Introduction
Reactive oxygen species (ROS) are produced normally
by metabolism, inflammation, phagocytosis and other physiological
biological processes. ROS can be harmful when in excess; however,
some levels are required to maintain cellular homeostasis through
redox cell signalling (1). When
the intracellular concentration of free radicals is excessive, they
interact with and cause oxidative damage to proteins, lipids and
DNA (2). Oxidative stress has been
associated with a variety of pathological conditions, including
cancer, diabetes, obesity and neurodegenerative diseases (3,4). In
addition, it occurs frequently in muscle tissue and especially
during intense exercise (5,6).
Furthermore, oxidative stress-induced damage of the vascular
endothelium is one of the most important factors regulating
aetiology of cardiovascular diseases (7,8).
Indeed, oxidative stress in endothelial cells induces acute and
chronic phases of leukocyte adhesion to the endothelium (9). The interaction between ROS and nitric
oxide sets off a vicious cycle, which results in inflammation
(7). Furthermore, ROS, such as
H2O2, can be diffused throughout endothelial
cells and react with cysteine groups of proteins modifying their
function (10). Thus, in an
oxidative stress context, endothelial cells may lose integrity,
progress to senescence and detach into the circulation (11).
Every organism that dwells in the presence of
oxygen, contains a variety of endogenous antioxidant mechanisms
including both enzymes and non-enzymatic metabolites (12). The most significant intracellular
antioxidant compound is reduced glutathione (GSH), a tripeptide
consisting of glutamic acid, glycine and cysteine, with the latter
containing a sulphydryl group responsible for its antioxidant
properties (13). Apart from the
endogenous mechanisms, diet is a frequent supplier of antioxidant
compounds, including polyphenols present in plants (14). Polyphenolic compounds are products
of plant secondary metabolism playing an important role in cellular
functions. When plant foods are consumed, the absorbed polyphenols
may elicit a variety of important bioactivities having beneficial
effects on human health (15,16).
Polyphenolic compounds can also be found in coffee, one of the most
popular beverage throughout the world due to its pleasant taste and
aroma as well as its stimulating effect (17). Coffee annual production exceeds 8
Mt and the average daily consumption is approximately 2.3 billion
cups (18). Traditionally, the
beneficial effects of coffee consumption on human health have been
mainly attributed to caffeine, which is its most widely
investigated ingredient. Nevertheless, there also other components,
such as chlorogenic acids (CGA) that contribute to its valuable
antioxidant properties (19).
Several studies investigated the quantity, and have reported the
potent antioxidant and disease-related beneficial properties of
CGAs (20–22).
However, despite the fact that coffee beans undergo
roasting before their consumption, there is not much evidence on
the effects of roasting on coffee composition, nor the differences
between green and roasted coffee beans regarding their antioxidant
activity (23–25). Indeed, it is known that the
roasting procedure, which differs between coffee varieties, greatly
affects CGAs leading to their breakdown and the formation of
products, which may alter the antioxidant capacity of coffee beans
(23,26,27).
Therefore, the aim of the present study was to examine the effects
of a green and a roasted C. arabica extract on nuclear
factor erythroid 2-related factor 2 (Nrf2)-target gene expression
in two cell lines.
Materials and methods
Coffee beans and roasting
conditions
Both the green and roasted coffee bean varieties
were provided from our collaborators (Coffee Island SA, Athens,
Greece). The roasted beans were roasted for 4 min at 215°C
(corresponding to a light roast).
Preparation of the extracts from
coffee beans
For both the green and roasted beans a 10% w/v
extract in H2O was prepared. The beans were grounded
using mortar and pestle, then a brief sonication treatment was
applied for 20 min (70% amplitude, 0.7 sec cycle) and finally they
were stirred for 20 min in moderate heat (35°C). After
centrifugation (7,000 × g, 10 min, 10°C), each extract was stored
in aliquots and kept at −80°C for further analysis.
Cell culture conditions
The endothelial EA.hy926 cells were donated by
Professor Koukoulis (University of Thessaly, Larissa, Greece),
while the C2C12 muscle cells were donated by Professor Koutsilieris
(National and Kapodistrian University of Athens, Athens, Greece).
The cells were cultured in normal Dulbecco's modified Eagle's
medium (DMEM) containing 10% (v/v) fetal bovine serum (FBS), 2 mM
L-glutamine, 100 U/ml of penicillin and 100 U/ml of streptomycin
(all purchased from Gibco, Paisley, UK) in plastic disposable
tissue culture flasks at 37°C in 5% carbon dioxide.
