Introduction
Nucleotide-binding domain leucine-rich
repeat-containing receptors (NLRs) are the main constituents of
inflammasomes and serve a role in innate immunity and inflammation
(1–3). NLR family pyrin domain containing
proteins (NLRPs) are the largest subfamily of NLRs (4,5).
Activation of NLRs results in recruitment of caspase-1 to
inflammasomes via an adaptor protein that contains a pyrin domain
(PYD) and caspase recruitment domain [CARD; PYD and CARD domain
containing (PYCARD) protein] and caspase-1 subsequently cleaves
pro-interleukin (IL)-1β and pro-IL-18 to produce their active forms
(1–3).
Previous studies revealed that deregulated
inflammasomes serve an important role in the pathogenesis of
autoinflammatory diseases (AIDs) (6–8).
AIDs are characterized by episodes of abnormally increased
inflammation without production of high-titer autoantibodies or
antigen-specific T lymphocytes, but with symptoms including spiking
fever, erythema, arthropathy, serositis and vasculopathy (9–11).
Adult-onset Still's disease (AOSD) is an inflammatory disorder
characterized by spiking fever, rash, arthritis,
hepatosplenomegaly, variable multisystemic involvement and
increases in levels of acute phase reactants (12,13).
AOSD has been classified as an AID as it demonstrates similar
phenotypes without significantly elevated levels of autoantibodies
(11,14). Previous studies have revealed
increased levels of serum IL-1β and IL-18 in patients with AOSD
(15–18). We further hypothesized that
inflammasome-dependent processing of cytokines may have a role in
in AOSD pathogenesis. However, to the best of our knowledge there
is no data available on the pathogenic role of inflammasomes in
AOSD.
Human parvovirus B19 (B19) is a DNA virus
responsible for a wide range of human diseases (19). It consists of two structural
proteins, VP1 and VP2, which are identical except for a 227-amino
acids long amino-terminal end of VP1, known as the VP1-unique
region (VP1u) (20). B19-VP1u is
essential for cytotoxicity and infectivity, while B19
non-structural protein-1 (B19-NS1) exhibits apoptotic activity
(19,20). B19 has been hypothesized to be
associated with rheumatic diseases including AOSD (21,22).
The present study aimed to investigate whether B19 serves a role as
an activator of NLRP3-inflammasome in AOSD.
The present study investigated whether mRNA levels
of NLRP-inflammasomes differ significantly between patients with
AOSD and healthy controls, whether inflammasome inhibitors
influence the expression of NLRP3-inflammasome signaling components
and whether B19-associated antigens serve a role as NLRPs
activators and affect the expressions of inflammasome components
and downstream cytokines from peripheral blood mononuclear cells
(PBMCs) of patients with AOSD.
Materials and methods
Patients and ethical approval
A total of 34 patients with active untreated AOSD
(mean age ± standard deviation, 32.7±11.1 years; 26 women and 8
men) fulfilling the Yamaguchi criteria (14) were enrolled in Taichung Veterans
General Hospital, Taiwan, between April 2013 and March 2015.
Patients with infections, malignancies or other rheumatic diseases
were excluded. Data were obtained on the presenting clinical
features using the following definitions: i) Spiking fever was
defined as an elevation of tympanic temperature above 39°C with
daily reduction in temperature into normal range or below 39°C; ii)
typical rash was presented as salmon-red maculopapular evanescent
skin lesion; iii) arthritis was defined as the presence of
swelling, local warmth and tenderness over the affected joints; iv)
sore throat constituted subjective symptoms without redness or
exudates in pharyngeal examination and v) the presence of
lymphadenopathy was defined as palpable tender lymph nodes which
most commonly occurred in the cervical region. Following blood
sampling for the determination of NLRPs expression, all patients
with AOSD received corticosteroids, non-steroidal anti-inflammatory
drugs and disease-modifying anti-rheumatic drugs. A total of 14
healthy volunteers (33.4±5.7 years of age; 12 women and 2 men),
with no rheumatic diseases, were enrolled as controls. The present
study was approved by the Institutional Review Board of Taichung
Veterans General Hospital (Taichung, Taiwan; approval no. CF11309)
and all participants submitted written informed consent according
to guidelines of the ethical principles for medical research
involving human subjects of the Declaration of Helsinki.
Determination of mRNA levels of NLRPs
in PBMCs using reverse transcription-quantitative polymerase chain
reaction (RT-qPCR)
PBMCs were isolated from venous blood using
Ficoll-Paque PLUS (GE Healthcare, Chicago, IL, USA) density
gradient centrifugation. Total RNA was obtained from PBMCs using
the guanidinium isothiocyanate method, as previously described
(23). A total of 2.5 µg RNA was
reverse transcribed using 200 U Moloney murine leukemia virus
reverse transcriptase (cat. no. 28025013, Fermentas; Thermo Fisher
Scientific, Inc., Waltham, MA, USA), which was incubated 1 h at
37°C with 0.025 mM oligo(dT) primer and Reverse Transcriptase
Buffer (18057018; Thermo Fisher Scientific, Inc.). mRNA levels of
NLRP1-NLRP14 were determined by qPCR using a TaqMan Universal
Master Mix (cat. no. 4440040, Applied Biosystems; Thermo Fisher
Scientific, Inc.). The catalog numbers of primers and probes for
each gene were as follows: NLRP1 (Hs00248187_m1), NLRP2
(Hs01546932_m1), NLRP3 (Hs00918082_m1), NLRP4 (Hs00370499_m1),
NLRP5 (Hs00411266_m1), NLRP6 (Hs00373246_m1), NLRP7
(Hs00373683_m1), NLRP8 (Hs00603419_m1), NLRP9 (Hs00603423_m1),
NLRP10 (Hs00738590_m1), NLRP11 (Hs00935472_m1), NLRP12
(Hs00536435_m1), NLRP13 (Hs00603406_m1), NLRP14 (Hs00698226_m1),
ASC (PYCARD) (Hs00741684_g1), caspase-1 (Hs00354832_m1), IL-1β
(Hs01555410_m1), and IL-18 (Hs01038788_m1) (Applied Biosystems;
Thermo Fisher Scientific, Inc.). Amplification cycles were
conducted for 95°C for 10 min, followed by 40 cycles of
denaturation at 95°C for 15 sec, then annealing and extension at
60°C for 1 min. To standardize mRNA levels of inflammasome
components and downstream cytokines, mRNA levels of the
housekeeping gene GAPDH (Hs99999905_m1) were also determined in
parallel for each sample. Relative mRNA levels of NLRPs were
calculated using the comparative threshold cycle (Cq) method,
2−ΔΔCq (24), using the
following equation: DDCq=patient (Cq NLRPs
gene-Cq GAPDH)-mean of controls (Cq
NLRPs gene-Cq GAPDH).
