Introduction
The kidney, an organ particularly prone to ischemic
injury, will undergo a period of ischemia during some urologic
surgeries such as renal partial nephrectomy or kidney
transplantation. Ischemic injury leads to necrosis of tubular
epithelial cells and acute renal failure, which is a major cause of
morbidity and mortality. Ischemia-reperfusion injury (IRI) led to
inevitable renal damage. Thus, reducing the damage to ischemic
tissues has been an important therapeutic goal. Various potentially
attractive measures to achieve this goal have been discovered and
applied to protect organs from IRI. In 1986, the phenomenon of
ischemic preconditioning (IPC) which could obviously reduce
myocardial infarct size, was discovered (1). In 1993, an update version-remote
ischemic preconditioning (RIPC)-was initially described by
Przyklenk et al that decreased infarct area (2). Considering these reviews, using RIPC
as a less invasive method, brief cycles of limb ischemia and
reperfusion as a remote precondition stimulus achieved by applying
a simple tourniquet around the lower or upper limb was a
noninvasive and comparatively inexpensive procedure (3,4).
However, results of RIPC were controversial (5,6).
Results from animal experiments and clinical trials indicated that
RIPC might offer neuroprotective effects by regulating multiple
cellular processes (3). RIPC was
previously shown to have a protective effect from renal IRI in rats
preexposed to chronic hypoxia via hypoxia-inducible factor-1α
(HIF-1α). RIPC is extensively documented as a method of protecting
organs, including the kidney, from IRI.
Currently, the mechanisms of RIPC have been
considered to involve pathways, comparatively similar to IPC:
(1) Neuronal pathway, (2) systemic response, (3) humoral pathway (7,8). The
neuronal and systemic response pathway have been studied in depth
whereas the humoral factor that conveyed the preconditioning signal
from the remote to the target has not yet been identified. The
effective endogenous factors currently confirmed are adenosine
(9), bradykinin (10), opioids (11), and calcitonin gene-related peptide
(12), which is released from the
remote organ or tissue during the preconditioning treatment and
conveyed to the target organ through the bloodstream.
Furthermore, the transcription factor HIF-1α has
recently been revealed to be a potential mediator of RIPC-induced
protection (13). Evidence
indicates that HIF-1α played an important role in the kidney and
other organs involved in mediating protection. HIF-1α activation
offered obvious protection to the kidney from IRI for the survival
of renal epithelial cells.
The protective effects of HIF-1α activation in renal
IRI might be mediated by a number of different pathways. Studies
have shown that heme oxygenase-1 and erythropoietin are involved in
this process (14). However, the
exact humoral candidate remained unknown; it appears that the
protective factor is thermolabile, hydrophobic and has a molecular
mass range from 3.5 to 8 kDa (15,16).
Adrenomedullin (ADM), a biologically active peptide,
was first isolated from human pheochromocytoma tissue extracts, and
later affirmed to be present extensively in vascular endothelium,
heart, lung and kidney (17). It
had an important effect on heart and vascular protection,
angiogenesis, central nervous system protection, tumor
growth-promoting action, anti-inflammation, receptors, and
intracellular signalling system (18). In addition, it was regulated by
HIF-1α and its molecular mass was approximately 5 kDa in the
humoral candidate range (19,20).
Thus, the purpose of this study was to investigate
whether RIPC offered protection to kidneys with ischemia
reperfusion injury and to determine whether ADM was the humoral
candidate in RIPC based on the former result.
Materials and methods
Animals
In accordance with principles and guidelines of the
Guide for the Care and Use of Laboratory Animals (US National
Institutes of Health Publication), all experiments were performed
in the laboratory of the Department of Pharmacology at Shanghai
Jiao Tong University School of Medicine. Male SD rats weighing
210±20 g were given standard rat chow with free access to water and
fasted, but allowed water ad libitum 12 h prior to experimentation.
The present study was approved by the Ethics committee of Xinhua
Hospital affiliated to Shanghai Jiao Tong University School of
Medicine (Shanghai, China).
There are two phases of the study: Part 1, a
comparing phase, in which we applied RIPC in a rat model and
compared whether RIPC offered renal protection; and part 2, an
intervention phase, in which we used drugs such as agonists and
antagonists to confirm our assumption of the humoral candidate in
RIPC.
Part 1: Twenty-four rats were randomised into four
groups (n=6 per group): Sham group, ischemia/reperfusion (IR)
group, acute phase RIPC group, and delayed phase RIPC group. The
sham group had sham surgery. The IR group underwent resection
surgery of the right kidney and a microvascular clamp was placed on
the left renal pedicle for 45 min, and then reperfusion resumed
(21). After 30 min of RIPC
(22), the acute RIPC and delayed
RIPC groups underwent the same step as the IR group. The difference
between the acute and delayed groups was that the delayed group
underwent IR surgery after a 24-hour period. All animals were
sacrificed for specimen collection 24 h after surgery.
