Introduction
Human esophageal squamous cell carcinoma (ESCC) is
one of the most aggressive types of cancer and is ranked as the
sixth most frequent cause of cancer-associated mortality in the
world, with a high incidence in northern China, South Africa,
Turkey and Iran (1–3). Furthermore, ESCC constitutes 7% of all
gastrointestinal cancers and is the predominant histological
subtype of esophageal cancer, comprising ~70% of cases worldwide.
Recently, progress in early diagnosis, surgery, and chemo- and
radiotherapy has led to an increase in the eight-year overall
survival rate of ESCC patients, however, improving the prognosis of
ESCC patients remains a challenge.
Tumor suppressor p53, encoded by the p53 gene
located at chromosome 17q13.1, is highly associated with a poor
prognosis in human cancers (4,5). It is
well known that the p53 protein may induce cell apoptosis and
regulate cell proliferation. Mutation of the p53 gene
results in the loss of its ability to induce cell death, which
leads to uncontrolled cell growth, thus, promoting tumorigenesis
(6,7).
In the present study, the overexpression of p53 in
the nucleus of the ESCC patient tissues was examined via tissue
microarray (TMA), which incorporated 118 ESCC specimens, as well as
using western blotting to analyze 64 samples of freshly frozen
tissues from ESCC patients.
The correlation between the p53 protein expression
level, and tumor progression and prognosis of ESCC patient was
evaluated, which may provide further data for predicting the
progression and prognosis in patients with ESCC.
Patients and methods
Patients and tissue samples
A total of 64 paired tissue samples, including tumor
tissue and the adjacent non-cancerous tissue, were collected from
ESCC patients who underwent surgery at the Department of
Cardiothoracic Surgery, the First Affiliated Hospital of Wenzhou
Medical University (Wenzhou, China) between May 2012 and September
2013. The tissues were immediately frozen in liquid nitrogen
following surgery and stored at −80°C until undergoing western blot
analysis to detect p53 expression levels. Written informed consent
for experimental use of the specimens was obtained from all
patients and the study was approved by the Board and Ethics
Committee of Wenzhou Medical University (Wenzhou, China). All the
patients were clinically and pathologically confirmed to exhibit
ESCC, and the tumor tissues were classified according to the
American Joint Committee on Cancer/Union Internationale Contre le
Cancer and were histologically graded in accordance with the World
Health Organization classification (8,9).
Protein extraction and western blot
analysis
The total protein from the 64 paired tissue samples
was homogenized using a homogenizer (Polytron PT-MR2100; Kinematica
AG, Luzern, Switzerland) in 1.5 ml tissue radio-immunoprecipitation
assay lysis buffer (50 mM Tris [pH 7.4], 150 mM NaCl, 1.0% Triton
X-100, 1% sodium deoxycholate and 0.1% SDS; Beyotime Institue of
Biotechnology, Shanghai, China) containing protease inhibitor
cocktail (Roche Applied Science, Indianapolis, IN, USA), 1 mM NaF
and 1 mM Na3VO4. Tissue homogenates were
incubated on ice for 15 min, centrifuged (Centrifuge 5417R;
Eppendorf, Hauppauge, NY, USA) at 18,000 × g for 20 min at 4°C and
the supernatants were collected. The protein concentration was
subsequently quantified using a BCA Protein assay kit (Thermo
Fisher Scientific, Waltham, MA, USA). A total of 20 mg protein from
each sample was separated by 10% SDS-PAGE (Bio-Rad, Hercules, CA,
USA) and transferred onto a nitrocellulose membrane (Bio-Rad).
Immunoblot analysis was subsequently performed with monoclonal
rabbit anti-human p53 (Proteintech Group, Wuhan, China) and
monoclonal mouse anti-human actin (Abmart Inc., Shanghai, China)
antibodies. The horseradish peroxidase-conjugated secondary
antibodies were obtained from Abmart Inc. The signals were
visualized using an electrochemiluminescence system (Thermo Fisher
Scientific) according to the manufacturer’s instructions and the
optical density was quantified using the National Institutes of
Health ImageJ software (http://imagej.nih.gov/ij/download/).
TMA and immunohistochemistry (IHC)
An ESCC TMA, containing a total of 118
formalin-fixed paraffin-embedded tissue samples, was constructed
according to a previously described method (10). IHC was also performed according to a
previously described method (11).
