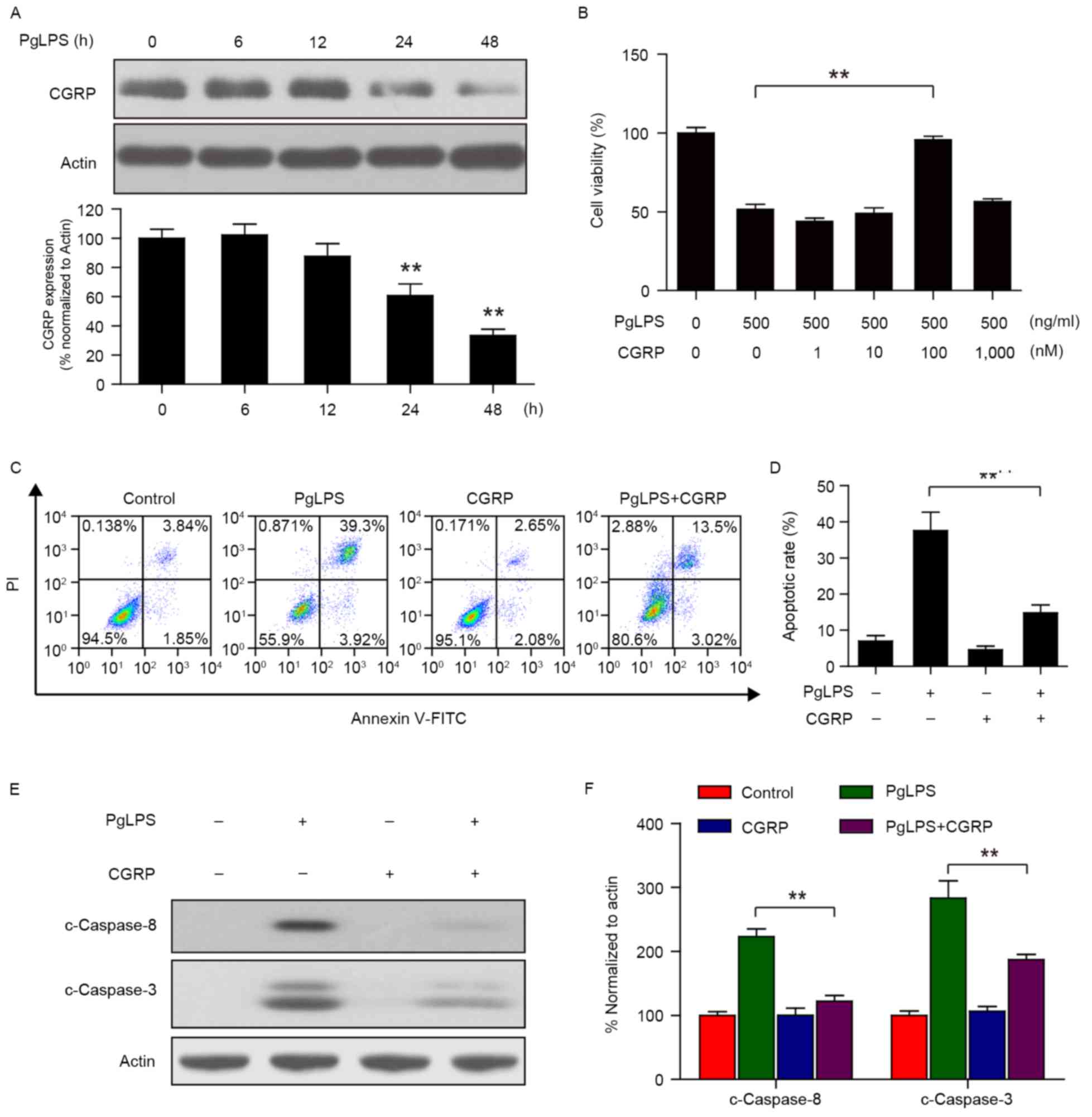 | Figure 2.Effects of CGRP pretreatment on
PgLPS-induced cell viability inhibition and apoptosis. (A)
Osteoblasts were treated with 500 ng/ml PgLPS for 0, 6, 12, 24 and
48 h, and the expression of CGRP protein levels were detected by
western blot analysis. Actin was used as a loading control; band
intensities normalized to actin are represented as the mean ±
standard deviation from three separate experiments; **P<0.01 vs.