Treatment of the cells with the coffee
extracts
Cells were seeded in culture flasks and incubated
for 24 h. The medium was then removed and replaced with serum-free
medium containing the coffee extracts at non-cytotoxic
concentrations. The cells were treated with the extracts (or just
with serum-free medium for the control flasks) for 24 h and were
then trypsinised, collected and centrifuged twice (300 × g, 5 min,
5°C). At the end of the first centrifugation, the supernatant fluid
was discarded and the cellular pellet was kept at −80°C until
further analysis.
qPCR of Nrf2 target genes
RNA was extracted from the cell pellet as mentioned
earlier using an RNA isolation kit (PureLink™ RNA kit; Invitrogen;
Thermo Fisher Scientific, Inc., Waltham, MA, USA). RNA was
quantified and its purity was confirmed by measuring the
OD260/280, with a value >1.8 indicating lack of
protein contamination. The extracted RNA (~10 µg) was treated with
DNase (RQ1 RNase-Free DNase, 1 U/µl; Promega Corporation, Madison,
WI, USA). DNA-free RNA was then reverse transcribed to obtain cDNA
(Superscript II Reverse Transcriptase) using oligo (dT) 12–18
primers (both from Invitrogen; Thermo Fisher Scientific, Inc.).
Amplification of cDNAs for the Nrf2 target genes (cat, sod1,
txn, hmox1, nrf2, nqo1, gclc, gsr, gpx1 and gsta2) and
for the gapdh gene was performed in 10 µl reactions
containing SYBR® Select Master mix (2X; Applied
Biosystems; Thermo Fisher Scientific, Inc.), 0.25 µΜ of each
primer, 50 nM ROX Low and 25 ng cDNA for the amplification of all
tested genes. The utilised primers were based on the literature
(Tables I and II). The thermocycling conditions used
for the amplification of the aforementioned genes were: 3 min at
95°C; 45 cycles of 15 sec at 95°C, 30 sec at 55°C for all the genes
in the myoblasts and the majority of genes in the endothelial cells
with the exception of cat, and gsta2 for which 30 sec
at 53°C were used, followed by 30 sec at 72°C. Finally, a melting
curve was carried out from 53 to 95°C to check the specificity of
the products. qPCR was performed on a MX3005P system (Stratagene,
UK). Amplification efficiencies were >87% with r2
values >0.982 for the genes.
 | Table I.Primers for nuclear factor erythroid
2-related factor 2 target genes in myoblasts. |
Table I.
Primers for nuclear factor erythroid
2-related factor 2 target genes in myoblasts.
| Gene | Gene ID | Primer (5′-3′) |
|---|
| cat | 12359 | Forward:
TGAGAAGCCTAAGAACGCAATTC |
|
|
| Reverse:
CCCTTCGCAGCCATGTG |
| sod1 | 20655 | Forward:
GTGATTGGGATTGCGCAGTA |
|
|
| Reverse:
TGGTTTGAGGGTAGCAGATGAGT |
| txn | 22166 | Forward:
CCGCGGGAGACAAGCTT |
|
|
| Reverse:
GGAATGGAAGAAGGGCTTGATC |
| hmox1 | 15368 | Forward:
CACGCATATACCCGCTACCT |
|
|
| Reverse:
CCAGAGTGTTCATTCGAGCA |
| nrf2 | 18024 | Forward:
CGAGATATACGCAGGAGAGGTAAGA |
|
|
| Reverse:
GCTCGACAATGTTCTCCAGCTT |
| nqo1 | 18104 | Forward:
TATCCTTCCGAGTCATCTCTAGCA |
|
|
| Reverse:
TCTGCAGCTTCCAGCTTCTTG |
| gclc | 14629 | Forward:
ATCTGCCAAGGCGGCAAC |
|
|
| Reverse:
ACTCCTCTGCAGCTGGCTC |
| gsr | 14782 | Forward:
GCTATGCAACATTCGCAGATG |
|
|
| Reverse:
AGCGGTAAACTTTTTCCCATTG |
| gpx1 | 14775 | Forward:
GAAGAACTTGGGCCATTTGG |
|
|
| Reverse:
TCTCGCCTGGCTCCTGTTT |
| gsta2 | 14858 | Forward:
CGTCCACCTGCTGGAACTTC |
|
|
| Reverse:
GCCTTCAGCAGAGGGAAAGG |
| gapdh | 14433 | Forward:
AACGACCCCTTCATTGAC |
|
|
| Reverse:
TCCACGACATACTCAGCAC |
 | Table II.Primers for nuclear factor erythroid
2-related factor 2 target genes in endothelial cells. |
Table II.
