Introduction
In recent years, the incidence of infertility has
been increasing annually. According to statistics from the World
Health Organization (WHO), the global infertility rate among
couples has reached ~15%; therefore, infertility is a pressing
issue in the field of reproductive medicine, which severely impacts
social development (1). Global
research has revealed that male infertility factors contribute to
~50% of infertility cases among couples, with abnormal sperm
quality being one of the significant causes of male infertility
(2). Reduced sperm motility, known
clinically as asthenozoospermia (AS), is the most common cause of
abnormal sperm quality (3). Sperm
motility is crucial for sperm to penetrate the cervical mucus,
reach the site of fertilization in the fallopian tubes and enter
the oocyte to form a fertilized egg, thus completing fertilization.
According to the WHO sixth edition semen analysis criteria
(4), a diagnosis of AS can be
considered when the progressive motility of sperm is <32% in two
or more semen samples, when other sperm parameters are within the
normal range (5). Currently, there
are no definitive treatments for AS, and a number of infertile
couples often rely on assisted reproductive techniques. Therefore,
investigating the pathogenesis of AS will help in early
intervention and improve the quality of life for patients.
In recent years, with the advancement of genomic
technologies, a large number of non-coding genes have been
discovered in the human genome. The transcripts of these genes are
called non-coding RNAs, with RNA molecules >200 nucleotides
referred to as long non-coding RNAs (lncRNAs) (6). Increasing evidence has suggested that
lncRNAs serve important roles in the biological processes of sperm,
including sperm formation, maturation and function (7). Zhang et al (8) reported significant differences in the
expression profiles of lncRNAs between normal sperm and
asthenozoospermic sperm, which may be related to sperm motility.
Therefore, further investigation of these differences may improve
understanding of the pathogenesis of AS. Saberiyan et al
(9) demonstrated that ANO1-AS2 may
affect sperm function in patients with AS and teratozoospermia by
regulating the expression of the ANO1 gene. Another study
discovered that long intergenic noncoding RNA 00574 (Linc00574) may
affect sperm motility through the regulation of TCTE3 expression
(10). These findings further
confirm the significant role of lncRNAs in the pathogenesis of
AS.
In our previous study, RNA sequencing and
bioinformatics analysis were used to determine the interaction
between lncRNAs and mRNAs in AS (11). Numerous differentially expressed
lncRNAs were identified and validated through quantitative PCR
(qPCR). Notably, Linc00893 (official gene name: EOLA1-DT) was
revealed to be significantly downregulated in the seminal plasma of
patients with AS, thus suggesting its association with the
pathogenesis of AS. However, the pathological relationship between
Linc00893 expression and AS, as well as its impact on cellular
function and mechanisms, remains unclear. The present study aimed
to explore the relationship between Linc00893 and AS, as well as to
investigate its cellular function and molecular mechanisms. The
findings may contribute to revealing the potential role of
Linc00893 in sperm development and function, providing novel
insights and approaches for the prevention and treatment of AS.
Materials and methods
Participants
For the present study, a total of 27 semen samples
from patients with AS and 31 samples from a control group, which
consisted of healthy individuals, were collected. The sample
collection took place between November 2021 and May 2023. Sperm
parameters were gathered from clinical microscopic examinations and
semen analysis data. All specimens were obtained from the Hainan
Women and Children's Medical Center (Haikou, China). The present
study was approved by the Ethics Committee of the Hainan Women and
Children's Medical Center (approval no. 2021-033) and conducted in
accordance with the principles outlined in The Declaration of
Helsinki. Written informed consent was obtained from all
participants after they were provided with detailed information
about the experiment.
The inclusion criteria for the AS group were as
follows: i) Two consecutive semen analyses showing progressive
motility <32%; ii) normal levels of hormones, such as
follicle-stimulating hormone, testosterone and estradiol; iii) no
abnormalities found during physical examination of the
genitourinary system; iv) availability of complete medical records
and follow-up data; v) in the absence of contraception, despite
having regular sexual intercourse (at least 2–3 times per week),
they have not been able to conceive within 1 year. The exclusion
criteria were as follows: i) Abnormal parameters, such as fructose,
acid phosphatase, liquefaction time, pH, sperm morphology and
α-glucosidase in seminal plasma; ii) incomplete medical records and
follow-up data; iii) refusal to provide informed consent. Inclusion
criteria for the control group were as follows: i) Two consecutive
routine semen analyses both indicating progressive motility
>32%; ii) a normal semen analysis report within the past month
and all other examinations showing normal results; iii)
physiological parameters, such as age and body mass index (BMI),
comparable to those of the AS group. Exclusion criteria included:
i) Presence of acute or chronic diseases, or long-term medication
use; ii) absence of complete medical records and follow-up data;
iii) unwillingness to sign an informed consent form. There were no
statistically significant differences in physiological parameters,
such as age and BMI, between the AS group and the control group, as
shown in Table I. Notably,
immotile sperm rate was calculated as 100% minus the total sperm
motility (%), and total sperm motility (%) is the sum of
progressive motility and non-progressive motility.
 | Table I.Subject characteristics. |
Table I.
Subject characteristics.
| Characteristic | Control (n=31) | AS (n=27) | P-value |
|---|
| Median age, years
(IQR) | 33 (31, 35) | 33 (31, 37.5) | 0.975 |
| Median BMI,
kg/m2 (IQR) | 21.7 (20.85,
22.85) | 21.7 (20.5,
23.4) | 0.925 |
| Median sperm
concentration, 106/ml (IQR) | 125.6 (72.25,
158.05) | 24 (12.75,
55.05) | <0.001 |
| Mean ± SD total
sperm, 106 | 398.58±204.4 | 134.81±100.97 | <0.001 |
| Mean ± SD total
sperm motility, % | 53.581±8.4094 | 16.804±11.292 | <0.001 |
| Median non-forward
motility sperm rate, % (IQR) | 7 (6, 10) | 5 (3, 9) | 0.023 |
| Median forward
motility sperm rate, % (IQR) | 45 (39.5, 51) | 9 (3, 15.5) | <0.001 |
| Mean ± SD inactive
sperm rate, % | 46.419±8.4094 | 83.196±11.292 | <0.001 |
| Median pH, %
(IQR) | 7.2 (7.2, 7.2) | 7.2 (7.2, 7.2) | 0.445 |
| Median abstinence,
days (IQR) | 4 (3, 6.5) | 5 (3, 5.5) | 0.820 |
| Median normal
morphology rate, % (IQR) | 7.7 (5.45,
10.25) | 3.9 (2.4,
5.35) | <0.001 |
| Median semen
volume, ml (IQR) | 3.2 (2.45,
4.2) | 3.6 (2.9, 4.6) | 0.408 |
Human SSC culture
Testicular biopsies from patients with obstructive
azoospermia were employed for the generation of SSCs. Specifically,
tissue was collected from a 35-year-old patient diagnosed with
obstructive azoospermia in December 2021. The isolated SSCs from
this biopsy were utilized in this study and in subsequent studies.
Human immortalized SSCs were prepared according to a previously
published protocol (12). The
cells were cultured in a CO2 incubator (cat. no. 3111;
Thermo Fisher Scientific, Inc.) at 37°C with 5% CO2. The
cells were cultured in Dulbecco's Modified Eagle's Medium/F12 (cat.
no. 11320033; Gibco; Thermo Fisher Scientific, Inc.) supplemented
with 10% fetal bovine serum (cat. no. 10099-141; Gibco; Thermo
Fisher Scientific, Inc.) and 1% penicillin-streptomycin. Passaging
was performed at a ratio of 1:4, with medium changes 2–3 times per
week.
Reverse transcription-qPCR
(RT-qPCR)
Approximately 106 spermatogonial stem
cells (SSCs) or >500 µl seminal plasma samples were collected.