Ex vivo cytokine production in PBMCs
treated with inflammasome inhibitors
To verify the association between NLRP3-inflammasome
and downstream inflammatory responses, alterations were determined
in mRNA expression and supernatant levels of downstream cytokines
in PBMCs treated with inflammasome inhibitors. PBMC samples were
incubated at 37°C in a 5% CO2 humidified atmosphere for
24 h in dimethyl sulfoxide (D2650, Sigma-Aldrich; Merck KGaA,
Darmstadt, Germany) as a solvent or in the presence of inflammasome
inhibitors. The inhibitors used in the present study included the
NLRP3 inhibitor glibenclamide (20 µM; IMI-2319, Novus Biologicals
LLC, Littleton, CO, USA) and caspase-1 inhibitor VX-765 (60 µM;
orb61303, Biorbyt Ltd. Cambridge, UK) at 37°C in a 5%
CO2 humidified atmosphere for 24 h. The mRNA expression
levels of NLRP3 were determined as described in the previous
section. Cell-free supernatant was harvested by centrifugation at
4°C for 15 min at 500 × g and stored at −70°C until used for the
determination of cytokine levels. Levels of IL-1β and IL-18 were
determined using ELISA kits (cat. no. ELH-IL1b-1, RayBiotech Inc.,
Norcross, GA, USA; and cat. no. 7620, MBL International Co.,
Woburn, MA, USA, respectively) according to the manufacturers'
instructions.
Ex vivo assay to investigate the
effect of B19-associated antigens on mRNA expression levels of
NLRP3 and downstream cytokines
PBMCs from 14 patients with AOSD and 4 healthy
volunteers were re-suspended in RPMI 1640 medium (Thermo Fisher
Scientific, Inc.) supplemented with 100 units/ml penicillin, 100
µg/ml streptomycin and 10% fetal bovine serum (Thermo Fisher
Scientific Inc.) at a final concentration 5×106
cells/well. Expression levels of NLRP3 were examined in PBMCs
treated with B19-associated antigens, including 1.5 µg/ml B19-NS1
and 1.5 µg/ml B19-VP1 u for 24 h, purified as previously described
(25). Fold change values
represent the ratio of mRNA expression levels in cells treated with
B19 antigens, compared with mRNA levels in cells treated with
medium only. Data are presented as the mean ± standard deviation.
mRNA levels of NLRP3 and supernatant levels of IL-1β and IL-18 were
determined as described above.
Ex vivo assay to investigate the
effect of B19-associated antigens on protein expression levels of
NLRP3 and downstream cytokines
The PBMCs were treated with Protein Extraction
Solution (cat. no. 17081, iNtRON Biotechnology, Seongnam, South
Korea) for 20 min on ice and centrifuged at 10,000 × g for 30 min
at 4°C, proteins were quantified with a Bicinchoninic Acid Protein
Assay (cat. no. 786571, Geno Technology Inc., Louis, MO, USA).
Total protein extraction (10 µg) was performed from PBMCs treated
with 1.5 µg/ml B19-NS1 and 1.5 µg/ml B19-VP1u. Proteins were
separated by 10% SDS-PAGE in running buffer (25 mM Tris, 192 mM
glycine, 0.1% SDS). The gel was run at 70 V 30 min at room
temperature, then at 100 V until the blue-dye front approached the
end of the gel. The gel was transferred to polyvinylidene fluoride
membranes (1620177; Bio-Rad Laboratories, Inc., Hercules, CA, USA),
which were blocked with transfer buffer (50 mM Tris, 384 mM
glycine, 20% methanol) at 4°C for 300 mA 1 h with the Mini TBC
Electrophoretic Transfer Cell (170–3930, Bio-Rad Laboratories,
Inc.). The membranes were blocked with 5% BSA (A7906;
Sigma-Aldrich; Merck KGaA) in TBST [150 mM NaCl, 20 mM Tris-HCl (pH
7.4), 0.1% Tween-20] at room temperature for 1 h. Western blot
analyses were performed using primary antibodies against NLRP3
(1:1,000; ab16097, Abcam, Cambridge, MA, USA), caspase-1 (1:1,000;
NB100-56565SS, Novus Biologicals, LLC), IL-1β (1:1,000;
NBS2-27342SS, NB100-56565SS, Novus Biologicals, LLC), IL-18
(1:1,000; PM014, MBL International Co.), and GAPDH (1:5,000;
sc-32233, Santa Cruz Biotechnology, Inc., Dallas, TX, USA) at 4°C
overnight. Horseradish peroxidase-conjugated anti-mouse (1:10,000;
sc-2005, Santa Cruz Biotechnology, Inc.) or rabbit immunoglobulin G
was used as a secondary antibody (1:10,000; sc-2004, Santa Cruz
Biotechnology, Inc.) for 1 h at room temperature. Expression levels
of GAPDH served as an internal control. Immunoreactive bands were
detected on radiographic films using the enhanced chemiluminescence
detection system (Advansta Inc., Menlo Park, CA USA). ImageJ v1.44p
software (National Institutes of Health, Bethesda, MD, USA) was
used for densitometry and values were normalized to GAPDH
expression levels. The protein expression levels of NLRP3 and
downstream cytokines were normalized to GAPDH and values are
expressed relative to the control group.