Primers for nuclear factor erythroid
2-related factor 2 target genes in endothelial cells.
| Gene | Access no. | Primer (5′-3′) |
|---|
| cat |
847 | Forward:
CCAGAAGAAAGCGGTCAAGAA |
|
|
| Reverse:
TGGATGTGGCTCCCGTAGTC |
| sod1 | 6647 | Forward: AGGGCATCA
TCAATTTCGAG |
|
|
| Reverse:
GGGCCTCAGACTACATCCAA |
| txn | 7295 | Forward:
TTTCCATCGGTCCTTACAGC |
|
|
| Reverse:
TTGGCTCCAGAAAATTCACC |
| hmox1 | 3162 | Forward:
GGCCTGGCCTTCTTCACCTT |
|
|
| Reverse:
GAGGGGCTCTGGTCCTTGGT |
| nrf2 | 4780 | Forward:
ATTGCCTGTAAGTCCTGGTCA |
|
|
| Reverse:
ACTGCTCTTTGGACATCATTTCG |
| nqo1 | 1728 | Forward:
GGGCAAGTCCATCCCAACTG |
|
|
| Reverse:
GCAAGTCAGGGAAGCCTGGA |
| gclc | 2729 | Forward:
GAAGAAGATATTTTTCCTGTCATTGAT |
|
|
| Reverse:
CCATTCATGTATTGAAGAGTGAATTT |
| gsr | 2936 | Forward:
CCAGCTTAGGAATAACCAGCGATGG |
|
|
| Reverse:
GTCTTTTTAACCTCCTTGACCTGGGAGAAC |
| gpx1 | 2876 | Forward:
CGCTTCCAGACCATTGACATC |
|
|
| Reverse:
CGAGGTGGTATTTTCTGTAAGATCA |
| gapdh | 2597 | Forward:
TGCACCACCAACTGCTTAG |
|
|
| Reverse:
GATGCAGGGATGATGTTC |
Statistical analysis
Results were analysed by one-way ANOVA followed by
Tukey's test for multiple pair-wise comparisons. All the results
were expressed as mean ± SD. Differences were considered
significant at P<0.05. Data were analysed using SPSS, version
20.0 (SPSS, Inc., Chicago, IL, USA).
Results and Discussion
This is a follow-up study from a previous one where
nine coffee extracts were analyzed regarding their composition and
effect on cellular redox status (28). According to the results, three
mono-caffeoylquinic acid isomers (CQAs), namely 3-, 4- and 5-CQA
were identified as the main polyphenolic compounds of the extracts,
accounting for up to 30.23 mg/g of coffee with 3-CQA being the most
abundant. Furthermore, upon administration to myoblasts and
endothelial cells, differential toxicity was observed since green
coffee extracts demonstrated higher toxicity towards the myoblasts,
while in the endothelial cell line the opposite was observed.
Nevertheless, all the extracts resulted in increased reduced GSH
levels, an important endogenous antioxidant tripeptide due to its
reactive sulfhydryl group (29).
Coffee affected GSH levels differently in the two cell lines as in
the myoblasts GSH was increased up to approximately 70%, while in
the endothelial cells it was increased up to approximately 30%. An
interesting observation was that GSH reached a peak concentration
at an intermediate coffee extract concentration, while at higher
extract concentration GSH was decreased. This finding could be
probably attributed to the fact that polyphenols, depending on
their concentration, induce a shift of their antioxidant activity
towards prooxidant effects (30).
Endogenous ROS levels were not affected by the tested extracts,
while lipid and protein oxidation levels were slightly reduced,
especially in the myoblasts (26).
In the current study, two extracts were selected for
further analysis in order to shed light on coffee's mechanism of
action. According to the literature, coffee constituents are able
to cause Nrf2 derepression (i.e., translocation to the nucleus
following detachment from the cytosol-localized Keap1) and
concomitantly antioxidant gene expression (31–33).
Therefore, the lightly (i.e., for 4 min) roasted C. arabica
coffee extract was selected since it has previously demonstrated
the highest antioxidant potency in various assays (34). In addition, its corresponding green
extract was also examined due to its peculiar toxicity pattern
(i.e., 320-fold more toxic in the myoblasts compared with the
roasted one).
Effect of coffee on Nrf2-target gene expression. To
test the hypothesis whether Nrf2 derepression is a potential
molecular mechanism of extract action, the green and roasted
extracts were administered in the two cell lines at concentrations
that caused the maximum increase in GSH levels and sequentially
were assessed for their effect on gene expression levels of Nrf2
target genes by qPCR. These genes encode for catalase, superoxide
dismutase 1, thioredoxin, heme oxygenase 1, Nrf2, NAD(P)H quinone
dehydrogenase 1, γ-glutamyl cysteine ligase catalytic subunit,
glutathione reductase, glutathione peroxidase 1 and glutathione
S-transferase α 2. The expression of all these genes is driven by
an antioxidant response element (ARE) in their promoter region,
recognized by the nuclear localised Nrf2. These proteins are a part
of the complex antioxidant system that protects cells from
oxidative damage by neutralising free radicals and oxidising
agents, therefore extracts or compounds that upregulate their
expression may be used as potential antioxidant supplements. In a
previous study, the roasted extract was administered in myoblasts
and a microarray analysis was conducted, indicating that coffee
constituents may act through the Nrf2 pathway (35). As for the used extract
concentrations, the roasted extract was administered at 400 µg/ml
and the green at 2.5 µg/ml in the myoblasts, while both extracts
were administered at 100 µg/ml in the endothelial cells.
According to the qPCR results, in the two cell lines
the roasted extract had a more profound effect on gene expression,
upregulating most genes compared to the green extract.