Part 2: We compared two phases of RIPC and acquired
the better RIPC protocol in part 1. In part 2, 48 rats were
randomly divided into eight groups based on the results in part 1
and the following drugs were used (n=6 per group): Sham+Vehicle
(21,23), IR+Vehicle (21), Delayed+Vehicle, Delayed+YC-1 (2
mg/kg) (23),
Delayed+2-Methoxyestradiol (2-MeOE2) (20 mg/kg) (24), Delayed+DMOG (40 mg/kg) (25), Delayed+Molidustat (BAY 85–3934)
(2.5 mg/kg) (26), IR+ADM (12
µg/kg) (27). All the injections
were given intraperitoneally 2 h before the time of RIPC with an
injection volume of 0.5 ml. The RIPC protocol adopted was on the
basis of part 1 (4,22). YC-1 and 2-MeOE2 were agents that
inhibited HIF-1 activity in vivo and in vitro
(24,28). As a result, YC-1 could also
decrease the expression level of the HIF-1 inducible genes,
including ADM (29,30). DMOG and BAY 85–3934, known as the
prolyl hydroxylase (PHD) inhibitor, slowed the degradation of the
HIF-1α, relatively increasing the expression of HIF-1 (26,31).
Biological samples and tissue
handling
Blood was obtained from the renal artery before
right nephrectomy and left kidney harvest. Urine was collected in
24 h after surgery. The left kidney was harvested for biochemical
analysis, and immunohistochemical and histopathological use. The
levels of urine microalbumin (mALB), serum beta2-microglobulin
(β2-MG), creatinine (Cr), blood urea nitrogen (BUN), together with
superoxide dismutase (SOD), malondialdehyde (MDA), and
myeloperoxidase (MPO), the three of which originated from kidney
tissue were tested by commercial assay kits (Jiancheng
Bioengineering Institute, Nanjing, China). ADM in blood and kidney
tissue was detected by an ELISA kit (Phoenix Pharmaceuticals, Inc.,
Burlingame, CA, USA). Western blot analyses were conducted by using
HIF-1α antibody (1:500; Novus Biologicals, Littleton, CO, USA). To
detect HIF-1α and ADM expression, immunohistochemical staining was
performed by the use of HIF-1α (1:50; Novus Biologicals), ADM
(1:200; Santa Cruz Biotechnology, Inc., Dallas, TX, USA)
antibodies. The histopathological findings on the kidney tissue
stained with hematoxylin and eosin were measured by the tubular
score method (32,33).
Data analysis
Data were given as mean ± standard deviation.
Software used for statistical analysis was Graphpad
Prism® (v5.01 for Windows; GraphPad Software, Inc., La
Jolla, CA, USA) and IBM SPSS Statistics (v19.0 for Windows; IBM
Corp., Armonk, NY, USA). All data were tested using a one-way
analysis of variance with the Tukey multiple comparison test.
P<0.05 was considered to indicate a statistically significant
difference.
Results
Prevention of IR induced renal
dysfunction and oxidative damage by RIPC
A significant increase in levels of Cr, BUN, mALB,
β2-MG, MPO, MDA and decreased activity of SOD were shown in the IR
group, indicating an obvious renal dysfunction and oxidative
damage. Treatment with acute and delayed RIPC significantly
decreased the renal damage with a lower level of Cr, BUN, mALB,
β2-MG, MPO, MDA and increased level of SOD compared with the IR
group. In addition, the indexes of renal dysfunction and oxidative
damage indicators induced by IR were reversed more significantly in
the delayed RIPC, which seemed to have a better protective effect
than the acute phase in part 1 as outlined in Table I (P<0.05). These results
strongly suggested that delayed RIPC produced marked beneficial
profiles against IR-induced renal injury.
 | Table I.The results about indexes of renal
function and oxidative injury indicators. |
Table I.
The results about indexes of renal
function and oxidative injury indicators.