Statistical analysis
Data (n=2) are presented as the mean ± standard
deviation or the median and interquartile range. Nonparametric
Mann-Whitney U test was used for between-group comparisons of
expression levels of NLRPs and downstream cytokines. Nonparametric
one-way analysis of variance with Dunn-Bonferroni post-hoc tests
were used for between-group comparisons of alterations in protein
expression levels of NLRP3 in PBMCs stimulated with B19-VP1u,
B19-NS1 or the control group. Association coefficient was
calculated using the nonparametric Spearman's rank association
test. Wilcoxon signed rank test was used to compare expression
levels of NLRP3 and downstream cytokines prior to and following
treatment. P<0.05 was considered to indicate a statistically
significant difference.
Results
Clinical characteristics of patients
with AOSD
Based on the findings of physical examination, the
presence of spiking fever (body temperature ≥39°C), evanescent
rash, arthritis, sore throat and lymphadenopathy were observed in
33, 30, 25, 24 and 13 of 34 patients with AOSD, respectively.
Neither the age at entry nor the proportion of females differed
significantly between the patients with AOSD and the healthy
controls.
Comparison of mRNA levels of NLRPs
between patients with AOSD and controls
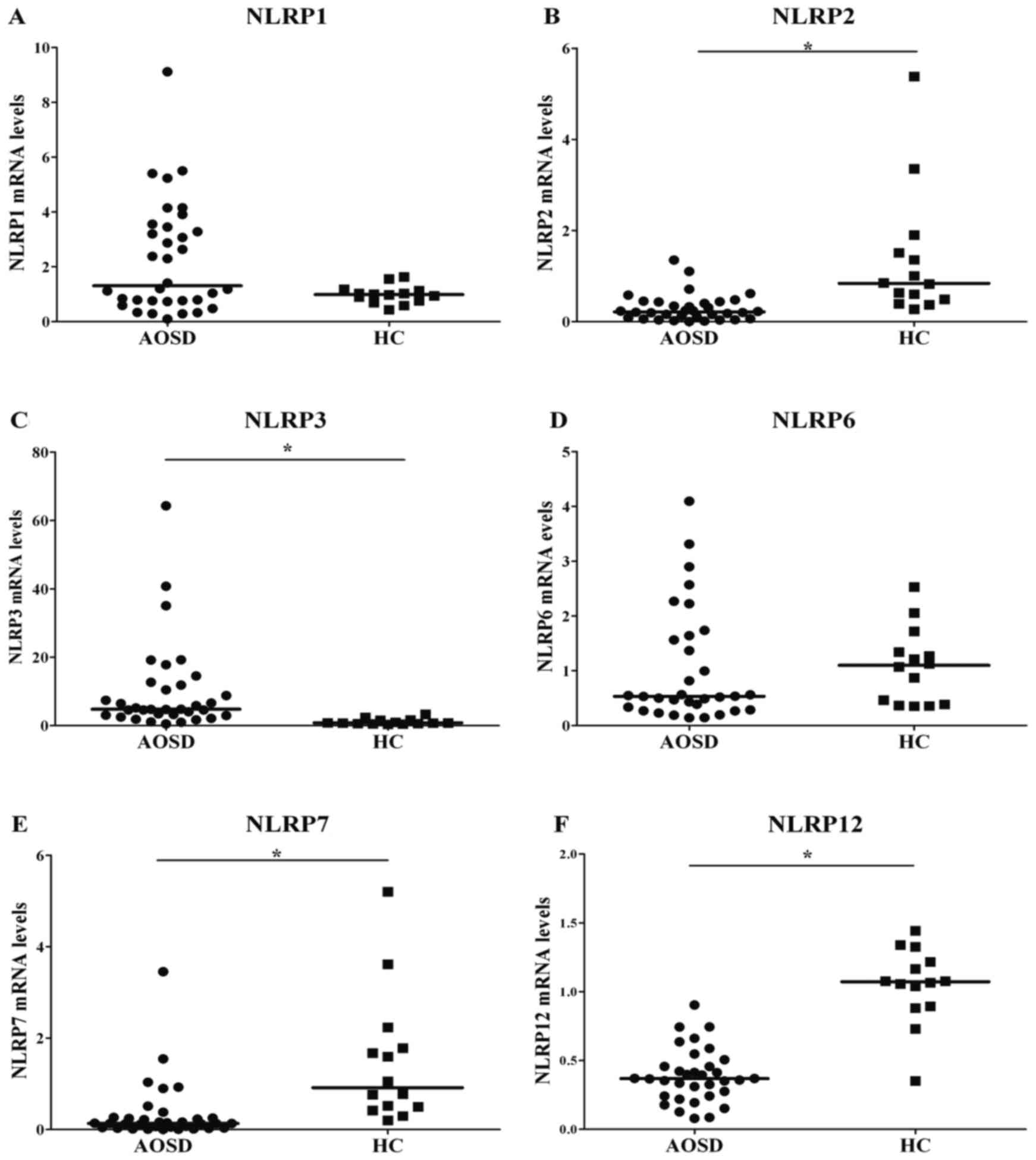
Significantly increased mRNA levels of NLRP3 were
observed in patients with AOSD compared with healthy patients
(P<0.001; Fig. 1 and Table I). By contrast, significantly lower
transcript levels of NLRP2, 7 and 12 were observed in patients with
AOSD compared with the healthy patients. Transcript levels of NLRP1
and 6 did not differ significantly in patients with AOSD compared
with the control group. The mRNA levels of NLRP4, 5, 8, 9, 10, 11,
13 and 14 were too low to be detected by qPCR in patients with AOSD
or healthy controls.