Subsequently, 1 ml TRIzol® (cat. no. 15596018;
Invitrogen; Thermo Fisher Scientific, Inc.) was added, followed by
vigorous shaking and a 5 min incubation at 4°C. Subsequently, 0.2
ml chloroform was added, and the mixture was vigorously shaken for
15 sec, followed by a 3 min incubation at 4°C. The sample was then
centrifuged at 24,148.8 × g for 15 min at 4°C, and the upper
aqueous phase was transferred to a new tube. An equal volume of
isopropanol was added, mixed and incubated at −20°C for 20 min,
followed by centrifugation at 24,148.8 × g for 15 min at 4°C to
remove the supernatant. The pellet was then washed with 1 ml 75%
DEPC ethanol and centrifuged at 10,732.8 × g for 5 min at 4°C, and
the liquid was discarded to obtain RNA. RT was then performed
according to the instructions provided with the Bestar™ qPCR RT Kit
(cat. no. DBI-2220; DBI Bioscience) or the miRNA RT Reagent Kit
(cat. no. 600036; Agilent Technologies, Inc.); briefly, 2 µg total
RNA was used as a template and heated at 65°C for 5 min, followed
by immediate cooling on ice. The reaction mixture, including gDNA
Remover, 10X gDNA Remover Buffer, RNA and RNase-free water, was
prepared according to the provided protocol. The mixture was
incubated at 37°C for 5 min, followed by cooling on ice.
Subsequently, another reaction mixture, including the
aforementioned reaction mixture, 5X RT Buffer, RT Enzyme Mix and
Primer Mix, was prepared. The first-strand cDNA was synthesized at
37°C for 15 min and 98°C for 5 min, and then collected for further
use. Subsequently, qPCR amplification was performed. The reaction
system, with a total volume of 20 µl, was prepared according to the
protocol provided with 2X Taq PCR Master Mix (cat. no. DBI-2030;
DBI Bioscience). The qPCR conditions were as follows: Initial
denaturation at 95°C for 2 min, followed by 40 cycles of
denaturation at 94°C for 20 sec, annealing at 58°C for 20 sec and
extension at 72°C for 20 sec. Melting curve analysis was performed
as follows: 94°C for 30 sec, 65°C for 30 sec and 94°C for 30 sec.
Each sample was repeated three times. U6 was employed as the
internal reference gene for miRNA expression, whereas GAPDH served
as the internal reference gene for mRNA expression. The
experimental data were analyzed using the 2−ΔΔCq method
(13). The primer sequences are
provided in Table II.
 | Table II.Reverse transcription-quantitative
PCR primer sequences. |
Table II.
Reverse transcription-quantitative
PCR primer sequences.
| Name | Sequence,
5′-3′ |
|---|
| GAPDH | F:
CACCATCTTCCAGGAGCGAG |
|
| R:
AAATGAGCCCCAGCCTTCTC |
| Linc00893 | F:
CAGAATTCAGGCCTCGTGGT |
|
| R:
GGGAGAAGTAGGCGCATCTC |
| miR-107 | F:
ACACTCCAGCTGGGAGCAGC |
|
| ATTGTACAGGG |
|
| R:
CTCAACTGGTGTCGTGGAGTC |
|
|
GGCAATTCAGTTGAGCCGATAGT |
| U6 | F:
CTCGCTTCGGCAGCACA |
|
| R:
AACGCTTCACGAATTTGCGT |
Vector construction and
transfection
Linc00893 small interfering (si)RNA was designed and
synthesized by Guangzhou Anernor Biotechnology Co., Ltd. The
following three siRNAs were designed: si-Linc00893 #1,
5′-TTGACTTCATAACCAAGTTCT-3′; si-Linc00893 #2,
5′-GGCTGTTTTGAAGTCAGTATT-3′; si-Linc00893 #3,
5′-CTGTTTTGAAGTCAGTATTCA-3′; negative control (NC),
5′-TTCTCCGAACGAGTCACGTTT-3′. siRNA (50 nM) transfection was
performed at 37°C using Lipofectamine® 3000 (cat. no.
L3000-015; Invitrogen; Thermo Fisher Scientific, Inc.) at a ratio
of 1:2 (siRNA:Lipofectamine 3000) when the cell density reached
30%. After transfection of SSCs for 6 h, the culture medium was
replaced with fresh medium, and the cell culture plates were
incubated at 37°C with 5% CO2 for 24 h before subsequent
experiments.
The full-length human myosin heavy chain 9 (MYH9)
cDNA (NM_002473.5) was amplified using PCR with PrimeSTAR HS DNA
polymerase (cat. no. R010B; Takara Bio, Inc.). The primer sequences
were as follows: Forward
5′-GTGGATCCGAGCTCGGTACCCGCCACCATGGCACAGCAAGCTGCCGATAAG-3′ and
reverse
5′-GAAAATAAAGATATTTTATTACCGGTTTAATTAATTATTCGGCAGGTTTGGCCTCAG-3′.
The PCR amplification conditions were segmented into three distinct
phases: Initial denaturation for one cycle at 98°C for 5 min;
followed by 30 cycles, each consisting of a denaturation step at
98°C for 10 sec, annealing at 55°C for 10 sec and extension at 72°C
for 90 sec; the amplification process was finalized with one cycle
of final extension at 72°C for 8 min. The amplified product was
subsequently cloned into the pLVX-Puro lentiviral vector (cat. no.
GV358; Shanghai GeneChem Co., Ltd.) between EcoRI and
BamHI sites to generate the recombinant lentiviral
expression vector pLVX-Puro-MYH9. A negative control vector,
containing the vector components but lacking the gene-coding
sequence, was also used for comparison. The 3rd generation
lentiviral packaging system, including 4 µg pCMV–VSV-G (Shanghai
GeneChem Co., Ltd), 3 µg pCMV-ΔR8.91 (Shanghai GeneChem Co., Ltd)
and 1 µg pLVX-Puro-MYH9 was transfected into 293T cells (The Cell
Bank of Type Culture Collection of The Chinese Academy of Sciences)
using Lipofectamine 3000 at 37°C, with the transfection process
sustained for 8 h. The supernatant containing lentiviral particles
was collected 48 h post-transfection, filtered through 0.45 µm
filters and concentrated by ultracentrifugation at 66,125 × g for
2.5 h at 4°C. For lentiviral transduction, SSCs (30% confluence)
were infected with lentiviral particles at a multiplicity of
infection of 50 in the presence of 2 µg/ml polybrene. Cells were
transduced for 12 h at 37°C, after which the medium was replaced
with regular culture medium, and subsequent experiments were
carried out 96 h after transduction. When co-transfecting cells
with the MYH9 overexpression vector and si-Linc00893, the MYH9
overexpression lentivirus was transduced first, followed by
transfection with the siRNA.
Cell Counting Kit 8 (CCK8) assay
The cells were seeded at a suitable density of
10,000 cells/well in a 96-well plate, with 100 µl culture medium
added to each well. The plate was then placed in a cell culture
incubator at 37°C and 5% CO2 for 24 h to allow cell
attachment. On days 1, 2, 3, 4 and 5 after siRNA transfection, 10
µl CCK-8 reagent (cat. no. CK04; Dojindo Laboratories, Inc.) was
added to the corresponding wells. The plate was gently shaken to
ensure thorough mixing of the CCK-8 reagent with the culture
medium. The 96-well plate was then returned to the cell culture
incubator at 37°C and 5% CO2 for an additional 3 h. The
absorbance of each well was measured at a wavelength of 450 nM
using an ELISA reader (cat. no. ELx800; BioTek Instruments,
Inc.).
EdU assay
The cells were seeded at a suitable density of
10,000 cells/well in a 96-well plate. After siRNA transfection and
according to the instructions provided with the EdU assay kit (cat.
no. C0078S; Beyotime Institute of Biotechnology), EdU at a final
concentration of 10 nM was added to the culture medium. The cells
were then cultured at 37°C for an additional 4 h to allow EdU to be
taken up by the cells and incorporated into newly synthesized DNA.
The culture medium was then removed, and the cells were fixed with
4% formaldehyde at room temperature for 20 min. Subsequently, 0.5%
Triton X-100 was used to permeabilize the cells at room temperature
for 10 min. The permeabilization solution was removed, and
fluorescently labeled copper sulfate was added, followed by
incubation at room temperature for 30 min. The cells were incubated
with DAPI for nuclear staining at room temperature for 10 min, and
then washed with PBS three times to remove excess dyes and
reagents. Finally, the cells were observed, images were captured
under a fluorescence microscope (DS-Fi3; Nikon Corporation) and the
proliferation of cells was assessed by calculating the ratio of
EdU-positive cells to total cells.
