Introduction
Previous studies have demonstrated that septins are
involved in various biological processes, including cell mitosis,
polarity determination, vesicle trafficking and apoptosis (1–4). It
has been demonstrated that septin 7 (SEPT7) inhibits cell cycle
progression in yeast, due to its high degree of homology to cell
division control protein 10 (5).
Furthermore, it has been revealed that SEPT7 inhibits glioma cell
proliferation and invasion, and induces apoptosis, suggesting that
SEPT7 inhibits the progression of glioma (6).
A previous study demonstrated that melatonin can
induce cell apoposis in hFOB 1.19 human osteoblastic cells by
activating the endoplasmic reticulum stress (ERS)-associated
eukaryotic translation initiation factor 2α (eIF2α)-cyclic
AMP-dependent transcription factor ATF-4 (ATF4) pathway and
subsequently triggered the cascade effects of DNA damage-inducible
transcript 3 protein, caspase-3 and mitogen-activated protein
kinase 8 (7). It has also been
previously demonstrated that the expression level of 78 kDa
glucose-regulated protein homolog (GRP78) increases when ERS occurs
(8).
Zhang et al (9) demonstrated that SEPT7 silencing
inhibits the proliferation and promotes the apoptosis of human
breast cancer cells, which indicated that the present of SEPT7 can
promote proliferation and inhibit apoptosis. Furthermore, miR-127
is able to inhibit the proliferation of hepatocellular carcinoma
cells (HCC) by suppressing the expression of SEPT7, indicating that
SPET7 can promote proliferation and inhibit apoptosis (10). The aforementioned studies both
demonstrated that in tumor cells, SEPT7 can promote proliferation
and inhibit apoptosis, but in normal cells, especially in human
osteoblasts, its function remains to be elucidated. The present
study aims to elucidate the underlying mechanism of SEPT7 in
melatonin-induced cell apoptosis in human osteoblast cell line hFOB
1.19.
MicroRNAs (miRNAs) are small, non-coding RNAs that
are 19–22 nucleotides long and post-transcriptionally regulate
protein expression by targeting protein-coding genes involved in
the proliferation, differentiation, apoptosis and migration of
cancer cells (11–16). Previous studies have demonstrated
that miR-590-3p inhibits hepatocellular carcinoma cell growth by
targeting transcriptional enhancer factor TEF-1, promotes
osteogenic differentiation by suppressing expression of adenomatous
polyposis coli protein and stabilizing β-catenin, and contributes
to the development of radioresistance in human glioblastoma cells
by directly targeting leucine-rich repeats and immunoglobulin-like
domains protein 1 (17–19). Although miR-590 serves potential
roles in cancer development, to the best of our knowledge, it
remains unknown whether miR-590 affects normal human osteoblasts.
In the present study, miR-590-3p was used following the
identification of overlapping microRNAs that target SEPT7 using
different databases (miRDB, DIANA and Targetscan). Experiments were
performed to elucidate whether SEPT7 is a novel direct target by
which miR-590-3p exerts its effect, including in vitro
experiments to investigate the role of miR-590-3p in
melatonin-induced apoptosis in the human fetal osteoblastic cell
line hFOB 1.19.
Abnormal proliferation and apoptosis of osteoblasts
may occur in patients with osteoporosis (20). Melatonin, which simulates
apoptosis, may be used in future studies to establish a novel type
of osteoporosis model and further investigate the mechanism
underlying osteoporosis. The present study improved the
understanding of the mechanism underlying ERS and apoptosis induced
by melatonin in hFOB 1.19 cells, providing a theoretical basis for
its subsequent applications of melatonin.
Materials and methods
In silico analysis
The miRNAs which target SEPT7 were predicted by
identifying overlapping microRNAs across different databases
(Targetscan, http://www.targetscan.org/; miRDB, http://mirdb.org/; and DIANA, http://diana.imis.athena-innovation.gr/DianaTools/index.php?r=site/index).
It was observed that miR-590-3p was the only microRNA which
overlapped in all three databases (Fig. 1).
Cell culture and reagents
The hFOB 1.19 normal human fetal osteoblastic cell
line was provided by the Department of Biochemistry and Molecular
Biology, Mayo Clinic (Rochester, MN, USA) and established as
previously described (21), and
was maintained in a 1:1 mixture of Dulbecco's minimum essential
(DME) and F12 medium without phenol red (Hyclone; GE Healthcare
Life Sciences, Logan, UT, USA), supplemented with 10% fetal bovine
serum (FBS; Clark Bioscience, Richmond, VA, USA) in 5%
CO2 at 37°C. The medium was replaced every other day.
Cells were used for further experiments when they reached passages
8–11. Melatonin (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) was
dissolved in 0.2% dimethyl sulfoxide; cells were subsequently
treated at the indicated doses of melatonin (2, 4 and 6 mM) or
vehicle media (DME and F12) containing 10% FBS for 24 h. Melatonin
was obtained from Sigma-Aldrich; Merck KGaA (Darmstadt, Germany).
Primary monoclonal antibodies against proteins were purchased from
Abcam (Cambridge, MA, USA). Goat anti-rabbit secondary antibodies
were obtained from OriGene Technologies, Inc. (Beijing, China).
Luciferase assay
The wild-type or mutant SEPT7 3′-untranslated
regions (3′-UTR; Shanghai GeneChem Co., Ltd., Shanghai, China) were
cloned into the pmirGLO luciferase reporter vector (Promega
Corporation, Madison, WI, USA) and co-transfected with either
miR-590-3p mimic or miR-negative control (NC) (Shanghai GeneChem
Co., Ltd.) into 293T cells (Scientific Center of China Medical
University, Shenyang, China), using Lipofectamine® 2000
(Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA, USA). The
pRL-TK Renilla luciferase reporter vector was transfected
into cells as an internal control. A total of 24 h following
transfection, luciferase activity was measured using a
Dual-Luciferase® Reporter assay system (Promega
Corporation), following cell lysis with Passive Lysis buffer,
included in the Reporter Assay System. Relative luciferase activity
was calculated as the ratio of firefly luciferase activity to
Renilla luciferase activity.
Annexin V-fluorescein isothiocyanate
(FITC)/propidium iodide (PI) staining and analysis using flow
cytometry
An Annexin V FITC/PI staining kit (Dojindo Molecular
Technologies, Inc., Kumamoto, Japan) was used to assess cell
apoptosis following the manufacturer's protocol. Briefly, cells
were trypsinized, washed in PBS, and resuspended in binding buffer.