Briefly, the sections were deparaffinized in xylene and rehydrated
through a gradient concentration of alcohol. The endogenous
peroxidase activity was inactivated, non-specific staining was
blocked by 5% normal goat serum and all sections were incubated
with anti-p53 antibody (1:100; Abmart Inc.) overnight at 4°C. The
slides were incubated with biotin-labeled goat anti-rabbit
immunoglobulin G and further incubated with streptavidin peroxidase
solution (SABC kit, Boster Biological Technology, Ltd., Wuhan,
China). The staining was visualized by reaction with 3,
3′-di-aminobenzidine (Boster Biological Technology, Ltd.) in
phosphate-buffered saline [PBS; Dycent Biotech (Shanghai) Co. Ltd.,
Shanghai, China] with 0.05% H2O2 for 5 min at
room temperature. Control staining was performed by staining the
same TMA (duplicate) with PBS rather than anti-p53 and no
immunostaining was observed. The slides were counter-stained with
hematoxylin, washed in double-distilled H2O and mounted
with resinous mounting medium. The TMA were scored separately by
two pathologists who had no prior knowledge of the
clinicopathological status of the specimens on the TMA.
Assessment of IHC
Histopathological sections were microscopically
examined (Nikon ECLIPSE 80i; Nikon Corporation, Tokyo, Japan) and
scored by two independent pathologists, who were blinded to the
clinical data pertaining to the patients. The IHC staining of
mutant (MT)p53 was assessed according to the immune-reactive score
(IRS) as described previously (12,13)
with slight adjustments, which evaluated the percentage of positive
cells and the staining intensity. The percentage of positive cells
was scored as follows: 1, ≤10% positive cells; 2, 11–49%; 3,
50–79%; and 4, ≥80% (14). The
staining intensity was graded as 0, negative; 1, weak; 2, moderate;
and 3, strong. The two scores were multiplied and the IRS (a value
between 0 and 12) was determined as low or high, which corresponded