 | Table I.Relative mRNA levels of NLRPs in
patients with AOSD and healthy controls. |
Table I.
Relative mRNA levels of NLRPs in
patients with AOSD and healthy controls.
| NLRP | Relative mRNA
expression levels [median (IQR) fold] |
|---|
| NLRP1 |
|
|
AOSD | 1.31
(0.76–3.48) |
|
Control | 0.99
(0.74–1.14) |
| NLRP2 |
|
|
AOSD | 0.22
(0.08–0.44)a |
|
Control | 0.84
(0.47–1.61) |
| NLRP3 |
|
|
AOSD | 4.79
(3.04–2.05)a |
|
Control | 0.79
(0.61–1.58) |
| NLRP6 |
|
|
AOSD | 0.53
(0.30–1.62) |
|
Control | 1.10
(0.38–1.44) |
| NLRP7 |
|
|
AOSD | 0.13
(0.04–0.25)a |
|
Control | 0.91
(0.48–1.89) |
| NLRP12 |
|
|
AOSD | 0.37
(0.24–0.47)a |
|
Control | 1.07
(0.89–1.24) |
Association between NLRP3 mRNA
expression levels and clinical features in AOSD
Higher levels of NLRP3 expression were observed in
AOSD patients with skin rash or arthritis compared with in those
without (10.91±13.84 vs. 3.66±2.09; 12.11±14.82 vs. 4.37±3.20;
respectively); statistical significance was not observed (P=0.309,
P=0.134, respectively). In addition, no significant differences in
NLRP3 expression levels between AOSD patients with and without sore
throats were observed (11.39±15.10 vs. 6.87±6.38, P=0.372), or
between AOSD patients with and without lymphadenopathy (11.59±12.68
vs. 9.11±13.73, P=0.601).
Correlation between mRNA levels of
NLRPs and expression of downstream inflammasome signaling
components in patients with AOSD
NLRP3 transcript levels were positively associated
with expression levels of inflammasome signaling components and
downstream cytokines including PYCARD (correlation coefficient
r=0.343, P<0.05), caspase-1 (r=0.445, P<0.01), IL-1β
(r=0.379, P<0.05) and IL-18 (r=0.482, P<0.01) in patients
with AOSD. Conversely, NLRP2 transcript levels were inversely
correlated with expression levels of PYCARD (r=−0.389, P<0.05),
caspase-1 (r=−0.515, P<0.01), IL-1β (r=−0.351, P<0.05) and
IL-18 (r=−0.481, P<0.01) in patients with AOSD.
NLRP3 and caspase-1 are associated
with inflammasome signaling in AOSD
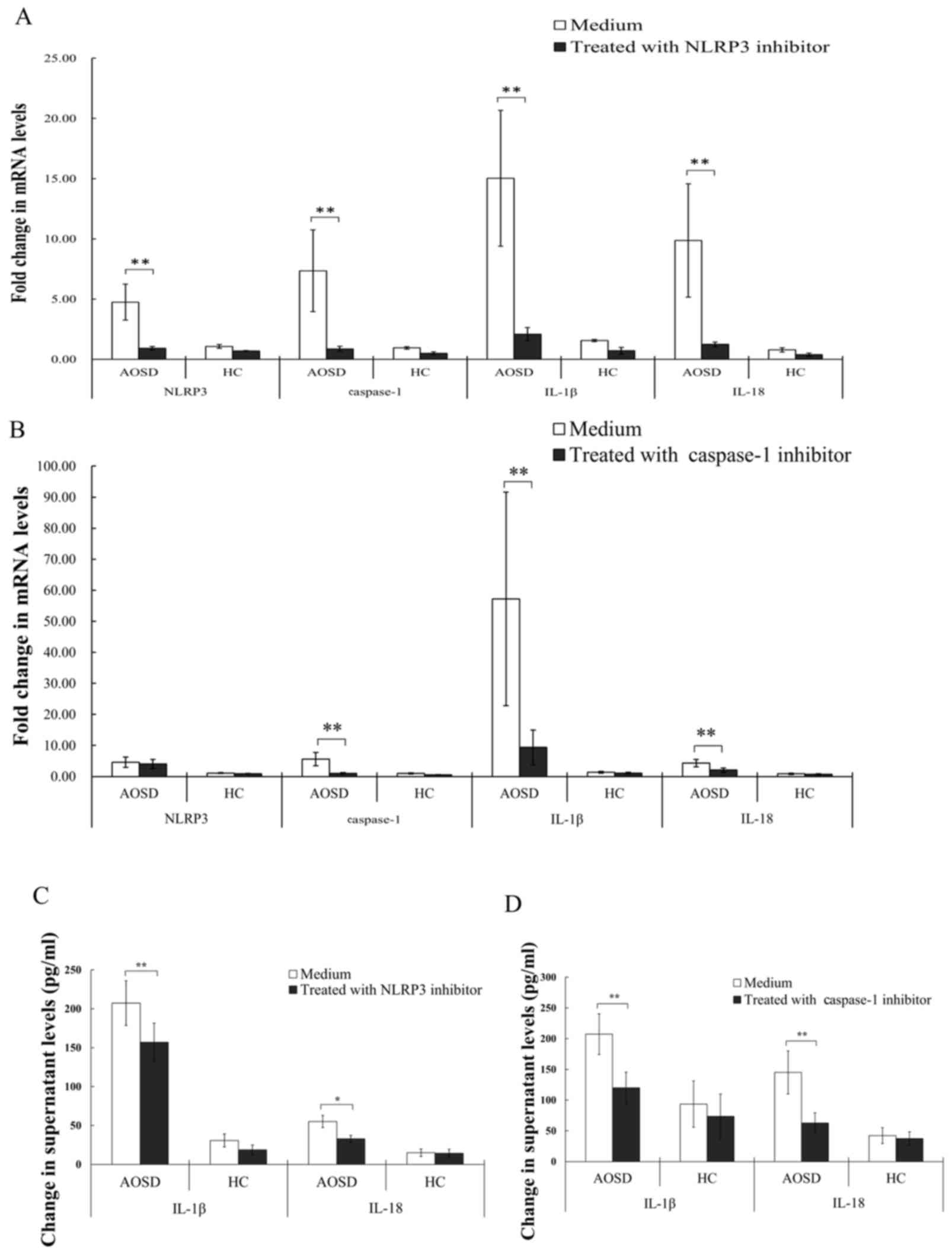
Inflammatory response to inflammasome inhibitors was
evaluated to analyze the NLRP3-inflammasome signaling pathway. The
results obtained indicated that treatment with NLRP3 inhibitor
significantly downregulated mRNA levels of NLRP3, caspase-1, IL-1β
and IL-18 (all P<0.005) in PBMCs from patients with AOSD
(Fig. 2A). Similarly, treatment
with caspase-1 inhibitor significantly reduced mRNA levels of
caspase-1, IL-1β and IL-18 (all P<0.005; Fig. 2B). Treatment of PBMCs from patients
with AOSD with NLRP3 inhibitor significantly reduced IL-1β and