untreated control. (B) Osteoblasts were pretreated with 0, 1, 10,
100 and 1,000 nM CGRP for 1 h, followed by treatment with or
without 500 ng/ml PgLPS for 48 h, and cell viability was measured
by Cell Counting kit-8 assay. Data are presented as the mean ±
standard deviation (n=3) and normalized to the untreated control
group; **P<0.01. (C and D) Osteoblasts were pretreated with 100
nM CGRP for 30 min, followed by treatment with 500 ng/ml PgLPS for
48 h. Apoptotic rates were measured by flow cytometry and data are
present the mean ± standard deviation from three separate
experiments; **P<0.01 vs. 0 h control. (E and F) Cells were
treated as indicated in C; whole cell lysates were prepared and
subjected to immunoblotting using antibodies against c-Caspase-8,
c-Caspase3 and Actin. Band intensities were quantified by
densitometric analysis and normalized to Actin. Data are presented
as the mean ± standard deviation; **P<0.01. C, cleaved; CGRP,
calcitonin gene-related peptide; FITC, fluorescein isothiocyanate;
PgLPS, Porphyromonas gingivalis lipopolysaccharide; PI,
propidium iodide. |
Osteoblasts (1×104 cells/well) were
pretreated with 100 nM CGRP at 37°C for 30 min, followed by
treating with 500 ng/ml PgLPS at 37°C for 48 h. Results from the
CCK-8 assay indicated that PgLPS-stimulated (500 ng/ml) osteoblasts
pretreated with CGRP (100 nM) significantly reduced the cytostatic
activity of PgLPS on osteoblasts (Fig.
2B). Apoptotic rates were measured by flow cytometry and,
consistent with the above results, 100 nM CGRP pretreatment
markedly suppressed the 500 ng/ml PgLPS-induced apoptosis in
osteoblasts (Fig. 2C and D). In
addition, CGRP (100 nM) pretreatment was demonstrated to suppress
the PgLPS-induced upregulation of c-Caspase-3 and c-Caspase 8
protein expression levels (Fig. 2E and
F).
CGRP blocks PgLPS-induced TNF-α
expression in osteoblasts
PgLPS is a classic endotoxin and has long been
considered a trigger of periodontal diseases (27). PgLPS can also induce cells to
release large amounts of inflammatory cytokines, such as TNF-α,
IL-1β and IL-6, and cause a series of inflammatory reactions
(28). Cells (1×104
cells/well) were treated with PgLPS (500 ng/ml) in the
aforementioned culture medium for different lengths of time (0, 6,