Specifically, in the myoblasts the roasted extract caused the
upregulation of nqo1 3.1-, 5.4- and 4.9-fold at 3, 12 and 24
h, respectively (Fig. 1),
gclc expression was increased 3.3- and 3.4-fold at 3 and 24
h, respectively, gpx1 was upregulated 2.5-, 2.8- and
5.1-fold at 3, 12 and 24 h, respectively, while gsta2
increased mRNA levels by 2.7-, 3.0- and 8.0-fold at 3, 12 and 24 h,
respectively. On the other hand, sod1 levels were decreased
by 78%. No significant differences were observed for cat, txn,
hmox1, nrf2 and gsr.
The C. arabica green extract caused the
downregulation of six genes at various time points without
upregulating any of the genes (Fig.
1). More specifically, sod1 was downregulated by 71% at
24 h, hmox1 was less abundant by 65% at 3 h, nrf2
mRNA was 71% less compared to the control at 12 h, gsr by 82
and 62% at 12 and 24 h, respectively, gpx1 by 80% at 3 h and
gsta2 by 67 and 91% at 3 and 24 h, respectively. No
statistically significant changes were observed for cat, txn,
nqo1 and gclc.
In the flow cytometric analysis of the previous
study, both the roasted and the green coffee extracts caused a
similar increase in GSH levels (i.e., 70 and 66%, respectively) at
the concentrations used in the qPCR. However, the latter analysis
showed that only the roasted extract upregulated the expression of
Nrf2 target genes and especially those that partake in GSH
metabolism (i.e., gclc, gpx1 and gsta2). The
increased abundance of these mRNAs, if translated into increased
protein levels, may justify the increased GSH levels. In addition,
the upregulation of nqo1, which encodes for an important
antioxidant enzyme that is involved in quinone detoxification, has
been displayed in previous studies with coffee (32). Therefore, it can be hypothesised
that the roasted extract causes the derepression of Nrf2. This can
be attributed to coffee polyphenols such as the 5-caffeoylquinic
acid that has been shown to stimulate Nrf2 gene expression
previously, through oxidation of KEAP1 cysteine residues (e.g.,
Cys151) that result in the release of Nrf2, which subsequently
enters the nucleus and induces gene expression (36). These results are in agreement with
those of another study in which the same extract was administered
in the same myoblast cell line at an equal concentration and was
subjected to microarray analysis to examine alterations in gene
expression levels (35).
By contrast, the green coffee extract resulted in
reduced gene expression for some antioxidant enzyme-coding genes.