IL-18 levels (P<0.005 and P<0.05, respectively) in the
supernatant, compared with the control treatment group (Fig. 2C). Similarly, treatment of PBMCs
from patients with AOSD with caspase-1 inhibitor significantly
reduced levels of IL-1β and −18, compared with the control group
(both P<0.005; Fig. 2D).
Expression levels of
NLRP3-inflammasome signaling components in PBMCs are stimulated by
parvovirus B19 antigens
To investigate the effect of B19 antigens on NLRP3
activation, PBMC cells were treated with B19 antigens and fold
changes relative to control in transcript levels of NLRP3 and
supernatant levels of downstream cytokines were determined. The
results of the present study revealed that stimulation of PBMCs
from patients with AOSD with B19-NS1 induced significant increases
in NLRP3, caspase-1 and IL-1β compared with healthy patients (all
P<0.0005; Fig. 3A). However,
transcript levels of NLRP3 and caspase-1 in B19-VP1u-stimulated
PBMCs did not differ significantly between patients with AOSD and
healthy controls. B19-NS1 treatment significantly increased
supernatant levels of IL-1β in patients with AOSD compared with
healthy controls (P<0.05). The difference in IL-18 levels was
not statistically different (Fig.
3B).
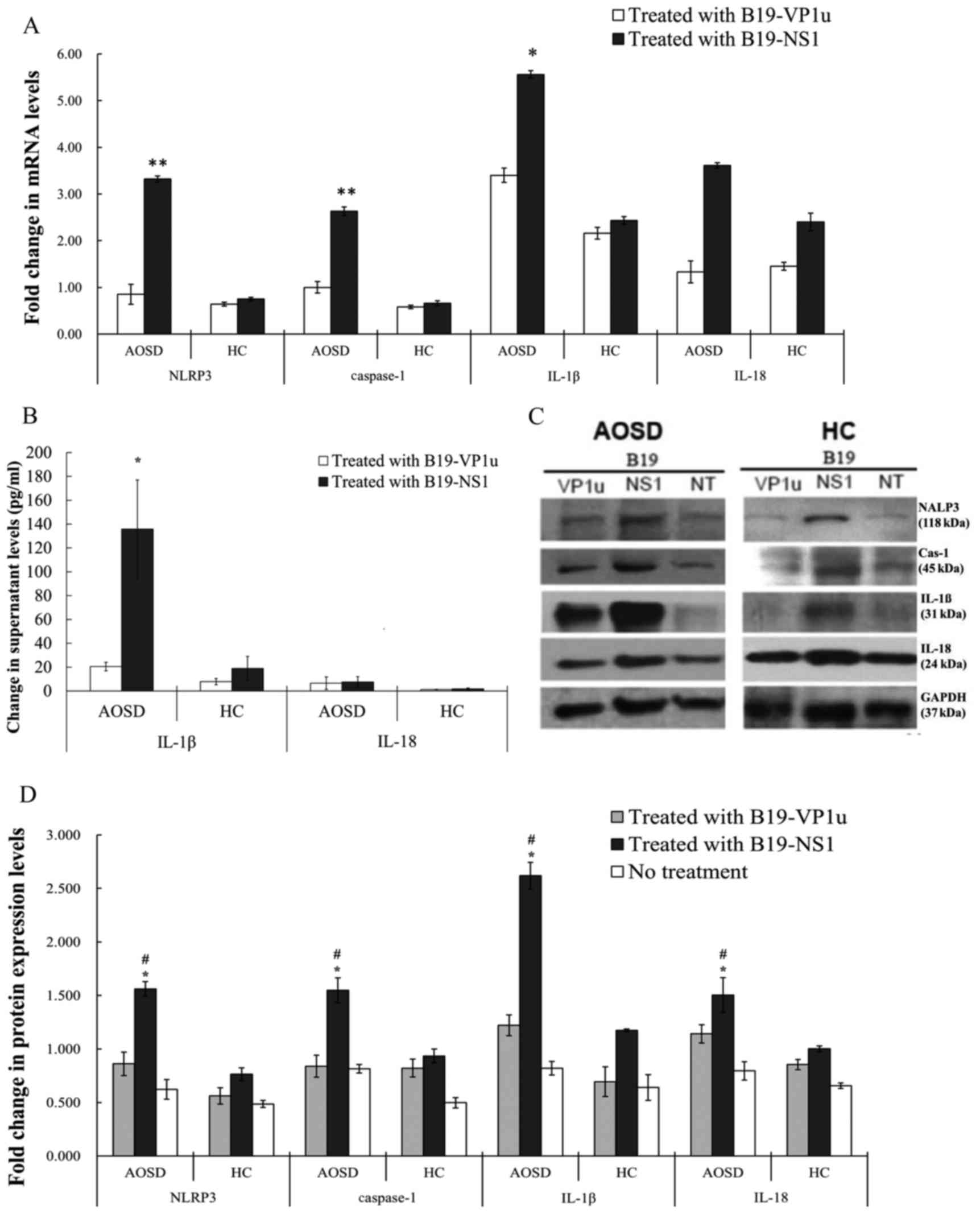 | Figure 3.Alterations in mRNA and protein
expression levels following treatment with B19-VP1u and B19-NS1.
(A) Fold-changes, relative to the healthy control group, in
transcript levels of NLRP3, caspase-1, IL-1β and-18 in PBMCs
stimulated with B19-VP1u and B19-NS1 in patients with AOSD and HCs.
(B) Supernatant levels of IL-1β and IL-18 in PBMCs stimulated with
B19-VP1u and B19-NS1 in patients with AOSD and HCs. (C) Western
blot analysis of expression of NLRP3, Cas-1, IL-1β and-18 and
control GAPDG proteins in PBMC lysates obtained from one AOSD
patient and one HC. (D) Protein expression levels (fold-changes
relative to healthy control group) of NLRP3, caspase-1, IL-1β
and-18 in PBMCs stimulated with B19-VP1u and B19-NS1 in patients
with AOSD and HC. Data are presented as the mean ± standard
deviation. *P<0.05 and **P<0.005 vs. the respective HC group,
#P<0.05, vs. the respective no treatment group. PBMC,
peripheral blood mononuclear cell; AOSD, adult-onset Still's
disease; HC, healthy control; IL, interleukin; NLRP3, NLR family
pyrin domain containing 3; Cas-1, caspase-1; B19-VP1u, human
parvovirus B19 VP1-unique region; B19-NS1, human parvovirus B19
non-structural protein-1. |
Patients with AOSD exhibit increased protein
expression of NLRP3-inflammasome components in PBMC lysates