to IRS values of ≤6 and >6, respectively.
Statistical analysis
The optical density of the western blot signals was
quantified using the National Institutes of Health ImageJ software
and all statistical analyses were carried out using the SPSS 13.0
statistical software package (SPSS Inc., Chicago, IL, USA). The
expression level of p53 was quantified relative to β-actin and the
differences between the cancer tissues and adjacent normal tissues
in p53 protein expression levels were compared using Student’s
t-test. The χ2 test was performed to evaluate the
correlation between the clinicopathological features of the
patients and the p53 expression level, which was observed by IHC.
Kaplan-Meier survival analysis was used to evaluate the patient
prognosis and the eight-year survival rate of the ESCC patients was
obtained using the life table method. A univariate analysis was
plotted using the Kaplan-Meier method and Cox regression analysis
was used to evaluate the correlation between risk of ESCC and
clinicopathological parameters, including p53 expression. P≤0.05
was considered to indicate a statistically significant
difference.
Results
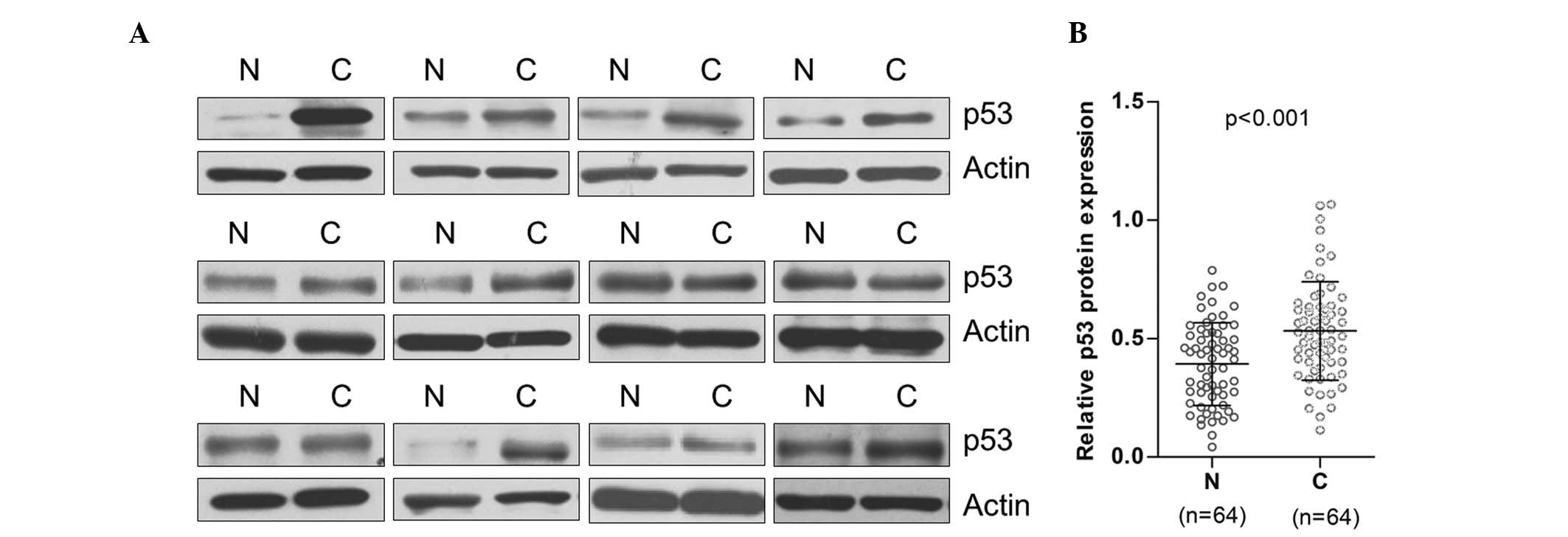
Increased level of p53 expression was
observed in ESCC tissues when compared with paired non-neoplastic
tissues
In the present study, the p53 protein expression
level of 64 paired tumor tissues and non-neoplastic tissues was
analyzed using western blot. The result demonstrated that the p53
expression level in the ESCC tissues was significantly higher than
that in the matched non-neoplastic tissues. The p53 protein in the
tumor tissue of the ESCC patients was found to be 1.89 times that
of the matched non-neoplastic tissues (n=64, P<0.001; Fig. 1A and B).
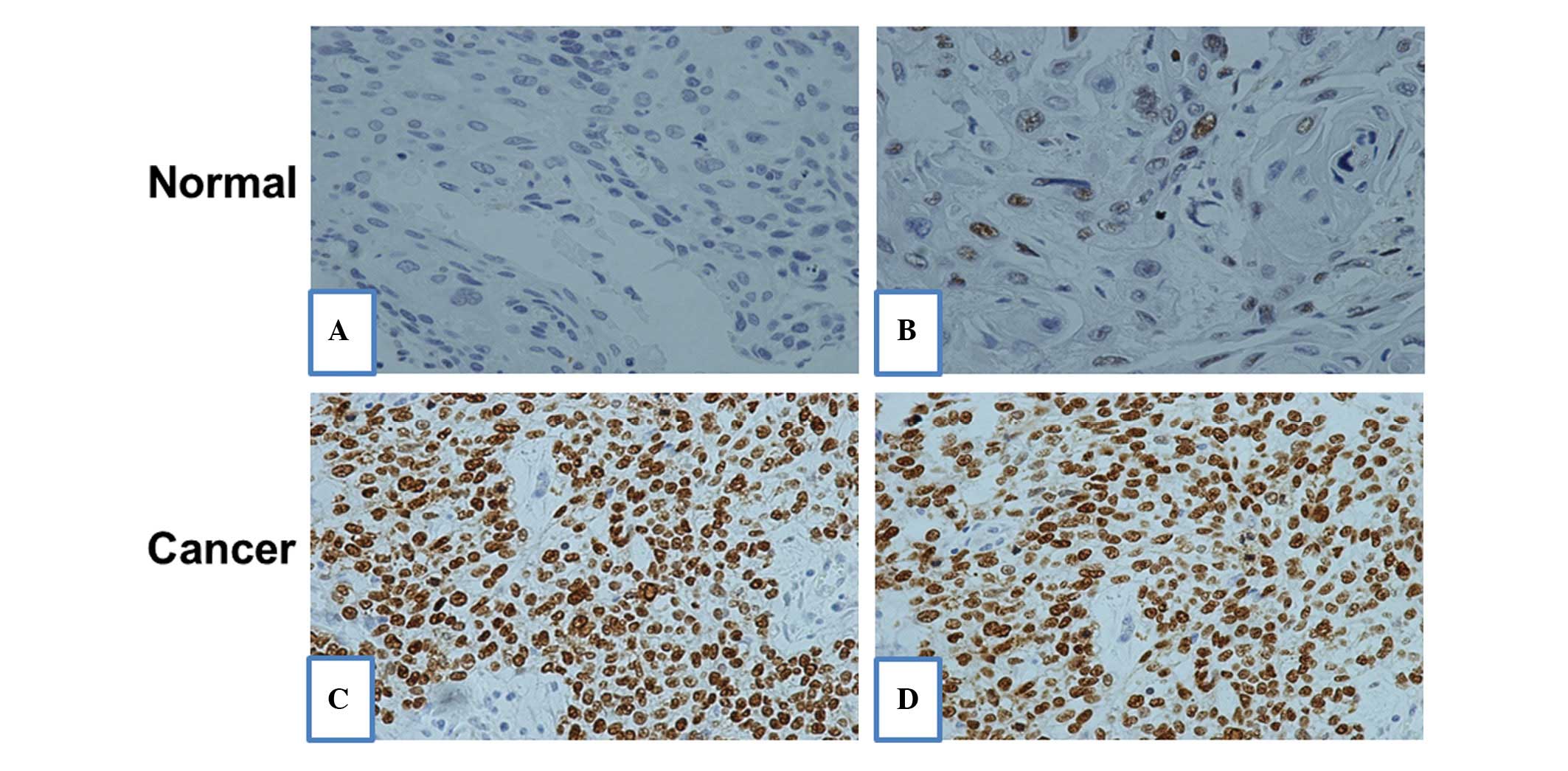
IHC of p53 expression levels in ESCC and
matched non-neoplastic tissues
IHC of ESCC TMA was conducted to further evaluate