Cells were stained with FITC-conjugated Annexin V and PI, and
analyzed using a BD LSRFortessa™ cell analyzer (BD
Biosciences, Franklin Lakes, NJ, USA) and quantified using BD
FACSDiva (version 6.2; BD Biosciences). Annexin V positive cells
were defined as apoptotic.
Overexpression plasmid construction
and rescue experiment
SEPT7 was cloned into GV230 plasmids (200 ng;
Shanghai GeneChem Co., Ltd., Shanghai, China) between
XhoI/KpnI sites to overexpress SEPT7 in hFOB 1.19
cells. Full-length SEPT7 gene (4377 bp; reference sequence
NM_001011553) was amplified by polymerase chain reaction (PCR). The
following primers were used for PCR: Forward,
5′-CTGCTCACAATAGTTGATACCCC-3′ and reverse,
5′-TGTTCACTCGTGATTCTGCATT-3′. The PrimeSTAR HS DNA polymerase, was
obtained from Shanghai GeneChem Co., Ltd., and cycled for 30 cycles
following initial denaturation (98°C for 5 min) with the following
parameters: 98°C for 10 sec, 55°C for 10 sec, 72°C for 90 sec; and
72°C for 8 min. Following enzyme digestion (Exnase™ II, 1 µl) using
ClonExpress II One Step Cloning kit (Vazyme, Piscataway, NJ, USA)
and sequencing, the PCR product was cloned into the
XhoI/KpnI sites of the GV230 expression vector. The
recombinant GV230-SEPT7 plasmid was confirmed via endonuclease
digestion and DNA sequencing (Shanghai GeneChem Co., Ltd.) prior to
transfection into hFOB 1.19 cells using Lipofectamine
2000® (Invitrogen; Thermo Fisher Scientific, Inc.,
Waltham, MA, USA). The rescue experiment was preformed following
transfection of the SEPT7 siRNA and the concentration of the SEPT7
overexpression plasmid was 45 nM. Time interval between
transfection and subsequent experimentation was 24 h.
SEPT7 small interfering (si)RNA
transfection
Cells were cultured in DME and F12 medium
supplemented with 10% FBS in a humidified incubator at 37°C and 5%
CO2. At 70–80% confluence, cells were transfected with
60 nM SEPT7 siRNA (forward, 5′-CGACUACAUUGAUAGUAAAUU-3′ and
reverse, 5′-UUUACUAUCAAUGUAGUCGAU-3′; Shanghai Genechem Co., Ltd.)
using Lipofectamine® 2000 according to the
manufacturer's protocol (Invitrogen; Thermo Fisher Scientific,
Inc.). There were three control groups: Blank control, transfection
reagent control (to control for potentially toxic influence of
Lipofectamine® 2000 and the influence of Lipofectamine
2000 on the expression of the target gene) and scramble siRNA
control (5′-GAAATTTATAACGATCAGTCT-3′) (Shanghai Genechem Co.,
Ltd.). The time interval between transfection and subsequent
experimentation was 24 h.
Western blotting
The cells were divided into 5 treatment groups
(control; scramble siRNA; miR-590-3p inhibitor (Shanghai Genechem
Co., Ltd., 60 nM, 37°C for 24 h) + scramble siRNA + 4 mM melatonin;
miR-590-3p inhibitor + SEPT7 siRNA + 4 mM melatonin; 4 mM
melatonin), 4 mM melatonin was selected as the experiment condition
according to a previous study (7).
Following treatment, proteins were extracted from cells using
radioimmunoprecipitation assay lysis buffer (Beyotime Institute of
Biotechnology, Shanghai, China), for 30 min at 4°C. The supernatant
containing total protein was harvested and proteins were quantified
using the bicinchoninic acid method. Aliquots containing 50 µg
proteins were separated on 12% SDS-PAGE gels and transferred to
polyvinylidene fluoride membranes at 60 V for 2 h at 4°C. Following
transfer, membranes were immediately soaked in blocking buffer,
containing 25 mg bovine serum albumin (Beyotime Institute of
Biotechnology) in TBS to final volume of 0.5 l, at 4°C for 2 h.
Subsequently, proteins were incubated with primary antibodies
against GRP78 (cat. no. ab21685); phosphorylated (p)-eIF2α (cat.
no. ab4837), SEPT7 (cat. no. ab186021) and β-actin (cat. no.
ab8226; all 1:5,000 dilution) overnight at 4°C. Proteins were then
incubated with goat anti-rabbit immunoglobulin G horseradish
peroxidase-conjugated secondary antibody (cat. no. ab6721) at
1:10,000 dilution, for 2 h at room temperature. The DNR Imaging
System (DNR Bio-Imaging Systems, Ltd., Neve Yamin, Israel) was used
to visualize specific bands using the BeyoECL Plus kit (Beyotime
Institute of Biotechnology) according to the manufacturer's
protocol and the optical density of each band was measured using
Image J software (version 1.51; National Institutes of Health,
Bethesda, MD, USA).
