Introduction
Acute lymphoblastic leukemia (ALL), with a high
invasion, is a malignant heterogeneous disease occurring in
lymphoid hematopoietic cells (1).
The etiology and pathogenesis of ALL remain to be investigated.
There is a great heterogeneity between the treatment and the
prognosis of ALL. ALL often occurs in children, accounting for
70–80% of children with acute leukemia (2). Patients with ALL usually have a poor
prognosis due to variable pathogenesis, complex etiology, severe
clinical symptoms and complex treatment plans, which may place a
burden on children's physical and mental health. With the
progression of clinical treatment methods, the therapeutic effect
of ALL treatment has increased. For children, the five-year overall
survival rate has reached 92.3%, while the rate was only ~10% in
the 1960s; the five-year disease-free survival (DFS) rate has also
increased to 80–85%. In addition, the total remission rate of
simple chemotherapy in children is >90%, and the DFS is ≤70–80%
(3,4). In the 1990s, the five-year survival
rate of children with ALL in developed countries was reported to be
70–80%, and the overall cure rate reached 80%. The cure rate of
children with leukemia is predicted to be >90% in China in the
near future with the development of medical technology (5). The incidence rate of ALL in adults is
lower compared with children, but adults with ALL exhibit poor
effects of treatment, ≤40% of relapse rate and only 30–40% DFS
(6). For adults, conventional
chemotherapy serves a poor effect; ~70–80% patients may not survive
for a long period or be cured, requiring long-term treatment
(7).
The primary clinical treatment method for ALL is
combination chemotherapy. Targeted therapy and hematopoietic stem
cell transplantation are also methods of treatment. In addition to
chemotherapy, hematopoietic stem cell transplantation is another
important method of treatment for patients with ALL. Simple
high-dose chemotherapy is not able to serve a myeloablative effect.
Due to the limitations of chemotherapeutic drugs, it is difficult
to eliminate tumor cells detected in sheltered areas by
chemotherapy, leading to tumor cell recurrence (6). Chemotherapy combined with
radiotherapy has become an important method of pretreatment prior
to hematopoietic stem cell transplantation with developing
radiotherapy technology. It is clinically confirmed that the
application of total body irradiation (TBI) may improve the success
rate of hematopoietic stem cell transplantation and decrease the
rate of recurrence (8).
TBI is an important method of pretreatment prior to
hematopoietic stem cell transplantation (9,10).
The effective pretreatment method of hematopoietic stem cell
transplantation is large-dose chemotherapy combined with TBI, which
prompts successful transplantation (11). The Northern European Bone Marrow
Transplant Organization, via relevant clinical trials, have
demonstrated that chemotherapy combined with TBI may exhibit an
increased effect of pretreatment compared with simple chemotherapy
for children or adults with acute leukemia, and that chemotherapy
combined with TBI may provide improved survival conditions for
patients (12–15). The three-year overall survival rate
of patients treated with chemotherapy combined with TBI following
transplantation is 76%, which is higher than 62% simple
chemotherapy, and the patients have a long DFS and a low recurrence
rate (12–15). As of the 20th century, chemotherapy
combined with TBI has demonstrated improved effects for children or
adults with leukemia compared with simple chemotherapy (16–18).
Linsenmeier et al (19)
indicated that TBI was a safe and effective pretreatment method
prior to hematopoietic stem cell transplantation; TBI prior to
high-dose chemotherapy may maintain the same effects of
pretreatment and effectively reduce notable untoward effects or
discomfort, the preparation workload of radiotherapy and the
untoward effects of radiation (19). Bhatia et al (20,21)
revealed that TBI had a preventive effect on the late mortality for
transplantation patients and that patients treated with TBI had
lower rates of late mortality by ≥50% compared with patients
without TBI.
However, with the increase and development of
clinical application, limitations of TBI have been reported.
Clinical experience has demonstrated that it is inadvisable to rely
solely on increasing doses of TBI, which can lead to an increase of
the dose to organs at risk including the lungs, livers and kidneys,
enhancing the incidence of complications, and an unimproved overall
survival rate for patients with ALL, therefore, a more targeted TBI
should be developed to lessen toxic reactions while increasing
doses (22). The application of
total marrow and lymphatic irradiation (TMLI) to the whole-body
skeleton, main lymph node chain and the spleen may evoke a
sufficient immunosuppression response to the transplant (23). Studies have revealed that patients
treated with TMLI and TBI have an equivalent extramedullary relapse
rate following hematopoietic stem cell transplantation; however,
TMLI is a more conservative targeted radiotherapy and does not
increase the risk of extramedullary relapse (23–27).
TMLI may reduce toxic reactions while increasing doses to target
volumes, which provide the possibility of treatment to the elderly
and patients with complications who poorly suit to conventional TBI
(28,29). Tomotherapy accelerators have
gradually replaced the intensity-modulated radiotherapy (IMRT) of
the conventional accelerator.
By comparing the target volume, clinical reaction
and effects of transplantation among helical tomotherapy (HT)-TBI,
HT-TMLI, IMRT-TBI and IMRT-TMLI, the present study evaluated the
advantages and disadvantages of these methods of treatment and
analyzed the possibility of replacing TBI with TMLI in clinical
treatment, which may provide a theoretical basis to clinical
treatment.