stimulated with B19-NS1, compared with samples treated with
B19-VP1u and medium only. Stimulation of PBMCs with B19-NS1 induced
an increase in the expression of NLRP3, caspase-1, IL-1β and IL-18
in patients with AOSD compared with healthy controls (all
P<0.05; Fig. 3C and D).
Discussion
Different types of inflammasomes contain different
NLRPs responsible for variable regulation of inflammatory responses
(2,4,26).
The present study demonstrated markedly increased NLRP3 transcript
levels in patients with AOSD compared with healthy controls. NLRP3
expression levels were positively associated with levels of
inflammasome components and downstream cytokines in patients with
AOSD. As previously demonstrated, the results of the present study
confirmed that activation of NLRP3 and caspase-1 is essential for
the inflammatory response in AOSD (27). The above observations suggest
involvement of NLRP3-inflammasome signaling in AOSD pathogenesis.
Parvovirus B19 infection has been previously associated with AOSD
and B19-NS1 has been hypothesized to serve role in NLRP3 activation
in patients with AOSD (22).
Following activation of NLRP3 and caspase-1, release
of IL-1β and IL-18 is the final stage of pathogenesis of
inflammasome-associated AIDs. NLRP3 transcript levels were
positively correlated with the expression levels of inflammasome
signaling components in patients with AOSD. Furthermore, NLRP3 and
caspase-1 inhibitors markedly suppressed expression of IL-1β and
IL-18, indicating that NLRP3-associated caspase-1-dependent
mechanism may mediate the production of downstream cytokines in
AOSD.
mRNA levels of NLRP2, a member of negative
regulatory NLRPs, were inversely correlated with the expression
levels of downstream inflammasome signaling components in patients
with AOSD. In addition, a negative correlation was identified
between NLRP2 levels and NLRP3 levels in patients with AOSD (data
not presented), supporting a hypothesis that NLRP2 competes with
NLRP3 in binding to PYCARD and subsequently activating caspase-1
(28,29). The aforementioned observations
suggest that NLRP2 may serve a role as a negative regulator of
immune responses in AOSD. However, the role of NLRP2-inflammasome
in rheumatic patients remains to be elucidated.
mRNA levels of NLRP7 and NLRP12 were lower in
patients with AOSD compared with healthy controls. Although it
remains to be elucidated whether NLRP7 is an activator or an
inhibitor of inflammasome, it has been hypothesized that NLRP7 can
inhibit NLRP3 and caspase-1-mediated release of IL-1β (30–32).