12, 24 or 48 h). Cells cultured in medium with 0 ng/ml PgLPS or for
0 h incubation were used as the controls. Results from the ELISA
assays demonstrated that PgLPS (500 ng/ml) treatment promoted the
expression of TNF-α, IL-1β, IL-6 and MCP-1 production in
osteoblasts in a time-dependent manner (Fig. 3A). Although no significant changes
were identified for MCP-2 expression between 0 and 6 h, expression
significantly increased at 12, 24 and 48 h following PgLPS
stimulation. In addition, pretreatment with CGRP did not
effectively reduce IL-1β, IL-6, MCP-1 and MCP-2 production in
osteoblasts treated with PgLPS, whereas TNF-α production was
significantly inhibited (Fig. 3B).
Osteoblasts (1×104 cells/well) were pretreated with 100
nM CGRP at 37°C for 30 min, followed by treating with 500 ng/ml
PgLPS at 37°C for 48 h. Western blot analysis also demonstrated
that the PgLPS-induced increase in TNF-α protein expression was
suppressed by CGRP pretreatment (Fig.
3C and D).
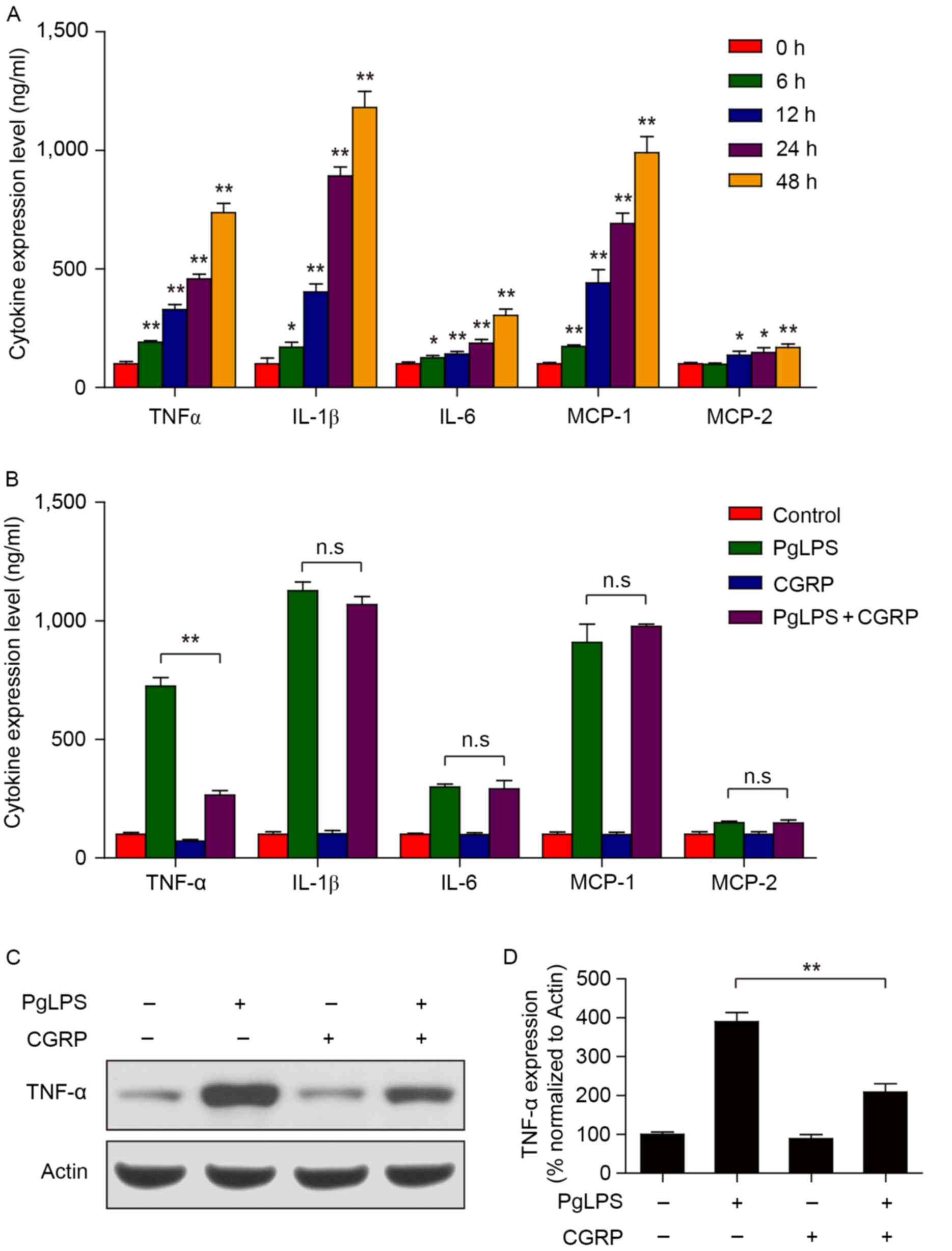 | Figure 3.Effects of CGRP pretreatment on
PgLPS-induced cytokine expression. (A) Osteoblasts were treated
with 500 ng/ml PgLPS for 0, 6, 12, 24 and 48 h. The protein
expression levels of TNF-α, IL-1β, IL-6, MCP-1 and MCP-2 in cell
culture supernatants were detected by ELISA. Data are presented as
the mean ± standard deviation; *P<0.05 and **P<0.01 vs. 0 h
control. (B) Cells were pretreated with 100 nM CGRP for 30 min,
followed by treatment with 500 ng/ml PgLPS for 48 h. The expression
of TNF-α, IL-1β, IL-6, MCP-1 and MCP-2 in cell culture supernatants
were detected by ELISA. Data are presented as the mean ± standard
deviation; **P<0.01. (C and D) Cells were treated as indicated
in B, the expression of TNF-α was detected by western blot
analysis. Band intensities were quantified by densitometric
analysis and normalized to actin. Data are presented as the mean ±
standard deviation; **P<0.01. CGRP, calcitonin gene-related
peptide; IL, interleukin; MCP, monocyte chemotactic protein; n.s.,
not significant; PgLPS, Porphyromonas gingivalis
lipopolysaccharide; TNF, tumor necrosis factor. |
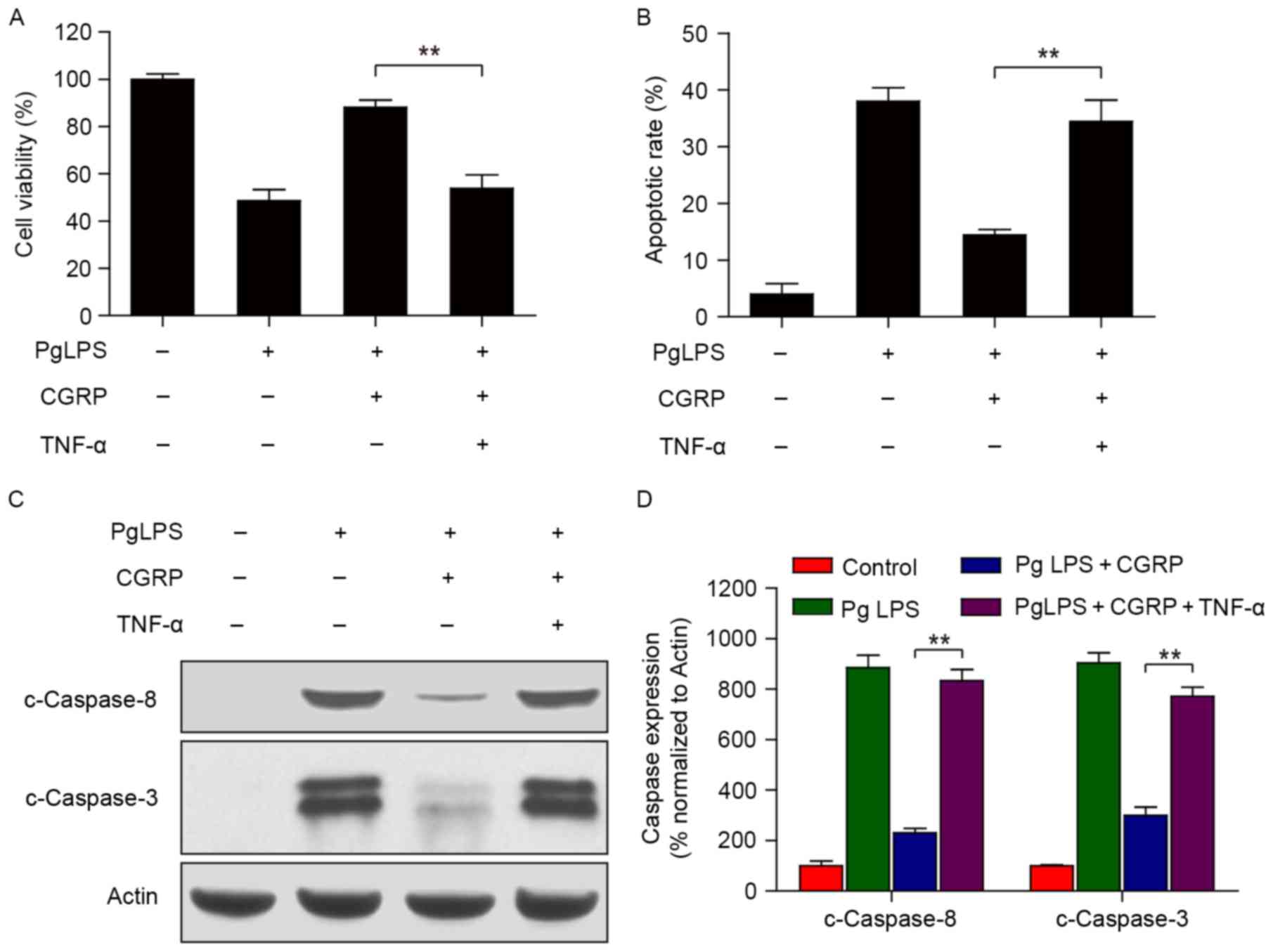
TNF-α is a key molecule in osteoblasts
viability inhibition and apoptosis induced by PgLPS and reversed by