Materials and methods
Patients
A total of 18 patients with ALL treated with total
body irradiation prior to hematopoietic stem cell transplantation
in the First Affiliated Hospital of Zhengzhou University
(Zhengzhou, China) from May 2016 to December 2016 were selected. In
the present study, 10 males and eight females, with an age range of
4–34 years old were enrolled. The Karnofsky performance status was
>100 (30). According to
pathological anatomy of the 18 patients, there were nine patients
with T-cell acute lymphoblastic leukemia and nine patients with
B-cell acute lymphoblastic leukemia. Prior to radiotherapy, all
patients and their families were notified of the requirement of
treatment and possible untoward effects, and signed written
informed consent was obtained. All patients prior to radiotherapy
underwent systemic high-dose chemotherapy plus intrathecal
injection. Following radiotherapy, patients entered into the
isolation room on the same day; after 1- to 2-day preparation,
myeloablative transplantation therapy was performed. The inclusion
criteria were as follows: i) Definite pathological diagnosis; ii)
systemic chemotherapy plus intrathecal injection; and iii) normal
heart, liver and kidney function and Karnofsky performance status
≥80. The exclusion criteria were as follows: i) severe damage of
heart, liver, kidney and lung function; ii) severe or uncontrolled
infection; and iii) patients with a history of severe mental
disorder that are not compliant to treatment. The present study was
approved by the Ethics Committee of the First Affiliated Hospital
of Zhengzhou University.
Computed tomography (CT) localization,
scan and image registration
A thermoplastic mask and vacuum pad were provided to
patients, according to the CT localization requirement of
radiotherapy, and the lead points were affixed at three points. For
patients with the height of >120 cm, there were three lead
points, each on two sets of images, respectively located 5–10 cm
above the navel and 5–10 cm below the knee. A continuous flat scan
was performed with Definition AS large aperture CT analog
positioning machine (Siemens AG, Munich, Germany). CT images of the
phase during normal respiration were acquired. As the maximum
length of the tomotherapy machine was 160 cm, a segmented scan was
performed for patients with a height >120 cm.
Delineating the clinical target volume
(CTV) in CT location images (segmented delineation for patients
with a height >120 cm)
CTV of TBI constituting the whole body except the
organs at risk, usually with 3 mm of skin adduction as CTV; CTV of
TMLI constituted the whole body bone marrow (sternum, vertebra,
rib, to delineate conveniently target volume), main lymph node
chains (neck and mediastinal lymph nodes) and the spleen.
Considering the reproduction of patients, and the intentions and
chemotherapy plans of the patient and their families in the
clinical treatment, the testes were not irradiated. Planning target
volume (PTV) was delineated by externally extending 5 mm from every
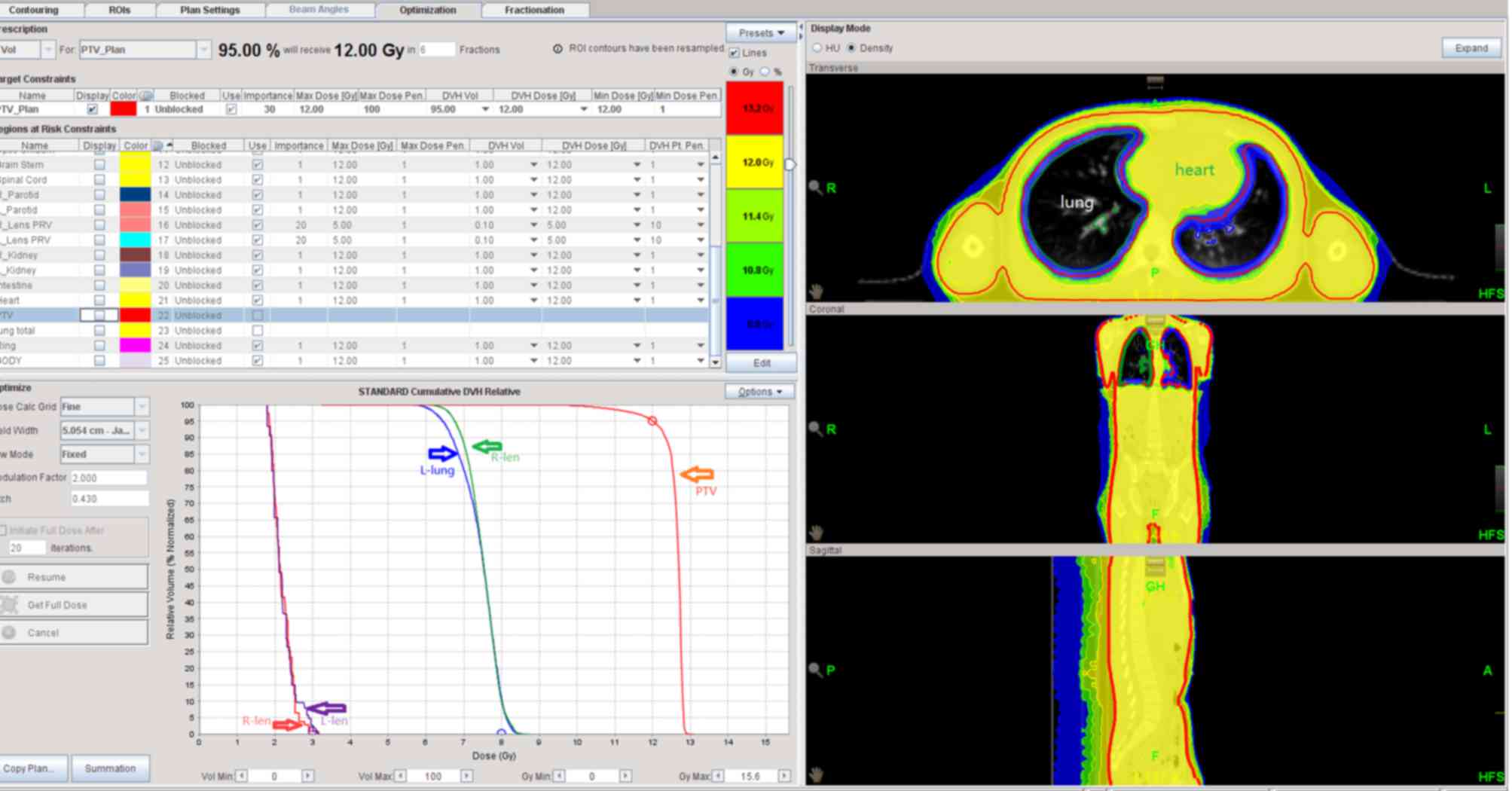
direction of CTV. Organs at risk included bilateral crystalline
lens and bilateral lungs (Fig.
1).
Prescription dose setting for target
volume, organs at risk and PTV
2 Gy were administered each time, two times a day,
with an interval of 6–8 h and 6 total doses of 12 Gy.
Dose constraint for important organs
at risk