Apoptosis detection
Following siRNA transfection and vector
transduction, cells were digested with 0.25% trypsin-EDTA solution
and washed with PBS. Subsequently, the cell count was determined
using a cell counter, the cells were centrifuged at 300 × g for 5
min at room temperature. and the supernatant was discarded. The
cells were then washed twice with PBS and were suspended in 1X
binding buffer at a concentration of 1×106 cells/100 µl.
According to the instructions provided by the apoptosis detection
kit (cat. no. 556547; BD Biosciences), 5 µl Annexin V-FITC and 5 µl
propidium iodide (PI) were added to each cell sample, gently mixed
and incubated at room temperature in the dark for 15 min. After
incubation, 400 µl 1X binding buffer was added to each sample and
gently mixed. The stained cell samples were analyzed using a flow
cytometer (FACSCalibur; BD Biosciences). The fluorescence signal of
Annexin V was detected using the FITC channel, and the fluorescence
signal of PI was detected using the PE channel. FlowJo V10 software
(FlowJo, LLC) was used to analyze the apoptotic rate of the
cells.
Fluorescence in situ hybridization
(FISH) detection
SSCs were cultured to 70–80% confluence, then washed
with PBS and fixed with 4% formaldehyde at room temperature for 15
min. After fixation, the cells were washed with PBS again and
permeabilized with 0.5% Triton X-100 for 10 min at room
temperature, to enhance cell membrane permeability. Following
permeabilization, the cells were washed with PBS again. The FISH
detection kit was purchased from Guangzhou Ribobio Co., Ltd. (cat.
no. C10910). The cells were incubated in pre-hybridization buffer
at 37°C for 30 min, followed by replacement with hybridization
solution containing Linc00893 FISH probe, with a concentration of 1
µM. The cells were hybridized for ≥16 h at 37°C. After
hybridization, the cells were washed with wash buffer and treated
with anti-fluorescence quenching reagent Fluoromount-G (cat. no.
0100-01; SouthernBiotech). Subsequently, blocking was performed
using a blocking solution containing 5% bovine serum albumin (BSA;
cat. no. 9048-46-8; AbMole Bioscience Inc.) at room temperature for
30 min to minimize nonspecific binding. Following treatment, the
cells were washed with wash buffer again. The nuclei were stained
with DAPI (cat. no. D9542; MilliporeSigma) at room temperature for
10 min, and the cells were washed with PBS once more. The cells
were observed under a fluorescence microscope and images were
captured. The Linc00893 FISH probe, with the following sequence:
5′-TAACACAAAGCTCTTTGCCTGCCCTCTAGCCTTCTTAACC-3′, was designed and
synthesized by Huzhou Hippo Biotechnology Co., Ltd.
Dual-luciferase assay
The binding site prediction of Linc00893 and
microRNA (miR)-107 was conducted using starBase (https://rnasysu.com/encori/), whereas the predicted
binding site between miR-107 and MYH9 3′UTR was performed using
TargetScan 7.0 (http://www.targetscan.org/). Based on the identified
binding sites, corresponding wild-type and mutant sequences of
Linc00893 and MYH9were designed and constructed into the psiCHECK-2
vector (Shanghai GeneChem Co., Ltd). The constructed vectors, along
with the miR-107 mimic (sequence: 5′-ACUAUCGGGACAUGUUACGACGA-3′)
and NC (sequence: 5′-UUUGUACUACACAAAAGUACUG-3′) designed and
synthesized by Huzhou Hippo Biotechnology Co., Ltd., were
co-transfected into SSCs (30% confluence) cultured in a 24-well
plate using Lipofectamine 3000 transfection reagent. After 6 h of
incubation at 37°C, under 5% CO2, the culture medium was
replaced with complete culture medium. According to the
instructions of the Dual-Glo Luciferase Assay Kit (cat. no. E1910;
Promega Corporation), the cells were lysed using a lysis buffer and
the supernatant was collected by centrifugation at 15,000 × g for 5
min at room temperature. Subsequently, 100 µl supernatant was mixed
with 100 µl luciferase assay reagent and the firefly luciferase and
Renilla luciferase activity was measured using a microplate
reader (ReadMax 1200; Shanghai Shanpu Biotechnology Co., Ltd.).
Normalization of the firefly luciferase results by comparison with
Renilla luciferase activity.
RNA immunoprecipitation (RIP)
experiment
The RIP assay kit was purchased from Guangzhou
BersinBio Biotechnology Co., Ltd. (cat. no. Bes5101). Briefly,
~4×107 cells were scraped and collected in an RNase-free
Eppendorf tube. After centrifugation at 4°C and 16,837.5 × g for 5
min, the supernatant was discarded and the cells were washed with
PBS 1–2 times. Subsequently, the cells were lysed in 1 ml cell
lysis buffer containing 10 µl protease inhibitor, 10 µl phosphatase
inhibitor and 12 µl PMSF, and incubated at 4°C for 1–2 h. Finally,
the cell lysate was centrifuged at 4°C and 24,148.8 × g for 15 min,
and the supernatant was transferred to a new 1.5 ml Eppendorf tube
and labeled. Magnetic beads (50 µl) were washed with 500 µl RIP
buffer and centrifuged at 4°C and 1,509.7 × g for 1 min; this step
was repeated twice. Subsequently, the beads were resuspended in 500
µl RIP buffer, and added to 5 µl Argonaute 2 (Ago2; 1:50; cat. no.
ab186733; Abcam) or 5 µl IgG (1:50; cat. no. ab172730; Abcam)
antibodies, followed by incubation at 4°C for 8 h. After
centrifugation of the bead-antibody mixture at 4°C and 1,509.7 × g
for 2 min, the supernatant was discarded, and the beads were washed
once with 500 µl RIP buffer. Subsequently, 300 µl cell lysate was
added to the mixture and incubated overnight at 4°C, while 30 µl
lysate was taken as the input group. After incubation, the
bead-antibody mixture was centrifuged at 4°C and 1,509.7 × g for 2
min, the supernatant was discarded and the beads were washed six
times with 1 ml RIP buffer. Finally, the purified RNA was obtained
and stored at −80°C for qPCR detection of miR-107 and Linc00893
expression.
Western blotting
For protein lysis of SSCs, RIPA buffer (cat. no.
89901; Thermo Fisher Scientific, Inc.) was used. Protein
quantification was performed using the BCA (cat. no. 23225; Thermo
Fisher Scientific, Inc.) method. For SDS-PAGE (cat. no. 1610184;
Bio-Rad Laboratories, Inc.), glass plates were cleaned and set up
in a casting stand. A 10% separating gel was prepared using a
mixture of acrylamide, Tris, SDS, ammonium persulfate, TEMED and
distilled water. After polymerization, a 5% stacking gel was added.
Samples containing 20 µg protein and 3.5 µl marker were loaded and
separated by SDS-PAGE. For protein transfer, a PVDF membrane (cat.
no. IPVH00010; MilliporeSigma) was equilibrated and proteins were
transferred at a constant current of 300 mA. The membrane was
subsequently blocked with 5% BSA at room temperature for 30 min and
incubated with MYH9 (1:1,000; cat. no. ab138498; Abcam) or GAPDH
(1:1,000; cat. no. ab8245; Abcam) primary antibodies at room
temperature for 1 h, followed by incubation with HRP-conjugated
secondary antibodies (anti-rabbit, 1:10,000, cat. no. ab6721,
Abcam; anti-mouse, 1:5,000, cat. no. ab6728, Abcam) at room
temperature for 1 h. Chemiluminescence detection was performed
using a mixture of reagents A and B (cat. no. 32106; Thermo Fisher
Scientific, Inc.), and the signal was captured on X-ray film. The
gel images were scanned and band intensities were semi-quantified
using ImageJ software (v1.8.0; National Institutes of Health).