the level of p53 protein expression in the ESCC tissues. IHC
revealed that the p53 protein was predominantly localized in the
nucleus (Fig. 2A) and the
expression level of p53 in the ESCC tissue was identified to be
significantly higher when compared with that in the adjacent normal
tissues (Fig. 2A).
Association of the p53 protein expression
level with clinicopathological features
A total of 118 ESCC patient tissue samples were used
to construct the TMA, including 96 males and 22 females (age range,
26–79 years; median, 63 years). The IHC staining for p53 (MTp53)
demonstrated low and high levels of p53 expression in 60 (50.8%)
and 58 (49.15%) samples, respectively. The level of p53 protein
expression was found to correlate with the pathological grade
(P<0.001) and N stage (P=0.007), however, not with patient age,
gender, history of alcohol consumption and smoking, T stage or TNM
stage (Table I).
 | Table IAssociation between p53 expression and
various clinicopathological factors of esophageal squamous cell
carcinoma patients. |
Table I
Association between p53 expression and
various clinicopathological factors of esophageal squamous cell
carcinoma patients.
| Characteristic | Total, n=118 | p53 protein
expression | P-value |
|---|
|
|---|
| Low, n=60 | High, n=58 |
|---|
| Gender | | | | 0.391 |
| Male | 96 | 47 | 49 | |
| Female | 22 | 13 | 9 | |
| Age | | | | 0.732 |
| <60 | 49 | 24 | 25 | |
| ≥60 | 69 | 36 | 33 | |
| Smoker | | | | 0.721 |
| Yes | 63 | 33 | 30 | |
| No | 55 | 27 | 28 | |
| Alcohol consumer | | | | 0.732 |
| Yes | 69 | 36 | 33 | |
| No | 49 | 24 | 25 | |
| Pathological
grade | | | | <0.001b |
| G1 | 37 | 29 | 8 | |
| G2 | 56 | 27 | 29 | |
| G3 | 25 | 4 | 21 | |
| T stagea | | | | 0.062 |
| T1 | 23 | 17 | 6 | |
| T2 | 36 | 14 | 22 | |
| T3 | 56 | 28 | 28 | |
| T4 | 3 | 1 | 2 | |
| N stagea | | | | 0.010b |
| N0 | 67 | 41 | 26 | |
| N≥1 | 51 | 19 | 32 | |