According to the requirement of the United States
Radiation Therapy Oncology Group (31); dose constraint in the main normal
tissues is presented in Table
I.
 | Table I.Radiotherapy dose constraint in the
normal tissues. |
Table I.
Radiotherapy dose constraint in the
normal tissues.
| Organ at risk | Dose
constraint | PRV |
|---|
| Lung | Mean dose <8
Gy |
|
| Left Lens | Max ≤9 Gy | PRV external
expansion |
|
|
| 3 mm, Max <9
Gy |
| Right Lens | Max ≤9 Gy | PRV external
expansion |
|
|
| 3 mm, Max <9
Gy |
Radiotherapy planning
CT images of the target volume and organs at risk
were transferred to the Tomotherapy and Pinnacle V9.8.6008
treatment planning system and the same physicist conducted the
radiotherapy plans according to the requirements of the clinician.
IMRT of the conventional accelerator was conducted using a Primus
linear accelerator with 6 megavoltage (MV) X rays and 3–7 radiation
fields. HT was implemented with 6 MV X rays; most lung tissues on
one side or on dorsal and ventral sides avoided exposure to
radiation.
Patients 1–10 were treated with HT plans and
assigned to groups randomly; patients 1–6 target volume delineation
was via HT-TBI, and patients 7–10 via HT-TMLI. Patients 11–18 were
treated with IMRT plans; patients 11–14 target volume delineation
was via IMRT-TBI, and patients 15–18 via IMRT-TMLI. The associated
indexes of target volume and organs at risk in different
radiotherapy plans were recorded and compared.
Dosimetry evaluation indexes
According to the dose volume histogram (DVH) of each
plan, the dosimetry evaluation indexes in target volume and organs
at risk included: Dmax, maximum dose to target volume or
organs at risk (cGy); Dmean, average dose to target
volume or organs at risk (cGy); D98, dose to 98% of
target volume or organs at risk (cGy); D95, dose to 95%
of target volume or organs at risk (cGy); D50, dose to
50% of target volume or organs at risk (cGy); D02, dose
to 2% of target volume or organs at risk (cGy) and V12,
volume ratio of target volume occupied by 12 Gy.
Dose to double lungs prior to hematopoietic stem
cell transplantation revealed close associations with the
complications following transplantation. Evaluation indexes for
double lungs included Dmax and Dmean;
however, considering the specificity of TBI in target volume, the
average dose to the lung should be lower than 8 Gy when evaluating
the dose to the lung during the treatment.
Evaluation indexes for the crystalline lens included
Dmax and Dmean. In addition, conformity index
(CI) and homogeneity index (HI) of target volume in HI and IMRT
plans were compared.
Clinical untoward effects and
transplantation effects
The main untoward effects occurred during the
treatment, including nausea, emesis, inappetence, xerostomia,
fever, diarrhea and rash, among of which nausea, emesis,
inappetence and xerostomia were the most common reactions. All
reactions were at I–II level (32). Symptomatic treatment was performed,
according to the patients' specific condition. Patients suffering
from fever, received treatment; physical cooling for patients with
low fever and aspirin-DL-lysine for patients with a body
temperature >38.5°C.
Transplantation effects were assessed by whether the
18 patients had successful transplantation or not. The number of
survivors and those who had succumbed to mortality, and the cause
of mortality during long-term follow-up was recorded.