NLRP12 has been previously reported to suppress the production of
proinflammatory cytokines by inhibiting nuclear factor-κB via a
proteasome-dependent pathway and has been identified as a negative
regulator of inflammation (33).
Although in a previous report NLRP9 has been
implicated in the pathogenesis of AOSD and NLRP10 has been
previously identified as an inflammasome inhibitor (34,35),
mRNA levels of NLRP4, 5, 8, 9, 10, 11, 13, and 14 were undetectable
in the present study. This observation supports the hypothesis that
only a few mammalian NLRPs directly participate in inflammatory
responses and certain NLRPs may be involved in other non-immune
pathways including embryogenesis (36). Another possibility is that the
assay used in the present study demonstrated limited sensitivity
and therefore failed to detect certain NLRPs.
Certain pathogen associated molecular patterns have
been identified as NLRP3 activators (1,3) but
specific ligands of NLRP3 in AOSD remain to be elucidated. Based on
the association of parvovirus B19 infection with AOSD pathogenesis,
we hypothesized that B19-associated antigens may activate the
expression of NLRP3-inflammasome associated signaling (22). Ex vivo experiments included
in the present study are, to the best of our knowledge, the first
to reveal that B19-NS1 can up-regulate the expression of mRNA and
protein levels of NLRP3, indicating that B19-NS1 may be an
activator of NLRP3 in AOSD. The results of the present study
confirm previous hypotheses that adenoviruses (non-enveloped DNA
viruses) can activate NLRP3 and that NLRP3 may be implicated in
immune responses to viral infection (1,37).
There were certain limitations of the present study.
Due to difficulties associated with obtaining biopsy tissues,
expression of NLRPs was not investigated in lesion specimens from
patients with AOSD. Lack of association between expression levels
of NLRP3 and clinical features may be due to the small sample size
in this clinically heterogeneous and uncommon disease. Association
between NLRP3 levels and parvovirus B19 DNA was not investigated in
the present study.
Enhanced expression of NLRP3 resulting in elevated
levels of downstream cytokines may be involved in pathogenesis of
AOSD. Since activators of NLRP3 in AOSD remain to be elucidated,
the presented study provides novel insights into the innate
immunity of AOSD.
Acknowledgements
The present study was supported by the National
Science Council of Taiwan (grant nos. NSC-101-2314-B-010-030-MY3
and NSC-101-2314-B-040-008) and by the Taichung Veterans General
Hospital and National Yang-Ming University (grant. no.
TCVGH-YM1010204). The authors of the present study would also like
to thank Mr. Ted Knoy (Writing Center, Hsinchu, Taiwan, R.O.C.) for
his editorial assistance.
References
|
1
|
Muruve DA, Pétrilli V, Zaiss AK, White LR,
Clark SA, Ross PJ, Parks RJ and Tschopp J: The inflammasome
recognizes cytosolic microbial and host DNA and triggers an innate
immune response. Nature. 452:103–107. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Martinon F, Gaide O, Pétrilli V, Mayor A
and Tschopp J: NALP inflammasome: A central role in innate
immunity. Semin Immunopathol. 29:213–229. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Schroder K and Tschopp J: The
inflammasomes. Cell. 140:821–832. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tschopp J, Martinon F and Burns K: NALPs:
A novel protein family involved in inflammation. Nat Rev Mol Cell
Biol. 4:95–104. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ting JP, Lovering RC, Alnemri ES, Bertin
J, Boss JM, Davis BK, Flavell RA, Girardin SE, Godzik A, Harton JA,
et al: The NLR gene family: A standard nomenclature. Immunity.
28:285–287. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Sidiropoulos PI, Goulielmos G, Voloudakis
GK, Petraki E and Boumpas DT: Inflammasomes and rheumatic diseases:
Evolving concepts. Ann Rheum Dis. 67:1382–1389. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Busso N and So A: Microcrystals as DAMPs
and their role in joint inflammation. Rheumatology (Oxford).
51:1154–1160. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Varghese GP, Uporova L, Halfvarson J,
Sirsjö A and Fransén K: Polymorphism in the NLRP3
inflammasome-associated EIF2K2 gene and inflammatory bowel disease.
Mol Med Rep. 11:4579–4584. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
McDermott MF, Aksentijevich I, Galon J,
McDermott EM, Ogunkolade BW, Centola M, Mansfield E, Gadina M,
Karenko L, Pettersson T, et al: Germline mutations in the
extracellular domains of the 55 kDa TNF receptor, TNFR1, define a
family of dominantly inherited autoinflammatory syndromes. Cell.
97:133–144. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Masters SL, Simon A, Aksentijevich I and
Kastner DL: Horror autoinflammaticus: The molecular pathophysiology
of autoinflammatory disease (*). Annu Rev Immunol. 27:621–668.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kastner DL, Aksentijevich I and
Goldbach-Mansky R: Autoinflammatory disease reloaded: A clinical
perspective. Cell. 140:784–790. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Gerfaud-Valentin M, Jamilloux Y, Iwaz J
and Sève P: Adult-onset Still's disease. Autoimmun Rev. 13:708–722.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kadavath S and Efthimiou P: Adult-onset
Still's disease-pathogenesis, clinical manifestations, and new
treatment options. Ann Med. 47:6–14. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yamaguchi M, Ohta A, Tsunematsu T,
Kasukawa R, Mizushima Y, Kashiwagi H, Kashiwazaki S, Tanimoto K,
Matsumoto Y, Ota T, et al: Preliminary criteria for classification
of adult Still's disease. J Rheumatol. 19:424–430. 1992.PubMed/NCBI
|
|
15
|
Kawashima M, Yamamura M, Taniai M,
Yamauchi H, Tanimoto T, Kurimoto M, Miyawaki S, Amano T, Takeuchi T
and Makino H: Levels of interleukin-18 and its binding inhibitors
in the blood circulation of patients with adult-onset Still's
disease. Arthritis Rheum. 44:550–560. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Choi JH, Suh CH, Lee YM, Suh YJ, Lee SK,
Kim SS, Nahm DH and Park HS: Serum cytokine profiles in patients
with adult onset Still's disease. J Rheumatol. 30:2422–2427.