This finding, to the best of our knowledge, has not been previously
reported in the literature. Despite the fact that the green extract
at this concentration (i.e., 2.5 µg/ml) resulted in increased GSH
levels, this finding cannot be attributed to increased mRNA
abundance of GSH metabolism-related genes. However, it is known
that polyphenols exert antioxidant activity through a direct free
radical scavenging capacity (37).
Therefore, it is possible that this scavenging activity of the
green coffee extract polyphenols, ‘spares’ endogenous GSH levels
leading to the observed increase. These results, in combination
with the high cytotoxicity of the green extract towards myoblasts
(at just 20 µg/ml) indicate that the commercial green coffee
extracts should be revisited for potential controversial
effects.
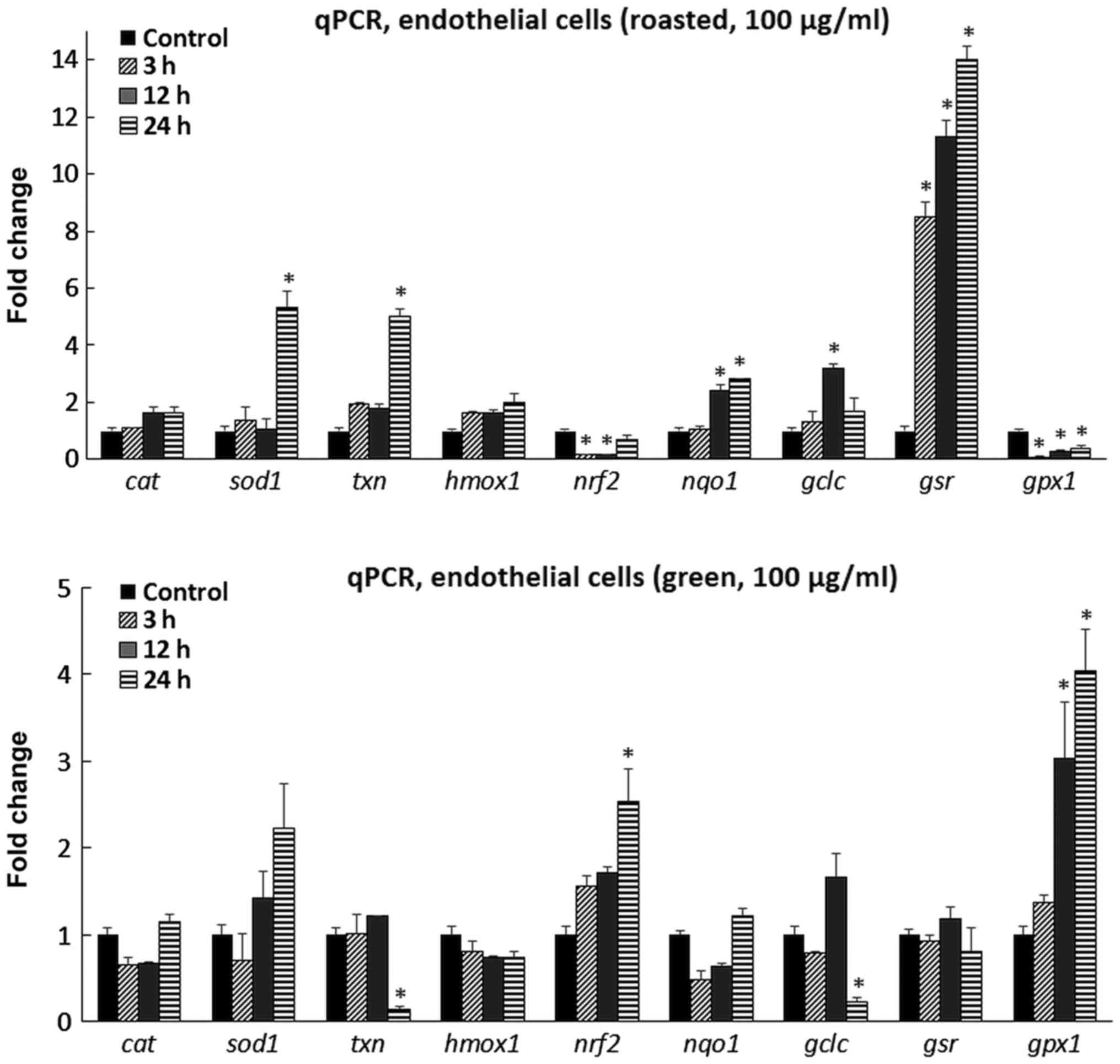
In the endothelial cells, the roasted extract caused
the increased abundance of sod1 mRNA 5.3-fold at 24 h
(Fig. 2). In addition, txn
was also upregulated 5.0-fold at 24 h, while nqo1 mRNA
levels were increased by 2.4- and 2.82-fold at 12 and 24 h,
respectively. The expression of the gclc gene was increased
by 3.2-fold at 12 h, while gsr abundance was increased by
8.5-, 11.3- and 14.0-fold at 3, 12 and 24 h, respectively. On the
other hand, nrf2 mRNA levels were decreased by 84% at 12 h
and 85% at 24 h compared with the control, while gpx1 was
downregulated by 92% at 3 h, 75% at 12 h and 64% at 24 h. No
significant alterations were observed for cat and
hmox1, although a rising trend was evident, particularly, at
24 h. The roasted extract exhibited a distinct profile of affecting
the tested genes in the myoblasts versus the endothelial cells. As
for the green extract, it caused a 2.5-fold increase in nrf2
mRNA levels at 24 h and a 3.0- and 4.0-fold increase for
gpx1 at 12 and 24 h, respectively (Fig. 2). By contrast, lower mRNA levels
were observed for txn by 86% at 24 h and gclc by 77%
at 24 h. No significant changes were evident for the other examined
genes.
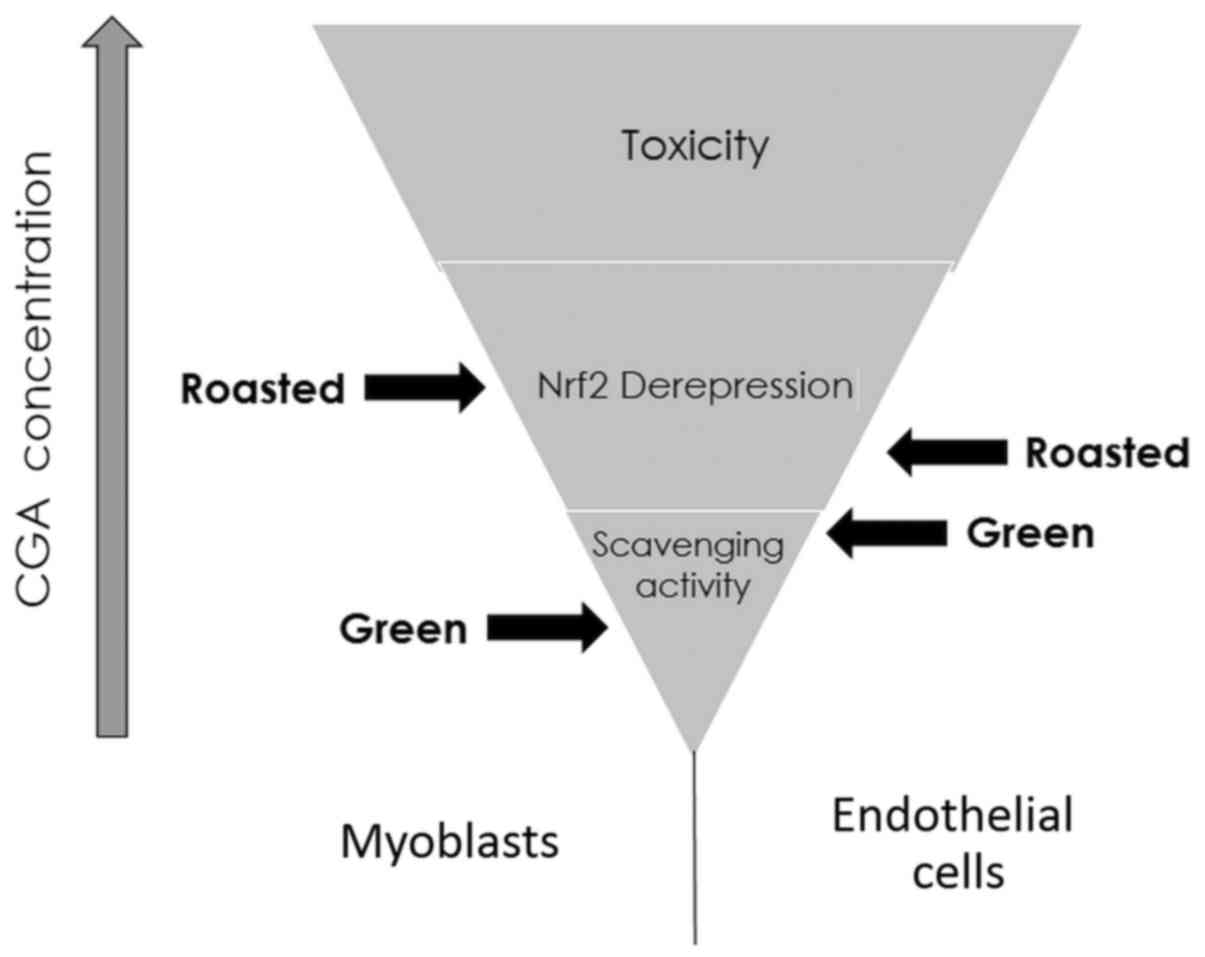
Taking into consideration the qPCR data along with
the dose-dependent, hormetic effect of polyphenols (i.e., direct
scavenging/antioxidant at low concentrations - pro-oxidant activity
at higher doses, potentially leading to Nrf2 derepression - and
toxic past a concentration threshold) (38), a potential mechanism of action for
the aforementioned green and roasted extracts can be deduced for
both cell lines (Fig. 3).