Statistical analysis
The statistical software SPSS (version 17.0; SPSS,
Inc., Chicago, IL, USA) was employed to analyze the data in the
present study. All data were presented as the mean ± standard
deviation. Provided that the difference between the groups followed
the normal distribution, the data were analyzed by a Student's
t-test. If the difference between groups did not follow the normal
distribution, the data were analyzed by a Wilcoxon rank-sum test.
Multiple comparisons were analyzed by using one-way analysis of
variance with the post hoc Student-Newman-Keuls test. P<0.05 was
considered to indicate a significant difference.
Results
Radiotherapy plans comparison
Basic clinical data of the 18 patients were
presented in Table II. Patients
1–6 target volume delineation was according to HT-TBI and patients
7–10 via HT-TMLI. Patients 11–14 target volume delineation was
according to IMRT-TBI and patients 15–18 via IMRT-TMLI.
 | Table II.Basic data of patients. |
Table II.
Basic data of patients.
| Patient number | Sex | Age | Pathological
type | Complications |
|---|
| 1 | Female | 7 | B-cell ALL | No |
| 2 | Male | 7 | B-cell ALL | No |
| 3 | Male | 13 | T-cell ALL | No |
| 4 | Female | 12 | B-cell ALL | No |
| 5 | Male | 10 | T-cell ALL | No |
| 6 | Male | 9 | T-cell ALL | No |
| 7 | Female | 19 | T-cell ALL | No |
| 8 | Female | 34 | B-cell ALL | No |
| 9 | Male | 12 | B-cell ALL | No |
| 10 | Male | 6 | T-cell ALL | No |
| 11 | Female | 8 | T-cell ALL | No |
| 12 | Female | 13 | B-cell ALL | No |
| 13 | Male | 16 | T-cell ALL | No |
| 14 | Male | 21 | B-cell ALL | No |
| 15 | Male | 18 | B-cell ALL | No |
| 16 | Female | 10 | T-cell ALL | No |
| 17 | Male | 11 | B-cell ALL | No |
| 18 | Female | 7 | T-cell ALL | No |
Dose distribution in the target volume of HT and
IMRT radiotherapy plans met dose requirements. CI in HT plans was
significantly higher compared with IMRT plans (P<0.05).
Additionally, there was no significant difference in other indexes
(P>0.05). The target volume of HT plans revealed a more
homogeneous dose distribution and increased conformity compared
with IMRT plans with the conventional accelerator.
As presented in Table
III, D95 of PTV in each radiotherapy plan was
approximately equal to the prescribed dose of 12 Gy. The target
volume of TBI was larger, which met the requirement of radiotherapy
plans. There were no significant differences in Dmax,
D98, D02 and Dmean among the four
radiotherapy plans (P>0.05).
 | Table III.The dose distribution in PTV between
HT and IMRT plans (cGy). |
Table III.
The dose distribution in PTV between
HT and IMRT plans (cGy).
| A, TBI |
|---|
|
|---|
| Variable | HT | IMRT | t-test | P-value |
|---|
|
Dmax | 12.250±0.606 | 12.050±0.592 | 0.35 | 0.75 |
| D98 | 11.128±0.176 | 11.124±0.181 | 0.08 | 0.95 |
| D95 | 11.940±0.689 | 11.923±0.653 | 0.32 | 0.73 |
|
Dmean | 12.453±0.092 | 12.782±0.085 | −0.32 | 0.77 |
| D50 | 12.527±0.124 | 12.432±0.119 | 0.05 | 0.96 |
| D02 | 11.370±4.167 | 11.115±4.059 | 0.33 | 0.72 |
| V12 | 94.541±0.979 | 92.521±0.955 | 0.98 | 0.338 |
| HI | 1.100±0.050 | 1.110±0.060 | −0.78 | 0.423 |
| CI | 0.770±0.030 | 0.710±0.025 | 4.41 | <0.001 |
|
| B, TMLI |
|
|
Variable | HT | IMRT | t-test | P-value |
|
|
Dmax | 13.930±0.336 | 13.890±0.351 | 0.33 | 0.88 |
| D98 | 10.931±0.452 | 11.124±0.449 | −0.32 | 0.74 |
| D95 | 11.922±0.174 | 11.920±0.158 | 0.88 | 0.92 |
|
Dmean | 12.720±0.202 | 12.698±0.211 | 0.35 | 0.85 |
| D50 | 12.910±0.297 | 12.831±0.287 | 0.75 | 0.59 |
| D02 | 13.283±0.095 | 12.950±0.096 | 0.36 | 0.68 |
| V12 | 94.360±0.961 | 93.537±0.954 | 0.41 | 0.78 |
| HI | 1.120±0.040 | 1.110±0.030 | 0.75 | 0.42 |
| CI | 0.810±0.030 | 0.770±0.050 | 4.56 | <0.001 |
Table IV presented
the results of dosimetry comparison in bilateral lung and bilateral
crystalline lens among four radiotherapy plans. The average dose to
the lung in each plan may be controlled under 8 Gy. TMLI plans
reduced the average dose to both lung and the volume ratio of
target volume occupied by 5 Gy (V5) compared with TBI
plans, and the other indexes were within the dose limitation of
normal tissue, without significant differences. Dmax,
Dmean and V5 of bilateral lungs and
Dmax and Dmean of bilateral crystalline lens
in IMRT plans were significantly increased compared with HT plans
(P<0.05).
 | Table IV.Differences of doses in organs at
risk between TBI plans and TMLI plans (cGy). |
Table IV.