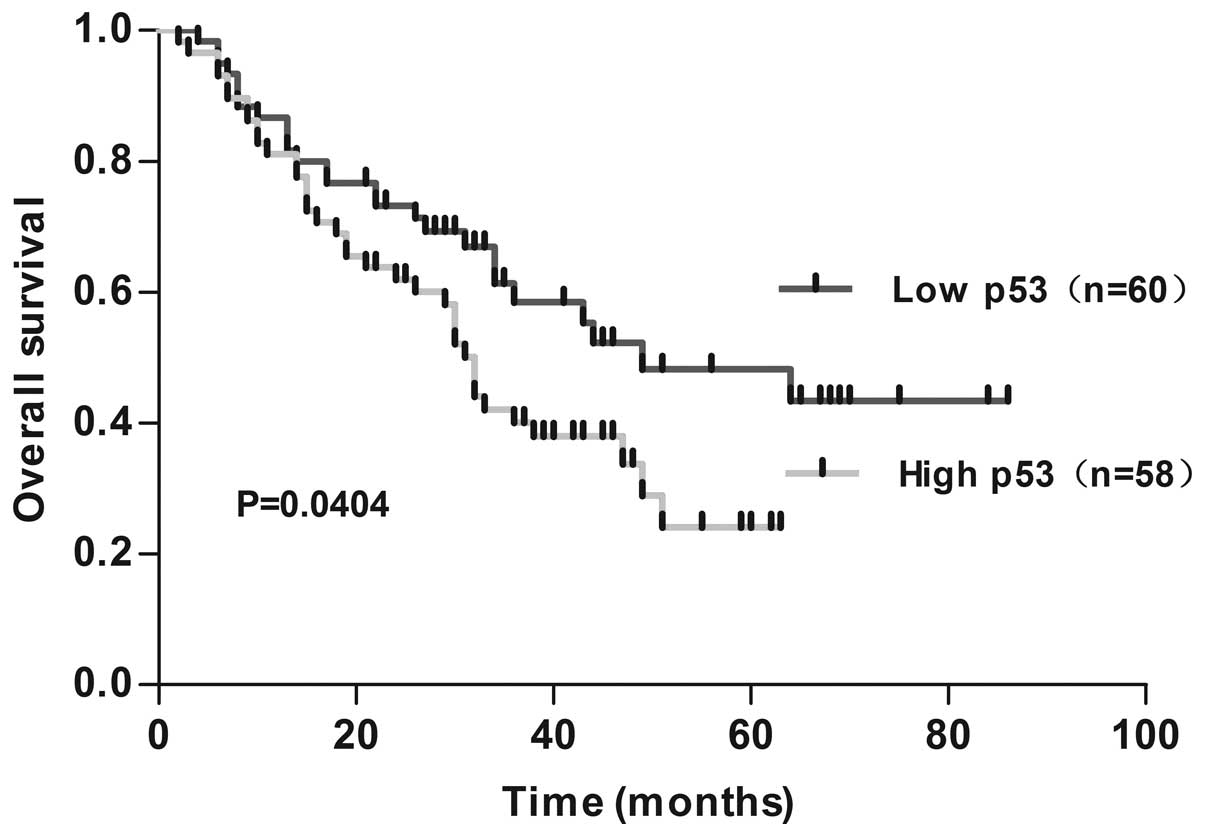
Survival analysis
The patients with clear follow-up data were used for
the survival analysis. Out of 118 patients, there were 58 cases
that exhibited a high expression level of p53 (49.15%), and 60
cases (50.85%) that exhibited a low expression level of p53. The
eight-year survival rate was 36.21% (21/58), 56.67% (34/60), in the
high p53 expression group and low p53 expression group,
respectively. Survival curves were obtained using the Kaplan-Meier
analysis and the log-rank test was used to compare differences in
survival between the two groups. According to the survival
analysis, it was found that the eight-year survival rate of the
group with low levels of p53 protein expression was higher than the
group with high levels of p53 protein expression (P=0.0404;
Fig. 3).
Assessment of ESCC risk factors
The univariate analysis and multivariate analysis
were used to evaluate the influence of various parameters on the
disease-free survival rate of ESCC patients. The ESCC risk factors,
including the p53 expression level, patient age, gender, TNM stage,
pathological grade, N stage, T stage, and history of smoking and
alcohol consumption were taken into account (Table II). The univariate analysis
demonstrated that the survival time of patients was significantly
correlated with the T stage (RR=3.886, P<0.001), N stage
(RR=3.620, P<0.001) and TNM stage (RR=3.576, P<0.001).
Furthermore, multivariate analysis revealed that the T stage
(RR=3.988, P<0.001) and N stage (RR=4.240, P=0.004) were
significant and independent prognostic factors for ESCC
patients.
 | Table IIUnivariate analysis and multivariate
analysis identifies the factors that influence the overall survival
rate of esophageal squamous cell carcinoma patients. |
Table II
Univariate analysis and multivariate
analysis identifies the factors that influence the overall survival
rate of esophageal squamous cell carcinoma patients.