| Group | Cr (µmol/l) | BUN (mmol/) | mALB
(µg/ml) | β2-MG (ng/ml) | MPO (U/g) | MDA (nmol/mg
prot) | SOD (U/mg
prot) |
|---|
| Sham | 49.94±7.14 | 10.72±1.50 | 31.46±4.62 | 327.48±83.79 | 0.13±0.03 | 1.15±0.08 | 206.36±23.92 |
| IR | 408.12±38.48 | 36.11±3.29 | 105.20±8.66 | 1060.88±79.25 | 0.52±0.10 | 2.77±0.24 | 252.16±18.65 |
| Acute |
338.47±37.64a |
29.50±2.30a |
89.23±7.99a |
792.31±61.49a |
0.32±0.08a |
2.42±0.32a |
313.82±21.93a |
| Delayed |
299.66±39.37a |
22.83±3.30a,b |
80.79±9.77a |
676.64±54.80a,b |
0.26±0.05a |
2.33±0.14a |
384.05±41.63a,b |
| Sham+Vehicle | 51.97±11.41 | 13.08±1.55 | 32.05±6.59 | 284.86±92.68 | 0.07±0.03 | 1.19±0.16 | 204.58±15.34 |
| IR+Vehicle | 403.11±19.81 | 35.89±3.00 | 113.80±14.20 | 1026.38±64.49 | 0.48±0.08 | 3.00±0.24 | 220.26±12.19 |
|
Delayed+Vehicle |
286.60±35.75c |
23.02±1.08c |
87.46±7.19c |
698.72±168.23c |
0.23±0.04c |
2.11±0.14c |
315.95±4.86c |
| Delayed+YC-1 |
374.30±29.32d |
35.24±1.63d |
93.55±15.47c |
735.21±197.35c |
0.28±0.03c |
2.65±0.20c,d |
257.50±4.02c,d |
|
Delayed+2-MeOE2 |
382.64±26.12d |
35.77±3.06d | 103.15±11.89 | 846.70±188.84 |
0.29±0.045c |
2.80±0.15d |
231.14±41.88d |
| Delayed+DMOG |
207.62±40.91c,d |
20.62±0.84c |
83.91±7.34c |
637.16±115.27c |
0.14±0.03c,d |
1.72±0.11c,d |
331.26±3.89c |
| Delayed+BAY
85–3934 |
183.16±7.33c,d |
20.86±1.98c |
81.10±6.74c |
598.06±107.90c |
0.16±0.021c |
1.74±0.069c,d |
334.69±20.88c |
| IR+ADM |
397.53±32.37d |
33.10±2.87d |
119.54±9.20d | 833.66±145.21 |
0.33±0.02c,d |
2.54±0.18c,d |
244.74±5.18d |
Modulated effects of RIPC on the renal
ischemic injury related to HIF-1α
Effects of RIPC on the renal ischemic injury related
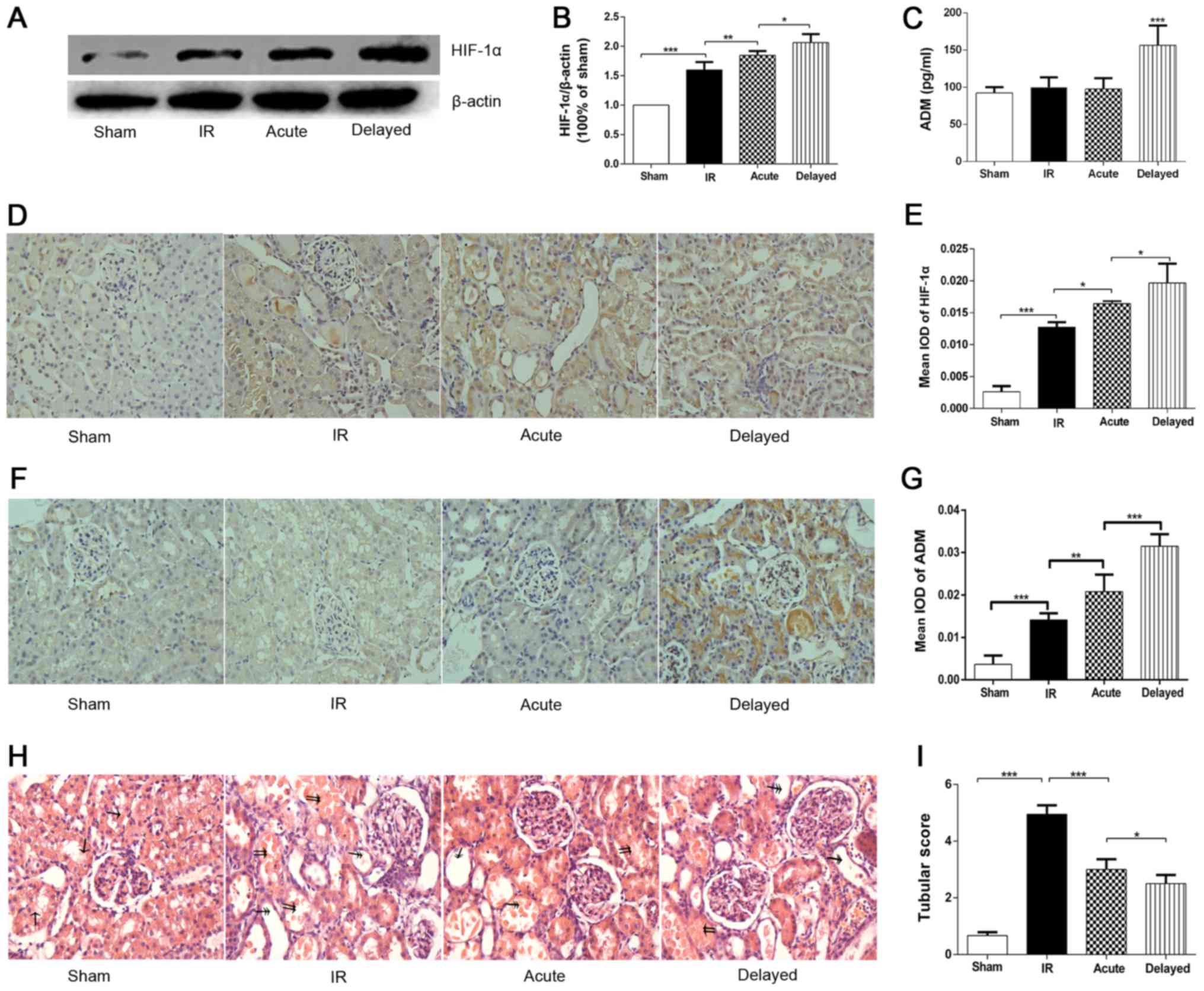
to HIF-1α were assessed (Fig. 1).