Gene Expression Omnibus (GEO) database
analysis
The GSE160749 dataset from the GEO database
(https://www.ncbi.nlm.nih.gov/geo/)
was extracted using the Aclbi platform (https://www.aclbi.com/static/index.html#/geo) to
compare the expression difference of MYH9 between AS and control
(healthy individuals) semen samples. Utilizing the GSE160749
dataset, AS and control semen samples were selected; subsequently,
the present study focused on MYH9 gene expression, Utilizing the
Aclbi platform for our analyses, boxplot diagrams were employed to
visualize the expression differences between the two groups.
Statistical analysis
Statistical analysis and data visualization were
performed using GraphPad Prism 9.0 software (Dotmatics).
Differences between two groups were compared using an independent
samples t-test. For datasets containing multiple groups,
differences among the groups were compared using ANOVA (for
parametric data) or Kruskal-Wallis test (for non-parametric data).
In the event of a significant result, a suitable post hoc multiple
comparisons, test such as Tukey (for parametric data) or Dunn's
(for non-parametric data) was employed. The Wilcoxon rank-sum test
was utilized to examine differential expression of MYH9 in the
GSE160749 dataset. Pearson correlation analysis was utilized to
assess the linear relationship between two variables. Normally
distributed data are presented as the mean ± standard deviation,
while non-normally distributed data are presented as the median and
interquartile range and the Mann-Whitney U test was employed for
analyses. P<0.05 was considered to indicate a statistically
significant difference.
Results
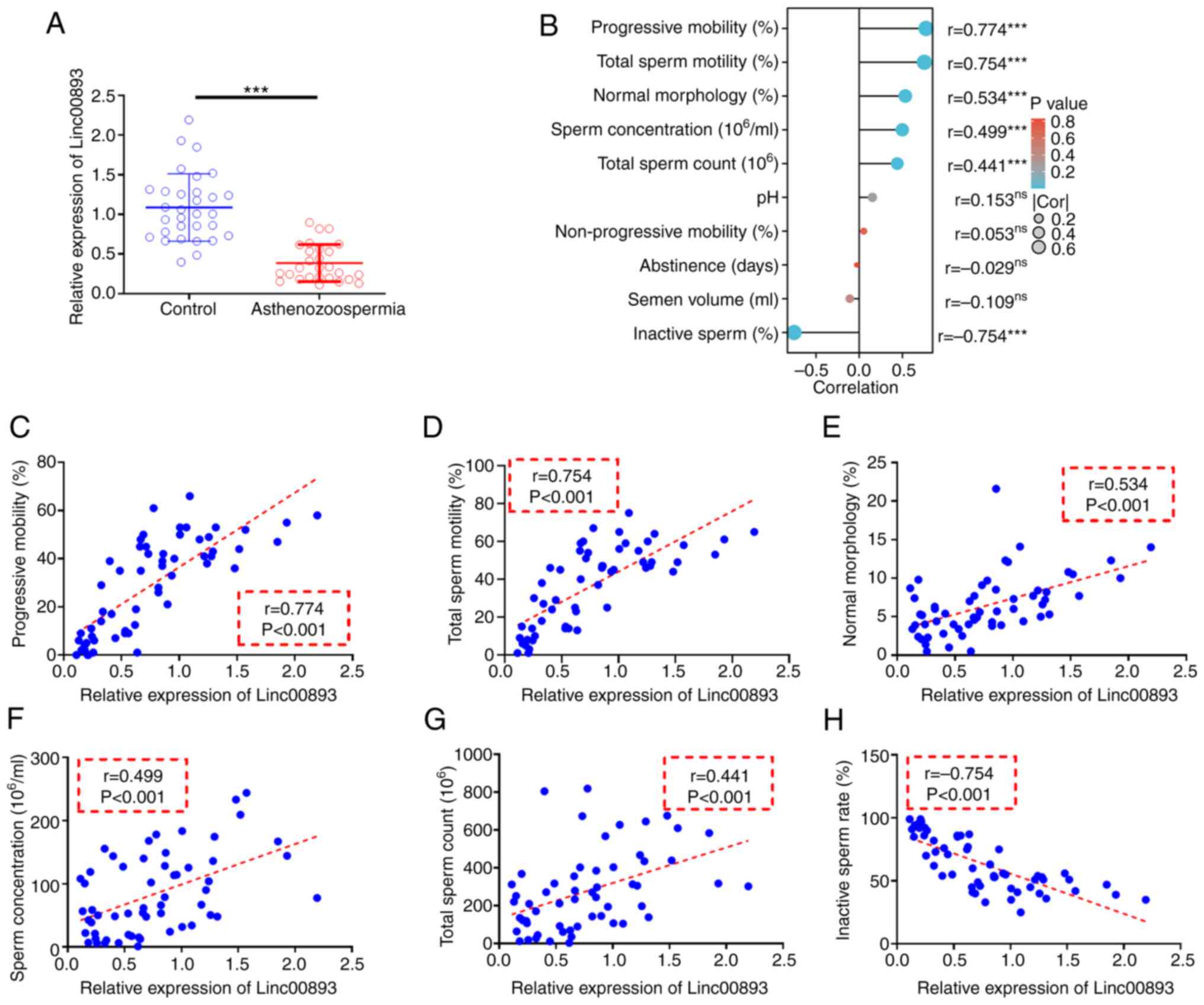
Linc00893 is downregulated in AS semen
samples and is positively correlated with sperm motility
To investigate the expression of Linc00893 in AS and
control semen samples, as well as its correlation with the
pathology of AS, RT-qPCR was used to assess the expression levels
of Linc00893 in semen. The results of RT-qPCR of clinical semen
samples revealed that the expression levels of Linc00893 were
significantly lower in the AS group compared with those in the
control group (Fig. 1A). Pearson
correlation analysis (Fig. 1B)
demonstrated significant positive correlations between Linc00893
expression in semen samples and progressive mobility sperm rate
(Fig. 1C), total sperm motility
(Fig. 1D), normal morphology rate
(Fig. 1E), sperm concentration
(Fig. 1F) and total sperm count
(Fig. 1G) (all r>0.3).
Moreover, Linc00893 expression showed a significant negative
correlation with inactive sperm rate (r<-0.3; Fig. 1H). These findings suggested that
Linc00893 may be significantly downregulated in AS semen samples,
and its expression is positively associated with various parameters
of sperm motility, indicating its potential role in the pathology
of AS, and its relevance as a biomarker for sperm motility and
quality.