Differences of doses in organs at
risk between TBI plans and TMLI plans (cGy).
|
| HT | IMRT |
|---|
|
|
|
|
|---|
| Variable | TBI | TMLI | TBI | TMLI |
|---|
| Left lung |
|
Dmax | 12.208±1.835 | 12.335±0.385 |
13.587±1.796a |
13.956±0.652b |
|
Dmean | 7.637±0.400 |
7.243±0.290c |
8.356±0.389a |
7.961±0.286b |
|
V5 | 84.486±5.377 | 80.417±2.980 |
88.354±5.255a |
83.441±2.756b |
| Right lung |
|
Dmax | 12.045±1.665 | 13.023±0.462 |
12.958±1.586a |
13.156±0.432b |
|
Dmean | 7.540±0.491 |
6.960±0.420c |
8.012±0.512a |
7.223±0.410b |
|
V5 | 82.392±2.565 |
75.710±2.262c |
87.256±2.554a |
80.004±2.353b |
| Left lens |
|
Dmax | 2.002±0.573 | 1.856±0.301 |
2.520±0.551a |
1.958±0.334b |
|
Dmean | 1.678±0.221 | 1.670±0.240 |
1.785±0.201a |
1.754±0.231b |
| Right lens |
|
Dmax | 2.012±0.564 | 1.900±0.138 |
2.112±0.560a |
1.956±0.135b |
|
Dmean | 1.678±0.243 | 1.713±0.210 |
1.681±0.253a |
1.799±0.230b |
By comparing the dose to PTV and organs at risk
among the four different radiotherapy plans, all plans were able to
meet dosimetry requirements in target volume and organs at risk. In
addition, the dose to bilateral lung and bilateral crystalline lens
in HT-TMLI was decreased compared with other plans. HT-TMLI
exhibited less damage to the bilateral lung and bilateral
crystalline lens.
Comparison of clinical untoward
effects
Each patient prior to treatment was administered
anti-emetic and antipyretic drugs to prevent emesis and fever. The
untoward effects of 18 patients during treatment were recorded.
Symptomatic treatment was performed, according to patients'
specific situation.
Nausea and emesis
Nausea and emesis was observed in 18 patients.
Emesis occurred following the second treatment, with I–II level.
Emesis was improved following the administration of anti-emetic
drugs, which did not affect the implementation of radiotherapy
plan.
Inappetence
There was mild inappetence in 18 patients.
Inappetence occurred following the third treatment. Inappetence
improved following fluid replacement therapy, which did not affect
the implementation of the radiotherapy plan.
Fever
There was fever in four patients (two treated with
IMRT-TBI, one with IMRT-TMLI and one with HT-TMLI) at body
temperature of 37.9–38.2°C in the first and second treatment. Fever
had improved following the treatment with physical cooling at low
fever or aspirin-DL-lysine at body temperature >38.5°C, which
did not affect the implementation of radiotherapy plan.
Xerostomia
There was slight to moderate xerostomia in 18
patients, and red and swollen oral mucosa in six patients.
Xerostomia occurred following the second treatment. Symptomatic
treatment was performed and patients were instructed to drink more
water and to maintain oral hygiene. All patients demonstrated
tolerance to xerostomia, which did not affect the implementation of
radiotherapy plan.
Diarrhea
Mild diarrhea was reported in five patients (two
treated with IMRT-TBI, one with IMRT-TMLI, one with HT-TBI and one
with HT-TMLI) following the treatment. Diarrhea was improved
following the administration of antidiarrheal drugs, which did not
affect the implementation of radiotherapy plan.
Rash
Mild rashes were recorded in two patients treated
with TBI (one with HT-TBI and one with IMRT-TBI) following the
fourth treatment. Rashes improved following the radiotherapy plan
and did not affect the implementation of radiotherapy plan.
According to the reactions of 18 patients following
treatment, the four radiotherapy plans revealed similar untoward
effects. Following symptomatic treatment, all untoward effects did
not affect the implementation of radiotherapy plan.
Comparison of transplantation
effects
All radiotherapy plans were completed on time in 18
patients, with a total dose of 12 Gy. All patients successfully
completed transplantation. As of March 2017, the duration of
follow-up was 3–8 months and the median duration of follow-up was 7
months. There were five survivors and one case of mortality (severe
rejection reaction following transplantation; mortality due to
severe infection) among the six patients treated with HT-TBI. A
total of four patients treated with HT-TMLI survived without
exhibiting a rejection response following transplantation. In
addition, four patients treated with IMRT-TBI and four patients
treated with IMRT-TMLI survived without rejection following
transplantation. Furthermore, the remaining 17 patients were
examined regularly and no severe untoward effects were observed
(Figs. 2–5).