CGRP
CCK-8 cell viability assay and flow cytometric
results revealed that CGRP pretreatment reversed the PgLPS-induced
inhibition of osteoblast cell viability and increase in apoptosis;
however, these effects were not observed in the additional presence
of exogenous TNF-α (Fig. 4A and
B). Furthermore, western blot protein expression analysis
confirmed that CGRP only inhibited the PgLPS-induced upregulation
of c-Caspase-8 and c-Caspase-3 levels; whereas, CGRP treatment was
not able to inhibit the upregulation of c-Caspase-8 and c-Caspase-3
expression in the presence of both PgLPS and TNF-α. TNF-α is an
important pro-inflammatory cytokine and a major bone resorption
factor. TNF-α mainly acts on osteoclasts and osteoblasts, and it
may cause osteoblast apoptosis (29). The current results demonstrate that
CGRP inhibited PgLPS-induced apoptosis; however, this phenomenon
was reversed by TNF-α expression. The present study hypothesized
that TNF-α was the key factor serving an opposing role in the
CGRP-induced inhibition of PgLPS-stimulated osteoblast
apoptosis.
Discussion
The present study assessed the effects of CGRP on
PgLPS-induced osteoblast apoptosis in vitro. The results
revealed that PgLPS may inhibit osteoblast viability and promote
apoptosis in a time- and concentration-dependent manner. CGRP
expression was demonstrated to reduce PgLPS-induced cytostatic
activity and apoptosis in osteoblasts, suggesting that CGRP may be
a potential agent for the prevention and treatment of
periodontitis.
A number of previous reports have focused on the
effects of CGRP on cultured osteoblasts and have revealed that CGRP
can regulate bone metabolism and stimulate osteoblasts
differentiation (17,30,31).
Nevertheless, only a few reports have reported on LPS-induced
osteoblast apoptosis. A study published in 1997 (32) demonstrated that CGRP can inhibit
LPS-induced TNF-α production in osteoblasts, which is in line with
the results of the present study. However, the present study also
further assessed the expression of several cytokines induced by LPS
and evaluated the effect of CGRP on them. The present study
successfully established a LPS-treated osteoblast cell model and
elucidated more mechanisms associated with apoptosis.