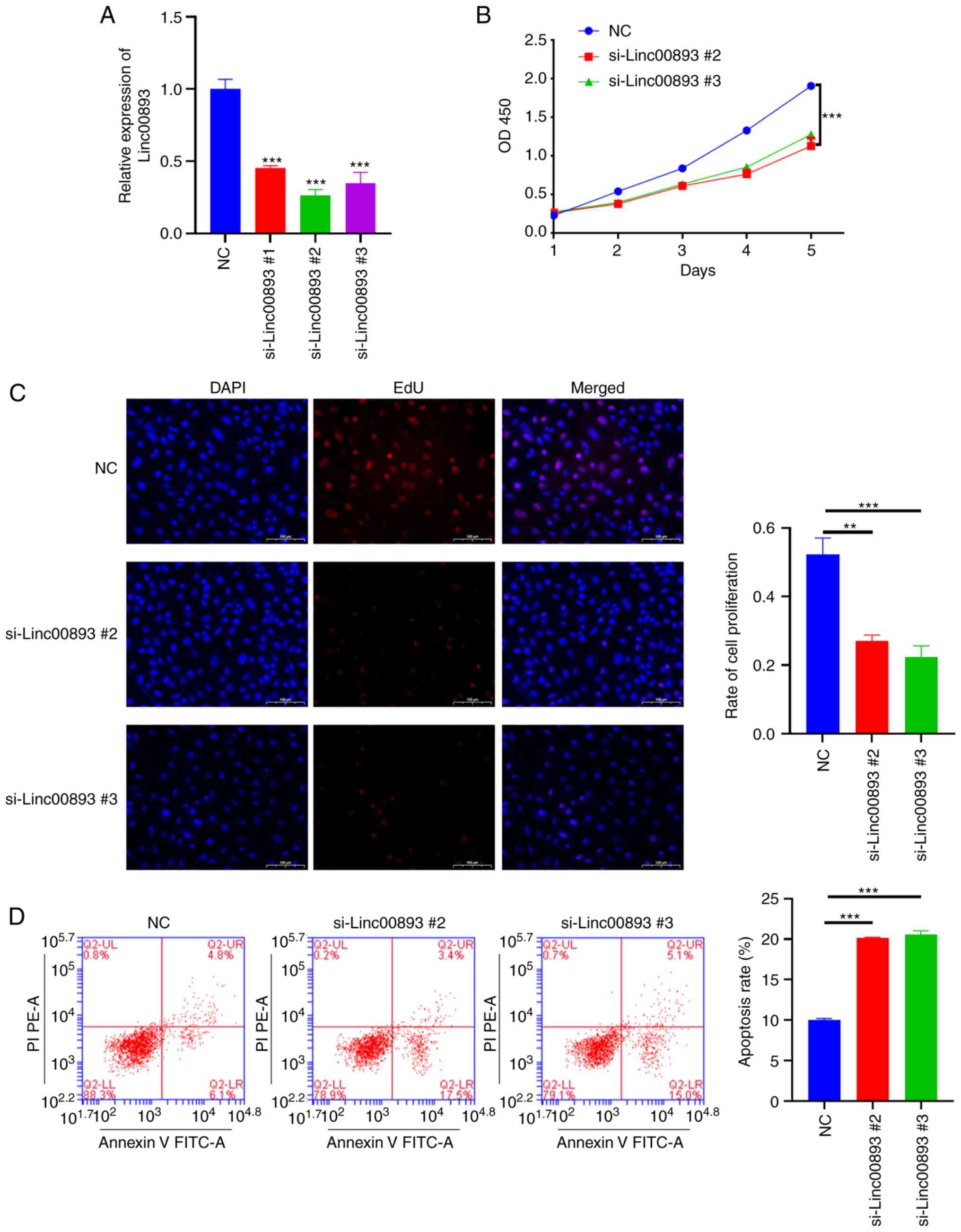
Knockdown of Linc00893 suppresses SSC
vitality
To investigate the effect of Linc00893 expression on
SSCs, siRNA was used to knockdown Linc00893. To select the siRNA
with the highest knockdown efficiency, three siRNA targets were
designed. After transfecting SSCs with siRNAs targeting Linc00893,
the results demonstrated a significant reduction in Linc00893
expression levels compared with those in the NC group, with all
three siRNAs showing efficacy (Fig.
2A). Based on the knockdown efficiency, si-Linc00893 #2 and
si-Linc00893 #3 were chosen for further experimentation. The CCK8
assay was performed to assess cell proliferation and revealed a
significant inhibition of SSC proliferation in the si-Linc00893
groups compared with that in the NC group (Fig. 2B). In addition, EdU staining was
used to assess cell proliferation, and the results demonstrated a
significant suppression of SSC proliferation in the si-Linc00893
groups compared with that in the NC group (Fig. 2C). Additionally, flow cytometry was
performed to examine cell apoptosis, which revealed a significant
increase in the apoptotic rate (including both early and late
apoptosis) of SSCs in the si-Linc00893 groups compared with that in
the NC group (Fig. 2D). These
results collectively indicated that knockdown of Linc00893 in SSCs
may lead to a marked decrease in cell viability and proliferation,
and an increase in apoptosis, highlighting the critical role of
Linc00893 in maintaining SSC vitality.
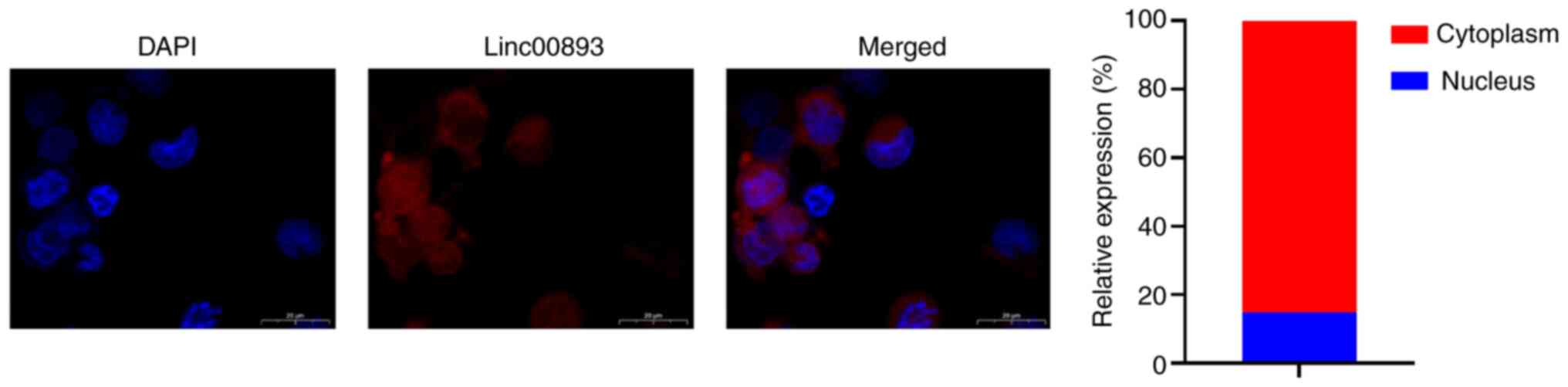
Linc00893 inhibits MYH9 expression
through competitive binding to miR-107
FISH was performed to determine the subcellular
localization of Linc00893 in SSCs. FISH analysis of Linc00893
expression in SSCs revealed predominantly cytoplasmic localization
(Fig. 3). Cytoplasmic expression
of lncRNAs can potentially influence the expression of target genes
through competitive endogenous RNA binding to miRNAs (14). Therefore, the miRNAs that bind to
Linc00893 were predicted using the starBase database and miR-107
was selected, which has been reported to be present in sperm
(15). The binding sites between
Linc00893 and miR-107 were further identified, as elucidated
through the starBase database (Fig.
4A). This interaction was verified using a dual-luciferase
reporter assay, wherein the wild-type Linc00893 sequence was found
to bind to miR-107, whereas the mutant Linc00893 sequence failed to
bind (Fig. 4B). Additionally,
using the TargetScan database, the target genes of miR-107 were
predicted and MYH9 was selected, which has been reported to be
expressed in sperm and to influence sperm function (16). The predicted binding sites between
miR-107 and MYH9 were identified using the same database (Fig. 4C), and this interaction was
confirmed using a dual-luciferase reporter assay, where the
wild-type MYH9 sequence was found to bind to miR-107, whereas the
mutant MYH9 sequence did not bind (Fig. 4D). After co-immunoprecipitation
using an Ago2-specific antibody, the results of the RIP assay
demonstrated significant enrichment of Linc00893 and miR-107
compared with that in the IgG control group (Fig. 4E). RT-qPCR analysis revealed that
the mRNA expression levels of MYH9 were significantly decreased
whereas miR-107 expression was significantly increased in the
si-Linc00893 group compared with those in the NC group (Fig. 4F). Furthermore, western blot
analysis revealed a significant decrease in MYH9 protein expression
in the si-Linc00893 group compared with that in the NC group
(P<0.001; Fig. 4G). Analysis of
the GSE160749 dataset from the GEO database showed significant
downregulation of MYH9 protein expression in AS samples (Fig. 4H); this finding is consistent with
the downregulation trend of Linc00893 and further corroborates the
mechanism where downregulated Linc00893 in AS inhibits MYH9
expression via reduced competitive binding to miR-107. These
experimental outcomes suggested that Linc00893 regulates MYH9
expression by competitively binding to miR-107, thus playing a
significant role in the molecular dynamics associated with AS.