Western blot analysis and immunohistochemistry staining of renal
tissue showed an increase in HIF-1α expression of renal tissue in
acute and delayed RIPC groups in comparison with the IR group, and
the level of HIF-1α expression in the delayed RIPC group was higher
than in the acute group (Fig. 1A, B,
D, E; P<0.05). RIPC significantly increased the activity of
SOD and decreased the production of MPO and MDA in the two RIPC
groups compared with the IR group. It was also found that SOD in
the delayed RIPC group was higher than in the acute group as
outlined in Table I (P<0.05).
Interestingly, the blood levels of ADM were increased in the
delayed RIPC group, whereas there was no significant change in the
acute group (Fig. 1C). What's
more, the expression of ADM in delayed RIPC group was also higher
than the acute one (Fig. 1F, G).
In addition, the histopathological results and tubular score also
These results suggested a better protective effect of delayed
rather than acute RIPC (Fig. 1H,
I).
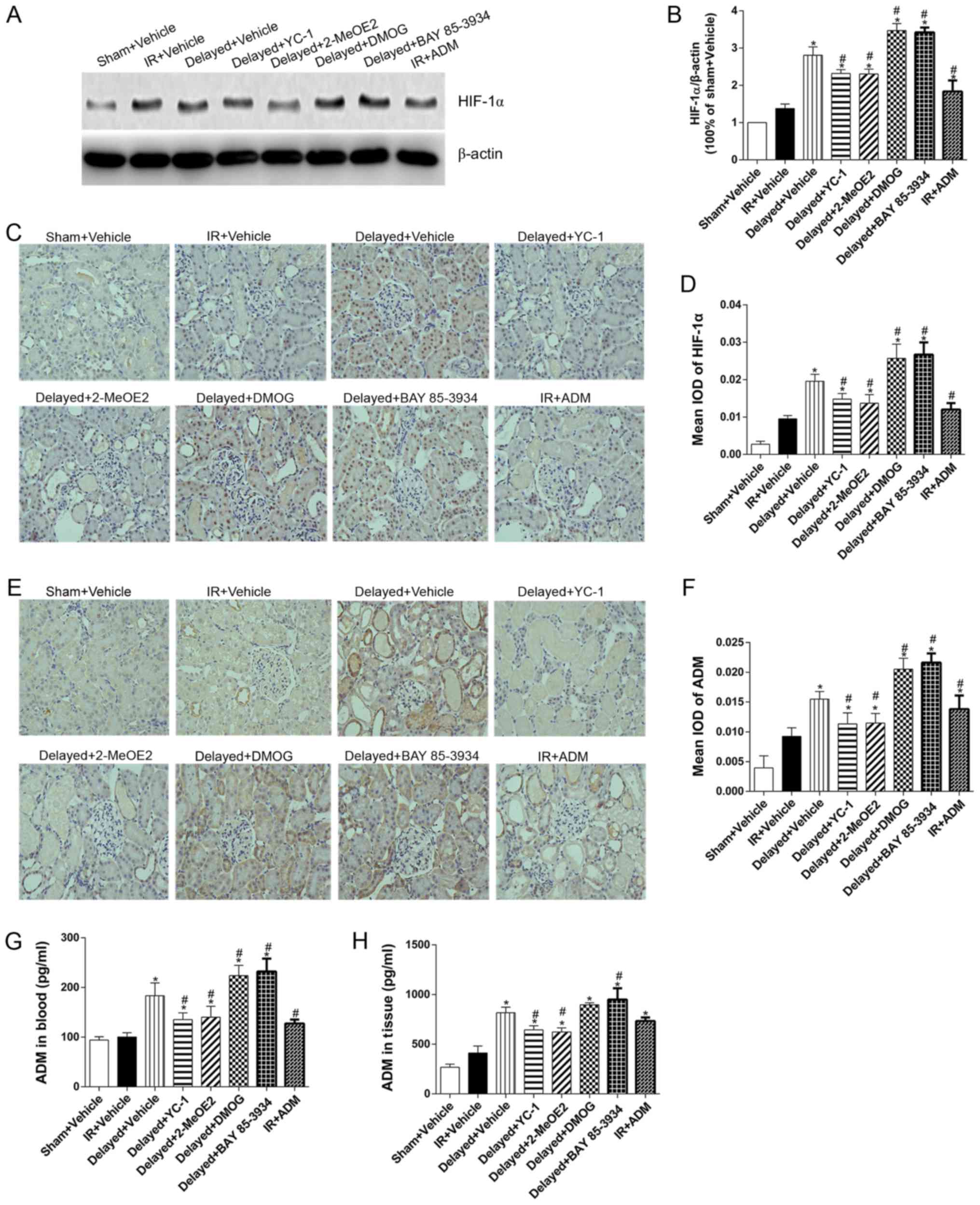
Based on these results, delayed RIPC was chosen as
the effective measure to prevent renal damage in the following
experiments as Delayed+Vehicle group. By pre-treating with YC-1 and
2-MeOE2, both HIF-1α antagonists, the expression of HIF-1α was
significantly downregulated in comparison with the Delayed+Vehicle
group. DMOG and BAY 85–3934 pretreatment could markedly upregulate
HIF-1α expression in the Delayed+DMOG and Delayed+BAY 85–3934 group
than in the Delayed+Vehicle group (Fig. 2; P<0.05). In the Delayed+ YC-1
and Delayed+2-MeOE2 group, the renal injury was obviously
accentuated after YC-1 and 2-MeOE2 pretreatment with a higher
tubular score, increased levels of Cr, BUN, mALB, β2-MG, MPO, and
MDA, and a marked decline in SOD activity that almost counteracted
the RIPC protection (Table I).