According to the data obtained from the current and previous
studies, it can be assumed that moderate consumption (e.g., 2–4
cups per day) of lightly roasted coffee (as higher amounts may
exhibit tissue-specific cytotoxicity) could elicit potentially
beneficial effects through the expression of Nrf2 target
genes. In order to validate these in vitro results, an in
vivo study is required so that the effects of bioavailability
and metabolism on the potential bioactive compounds will also be
taken into consideration.
Τo conclude, the two coffee extracts differentially
affected gene expression in the two tested cell lines. On the one
hand, the roasted extract (at 400 µg/ml) caused the upregulation of
four genes (and especially those involved in GSH metabolism) and
downregulation in one, while the green extract (at 2.5 µg/ml)
resulted in the downregulation of six genes in the myoblasts. On
the other hand, with respect to the endothelial cells, both
extracts were administered at the same concentration (i.e, 100
µg/ml) as in that particular concentration maximum GSH levels were
exhibited by flow cytometry. Again, the roasted extract had a more
profound effect, as five genes were upregulated, including
gclc and gsr that may explain the increased GSH
levels and two were downregulated, including gpx1 that could
potentially lead to reduced GSH depletion as it utilises this
tripeptide as a substrate. The green coffee extract caused an
upregulation of only two genes whereas, it downregulated another
two. These results highlight the complexity of coffee's molecular
mechanism of action, which could partly be explained by the
intricate regulation of the antioxidant mechanisms and the
interplay between them. Specifically, when some antioxidant
mechanisms are activated, some others remain inactive as a
compensation adaptive cell response (39).
Acknowledgements
Not applicable.
Funding
AP received funding by the Hellenic General
Secretariat for Research and Technology (GSRT) and the Hellenic
Foundation for Research and Innovation (HFRI). This study was
funded by a grant (no. 5042; ‘Assessment of antioxidant and
anticarcinogenic activity of green and roasted coffee varieties’)
from Coffee Island SA to DK.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
AP and ASV wrote the manuscript, AP and AEAT
conducted the assays. DS collaborated with DK in reviewing the
experimental design and the discussion. DK supervised the study
while all authors reviewed the manuscript.