Discussion
The pathogenesis of ALL is unclear due to
complicated etiology, and, therefore treatment is difficult to
administer. There is a notable heterogeneity between the treatment
and prognosis of ALL. The primary clinical therapy of ALL is
combinational chemotherapy. Targeted therapy and hematopoietic stem
cell transplantation are additional methods of treatment.
Hematopoietic stem cell transplantation via high-dose
chemo-radiotherapy or other immunosuppression pretreatment, removes
tumor cells and abnormal clone cells in vivo of patients,
and subsequent hematopoietic and immune function via the use of
hematopoietic stem cells from the donor was reconstructed (33). The present clinical study
demonstrated that hematopoietic stem cell transplantation may be
considered as the only cure for ALL.
TBI, as an important pretreatment method of
hematopoietic stem cell transplantation (10,11),
concerns the success or failure of transplantation (12). Tomotherapy accelerators have been
used in clinical treatment in the First Affiliated Hospital of
Zhengzhou University for a number of patients against solid tumors
in combination with TBI, particularly for patients with complex
target volumes or who do not qualify for conventional linear
accelerators therapy due to their condition. Compared with the
conventional IMRT plan, the tomotherapy plan exhibits a higher
conformity and increased protection to lung tissue, which may
effectively reduce the incidence of radiation pneumonia (34). As of May 2016, tomotherapy
accelerator in the First Affiliated Hospital of Zhengzhou
University has treated numerous patients with ALL, aplastic anemia
or lymphoma. A total of 18 patients with ALL in the present study
were treated with HT-TBI, HT-TMLI, IMRT-TMLI and IMRT-TBI. Various
radiotherapy plans were established depending on different target
volumes. The results of indexes in each radiotherapy plan revealed
that all plans had high conformity and homogeneity, and that doses
to target volumes may meet the requirement of 12 Gy. The doses to
organs at risk were controlled below 12 Gy; in particular, the
average dose to the lung was controlled below 8 Gy. HT plans were
significantly different from IMRT plans. HT-TMLI demonstrated more
advantages than the aforementioned plans. The DVH revealed that
lower doses were administered to the lungs in the HT-TMLI plan.
Considering the key organ of the lung and the severity of
interstitial pneumonia, the reduction of dosage to the lung may be
beneficial to patients. There was no difference in other organs at
risk among the four plans, and all plans were able to protect the
organs at risk. All patients exhibited similar untoward effects
during treatment, including mild-moderate nausea, emesis,
inappetence and xerostomia. These untoward effects improved
following clinical symptomatic treatment, and all patients revealed
tolerance to the symptomatic drugs, which did not affect the
implementation of the radiotherapy plans. In the preset study,
successful transplantation of the 18 patients was reported.
Following transplantation, one patient succumbed to mortality due
to severe infection; no severe untoward effects occurred in the
remaining patients. However, the long-term effects required
continuous follow-up observation.
By comparing the target volumes, clinical reactions
and transplant effects among HT-TBI, HT-TMLI, IMRT-TBI and
IMRT-TMLI, the clinical reactions of all plans were similar for
patients with ALL. The CI of HT plans was increased compared with
IMRT plans; HT-TMLI revealed higher conformity in the target volume
and lower doses to organs at risk. In accordance with a previous
study, the present study reported that HT-TMLI did not increase the
risk of extramedullary relapse (23). Therefore, the use of HT-TMLI may be
preferred over the aforementioned plans. This laid a theoretical
foundation for the implementation of TBI for ALL; however, due to
the relatively small number of patients and short duration of
follow-up, further investigation is required to observe the
long-term effects, transplantation complications and DFS to provide
more accurate information for clinical treatment.
Clinical comparison of HT-TBI, HT-TMLI, IMRT-TBI and
IMRT-TMLI among the 18 patients with ALL in the present study
demonstrated that HT plans exhibited increased conformity compared
with IMRT plans; HT-TMLI may reduce doses exposed to key organs
without increasing the recurrence rate following hematopoietic stem
cell transplantation, as well as attaining the requirement of
conformity and dose distribution in target volume. In addition,
with reference to another study (35), HT-TMLI may increase the doses for
target volumes and provide improved protection for organs at risk.
HT-TBI did not serve such a role; HT-TMLI did not increase the risk
of extramedullary relapse. Furthermore, the four radiotherapy plans
demonstrated similar clinical untoward effects and rate of
transplantation success, and it may be more feasible in dosimetry
to adopt HT-TMLI than the other aforementioned plans for patients
with ALL. Long-term effects, complications, transplantation
recurrence and long-term survival rates require further
investigation with an increased number of patients to follow up, in
addition to prolonged follow-up time.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during the present
study are included in this published article.
Authors' contributions
YL and FK contributed to the design and concept of
the present study. HL, DX, FJ and XZ acquired the data and drafted
the paper. YL, FK, BW and GL analyzed and interpreted the patient
data, and critically revised the paper. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of the First Affiliated Hospital of Zhengzhou University
(Zhengzhou, China). Prior to radiotherapy, all patients and their
families were notified of the requirement of treatment and possible
untoward effects, and signed written informed consent was
obtained.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ye T, Ma L and Luo S: Advances in the
treatment of acute lymphoblastic leukemiaProceedings of the Fifth
National Anatomy and Technology Conference. Three Gorges
University; Fuzhou: pp. 301–302. 2015
|
|
2
|
Zheng HY: Advancement of treatment of
acute lymphoblastic leukemia in children. J Appl Clin Pediat.