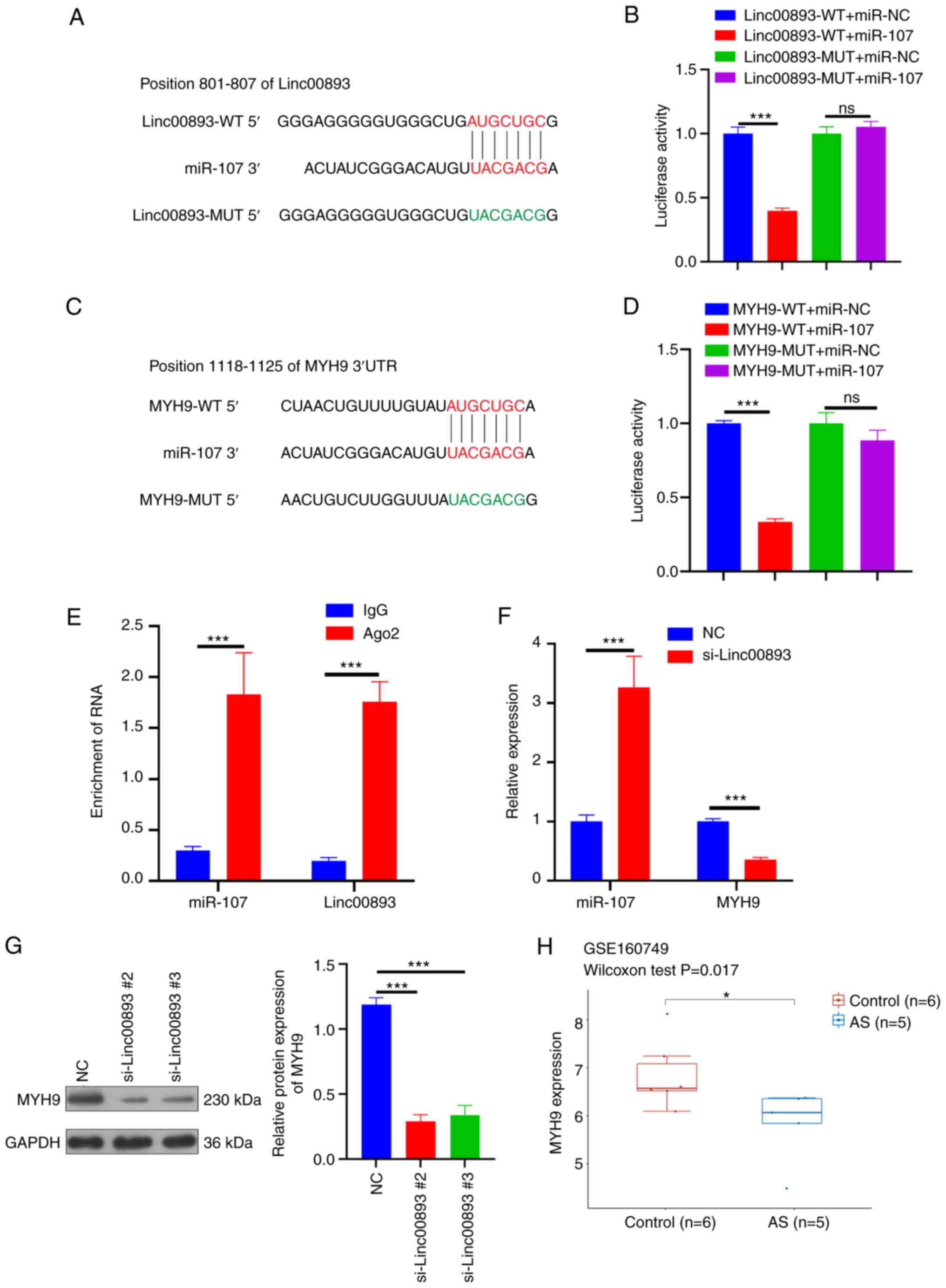 | Figure 4.Linc00893 regulates MYH9 expression
through competitive binding with miR-107. (A) Predicted binding
sites between Linc00893 and miR-107. (B) Dual-luciferase reporter
assay confirming the interaction between Linc00893 and miR-107. (C)
Predicted binding sites between miR-107 and MYH9. (D)
Dual-luciferase reporter assay confirming the interaction between
MYH9 and miR-107. (E) RNA immunoprecipitation assay results. (F)
Reverse transcription-quantitative PCR results. (G) Western
blotting results. (H) Analysis of the GSE160749 dataset. The
experiment was repeated three times, and data are presented as the
mean ± standard deviation. ns, no significance; *P<0.05;
***P<0.001. Ago2, Argonaute 2; AS, asthenozoospermia; Linc00893,
long intergenic noncoding RNA 00893; miR, microRNA; MUT, mutant;
MYH9, myosin heavy chain 9; NC, negative control; si, small
interfering; WT, wild type. |
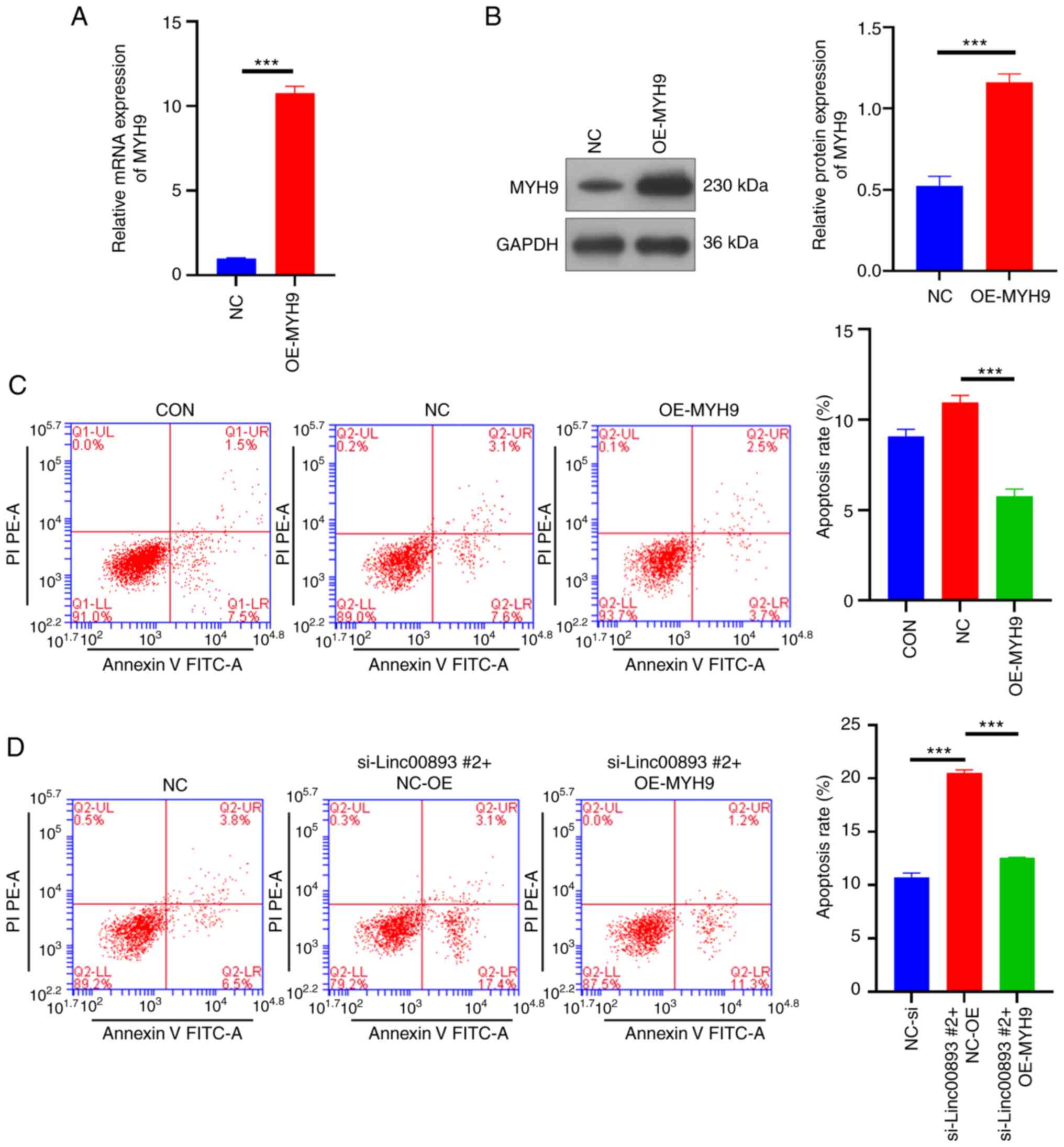
Overexpression of MYH9 inhibits SSC
apoptosis and reverses apoptosis induced by Linc00893
knockdown
To confirm the impact of MYH9 on SSCs, a lentiviral
vector overexpressing MYH9 and a NC vector were constructed, and
were transduced into SSCs. The RT-qPCR results showed that the mRNA
expression levels of MYH9 in the MYH9 overexpression (OE-MYH9)
group were significantly higher compared with those in the NC group
(Fig. 5A). Similarly, western blot
analysis revealed that the protein expression levels of MYH9 were
significantly increased in the OE-MYH9 group (P<0.001; Fig. 5B). These results suggested that
MYH9 was successfully overexpressed in SSCs. The flow cytometry
apoptosis assay indicated that overexpression of MYH9 inhibited SSC
apoptosis (Fig. 5C), and reversed
the apoptosis caused by knockdown of Linc00893 (Fig. 5D). These results indicated that
overexpressing MYH9 not only reduces the apoptosis of SSCs, but
also counteracts the apoptosis induced by Linc00893 knockdown,
indicating the crucial role of MYH9 in modulating SSC survival and
its involvement in the apoptotic pathways influenced by
Linc00893.