| Univariate
analysis | Multivariate
analysis |
|---|
|
|
|
|---|
| Variable | RR | 95% CI | P-value | RR | 95% CI | P-value |
|---|
| p53 | 1.646 | 0.987–2.745 | 0.056 | 1.282 | 0.736–2.233 | 0.381 |
| Age | 1.007 | 0.979–1.035 | 0.652 | 1.012 | 0.984–1.040 | 0.408 |
| Gender | 1.282 | 0.651–2.523 | 0.472 | 1.160 | 0.501–2.685 | 0.728 |
| Smoker | 1.095 | 0.661–1.811 | 0.725 | 1.755 | 0.756–4.077 | 0.191 |
| Alcohol
consumer | 0.948 | 0.570–1.577 | 0.948 | 0.531 | 0.224–1.259 | 0.151 |
| Pathological
grade | 1.342 | 0.749–2.404 | 0.323 | 1.057 | 0.571–1.955 | 0.860 |
| T stagea | 3.886 | 2.256–6.696 | <0.001 | 3.988 | 1.969–8.077 | <0.001 |
| N stagea | 3.620 | 2.149–6.099 | <0.001 | 4.240 | 1.580–11.378 | 0.004 |
| TNM stagea | 3.576 | 2.144–5.963 | <0.001 | 0.596 | 0.207–1.716 | 0.381 |
Discussion
The human p53 gene is located at chromosome
17p13.1 and encodes the p53 protein, which is composed of 393 amino
acids. The p53 gene is a member of a highly conserved family
that contains at least another two genes, p63 and
p73. The wild-type (WT)p53 protein contains 393 amino acids
and p53 is a tumor suppressor that has a close association with
numerous types of human cancer; the mutation or loss of the
p53 gene can be identified in >50% of all human cancers
(15,16). p53 is involved in the regulation of
the cell cycle, as well as inducing a variety of activities to
maintain the genomic stability, cellular senescence (17,18)
and apoptosis (19). Under normal
conditions, p53 protein levels are maintained at a very low level
unless the cells are activated by signals from DNA damage, as well
as certain other cellular stresses (20). The response to DNA damage and
cellular stresses is the upregulation of the p53 protein expression
level, which leads to cell cycle arrest, DNA repair or apoptosis.
Thus, p53 is critical in the inhibition of malignant cancer cell
division.
There are two types of p53 proteins, WTp53 and
MTp53. WTp53 is a tumor suppressor, which prevents the
proliferation of tumor cells; MTp53 causes issues with the
regulation of the cell cycle, resulting in uncontrolled cell growth
that promotes tumorigenesis. WTp53 has a particularly short
half-life and is difficult to detect in normal cells. Conversely,
MTp53 is markedly more stable, with a longer half-life, which
favors detection by IHC. Previous studies have detected the p53
mutation using IHC and were able to define the tissue via the
strong staining of the p53 protein as MTp53 (21–23).
Based on this finding, the point mutation in the p53 gene
was associated with p53 protein stabilization. The majority of
human cancers may be detected via the upregulation of the p53
protein, including liposarcoma (24), colorectal cancer (25), breast carcinomas (26) and endometrial carcinomas (27). Recently, Zhu et al (28) demonstrated that the knock-down of
MTp53 using small interfering RNA induced cell cylce arrest and
triggered apoptosis in bladder cancer cells.
Chava et al (29) performed IHC in archival tissue
samples to evaluate the expression levels of fragile histidine
triad (FHIT) and the p53 protein. The study indicated that the
level of p53 gene expression was eight times that which was
observed in the normal tissues. The results showed that FHIT and
p53 were well correlated with SCC. As the study only
involved 23 ESCC samples to perform the IHC, a greater number of
samples are required to improve the evaluation of the correlation
of the p53 protein with ESCC. In the present study, western blot
analysis was performed using tissues, which were snap-frozen in
liquid nitrogen and stored at −80°C. The results demonstrated that
the expression level of p53 protein in the cancer tissues was 1.89
times than that in the normal tissue. In addition, IHC analysis was
conducted and the results demonstrated that the tumor tissues of
the ESCC patients exhibited strong p53 protein staining, whereas
the matched tumor-adjacent tissues exhibited weak p53 staining.
Therefore, the upregulation of p53 in ESCC patient tissues has a
significant role in esophageal carcinomaproliferation.
ESCC tumorigenesis is a complex process, which is
affected by various factors. The pathogenesis of ESCC remains
unclear, and numerous studies indicate that ESCC is associated with
multi-factor and multi-gene mutations. However, previous studies
have shown that environmental and lifestyle factors, such as
smoking, alcohol consumption, lack of fruit and vegetable intake,
or an excess of pickled foods are potential factors that may lead
to esophageal cancer (29–33). In the present study, the Cox
proportional hazards model was used during the statistical
analysis, and revealed that the patient age, gender, clinical and
pathological stages, and the presence or absence of a history of
alcohol and tobacco use did not result in significant differences
with regard to an association with prognosis; however, the results
of univariate analysis showed that the T stage, N stage and TNM
stage were significantly correlated with the prognosis of patient
survival. Additionally, multivariate analysis revealed that the T
and N stages correlated with ESCC patient survival.