However, the protective effect of RIPC could be dramatically
augmented by DMOG in the Delayed+DMOG group and BAY 85–3934 in the
Delayed+BAY 85–3934 group with slighter renal injury than in the
Delayed+Vehicle group by decreasing tubular score and production of
Cr, MPO, and MDA (Table I,
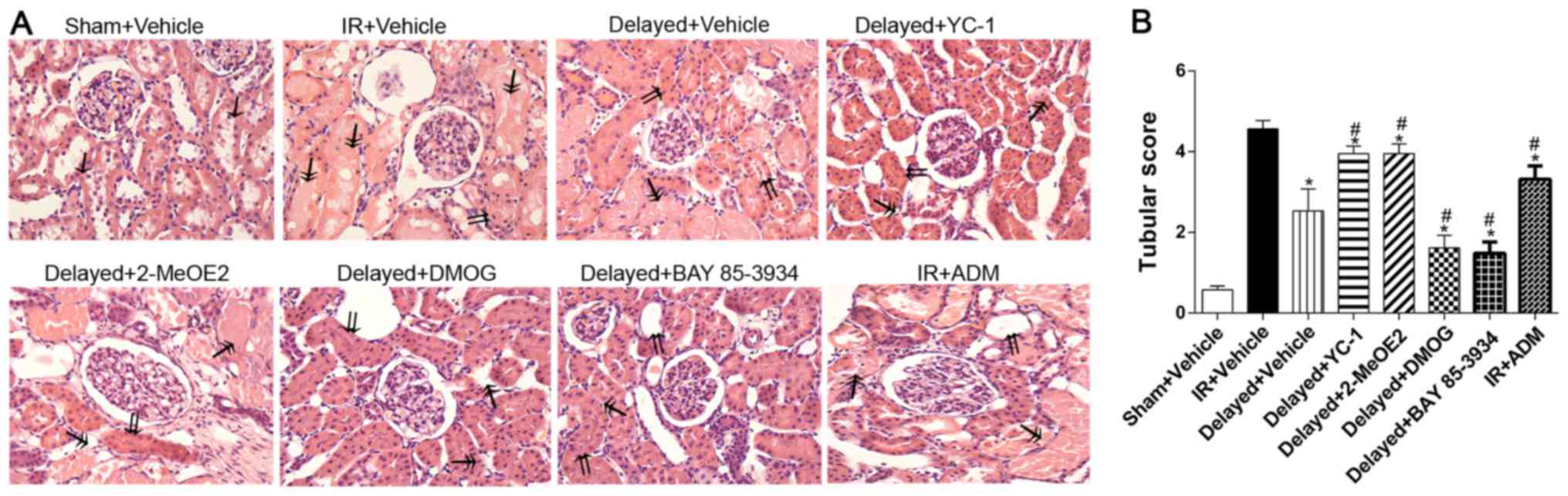
Fig. 3A, B).
Protective effects of RIPC against IR
induced renal damage mediated by ADM
Immunochemistry staining and ELISA detection of
renal tissue revealed that RIPC could markedly upregulate ADM
expression, accompanied by HIF-1α, in the kidney. After receiving
exogenous ADM, the renal tissues of the IR+ADM group showed notably
increased ADM expression but not in the blood compared with the
IR+Vehicle group (Fig. 2E-H).
Treatment with ADM also led to increased activity of SOD, lowering
the levels of MPO and MDA and improving tubular score for renal
histopathological changes (Table
I, Fig. 3A, B). All of these
results revealed that ADM offered slight protection to the kidney
than did the IR+Vehicle group, indicating this humoral factor was
involved in the process of RIPC's protection against IR-induced
renal injury. In addition, drug intervention targeting HIF-1 also
regulated the expression of ADM. Pretreatment with YC-1, the
antagonist of HIF-1, in the Delayed+YC-1 group visibly
downregulated the production of ADM with accelerated renal damage.
So did the Delayed+2-MeOE2 group. However, DMOG and BAY 85–3934,
the agonist of HIF-1, significantly increased the expression of ADM
(Fig. 2E, F, G, H) and improved
renal function (Table I). The
histopathological findings in (Fig.
3A, B) showed a few of tubular epithelial cell flattening,
brush border loss, and apoptosis were observed in the Sham+Vehicle
group. Consequently, the Sham+Vehicle group got the lowest score.