Discussion
AS is a common cause of male infertility,
characterized by reduced or lack of sperm motility (3). lncRNAs serve a critical role in male
infertility, but their specific mechanisms in AS are not fully
understood (7). The present study
confirmed the downregulation of Linc00893 in asthenozoospermic
samples, which aligns with our previous RNA sequencing results
(11). This discovery suggests
that Linc00893 may play an important regulatory role in the
pathological process of AS. Furthermore, the expression of
Linc00893 was positively correlated with progressive mobility,
total sperm motility, normal morphology rate, sperm concentration
and total sperm count, indicating that Linc00893 may be involved in
multiple biological processes related to sperm quality. However,
the sample size of 27 cases of AS is limited, precluding any
subgroup analysis. AS exhibits significant heterogeneity, with
varying degrees of sperm motility deficiency (17); therefore, further validation with a
larger clinical sample size is necessary. The current study
preliminarily indicated that Linc00893 is significantly
downregulated in AS samples; given the current sample size, this
preliminary conclusion can still be drawn.
SSCs are undifferentiated spermatogonia that serve
as the foundation for spermatogenesis, which is responsible for the
continuous production of spermatozoa (18). Additionally, SSCs undergo
self-renewal throughout the lifespan of mammals, differentiating
into spermatocytes and mature spermatozoa. The vitality of SSCs can
influence the motility of the resulting sperm. Consequently, they
serve as an in vitro model for the study of AS (13). Numerous studies have indicated that
the self-renewal and proliferation of SSCs are regulated by various
factors, including lncRNAs (19,20).
Recent research has revealed that the expression of Linc00893 is
inversely correlated with cell proliferation in various types of
cancer and that it serves a pivotal role in inhibiting tumor
progression (21–24). However, the relationship between
Linc00893 and SSCs remains unclear. By knocking down the expression
of Linc00893, the present study observed that Linc00893 knockdown
inhibited SSC vitality and promoted their apoptosis. Drawing upon
the present research findings from clinical samples, a positive
correlation was observed between the expression of Linc00893 and
the total sperm count. Furthermore, the knockdown of Linc00893
inhibited the proliferation of SSCs. Based on these observations,
it may be hypothesized that reduced expression of Linc00893 can
diminish male fertility by reducing sperm count and sperm vitality.
Consequently, enhancing the expression of Linc00893 may be a
pivotal strategy to improve the quality of male sperm.
In previous studies, the mechanism of Linc00893 has
been mainly associated with its interaction with miRNAs. For
example, in prostate cancer, Linc00893 interacts with miR-3173-5p,
leading to the downregulation of miR-3173-5p expression. This
inhibition subsequently suppresses the activation of the JAK2/STAT3
signaling pathway, thereby impeding prostate cancer progression
(23). Cytoplasmic lncRNAs
primarily exert their functions through interactions with miRNAs
(14), which is consistent with
the present finding that Linc00893 is predominantly localized in
the cytoplasm. Ago2 is a central component of the RNA-induced
silencing complex (RISC) (25).
Within the RISC, Ago2 associates with mature miRNAs, guiding the
complex to its target mRNAs, and resulting in their degradation or
translational suppression (26).
Consequently, the demonstration of lncRNA binding to Ago2 provides
evidence for its involvement in the competitive inhibition process
(27,28). The present study revealed that
Linc00893 was downregulated in AS, and could reduce competitive
binding to miR-107 and inhibit the expression of MYH9. MYH9, a
protein encoded by the gene of the same name, is involved in cell
motility and regulation of cell adhesion, and has crucial roles in
diseases, such as platelet dysfunction, tumor development and
hypertension (29,30). Studies have demonstrated that MYH9
is one of the key proteins involved in the binding of sperm to
oviductal glycoproteins (31,32).
MYH9, along with its non-muscle myosin IIB family, is essential for
cytoplasmic division during male meiosis (16). However, to the best of our
knowledge, there have been no reports linking MYH9 to AS. The
present findings indicated that overexpression of MYH9 inhibited
SSC apoptosis, suggesting a protective role for MYH9. This is
consistent with the majority of literature, which has reported that
MYH9 promotes cell proliferation (33–35).
Therefore, it may be hypothesized that the downregulation of
Linc00893, through its reduced competitive binding to miR-107,
inhibits MYH9, subsequently reducing SSC viability, and
contributing to the occurrence and progression of AS. The present
findings may enhance understanding of the molecular mechanisms
underlying AS. This knowledge is of significant importance for
further elucidating the regulatory mechanisms of SSCs and
developing therapeutic strategies for AS.
Although the present study has made some important
discoveries, there are still limitations that should be
acknowledged. Firstly, further research is needed to elucidate the
precise mechanisms of Linc00893 in AS. Secondly, the present study
mainly relied on in vitro experiments and lacked validation
in animal models in vivo. Additionally, functional
regulatory relationships between Linc00893, miR-107 and MYH9 need
to be investigated through separate interventions. Finally, the
clinical relevance of Linc00893 in relation to AS warrants further
investigation. Large-scale clinical studies involving different
populations with AS would help determine the diagnostic and
prognostic value of Linc00893 in AS. Unresolved issues will be
addressed in future research endeavors.
In conclusion, the present study detected the
downregulation of Linc00893 in AS, and its association with sperm
motility, SSC viability and MYH9 expression. Furthermore, Linc00893
may inhibit MYH9 expression through reduced competitive binding
with miR-107. These discoveries provide novel insights into the
pathogenesis of AS, and offer potential targets for the diagnosis
and treatment of related disorders.
Acknowledgements
Not applicable.
Funding
This work was funded by the Hainan Provincial Natural Science
Foundation (grant no. 822RC857) and the Key R&D Program of
Hainan Province (grant no. ZDYF2023SHFZ093).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
HL and DX were involved in cell-related experiments
and manuscript writing. LZ performed cell culture and material
procurement. HR performed FISH detection. AW performed western
blotting and RT-qPCR detection. JH performed dual-luciferase assay
and RIP. MX performed the EdU assay and reviewed the manuscript. WL
assessed cell apoptosis and reviewed the manuscript. HL and DX
confirm the authenticity of all the raw data. All authors read and
approved the manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of the Affiliated Obstetrics and Gynecology Hospital of
Hainan Medical College (Children's Hospital) (approval no.
2021-033) and was conducted in accordance with the principles
outlined in The Declaration of Helsinki. Written informed consent
was obtained from all participants (including the donor of
testicular biopsies for SSC generation) after they were provided
with detailed information about the experiment.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Carson SA and Kallen AN: Diagnosis and
management of infertility: A review. JAMA. 326:65–76. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Eisenberg ML, Esteves SC, Lamb DJ,
Hotaling JM, Giwercman A, Hwang K and Cheng YS: Male infertility.