22:167–169. 2007.
|
|
3
|
Pui CH, Mullighan CG, Evans WE and Relling
MV: Pediatric acute lymphoblastic leukemia: Where are we going and
how do we get there? Blood. 120:1165–1174. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hunger SP, Loh ML, Whitlock JA, Winick NJ,
Carroll WL, Devidas M and Raetz EA: COG Acute Lymphoblastic
Leukemia Committee: Children's Oncology Group's 2013 blueprint for
research: Acute lymphoblastic leukemia. Pediatr Blood Cancer.
60:957–963. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sun Q, Tang J and Huang MY: Advancement of
treatment plan of acute lymphoblastic leukemia in children. Med
Inform. 28:389. 2015.
|
|
6
|
Hu YY and Ge FM: Advancement of treatment
of acute lymphoblastic leukemia in adult. J Pract Oncol.
29:591–594. 2014.
|
|
7
|
Linker CA, Levitt LJ, O'Donnell M, Forman
SJ and Ries CA: Treatment of adult acute lymphoblastic leukemia
with intensive cyclical chemotherapy: A follow-up report. Blood.
78:2814–2822. 1991.PubMed/NCBI
|
|
8
|
Zhang QG, Xie ST, Zeng QF, Xiao Y and Liu
X: Effect and toxic and side effects of TBI treating refractory
lymphoma in APBSCT. Contemp Med. 34:1–3. 2014.
|
|
9
|
Kim I, Park S, Kim BK, Chang HM, Bang SM,
Byun JH, Kim DJ, Min WS, Kim HJ and Kim CC: Allogeneic bone marrow
transplantation for chronic myeloid leukemia: A retrospective study
of busulfan-cytoxan versus total body irradiation-cytoxan as
preparative regimen in Koreans. Clin Transplant. 15:167–172. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Nakasone H, Fukuda T, Kanda J, Mori T,
Yano S, Kobayashi T, Miyamura K, Eto T, Kanamori H, Iwato K, et al:
Impact of conditioning intensity and TBI on acute GVHD after
hematopoietic cell transplantation. Bone Marrow Transplant.
50:559–565. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Valente M, Denis J, Grenier N, Arvers P,
Foucher B, Desangles F, Martigne P, Chaussard H, Drouet M, Abend M
and Hérodin F: Revisiting biomarkers of total-body and partial-body
exposure in a baboon model of irradiation. PLoS One.
10:e01321942015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ringdén O, Ruutu T, Remberger M,
Nikoskelainen J, Volin L, Vindeløv L, Parkkali T, Lenhoff S,
Sallerfors B, Ljungman P, et al: A randomized trial comparing
busulfan with total body irradiation as conditioning in allogeneic
marrow transplant recipients with leukemia: A report from the
Nordic Bone Marrow Transplantation Group. Blood. 83:2723–2730.
1994.PubMed/NCBI
|
|
13
|
Blaise D, Maraninchi D, Michallet M,
Reiffers J, Jouet JP, Milpied N, Devergie A, Attal M, Sotto JJ,
Kuentz M, et al: Long-term follow-up of a randomized trial
comparing the combination of cyclophosphamide with total body
irradiation or busulfan as conditioning regimen for patients
receiving HLA-identical marrow grafts for acute myeloblastic
leukemia in first complete remission. Blood. 97:3669–3671. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Dusenbery KE, Daniels KA, McClure JS,
McGlave PB, Ramsay NK, Blazar BR, Neglia JP, Kersey JH and Woods
WG: Randomized comparison of cyclophosphamide-total body
irradiation versus busulfan-cyclophosphamide conditioning in
autologous bone marrow transplantation for acute myeloid leukemia.
Int J Radiat Oncol Biol Phys. 31:119–128. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bunin N, Aplenc R, Kamani N, Shaw K, Cnaan
A and Simms S: Randomized trial of busulfan vs total body
irradiation containing conditioning regimens for children with
acute lymphoblastic leukemia: A Pediatric Blood and Marrow
Transplant Consortium study. Bone Marrow Transplant. 32:543–548.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Farese AM, Brown CR, Smith CP, Gibbs AM,
Katz BP, Johnson CS, Prado KL and MacVittie TJ: The ability of
filgrastim to mitigate mortality following LD50/60 total-body
irradiation is administration time-dependent. Health Phys.
106:39–47. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Burnett AF, Biju PG, Lui H and
Hauer-Jensen M: Oral interleukin 11 as a countermeasure to lethal
total-body irradiation in a murine model. Radiat Res. 180:595–602.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Nunamaker EA, Anderson RJ, Artwohl JE,
Lyubimov AV and Fortman JD: Predictive observation-based endpoint
criteria for mice receiving total body irradiation. Comp Med.
63:313–322. 2013.PubMed/NCBI
|
|
19
|
Linsenmeier C, Thoennessen D, Negretti L,
Bourquin JP, Streller T, Lütolf UM and Oertel S: Total body
irradiation (TBI) in pediatric patients. A single-center experience
after 30 years of low-dose rate irradiation. Strahlenther Onkol.