However, the IR+Vehicle group which scored the highest, in addition
to the histopathological changes mentioned for the Sham+Vehicle
group, had the pathological changes of tubular lumen multifocal
necrosis and debris, lumen flat expansion and cast. In the
Delayed+Vehicle, Delayed+YC-1, Delayed+2-MeOE2, Delayed+DMOG,
Delayed+BAY 85–3934 groups, the lumen necrosis and flat expansion
and cast were also observed, but the damage and score was less than
the IR+Vehicle group. All these results indicated that ADM might
act as a downstream humoral factor regulated by HIF-1α. The
relationship between these results and the complete mechanism needs
further investigation.
In summary, we found RIPC could offer a protective
effect against IR-induced renal injury by reversing the renal
dysfunction and oxidative damage indicators in this study. Results
also showed that the effect of RIPC was mediated by HIF-1α and a
downstream humoral factor ADM. However, molecular mechanism is
still unclear and further investigation is needed.
Discussion
RIPC is a less invasive method for reducing
myocardial infarct size developed from ischemic preconditioning
(IPC), offering similar protection by applying ischemia reperfusion
to the organs or tissue far away from the protected target
(3). RIPC was first and frequently
applied during cardiac surgery in the clinic (2). However, it was rarely used in
urology. Currently, the protection offered by RIPC remains
controversial (3,5,6) and
the mechanism is unclear. This was also one of the reasons we
conducted the study.
Similar to IPC, the protective effect of RIPC had
been demonstrated to have two phases or windows, with the first
period of protection occurring and lasting 1 to 4 h immediately
after precondition or acute RIPC, and subsequently the delayed RIPC
appearing at 24 h and lasting 72 h (34). In part 1, the two phases of RIPC
both could reduce the ischemia reperfusion damage and improve renal
function indicators, and the delayed phase offered better
protection that was adopted as RIPC protocol in part 2 (Table I, Fig.
1I).
The results of our experiment suggested HIF-1α plays
a key role in the renal protection offered by RIPC. HIF-1,
consisting of an inducible α subunit and constitutive β subunit,
was a transcriptional factor whose expression was affected by
oxygen concentration in the environment, regulating hundreds of
downstream target gene expressions, including ADM (20). It played an important role in basic
metabolism so that mice would die if the HIF-1α gene was knocked
down (35). HIF-1 also was
involved in cancer metabolism and metastasis (36). First, the expression of HIF-1α was
significantly increased which was a positive correlation for renal
protection. Second, in the Delayed+DMOG group receiving DMOG
injection and the Delayed+BAY 85–3934 group receiving BAY 85–3934
injection, both of the two agents slowed the degradation of the
HIF-1α, relatively increased the expression of the HIF-1,
significantly improved the indicators of Cr, BUN, MPO, MDA, lowered
the tubular score, and further strengthened the protection of
RIPC.
However, in the Delayed+YC-1 group and the
Delayed+2-MeOE2, the expression of HIF-1α was obviously inhibited
and the expression of ADM in blood and tissue was decreased. In
addition, the renal function indicators Cr and BUN, and oxidative
damage indicators MPO, MDA, and tubular score in the Delayed+YC-1
group were worse than in the Delayed + Vehicle group. All the
aforementioned findings suggested changing the expression of HIF-1α
could significantly strengthen or weaken the protective effects of
RIPC.
ADM, a biologically actively peptide regulated by
HIF-1, was first isolated from human pheochromocytoma tissue
extracts (17). It has an
important effect on heart and vascular protection, angiogenesis,
central nervous system protection, tumor growth-promoting action,
anti-inflammation, receptors, and intracellular signaling systems
(18). Circulating ADM was rapidly
metabolized with a half-life of about 20 min, but exogenous ADM
could increase the concentration in plasma and tissue (37). We found that when the expression of
HIF-1α increased or decreased, accordingly the expression of ADM
also changed. High expression of HIF-1α upregulated the
concentration of ADM in blood and tissue. Elevated ADM might play
an anti-inflammatory and antioxidant role, which could reduce the
inflammatory infiltration and lipid peroxidation in the tissue
suffering ischemia reperfusion, and provide protection to the
microvasculature. However, the elevation of ADM was only
significant in the delayed phase of RIPC. Furthermore, in the
presence of exogenous ADM, the expression of HIF-1α was also
increased.
Thus, we inferred the protection in early or acute
phase of RIPC was offered by the existent ADM, SOD, and other
proteins that played an important role in antioxidation. Later in
the delayed phase, the expression of HIF-1α upregulated the
transcription of these proteins, and the system could synthesize
more antioxidants to fight against IRI. Furthermore, there might
also be some positive feedback regulation mechanism between ADM and
HIF-1α that significantly increased the expression of ADM and thus
help play a stronger role in the protection of renal function in
the delayed phase rather than the acute phase. However, more
research on how the positive feedback regulation mechanism worked
is needed.
We found that ADM might play an important role in
the protective effect of RIPC in renal function. After exogenous
ADM was received, the concentration of ADM in blood was not
significantly increased although it was obviously increased in
renal tissue. Exogenous ADM improved the activity of SOD, lowered
the activity of MPO, MDA, and improved tubular score. However,
simply receiving ADM could not take place of the role of RIPC in
renal protection.