In conclusion, the present results further
demonstrated that p53 (MTp53) is overexpressed in the tumor tissue
of ESCC patients, which leads to transcriptional regulation
dysfunction and uncontrolled cell growth. Therefore, p53 may be
used as a specific therapeutic target for the treatment of ESCC and
as a biomarker for the diagnosis of ESCC.
Acknowledgements
The authors would like to thank Dr Charles Reichman
for the critical review of the manuscript. The current study was
partially supported by the National Natural Science Foundation of
China (grant nos. 31070710 and 31171345), the Zhejiang Qianjiang
Talent Project B Grant (grant no. 2010R10045), which was awarded to
Professor Bin Lu, and the Natural Science Foundation of Zhejiang
Province (grant no. Y2110097), which was awarded to Professor
Yongzhang Liu.
References
|
1
|
Song QK, Li J, Jiang HD, He YM, Zhou XQ
and Huang CY: Esophageal cancer mortality during 2004–2009 in
Yanting County, China. Asian Pac J Cancer Prev. 13:5003–5006.
2012.
|
|
2
|
Blot WJ: Esophageal cancer trends and risk
factors. Semin Oncol. 21:403–410. 1994.
|
|
3
|
Brooks-Brunn JA: Esophageal cancer: an
overview. Medsurg Nurs. 9:248–254. 2000.
|
|
4
|
Cho H, Ha SY, Park SH, Park K and Chae YS:
Role of p53 gene mutation in tumor aggressiveness of intracranial
meningiomas. J Korean Med Sci. 14:199–205. 1999.
|
|
5
|
Murata A, Baba Y, Watanabe M, et al: p53
immunohistochemical expression and patient prognosis in esophageal
squamous cell carcinoma. Med Oncol. 30:7282013.
|
|
6
|
Cardin R, Piciocchi M, Tieppo C, et al:
Oxidative DNA damage in Barrett mucosa: correlation with telomeric
dysfunction and p53 mutation. Ann Surg Oncol. 20(Suppl 3):
S583–S589. 2013.
|
|
7
|
Di Agostino S, Strano S and Blandino G:
Gender, mutant p53 and PML: a growing ‘affaire’ in tumor
suppression and oncogenesis. Cell Cycle. 12:1824–1825. 2013.
|
|
8
|
Guinan P, Sobin LH, Algaba F, et al: TNM
staging of renal cell carcinoma: Workgroup No 3. Union
International Contre le Cancer (UICC) and the American Joint
Committee on Cancer (AJCC). Cancer. 80:992–993. 1997.
|
|
9
|
Sobin LH and Fleming ID: TNM
Classification of Malignant Tumors, fifth edition (1997). Union
Internationale Contre le Cancer and the American Joint Committee on
Cancer. Cancer. 80:1803–1804. 1997.
|
|
10
|
Nocito A, Bubendorf L, Tinner EM, et al:
Microarrays of bladder cancer tissue are highly representative of
proliferation index and histological grade. J Pathol. 194:349–357.
2001.
|
|
11
|
Nie X, Li M, Lu B, et al: Down-regulating
overexpressed human Lon in cervical cancer suppresses cell
proliferation and bioenergetics. PloS One. 8:e810842013.
|
|
12
|
Remmele W and Stegner HE: Recommendation
for uniform definition of an immunoreactive score (IRS) for
immunohistochemical estrogen receptor detection (ER-ICA) in breast
cancer tissue. Pathologe. 8:138–140. 1987.(In German).
|
|
13
|
Cheng AN, Jiang SS, Fan CC, et al:
Increased Cdc7 expression is a marker of oral squamous cell
carcinoma and overexpression of Cdc7 contributes to the resistance
to DNA-damaging agents. Cancer Lett. 337:218–225. 2013.
|
|
14
|
Bolander A, Agnarsdóttir M, Strömberg S,
et al: The protein expression of TRP-1 and galectin-1 in cutaneous
malignant melanomas. Cancer genomics Proteomics. 5:293–300.