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
miRNAs were extracted from hFOB 1.19 cells using the
E.Z.N.A.® Total RNA Midi kit (Omega Bio-Tek, Norcross,
GA, USA) according to the manufacturer's protocol and quantified
spectrophotometrically at a wavelength of 260 nm, with acceptable
CCLX/280 ratios between 1.8 and 2.0. RNA quality was also verified
by 1% agarose gel electrophoresis and staining with 1 µg/ml
ethidium bromide. The reagent used for reverse transcription was
purchased from the Promega Corporation. Reverse transcription was
conducted in a total volume of 20 µl [1.0 µg total RNA, 1.0 µl
forward primer, 1.0 µl reverse primer, 9.5 µl H2O,
mixing at 85°C for 5 min, mixing with 2.0 µl 10 mM dNTP, 0.5 µl
RNase inhibitor, 0.5 µl U6 primer, 4.0 µl 5X buffer and 0.5 µl
M-MLV reverse transcriptase (200 units; cat. no. M1701) at 30°C for
10 min, 43°C for 50 min and 85°C for 10 min], the U6 primers were:
Forward, 5′-CTCGCTTCGGCAGCACA-3′ and reverse,
5′-AACGCTTCACGAATTTGCGT-3′. Subsequently, qPCR was performed on
LightCycler® 480 High-Resolution Melting Master (Roche
Diagnostics, Basel, Switzerland). Specific primers for miR-590-3p
(forward, 5′-TAATTTTATGTATAAGCTAGT-3′ and reverse,
5′-GCTGAGGTGCTGTGGT-3′) were obtained from Shanghai GeneChem Co.,
Ltd. (Shanghai, China). Amplifications were conducted in a total
volume of 20 µl [10.0 µl of 2X SYBR Premix Ex Taq II (Takara
Biotechnology Co., Ltd.) 0.8 µl forward primer, 0.8 µl reverse
primer, 2.0 µl DNA template, 6.4 µl double distilled
H2O]. The following thermocycling conditions were used
for PCR: Initial denaturation at 95°C for 30 sec; 40 cycles of 95°C
for 5 sec and 60°C for 30 sec. β-actin (forward,
5′-CAGGGCGTGATGGTGGGCA-3′ and reverse,
5′-CAAACATCATCTGGGTCATCTTCTC-3′) was obtained from Shanghai
GeneChem Co., Ltd. (Shanghai, China) and used as an internal
control. Analysis of melting curve was used to support the
reliability of the results. Relative expression was calculated
using the 2−ΔΔCq method (22).
Statistical analysis
SPSS software (version 20.0; IBM Corp., Armonk, NY,
USA) was used for data processing. An independent samples t-test or
one-way analysis of variance, followed by the Student-Newman-Keuls
test, were used to evaluate the differences between groups. The
results are presented as the mean ± standard error of the mean. The
N-fold gene expression values in gene expression ≤0.5 and >2
were considered to be significant, compared with the values
obtained for the control genes. P<0.05 was considered to
indicate a statistically significant difference.
Results
miR-590-3p expression following
treatment with melatonin
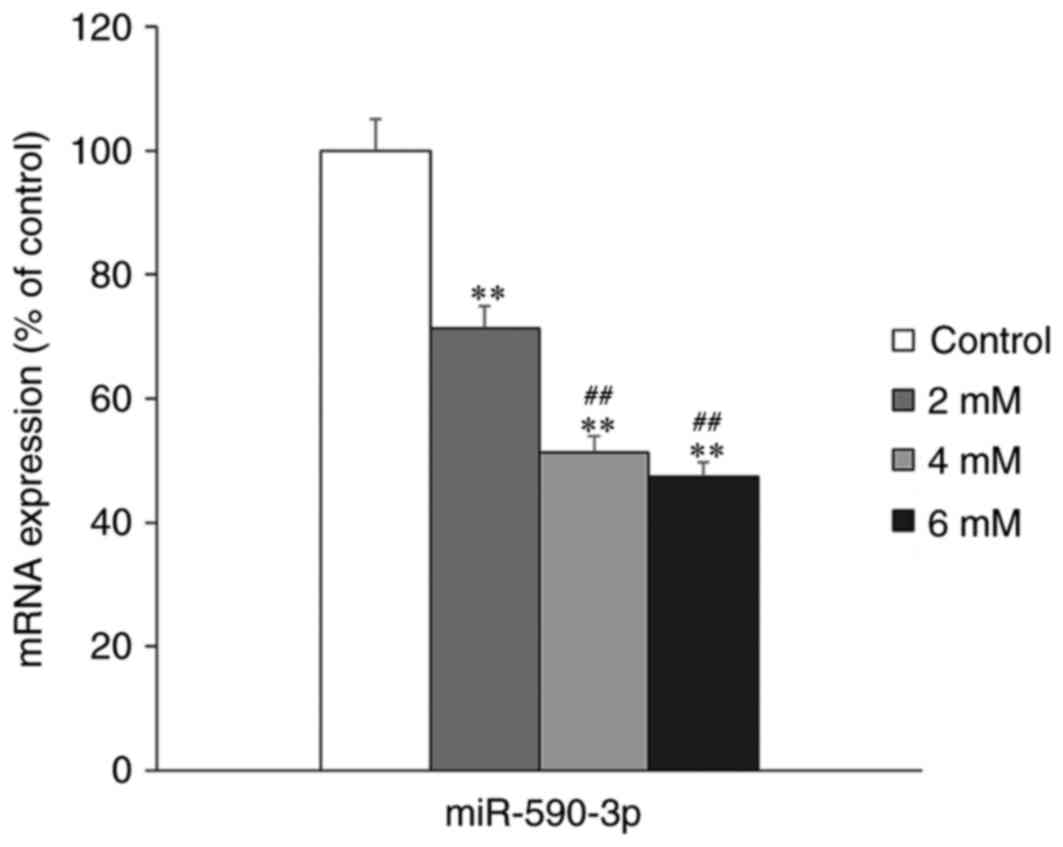
The effect of melatonin treatment on miR-590-3p
expression in hFOB 1.19 cells was examined using RT-qPCR. The
expression of miR-590-3p was significantly decreased in the
melatonin-treated groups compared with the control group
(P<0.01; Fig. 2). Furthermore,
it was determined that miR-590-3p expression was significantly
lower in the high concentration melatonin-treated groups (4 and 6
mM), compared with the low melatonin concentration group (2 mM;
P<0.01; Fig. 2).