Ethics approval and consent to
participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
D.A. Spandidos is the Editor-in-Chief for the
journal, but had no personal involvement in the reviewing process,
or any influence in terms of adjudicating on the final decision,
for this article.
Glossary
Abbreviations
Abbreviations:
|
ROS
|
reactive oxygen species
|
|
GSH
|
reduced form of glutathione
|
|
TPC
|
total polyphenolic content
|
|
Nrf2
|
nuclear factor erythroid 2-related
factor 2
|
References
|
1
|
Ray PD, Huang BW and Tsuji Y: Reactive
oxygen species (ROS) homeostasis and redox regulation in cellular
signaling. Cell Signal. 24:981–990. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Halliwell B: Free radicals and other
reactive species in diseaseeLS. John Wiley & Sons, Ltd.;
Hoboken, NJ: 2001
|
|
3
|
Sosa V, Moliné T, Somoza R, Paciucci R,
Kondoh H and LLeonart ME: Oxidative stress and cancer: An overview.
Ageing Res Rev. 12:376–390. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rochette L, Zeller M, Cottin Y and Vergely
C: Diabetes, oxidative stress and therapeutic strategies. Biochim
Biophys Acta. 1840:2709–2729. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Veskoukis AS, Goutianos G, Paschalis V,
Margaritelis NV, Tzioura A, Dipla K, Zafeiridis A, Vrabas IS,
Kyparos A and Nikolaidis MG: The rat closely mimics oxidative
stress and inflammation in humans after exercise but not after
exercise combined with vitamin C administration. Eur J Appl
Physiol. 116:791–804. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Spanidis Y, Goutzourelas N, Stagos D,
Mpesios A, Priftis A, Bar-Or D, Spandidos DA, Tsatsakis AM, Leon G
and Kouretas D: Variations in oxidative stress markers in elite
basketball players at the beginning and end of a season. Exp Ther
Med. 11:147–153. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Deanfield JE, Halcox JP and Rabelink TJ:
Endothelial function and dysfunction: Testing and clinical
relevance. Circulation. 115:1285–1295. 2007.PubMed/NCBI
|
|
8
|
Victor VM, Rocha M, Solá E, Bañuls C,
Garcia-Malpartida K and Hernández-Mijares A: Oxidative stress,
endothelial dysfunction and atherosclerosis. Curr Pharm Des.
15:2988–3002. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kokura S, Wolf RE, Yoshikawa T, Granger DN
and Aw TY: Molecular mechanisms of neutrophil-endothelial cell
adhesion induced by redox imbalance. Circ Res. 84:516–524. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Rhee SG: Cell signaling. H2O2, a necessary
evil for cell signaling. Science. 312:1882–1883. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Woywodt A, Bahlmann FH, De Groot K, Haller
H and Haubitz M: Circulating endothelial cells: Life, death,
detachment and repair of the endothelial cell layer. Nephrol Dial
Transplant. 17:1728–1730. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Birben E, Sahiner UM, Sackesen C, Erzurum
S and Kalayci O: Oxidative stress and antioxidant defense. World
Allergy Organ J. 5:9–19. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Pastore A, Federici G, Bertini E and
Piemonte F: Analysis of glutathione: Implication in redox and
detoxification. Clin Chim Acta. 333:19–39. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Landete JM: Dietary intake of natural
antioxidants: Vitamins and polyphenols. Crit Rev Food Sci Nutr.
53:706–721. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Pandey KB and Rizvi SI: Plant polyphenols
as dietary antioxidants in human health and disease. Oxid Med Cell
Longev. 2:270–278. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Goutzourelas N, Stagos D, Spanidis Y,
Liosi M, Apostolou A, Priftis A, Haroutounian S, Spandidos DA,
Tsatsakis AM and Kouretas D: Polyphenolic composition of grape stem
extracts affects antioxidant activity in endothelial and muscle
cells. Mol Med Rep. 12:5846–5856. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Heckman MA, Weil J and de Mejia Gonzalez
E: Caffeine (1, 3, 7-trimethylxanthine) in foods: A comprehensive
review on consumption, functionality, safety, and regulatory
matters. J Food Sci. 75:R77–R87. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Higdon JV and Frei B: Coffee and health: A
review of recent human research. Crit Rev Food Sci Nutr.
46:101–123. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Murthy PS and Naidu MM: Recovery of
phenolic antioxidants and functional compounds from coffee industry
by-productsFood and Bioprocess Technology. 5. Springer; Berlin: pp.
897–903. 2010, View Article : Google Scholar
|
|
20
|
Henry-Vitrac C, Ibarra A, Roller M,
Mérillon JM and Vitrac X: Contribution of chlorogenic acids to the
inhibition of human hepatic glucose-6-phosphatase activity in vitro
by Svetol, a standardized decaffeinated green coffee extract. J
Agric Food Chem. 58:4141–4144. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Park JB: Isolation and quantification of
major chlorogenic acids in three major instant coffee brands and
their potential effects on H2O2-induced
mitochondrial membrane depolarization and apoptosis in PC-12 cells.