Nat Rev Dis Primers. 9:492023. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Tu C, Wang W, Hu T, Lu G, Lin G and Tan
YQ: Genetic underpinnings of asthenozoospermia. Best Pract Res Clin
Endocrinol Metab. 34:1014722020. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Björndahl L and Brown JK; other Editorial
Board Members of the WHO Laboratory Manual for the Examination and
Processing of Human Semen, : The sixth edition of the WHO
Laboratory manual for the examination and processing of human
semen: Ensuring quality and standardization in basic examination of
human ejaculates. Fertil Steril. 117:246–251. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Pan MM, Hockenberry MS, Kirby EW and
Lipshultz LI: Male infertility diagnosis and treatment in the era
of in vitro fertilization and intracytoplasmic sperm injection. Med
Clin North Am. 102:337–347. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Herman AB, Tsitsipatis D and Gorospe M:
Integrated lncRNA function upon genomic and epigenomic regulation.
Mol Cell. 82:2252–2266. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kyrgiafini MA, Sarafidou T and Mamuris Z:
The role of long noncoding RNAs on male infertility: A systematic
review and in silico analysis. Biology (Basel).
11:15102022.PubMed/NCBI
|
|
8
|
Zhang X, Zhang P, Song D, Xiong S, Zhang
H, Fu J, Gao F, Chen H and Zeng X: Expression profiles and
characteristics of human lncRNA in normal and asthenozoospermia
sperm†. Biol Reprod. 100:982–993. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Saberiyan M, Mirfakhraie R, Gholami D,
Dehdehi L and Teimori H: Investigating the regulatory function of
the ANO1-AS2 on the ANO1 gene in infertile men with
asthenozoospermia and terato-asthenozoospermia. Exp Mol Pathol.
117:1045282020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Saberiyan M, Mirfakhraie R, Moghni M and
Teimori H: Study of Linc00574 regulatory effect on the TCTE3
expression in sperm motility. Reprod Sci. 28:159–165. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lu H, Xu D, Wang P, Sun W, Xue X, Hu Y,
Xie C and Ma Y: RNA-sequencing and bioinformatics analysis of long
noncoding RNAs and mRNAs in the asthenozoospermia. Biosci Rep.
40:BSR201940412020. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hou J, Niu M, Liu L, Zhu Z, Wang X, Sun M,
Yuan Q, Yang S, Zeng W, Liu Y, et al: Establishment and
characterization of human germline stem cell line with unlimited
proliferation potentials and no tumor formation. Sci Rep.
5:169222015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wang L, Cho KB, Li Y, Tao G, Xie Z and Guo
B: Long noncoding RNA (lncRNA)-mediated competing endogenous RNA
networks provide novel potential biomarkers and therapeutic targets
for colorectal cancer. Int J Mol Sci. 20:57582019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Güngör BH, Tektemur A, Arkali G, Dayan
Cinkara S, Acisu TC, Koca RH, Etem Önalan E, Özer Kaya S, Kizil M,
Sönmez M, et al: Effect of freeze-thawing process on lipid
peroxidation, miRNAs, ion channels, apoptosis and global DNA
methylation in ram spermatozoa. Reprod Fertil Dev. 33:747–759.
2021. View
Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yang F, Wei Q, Adelstein RS and Wang PJ:
Non-muscle myosin IIB is essential for cytokinesis during male
meiotic cell divisions. Dev Biol. 369:356–361. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Al-Malki AH, Alrabeeah K, Mondou E,
Brochu-Lafontaine V, Phillips S and Zini A: Testicular sperm
aspiration (TESA) for infertile couples with severe or complete
asthenozoospermia. Andrology. 5:226–231. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lord T and Nixon B: Metabolic changes
accompanying spermatogonial stem cell differentiation. Dev Cell.
52:399–411. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hu K, Zhang J and Liang M: LncRNA AK015322
promotes proliferation of spermatogonial stem cell C18-4 by acting
as a decoy for microRNA-19b-3p. In Vitro Cell Dev Biol Anim.
53:277–284. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Li L, Wang M, Wang M and Wu X, Geng L, Xue
Y, Wei X, Jia Y and Wu X: LncRNA analysis of mouse spermatogonial
stem cells following glial cell-derived neurotrophic factor
treatment. Genom Data. 5:275–278. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li S, Zhang Y, Dong J, Li R, Yu B, Zhao W
and Liu J: LINC00893 inhibits papillary thyroid cancer by
suppressing AKT pathway via stabilizing PTEN. Cancer Biomark.
30:277–286. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ou X, Zhou X, Li J, Ye J, Liu H, Fang D,
Cai Q, Cai S, He Y and Xu J: p53-Induced LINC00893 regulates RBFOX2
stability to suppress gastric cancer progression. Front Cell Dev
Biol. 9:7964512022. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yu C, Fan Y, Zhang Y, Liu L and Guo G:
LINC00893 inhibits the progression of prostate cancer through
miR-3173-5p/SOCS3/JAK2/STAT3 pathway. Cancer Cell Int. 22:2282022.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhu J, Jiang C, Hui H, Sun Y, Tao M, Liu Y
and Qian X: Overexpressed lncRNA LINC00893 suppresses progression
of colon cancer by binding with miR-146b-3p to upregulate PRSS8. J
Oncol. 2022:80023182022. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Li X, Wang X, Cheng Z and Zhu Q: AGO2 and
its partners: A silencing complex, a chromatin modulator, and new
features. Crit Rev Biochem Mol Biol. 55:33–53. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Barbu MG, Thompson DC, Suciu N, Voinea SC,
Cretoiu D and Predescu DV: The roles of MicroRNAs in male
infertility. Int J Mol Sci. 22:29102021. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang Y, Yang L, Chen T, Liu X, Guo Y, Zhu
Q, Tong X, Yang W, Xu Q, Huang D and Tu K: A novel lncRNA
MCM3AP-AS1 promotes the growth of hepatocellular carcinoma by
targeting miR-194-5p/FOXA1 axis. Mol Cancer. 18:282019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhao X, Su L, He X, Zhao B and Miao J:
Long noncoding RNA CA7-4 promotes autophagy and apoptosis
via sponging MIR877-3P and MIR5680 in high
glucose-induced vascular endothelial cells. Autophagy. 16:70–85.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Pecci A, Ma X, Savoia A and Adelstein RS:
MYH9: Structure, functions and role of non-muscle myosin IIA in
human disease. Gene. 664:152–167. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Savoia A and Pecci A: MYH9-related
disease. GeneReviews®. Adam MP, Mirzaa GM, Pagon RA,
Wallace SE, Bean LJH, Gripp KW and Amemiya A: University of
Washington; Seattle, USA: (Online).
|
|
31
|
Kadam KM, D'Souza SJ, Bandivdekar AH and
Natraj U: Identification and characterization of oviductal
glycoprotein-binding protein partner on gametes: Epitopic
similarity to non-muscle myosin IIA, MYH 9. Mol Hum Reprod.
12:275–282. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Lamy J, Nogues P, Combes-Soia L, Tsikis G,
Labas V, Mermillod P, Druart X and Saint-Dizier M: Identification
by proteomics of oviductal sperm-interacting proteins.
Reproduction. 155:457–466. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Gao S, Wang S, Zhao Z, Zhang C, Liu Z, Ye
P, Xu Z, Yi B, Jiao K, Naik GA, et al: TUBB4A interacts with MYH9
to protect the nucleus during cell migration and promotes prostate
cancer via GSK3β/β-catenin signalling. Nat Commun. 13:27922022.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Hu S, Ren S, Cai Y, Liu J, Han Y, Zhao Y,
Yang J, Zhou X and Wang X: Glycoprotein PTGDS promotes
tumorigenesis of diffuse large B-cell lymphoma by MYH9-mediated
regulation of Wnt-β-catenin-STAT3 signaling. Cell Death Differ.
29:642–656. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Lin X, Li AM, Li YH, Luo RC, Zou YJ, Liu
YY, Liu C, Xie YY, Zuo S, Liu Z, et al: Silencing MYH9 blocks
HBx-induced GSK3β ubiquitination and degradation to inhibit tumor
stemness in hepatocellular carcinoma. Signal Transduct Target Ther.
5:132020. View Article : Google Scholar : PubMed/NCBI
|