Expression of SEPT and the association
between SEPT7 and miR-590-3p
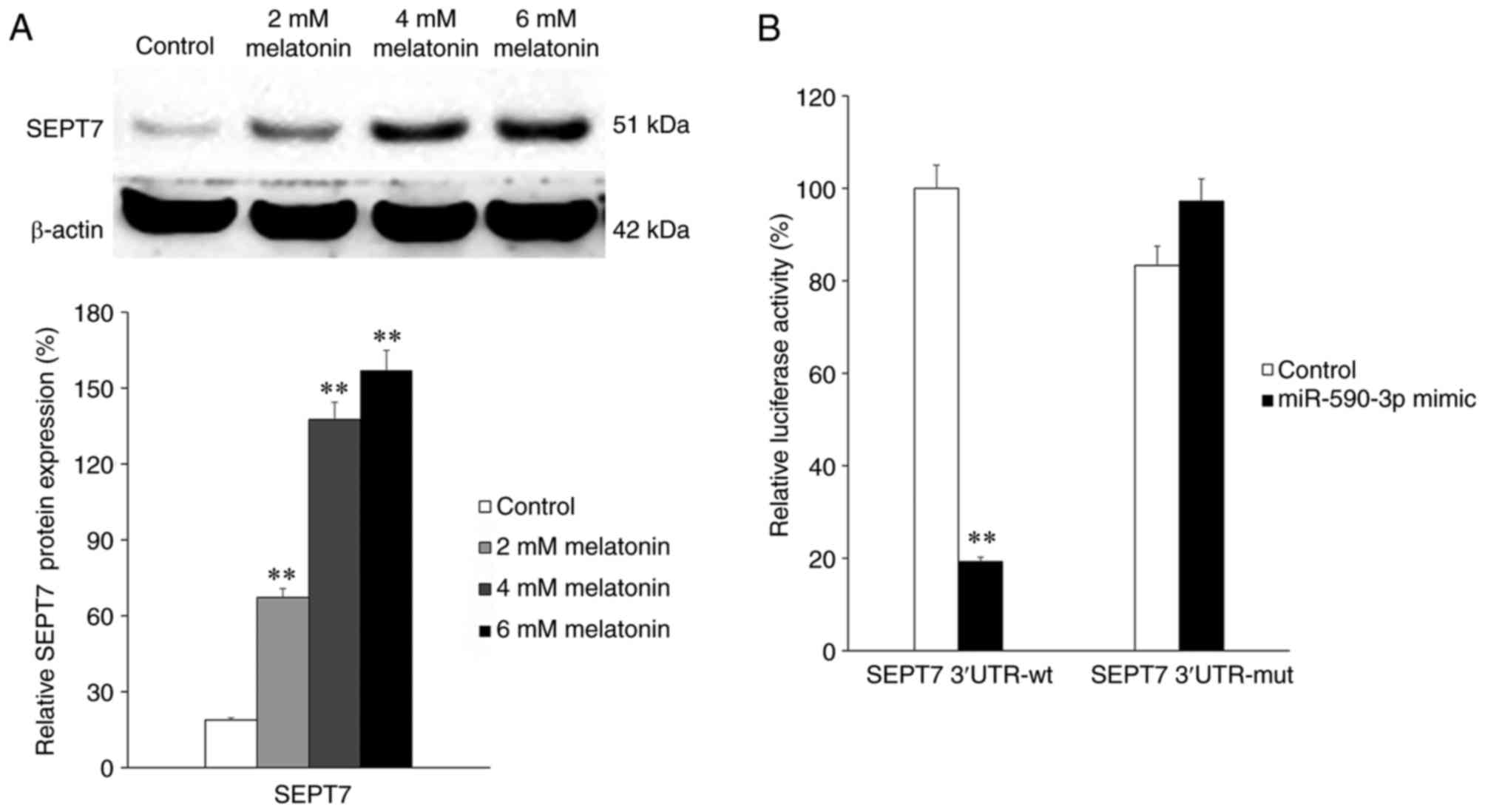
Subsequently, SEPT7 expression in groups treated
with different concentrations of melatonin was determined. The
results indicated that the expression of SEPT7 significantly
increased following treatment with melatonin in a
concentration-dependent manner (all P<0.01; Fig. 3A). This indicates that melatonin
treatment is positively associated with SEPT7 expression. To
elucidate the association between miR-590-3p and SEPT7, a
luciferase reporter assay was performed. SEPT7 3′-UTRs containing
wild-type or mutant potential target sites of miR-590-3p were
constructed and co-transfected with the miR-590-3p mimic into 293T
cells. The results demonstrated that the miR-590-3p mimic