Inflammation is characterized by an increase in the
expression of inflammatory cytokines produced by cells of the
activated innate and adaptive immune systems. It has been
previously reported that the inflammatory response is closely
related to the extent of systemic and local bone loss (33). LPS exposure was reported to
stimulate osteoclastic bone resorption in vivo and inhibit
osteoblast differentiation (34).
An increasing amount of data has also demonstrated that PgLPS may
directly induce cell death or apoptosis in osteoblasts (10,34).
Additional studies have demonstrated that apoptosis serves an
essential role in the development and progression of infectious
diseases, autoimmune diseases and tumors (35). Osteoblasts cells are capable of
secreting matrix and mineralizing into bone tissue; they are the
major cells involved in bone remodeling (36). Osteoblast proliferation and
apoptosis are of great importance to maintaining the balance of
bone formation (37). In the
course of periodontitis, bacterial-induced osteoblast apoptosis may
be a major contributor to bone loss. A number of studies have
revealed that LPS is present in plaque, saliva, gingival crevicular
fluid, inflammatory cavities and diseased cementum, with high
toxicity to periodontal tissues (38–40).
In addition, a previous study suggested that during alveolar bone
resorption and periodontitis, bacteria do not directly invade
gingival tissue; instead, the destruction of gingival tissue may be
mediated by LPS cytotoxicity in the gingival crevice (37). A number of inflammatory cytokines
are synthesized and secreted by periodontal tissues and cells in
response to LPS exposure, and research has shown that LPS may
inhibit osteoblast proliferation and differentiation, and suppress
bone formation (34). A previous
study reported that PgLPS was able to significantly inhibit
alkaline phosphatase (ALP) activity in osteoblasts and decrease the
formation of mineralization nodules in a dose- and time-dependent
manner (7). Additionally, PgLPS
was revealed to increase the protein expression levels of CD14 and
LPS receptors, and the mRNA expression level of the bone resorption
factor IL-1β. Similarly, LPS exposure was demonstrated to
significantly inhibit ALP activity and collagen synthesis in the
osteoblast cell line MC3T3-E1 (34). LPS expression has also been
reported to facilitate osteoclast differentiation and activation by
activating the mitogen-activated protein kinase signaling pathway
(41), as well as induce
osteoblast apoptosis by activating the c-Jun N-terminal kinase
pathway (42). The present study
demonstrated that PgLPS stimulation inhibited the viability and
induced apoptosis in osteoblasts, which was in agreement with
previous reports. In addition, CGRP pretreatment was revealed to
reduce apoptotic rates that were induced by PgLPS exposure.
Following PgLPS stimulation, osteoblasts released a large amount of
inflammatory cytokines, including TNF-α, IL-1β, IL-6, MCP-1 and
MCP-2; PgLPS stimulation also increased the expression of apoptotic
signals c-Caspase-3 and c-Caspase-8. A number of previous studies
reported that the change in cytokine expression levels, including
IL-1β, IL-6, TNF-α, interferon-γ and leukemia inhibitory factor,
was an important cause of bone loss (43,44).
Among these, the increase of TNF-α expression was identified as one
of the characteristics of bone loss (45). It is worth noting that, results
from the present study demonstrated that TNF-α was markedly
suppressed by CGRP pretreatment, whereas CGRP inhibited the
PgLPS-activated caspase signal, which ultimately led to apoptosis.
These results indicated that CGRP was able to block PgLPS-induced
initiation of inflammation and thus may have a protective effect on
osteoblasts.
In the serum of patients suffering from
postmenopausal osteoporosis, the level of TNF-α expression was
reportedly increased (46).
Similarly, TNF-α expression was also revealed to be increased in
the serum of patients with type 2 diabetes, which indicated that
the change in TNF-α level may be an important cause for bone
metabolism diseases (47). TNF-α
has many physiological functions in the regulation of cell
proliferation, differentiation and survival. In models of
pathological bone loss, such as senile osteoporosis and bone
resorption caused by chronic inflammation, the concentrations of
TNF-α are increased, indicating that TNF-α may also be an important
factor in regulating bone metabolism (48). Indeed, TNF-α alone was reported to
cause osteoblast apoptosis and, in addition, TNF-α was revealed to
inhibit collagen synthesis, ALP activity and osteocalcin synthesis
(49). TNF-α may directly promote
osteoclast maturation and differentiation by inducing the
expression of macrophage colony-stimulating factor and receptor
activator of nuclear factor κB ligand (RANKL) in osteoblasts
(50). Additionally, TNF-α may
directly promote RANKL-exposed precursor cells to differentiate
into osteoclasts, activate mature osteoclasts and inhibit
osteoclast apoptosis (51).