186:614–620. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Bhatia S, Francisco L, Carter A, Sun CL,
Baker KS, Gurney JG, McGlave PB, Nademanee A, O'Donnell M, Ramsay
NK, et al: Late mortality after allogeneic hematopoietic cell
transplantation and functional status of long-term survivors:
Report from the Bone Marrow Transplant Survivor Study. Blood.
110:3784–3792. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Bhatia S, Robison LL, Francisco L, Carter
A, Liu Y, Grant M, Baker KS, Fung H, Gurney JG, McGlave PB, et al:
Late mortality in survivors of autologous hematopoietic-cell
transplantation: Report from the Bone Marrow Transplant Survivor
Study. Blood. 105:4215–4222. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Appelbaum FR, Badger CC, Bernstein ID,
Buckner CD, Deeg HJ, Eary JF, Matthews DC, Press OW, Storb R and
Thomas ED: Is there a better way to deliver total body irradiation?
Bone Marrow Transplant. 10 Suppl 1:S77–S81. 1992.
|
|
23
|
Kim JH, Stein A, Tsai N, Schultheiss TE,
Palmer J, Liu A, Rosenthal J, Forman SJ and Wong JY: Extramedullary
relapse following total marrow and lymphoid irradiation in patients
undergoing allogeneic hematopoietic cell transplantation. Int J
Radiat Oncol Biol Phys. 89:75–81. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lee KH, Lee JH, Choi SJ, Lee JH, Kim S,
Seol M, Lee YS, Kim WK, Seo EJ, Park CJ, et al: Bone marrow vs
extramedullary relapse of acute leukemia after allogeneic
hematopoietic cell transplantation: Risk factors and clinical
course. Bone Marrow Transplant. 32:835–842. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Chong G, Byrnes G, Szer J and Grigg A:
Extramedullary relapse after allogeneic bone marrow transplantation
for haematological malignancy. Bone Marrow Transplant.
26:1011–1015. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Solh M, DeFor TE, Weisdorf DJ and Kaufman
DS: Extramedullary relapse of acute myelogenous leukemia after
allogeneic hematopoietic stem cell transplantation: Better
prognosis than systemic relapse. Biol Blood Marrow Transplant.
18:106–112. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Mortimer J, Blinder MA, Schulman S,
Appelbaum FR, Buckner CD, Clift RA, Sanders JE, Storb R and Thomas
ED: Relapse of acute leukemia after marrow transplantation: Natural
history and results of subsequent therapy. J Clin Oncol. 7:50–57.
1989. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wong JY, Forman S, Somlo G, Rosenthal J,
Liu A, Schultheiss T, Radany E, Palmer J and Stein A: Dose
escalation of total marrow irradiation with concurrent chemotherapy
in patients with advanced acute leukemia undergoing allogeneic
hematopoietic cell transplantation. Int J Radiat Oncol Biol Phys.
85:148–156. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Rosenthal J, Wong J, Stein A, Qian D, Hitt
D, Naeem H, Dagis A, Thomas SH and Forman S: Phase 1/2 trial of
total marrow and lymph node irradiation to augment
reduced-intensity transplantation for advanced hematologic
malignancies. Blood. 117:309–315. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Karnofsky DA and Burchenal JH: The
clinical evaluation of chemotherapeutic agents in cancerEvaluation
of Chemotherapeutic Agents. Macleod CM: Columbia University Press;
New York, NY: pp. 1911949
|
|
31
|
Trotti A, Byhardt R, Stetz J, Gwede C,
Corn B, Fu K, Gunderson L, McCormick B, Morrisintegral M, Rich T,
et al: Common toxicity criteria: Version 2.0. an improved reference
for grading the acute effects of cancer treatment: Impact on
radiotherapy. Int J Radiat Oncol Biol Phys. 47:13–47. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
U.S. Department of Health and Human
Services, . The National Institutes of Health and National Cancer
Institute: Common Terminology Criteria for Adverse Events (CTCAE).
Version 4.0.3. https://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03/CTCAE_4.03_2010-06-14_QuickReference_5×7.pdfJune
14–2010
|
|
33
|
Storek J, Geddes M, Khan F, Huard B, Helg
C, Chalandon Y, Passweg J and Roosnek E: Reconstitution of the
immune system after hematopoietic stem cell transplantation in
humans. Semin Immunopathol. 30:425–437. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Moon SH, Shin KH, Kim TH, Yoon M, Park S,
Lee DH, Kim JW, Kim DW, Park SY and Cho KH: Dosimetric comparison
of four different external beam partial breast irradiation
techniques: Three-dimensional conformal radiotherapy,
intensity-modulated radiotherapy, helical tomotherapy, and proton
beam therapy. Radiother Oncol. 90:66–73. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Schultheiss TE, Wong J, Liu A, Olivera G
and Somlo G: Image-guided total marrow and total lymphatic
irradiation using helical tomotherapy. Int J Radiat Oncol Biol
Phys. 67:1259–1267. 2007. View Article : Google Scholar : PubMed/NCBI
|