significantly decreased the luciferase activity of wild-type SEPT7
3′-UTR compared with the control (P<0.01), whereas the
luciferase activity of the mutant SEPT7 3′-UTR was not
significantly affected by the miR-590-3p mimic (Fig. 3B).
miR-590-3p expression and cell
apoptosis in the human osteoblast cell line hFOB 1.19
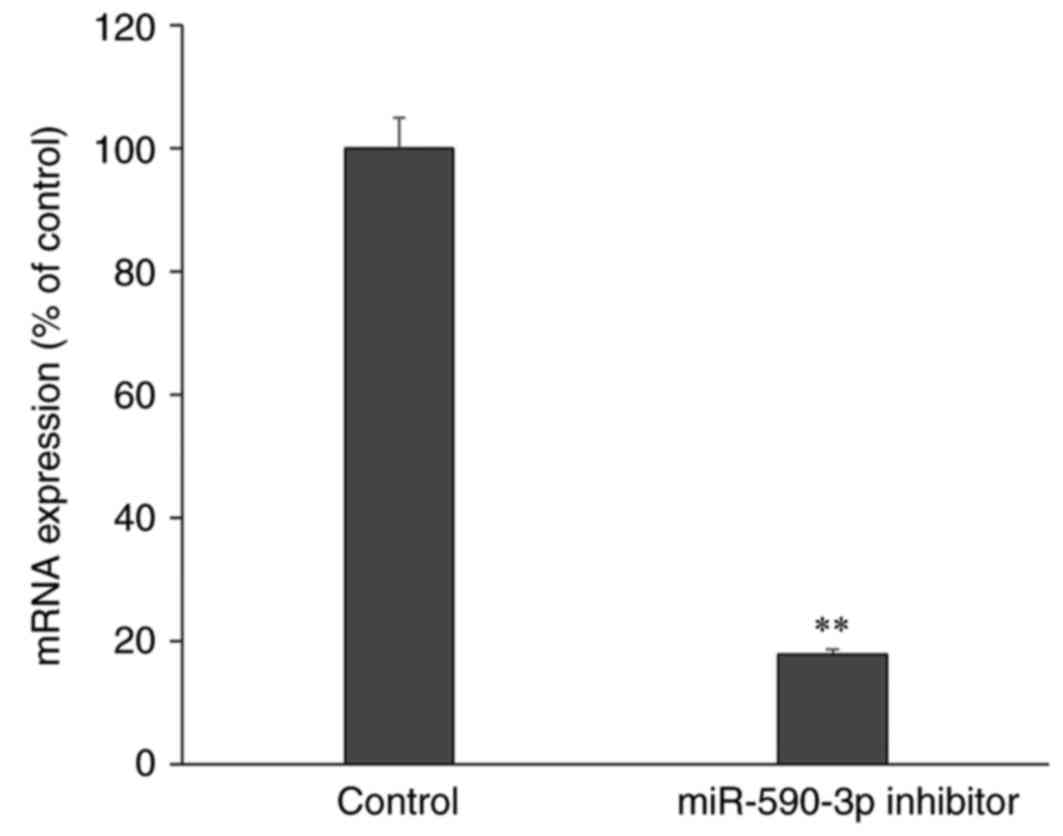
Following transfection with miR-590-3p inhibitor,
the expression of miR-590-3p was significantly decreased compared
with control cells that did not undergo transfection, which
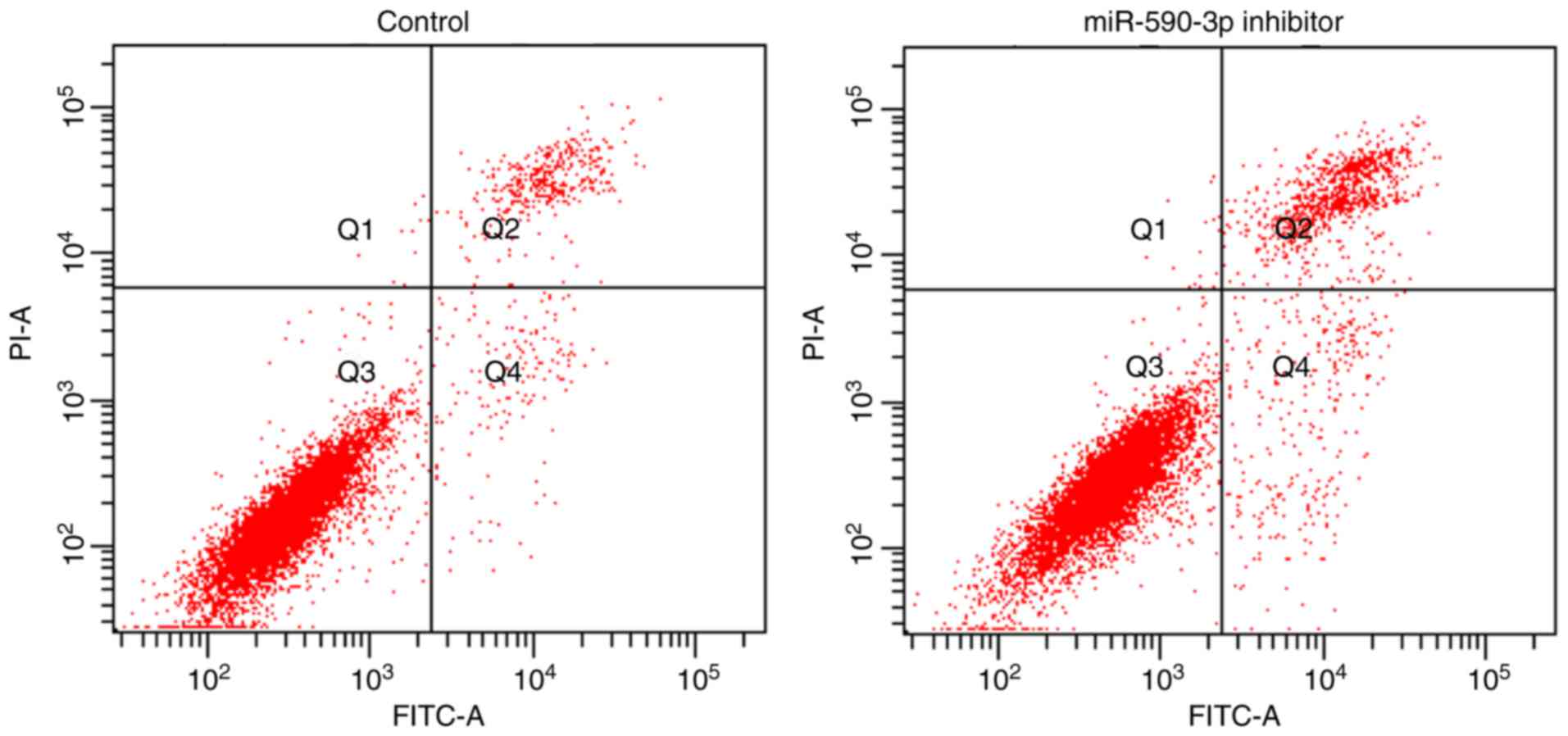
confirmed a successful miR-590-3p inhibition (P<0.01; Fig. 4). Furthermore, the apoptosis rate
was increased following transfection with the miR-590-3p inhibitor
compared with the control group (P<0.01; Fig. 5).