In summary, RIPC could provide effective renal
protection against ischemia reperfusion injury. The upregulated
expression of HIF-1α was the key to RIPC's protection. With the
help of HIF-1, a series of antioxidant proteins was upregulated,
which ultimately played a role in fighting against IRI. ADM was an
important member of a series of downstream target proteins
regulated by HIF-1, and it might form a positive feedback
regulation pathway with HIF-1α, which made the delayed phase of
RIPC exhibit stronger renal protection.
Acknowledgements
The present study was sponsored by the Natural
Science Foundation of Shanghai (grant no. 14ZR1426900).
References
|
1
|
Murry CE, Jennings RB and Reimer KA:
Preconditioning with ischemia: A delay of lethal cell injury in
ischemic myocardium. Circulation. 74:1124–1136. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Przyklenk K, Bauer B, Ovize M, Kloner RA
and Whittaker P: Regional ischemic ‘preconditioning’ protects
remote virgin myocardium from subsequent sustained coronary
occlusion. Circulation. 87:893–899. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Tapuria N, Kumar Y, Habib MM, Abu Amara M,
Seifalian AM and Davidson BR: Remote ischemic preconditioning: A
novel protective method from ischemia reperfusion injury-a review.
J Surg Res. 150:304–330. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wever KE, Warlé MC, Wagener FA, van der
Hoorn JW, Masereeuw R, van der Vliet JA and Rongen GA: Remote
ischaemic preconditioning by brief hind limb ischaemia protects
against renal ischaemia-reperfusion injury: The role of adenosine.
Nephrol Dial Transplant. 26:3108–3117. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Remote Preconditioning Trialists' Group, ;
Healy DA, Khan WA, Wong CS, Moloney MC, Grace PA, Coffey JC, Dunne
C, Walsh SR, Sadat U, et al: Remote preconditioning and major
clinical complications following adult cardiovascular surgery:
Systematic review and meta-analysis. Int J Cardiol. 176:20–31.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yang Y, Lang XB, Zhang P, Lv R, Wang YF
and Chen JH: Remote ischemic preconditioning for prevention of
acute kidney injury: A meta-analysis of randomized controlled
trials. Am J Kidney Dis. 64:574–583. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hausenloy DJ and Yellon DM: Remote
ischaemic preconditioning: Underlying mechanisms and clinical
application. Cardiovasc Res. 79:377–386. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Saxena P, Newman MA, Shehatha JS,
Redington AN and Konstantinov IE: Remote ischemic conditioning:
Evolution of the concept, mechanisms, and clinical application. J
Card Surg. 25:127–134. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Pell TJ, Baxter GF, Yellon DM and Drew GM:
Renal ischemia preconditions myocardium: Role of adenosine
receptors and ATP-sensitive potassium channels. Am J Physiol.
275:H1542–H1547. 1998.PubMed/NCBI
|
|
10
|
Schoemaker RG and van Heijningen CL:
Bradykinin mediates cardiac preconditioning at a distance. Am J
Physiol Heart Circ Physiol. 278:H1571–H1576. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Patel HH, Moore J, Hsu AK and Gross GJ:
Cardioprotection at a distance: Mesenteric artery occlusion
protects the myocardium via an opioid sensitive mechanism. J Mol
Cell Cardiol. 34:1317–1323. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tang ZL, Dai W, Li YJ and Deng HW:
Involvement of capsaicin-sensitive sensory nerves in early and
delayed cardioprotection induced by a brief ischaemia of the small
intestine. Naunyn Schmiedebergs Arch Pharmacol. 359:243–247. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kant R, Diwan V, Jaggi AS, Singh N and
Singh D: Remote renal preconditioning-induced cardioprotection: A
key role of hypoxia inducible factor-prolyl 4-hydroxylases. Mol
Cell Biochem. 312:25–31. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hill P, Shukla D, Tran MG, Aragones J,
Cook HT, Carmeliet P and Maxwell PH: Inhibition of hypoxia
inducible factor hydroxylases protects against renal
ischemia-reperfusion injury. J Am Soc Nephrol. 19:39–46. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Serejo FC, Rodrigues LF Jr, da Silva
Tavares KC, de Carvalho AC and Nascimento JH: Cardioprotective
properties of humoral factors released from rat hearts subject to
ischemic preconditioning. J Cardiovasc Pharmacol. 49:214–220. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Shimizu M, Tropak M, Diaz RJ, Suto F,
Surendra H, Kuzmin E, Li J, Gross G, Wilson GJ, Callahan J and
Redington AN: Transient limb ischaemia remotely preconditions
through a humoral mechanism acting directly on the myocardium:
Evidence suggesting cross-species protection. Clin Sci (Lond).