2003.PubMed/NCBI
|
|
17
|
Chen DY, Chen YM, Lan JL, Lin CC, Chen HH
and Hsieh CW: Potential role of Th17 cells in the pathogenesis of
adult-onset still's disease. Rheumatology (Oxford). 49:2305–2312.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Priori R, Barone F, Alessandri C,
Colafrancesco S, McInnes IB, Pitzalis C, Valesini G and Bombardieri
M: Markely increased IL-18 liver expression in adult-onset Still's
disease-related hepatitis. Rheumatology (Oxford). 50:776–780. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Young NS and Brown KE: Parvovirus B19. N
Engl J Med. 350:586–597. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Dorsch S, Liebisch G, Kaufmann B, von
Landenberg P, Hoffmann JH, Drobnik W and Modrow S: The VP1 unique
region of parvovirus B19 and its constituent phospholipase A2-like
activity. J Virol. 76:2014–2018. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Adamson-Small LA, Ignatovich IV,
Laemmerhirt MG and Hobbs JA: Persistent parvovirus B19 infection in
non-erythroid tissues: Possible role in the inflammatory and
disease process. Virus Res. 190:8–16. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chen DY, Chen YM, Lan JL, Tzang BS, Lin CC
and Hsu TC: Significant association of past parvovirus B19
infection with cytopenia in both adult-onset Still's disease and
systemic lupus erythematosus patients. Clin Chim Acta. 413:855–860.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chomczynski P and Sacchi N: Single-step
method of RNA isolation by acid guanidinium
thiocyanate-phenol-chloroform extraction. Anal Biochem.
162:156–159. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Tzang BS, Tsay GJ, Lee YJ, Li C, Sun YS
and Hsu TC: The association of VP1 unique region protein in acute
parvovirus B19 infection and anti-phospholipid antibody production.
Clin Chim Acta. 378:59–65. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Pontillo A, Girardelli M, Kamada AJ,
Pancotto JA, Donadi EA, Crovella S and Sandrin-Garcia P:
Polymorphisms in inflammasome genes are involved in the
predisposition to systemic lupus erythematosus. Autoimmunity.
45:271–278. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Tschopp J and Schroder K: NLRP3
inflammasome activation: The convergence of multiple signaling
pathways on ROS production. Nat Rev Immunol. 10:210–215. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Bostanci N, Emingil G, Saygan B, Turkoglu
O, Atilla G, Curtis MA and Belibasakis GN: Expression and
regulation of the NALP3 inflammasome complex in periodontal
diseases. Clin Exp Immunol. 157:415–422. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Bruey JM, Bruey-Sedano N, Newman R,
Chandler S, Stehlik C and Reed JC: PAN1/NALP2/PYPAF2, an inducible
inflammatory mediator that regulates NF-kappaB and caspase-1
activation in macrophages. J Biol Chem. 279:51897–51907. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Radian AD, de Almeida L, Dorfleutner A and
Stehlik C: NLRP7 and related inflammasome activating pattern
recognition receptors and their function in host defense and
disease. Microbes Infect. 15:630–639. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Khare S, Dorfleutner A, Bryan NB, Yun C,
Radian AD, de Almeida L, Rojanasakul Y and Stehlik C: An
NLRP7-containing inflammasome mediates recognition of microbial
lipopeptides in human macrophages. Immunity. 36:464–476. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kinoshita T, Wang Y, Hasegawa M, Imamura R
and Suda T: PYPAF3, a PYRIN-containing APAF-1-like protein, is a
feedback regulator of caspase-1-dependent interleukin-1beta
secretion. J Biol Chem. 280:21720–21725. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Pinheiro AS, Eibl C, Ekman-Vural Z,
Schwarzenbacher R and Peti W: The NLRP12 pyrin domain: Structure,
dynamics, and functional insights. J Mol Biol. 413:790–803. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Tadaki H, Saitsu H, Nishimura-Tadaki A,
Imagawa T, Kikuchi M, Hara R, Kaneko U, Kishi T, Miyamae T, Miyake
N, et al: De novo 19q13.42 duplications involving NLRP gene cluster
in a patient with systemic-onset juvenile idiopathic arthritis. J
Hum Genet. 56:343–347. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wang Y, Hasegawa M, Imamura R, Kinoshita
T, Kondo C, Konaka K and Suda T: PYNOD, a novel Apaf-1/CED4-like
protein is an inhibitor of ASC and caspase-1. Int Immunol.
16:777–786. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Fernandes R, Tsuda C, Perumalsamy AL,
Naranian T, Chong J, Acton BM, Tong ZB, Nelson LM and Jurisicova A:
NLRP5 mediates mitochondrial function in mouse oocytes and embryos.
Biol Reprod. 86:138, 1–10. 2012. View Article : Google Scholar
|
|
37
|
Barlan AU, Griffin TM, McGuire KA and
Wiethoff CM: Adenovirus membrane penetration activates the NLRP3
inflammasome. J Virol. 85:146–155. 2011. View Article : Google Scholar : PubMed/NCBI
|