According to the present results, CGRP pretreatment inhibited
PgLPS-induced osteoblast apoptosis; however, this phenomenon was
reversed by the addition of TNF-α. Although an accurate explanation
of this phenomenon is difficult, it may be speculated that TNF-α
serves a pivotal role in CGRP inhibition of PgLPS-induced apoptosis
in osteoblasts; however, the exact mechanism involved requires
further investigation.
Current treatments for periodontitis mainly consist
of antibiotics; despite the therapeutic effects, long-term use of
antibiotics may lead to oral dysbiosis, increased bacterial
resistance to these antibiotics and gastrointestinal irritation,
among other side effects. These factors have affected the clinical
application of antibiotics for periodontitis (52). The present in vitro study
demonstrated that the vasoactive peptide CGRP reduced the
expression of inflammatory cytokines and osteoblast apoptosis. In
particular, its significant inhibition of TNF-α suggested a new
potential target of action for this peptide in the treatment of
periodontitis. In addition, it may be also interesting to integrate
the present results with the effects of the neuropeptidergic
innervation in dental pulp as, in pulpitis, CGRP-positive fibers in
the pulp increased, which also suggested that CGRP may serve a
potential role in the regulation of this process. The present study
on CGRP may also lead to a new direction for the treatment of
pulpitis, which will be the focus of future research.
References
|
1
|
Boyce BF, Rosenberg E, de Papp AE and
Duong LT: The osteoclast, bone remodelling and treatment of
metabolic bone disease. Eur J Clin Invest. 42:1332–1341. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Di Benedetto A, Gigante I, Colucci S and
Grano M: Periodontal disease: Linking the primary inflammation to
bone loss. Clin Dev Immunol. 2013:5037542013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Song B, Zhou T, Yang WL, Liu J and Shao
LQ: Programmed cell death in periodontitis: Recent advances and
future perspectives. Oral Dis. 23:609–619. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Nakao J, Fujii Y, Kusuyama J, Bandow K,
Kakimoto K, Ohnishi T and Matsuguchi T: Low-intensity pulsed
ultrasound (LIPUS) inhibits LPS-induced inflammatory responses of
osteoblasts through TLR4-MyD88 dissociation. Bone. 58:17–25. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ossola CA, Surkin PN, Pugnaloni A, Mohn
CE, Elverdin JC and Fernandez-Solari J: Long-term treatment with
methanandamide attenuates LPS-induced periodontitis in rats.
Inflamm Res. 61:941–948. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bandow K, Maeda A, Kakimoto K, Kusuyama J,
Shamoto M, Ohnishi T and Matsuguchi T: Molecular mechanisms of the
inhibitory effect of lipopolysaccharide (LPS) on osteoblast
differentiation. Biochem Biophys Res Commun. 402:755–761. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kadono H, Kido J, Kataoka M, Yamauchi N
and Nagata T: Inhibition of osteoblastic cell differentiation by
lipopolysaccharide extract from Porphyromonas gingivalis. Infect
Immun. 67:2841–2846. 1999.PubMed/NCBI
|
|
8
|
Kato H, Taguchi Y, Tominaga K, Umeda M and
Tanaka A: Porphyromonas gingivalis LPS inhibits osteoblastic
differentiation and promotes pro-inflammatory cytokine production
in human periodontal ligament stem cells. Arch Oral Biol.
59:167–175. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Li Y, Shibata Y, Zhang L, Kuboyama N and
Abiko Y: Periodontal pathogen aggregatibacter actinomycetemcomitans
LPS induces mitochondria-dependent-apoptosis in human placental
trophoblasts. Placenta. 32:11–19. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Thammasitboon K, Goldring SR and Boch JA:
Role of macrophages in LPS-induced osteoblast and PDL cell
apoptosis. Bone. 38:845–852. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Melvin KE, Miller HH and Tashjian AH Jr:
Early diagnosis of medullary carcinoma of the thyroid gland by
means of calcitonin assay. N Engl J Med. 285:1115–1120. 1971.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Broad PM, Symes AJ, Thakker RV and Craig
RK: Structure and methylation of the human calcitonin/alpha-CGRP
gene. Nucleic Acids Res. 17:6999–7011. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Baillie LD, Schmidhammer H and Mulligan
SJ: Peripheral mu-opioid receptor mediated inhibition of calcium
signaling and action potential-evoked calcium fluorescent
transients in primary afferent CGRP nociceptive terminals.
Neuropharmacology. 93:267–273. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Pereira RT, Costa LS, Oliveira IR, Araújo
JC, Aerts M, Vigliano FA and Rosa PV: Relative distribution of
gastrin-, CCK-8-, NPY- and CGRP-immunoreactive cells in the
digestive tract of dorado (Salminus brasiliensis). Tissue Cell.
47:123–131. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Uddman R, Luts A and Sundler F: Occurrence
and distribution of calcitonin gene-related peptide in the
mammalian respiratory tract and middle ear. Cell Tissue Res.