The association between miR-590-3p and
SEPT7
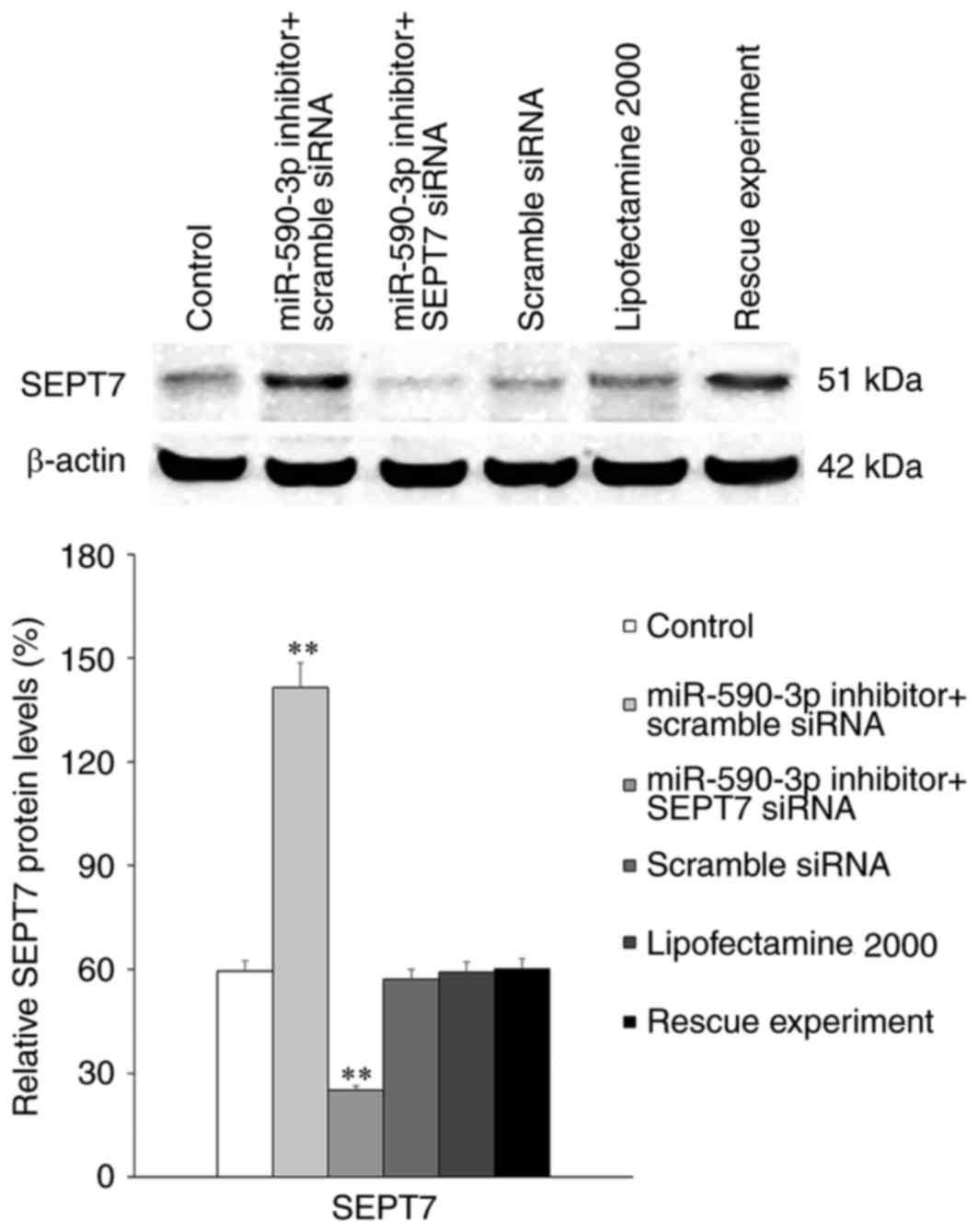
Transfection of the miR-590-3p inhibitor
significantly increased the expression of SEPT7 in hFOB 1.19 cells
compared with the control group (P<0.01; Fig. 6). However, this increased SEPT7
expression was abolished following transfection of SEPT7 small
interfering (si)RNA (P<0.01).
The effects of miR-590-3p, SEPT7 and
melatonin treatment on apoptosis and the expression of
ERS-associated proteins
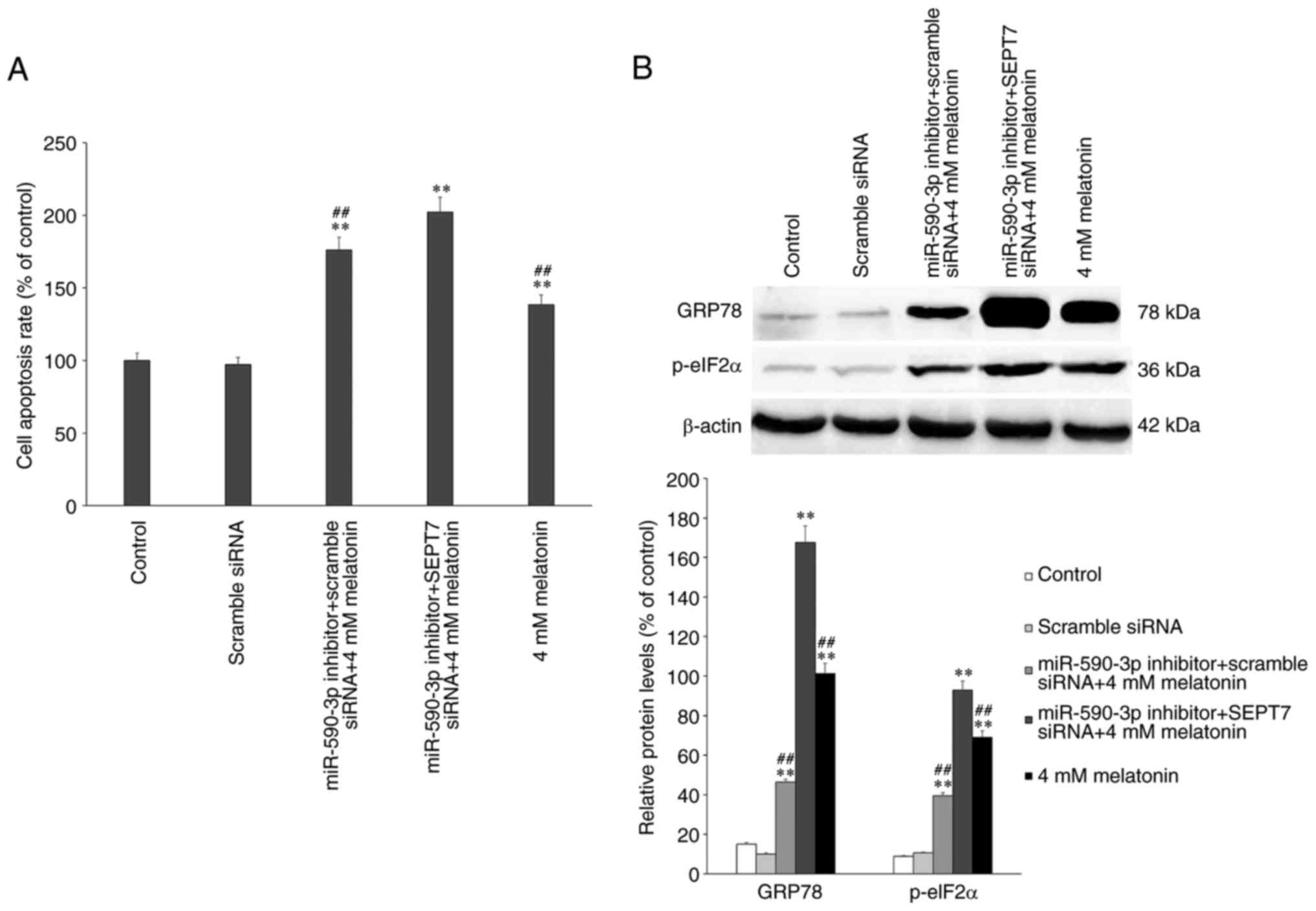
The expression of ERS- and apoptosis-associated
proteins were assessed by western blotting following treatment with
4 mM melatonin alone or in combination of miR-590-3p inhibitor
and/or SEPT7 siRNA. The results demonstrated that, in the 4 mM
melatonin treatment group, apoptosis occurred as previously
described and was significantly increased compared with the control
group (P<0.01; Fig. 7A)
(7), while in the miR-590-3p
inhibitor treatment groups, the rate of apoptosis was significantly
increased compared with the control and the melatonin-treated group
(P<0.01; Fig. 7A). The
apoptotic rate and ERS-associated protein expression levels
increased significantly in the miR-590-3p inhibitor + SEPT7 siRNA +
4 mM melatonin treatment group, compared with the other groups
(Fig. 7). The expression of
ERS-associated proteins was assessed and it was demonstrated that
the expression of GRP78 and p-eIF2α were significantly increased in
the miR-590-3p inhibitor + SEPT7 siRNA group compared with the
other groups (P<0.01) (Fig.