117:191–200. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kitamura K, Kangawa K, Kawamoto M, Ichiki
Y, Nakamura S, Matsuo H and Eto T: Adrenomedullin: A novel
hypotensive peptide isolated from human pheochromocytoma. Biochem
Biophys Res Commun. 192:553–560. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kato J and Kitamura K: Bench-to-bedside
pharmacology of adrenomedullin. Eur J Pharmacol. 764:140–148. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kubo K, Tokashiki M, Kuwasako K, Tamura M,
Tsuda S, Kubo S, Yoshizawa-Kumagaye K, Kato J and Kitamura K:
Biological properties of adrenomedullin conjugated with
polyethylene glycol. Peptides. 57:118–121. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Majmundar AJ, Wong WJ and Simon MC:
Hypoxia-inducible factors and the response to hypoxic stress. Mol
Cell. 40:294–309. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ateş E, Genç E, Erkasap N, Erkasap S,
Akman S, Firat P, Emre S and Kiper H: Renal protection by brief
liver ischemia in rats. Transplantation. 74:1247–1251. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Oxman T, Arad M, Klein R, Avazov N and
Rabinowitz B: Limb ischemia preconditions the heart against
reperfusion tachyarrhythmia. Am J Physiol. 273:H1707–H1712.
1997.PubMed/NCBI
|
|
23
|
Zhang Z, Yan J, Taheri S, Liu KJ and Shi
H: Hypoxia-inducible factor 1 contributes to N-acetylcysteine's
protection in stroke. Free Radic Biol Med. 68:8–21. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Schaible EV, Windschügl J, Bobkiewicz W,
Kaburov Y, Dangel L, Krämer T, Huang C, Sebastiani A, Luh C, Werner
C, et al: 2-Methoxyestradiol confers neuroprotection and inhibits a
maladaptive HIF-1α response after traumatic brain injury in mice. J
Neurochem. 129:940–954. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhao X, Jin Y, Li H, Wang Z, Zhang W and
Feng C: Hypoxia-inducible factor 1 alpha contributes to pulmonary
vascular dysfunction in lung ischemia-reperfusion injury. Int J
Clin Exp Pathol. 7:3081–3088. 2014.PubMed/NCBI
|
|
26
|
Flamme I, Oehme F, Ellinghaus P, Jeske M,
Keldenich J and Thuss U: Mimicking hypoxia to treat anemia:
HIF-stabilizer BAY 85–3934 (Molidustat) stimulates erythropoietin
production without hypertensive effects. PLoS One. 9:e1118382014.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Dwivedi AJ, Wu R, Nguyen E, Higuchi S,
Wang H, Krishnasastry K, Marini CP, Ravikumar TS and Wang P:
Adrenomedullin and adrenomedullin binding protein-1 prevent acute
lung injury after gut ischemia-reperfusion. J Am Coll Surg.
205:284–293. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yeo EJ, Chun YS, Cho YS, Kim J, Lee JC,
Kim MS and Park JW: YC-1: A potential anticancer drug targeting
hypoxia-inducible factor 1. J Natl Cancer Inst. 95:516–525. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zudaire E, Martı́nez A and Cuttitta F:
Adrenomedullin and cancer. Regul Pept. 112:175–183. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hu CJ, Wang LY, Chodosh LA, Keith B and
Simon MC: Differential roles of hypoxia-inducible factor 1alpha
(HIF-1alpha) and HIF-2alpha in hypoxic gene regulation. Mol Cell
Biol. 23:9361–9374. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Yuan Q, Bleiziffer O, Boos AM, Sun J,
Brandl A, Beier JP, Arkudas A, Schmitz M, Kneser U and Horch RE:
PHDs inhibitor DMOG promotes the vascularization process in the AV
loop by HIF-1a up-regulation and the preliminary discussion on its
kinetics in rat. BMC Biotechnol. 14:1122014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Paller MS, Hoidal JR and Ferris TF: Oxygen
free radicals in ischemic acute renal failure in the rat. J Clin
Invest. 74:1156–1164. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Yurdakul T, Kulaksizoglu H, Pişkin MM,
Avunduk MC, Ertemli E, Gokçe G, Barişkaner H, Byükbaş S and Kocabas
V: Combination antioxidant effect of α-tocoferol and erdosteine in
ischemia-reperfusion injury in rat model. Int Urol Nephrol.
42:647–655. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Loukogeorgakis SP, Panagiotidou AT,
Broadhead MW, Donald A, Deanfield JE and MacAllister RJ: Remote
ischemic preconditioning provides early and late protection against
endothelial ischemia-reperfusion injury in humans: Role of the
autonomic nervous system. J Am Coll Cardiol. 46:450–456. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Cai Z, Luo W, Zhan H and Semenza GL:
Hypoxia-inducible factor 1 is required for remote ischemic
preconditioning of the heart. Proc Natl Acad Sci USA. 110:pp.
17462–17467. 2013; View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Tsai YP and Wu KJ: Hypoxia-regulated
target genes implicated in tumor metastasis. J Biomed Sci.
19:1022012. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kitamura K, Kangawa K, Kawamoto M, Ichiki
Y, Nakamura S, Matsuo H and Eto T: Adrenomedullin: A novel
hypotensive peptide isolated from human pheochromocytoma. 1993.
Biochem Biophys Res Commun. 425:548–555. 2012. View Article : Google Scholar : PubMed/NCBI
|