241:551–555. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sun T, Guo Z, Liu CJ, Li MR, Li TP, Wang X
and Yuan DJ: Preservation of CGRP in myocardium attenuates
development of cardiac dysfunction in diabetic rats. Int J Cardiol.
220:226–234. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
He H, Chai J, Zhang S, Ding L, Yan P, Du W
and Yang Z: CGRP may regulate bone metabolism through stimulating
osteoblast differentiation and inhibiting osteoclast formation. Mol
Med Rep. 13:3977–3984. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Tian G, Zhang G and Tan YH: Calcitonin
gene-related peptide stimulates BMP-2 expression and the
differentiation of human osteoblast-like cells in vitro. Acta
Pharmacol Sin. 34:1467–1474. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Holzmann B: Modulation of immune responses
by the neuropeptide CGRP. Amino Acids. 45:1–7. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Holzmann B: Antiinflammatory activities of
CGRP modulating innate immune responses in health and disease. Curr
Protein Pept Sci. 14:268–274. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Streilein JW: Ocular immune privilege:
Therapeutic opportunities from an experiment of nature. Nat Rev
Immunol. 3:879–889. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
22
|
Carucci JA, Ignatius R, Wei Y, Cypess AM,
Schaer DA, Pope M, Steinman RM and Mojsov S: Calcitonin
gene-related peptide decreases expression of HLA-DR and CD86 by
human dendritic cells and dampens dendritic cell-driven T
cell-proliferative responses via the type I calcitonin gene-related
peptide receptor. J Immunol. 164:3494–3499. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Harzenetter MD, Novotny AR, Gais P, Molina
CA, Altmayr F and Holzmann B: Negative regulation of TLR responses
by the neuropeptide CGRP is mediated by the transcriptional
repressor ICER. J Immunol. 179:607–615. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Nagata S: Apoptosis by death factor. Cell.
88:355–365. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kroeger I, Erhardt A, Abt D, Fischer M,
Biburger M, Rau T, Neuhuber WL and Tiegs G: The neuropeptide
calcitonin gene-related peptide (CGRP) prevents inflammatory liver
injury in mice. J Hepatol. 51:342–353. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Orriss IR, Hajjawi MO, Huesa C, MacRae VE
and Arnett TR: Optimisation of the differing conditions required
for bone formation in vitro by primary osteoblasts from mice and
rats. Int J Mol Med. 34:1201–1208. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
DeLeon-Pennell KY, de Castro Brás LE, Iyer
RP, Bratton DR, Jin YF, Ripplinger CM and Lindsey ML: P. gingivalis
lipopolysaccharide intensifies inflammation post-myocardial
infarction through matrix metalloproteinase-9. J Mol Cell Cardiol.
76:218–226. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ding PH, Darveau RP, Wang CY and Jin L:
3LPS-binding protein and its interactions with P. gingivalis LPS
modulate pro-inflammatory response and toll-like receptor signaling
in human oral keratinocytes. PLoS One. 12:e01732232017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wu X, Feng X, He Y, et al: IL-4
administration exerts preventive effects via suppression of
underlying inflammation and TNF-α-induced apoptosis in
steroid-induced osteonecrosis. Osteoporos Int. 27:1827–1837. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Liang W, Zhuo X, Tang Z, Wei X and Li B:
Calcitonin gene-related peptide stimulates proliferation and
osteogenic differentiation of osteoporotic rat-derived bone
mesenchymal stem cells. Mol Cell Biochem. 402:101–110. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ma W, Zhang X, Shi S and Zhang Y:
Neuropeptides stimulate human osteoblast activity and promote gap
junctional intercellular communication. Neuropeptides. 47:179–186.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Millet I and Vignery A: The neuropeptide
calcitonin gene-related peptide inhibits TNF-alpha but poorly
induces IL-6 production by fetal rat osteoblasts. Cytokine.
9:999–1007. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Redlich K and Smolen JS: Inflammatory bone
loss: Pathogenesis and therapeutic intervention. Nat Rev Drug
Discov. 11:234–250. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
34
|
Guo C, Yuan L, Wang JG, Wang F, Yang XK,
Zhang FH, Song JL, Ma XY, Cheng Q and Song GH: Lipopolysaccharide
(LPS) induces the apoptosis and inhibits osteoblast differentiation
through JNK pathway in MC3T3-E1 cells. Inflammation. 37:621–631.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Favaloro B, Allocati N, Graziano V, Di
Ilio C and De Laurenzi V: Role of apoptosis in disease. Aging
(Albany NY). 4:330–349. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Meshcheryakova A, Mechtcheriakova D and
Pietschmann P: Sphingosine 1-phosphate signaling in bone
remodeling: Multifaceted roles and therapeutic potential. Expert
Opin Ther Targets. 21:725–737. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kesavalu L, Sathishkumar S, Bakthavatchalu
V, Matthews C, Dawson D, Steffen M and Ebersole JL: Rat model of
polymicrobial infection, immunity and alveolar bone resorption in
periodontal disease. Infect Immun. 75:1704–1712. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Bai Y, Wei Y, Wu L, Wei J, Wang X and Bai
Y: C/EBP β mediates endoplasmic reticulum stress regulated
inflammatory response and extracellular matrix degradation in
LPS-stimulated human periodontal ligament cells. Int J Mol Sci.