7B).
Discussion
Melatonin serves roles in aging, reproduction, tumor
growth and various biological progresses (23–25).
A previous research conducted by the authors of the present study
demonstrated that melatonin may cause apoptosis by inducing ERS in
hFOB 1.19 cells and that during melatonin-induced apoptosis, the
expression of proteins associated with the classic apoptotic
pathway, including p-eIF2α, are upregulated, which is indicative of
the occurrence and extent of melatonin-induced apoptosis (7).
Septins are proteins that belong to a protein
superfamily. They weigh 30–65 kDa and exhibit conserved structures
(26). Studies have demonstrated
that septins, along with tubulin, microfilaments and intermediate
fibrin, make up the cytoskeleton and serve roles in the
transportation of intracellular substances, regulation of cell
division and the cell cycle and physiological processes including
apoptosis (27–30). The results of the present study
demonstrated that miR-590-3p expression was downregulated in the
human fetal osteoblastic cell line hFOB 1.19 following treatment
with melatonin. This decrease occurred in a dose-dependent manner,
indicating that increasing concentrations of melatonin may decrease
the expression of miR-590-3p. This means that melatonin
concentration-associated increases in the expression of SEPT7 may
be negatively associated with miR-590-3p expression. Furthermore,
the results of the luciferase reporter assay indicated that SEPT7
was a direct target of miR-590-3p. An undergoing study performed by
the authors of the present study suggests that SEPT7 upregulation
inhibits melatonin-induced cell apoptosis by suppressing ERS in
hFOB 1.19 cells. In the present study, SEPT7 expression was
significantly increased following transfection with the miR-590-3p
inhibitor compared with the control group; therefore, it was
hypothesized that miR-590-3p inhibition may protect hFOB 1.19 cells
from melatonin-induced apoptosis. However, the results of the
current study contradict this hypothesis; the inhibition of
miR-590-3p expression increased the rate of apoptosis rather than
decreasing it. Following treatment with 4 mM melatonin for 24 h,
the miR-590-3p inhibitor and/or SEPT7 siRNA were transfected. The
results demonstrated that in the 4-mM melatonin-treated group, the
expression of GRP78 and p-eIF2α increased, as did the rate of
apoptosis. The rate of apoptosis significantly increased in the
miR-590-3p inhibitor + scramble siRNA + 4 mM melatonin-treated
group and peaked in the miR-590-3p inhibitor + SEPT7 siRNA + 4 mM
melatonin-treated group. Furthermore, in the miR-590-3p inhibitor +
scramble siRNA + 4 mM melatonin group, the expression of the
ERS-associated proteins GRP78 and p-eIF2α was the lowest of all the
treatment groups, which indicated that SEPT7 may be a protective
factor in melatonin-induced apoptosis. ERS expression peaked in the
miR-590-3p inhibitor + SEPT7 siRNA + 4 mM melatonin-treated group
compared with the 4 mM melatonin-treated group. An undergoing study
suggests that, increased expression of SEPT7 may suppress ERS.
Consequently, miR-590-3p inhibition may active other factors that
promote apoptosis via non-ERS pathways, to oppose the inhibitory
effect of SEPT7 on apoptosis.
In conclusion, the present study indicated that
miR-590-3p is a potential regulator of melatonin-induced apoptosis.
SEPT7, target gene of miR-590-3p, was inhibited by the presence of
miR-590-3p under normal circumstances. When cells were treated with
melatonin which induced apoptosis, the level of miR-590-3p
decreased leading an increase of SEPT7, which can exert its
protective effect on hFOB 1.19 cells. The inhibition of miR-590-3p
may initially be activated by treatment with high concentrations of
melatonin, upregulating the expression of SEPT7, which may induce
an anti-apoptotic effect. High concentration of melatonin in human
osteoblast cell line hFOB 1.19 leads to apoptosis (7). It can be hypothesized that miR-590-3p
may target proapoptotic genes and SEPT7 as a target of miR-590-3p
may prevent apoptosis. The results of the present study may improve
understanding of the association between miRNAs and cell apoptosis
in the human fetal osteoblast cell line hFOB 1.19. This may be used
to identify future methods of preventing and treating
osteoporosis.
Acknowledgements
The present study was supported by the National
Natural Science Foundation of China Grant (grant no. 81472044) and
the Shenyang Science and Technology Program, Population and Health
Special (grant no. 17-230-9-04). The authors of the present study
would also like to thank Dr M. Subramaniam (Department of
Biochemistry and Molecular Biology, Mayo Clinic, Rochester, MN,
USA) for providing the human fetal osteoblastic cell line hFOB
1.19.
References
|
1
|
Beites CL, Xie H, Bowser R and Trimble WS:
The septin CDCrel-1 binds syntaxin and inhibits exocytosis. Nat
Neurosci. 2:434–439. 1999. View
Article : Google Scholar : PubMed/NCBI
|
|
2
|
Field CM and Kellogg D: Septins:
Cytoskeletal polymers or signalling GTPases? Trends Cell Biol.