2008.
|
|
15
|
Grelewski PG and Bar JK: The role of p53
protein and MMP-2 tumor/stromal cells expression on progressive
growth of ovarian neoplasms. Cancer Invest. 31:472–479. 2013.
|
|
16
|
Lee JY, Kim HJ, Yoon NA, et al: Tumor
suppressor p53 plays a key role in induction of both
tristetraprolin and let-7 in human cancer cells. Nucleic Acids Res.
41:5614–5625. 2013.
|
|
17
|
Krell J, Frampton AE, Colombo T, et al:
The p53 miRNA interactome and its potential role in the cancer
clinic. Epigenomics. 5:417–428. 2013.
|
|
18
|
Gu Z, Jiang J, Tan W, et al: p53/p21
Pathway involved in mediating cellular senescence of bone
marrow-derived mesenchymal stem cells from systemic lupus
erythematosus patients. Clin Dev Immunol. 2013:1342432013.
|
|
19
|
Zhou Y and Ho WS: Combination of
liquiritin, isoliquiritin and isoliquirigenin induce apoptotic cell
death through upregulating p53 and p21 in the A549 non-small cell
lung cancer cells. Oncol Rep. 31:298–304. 2014.
|
|
20
|
Formigari A, Gregianin E and Irato P: The
effect of zinc and the role of p53 in copper-induced cellular
stress responses. J Appl Toxicol. 33:527–536. 2013.
|
|
21
|
Biramijamal F, Allameh A, Mirbod P, Groene
HJ, Koomagi R and Hollstein M: Unusual profile and high prevalence
of p53 mutations in esophageal squamous cell carcinomas from
northern Iran. Cancer Res. 61:3119–3123. 2001.
|
|
22
|
Guimaraes DP and Hainaut P: TP53: a key
gene in human cancer. Biochimie. 84:83–93. 2002.
|
|
23
|
Taghavi N, Biramijamal F, Sotoudeh M, et
al: Association of p53/p21 expression with cigarette smoking and
prognosis in esophageal squamous cell carcinoma patients. World J
Gastroenterol. 16:4958–4967. 2010.
|
|
24
|
Chiarugi V and Ruggiero M: Role of three
cancer ‘master genes’ p53, bcl2 and c-myc on the apoptotic process.
Tumori. 82:205–209. 1996.
|
|
25
|
Huerta S, Gao X, Dineen S, Kapur P, Saha D
and Meyer J: Role of p53, Bax, p21, and DNA-PKcs in radiation
sensitivity of HCT-116 cells and xenografts. Surgery. 154:143–151.
2013.
|
|
26
|
Jung SY, Jeong J, Shin SH, et al:
Accumulation of p53 determined by immunohistochemistry as a
prognostic marker in node negative breast cancer; analysis
according to St Gallen consensus and intrinsic subtypes. J Surg
Oncol. 103:207–211. 2011.
|
|
27
|
Koshiyama M, Konishi I, Wang DP, et al:
Immunohistochemical analysis of p53 protein over-expression in
endometrial carcinomas: inverse correlation with sex steroid
receptor status. Virchows Arch A Pathol Anat Histopathol.
423:265–271. 1993.
|
|
28
|
Zhu HB, Yang K, Xie YQ, Lin YW, Mao QQ and
Xie LP: Silencing of mutant p53 by siRNA induces cell cycle arrest
and apoptosis in human bladder cancer cells. World J Surg Oncol.
11:222013.
|
|
29
|
Chava S, Mohan V, Shetty PJ, et al:
Immunohistochemical evaluation of p53, FHIT, and IGF2 gene
expression in esophageal cancer. Dis Esophagus. 25:81–87. 2012.
|
|
30
|
Launoy G, Milan C, Day NE, Pienkowski MP,
Gignoux M and Faivre J: Diet and squamous-cell cancer of the
oesophagus: a French multicentre case-control study. Int J Cancer.
76:7–12. 1998.
|
|
31
|
Sammon AM and Alderson D: Diet, reflux and
the development of squamous cell carcinoma of the oesophagus in
Africa. Br J Surg. 85:891–896. 1998.
|
|
32
|
Morse DE, Pendrys DG, Katz RV, et al: Food
group intake and the risk of oral epithelial dysplasia in a United
States population. Cancer Causes Control. 11:713–720. 2000.
|
|
33
|
Mizobuchi S, Furihata M, Sonobe H, et al:
Association between p53 immunostaining and cigarette smoking in
squamous cell carcinoma of the esophagus. Jpn J Clin Oncol.
30:423–428. 2000.
|