17:3852016. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Jaedicke KM, Preshaw PM and Taylor JJ:
Salivary cytokines as biomarkers of periodontal diseases.
Periodontol 2000. 70:164–183. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Toyman U, Tuter G, Kurtis B, Kıvrak E,
Bozkurt Ş, Yücel AA and Serdar M: Evaluation of gingival crevicular
fluid levels of tissue plasminogen activator, plasminogen activator
inhibitor 2, matrix metalloproteinase-3 and interleukin 1-β in
patients with different periodontal diseases. J Periodontal Res.
50:44–51. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Hou GQ, Guo C, Song GH, Fang N, Fan WJ,
Chen XD, Yuan L and Wang ZQ: Lipopolysaccharide (LPS) promotes
osteoclast differentiation and activation by enhancing the MAPK
pathway and COX-2 expression in RAW264.7 cells. Int J Mol Med.
32:503–510. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Guo C, Wang SL, Xu ST, Wang JG and Song
GH: SP600125 reduces lipopolysaccharide-induced apoptosis and
restores the early-stage differentiation of osteoblasts inhibited
by LPS through the MAPK pathway in MC3T3-E1 cells. Int J Mol Med.
35:1427–1434. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Kırzıoğlu FY, Tözüm Bulut M, Doğan B,
Fentoğlu Ö, Özmen Ö, Çarsancaklı SA, Ergün AG, Özdem M and Orhan H:
Anti-inflammatory effect of rosuvastatin decreases alveolar bone
loss in experimental periodontitis. J Oral Sci. 59:247–255. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Yao Z, Lei W, Duan R, Li Y, Luo L and
Boyce BF: RANKL cytokine enhances TNF-induced osteoclastogenesis
independently of TNF receptor associated factor (TRAF) 6 by
degrading TRAF3 in osteoclast precursors. J Biol Chem.
292:10169–10179. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Tzach-Nahman R, Mizraji G, Shapira L,
Nussbaum G and Wilensky A: Oral infection with porphyromonas
gingivalis induces peri-implantitis in a murine model: Evaluation
of bone loss and the local inflammatory response. J Clin
Periodontol. 44:739–748. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Al-Daghri NM, Aziz I, Yakout S, Aljohani
NJ, Al-Saleh Y, Amer OE, Sheshah E, Younis GZ and Al-Badr FB:
Inflammation as a contributing factor among postmenopausal Saudi
women with osteoporosis. Medicine (Baltimore). 96:e57802017.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Chen YL, Qiao YC, Xu Y, Ling W, Pan YH,
Huang YC, Geng LJ, Zhao HL and Zhang XX: Serum TNF-α concentrations
in type 2 diabetes mellitus patients and diabetic nephropathy
patients: A systematic review and meta-analysis. Immunol Lett.
186:52–58. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Wang C, Yu X, Yan Y, Yang W, Zhang S,
Xiang Y, Zhang J and Wang W: Tumor necrosis factor-alpha: a key
contributor to intervertebral disc degeneration. Acta Biochim
Biophys Sin (Shanghai). 49:1–13. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Centrella M, McCarthy TL and Canalis E:
Tumor necrosis factor-alpha inhibits collagen synthesis and
alkaline phosphatase activity independently of its effect on
deoxyribonucleic acid synthesis in osteoblast-enriched bone cell
cultures. Endocrinology. 123:1442–1448. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Lam J, Takeshita S, Barker JE, Kanagawa O,
Ross FP and Teitelbaum SL: TNF-alpha induces osteoclastogenesis by
direct stimulation of macrophages exposed to permissive levels of
RANK ligand. J Clin Invest. 106:1481–1488. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Park J, Choi HM, Yang HI, Yoo MC and Kim
KS: Increased expression of IL-1 receptors in response to IL-1β may
produce more IL-6, IL-8, VEGF and PGE2 in senescent synovial cells
induced in vitro than in presenescent cells. Rheumatol Int.
32:2005–2010. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Santos RS, Macedo RF, Souza EA, Soares RS,
Feitosa DS and Sarmento CF: The use of systemic antibiotics in the
treatment of refractory periodontitis: A systematic review. J Am
Dent Assoc. 147:577–585. 2016. View Article : Google Scholar : PubMed/NCBI
|