9:387–394. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Larisch S, Yi Y, Lotan R, Kerner H, Eimerl
S, Tony Parks W, Gottfried Y, Birkey Reffey S, de Caestecker MP,
Danielpour D, et al: A novel mitochondrial septin-like protein,
ARTS, mediates apoptosis dependent on its P-loop motif. Nat Cell
Biol. 2:915–921. 2000. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kartmann B and Roth D: Novel roles for
mammalian septins: From vesicle trafficking to oncogenesis. J Cell
Sci. 114:839–844. 2001.PubMed/NCBI
|
|
5
|
Hou M, Liu X, Cao J and Chen B: SEPT7
overexpression inhibits glioma cell migration by targeting the
actin cytoskeleton pathway. Oncol Rep. 35:2003–2010. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Jia ZF, Pu PY, Kang CS, Wang GX, Zhang ZY,
Qiu MZ and Huang Q: Influence of SEPT7 on biological characters of
glioma cell line TJ905. Zhonghua Wai Ke Za Zhi. 45:1420–1423.
2007.(In Chinese). PubMed/NCBI
|
|
7
|
Meng X, Zhu Y, Tao L, Zhao S and Qiu S:
Periostin has a protective role in melatonin-induced cell apoptosis
by inhibiting the eIF2α-ATF4 pathway in human osteoblasts. Int J
Mol Med. 41:1003–1012. 2018.PubMed/NCBI
|
|
8
|
Pizarro JG, Yeste-Velasco M, Esparza JL,
Verdaguer E, Pallàs M, Camins A and Folch J: The antiproliferative
activity of melatonin in B65 rat dopaminergic neuroblastoma cells
is related to the downregulation of cell cycle-related genes. J
Pineal Res. 45:8–16. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhang NZ, Liu L, Fan N, Zhang Q, Wang W,
Zheng M, Ma L, Li Y and Shi L: The requirement of SEPT2 and SEPT7
for migration and invasion in human breast cancer via MEK/ERK
activation. Oncotarget. 7:61587–61600. 2016.PubMed/NCBI
|
|
10
|
Zhou J, Lu S, Yang S, Chen H, Shi H, Miao
M and Jiao B: MicroRNA-127 post-transcriptionally downregulates
Sept7 and suppresses cell growth in hepatocellular carcinoma cells.
Cell Physiol Biochem. 33:1537–1546. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Schickel R, Boyerinas B, Park SM and Peter
ME: MicroRNAs: Key players in the immune system, differentiation,
tumorigenesis and cell death. Oncogene. 27:5959–5974. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Iorio MV and Croce CM: MicroRNAs in
cancer: Small molecules with a huge impact. J Clin Oncol.
27:5848–5856. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Stefani G and Slack FJ: Small non-coding
RNAs in animal development. Nat Rev Mol Cell Biol. 9:219–230. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Akao Y, Nakagawa Y and Naoe T:
MicroRNA-143 and-145 in colon cancer. DNA Cell Biol. 26:311–320.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Akao Y, Nakagawa Y and Naoe T:
MicroRNAs-143 and −145 are possible common onco-microRNAs in human
cancers. Oncol Rep. 16:845–850. 2006.PubMed/NCBI
|
|
16
|
Wilusz JE, Sunwoo H and Spector DL: Long
noncoding RNAs: Functional surprises from the RNA world. Genes Dev.
23:1494–1504. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ge X and Gong L: MiR-590-3p suppresses
hepatocellular carcinoma growth by targeting TEAD1. Tumour Biol.
39:10104283176959472017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wu S, Liu W and Zhou L: MiR-590-3p
regulates osteogenic differentiation of human mesenchymal stem
cells by regulating APC gene. Biochem Biophys Res Commun.
478:1582–1587. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chen L, Wang W, Zhu S, Jin X, Wang J, Zhu
J and Zhou Y: MicroRNA-590-3p enhances the radioresistance in
glioblastoma cells by targeting LRIG1. Exp Ther Med. 14:1818–1824.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Huang CX, Lv B and Wang Y: Protein
phosphatase 2a mediates oxidative stress induced apoptosis in
osteoblasts. Mediators Inflamm. 2015:8042602015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Subramaniam M, Jalal SM, Rickard DJ,
Harris SA, Bolander ME and Spelsberg TC: Further characterization
of human fetal osteoblastic hFOB 1.19 and hFOB/ER alpha cells: Bone
formation in vivo and karyotype analysis using multicolor
fluorescent in situ hybridization. J Cell Biochem. 87:9–15. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Reiter RJ, Tan DX and Fuentes-Broto L:
Melatonin: A multi-tasking molecule. Prog Brain Res. 181:127–151.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Akbarzadeh M, Rahbarghazi R, Nabat E,
Movassaghpour AA, Shanehbandi D, Faramarzian Azimi Maragheh B,
Matluobi D, Barazvan B, Kazemi M, Samadi N and Nouri M: The impact
of different extracellular matrices on melatonin effect in
proliferation and stemness properties of ovarian cancer cells.
Biomed Pharmacother. 87:288–295. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Bavithra S, Selvakumar K, Sundareswaran L
and Arunakaran J: Neuroprotective effect of melatonin against PCBs
induced behavioural, molecular and histological changes in cerebral
cortex of adult male wistar rats. Neurochem Res. 42:428–438. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Mostowy S and Cossart P: Septins: The
forth component of the cytoskeleton. Nat Rev Mol Cell Biol.
13:183–194. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kremer BE, Haystead T and Macara IG:
Mammalian septins regulate microtubule stability through
interaction with the micro tubule-binding protein MAP4. Mol Biol
Cell. 16:4648–4659. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wloka C, Nishihama R, Onishi M, Oh Y,
Hanna J, Pringle JR, Krauss M and Bi E: Evidence that a septin
diffusion barrier is dispensable for cytokinesis in budding yeast.
Biol Chem. 392:813–829. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Shehadeh L, Mitsi G, Adi N, Bishopric N
and Papapetropoulos S: Expression of lewy body protein septin 4 in
postmortem brain of Parkinson's disease and contro subjects. Mov
Disord. 24:204–210. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Scott M, Hyland P, McGregor G, Hillan KJ,
Russell SE and Hall PA: Multimodality expression profiling shows
SEPT9 to be overexpressed in a wide range of human tumours.
Oncogene. 24:4688–4700. 2005. View Article : Google Scholar : PubMed/NCBI
|