Food Funct. 4:1632–1638. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Xu J-G, Hu Q-P and Liu Y: Antioxidant and
DNA-protective activities of chlorogenic acid isomers. J Agric Food
Chem. 60:11625–11630. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Jaiswal R, Matei MF, Golon A, Witt M and
Kuhnert N: Understanding the fate of chlorogenic acids in coffee
roasting using mass spectrometry based targeted and non-targeted
analytical strategies. Food Funct. 3:976–984. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Daglia M, Racchi M, Papetti A, Lanni C,
Govoni S and Gazzani G: In vitro and ex vivo antihydroxyl radical
activity of green and roasted coffee. J Agric Food Chem.
52:1700–1704. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Priftis A, Papikinos K, Koukoulanaki M,
Kerasioti E, Stagos D, Konstantinopoulos K, Spandidos DA,
Kermenidou M, Karakitsios S, Sarigiannis D, et al: Development of
an assay to assess genotoxicity by particulate matter extract. Mol
Med Rep. 15:1738–1746. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kamiyama M, Moon J-K, Jang HW and
Shibamoto T: Role of degradation products of chlorogenic acid in
the antioxidant activity of roasted coffee. J Agric Food Chem.
63:1996–2005. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Gawlik-Dziki U, Świeca M, Dziki D,
Kowalska I, Pecio Ł, Durak A and Sęczyk Ł: Lipoxygenase inhibitors
and antioxidants from green coffee - Mechanism of action in the
light of potential bioaccessibility. Food Res Int. 61:48–55. 2014.
View Article : Google Scholar
|
|
28
|
Priftis A, Panagiotou EM, Lakis K, Plika
C, Halabalaki M, Ntasi G, Veskoukis AS, Stagos D, Skaltsounis LA
and Kouretas D: Roasted and green coffee extracts show antioxidant
and cytotoxic activity in myoblast and endothelial cell lines in a
cell specific manner. Food Chem Toxicol. 114:119–127. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Aquilano K, Baldelli S and Ciriolo MR:
Glutathione: New roles in redox signaling for an old antioxidant.
Front Pharmacol. 5:1962014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Sakihama Y, Cohen MF, Grace SC and
Yamasaki H: Plant phenolic antioxidant and prooxidant activities:
Phenolics-induced oxidative damage mediated by metals in plants.
Toxicology. 177:67–80. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Volz N, Boettler U, Winkler S, Teller N,
Schwarz C, Bakuradze T, Eisenbrand G, Haupt L, Griffiths LR,
Stiebitz H, et al: Effect of coffee combining green coffee bean
constituents with typical roasting products on the Nrf2/ARE pathway
in vitro and in vivo. J Agric Food Chem. 60:9631–9641. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Higgins LG, Cavin C, Itoh K, Yamamoto M
and Hayes JD: Induction of cancer chemopreventive enzymes by coffee
is mediated by transcription factor Nrf2. Evidence that the
coffee-specific diterpenes cafestol and kahweol confer protection
against acrolein. Toxicol Appl Pharmacol. 226:328–337. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Boettler U, Sommerfeld K, Volz N, Pahlke
G, Teller N, Somoza V, Lang R, Hofmann T and Marko D: Coffee
constituents as modulators of Nrf2 nuclear translocation and ARE
(EpRE)-dependent gene expression. J Nutr Biochem. 22:426–440. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Priftis A, Stagos D, Konstantinopoulos K,
Tsitsimpikou C, Spandidos DA, Tsatsakis AM, Tzatzarakis MN and
Kouretas D: Comparison of antioxidant activity between green and
roasted coffee beans using molecular methods. Mol Med Rep.
12:7293–7302. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Priftis A, Goutzourelas N, Halabalaki M,
Ntasi G, Stagos D, Amoutzias GD, Skaltsounis LA and Kouretas D:
Effect of polyphenols from coffee and grape on gene expression in
myoblasts. Mech Ageing Dev. (In press). PubMed/NCBI
|
|
36
|
Liang N and Kitts DD: Role of chlorogenic
acids in controlling oxidative and inflammatory stress conditions.
Nutrients. 8:1–20. 2015. View Article : Google Scholar :
|
|
37
|
Sirota R, Gibson D and Kohen R: The role
of the catecholic and the electrophilic moieties of caffeic acid in
Nrf2/Keap1 pathway activation in ovarian carcinoma cell lines.
Redox Biol. 4:48–59. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Houghton CA, Fassett RG and Coombes JS:
Sulforaphane and other nutrigenomic Nrf2 activators: Can the
clinician's expectation be matched by the reality? Oxid Med Cell
Longev. 2016:78571862016. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Alvarez S and Boveris A: Antioxidant
adaptive response of human mononuclear cells to UV-B: Effect of
lipoic acid. J Photochem Photobiol B. 55:113–119. 2000. View Article : Google Scholar : PubMed/NCBI
|